Evidence of the Role of Inflammation and the Hormonal Environment in the Pathogenesis of Adrenal Myelolipomas in Congenital Adrenal Hyperplasia
Abstract
:1. Introduction
2. Results
2.1. Characteristics of the CAH Study Population
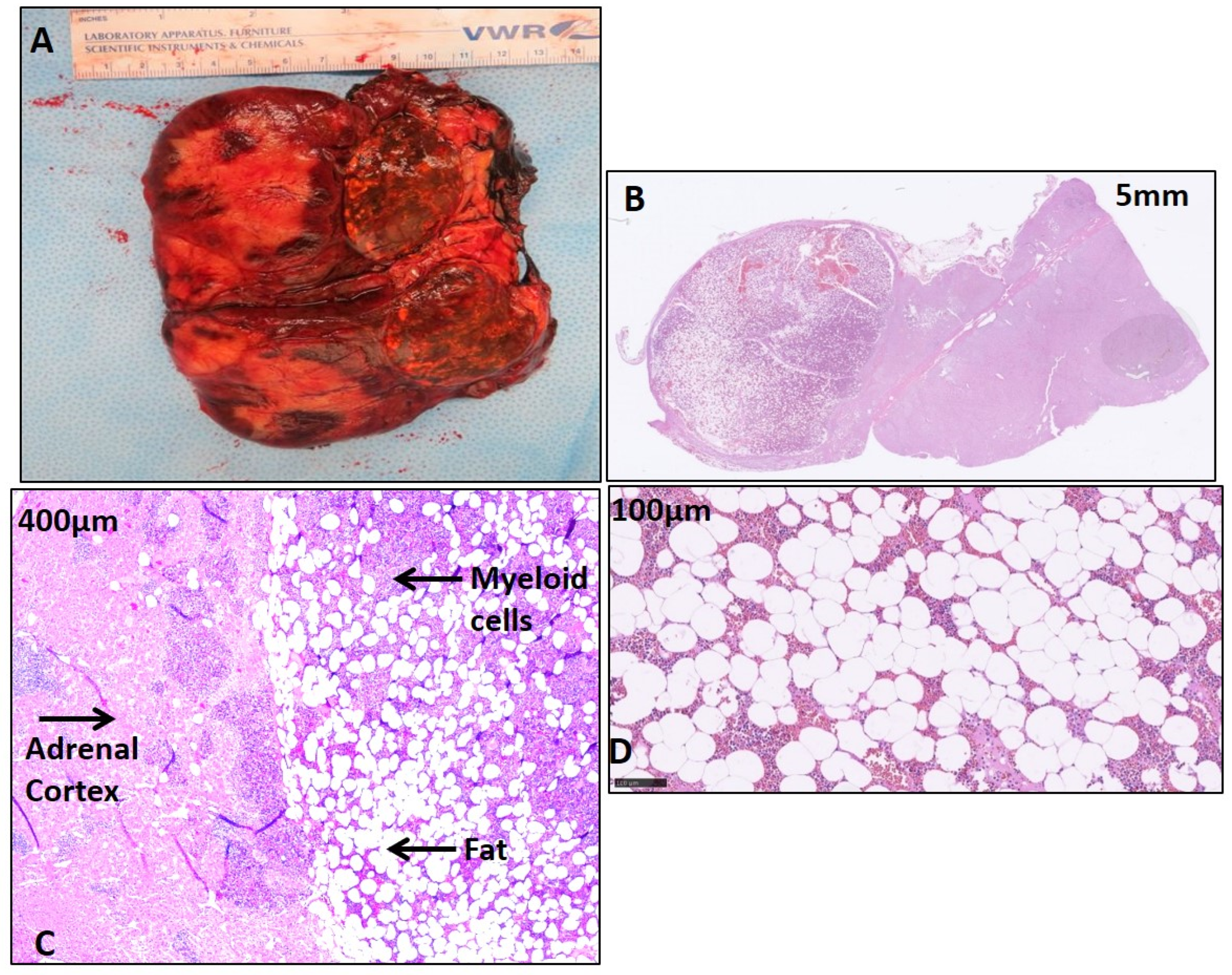
2.2. Histological Features of CAH Adrenal Myelolipoma
2.3. Gene Expression of Inflammatory and Lipogenic Genes by qRT-PCR
2.4. RNA-Sequencing Analysis
2.5. Cell Chemotactic Assay
3. Discussion
4. Materials and Methods
4.1. Tissue Collection
4.2. Histology and Immunohistochemistry
4.3. Gene Expression Analysis by qRT-PCR
4.4. Cell Culture
4.5. RNA-Sequencing and Differential Expression Testing
4.6. In Vitro Invasion and Transfilter Migration Assays
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Damjanov, I.; Katz, S.M.; Catalano, E.; Mason, D.; Schwartz, A.B. Myelolipoma in a heterotopic adrenal gland: Light and electron microscopic findings. Cancer 1979, 44, 1350–1356. [Google Scholar] [CrossRef]
- Shenoy, V.G.; Thota, A.; Shankar, R.; Desai, M.G. Adrenal myelolipoma: Controversies in its management. Indian J. Urol. 2015, 31, 94–101. [Google Scholar] [CrossRef]
- Lam, A.K. Lipomatous tumours in adrenal gland: WHO updates and clinical implications. Endocr.-Relat. Cancer 2017, 24, R65–R79. [Google Scholar] [CrossRef]
- Dann, P.H.; Krinsky, G.A.; Israel, G.M. Case 135: Presacral myelolipoma. Radiology 2008, 248, 314–316. [Google Scholar] [CrossRef] [PubMed]
- Schulte, K.M.; Simon, D.; Dotzenrath, C.; Scheuring, S.; Kohrer, K.; Roher, H.D. Sequence analysis of the MEN I gene in two patients with multiple cutaneous lipomas and endocrine tumors. Horm. Metab. Res. 2000, 32, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Bokhari, M.R.; Zulfiqar, H.; Garla, V.V. Adrenal Myelolipoma; StatPearls: Treasure Island, FL, USA, 2023. [Google Scholar]
- Feng, C.; Jiang, H.; Ding, Q.; Wen, H. Adrenal myelolipoma: A mingle of progenitor cells? Med. Hypotheses 2013, 80, 819–822. [Google Scholar] [CrossRef] [PubMed]
- Nermoen, I.; Rorvik, J.; Holmedal, S.H.; Hykkerud, D.L.; Fougner, K.J.; Svartberg, J.; Husebye, E.S.; Lovas, K. High frequency of adrenal myelolipomas and testicular adrenal rest tumours in adult Norwegian patients with classical congenital adrenal hyperplasia because of 21-hydroxylase deficiency. Clin. Endocrinol. 2011, 75, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Giacaglia, L.R.; Mendonca, B.B.; Madureira, G.; Melo, K.F.; Suslik, C.A.; Arnhold, I.J.; Bachega, T.A. Adrenal nodules in patients with congenital adrenal hyperplasia due to 21-hydroxylase deficiency: Regression after adequate hormonal control. J. Pediatr. Endocrinol. Metab. 2001, 14, 415–419. [Google Scholar] [CrossRef]
- Nermoen, I.; Falhammar, H. Prevalence and Characteristics of Adrenal Tumors and Myelolipomas in Congenital Adrenal Hyperplasia: A Systematic Review and Meta-Analysis. Endocr. Pract. 2020, 26, 1351–1365. [Google Scholar] [CrossRef]
- Merke, D.P.; Auchus, R.J. Congenital Adrenal Hyperplasia Due to 21-Hydroxylase Deficiency. N. Engl. J. Med. 2020, 383, 1248–1261. [Google Scholar] [CrossRef]
- Wen, J.; Chen, H.; Yi, W.; Zhou, X. Primary costal myelolipoma: A case report and review of the literature. Int. J. Clin. Exp. Pathol. 2015, 8, 2202–2204. [Google Scholar]
- Plaut, A. Myelolipoma in the adrenal cortex; myeloadipose structures. Am. J. Pathol. 1958, 34, 487–515. [Google Scholar]
- Selye, H.; Stone, H. Hormonally induced transformation of adrenal into myeloid tissue. Am. J. Pathol. 1950, 26, 211–233. [Google Scholar]
- Merke, D.P.; Chrousos, G.P.; Eisenhofer, G.; Weise, M.; Keil, M.F.; Rogol, A.D.; Van Wyk, J.J.; Bornstein, S.R. Adrenomedullary dysplasia and hypofunction in patients with classic 21-hydroxylase deficiency. N. Engl. J. Med. 2000, 343, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Kolli, V.; da Cunha, I.W.; Kim, S.; Iben, J.R.; Mallappa, A.; Li, T.; Gaynor, A.; Coon, S.L.; Quezado, M.M.; Merke, D.P. Morphologic and Molecular Characterization of Adrenals and Adrenal Rest Affected by Congenital Adrenal Hyperplasia. Front. Endocrinol. 2021, 12, 730947. [Google Scholar] [CrossRef] [PubMed]
- Marx, C.; Bornstein, S.R.; Wolkersdorfer, G.W. Cellular immune-endocrine interaction in adrenocortical tissues. Eur. J. Clin. Investig. 2000, 30 (Suppl. 3), 1–5. [Google Scholar] [CrossRef]
- Ehrhart-Bornstein, M.; Hinson, J.P.; Bornstein, S.R.; Scherbaum, W.A.; Vinson, G.P. Intraadrenal interactions in the regulation of adrenocortical steroidogenesis. Endocr. Rev. 1998, 19, 101–143. [Google Scholar] [CrossRef]
- Kanczkowski, W.; Sue, M.; Zacharowski, K.; Reincke, M.; Bornstein, S.R. The role of adrenal gland microenvironment in the HPA axis function and dysfunction during sepsis. Mol. Cell. Endocrinol. 2015, 408, 241–248. [Google Scholar] [CrossRef]
- Lefebvre, H.; Prevost, G.; Louiset, E. Autocrine/paracrine regulatory mechanisms in adrenocortical neoplasms responsible for primary adrenal hypercorticism. Eur. J. Endocrinol. 2013, 169, R115–R138. [Google Scholar] [CrossRef] [PubMed]
- White, P.C.; Speiser, P.W. Congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Endocr. Rev. 2000, 21, 245–291. [Google Scholar] [CrossRef]
- Abdellatif, A.B.; Fernandes-Rosa, F.L.; Boulkroun, S.; Zennaro, M.C. Vascular and hormonal interactions in the adrenal gland. Front. Endocrinol. 2022, 13, 995228. [Google Scholar] [CrossRef]
- Labrie, F. Adrenal androgens and intracrinology. Semin. Reprod. Med. 2004, 22, 299–309. [Google Scholar] [CrossRef]
- Mueller, J.W.; Vogg, N.; Lightning, T.A.; Weigand, I.; Ronchi, C.L.; Foster, P.A.; Kroiss, M. Steroid Sulfation in Adrenal Tumors. J. Clin. Endocrinol. Metab. 2021, 106, 3385–3397. [Google Scholar] [CrossRef]
- Speiser, P.W.; Arlt, W.; Auchus, R.J.; Baskin, L.S.; Conway, G.S.; Merke, D.P.; Meyer-Bahlburg, H.F.L.; Miller, W.L.; Murad, M.H.; Oberfield, S.E.; et al. Congenital Adrenal Hyperplasia Due to Steroid 21-Hydroxylase Deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2018, 103, 4043–4088. [Google Scholar] [CrossRef] [PubMed]
- Turcu, A.F.; Mallappa, A.; Elman, M.S.; Avila, N.A.; Marko, J.; Rao, H.; Tsodikov, A.; Auchus, R.J.; Merke, D.P. 11-Oxygenated Androgens Are Biomarkers of Adrenal Volume and Testicular Adrenal Rest Tumors in 21-Hydroxylase Deficiency. J. Clin. Endocrinol. Metab. 2017, 102, 2701–2710. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.M.; Chen, X.; Easton, J.; Finkelstein, D.; Liu, Z.; Pounds, S.; Rodriguez-Galindo, C.; Lund, T.C.; Mardis, E.R.; Wilson, R.K.; et al. Genomic landscape of paediatric adrenocortical tumours. Nat. Commun. 2015, 6, 6302. [Google Scholar] [CrossRef] [PubMed]
- Zeiger, M.A.; Thompson, G.B.; Duh, Q.Y.; Hamrahian, A.H.; Angelos, P.; Elaraj, D.; Fishman, E.; Kharlip, J.; American Association of Clinical, E.; American Association of Endocrine, S. The American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons medical guidelines for the management of adrenal incidentalomas. Endocr. Pract. 2009, 15 (Suppl. 1), 1–20. [Google Scholar] [CrossRef]
- Fassnacht, M.; Dekkers, O.M.; Else, T.; Baudin, E.; Berruti, A.; de Krijger, R.; Haak, H.R.; Mihai, R.; Assie, G.; Terzolo, M. European Society of Endocrinology Clinical Practice Guidelines on the management of adrenocortical carcinoma in adults, in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 2018, 179, G1–G46. [Google Scholar] [CrossRef]
- Arlt, W.; Biehl, M.; Taylor, A.E.; Hahner, S.; Libe, R.; Hughes, B.A.; Schneider, P.; Smith, D.J.; Stiekema, H.; Krone, N.; et al. Urine steroid metabolomics as a biomarker tool for detecting malignancy in adrenal tumors. J. Clin. Endocrinol. Metab. 2011, 96, 3775–3784. [Google Scholar] [CrossRef]
- Fassnacht, M.; Allolio, B. Clinical management of adrenocortical carcinoma. Best Pract. Res. Clin. Endocrinol. Metab. 2009, 23, 273–289. [Google Scholar] [CrossRef]
- Calissendorff, J.; Juhlin, C.C.; Sundin, A.; Bancos, I.; Falhammar, H. Adrenal myelolipomas. Lancet Diabetes Endocrinol. 2021, 9, 767–775. [Google Scholar] [CrossRef]
- Lefebvre, H.; Thomas, M.; Duparc, C.; Bertherat, J.; Louiset, E. Role of ACTH in the Interactive/Paracrine Regulation of Adrenal Steroid Secretion in Physiological and Pathophysiological Conditions. Front. Endocrinol. 2016, 7, 98. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Gubbels Bupp, M.R.; Jorgensen, T.N. Androgen-Induced Immunosuppression. Front. Immunol. 2018, 9, 794. [Google Scholar] [CrossRef]
- Vancolen, S.; Sebire, G.; Robaire, B. Influence of androgens on the innate immune system. Andrology 2023, 11, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Jaillon, S.; Berthenet, K.; Garlanda, C. Sexual Dimorphism in Innate Immunity. Clin. Rev. Allergy Immunol. 2019, 56, 308–321. [Google Scholar] [CrossRef]
- Henze, L.; Schwinge, D.; Schramm, C. The Effects of Androgens on T Cells: Clues to Female Predominance in Autoimmune Liver Diseases? Front. Immunol. 2020, 11, 1567. [Google Scholar] [CrossRef] [PubMed]
- Angum, F.; Khan, T.; Kaler, J.; Siddiqui, L.; Hussain, A. The Prevalence of Autoimmune Disorders in Women: A Narrative Review. Cureus 2020, 12, e8094. [Google Scholar] [CrossRef]
- Ose, J.; Poole, E.M.; Schock, H.; Lehtinen, M.; Arslan, A.A.; Zeleniuch-Jacquotte, A.; Visvanathan, K.; Helzlsouer, K.; Buring, J.E.; Lee, I.M.; et al. Androgens Are Differentially Associated with Ovarian Cancer Subtypes in the Ovarian Cancer Cohort Consortium. Cancer Res. 2017, 77, 3951–3960. [Google Scholar] [CrossRef]
- Secreto, G.; Girombelli, A.; Krogh, V. Androgen excess in breast cancer development: Implications for prevention and treatment. Endocr. Relat. Cancer 2019, 26, R81–R94. [Google Scholar] [CrossRef]
- Kaaks, R.; Berrino, F.; Key, T.; Rinaldi, S.; Dossus, L.; Biessy, C.; Secreto, G.; Amiano, P.; Bingham, S.; Boeing, H.; et al. Serum sex steroids in premenopausal women and breast cancer risk within the European Prospective Investigation into Cancer and Nutrition (EPIC). J. Natl. Cancer Inst. 2005, 97, 755–765. [Google Scholar] [CrossRef]
- Quinkler, M.; Meyer, B.; Bumke-Vogt, C.; Grossmann, C.; Gruber, U.; Oelkers, W.; Diederich, S.; Bahr, V. Agonistic and antagonistic properties of progesterone metabolites at the human mineralocorticoid receptor. Eur. J. Endocrinol. 2002, 146, 789–799. [Google Scholar] [CrossRef]
- Mooij, C.F.; Parajes, S.; Pijnenburg-Kleizen, K.J.; Arlt, W.; Krone, N.; Claahsen-van der Grinten, H.L. Influence of 17-Hydroxyprogesterone, Progesterone and Sex Steroids on Mineralocorticoid Receptor Transactivation in Congenital Adrenal Hyperplasia. Horm. Res. Paediatr. 2015, 83, 414–421. [Google Scholar] [CrossRef]
- Pijnenburg-Kleizen, K.J.; Engels, M.; Mooij, C.F.; Griffin, A.; Krone, N.; Span, P.N.; van Herwaarden, A.E.; Sweep, F.C.; Claahsen-van der Grinten, H.L. Adrenal Steroid Metabolites Accumulating in Congenital Adrenal Hyperplasia Lead to Transactivation of the Glucocorticoid Receptor. Endocrinology 2015, 156, 3504–3510. [Google Scholar] [CrossRef]
- Solano, M.E.; Arck, P.C. Steroids, Pregnancy and Fetal Development. Front. Immunol. 2019, 10, 3017. [Google Scholar] [CrossRef]
- Engler, J.B.; Kursawe, N.; Solano, M.E.; Patas, K.; Wehrmann, S.; Heckmann, N.; Luhder, F.; Reichardt, H.M.; Arck, P.C.; Gold, S.M.; et al. Glucocorticoid receptor in T cells mediates protection from autoimmunity in pregnancy. Proc. Natl. Acad. Sci. USA 2017, 114, E181–E190. [Google Scholar] [CrossRef] [PubMed]
- Oettel, M.; Mukhopadhyay, A.K. Progesterone: The forgotten hormone in men? Aging Male 2004, 7, 236–257. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Ji, H.; Liu, S.; Xing, Q.; Zhu, B.; Wang, Y. Survival Prognosis, Tumor Immune Landscape, and Immune Responses of ADAMTS14 in Clear Cell Renal Cell Carcinoma and Its Potential Mechanisms. Front. Immunol. 2022, 13, 790608. [Google Scholar] [CrossRef] [PubMed]
- El-Masry, O.S.; Alamri, A.M.; Alzahrani, F.; Alsamman, K. ADAMTS14, ARHGAP22, and EPDR1 as potential novel targets in acute myeloid leukaemia. Heliyon 2022, 8, e09065. [Google Scholar] [CrossRef] [PubMed]
- Sedo, A.; Stremenova, J.; Busek, P.; Duke-Cohan, J.S. Dipeptidyl peptidase-IV and related molecules: Markers of malignancy? Expert Opin. Med. Diagn. 2008, 2, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Grigo, C.; Steinbeck, J.; von Horsten, S.; Amann, K.; Daniel, C. Soluble DPP4 originates in part from bone marrow cells and not from the kidney. Peptides 2014, 57, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Decmann, A.; Perge, P.; Toth, M.; Igaz, P. Adrenal myelolipoma: A comprehensive review. Endocrine 2018, 59, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.Q.; Kaupert, L.C.; Brito, L.P.; Lerario, A.M.; Mariani, B.M.; Ribeiro, M.; Monte, O.; Denes, F.T.; Mendonca, B.B.; Bachega, T.A. Increased expression of ACTH (MC2R) and androgen (AR) receptors in giant bilateral myelolipomas from patients with congenital adrenal hyperplasia. BMC Endocr. Disord. 2014, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Kiwaki, K.; Levine, J.A. Differential effects of adrenocorticotropic hormone on human and mouse adipose tissue. J. Comp. Physiol. B 2003, 173, 675–678. [Google Scholar] [CrossRef]
- Wu, T.; Hu, E.; Xu, S.; Chen, M.; Guo, P.; Dai, Z.; Feng, T.; Zhou, L.; Tang, W.; Zhan, L.; et al. clusterProfiler 4.0: A universal enrichment tool for interpreting omics data. Innovation 2021, 2, 100141. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolli, V.; Frucci, E.; da Cunha, I.W.; Iben, J.R.; Kim, S.A.; Mallappa, A.; Li, T.; Faucz, F.R.; Kebebew, E.; Nilubol, N.; et al. Evidence of the Role of Inflammation and the Hormonal Environment in the Pathogenesis of Adrenal Myelolipomas in Congenital Adrenal Hyperplasia. Int. J. Mol. Sci. 2024, 25, 2543. https://doi.org/10.3390/ijms25052543
Kolli V, Frucci E, da Cunha IW, Iben JR, Kim SA, Mallappa A, Li T, Faucz FR, Kebebew E, Nilubol N, et al. Evidence of the Role of Inflammation and the Hormonal Environment in the Pathogenesis of Adrenal Myelolipomas in Congenital Adrenal Hyperplasia. International Journal of Molecular Sciences. 2024; 25(5):2543. https://doi.org/10.3390/ijms25052543
Chicago/Turabian StyleKolli, Vipula, Emily Frucci, Isabela Werneck da Cunha, James R. Iben, Sun A. Kim, Ashwini Mallappa, Tianwei Li, Fabio Rueda Faucz, Electron Kebebew, Naris Nilubol, and et al. 2024. "Evidence of the Role of Inflammation and the Hormonal Environment in the Pathogenesis of Adrenal Myelolipomas in Congenital Adrenal Hyperplasia" International Journal of Molecular Sciences 25, no. 5: 2543. https://doi.org/10.3390/ijms25052543







