Membrane Transporter of Serotonin and Hypercholesterolemia in Children
Abstract
1. Introduction
2. Results
2.1. Laboratory and Instrumental Characteristics of the Study Groups
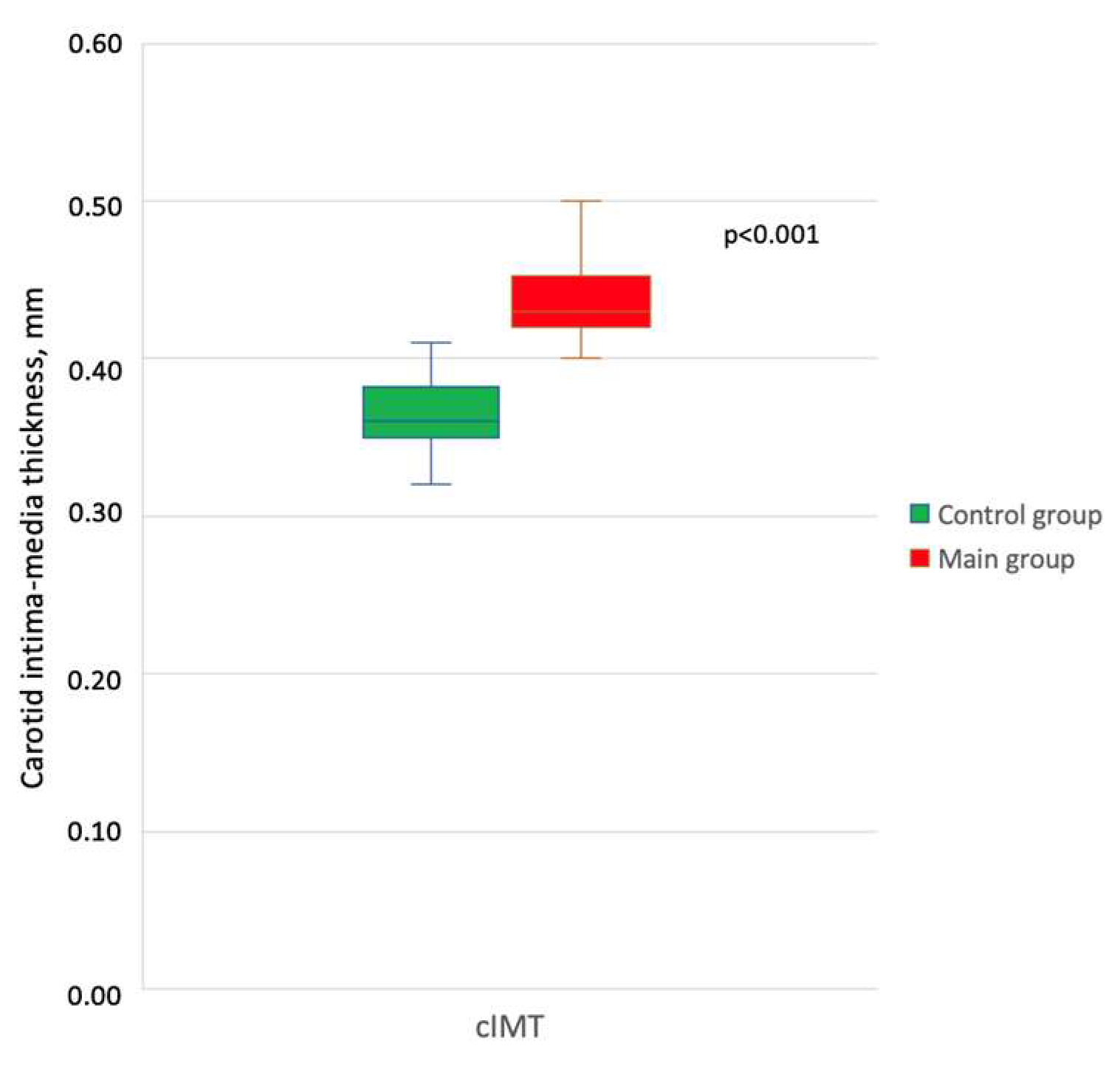
2.2. Main Results and Their Comparison
2.3. Spearman Correlation
3. Discussion
4. Materials and Methods
4.1. Recruitment of Study Groups
4.2. Instrumental Examination
4.3. Determination of Serotonin and Serotonin Membrane Transporter
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mercado, C.P.; Kilic, F. Molecular mechanisms of SERT in platelets: Regulation of plasma serotonin levels. Mol. Interv. 2010, 10, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Ziu, E.; Mercado, C.P.; Li, Y.; Singh, P.; Ahmed, B.A.; Freyaldenhoven, S.; Lensing, S.; Ware, J.; Kilic, F. Down-regulation of the serotonin transporter in hyperreactive platelets counteracts the pro-thrombotic effect of serotonin. J. Mol. Cell. Cardiol. 2012, 52, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Haney, M.; Calarge, C.; Bliziotes, M. Clinical Implications of Serotonin Regulation of Bone Mass. In Translational Endocrinology of Bone; Karsenty, G., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 16, pp. 189–198. [Google Scholar]
- Rudnick, G. Active transport of 5-hydroxytryptamine by plasma membrane vesicles isolated from human blood platelets. J. Biol. Chem. 1977, 252, 2170–2174. [Google Scholar] [CrossRef] [PubMed]
- Fraer, M.; Kilic, F. Serotonin: A different player in hypertension-associated thrombosis. Hypertension 2015, 65, 942–948. [Google Scholar] [CrossRef] [PubMed]
- Mindubayeva, F.; Niyazova, Y.; Nigmatullina, R.; Kabiyeva, S.; Salikhova, Y. The system of serotonin and its metabolism in platelets in children with congenital heart defects of an early age. Georgian Med. News 2020, 298, 42–46. [Google Scholar]
- Ni, W.; Watts, S.W. 5-hydroxytryptamine in the cardiovascular system: Focus on the serotonin transporter (SERT). Clin. Exp. Pharmacol. Physiol. 2006, 33, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Nigmatullina, R.R.; Matveeva, V.L.; Chibireva, M.D. The influence of selective serotonin re-uptake inhibitor fluoxetine on inotropic function of myocardium in the ontogenesis of rats. Rossiiskii fiziologicheskii zhurnal imeni IM Sechenova 2014, 100, 348–359. [Google Scholar]
- Gillis, C.N.; Pitt, B.R. The fate of circulating amines within the pulmonary circulation. Annu. Rev. Physiol. 1982, 44, 269–281. [Google Scholar] [CrossRef]
- Balkovetz, D.F.; Tiruppathi, C.; Leibach, F.H.; Mahesh, V.B.; Ganapathy, V. Evidence for an imipramine-sensitive serotonin transporter in human placental brush-border membranes. J. Biol. Chem. 1989, 264, 2195–2198. [Google Scholar] [CrossRef]
- Gabrielsen, M.; Ravna, A.W.; Kristiansen, K.; Sylte, I. Substrate binding and translocation of the serotonin transporter studied by docking and molecular dynamics simulations. J. Mol. Model. 2012, 3, 1073–1085. [Google Scholar] [CrossRef]
- Brenner, B.; Harney, J.T.; Ahmed, B.A.; Jeffus, B.C.; Unal, R.; Mehta, J.L.; Kilic, F. Plasma serotonin levels and the platelet serotonin transporter. J. Neurochem. 2007, 1, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, N.; Allaeys, I.; Marcoux, G.; Machlus, K.R.; Mailhot, B.; Zufferey, A.; Levesque, T.; Becker, Y.; Tessandier, N.; Melki, I.; et al. Platelets release pathogenic serotonin and return to circulation after immune complex-mediated sequestration. Proc. Natl. Acad. Sci. USA 2018, 115, 1550–1559. [Google Scholar] [CrossRef] [PubMed]
- Bjerregaard, H.; Severinsen, K.; Said, S.; Wiborg, O.; Sinning, S. A dualistic conformational response to substrate binding in the human serotonin transporter reveals a high affinity state for serotonin. J. Biol. Chem. 2015, 290, 7747–7755. [Google Scholar] [CrossRef] [PubMed]
- Deveau, C.M.; Rodriguez, E.; Schroering, A.; Yamamoto, B.K. Serotonin transporter regulation by cholesterol-independent lipid signaling. Biochem. Pharmacol. 2021, 183, 114349. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, M.; Masetti, M.; Recanatini, M.; Cavalli, A.; Bottegoni, G. Mapping Cholesterol Interaction Sites on Serotonin Transporter through Coarse-Grained Molecular Dynamics. PLoS ONE 2016, 11, e0166196. [Google Scholar] [CrossRef] [PubMed]
- Scanlon, S.M.; Williams, D.C.; Schloss, P. Membrane cholesterol modulates serotonin transporter activity. Biochemistry 2001, 40, 10507–10513. [Google Scholar] [CrossRef] [PubMed]
- Frishman, W.H.; Grewall, P. Serotonin and the heart. Ann. Med. 2000, 32, 195–209. [Google Scholar] [CrossRef]
- Kanova, M.; Kohout, P. Serotonin-Its Synthesis and Roles in the Healthy and the Critically Ill. Int. J. Mol. Sci. 2021, 22, 4837. [Google Scholar] [CrossRef]
- Neumann, J.; Hofmann, B.; Dhein, S.; Gergs, U. Cardiac Roles of Serotonin (5-HT) and 5-HT-Receptors in Health and Disease. Int. J. Mol. Sci. 2023, 24, 4765. [Google Scholar] [CrossRef]
- Tarbit, E.; Singh, I.; Peart, J.N.; Bivol, S.; Rose’Meyer, R.B. Increased release of serotonin from rat primary isolated adult cardiac myofibroblasts. Sci. Rep. 2021, 11, 20376. [Google Scholar] [CrossRef]
- Oldfield, C.J.; Moffatt, T.L.; O’Hara, K.A.; Xiang, B.; Dolinsky, V.W.; Duhamel, T.A. Muscle-specific sirtuin 3 overexpression does not attenuate the pathological effects of high-fat/high-sucrose feeding but does enhance cardiac SERCA2a activity. Physiol. Rep. 2021, 9, 14961. [Google Scholar] [CrossRef] [PubMed]
- Rieder, M.; Gauchel, N.; Bode, C.; Duerschmied, D. Serotonin: A platelet hormone modulating cardiovascular disease. J. Thromb. Thrombolysis 2021, 52, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liang, X.; Li, C.; Li, R.; Tong, X.; Zhang, R.; Shan, X.; Yang, J.; Ma, X.; Lu, W.; et al. 5-HT2A Receptor and 5-HT Degradation Play a Crucial Role in Atherosclerosis by Modulating Macrophage Foam Cell Formation, Vascular Endothelial Cell Inflammation, and Hepatic Steatosis. J. Atheroscler. Thromb. 2022, 29, 322–336. [Google Scholar] [CrossRef] [PubMed]
- Shimabukuro, M. Serotonin and Atheroscelotic Cardiovascular Disease. J. Atheroscler. Thromb. 2022, 29, 315–316. [Google Scholar] [CrossRef] [PubMed]
- Shan, J.; Khelashvili, G.; Mondal, S.; Mehler, E.L.; Weinstein, H. Ligand-dependent conformations and dynamics of the serotonin 5-HT(2A) receptor determine its activation and membrane-driven oligomerization properties. PLoS Comput. Biol. 2012, 8, e1002473. [Google Scholar] [CrossRef] [PubMed]
- Vikenes, K.; Farstad, M.; Nordrehaug, J.E. Serotonin is associated with coronary artery disease and cardiac events. Circulation 1999, 100, 483–489. [Google Scholar] [CrossRef]
- Ren, S.; Qian, S.; Wang, W.; Liu, J.; Liu, P. Prospective study of sarpogrelate hydrochloride on patients with arteriosclerosis obliterans. Ann. Thorac. Cardiovasc. Surg. 2013, 19, 30–34. [Google Scholar] [CrossRef]
- Di Taranto, M.D.; Fortunato, G. Genetic heterogeneity of Familial hypercholesterolemia: Repercussions for molecular diagnosis. Int. J. Mol. Sci. 2023, 24, 3224. [Google Scholar] [CrossRef]
- Sadykova, D.I.; Galimova, L.F. Family hypercholesterolaemia at children: Clinic, diagnostics, treatment. Rossiyskiy Vestnik Perinatologii i Pediatrii 2017, 62, 119–123. [Google Scholar] [CrossRef][Green Version]
- Ezhov, M.V.; Bazhan, S.S.; Ershova, A.I.; Meshkov, A.N.; Sokolov, A.A.; Kukharchuk, V.V.; Gurevich, V.S.; Voevoda, M.I.; Sergienko, I.V.; Shakhtshneider, E.V.; et al. Clinical guidelines for familial hypercholesterolemia. Ateroscleroz 2019, 15, 58–98. [Google Scholar]
- Beheshti, S.O.; Madsen, C.M.; Varbo, A.; Nordestgaard, B.G. Worldwide prevalence of Familial hypercholesterolemia: Meta-analyses of 11 million subjects. J. Am. Coll. Cardiol. 2020, 75, 2553–2566. [Google Scholar] [CrossRef] [PubMed]
- Mainieri, F.; Tagi, V.M.; Chiarelli, F. Recent advances on Familial Hypercholesterolemia in children and adolescents. Biomedicines 2022, 10, 1043. [Google Scholar] [CrossRef] [PubMed]
- Wiegman, A.; Gidding, S.S.; Watts, G.F. European Atherosclerosis Society Consensus Panel. Familial hypercholesterolaemia in children and adolescents: Gaining decades of life by optimizing detection and treatment. Eur. Heart J. 2015, 35, 2425–2437. [Google Scholar] [CrossRef] [PubMed]
- Berenson, G.S.; Srinivasan, S.R.; Bao, W.; Newman, W.P.; Tracy, R.E.; Wattigney, W.A. Association between multiple cardiovascular risk factors and atherosclerosis in children and young adults. The Bogalusa Heart Study. N. Engl. J. Med. 1998, 338, 1650–1656. [Google Scholar] [CrossRef] [PubMed]
- Plana, N.; Rodríguez-Borjabad, C.; Ibarretxe, D.; Ferré, R.; Feliu, A.; Caselles, A. Lipid and lipoprotein parameters for detection of familial hypercholesterolemia in childhood. The DECOPIN Project. Clin. Investig. Arterioscler. 2018, 30, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Townsend, R.R.; Wilkinson, I.B.; Schiffrin, E.L.; Avolio, A.P.; Chirinos, J.A.; Cockcroft, J.R.; Heffernan, K.S.; Lakatta, E.G.; McEniery, C.M.; Mitchell, G.F.; et al. Recommendations for improving and standardizing vascular research on arterial stiffness. A Scientific Statement from the American Heart Association. J. Hypertens. 2015, 66, 698–722. [Google Scholar] [CrossRef] [PubMed]
- Ravikumar, R.; Deepa, R.; Shanthirani, C.; Mohan, V. Comparison of carotid intima-media thickness, arterial stiffness, and brachial artery flow mediated dilatation in diabetic and nondiabetic subjects (The Chennai Urban-Population Study [CUPS-9]). Am. J. Cardiol. 2002, 90, 702–707. [Google Scholar] [CrossRef]
- Vasyuk, Y.A.; Ivanova, S.V.; Shkolnik, E.L.; Kotovskaya, Y.V.; Milyagin, V.A.; Oleynikov, V.E.; Orlova, Y.A.; Sumin, A.N.; Baranov, A.A.; Boytsov, S.A.; et al. Consensus of Russian experts on the evaluation of arterial stiffness in clinical practice. Cardiovasc. Ther. Prev. 2016, 15, 4–19. [Google Scholar] [CrossRef]
- Vallée, A.; Cinaud, A.; Protogerou, A.; Zhang, Y.; Topouchian, J.; Safar, M.E.; Blacher, J. Arterial stiffness and coronary ischemia: New aspects and paradigms. Curr. Hypertens. Rep. 2020, 22, 5. [Google Scholar] [CrossRef]
- Kim, D.A.; McClure, W.G.; Neighoff, J.B.; Vaidya, D.; Williams, M.S. Platelet response to serotonin in patients with stable coronary heart disease. Am. J. Cardiol. 2014, 114, 181–186. [Google Scholar] [CrossRef]
- Fouquet, G.; Coman, T.; Hermine, O.; Côté, F. Serotonin, hematopoiesis and stem cells. Pharmacol. Res. 2019, 140, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Ni, W.; Geddes, T.J.; Priestley, J.R.; Szasz, T.; Kuhn, D.M.; Watts, S.W. The existence of a local 5-hydroxytryptaminergic system in peripheral arteries. Br. J. Pharmacol. 2008, 154, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Chun, E.J.; Hur, J.H.; Min, S.H.; Lee, J.E.; Oh, T.J.; Kim, K.M.; Jang, H.C.; Han, S.J.; Kang, D.K.; et al. Effect of sarpogrelate, a selective 5-HT2A receptor antagonist, on characteristics of coronary artery disease in patients with type 2 diabetes. Atherosclerosis 2017, 257, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Aflyatumova, G.N.; Nigmatullina, R.R.; Sadykova, D.I.; Chibireva, M.D.; Fugetto, F.; Serra, R. Endothelin-1, nitric oxide, serotonin and high blood pressure in male adolescents. Vasc. Health Risk Manag. 2018, 14, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Watts, S.W. 5-HT in systemic hypertension: Foe, friend or fantasy? Clin. Sci. 2005, 108, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Chistiakov, D.A.; Sobenin, I.A.; Orekhov, A.N.; Bobryshev, Y.V. Human miR-221/222 in Physiological and Atherosclerotic Vascular Remodeling. BioMed Res. Int. 2015, 2015, 354517. [Google Scholar] [CrossRef]
- Jaminon, A.; Reesink, K.; Kroon, A.; Schurgers, L. The Role of Vascular Smooth Muscle Cells in Arterial Remodeling: Focus on Calcification-Related Processes. Int. J. Mol. Sci. 2019, 20, 5694. [Google Scholar] [CrossRef]
- Kovács, B.; Cseprekál, O.; Diószegi, Á.; Lengyel, S.; Maroda, L.; Paragh, G.; Harangi, M.; Páll, D. The Importance of Arterial Stiffness Assessment in Patients with Familial Hypercholesterolemia. J. Clin. Med. 2022, 11, 2872. [Google Scholar] [CrossRef]
- Morotti, A.; Barale, C.; Melchionda, E.; Russo, I. Platelet Redox Imbalance in Hypercholesterolemia: A Big Problem for a Small Cell. Int. J. Mol. Sci. 2022, 23, 11446. [Google Scholar] [CrossRef]
- Koba, S.; Pakala, R.; Watanabe, T.; Katagiri, T.; Benedict, C.R. Vascular smooth muscle proliferation: Synergistic interaction between serotonin and low-density lipoproteins. J. Am. Coll. Cardiol. 1999, 34, 1644–1651. [Google Scholar] [CrossRef]
- Han, D.; Choi, J.H.; Kim, S.; Park, S.M.; Shin, D.G.; Kang, M.K.; Choi, S.; Lee, N.; Cho, J.R. Changes in serum serotonin levels in patients with acute coronary syndrome and stable angina undergoing percutaneous coronary intervention. J. Int. Med. Res. 2020, 48, 300060520970104. [Google Scholar] [CrossRef] [PubMed]
- Ayme-Dietrich, E.; Aubertin-Kirch, G.; Maroteaux, L.; Monassier, L. Cardiovascular remodeling and the peripheral serotonergic system. Arch. Cardiovasc. Dis. 2017, 110, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Ban, Y.; Watanabe, T.; Miyazaki, A.; Nakano, Y.; Tobe, T.; Idei, T.; Iguchi, T.; Ban, Y.; Katagiri, T. Impact of increased plasma serotonin levels and carotid atherosclerosis on vascular dementia. Atherosclerosis 2007, 195, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Elkins, C.; Fruh, S.; Jones, L.; Bydalek, K. Clinical Practice Recommendations for Pediatric Dyslipidemia. J. Pediatr. Health Care 2019, 33, 494–504. [Google Scholar] [CrossRef]
- WHO. Obesity and Overweight. Available online: https://www.who.int/ru/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 18 November 2023).

| Main Group | Control Group | p 1 | |||
|---|---|---|---|---|---|
| Mean | CI | Mean | CI | ||
| TC, mg/dL 2 | 259.02 | 150.77–371.14 | 150.77 | 96.65–204.9 | <0.001 |
| HDL-cholesterol, mg/dL | 57.99 | 34.79–77.32 | 61.86 | 42.53–85.05 | 0.004 |
| LDL-cholesterol, mg/dL | 185.57 | 81.19–289.95 | 85.05 | 46.39–119.85 | <0.001 |
| TG, mg/dL | 87.5 | 43.75–131.25 | 70 | 17.5–131.25 | 0.003 |
| Index | Main Group | Control Group | p 1 | ||
|---|---|---|---|---|---|
| Median | Q1–Q3 | Median | Q1–Q3 | ||
| PWV min, m/s 2 | 4.1 | 3.5–5.6 | 3.3 | 2.8–4.1 | <0.001 |
| PWV, m/s | 6.9 | 5.5–8 | 4.4 | 3.7–5.1 | <0.001 |
| PWV max, m/s | 8.5 | 7.3–10.6 | 6.6 | 5.8–7.4 | <0.001 |
| AIxmin, % | −73.5 | −82–−67 | −75 | −84–−69 | 0.179 |
| Aixmean, % | −39.5 | −47–−35 | −51 | −58–−45 | <0.001 |
| AIxmax, % | 9.5 | 3–30 | 7.5 | −3–26 | 0.179 |
| AASI | 0.5 | 0.2–0.8 | 0.4 | 0.2–0.7 | <0.001 |
| Main Group | Control Group | p 1 | q | |||
|---|---|---|---|---|---|---|
| Median | Q1–Q3 | Median | Q1–Q3 | |||
| Plasma 5-HT (pmol/mL) 2 | 96.7 | 24.2–175.2 | 66.2 | 42.4–79.2 | <0.001 | <0.001 |
| Platelets 5-HT (pmol/109 platelets) | 0.03 | 0.01–0.5 | 0.02 | 0.01–0.03 | 0.007 | 0.014 |
| Mean | CI | Mean | CI | |||
| Platelets 5-HT (pmol/mL) | 8,530,370 | 2,326,366–14,734,712 | 6,235,403 | 723,560–11,747,293 | <0.001 | 0.004 |
| SERT, ng/mL | 0.04 | 0.02–0.07 | 0.03 | 0.01–0.06 | <0.001 | 0.002 |
| 5-HIAA in blood plasma (pmol/mL) | 27.2 | 14.2–40.1 | 20.9 | 8.4–33.5 | <0.001 | <0.001 |
| cIMT 1 | PWVmin | PWV | PWVmax | AASI | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ρ 2 | p | ρ | p | ρ | p | ρ | p | ρ | p | |
| 5-HT in plasma 3 | 0.5 | <0.001 | 0.4 | <0.001 | 0.6 | <0.001 | 0.5 | <0.001 | 0.3 | <0.001 |
| Platelets 5-HT | 0.4 | <0.001 | 0.3 | 0.001 | 0.5 | <0.001 | 0.4 | <0.001 | 0.3 | <0.001 |
| 5-HT in 1 platelet | 0.3 | <0.001 | 0.3 | 0.002 | 0.4 | <0.001 | 0.4 | <0.001 | 0.4 | <0.001 |
| 5-HIAA | 0.4 | <0.001 | 0.2 | 0.0026 | 0.4 | <0.001 | 0.4 | <0.001 | 0.3 | 0.001 |
| SERT | 0.4 | <0.001 | 0.3 | <0.001 | 0.5 | <0.001 | 0.4 | <0.001 | 0.3 | <0.001 |
| 5-HT in plasma 3 | 0.5 | <0.001 | 0.4 | <0.001 | 0.6 | <0.001 | 0.5 | <0.001 | 0.3 | <0.001 |
| TC 1 | LDL-Cholesterol | HDL-Cholesterol | ||||
|---|---|---|---|---|---|---|
| ρ 2 | p | ρ | p | ρ | p | |
| 5-HT in plasma 3 | 0.5 | <0.001 | 0.5 | <0.001 | −0.2 | 0.03 |
| Platelets 5-HT | 0.3 | <0.001 | 0.4 | <0.001 | ||
| 5-HT in 1 platelet | 0.3 | 0.003 | 0.3 | <0.001 | ||
| 5-HIAA | 0.5 | <0.001 | 0.4 | <0.001 | −0.3 | <0.001 |
| SERT | 0.3 | 0.003 | 0.3 | 0.005 | ||
| Main Group | Control Group | p 1 | |||
|---|---|---|---|---|---|
| Mean | CI | Mean | CI | ||
| Age, years | 10.5 | 2.8–18.1 | 10.5 | 2.8–18.1 | 1 |
| Height, cm | 143.1 | 98.4–177.7 | 141.7 | 95.4–187.9 | 0.758 |
| Weight, kg | 37.7 | 6.1–69.3 | 38.2 | 1.8–74.6 | 0.868 |
| z-score BMI | −0.18 | −1.9–1.5 | −0.07 | −2.1–2 | 0.544 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sadykova, D.; Nigmatullina, R.; Salakhova, K.; Slastnikova, E.; Galimova, L.; Khaliullina, C.; Valeeva, I. Membrane Transporter of Serotonin and Hypercholesterolemia in Children. Int. J. Mol. Sci. 2024, 25, 767. https://doi.org/10.3390/ijms25020767
Sadykova D, Nigmatullina R, Salakhova K, Slastnikova E, Galimova L, Khaliullina C, Valeeva I. Membrane Transporter of Serotonin and Hypercholesterolemia in Children. International Journal of Molecular Sciences. 2024; 25(2):767. https://doi.org/10.3390/ijms25020767
Chicago/Turabian StyleSadykova, Dinara, Razina Nigmatullina, Karina Salakhova, Evgeniia Slastnikova, Liliya Galimova, Chulpan Khaliullina, and Ildaria Valeeva. 2024. "Membrane Transporter of Serotonin and Hypercholesterolemia in Children" International Journal of Molecular Sciences 25, no. 2: 767. https://doi.org/10.3390/ijms25020767
APA StyleSadykova, D., Nigmatullina, R., Salakhova, K., Slastnikova, E., Galimova, L., Khaliullina, C., & Valeeva, I. (2024). Membrane Transporter of Serotonin and Hypercholesterolemia in Children. International Journal of Molecular Sciences, 25(2), 767. https://doi.org/10.3390/ijms25020767






