Identification, Biochemical Characterization, and In Vivo Detection of a Zn-Metalloprotease with Collagenase Activity from Mannheimia haemolytica A2
Abstract
:1. Introduction
2. Results
2.1. Zymography of Proteins Precipitated with Ammonium Sulfate (PpAs) from the CS of M. haemolytica A2
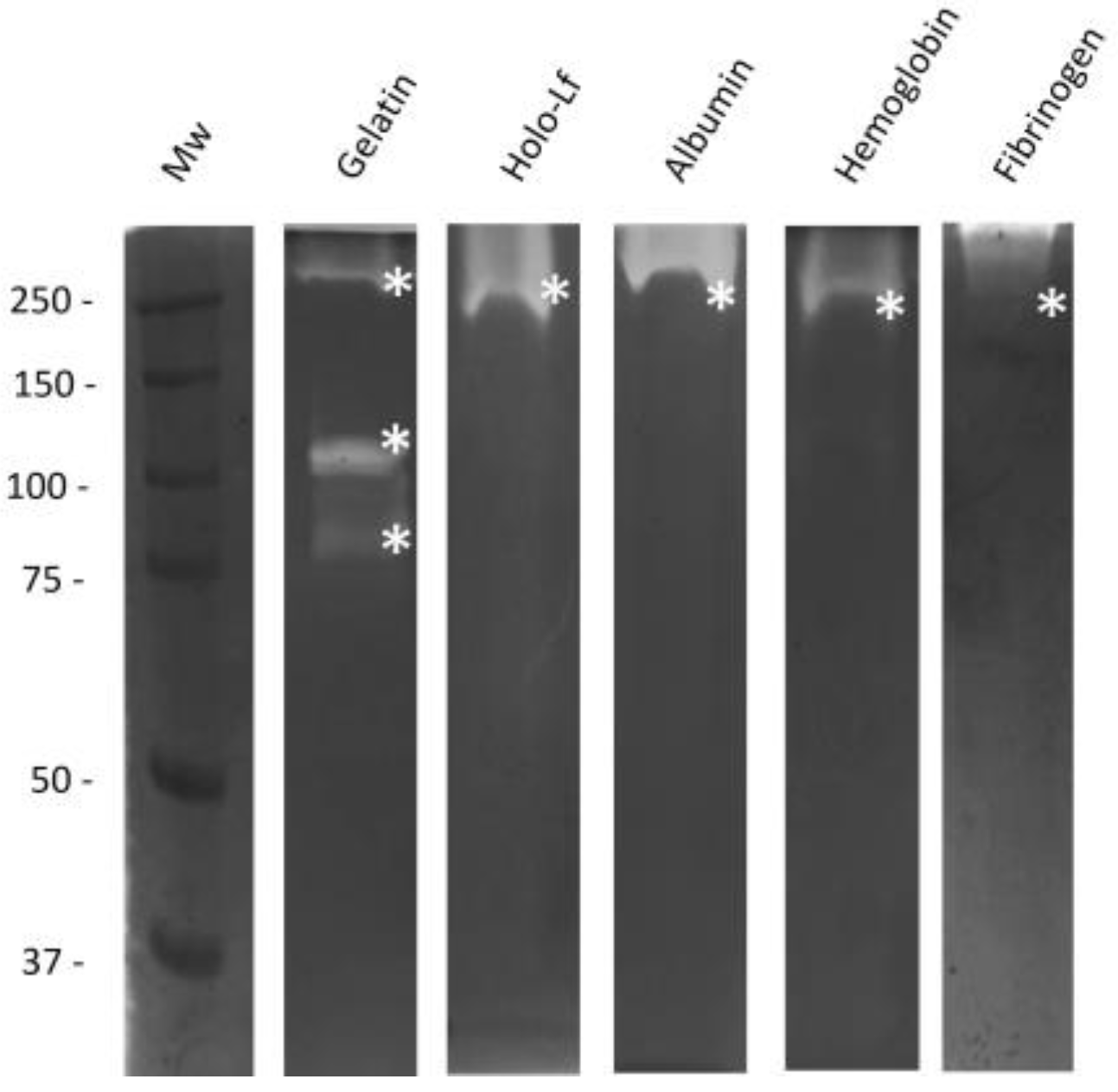
2.2. Determination of the Proteolytic Activity of 60% PpAs from M. haemolytica A2 against Several Mammalian Proteins
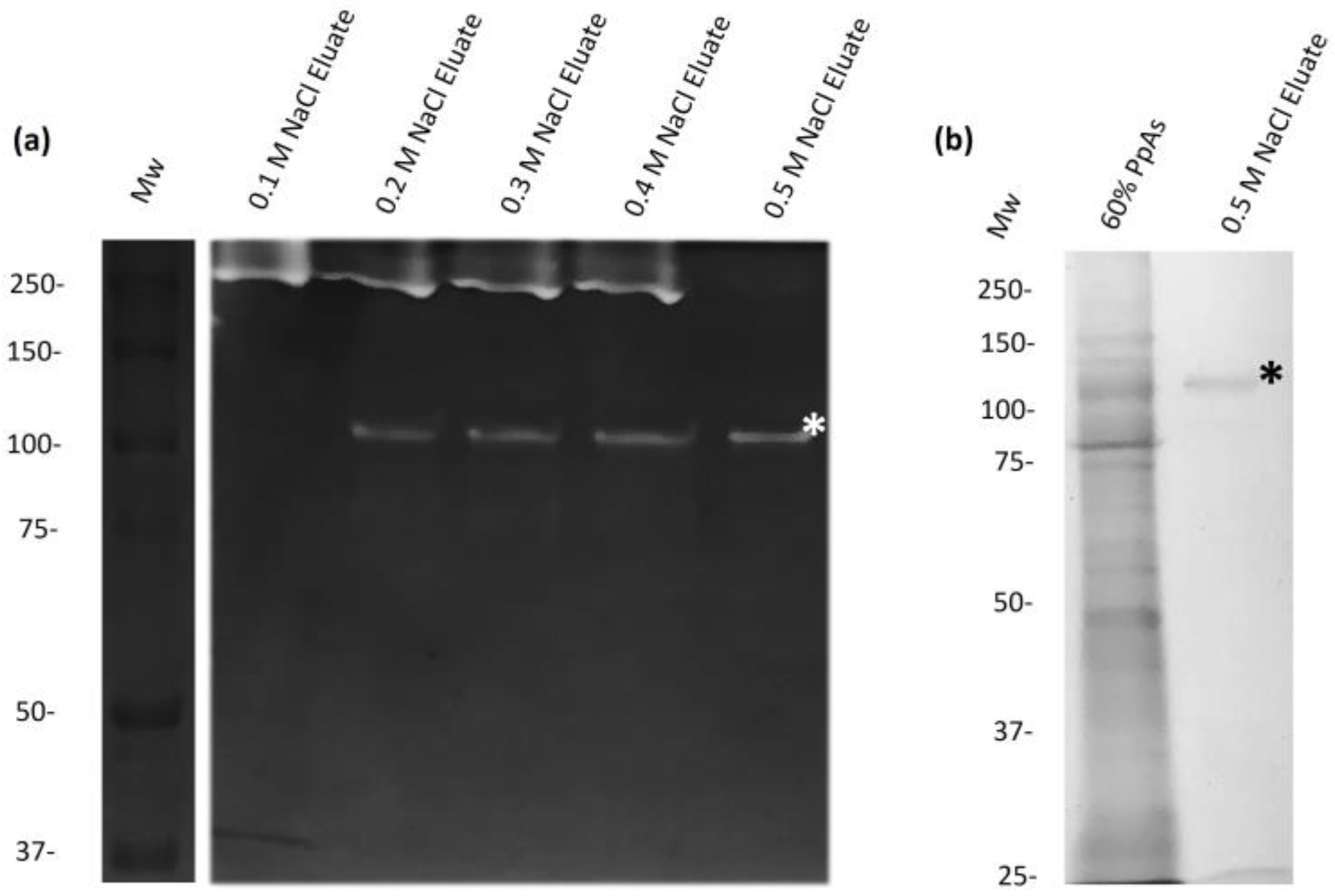
2.3. The 110-kDa Mh Protease Was Purified through Cationic Exchange Chromatography
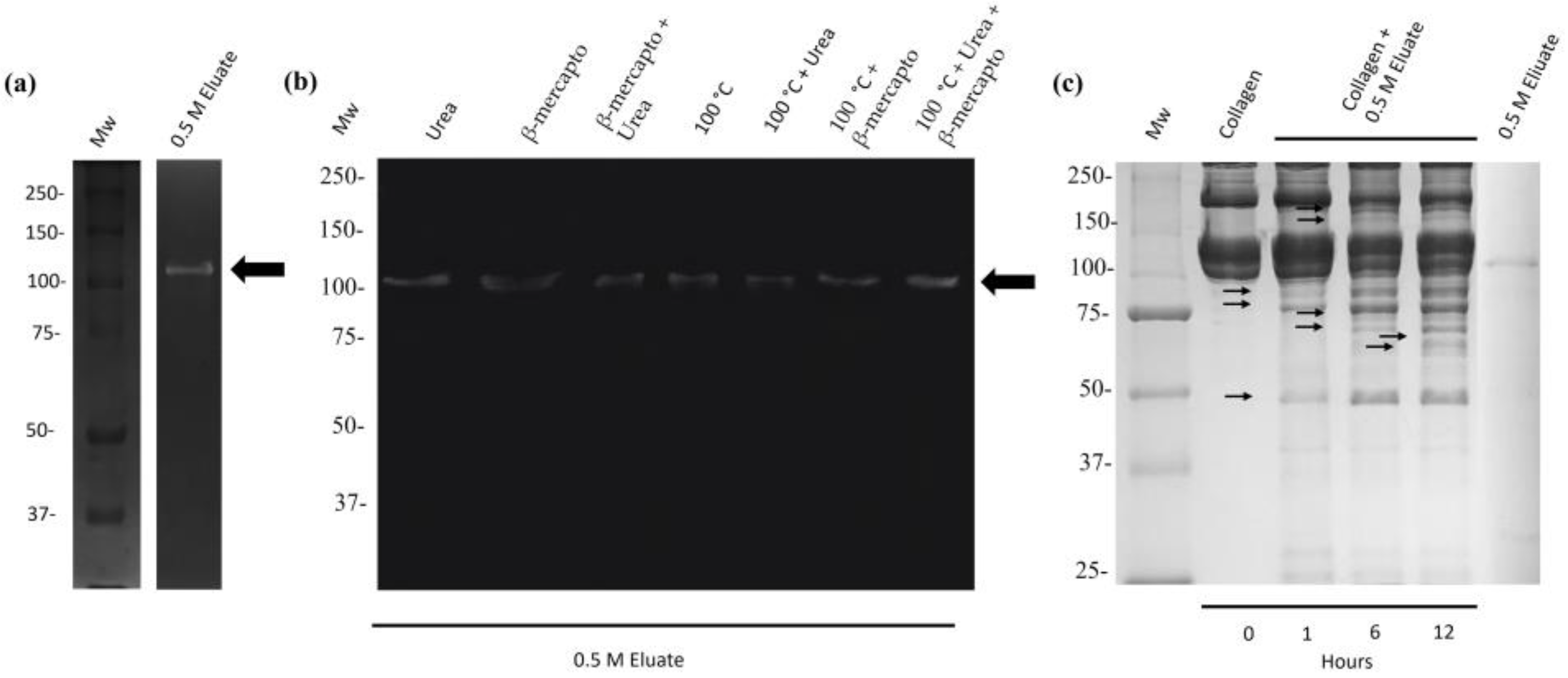
2.4. The 110 kDa Protease of M. haemolytica A2 Is a Heat-Resistant Enzyme That Possesses Collagenolytic Activity
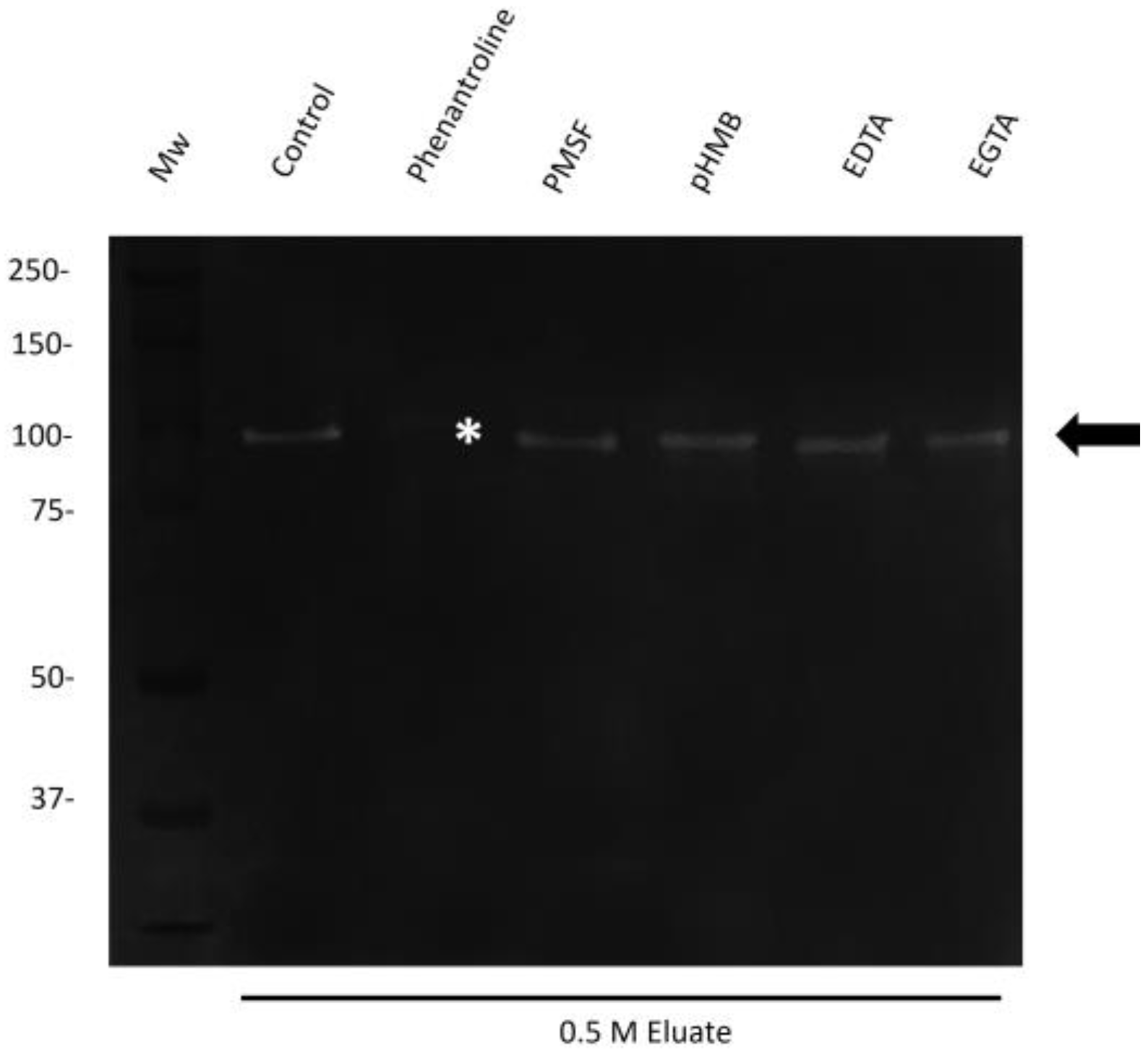
2.5. The 110 kDa Protease with Collagenase Activity Secreted by M. haemolytica A2 Belongs to the Zn-Dependent Family of Metalloproteases
2.6. The 110 kDa-Mh Protease Was Confirmed as a Zn-Dependent Metalloprotease through Mass Spectrometry
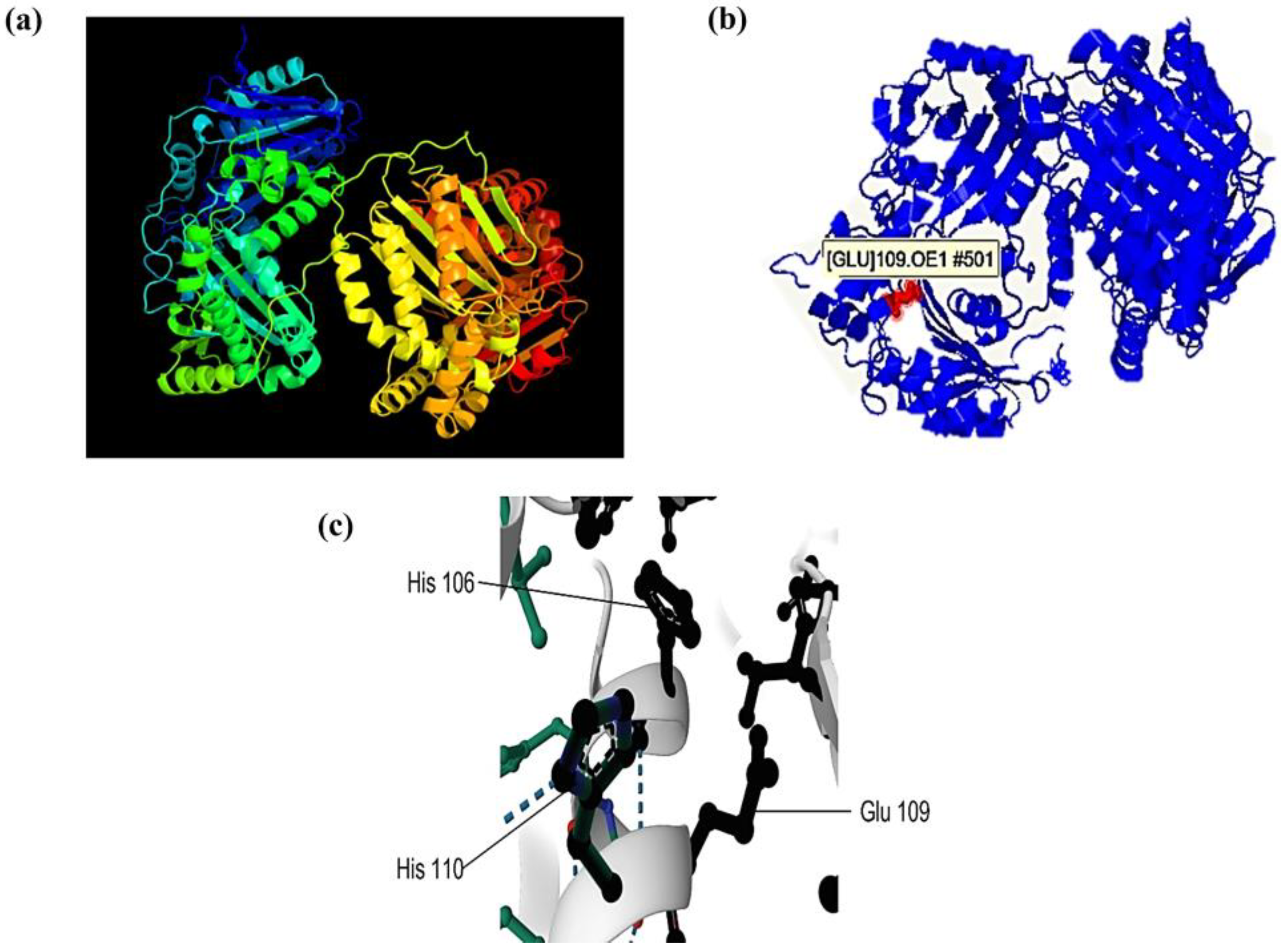
2.7. Prediction of the 3D Structure and Catalytic Site of the 110-Mh Metalloprotease
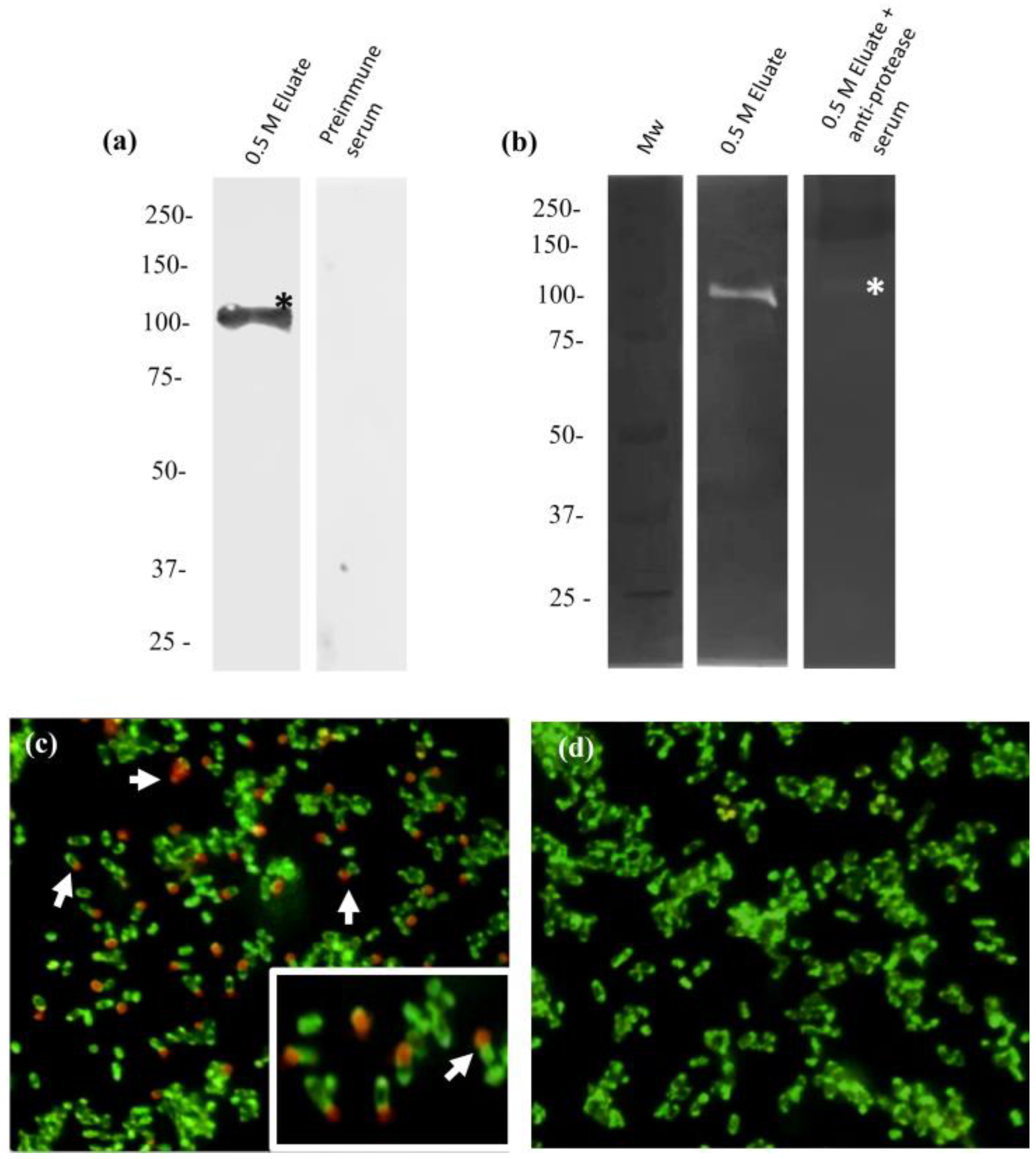
2.8. Anti-110-Mh Metalloprotease Antibodies Inhibit Its Proteolytic Activity
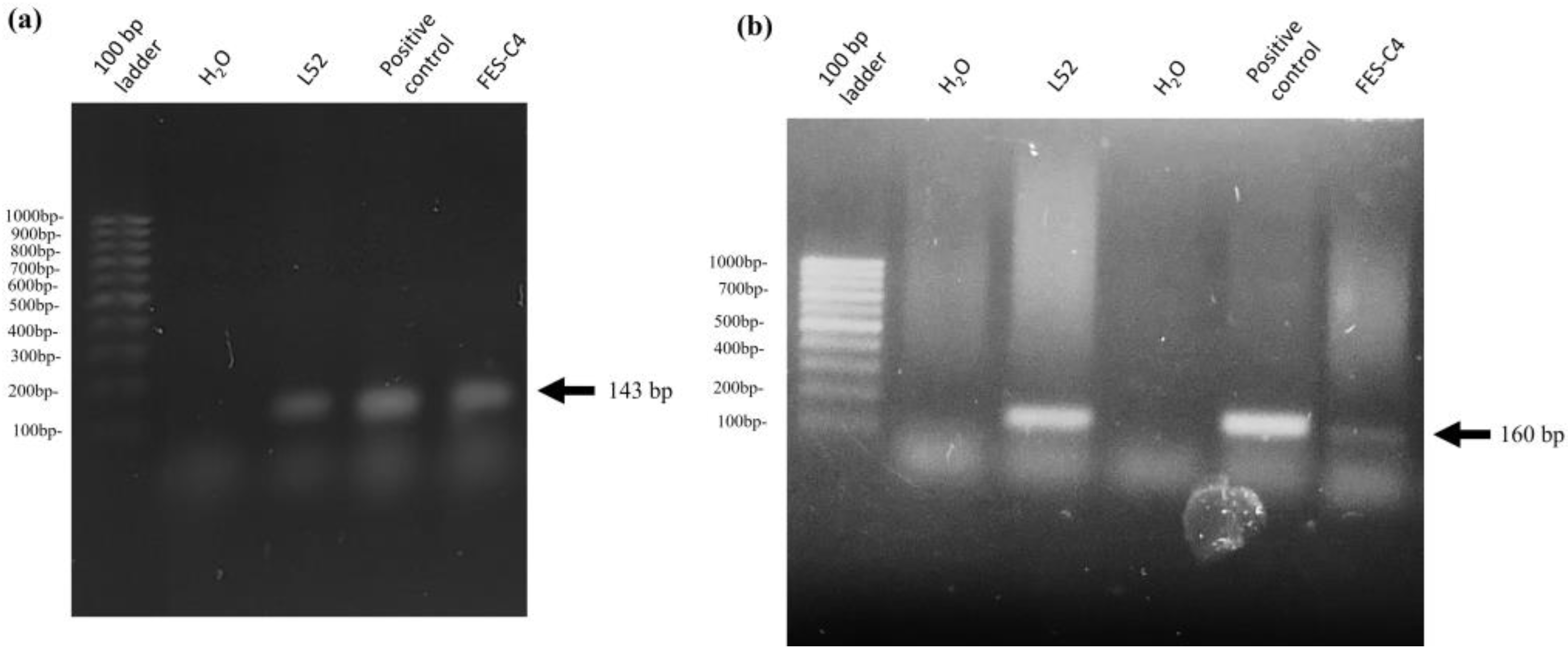
2.9. Identification of M. haemolytica A2 from a Suggestive Case of Mannheimiosis in Sheep through Histopathology and PCR
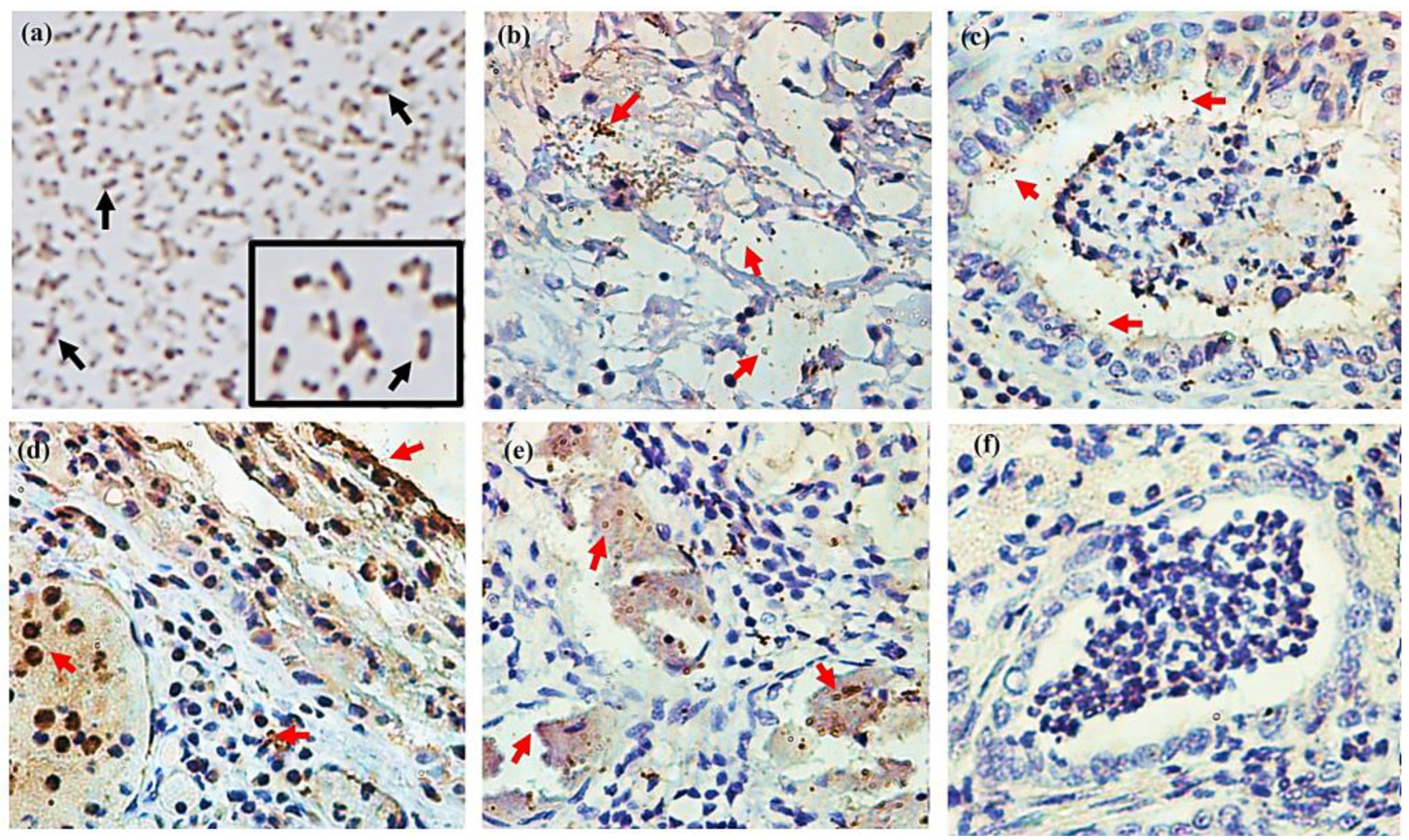
2.10. Detection of the 110-Mh Metalloprotease in Lung Samples from Sheep with Suggestive Pneumonic Damage of M. haemolytica
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains
4.2. Precipitation of Proteins from CS with (NH4)2SO4
4.3. Zymography Assays of the PpAs from CS of M. haemolytica A2
4.4. Purification of the 110 kDa Protease by Ion Exchange Chromatography
4.5. Proteolytic Degradation of Bovine Collagen Type I by the 110 kDa Protease of M. haemolytica A2
4.6. Characterization of the Proteolytic Activity of 110 kDa by Using Protease Inhibitors
4.7. Preparation of Sample for LC-MS/MS Mass Spectrometry
4.8. LC-MS/MS Mass Spectrometry
4.9. Database Search
4.10. Prediction of the 3D Structure and Catalytic Site of the 110-Mh Metalloprotease
4.11. Production of Anti-Protease Serum, Western Blotting, and Inhibition of 110-Mh Metalloprotease Activity by Antibodies
4.12. Determination and Characterization of M. haemolytica A2 Isolates from Lungs
4.13. Histopathology, Immunohistochemistry, and Immunofluorescence
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boukahil, I.; Czuprynski, C.J. Mannheimia haemolytica biofilm formation on bovine respiratory epithelial cells. Vet. Microbiol. 2016, 197, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Kirchhoff, J.; Uhlenbruck, S.; Goris, K.; Keil, G.M.; Herrler, G. Three viruses of the bovine respiratory disease complex apply different strategies to initiate infection. Vet. Res. 2014, 45, 20. [Google Scholar] [CrossRef] [PubMed]
- LeaMaster, B.R.; Evermann, J.F.; Lehmkuhl, H.D. Identification of ovine adenovirus types five and six in an epizootic of respiratory tract disease in recently weaned lambs. J. Am. Vet. Med. Assoc. 1987, 190, 1545–1547. [Google Scholar] [PubMed]
- Singh, K.; Ritchey, J.W.; Confer, A.W. Mannheimia haemolytica: Bacterial-host interactions in bovine pneumonia. Vet. Pathol. 2011, 48, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.P.; Janzen, E.D. A review of morbidity and mortality rates and disease occurrence in north american feedlot cattle. Can. Vet. J. 1986, 27, 496–500. [Google Scholar] [PubMed]
- Goodwin-Ray, K.A.; Stevenson, M.A.; Heuer, C.; Cogger, N. Economic effect of pneumonia and pleurisy in lambs in New Zealand. New Zealand Vet. J. 2008, 56, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Confer, A.W.; Ayalew, S. Mannheimia haemolytica in bovine respiratory disease: Immunogens, potential immunogens, and vaccines. Anim. Health Res. Rev. 2019, 19, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Duff, G.C.; Galyean, M.L. Board-invited review: Recent advances in management of highly stressed, newly received feedlot cattle. J. Anim. Sci. 2007, 85, 823–840. [Google Scholar] [CrossRef]
- Larson, R.L.; Step, D.L. Evidence-based effectiveness of vaccination against Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni in feedlot cattle for mitigating the incidence and effect of bovine respiratory disease complex. Vet. Clin. North Am. Food Anim. Pract. 2012, 28, 97–106. [Google Scholar] [CrossRef]
- Zecchinon, L.; Fett, T.; Desmecht, D. How Mannheimia haemolytica defeats host defence through a kiss of death mechanism. Vet. Res. 2005, 36, 133–156. [Google Scholar] [CrossRef]
- Zachary, J. Pathologic Basis of Veterinary Disease Expert Consult, 7th ed.; Elsevier: Amsterdam, The Netherlands, 2022; Volume 98, p. 496. [Google Scholar]
- Paulsen, D.B.; Confer, A.W.; Clinkenbeard, K.D.; Mosier, D.A. Pasteurella haemolytica lipopolysaccharide-induced cytotoxicity in bovine pulmonary artery endothelial monolayers: Inhibition by indomethacin. Vet. Pathol. 1995, 32, 173–183. [Google Scholar] [CrossRef]
- Belzer, C. Characterization and identification of the immunoreactive 35 kilodalton periplasmic iron-regulated protein of Mannheimia (Pasteurella) haemolvtica. Vet. Méx. 2001, 40, 293–314. [Google Scholar]
- Straus, D.C.; Jolley, W.L.; Purdy, C.W. Characterization of neuraminidases produced by various serotypes of Pasteurella haemolytica. Infect. Immun. 1993, 61, 4669–4674. [Google Scholar] [CrossRef]
- Samaniego-Barron, L.; Luna-Castro, S.; Pina-Vazquez, C.; Suarez-Guemes, F.; de la Garza, M. Two outer membrane proteins are bovine lactoferrin-binding proteins in Mannheimia haemolytica A1. Vet. Res. 2016, 47, 93. [Google Scholar] [CrossRef]
- Jeyaseelan, S.; Sreevatsan, S.; Maheswaran, S.K. Role of Mannheimia haemolytica leukotoxin in the pathogenesis of bovine pneumonic pasteurellosis. Anim. Health Res. Rev. 2002, 3, 69–82. [Google Scholar] [CrossRef]
- Ramírez-Rico, G.; Martínez-Castillo, M.; González-Ruíz, C.; Luna-Castro, S.; de la Garza, M. Mannheimia haemolytica A2 secretes different proteases into the culture medium and in outer membrane vesicles. Microb. Pathog. 2017, 113, 276–281. [Google Scholar] [CrossRef]
- Ayalew, S.; Murdock, B.K.; Snider, T.A.; Confer, A.W. Mannheimia haemolytica IgA-specific proteases. Vet. Microbiol. 2019, 239, 108487. [Google Scholar] [CrossRef]
- Barrett, A.J.; Rawlings, N.D.; Woessner, J.F. Handbook of Proteolytic Enzymes, 2nd ed.; Academic Press: Cambridge, MA, USA, 2004; Volume 2004. [Google Scholar]
- Abdullah, K.M.; Lo, R.Y.; Mellors, A. Distribution of glycoprotease activity and the glycoprotease gene among serotypes of Pasteurella haemolytica. Biochem. Soc. Trans. 1990, 18, 901–903. [Google Scholar] [CrossRef]
- Highlander, S.K. Molecular genetic analysis of virulence in Mannheimia (pasteurella) haemolytica. Front. Biosci. 2001, 6, D1128–D1150. [Google Scholar] [CrossRef]
- Lo, R.Y.; Strathdee, C.A.; Shewen, P.E. Nucleotide sequence of the leukotoxin genes of Pasteurella haemolytica A1. Infect. Immun. 1987, 55, 1987–1996. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.S.; Maheswaran, S.K.; Lin, G.; Townsend, E.L.; Ames, T.R. Induction of inflammatory cytokines in bovine alveolar macrophages following stimulation with Pasteurella haemolytica lipopolysaccharide. Infect. Immun. 1995, 63, 381–388. [Google Scholar] [CrossRef]
- Lafleur, R.L.; Abrahamsen, M.S.; Maheswaran, S.K. The biphasic mRNA expression pattern of bovine interleukin-8 in Pasteurella haemolytica lipopolysaccharide-stimulated alveolar macrophages is primarily due to tumor necrosis factor alpha. Infect. Immun. 1998, 66, 4087–4092. [Google Scholar] [CrossRef]
- Maheswaran, S.K.; Weiss, D.J.; Kannan, M.S.; Townsend, E.L.; Reddy, K.R.; Whiteley, L.O.; Srikumaran, S. Effects of Pasteurella haemolytica A1 leukotoxin on bovine neutrophils: Degranulation and generation of oxygen-derived free radicals. Vet. Immunol. Immunopathol. 1992, 33, 51–68. [Google Scholar] [CrossRef]
- Cudd, L.; Clarke, C.; Clinkenbeard, K.; Shelton, M.; Clinkenbeard, P.; Murphy, G. Role of intracellular calcium in Pasteurella haemolytica leukotoxin-induced bovine neutrophil leukotriene B4 production and plasma membrane damage. FEMS Microbiol. Lett. 1999, 172, 123–129. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Hirano, A.; Arakawa, T.; Shiraki, K. Mechanistic insights into protein precipitation by alcohol. Int. J. Biol. Macromol. 2012, 50, 865–871. [Google Scholar] [CrossRef]
- Avalos-Gomez, C.; Reyes-Lopez, M.; Ramirez-Rico, G.; Diaz-Aparicio, E.; Zenteno, E.; Gonzalez-Ruiz, C.; de la Garza, M. Effect of apo-lactoferrin on leukotoxin and outer membrane vesicles of Mannheimia haemolytica A2. Vet. Res. 2020, 51, 36. [Google Scholar] [CrossRef]
- Ramirez-Rico, G.; Martinez-Castillo, M.; Avalos-Gomez, C.; de la Garza, M. Bovine apo-lactoferrin affects the secretion of proteases in Mannheimia haemolytica A2. Access Microbiol. 2021, 3, 000269. [Google Scholar] [CrossRef]
- Schafer, F.Q.; Qian, S.Y.; Buettner, G.R. Iron and free radical oxidations in cell membranes. Cell Mol. Biol. 2000, 46, 657–662. [Google Scholar]
- Haney, E.F.; Nazmi, K.; Bolscher, J.G.; Vogel, H.J. Structural and biophysical characterization of an antimicrobial peptide chimera comprised of lactoferricin and lactoferrampin. Biochim. Biophys. Acta 2012, 1818, 762–775. [Google Scholar] [CrossRef] [PubMed]
- Periayah, M.H.; Halim, A.S.; Mat Saad, A.Z. Mechanism Action of Platelets and Crucial Blood Coagulation Pathways in Hemostasis. Int. J. Hematol. Oncol. Stem. Cell Res. 2017, 11, 319–327. [Google Scholar] [PubMed]
- Negrete-Abascal, E.; Tenorio, V.R.; Guerrero, A.L.; García, R.M.; Reyes, M.E.; De la Garza, M. Purification and characterization of a protease from Actinobacillus pleuropneumoniae serotype 1, an antigen common to all the serotypes. Can. J. Vet. Res. 1998, 62, 183–190. [Google Scholar] [PubMed]
- Beynon, R.J.; Bond, J.S. Proteolytic Enzymes: A Practical Approach, 2nd ed.; Oxford University Press: Oxford, UK, 2001; pp. 1–359. [Google Scholar]
- Jackson, R.J.; Lim, D.V.; Dao, M.L. Identification and analysis of a collagenolytic activity in Streptococcus mutans. Curr. Microbiol. 1997, 34, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Castillo, M.; Cardenas-Guerra, R.E.; Arroyo, R.; Debnath, A.; Rodriguez, M.A.; Sabanero, M.; Flores-Sanchez, F.; Navarro-Garcia, F.; Serrano-Luna, J.; Shibayama, M. Nf-GH, a glycosidase secreted by Naegleria fowleri, causes mucin degradation: An in vitro and in vivo study. Future Microbiol. 2017, 12, 781–799. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, S.; Shinoda, S. Microbial metalloproteases and pathogenesis. Microbes Infect. 2000, 2, 91–98. [Google Scholar] [CrossRef]
- Garcia Gonzalez, O.; Garcia, R.M.; de la Garza, M.; Vaca, S.; Paniagua, G.L.; Mejia, R.; Tenorio, V.R.; Negrete-Abascal, E. Actinobacillus pleuropneumoniae metalloprotease: Cloning and in vivo expression. FEMS Microbiol. Lett. 2004, 234, 81–86. [Google Scholar] [CrossRef]
- Shewen, P.E.; Lee, C.W.; Perets, A.; Hodgins, D.C.; Baldwin, K.; Lo, R.Y. Efficacy of recombinant sialoglycoprotease in protection of cattle against pneumonic challenge with Mannheimia (Pasteurella) haemolytica A1. Vaccine 2003, 21, 1901–1906. [Google Scholar] [CrossRef]
- Conlon, J.A.; Shewen, P.E.; Lo, R.Y. Efficacy of recombinant leukotoxin in protection against pneumonic challenge with live Pasteurella haemolytica A1. Infect. Immun. 1991, 59, 587–591. [Google Scholar] [CrossRef]
- McNeil, H.J.; Shewen, P.E.; Lo, R.Y.; Conlon, J.A.; Miller, M.W. Novel protease produced by a Pasteurella trehalosi serotype 10 isolate from a pneumonic bighorn sheep: Characteristics and potential relevance to protection. Vet. Microbiol. 2003, 93, 145–152. [Google Scholar] [CrossRef]
- Matsushita, O.; Koide, T.; Kobayashi, R.; Nagata, K.; Okabe, A. Substrate recognition by the collagen-binding domain of Clostridium histolyticum class I collagenase. J. Biol. Chem. 2001, 276, 8761–8770. [Google Scholar] [CrossRef]
- Mandl, I.; Maclennan, J.D.; Howes, E.L. Isolation and characterization of proteinase and collagenase from Cl. histolyticum. J. Clin. Invest 1953, 32, 1323–1329. [Google Scholar] [CrossRef]
- Awad, M.M.; Ellemor, D.M.; Bryant, A.E.; Matsushita, O.; Boyd, R.L.; Stevens, D.L.; Emmins, J.J.; Rood, J.I. Construction and virulence testing of a collagenase mutant of Clostridium perfringens. Microb. Pathog. 2000, 28, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Grab, D.J.; Kennedy, R.; Philipp, M.T. Borrelia burgdorferi possesses a collagenolytic activity. FEMS Microbiol. Lett. 1996, 144, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, S.; Nitanda, Y.; Fujii, K.; Kawahara, K.; Li, T.; Maehara, Y.; Ramamurthy, T.; Takeda, Y.; Shinoda, S. Differential gene expression and extracellular secretion of the collagenolytic enzymes by the pathogen Vibrio parahaemolyticus. FEMS Microbiol. Lett. 2008, 283, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Uitto, V.J.; Baillie, D.; Wu, Q.; Gendron, R.; Grenier, D.; Putnins, E.E.; Kanervo, A.; Firth, J.D. Fusobacterium nucleatum increases collagenase 3 production and migration of epithelial cells. Infect. Immun. 2005, 73, 1171–1179. [Google Scholar] [CrossRef] [PubMed]
- Fives-Taylor, P.M.; Meyer, D.H.; Mintz, K.P.; Brissette, C. Virulence factors of Actinobacillus actinomycetemcomitans. Periodontol. 2000 1999, 20, 136–167. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.K.; Wright, L.G. The involvement of collagenase in the necrosis induced by the bites of some spiders. Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1992, 102, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Kassegne, K.; Hu, W.; Ojcius, D.M.; Sun, D.; Ge, Y.; Zhao, J.; Yang, X.F.; Li, L.; Yan, J. Identification of Collagenase as a Critical Virulence Factor for Invasiveness and Transmission of Pathogenic Leptospira Species. J. Infect. Dis. 2013, 209, 1105–1115. [Google Scholar] [CrossRef]
- Burgstaller, G.; Oehrle, B.; Gerckens, M.; White, E.S.; Schiller, H.B.; Eickelberg, O. The instructive extracellular matrix of the lung: Basic composition and alterations in chronic lung disease. Eur. Respir. J. 2017, 50, 1601805. [Google Scholar] [CrossRef]
- Suki, B.; Ito, S.; Stamenovic, D.; Lutchen, K.R.; Ingenito, E.P. Biomechanics of the lung parenchyma: Critical roles of collagen and mechanical forces. J. Appl. Physiol. 2005, 98, 1892–1899. [Google Scholar] [CrossRef]
- Bosman, F.T.; Stamenkovic, I. Functional structure and composition of the extracellular matrix. J. Pathol. 2003, 200, 423–428. [Google Scholar] [CrossRef]
- Starr, A.E.; Dan, T.; Minhas, K.; Shewen, P.E.; Coomber, B.L. Potential involvement of gelatinases and their inhibitors in Mannheimia haemolytica pneumonia in cattle. Infect. Immun. 2004, 72, 4393–4400. [Google Scholar] [CrossRef] [PubMed]
- Duong-Ly, K.C.; Gabelli, S.B. Salting out of proteins using ammonium sulfate precipitation. Methods Enzym. 2014, 541, 85–94. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Negrete-Abascal, E.; Tenorio, V.R.; Serrano, J.J.; Garcia, C.; de la Garza, M. Secreted proteases from Actinobacillus pleuropneumoniae serotype 1 degrade porcine gelatin, hemoglobin and immunoglobulin A. Can. J. Vet. Res. 1994, 58, 83–86. [Google Scholar]
- Meneses-Romero, E.; Hernandez-Orihuela, L.; Pando-Robles, V.; Lopez, T.D.; Oses-Prieto, J.A.; Burlingame, A.L.; Batista, C.V.F. Quantitative proteomic analysis reveals high interference on protein expression of H9c2 cells activated with glucose and cardiotonic steroids. J. Proteom. 2020, 211, 103536. [Google Scholar] [CrossRef]
- Kelley, L.A.; Mezulis, S.; Yates, C.M.; Wass, M.N.; Sternberg, M.J. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protoc. 2015, 10, 845–858. [Google Scholar] [CrossRef]
- Jakubec, D.; Skoda, P.; Krivak, R.; Novotny, M.; Hoksza, D. PrankWeb 3: Accelerated ligand-binding site predictions for experimental and modelled protein structures. Nucleic. Acids. Res. 2022, 50, W593–W597. [Google Scholar] [CrossRef]
- Greenfield, E.A.; DeCaprio, J.; Brahmandam, M. Preparing Protein Antigens from Sodium Dodecyl Sulfate-Polyacrylamide Gels for Immunization. Cold Spring Harb. Protoc. 2019, 2019, pdb-prot100008. [Google Scholar] [CrossRef]
- Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc. Natl. Acad. Sci. USA 1979, 76, 4350–4354. [Google Scholar] [CrossRef]
- Klima, C.L.; Zaheer, R.; Briggs, R.E.; McAllister, T.A. A multiplex PCR assay for molecular capsular serotyping of Mannheimia haemolytica serotypes 1, 2, and 6. J. Microbiol. Methods 2017, 139, 155–160. [Google Scholar] [CrossRef]
- Edward Moore, E.; Arnscheidt, A.; Kruger, A.; Strömpl, C.; Mau, M. Simplified protocols for the preparation of genomic DNA from bacterial cultures. Mol. Microb. Ecol. Man. 2004, 1, 3–18. [Google Scholar]
- Kisiela, D.I.; Czuprynski, C.J. Identification of Mannheimia haemolytica adhesins involved in binding to bovine bronchial epithelial cells. Infect. Immun. 2009, 77, 446–455. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez-Rico, G.; Martinez-Castillo, M.; Ruiz-Mazón, L.; Meneses-Romero, E.P.; Palacios, J.A.F.; Díaz-Aparicio, E.; Abascal, E.N.; de la Garza, M. Identification, Biochemical Characterization, and In Vivo Detection of a Zn-Metalloprotease with Collagenase Activity from Mannheimia haemolytica A2. Int. J. Mol. Sci. 2024, 25, 1289. https://doi.org/10.3390/ijms25021289
Ramírez-Rico G, Martinez-Castillo M, Ruiz-Mazón L, Meneses-Romero EP, Palacios JAF, Díaz-Aparicio E, Abascal EN, de la Garza M. Identification, Biochemical Characterization, and In Vivo Detection of a Zn-Metalloprotease with Collagenase Activity from Mannheimia haemolytica A2. International Journal of Molecular Sciences. 2024; 25(2):1289. https://doi.org/10.3390/ijms25021289
Chicago/Turabian StyleRamírez-Rico, Gerardo, Moises Martinez-Castillo, Lucero Ruiz-Mazón, Erika Patricia Meneses-Romero, José Arturo Flores Palacios, Efrén Díaz-Aparicio, Erasmo Negrete Abascal, and Mireya de la Garza. 2024. "Identification, Biochemical Characterization, and In Vivo Detection of a Zn-Metalloprotease with Collagenase Activity from Mannheimia haemolytica A2" International Journal of Molecular Sciences 25, no. 2: 1289. https://doi.org/10.3390/ijms25021289





