Plasma Proteomic Biomarkers in Alzheimer’s Disease and Cardiovascular Disease: A Longitudinal Study
Abstract
:1. Introduction
2. Results
2.1. Baseline Descriptive Statistics
2.2. Multivariable LMM Analysis
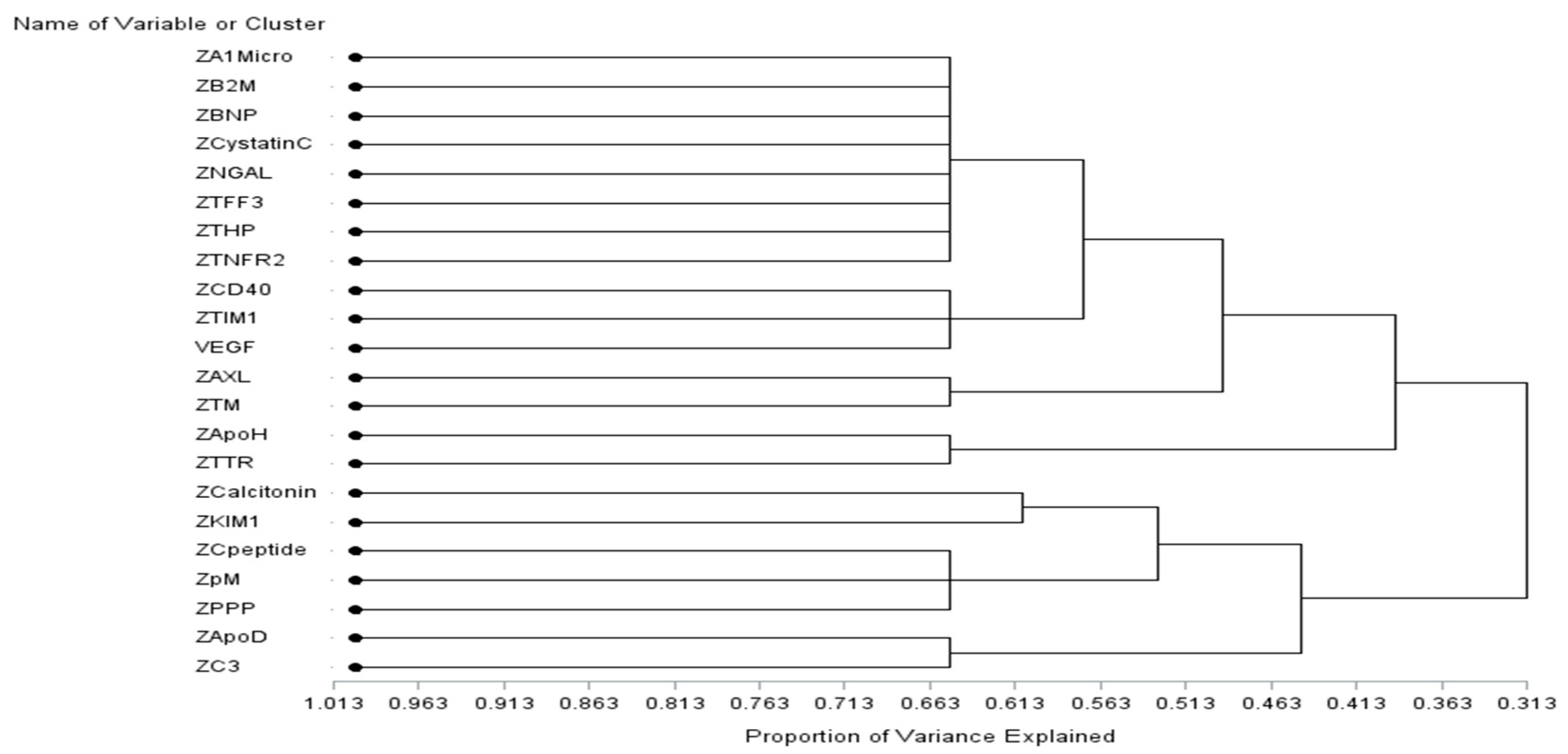
2.3. Correlation Analysis and Variable Cluster Analysis
3. Discussion
3.1. Proteomic Biomarkers Associated with AD, CVDs, and APOE-ε4
3.2. Proteomic Biomarkers Associated with MCI, CVDs, and APOE-ε4
3.3. Proteomic Biomarkers Associated with AD, MCI, CVDs, and APOE-ε4
3.4. Relationship among Shared Proteomic Biomarkers
3.5. Strengths and Limitations
4. Materials and Methods
4.1. Dataset
4.2. Measures
4.3. Statistical Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- 2023 Alzheimer’s Disease Facts and Figures. Alzheimer’s Dement 2023, 19, 1598–1695. [CrossRef] [PubMed]
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J Am Coll Cardiol 2020, 76, 2982–3021. [Google Scholar] [CrossRef]
- Waigi, E.W.; Webb, R.C.; Moss, M.A.; Uline, M.J.; McCarthy, C.G.; Wenceslau, C.F. Soluble and Insoluble Protein Aggregates, Endoplasmic Reticulum Stress, and Vascular Dysfunction in Alzheimer’s Disease and Cardiovascular Diseases. GeroScience 2023, 45, 1411–1438. [Google Scholar] [CrossRef] [PubMed]
- Virani, S.S.; Alonso, A.; Aparicio, H.J.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics—2021 Update: A Report From the American Heart Association. Circulation 2021, 143(8), e254–e743. [Google Scholar] [CrossRef]
- Carter, C.J.; France, J.; Crean, S.; Singhrao, S.K. The Porphyromonas Gingivalis/Host Interactome Shows Enrichment in GWASdb Genes Related to Alzheimer’s Disease, Diabetes and Cardiovascular Diseases. Front. Aging Neurosci. 2017, 9, 408. [Google Scholar] [CrossRef] [PubMed]
- Gong, S.; Su, B.B.; Tovar, H.; Mao, C.; Gonzalez, V.; Liu, Y.; Lu, Y.; Wang, K.-S.; Xu, C. Polymorphisms Within RYR3 Gene Are Associated With Risk and Age at Onset of Hypertension, Diabetes, and Alzheimer’s Disease. Am. J. Hypertens. 2018, 31, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Tini, G.; Scagliola, R.; Monacelli, F.; La Malfa, G.; Porto, I.; Brunelli, C.; Rosa, G.M. Alzheimer’s Disease and Cardiovascular Disease: A Particular Association. Cardiol. Res. Pract. 2020, 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Mur, J.; McCartney, D.L.; Walker, R.M.; Campbell, A.; Bermingham, M.L.; Morris, S.W.; Porteous, D.J.; McIntosh, A.M.; Deary, I.J.; Evans, K.L.; et al. DNA Methylation in APOE: The Relationship with Alzheimer’s and with Cardiovascular Health. AD Transl. Res. Clin. Interv. 2020, 6, e12026. [Google Scholar] [CrossRef] [PubMed]
- De Bruijn, R.F.; Ikram, M.A. Cardiovascular Risk Factors and Future Risk of Alzheimer’s Disease. BMC Med. 2014, 12, 130. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, U.K.; Bennet, A.M.; Gatz, M.; Dickman, P.W.; Pedersen, N.L. Nonstroke Cardiovascular Disease and Risk of Alzheimer Disease and Dementia. Alzheimer Dis. Assoc. Disord. 2010, 24, 213–219. [Google Scholar] [CrossRef]
- Kotze, M.J.; Van Rensburg, S.J. Pathology Supported Genetic Testing and Treatment of Cardiovascular Disease in Middle Age for Prevention of Alzheimer’s Disease. Metab. Brain Dis. 2012, 27, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Mahley, R.W. Apolipoprotein E: From Cardiovascular Disease to Neurodegenerative Disorders. J. Mol. Med. 2016, 94, 739–746. [Google Scholar] [CrossRef]
- Ray, M.; Ruan, J.; Zhang, W. Variations in the Transcriptome of Alzheimer’s Disease Reveal Molecular Networks Involved in Cardiovascular Diseases. Genome Biol. 2008, 9, R148. [Google Scholar] [CrossRef]
- Wang, K.; Lu, Y.; Morrow, D.F.; Xiao, D.; Xu, C.; The Alzheimer’s Disease Neuroimaging Initiative. Associations of ARHGAP26 Polymorphisms with Alzheimer’s Disease and Cardiovascular Disease. J. Mol. Neurosci. 2022, 72, 1085–1097. [Google Scholar] [CrossRef]
- Belloy, M.E.; Napolioni, V.; Greicius, M.D. A Quarter Century of APOE and Alzheimer’s Disease: Progress to Date and the Path Forward. Neuron 2019, 101, 820–838. [Google Scholar] [CrossRef]
- Xu, C.; Garcia, D.; Lu, Y.; Ozuna, K.; Adjeroh, D.A.; Wang, K.; on behalf of the Alzheimer’s Disease Neuroimaging Initiative. Levels of Angiotensin-Converting Enzyme and Apolipoproteins Are Associated with Alzheimer’s Disease and Cardiovascular Diseases. Cells 2021, 11, 29. [Google Scholar] [CrossRef]
- Broce, I.J.; Tan, C.H.; Fan, C.C.; Jansen, I.; Savage, J.E.; Witoelar, A.; Wen, N.; Hess, C.P.; Dillon, W.P.; Glastonbury, C.M.; et al. Dissecting the Genetic Relationship between Cardiovascular Risk Factors and Alzheimer’s Disease. Acta. Neuropathol. 2019, 137, 209–226. [Google Scholar] [CrossRef]
- Bessi, V.; Balestrini, J.; Bagnoli, S.; Mazzeo, S.; Giacomucci, G.; Padiglioni, S.; Piaceri, I.; Carraro, M.; Ferrari, C.; Bracco, L.; et al. Influence of ApoE Genotype and Clock T3111C Interaction with Cardiovascular Risk Factors on the Progression to Alzheimer’s Disease in Subjective Cognitive Decline and Mild Cognitive Impairment Patients. J. Pers. Med. 2020, 10, 45. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Mehta, V.; Singh, T.G. Alzheimer’s Disorder: Epigenetic Connection and Associated Risk Factors. Curr. Neuropharmacol. 2020, 18, 740–753. [Google Scholar] [CrossRef] [PubMed]
- Jammeh, E.; Zhao, P.; Carroll, C.; Pearson, S.; Ifeachor, E. Identification of Blood Biomarkers for Use in Point of Care Diagnosis Tool for Alzheimer’s Disease. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; IEEE: Orlando, FL, USA; pp. 2415–2418. [Google Scholar]
- Amatruda, J.G.; Estrella, M.M.; Garg, A.X.; Thiessen-Philbrook, H.; McArthur, E.; Coca, S.G.; Parikh, C.R.; Shlipak, M.G.; TRIBE-AKI Consortium. Urine Alpha-1-Microglobulin Levels and Acute Kidney Injury, Mortality, and Cardiovascular Events Following Cardiac Surgery. Am. J. Nephrol. 2021, 52, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-M.; Ma, Y.-H.; Gao, P.-Y.; Wang, Z.-B.; Huang, L.-Y.; Hou, J.-H.; Tan, L.; Yu, J.-T. Plasma Β2-Microglobulin and Cerebrospinal Fluid Biomarkers of Alzheimer’s Disease Pathology in Cognitively Intact Older Adults: The CABLE Study. Alzheimer's Res. Ther. 2023, 15, 69. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Y.; Wang, C.; Chen, W.; Chen, X.; Wu, F.; He, H. Copper Metabolism-Related Genes in Entorhinal Cortex for Alzheimer’s Disease. Sci. Rep. 2023, 13, 17458. [Google Scholar] [CrossRef]
- Kim, Y.; Lu, S.; Ho, J.E.; Hwang, S.; Yao, C.; Huan, T.; Levy, D.; Ma, J. Proteins as Mediators of the Association Between Diet Quality and Incident Cardiovascular Disease and All-Cause Mortality: The Framingham Heart Study. JAHA 2021, 10, e021245. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Sun, L.; Kaptoge, S. Association of Beta-2-Microglobulin and Cardiovascular Events and Mortality: A Systematic Review and Meta-Analysis. Atherosclerosis 2021, 320, 70–78. [Google Scholar] [CrossRef]
- Chen, F.; Liu, J.; Li, F.-Q.; Wang, S.-S.; Zhang, Y.-Y.; Lu, Y.-Y.; Hu, F.-F.; Yao, R.-Q. Β2-Microglobulin Exacerbates Neuroinflammation, Brain Damage, and Cognitive Impairment after Stroke in Rats. Neural. Regen. Res. 2023, 18, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Borné, Y.; Johnson, L.; Muhammad, I.F.; Persson, M.; Niu, K.; Engström, G. Comparing the Inflammatory Profiles for Incidence of Diabetes Mellitus and Cardiovascular Diseases: A Prospective Study Exploring the ‘Common Soil’ Hypothesis. Cardiovasc. Diabetol. 2018, 17, 87. [Google Scholar] [CrossRef] [PubMed]
- Hertle, E.; Van Greevenbroek, M.M.; Arts, I.C.; Van Der Kallen, C.J.; Geijselaers, S.L.; Feskens, E.J.; Jansen, E.H.; Schalkwijk, C.G.; Stehouwer, C.D. Distinct Associations of Complement C3a and Its Precursor C3 with Atherosclerosis and Cardiovascular Disease: The CODAM Study. Thromb. Haemost. 2014, 111, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Arguinzonis, M.; Diaz-Riera, E.; Peña, E.; Escate, R.; Juan-Babot, O.; Mata, P.; Badimon, L.; Padro, T. Alternative C3 Complement System: Lipids and Atherosclerosis. IJMS 2021, 22, 5122. [Google Scholar] [CrossRef]
- Sacks, F.M.; Furtado, J.D.; Jensen, M.K. Protein-Based HDL Subspecies: Rationale and Association with Cardiovascular Disease, Diabetes, Stroke, and Dementia. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2022, 1867, 159182. [Google Scholar] [CrossRef]
- Hu, W.T.; Watts, K.D.; Tailor, P.; Nguyen, T.P.; Howell, J.C.; Lee, R.C.; Seyfried, N.T.; Gearing, M.; Hales, C.M.; Levey, A.I.; et al. CSF Complement 3 and Factor H Are Staging Biomarkers in Alzheimer’s Disease. Acta Neuropathol. Commun. 2016, 4, 14. [Google Scholar] [CrossRef] [PubMed]
- Bonham, L.W.; Desikan, R.S.; Yokoyama, J.S. The Relationship between Complement Factor C3, APOE Ε4, Amyloid and Tau in Alzheimer’s Disease. Acta Neuropathol. Commun. 2016, 4, 65. [Google Scholar] [CrossRef]
- Wang, T.; Wang, X.; Yao, Y.; Zhao, C.; Yang, C.; Han, Y.; Cai, Y. Association of Plasma Apolipoproteins and Levels of Inflammation-Related Factors with Different Stages of Alzheimer’s Disease: A Cross-Sectional Study. BMJ Open 2022, 12, e054347. [Google Scholar] [CrossRef]
- Simonsen, A.H.; Hagnelius, N.-O.; Waldemar, G.; Nilsson, T.K.; McGuire, J. Protein Markers for the Differential Diagnosis of Vascular Dementia and Alzheimer’s Disease. Int. J. Proteom. 2012, 2012, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mathews, P.M.; Levy, E. Cystatin C in Aging and in Alzheimer’s Disease. Ageing Res. Rev. 2016, 32, 38–50. [Google Scholar] [CrossRef] [PubMed]
- McNicholas, K.; François, M.; Liu, J.-W.; Doecke, J.D.; Hecker, J.; Faunt, J.; Maddison, J.; Johns, S.; Pukala, T.L.; Rush, R.A.; et al. Salivary Inflammatory Biomarkers Are Predictive of Mild Cognitive Impairment and Alzheimer’s Disease in a Feasibility Study. Front. Aging Neurosci. 2022, 14, 1019296. [Google Scholar] [CrossRef] [PubMed]
- Gevorgyan, M.M.; Voronina, N.P.; Goncharova, N.V.; Kozaruk, T.V.; Russkikh, G.S.; Bogdanova, L.A.; Korolenko, T.A. Cystatin C as a Marker of Progressing Cardiovascular Events during Coronary Heart Disease. Bull Exp. Biol. Med. 2017, 162, 421–424. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Du, Q.; Dai, Q.; Ge, J.; Cheng, X. Cysteine Protease Cathepsins in Atherosclerotic Cardiovascular Diseases. J. Atheroscler. Thromb. 2018, 25, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Einwoegerer, C.F.; Domingueti, C.P. Association Between Increased Levels of Cystatin C and the Development of Cardiovascular Events or Mortality: A Systematic Review and Meta-Analysis. Arq. Bras. Cardiol. 2018, 111, 796–807. [Google Scholar] [CrossRef]
- West, M.; Kirby, A.; Stewart, R.A.; Blankenberg, S.; Sullivan, D.; White, H.D.; Hunt, D.; Marschner, I.; Janus, E.; Kritharides, L.; et al. Circulating Cystatin C Is an Independent Risk Marker for Cardiovascular Outcomes, Development of Renal Impairment, and Long-Term Mortality in Patients With Stable Coronary Heart Disease: The LIPID Study. JAHA 2022, 11, e020745. [Google Scholar] [CrossRef] [PubMed]
- Van Der Laan, S.W.; Fall, T.; Soumaré, A.; Teumer, A.; Sedaghat, S.; Baumert, J.; Zabaneh, D.; Van Setten, J.; Isgum, I.; Galesloot, T.E.; et al. Cystatin C and Cardiovascular Disease. J. Am. Coll. Cardiol. 2016, 68, 934–945. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, A.C.; Jansson, J.-H.; Söderberg, S.; Ruge, T.; Larsson, A.; Ärnlöv, J. Levels of Soluble Tumor Necrosis Factor Receptor 1 and 2, Gender, and Risk of Myocardial Infarction in Northern Sweden. Atherosclerosis 2018, 272, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Ain, Q.U.; Sarfraz, M.; Prasesti, G.K.; Dewi, T.I.; Kurniati, N.F. Confounders in Identification and Analysis of Inflammatory Biomarkers in Cardiovascular Diseases. Biomolecules 2021, 11, 1464. [Google Scholar] [CrossRef] [PubMed]
- Ajoolabady, A.; Bi, Y.; McClements, D.J.; Lip, G.Y.H.; Richardson, D.R.; Reiter, R.J.; Klionsky, D.J.; Ren, J. Melatonin-Based Therapeutics for Atherosclerotic Lesions and beyond: Focusing on Macrophage Mitophagy. Pharmacol. Res. 2022, 176, 106072. [Google Scholar] [CrossRef] [PubMed]
- Apte, R.S.; Chen, D.S.; Ferrara, N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell 2019, 176, 1248–1264. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.K.; Gracias, D.T.; Croft, M. TNF Activity and T Cells. Cytokine 2018, 101, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Cribbs, D.H.; Anderson, A.J.; Cummings, B.J.; Su, J.H.; Wasserman, A.J.; Cotman, C.W. The Induction of the TNF Death Domain Signaling Pathway in Alzheimer’s Disease Brain. Neurochem. Res. 2003, 28, 307–318. [Google Scholar] [CrossRef]
- Zhao, A.; Li, Y.; Deng, Y. TNF Receptors Are Associated with Tau Pathology and Conversion to Alzheimer’s Dementia in Subjects with Mild Cognitive Impairment. Neurosci. Lett. 2020, 738, 135392. [Google Scholar] [CrossRef]
- Ortí-Casañ, N.; Wu, Y.; Naudé, P.J.W.; De Deyn, P.P.; Zuhorn, I.S.; Eisel, U.L.M. Targeting TNFR2 as a Novel Therapeutic Strategy for Alzheimer’s Disease. Front. Neurosci. 2019, 13, 49. [Google Scholar] [CrossRef] [PubMed]
- Pillai, J.A.; Bebek, G.; Khrestian, M.; Bena, J.; Bergmann, C.C.; Bush, W.S.; Leverenz, J.B.; Bekris, L.M. TNFRSF1B Gene Variants and Related Soluble TNFR2 Levels Impact Resilience in Alzheimer’s Disease. Front. Aging Neurosci. 2021, 13, 638922. [Google Scholar] [CrossRef] [PubMed]
- Ortí-Casañ, N.; Zuhorn, I.S.; Naudé, P.J.W.; De Deyn, P.P.; Van Schaik, P.E.M.; Wajant, H.; Eisel, U.L.M. A TNF Receptor 2 Agonist Ameliorates Neuropathology and Improves Cognition in an Alzheimer’s Disease Mouse Model. Proc. Natl. Acad. Sci. USA 2022, 119, e2201137119. [Google Scholar] [CrossRef] [PubMed]
- Ortí-Casañ, N.; Wajant, H.; Kuiperij, H.B.; Hooijsma, A.; Tromp, L.; Poortman, I.L.; Tadema, N.; De Lange, J.H.E.; Verbeek, M.M.; De Deyn, P.P.; et al. Activation of TNF Receptor 2 Improves Synaptic Plasticity and Enhances Amyloid-β Clearance in an Alzheimer’s Disease Mouse Model with Humanized TNF Receptor 2. JAD 2023, 94, 977–991. [Google Scholar] [CrossRef] [PubMed]
- Steubl, D.; Buzkova, P.; Ix, J.H.; Devarajan, P.; Bennett, M.R.; Chaves, P.H.M.; Shlipak, M.G.; Bansal, N.; Sarnak, M.J.; Garimella, P.S. Association of Serum and Urinary Uromodulin and Their Correlates in Older Adults—The Cardiovascular Health Study. Nephrology 2020, 25, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Thielemans, R.; Speeckaert, R.; Delrue, C.; De Bruyne, S.; Oyaert, M.; Speeckaert, M.M. Unveiling the Hidden Power of Uromodulin: A Promising Potential Biomarker for Kidney Diseases. Diagnostics 2023, 13, 3077. [Google Scholar] [CrossRef]
- Devuyst, O.; Olinger, E.; Rampoldi, L. Uromodulin: From Physiology to Rare and Complex Kidney Disorders. Nat. Rev. Nephrol. 2017, 13, 525–544. [Google Scholar] [CrossRef]
- Steubl, D.; Schneider, M.P.; Meiselbach, H.; Nadal, J.; Schmid, M.C.; Saritas, T.; Krane, V.; Sommerer, C.; Baid-Agrawal, S.; Voelkl, J.; et al. Association of Serum Uromodulin with Death, Cardiovascular Events, and Kidney Failure in CKD. CJASN 2020, 15, 616–624. [Google Scholar] [CrossRef]
- Araújo, D.C.; Veloso, A.A.; Gomes, K.B.; De Souza, L.C.; Ziviani, N.; Caramelli, P. A Novel Panel of Plasma Proteins Predicts Progression in Prodromal Alzheimer’s Disease. JAD 2022, 88, 549–561. [Google Scholar] [CrossRef]
- Jack, C.R.; Bennett, D.A.; Blennow, K.; Carrillo, M.C.; Dunn, B.; Haeberlein, S.B.; Holtzman, D.M.; Jagust, W.; Jessen, F.; Karlawish, J.; et al. NIA-AA Research Framework: Toward a Biological Definition of Alzheimer’s Disease. Alzheimer’s Dement. 2018, 14, 535–562. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, R.M.; Paterson, R.W.; Warren, J.D.; Zetterberg, H.; O’Brien, J.T.; Fox, N.C.; Halliday, G.M.; Schott, J.M. Biomarkers in Dementia: Clinical Utility and New Directions. J. Neurol. Neurosurg. Psychiatry 2014, 85, 1426–1434. [Google Scholar] [CrossRef] [PubMed]
- Khoury, R.; Patel, K.; Gold, J.; Hinds, S.; Grossberg, G.T. Recent Progress in the Pharmacotherapy of Alzheimer’s Disease. Drugs Aging 2017, 34, 811–820. [Google Scholar] [CrossRef] [PubMed]
- Buchhave, P. Cerebrospinal Fluid Levels Ofβ-Amyloid 1-42, but Not of Tau, Are Fully Changed Already 5 to 10 Years Before the Onset of Alzheimer Dementia. Arch. Gen. Psychiatry 2012, 69, 98. [Google Scholar] [CrossRef]
- Villemagne, V.L.; Burnham, S.; Bourgeat, P.; Brown, B.; Ellis, K.A.; Salvado, O.; Szoeke, C.; Macaulay, S.L.; Martins, R.; Maruff, P.; et al. Amyloid β Deposition, Neurodegeneration, and Cognitive Decline in Sporadic Alzheimer’s Disease: A Prospective Cohort Study. Lancet Neurol. 2013, 12, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Jansen, W.J.; Ossenkoppele, R.; Knol, D.L.; Tijms, B.M.; Scheltens, P.; Verhey, F.R.J.; Visser, P.J.; Aalten, P.; Aarsland, D.; Alcolea, D.; et al. Prevalence of Cerebral Amyloid Pathology in Persons Without Dementia: A Meta-Analysis. JAMA 2015, 313, 1924. [Google Scholar] [CrossRef]
- Tibolla, G.; Norata, G.D.; Meda, C.; Arnaboldi, L.; Uboldi, P.; Piazza, F.; Ferrarese, C.; Corsini, A.; Maggi, A.; Vegeto, E.; et al. Increased Atherosclerosis and Vascular Inflammation in APP Transgenic Mice with Apolipoprotein E Deficiency. Atherosclerosis 2010, 210, 78–87. [Google Scholar] [CrossRef]
- Austin, S.A.; Sens, M.A.; Combs, C.K. Amyloid Precursor Protein Mediates a Tyrosine Kinase-Dependent Activation Response in Endothelial Cells. J. Neurosci. 2009, 29, 14451–14462. [Google Scholar] [CrossRef]
- De Meyer, G.R.Y.; De Cleen, D.M.M.; Cooper, S.; Knaapen, M.W.M.; Jans, D.M.; Martinet, W.; Herman, A.G.; Bult, H.; Kockx, M.M. Platelet Phagocytosis and Processing of β-Amyloid Precursor Protein as a Mechanism of Macrophage Activation in Atherosclerosis. Circ. Res. 2002, 90, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.-H.; Alexopoulos, P.; Wagenpfeil, S.; Kurz, A.; Perneczky, R. Plasma Proteomics for the Identification of Alzheimer Disease. Alzheimer Dis. Assoc. Disord. 2013, 27, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, V.; Kosian, S. Feature Selection and Dimension Reduction Techniques in SAS; EXL Service: New York, NY, USA, 2011. [Google Scholar]
- Muthén, B.; Kaplan, D. A Comparison of Some Methodologies for the Factor Analysis of Non-normal Likert Variables. Br. J. Math. Stat. 1985, 38, 171–189. [Google Scholar] [CrossRef]
| Variable | AD | MCI | CN | F/χ2, p | CVD | Non-CVD | t/χ2, p |
|---|---|---|---|---|---|---|---|
| Age (mean ± SD) | 74.8 ± 8.1 | 74.8 ± 7.7 | 75.1 ± 5.8 | 0.05, 0.9527 | 75.2 ± 7.2 | 73.9 ± 7.8 | 3.60, 0.0584 |
| Gender | |||||||
| Male | 65 | 248 | 30 | 4.39, 0.1113 | 258 | 85 | 4.38, 0.0364 * |
| Female | 46 | 135 | 28 | 140 | 69 | ||
| Education (mean ± SD) | 15.1 ± 3.2 | 15.6 ± 3.0 | 15.7 ± 2.8 | 1.31, 0.2699 | 15.4 ± 3.1 | 15.8 ± 2.8 | 1.70, 0.1930 |
| APOE- ε4 | |||||||
| 0 | 36 | 178 | 53 | 54.80, <0.0001 *** | 187 | 80 | 1.10, 0.2953 |
| 1+ | 75 | 205 | 5 | 211 | 74 |
| Variable | AD vs. CN (t, p) | MCI vs. CN (t, p) | CVD vs. Non-CVD (t, p) | APOE-ε4-1+ vs. 0 (t, p) | 12 Months (t, p) |
|---|---|---|---|---|---|
| A1Micro | 2.40, 0.0168 * | 1.80, 0.0720 | 4.68, <0.0001 *** | −2.01, 0.0454 * | −5.20, <0.0001 *** |
| ApoH | 2.20, 0.0279 * | −1.55, 0.1225 | 2.90, 0.0039 ** | −0.54, 0.5874 | −15.40, <0.0001 *** |
| β2M | 3.10, 0.0020 ** | 0.28, 0.7813 | 4.52, <0.0001 *** | −2.61, 0.0092 ** | −0.97, 0.3314 |
| BNP | 5.53, <0.0001 *** | 4.94, <0.0001 *** | 3.82, 0.0001 ** | −0.40, 0.6866 | 3.728, 0.0011 ** |
| Complement C3 | 2.66, 0.0080 ** | −1.44, 0.1504 | 2.07, 0.0391 * | −2.30, 0.0221 * | 2.94, 0.0034 ** |
| Cystatin C | 2.31, 0.0213 * | −1.24, 0.2157 | 5.78, <0.0001 *** | −2.49, 0.0130 * | −1.65, 0.1001 |
| KIM1 | −3.06, 0.0023 ** | 0.74, 0.4617 | 3.12, 0.0019 ** | 0.01, 0.9900 | −5.47, <0.0001 *** |
| NGAL | 2.83, 0.0048 ** | 0.60, 0.5465 | 2.53, 0.0117 * | −1.61 0.1080 | −3.94, <0.0001 *** |
| PPP | 3.53, 0.0005 ** | 3.08, 0.0022 ** | 2.15, 0.0322 * | 1.31, 0.1904 | 2.77, 0.0059 ** |
| TFF3 | 2.84, 0.0046 ** | 0.30, 0.7653 | 4.30, <0.0001 *** | −1.37, 0.1706 | 4.09, <0.0001 *** |
| THP | −3.51, 0.0005 ** | −2.91, 0.0038 ** | −4.10, <0.0001 *** | 2.90, 0.0038 ** | −9.98, <0.0001 *** |
| TIM1 | 3.29, 0.0011 ** | 0.68, 0.4999 | 2.14, 0.0328 * | −1.51, 0.1321 | 0.72, 0.4744 |
| TM | 2.14, 0.0324 * | 0.90, 0.3665 | 4.19, <0.0001 *** | −0.74, 0.4626 | −1.46, 0.1455 |
| VEGF | 3.63, 0.0003 ** | 2.40, 0.0169 * | 4.13, <0.0001 *** | −1.70, 0.0895 | 1.92, 0.0553 |
| Variable | AD vs. CN (t, p) | MCI vs. CN (t, p) | CVD vs. non-CVD (t, p) | APOE-ε4-1+ vs. 0 (t, p) | 12 Months (t, p) |
|---|---|---|---|---|---|
| ApoD | −1.84, 0.0670 | −3.12, 0.0019 ** | −2.00, 0.0456 * | −0.92, 0.3576 | −0.78, 0.4374 |
| AXL | 1.62, 0.1067 | 2.64, 0.0084 ** | 2.28, 0.0233 * | −0.25, 0.8027 | −5.62, <0.0001 *** |
| Calcitonin | 1.86, 0.0633 | 2.75, 0.0061 ** | 2.17, 0.0302 * | 2.51, 0.0124 * | −2.34, 0.0197 * |
| CD40 | 1.15, 0.2498 | −2.61, 0.0093 ** | −0.93, 0.3543 | −0.42, 0.6278 | 2.97, 0.0032 ** |
| C-peptide | 1.14, 0.2558 | 2.03, 0.0429 * | 4.77, <0.0001 *** | 0.22, 0.8281 | 1.05, 0.2947 |
| pM | 0.56, 0.5784 | 2.10, 0.0365 * | 3.63, 0.0003 ** | −1.15, 0.2525 | 1.95, 0.0517 |
| TNFR2 | 0.64, 0.5208 | −2.07, 0.0385 * | 3.96, <0.0001 *** | −2.38, 0.0175 * | −3.43, 0.0007 ** |
| TTR | −1.42, 0.1562 | −3.69, 0.0002 ** | 2.68, 0.0075 ** | 1.10, 0.2701 | −10.65, <0.0001 *** |
| Comparison | Visit | Difference ± SE | t, p |
|---|---|---|---|
| AD vs. CN | Baseline | −0.52 ± 0.16 | −3.16, 0.0048 ** |
| 12 months | −0.60 ± 0.16 | −3.66, 0.0008 ** | |
| MCI vs. CN | Baseline | −0.31 ± 0.14 | −2.22, 0.0695 |
| 12 months | −0.50 ± 0.14 | −3.53, 0.0013 ** | |
| AD vs. MCI | Baseline | −0.20 ± 0.10 | −1.97, 0.1219 |
| 12 months | −0.10 ± 0.11 | −0.99, 0.5863 | |
| CVD vs. non-CVD | Baseline | −0.39 ± 0.09 | −4.32, <0.0001 *** |
| 12 months | −0.33 ± 0.09 | −3.55, 0.0004 ** | |
| APOE-ε4-1+ vs. 0 | Baseline | 0.24 ± 0.09 | 2.73, 0.0066 ** |
| 12 months | 0.26 ± 0.09 | 2.94, 0.0034 ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Theeke, L.A.; Liu, Y.; Wang, S.; Luo, X.; Navia, R.O.; Xiao, D.; Xu, C.; Wang, K.; The Alzheimer and Disease Neuroimaging Initiative. Plasma Proteomic Biomarkers in Alzheimer’s Disease and Cardiovascular Disease: A Longitudinal Study. Int. J. Mol. Sci. 2024, 25, 10751. https://doi.org/10.3390/ijms251910751
Theeke LA, Liu Y, Wang S, Luo X, Navia RO, Xiao D, Xu C, Wang K, The Alzheimer and Disease Neuroimaging Initiative. Plasma Proteomic Biomarkers in Alzheimer’s Disease and Cardiovascular Disease: A Longitudinal Study. International Journal of Molecular Sciences. 2024; 25(19):10751. https://doi.org/10.3390/ijms251910751
Chicago/Turabian StyleTheeke, Laurie A., Ying Liu, Silas Wang, Xingguang Luo, R. Osvaldo Navia, Danqing Xiao, Chun Xu, Kesheng Wang, and The Alzheimer and Disease Neuroimaging Initiative. 2024. "Plasma Proteomic Biomarkers in Alzheimer’s Disease and Cardiovascular Disease: A Longitudinal Study" International Journal of Molecular Sciences 25, no. 19: 10751. https://doi.org/10.3390/ijms251910751







