Comprehensive Atlas of Alternative Splicing Reveals NSRP1 Promoting Adipogenesis through CCDC18
Abstract
:1. Introduction
2. Results
2.1. Genome-Wide Identification of Alternative Splicing Events (ASEs) in Different Tissues
2.2. Tissue-Specific Differentially Alternative Splicing Genes (DASGs) and Differentially Expressed Genes (DEGs)
2.3. Functional Analysis of DEGs Association with ASEs
2.4. Effects of Transcription Factors (TFs) on Alternative Splicing (AS)
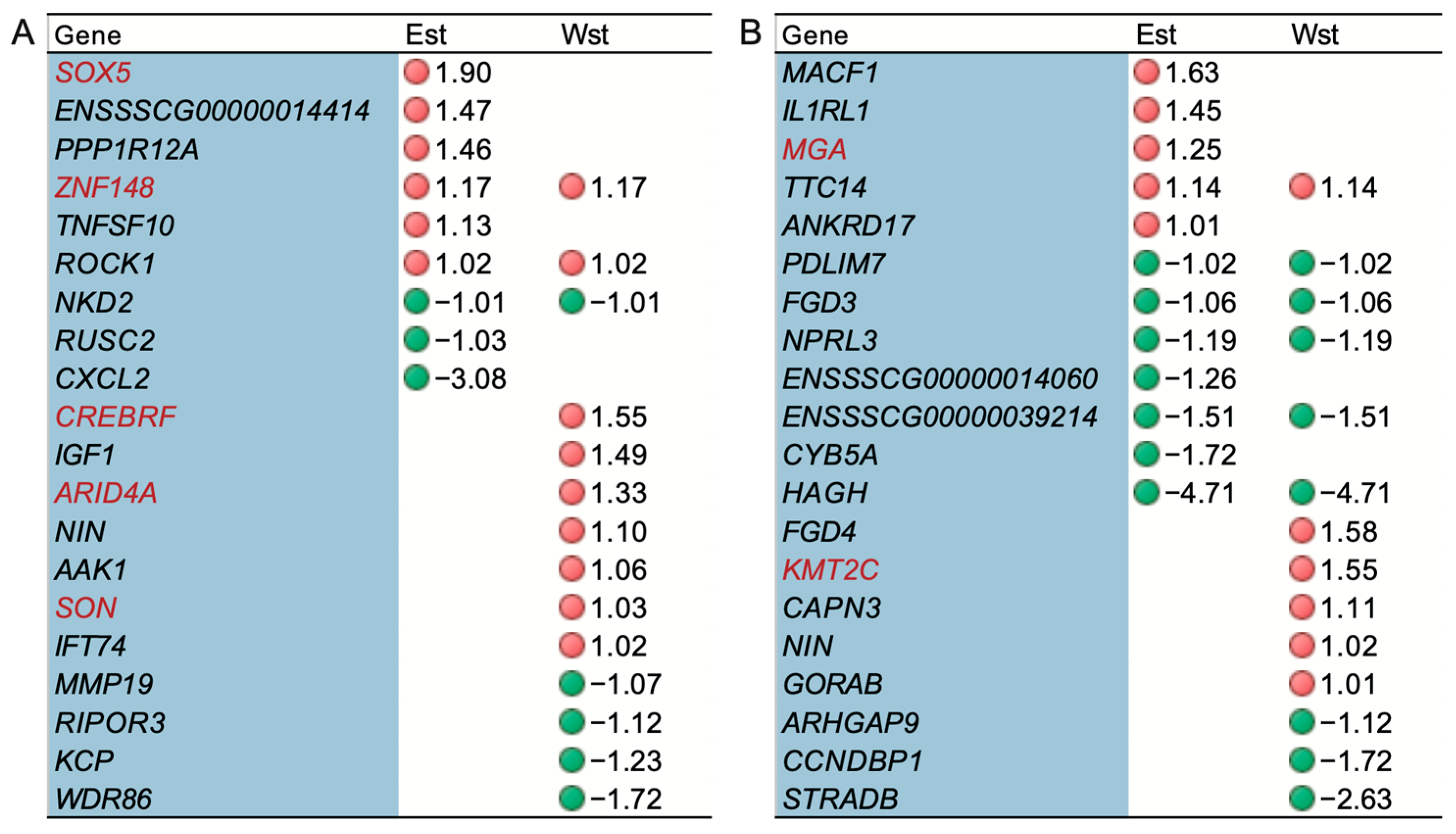
2.5. The Impact of Genes under Potential Selection on Alternative Splicing
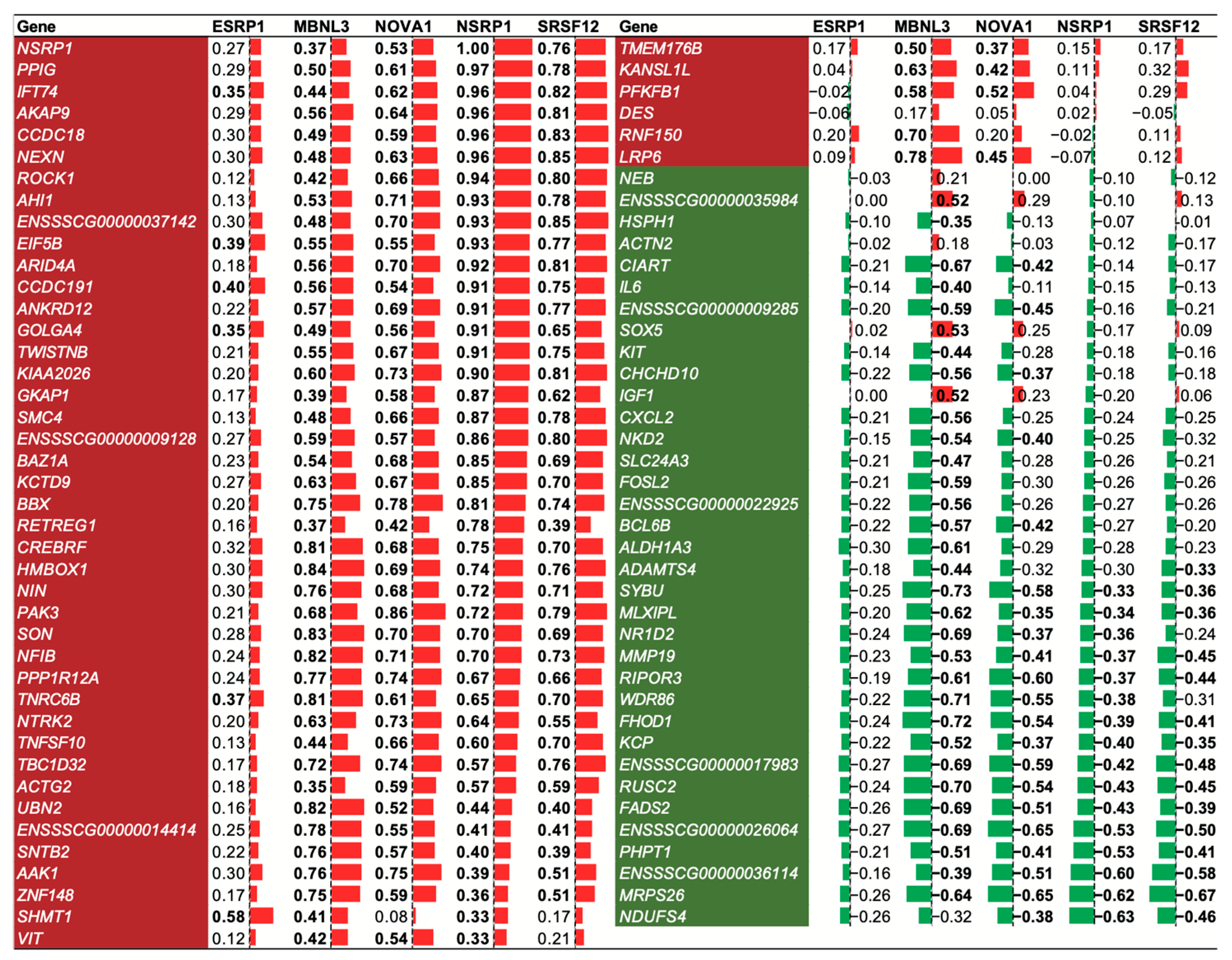
2.6. Effects of Splicing Factors (SFs) on Alternative Splicing
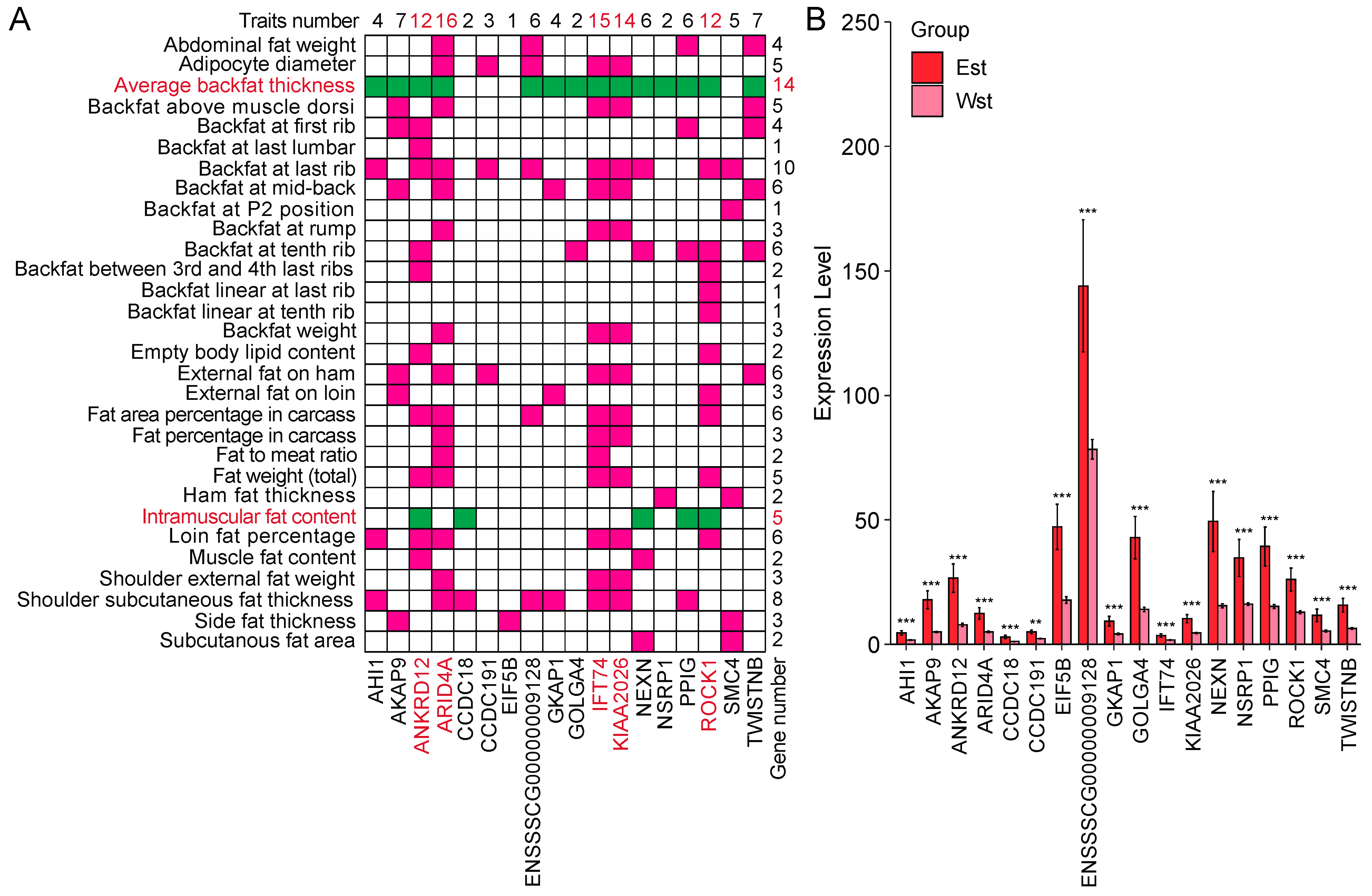
2.7. Splicing Factor NSRP1 and Correlated Genes Impact Adipose-Deposition Traits
2.8. Splicing Factor NSRP1 Regulates Adipogenesis
2.9. NSRP1 Promotes Adipogenesis by Regulating AS and Expression of CCDC18
3. Discussion
3.1. ASEs Identification and Gene Functional Enrichment Analysis
3.2. Associations between TFs and AS
3.3. ASGs under Potential Selection
3.4. SF NSRP1 Promotes Adipose Deposition
3.5. Limitations of the Study
3.6. Commercial Prospects of Marker Genes
4. Materials and Methods
4.1. Data Collection
4.2. Transcriptome Data Quality Control and Alignment
4.3. Identification and Differential Analysis of Alternative Splicing Events
4.4. Identification of Differentially Expressed Genes
4.5. Gene Functional Enrichment Analysis
4.6. Protein-Protein Interaction Network Analysis
4.7. Integrated Analysis of Transcription Factors, Selection Signals, Splicing Factors, and QTLs
4.8. Adipocyte Culture and Induced Differentiation
4.9. RNA Extraction and RT-qPCR
4.10. Western Blot Analysis
4.11. Cell Counting Kit-8 Proliferation Assay
4.12. 5-Ethynyl-2′-Deoxyuridine (EdU) Staining
4.13. Oil Red O Staining
4.14. Semiquantitative RT-PCR Analysis of Alternative Splicing Events
4.15. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- White, S. From globalized pig breeds to capitalist pigs: A study in animal cultures and evolutionary history. Environ. Hist. 2011, 16, 94–120. [Google Scholar] [CrossRef]
- Zhang, L.; Tian, H.; Shi, H.; Pan, S.; Qin, X.; Pan, N.; Dangal, S.R. Methane emissions from livestock in East Asia during 1961–2019. Ecosyst. Health Sustain. 2021, 7, 1918024. [Google Scholar] [CrossRef]
- Choi, S.K.; Lee, J.-E.; Kim, Y.-J.; Min, M.-S.; Voloshina, I.; Myslenkov, A.; Oh, J.G.; Kim, T.-H.; Markov, N.; Seryodkin, I. Genetic structure of wild boar (Sus scrofa) populations from East Asia based on microsatellite loci analyses. BMC Genet. 2014, 15, 85. [Google Scholar] [CrossRef]
- Larson, G.; Dobney, K.; Albarella, U.; Fang, M.; Matisoo-Smith, E.; Robins, J.; Lowden, S.; Finlayson, H.; Brand, T.; Willerslev, E. Worldwide phylogeography of wild boar reveals multiple centers of pig domestication. Science 2005, 307, 1618–1621. [Google Scholar] [CrossRef]
- Evin, A.; Cucchi, T.; Cardini, A.; Vidarsdottir, U.S.; Larson, G.; Dobney, K. The long and winding road: Identifying pig domestication through molar size and shape. J. Archaeol. Sci. 2013, 40, 735–743. [Google Scholar] [CrossRef]
- Liu, Y.; Fu, Y.; Yang, Y.; Yi, G.; Lian, J.; Xie, B.; Yao, Y.; Chen, M.; Niu, Y.; Liu, L.; et al. Integration of multi-omics data reveals cis-regulatory variants that are associated with phenotypic differentiation of eastern from western pigs. Genet. Sel. Evol. 2022, 54, 62. [Google Scholar] [CrossRef]
- Deng, L.; Li, W.; Liu, W.; Liu, Y.; Xie, B.; Groenen, M.A.M.; Madsen, O.; Yang, X.; Tang, Z. Integrative metabolomic and transcriptomic analysis reveals difference in glucose and lipid metabolism in the longissimus muscle of Luchuan and Duroc pigs. Front. Genet. 2023, 14, 1128033. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liang, G.; Niu, G.; Zhang, Y.; Zhou, R.; Wang, Y.; Mu, Y.; Tang, Z.; Li, K. Comparative analysis of DNA methylome and transcriptome of skeletal muscle in lean-, obese-, and mini-type pigs. Sci. Rep. 2017, 7, 39883. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.J.; Smith, C.W.; Jiggins, C.D. Alternative splicing as a source of phenotypic diversity. Nat. Rev. Genet. 2022, 23, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Gao, F.; He, M.; Ding, X.F.; Wong, A.M.; Sze, S.C.; Yu, A.C.; Sun, T.; Chan, A.W.H.; Wang, X. Long-read RNA sequencing identifies alternative splice variants in hepatocellular carcinoma and tumor-specific isoforms. Hepatology 2019, 70, 1011–1025. [Google Scholar] [CrossRef] [PubMed]
- Baralle, F.E.; Giudice, J. Alternative splicing as a regulator of development and tissue identity. Nat. Rev. Mol. Cell Biol. 2017, 18, 437–451. [Google Scholar] [CrossRef]
- Mazin, P.V.; Khaitovich, P.; Cardoso-Moreira, M.; Kaessmann, H. Alternative splicing during mammalian organ development. Nat. Genet. 2021, 53, 925–934. [Google Scholar] [CrossRef]
- Modrek, B.; Lee, C. A genomic view of alternative splicing. Nat. Genet. 2002, 30, 13–19. [Google Scholar] [CrossRef]
- Scotti, M.M.; Swanson, M.S. RNA mis-splicing in disease. Nat. Rev. Genet. 2016, 17, 19–32. [Google Scholar] [CrossRef]
- Han, H.; Braunschweig, U.; Gonatopoulos-Pournatzis, T.; Weatheritt, R.J.; Hirsch, C.L.; Ha, K.C.H.; Radovani, E.; Nabeel-Shah, S.; Sterne-Weiler, T.; Wang, J.; et al. Multilayered Control of Alternative Splicing Regulatory Networks by Transcription Factors. Mol. Cell 2017, 65, 539–553.e7. [Google Scholar] [CrossRef]
- Bielli, P.; Busa, R.; Di Stasi, S.M.; Munoz, M.J.; Botti, F.; Kornblihtt, A.R.; Sette, C. The transcription factor FBI-1 inhibits SAM68-mediated BCL-X alternative splicing and apoptosis. EMBO Rep. 2014, 15, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Long, J.C.; Caceres, J.F. The SR protein family of splicing factors: Master regulators of gene expression. Biochem. J. 2009, 417, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Baxter, T.; Muir, W.M.; Groenen, M.A.; Schook, L.B. Genetic resources, genome mapping and evolutionary genomics of the pig (Sus scrofa). Int. J. Biol. Sci. 2007, 3, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Li, P.J. Exponential growth, animal welfare, environmental and food safety impact: The case of China’s livestock production. J. Agric. Environ. Ethics 2009, 22, 217–240. [Google Scholar] [CrossRef]
- Nygard, A.-B.; Cirera, S.; Gilchrist, M.J.; Gorodkin, J.; Jørgensen, C.B.; Fredholm, M. A study of alternative splicing in the pig. BMC Res. Notes 2010, 3, 123. [Google Scholar] [CrossRef] [PubMed]
- Hao, W.; Yang, Z.; Sun, Y.; Li, J.; Zhang, D.; Liu, D.; Yang, X. Characterization of alternative splicing events in porcine skeletal muscles with different intramuscular Fat contents. Biomolecules 2022, 12, 154. [Google Scholar] [CrossRef]
- Shu, Z.; Wang, L.; Wang, J.; Zhang, L.; Hou, X.; Yan, H.; Wang, L. Integrative Analysis of Nanopore and Illumina Sequencing Reveals Alternative Splicing Complexity in Pig Longissimus Dorsi Muscle. Front. Genet. 2022, 13, 877646. [Google Scholar] [CrossRef]
- Bhadra, M.; Howell, P.; Dutta, S.; Heintz, C.; Mair, W.B. Alternative splicing in aging and longevity. Hum. Genet. 2020, 139, 357–369. [Google Scholar] [CrossRef]
- Jacobs, A.; Elmer, K.R. Alternative splicing and gene expression play contrasting roles in the parallel phenotypic evolution of a salmonid fish. Mol. Ecol. 2021, 30, 4955–4969. [Google Scholar] [CrossRef]
- Bush, S.J.; Chen, L.; Tovar-Corona, J.M.; Urrutia, A.O. Alternative splicing and the evolution of phenotypic novelty. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20150474. [Google Scholar] [CrossRef]
- Yang, Y.; Yuan, H.; Yang, Q.; Cai, Y.; Ren, Y.; Li, Y.; Gao, C.; Zhao, S. Post-transcriptional regulation through alternative splicing in the lungs of Tibetan pigs under hypoxia. Gene 2022, 819, 146268. [Google Scholar] [CrossRef]
- Tan, B.; Zeng, J.; Meng, F.; Wang, S.; Xiao, L.; Zhao, X.; Hong, L.; Zheng, E.; Wu, Z.; Li, Z. Comprehensive analysis of pre-mRNA alternative splicing regulated by m6A methylation in pig oxidative and glycolytic skeletal muscles. BMC Genom. 2022, 23, 804. [Google Scholar] [CrossRef]
- Jiang, W.; Chen, L. Tissue specificity of gene expression evolves across mammal species. J. Comput. Biol. 2022, 29, 880–891. [Google Scholar] [CrossRef]
- Mosthaf, L.; Grako, K.; Dull, T.; Coussens, L.; Ullrich, A.; McClain, D. Functionally distinct insulin receptors generated by tissue-specific alternative splicing. EMBO J. 1990, 9, 2409–2413. [Google Scholar] [CrossRef]
- Qu, A.; Bai, Y.; Zhang, X.; Zeng, J.; Pu, F.; Wu, L.; Xu, P.; Zhou, T. Tissue-Specific Analysis of Alternative Splicing Events and Differential Isoform Expression in Large Yellow Croaker (Larimichthys crocea) after Cryptocaryon irritans Infection. Mar. Biotechnol. 2022, 24, 640–654. [Google Scholar] [CrossRef]
- Yu, T.; Tian, X.; Li, D.; He, Y.; Yang, P.; Cheng, Y.; Zhao, X.; Sun, J.; Yang, G. Transcriptome, proteome and metabolome analysis provide insights on fat deposition and meat quality in pig. Food Res. Int. 2023, 166, 112550. [Google Scholar] [CrossRef]
- Shen, J.; Hao, Z.; Luo, Y.; Zhen, H.; Liu, Y.; Wang, J.; Hu, J.; Liu, X.; Li, S.; Zhao, Z. Deep small RNA sequencing reveals important miRNAs related to muscle development and intramuscular fat deposition in longissimus dorsi muscle from different goat breeds. Front. Vet. Sci. 2022, 9, 911166. [Google Scholar] [CrossRef]
- Wang, H.; Wang, X.; Li, M.; Wang, S.; Chen, Q.; Lu, S. Identification of key sex-specific pathways and genes in the subcutaneous adipose tissue from pigs using WGCNA method. BMC Genom. Data 2022, 23, 35. [Google Scholar] [CrossRef]
- Jiao, Y.; Liang, X.; Hou, J.; Aisa, Y.; Wu, H.; Zhang, Z.; Nuermaimaiti, N.; Zhao, Y.; Jiang, S.; Guan, Y. Adenovirus type 36 regulates adipose stem cell differentiation and glucolipid metabolism through the PI3K/Akt/FoxO1/PPARγ signaling pathway. Lipids Health Dis. 2019, 18, 70. [Google Scholar] [CrossRef]
- Chen, H.; Peng, T.; Shang, H.; Shang, X.; Zhao, X.; Qu, M.; Song, X. RNA-Seq analysis reveals the potential molecular mechanisms of puerarin on intramuscular fat deposition in heat-stressed beef cattle. Front. Nutr. 2022, 9, 817557. [Google Scholar] [CrossRef]
- Poklukar, K.; Čandek-Potokar, M.; Batorek Lukač, N.; Tomažin, U.; Škrlep, M. Lipid deposition and metabolism in local and modern pig breeds: A review. Animals 2020, 10, 424. [Google Scholar] [CrossRef]
- Ahmed, B.; Sultana, R.; Greene, M.W. Adipose tissue and insulin resistance in obese. Biomed. Pharmacother. 2021, 137, 111315. [Google Scholar] [CrossRef]
- Nedunchezhiyan, U.; Varughese, I.; Sun, A.R.; Wu, X.; Crawford, R.; Prasadam, I. Obesity, inflammation, and immune system in osteoarthritis. Front. Immunol. 2022, 13, 907750. [Google Scholar] [CrossRef]
- Zhao, G.-N.; Tian, Z.-W.; Tian, T.; Zhu, Z.-P.; Zhao, W.-J.; Tian, H.; Cheng, X.; Hu, F.-J.; Hu, M.-L.; Tian, S. TMBIM1 is an inhibitor of adipogenesis and its depletion promotes adipocyte hyperplasia and improves obesity-related metabolic disease. Cell Metab. 2021, 33, 1640–1654.e8. [Google Scholar] [CrossRef]
- Stadhouders, R.; Filion, G.J.; Graf, T. Transcription factors and 3D genome conformation in cell-fate decisions. Nature 2019, 569, 345–354. [Google Scholar] [CrossRef]
- Andrews, G.; Fan, K.; Pratt, H.E.; Phalke, N.; Consortium, Z.; Karlsson, E.K.; Lindblad-Toh, K.; Gazal, S.; Moore, J.E.; Weng, Z. Mammalian evolution of human cis-regulatory elements and transcription factor binding sites. Science 2023, 380, eabn7930. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, A.A.; Fayyaz, S.; Poltronieri, P.; Calin, G.; Mallardo, M. Epigenetic Deregulation in Cancer: Enzyme Players and Non-coding RNAs; Seminars in cancer biology, 2022; Elsevier: Amsterdam, The Netherlands, 2022; pp. 197–207. [Google Scholar]
- Milazzo, G.; Mercatelli, D.; Di Muzio, G.; Triboli, L.; De Rosa, P.; Perini, G.; Giorgi, F.M. Histone deacetylases (HDACs): Evolution, specificity, role in transcriptional complexes, and pharmacological actionability. Genes 2020, 11, 556. [Google Scholar] [CrossRef]
- Delcuve, G.P.; Khan, D.H.; Davie, J.R. Targeting class I histone deacetylases in cancer therapy. Expert. Opin. Ther. Targets 2013, 17, 29–41. [Google Scholar] [CrossRef]
- Grivas, P.D.; Papavassiliou, A.G. Transcriptional Corepressors in Cancer: Emerging Targets for Therapeutic Intervention; Wiley Online Library: Hoboken, NU, USA, 2013; Volume 119, pp. 1120–1128. [Google Scholar]
- Wu, M.-Y.; Eldin, K.W.; Beaudet, A.L. Identification of chromatin remodeling genes Arid4a and Arid4b as leukemia suppressor genes. J. Natl. Cancer Inst. 2008, 100, 1247–1259. [Google Scholar] [CrossRef]
- Liang, Y.K.; Han, Z.D.; Lu, J.M.; Liu, Z.Z.; Zhuo, Y.J.; Zhu, X.J.; Chen, J.X.; Ye, J.H.; Liang, Y.X.; He, H.C. Downregulation of ARID4A and ARID4B promote tumor progression and directly regulated by microRNA-30d in patient with prostate cancer. J. Cell. Biochem. 2018, 119, 7245–7255. [Google Scholar] [CrossRef]
- Huang, L.; Teng, D.; Wang, H.; Sheng, G.; Liu, T. Association of copy number variation in the AHI1 gene with risk of obesity in the Chinese population. Eur. J. Endocrinol. 2012, 166, 727–734. [Google Scholar] [CrossRef]
- Cho, Y.; Gutierrez, L.; Bordonaro, M.; Russo, D.; Anzelmi, F.; Hooven, J.T.; Cerra, C.; Lazarova, D.L. Effects of propolis and gamma-cyclodextrin on intestinal neoplasia in normal weight and obese mice. Cancer Med. 2016, 5, 2448–2458. [Google Scholar] [CrossRef]
- Deaton, A.M.; Dubey, A.; Ward, L.D.; Dornbos, P.; Flannick, J.; Consortium, A.-T.D.-G.; Yee, E.; Ticau, S.; Noetzli, L.; Parker, M.M. Rare loss of function variants in the hepatokine gene INHBE protect from abdominal obesity. Nat. Commun. 2022, 13, 4319. [Google Scholar] [CrossRef]
- Tan, X.; Liu, R.; Xing, S.; Zhang, Y.; Li, Q.; Zheng, M.; Zhao, G.; Wen, J. Genome-wide detection of key genes and epigenetic markers for chicken fatty liver. Int. J. Mol. Sci. 2020, 21, 1800. [Google Scholar] [CrossRef]
- Zappaterra, M.; Gioiosa, S.; Chillemi, G.; Zambonelli, P.; Davoli, R. Muscle transcriptome analysis identifies genes involved in ciliogenesis and the molecular cascade associated with intramuscular fat content in Large White heavy pigs. PLoS ONE 2020, 15, e0233372. [Google Scholar] [CrossRef]
- Pan, C.; Yang, C.; Wang, S.; Ma, Y. Identifying key genes and functionally enriched pathways of diverse adipose tissue types in cattle. Front. Genet. 2022, 13, 790690. [Google Scholar] [CrossRef]
- Peng, Y.; Shi, X.-e.; Huang, K.-l.; Yao, X.-p.; Chen, F.-f.; Li, X.; Yang, G.-s. Knock-down Sox5 suppresses porcine adipogenesis through BMP R-Smads signal pathway. Biochem. Biophys. Res. Commun. 2020, 527, 574–580. [Google Scholar] [CrossRef]
- Li, Z.; Liu, X.; Li, Y.; Wang, W.; Wang, N.; Xiao, F.; Gao, H.; Guo, H.; Li, H.; Wang, S. Chicken C/EBPζ gene: Expression profiles, association analysis, and identification of functional variants for abdominal fat. Domest. Anim. Endocrinol. 2021, 76, 106631. [Google Scholar] [CrossRef]
- Jiang, H.; Gao, Q.; Che, X.; Zhu, L.; Zhang, Z.; Chen, Y.; Dai, Y. Inhibition of penile tunica albuginea myofibroblasts activity by adipose-derived stem cells. Exp. Ther. Med. 2017, 14, 5149–5156. [Google Scholar] [CrossRef] [PubMed]
- Dou, Y.; Qi, K.; Liu, Y.; Li, C.; Song, C.; Wei, Y.; Zhang, Z.; Li, X.; Wang, K.; Li, X. Identification and Functional Prediction of Long Non-Coding RNA in Longissimus Dorsi Muscle of Queshan Black and Large White Pigs. Genes 2023, 14, 197. [Google Scholar] [CrossRef] [PubMed]
- Mota, L.F.; Lopes, F.B.; Fernandes Júnior, G.A.; Rosa, G.J.; Magalhães, A.F.; Carvalheiro, R.; Albuquerque, L.G. Genome-wide scan highlights the role of candidate genes on phenotypic plasticity for age at first calving in Nellore heifers. Sci. Rep. 2020, 10, 6481. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wei, S.; Deng, S.; Li, D.; Liu, K.; Shan, B.; Shao, Y.; Wei, W.; Chen, J.; Zhang, L. Genome-wide identification and comparison of mRNA s, lnc RNA s and circ RNA s in porcine intramuscular, subcutaneous, retroperitoneal and mesenteric adipose tissues. Anim. Genet. 2019, 50, 228–241. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Xiao, Y.; Xiong, Z.; Zhao, F.; Yin, C.; Zhang, Y.; Su, P.; Li, D.; Chen, Z.; Ma, X. MACF1, Versatility in Tissue-Specific Function and in Human Disease; Seminars in cell & developmental biology, 2017; Elsevier: Amsterdam, The Netherlands, 2017; pp. 3–8. [Google Scholar]
- Akhabir, L.; Sandford, A. Genetics of interleukin 1 receptor-like 1 in immune and inflammatory diseases. Curr. Genom. 2010, 11, 591–606. [Google Scholar] [CrossRef] [PubMed]
- Shaw, C.H.; Palti, Y.; Gao, G.; Wiens, G.D. Evolution of interleukin-1 receptor-like 1 and its role in rainbow trout (Oncorhynchus mykiss) resistance to Flavobacterium psychrophilum. J. Immunol. 2017, 198, 201–222. [Google Scholar] [CrossRef]
- Chen, Y.; Zeng, B.; Shi, P.; Xiao, H.; Chen, S. Comparative analysis of the Liver and Spleen transcriptomes between Holstein and Yunnan humped cattle. Animals 2019, 9, 527. [Google Scholar] [CrossRef]
- Sun, L.; Li, M.; Deng, L.; Niu, Y.; Tang, Y.; Wang, Y.; Guo, L. MGA Mutation as a novel biomarker for immune checkpoint therapies in non-squamous non-small cell lung cancer. Front. Pharmacol. 2021, 12, 625593. [Google Scholar] [CrossRef]
- Wang, J.; Shivakumar, S.; Barker, K.; Tang, Y.; Wallstrom, G.; Park, J.G.; Tsay, J.-C.J.; Pass, H.I.; Rom, W.N.; LaBaer, J. Comparative study of autoantibody responses between lung adenocarcinoma and benign pulmonary nodules. J. Thorac. Oncol. 2016, 11, 334–345. [Google Scholar] [CrossRef]
- Zhou, G.; Jiang, N.; Zhang, W.; Guo, S.; Xin, G. Biomarker identification in membranous nephropathy using a long non-coding RNA-mediated competitive endogenous RNA network. Interdiscip. Sci. Comput. Life Sci. 2021, 13, 615–623. [Google Scholar] [CrossRef]
- Kim, C.-H.; Park, S.-M.; Lee, S.-j.; Kim, Y.-D.; Jang, S.-H.; Woo, S.-M.; Kwon, T.-K.; Park, Z.-Y.; Chung, I.-J.; Kim, H.-R. NSrp70 is a lymphocyte-essential splicing factor that controls thymocyte development. Nucleic Acids Res. 2021, 49, 5760–5778. [Google Scholar] [CrossRef]
- Shi, Y. Mechanistic insights into precursor messenger RNA splicing by the spliceosome. Nat. Rev. Mol. Cell Biol. 2017, 18, 655–670. [Google Scholar] [CrossRef]
- Zeng, M.; Wang, B.; Liu, L.; Yang, Y.; Tang, Z. Genome-wide association study identifies 12 new genetic loci associated with growth traits in pigs. J. Integr. Agric. 2024, 23, 217–227. [Google Scholar] [CrossRef]
- Zha, A.; Li, W.; Wang, J.; Bai, P.; Qi, M.; Liao, P.; Tan, B.E.; Yin, Y. Trimethylamine oxide supplementation differentially regulates fat deposition in liver, longissimus dorsi muscle and adipose tissue of growing-finishing pigs. Anim. Nutr. 2024, in press. [Google Scholar] [CrossRef]
- Zhang, J.; Meng, S.; Wang, H.; Zhang, C.; Sun, Z.; Huang, L.; Miao, Z. Comparison of Growth Performance, Carcass Properties, Fatty Acid Profile, and Genes Involved in Fat Metabolism in Nanyang and Landrace Pigs. Genes 2024, 15, 186. [Google Scholar] [CrossRef]
- He, S.; Wang, Y.; Luo, Y.; Xue, M.; Wu, M.; Tan, H.; Peng, Y.; Wang, K.; Fang, M. Integrated analysis strategy of genome-wide functional gene mining reveals DKK2 gene underlying meat quality in Shaziling synthesized pigs. BMC Genom. 2024, 25, 30. [Google Scholar] [CrossRef]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef]
- Andrews, S. FastQC: A quality control tool for high throughput sequence data. In Babraham Bioinformatics; Babraham Institute: Cambridge, UK, 2010. [Google Scholar]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.; Park, J.W.; Lu, Z.-x.; Lin, L.; Henry, M.D.; Wu, Y.N.; Zhou, Q.; Xing, Y. rMATS: Robust and flexible detection of differential alternative splicing from replicate RNA-Seq data. Proc. Natl. Acad. Sci. USA 2014, 111, E5593–E5601. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Smyth, G.K.; Shi, W. featureCounts: An efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 2014, 30, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 1–21. [Google Scholar] [CrossRef]
- Yu, G.; Wang, L.-G.; Han, Y.; He, Q.-Y. clusterProfiler: An R package for comparing biological themes among gene clusters. Omics A J. Integr. Biol. 2012, 16, 284–287. [Google Scholar] [CrossRef]





| Adipose | Blood | Heart | Kidney | Lung | Muscle | Ovary | Spleen | |
|---|---|---|---|---|---|---|---|---|
| DASGs | 4187 | 2898 | 173 | 242 | 315 | 244 | 584 | 1721 |
| SDASGs | 1740 | 909 | 41 | 63 | 80 | 56 | 101 | 280 |
| SDASGs/DASGs | 41.56% | 31.37% | 23.70% | 26.03% | 25.40% | 22.95% | 17.29% | 16.27% |
| DEGs | 840 | 960 | 178 | 218 | 358 | 310 | 1348 | 620 |
| SDEGs | 504 | 604 | 69 | 109 | 210 | 161 | 880 | 311 |
| SDEGs/DEGs | 60% | 62.92% | 38.76% | 50% | 58.66% | 51.94% | 65.28% | 50.16% |
| coDASG_DEGs | 83 | 164 | 0 | 4 | 4 | 2 | 15 | 65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Wang, W.; Liu, W.; Li, X.; Yi, G.; Adetula, A.A.; Huang, H.; Tang, Z. Comprehensive Atlas of Alternative Splicing Reveals NSRP1 Promoting Adipogenesis through CCDC18. Int. J. Mol. Sci. 2024, 25, 2874. https://doi.org/10.3390/ijms25052874
Liu L, Wang W, Liu W, Li X, Yi G, Adetula AA, Huang H, Tang Z. Comprehensive Atlas of Alternative Splicing Reveals NSRP1 Promoting Adipogenesis through CCDC18. International Journal of Molecular Sciences. 2024; 25(5):2874. https://doi.org/10.3390/ijms25052874
Chicago/Turabian StyleLiu, Lei, Wei Wang, Weiwei Liu, Xingzheng Li, Guoqiang Yi, Adeyinka Abiola Adetula, Haibo Huang, and Zhonglin Tang. 2024. "Comprehensive Atlas of Alternative Splicing Reveals NSRP1 Promoting Adipogenesis through CCDC18" International Journal of Molecular Sciences 25, no. 5: 2874. https://doi.org/10.3390/ijms25052874






