Abstract
Breast cancer is the second leading contributor to the age-standardized mortality rate, for both sexes and all ages worldwide. In Europe and the United States, it is the second leading cause of mortality, with an incidence rate of about 2.6 million cases per year. Noscapine, a well-known alkaloid used as a cough suppressant, demonstrated anti-tumor effects by triggering apoptosis in various cancer cell lines and has the potential to become another ally against breast, ovarian, colon, and gastric cancer, among other types of malignancy. Apoptosis plays a crucial role in the treatment of cancer. Noscapine affected BAX, CASP8, CASP9, NFKBIA, and RELA gene and protein expression in the MCF-7 and MDA-MB-231 cell lines. Gene expression was higher in tumor than in normal tissue, including the BAX expression levels in lung, ovary, endometrium, colon, stomach, and glioblastoma patients; BCL2L1 expression in endometrium, colon, and stomach patients; CASP8 gene expression levels in lung, endometrium, colon, stomach, and glioblastoma patients; RELA in colon, stomach, and glioblastoma patients; and NFKBIA in glioblastoma patients. It can be concluded that noscapine affected genes and proteins related to apoptosis in cancer cell lines and several types of cancer patients.
1. Introduction
Breast cancer is the second leading contributor to the age-standardized mortality rate, for both sexes and all ages worldwide, and over 680 thousand deaths occurred due to this disease in 2020 []. In Europe and the United States, it is the second leading cause of mortality, with an incidence rate of about 2.6 million cases per year []. There are numerous risk factors known to contribute to the development of cancer, such as age, geographic area, and race [,]; other known or suspected risk factors for cancer are alcohol, chronic inflammation, hormones, radiation, sunlight, cancer-causing substances, infectious agents, obesity, tobacco, etc. Some of these risk factors can be avoided; however, aging cannot [].
Apoptosis is a highly regulated cellular process that takes place in both normal and abnormal conditions. It follows distinct biochemical and genetic pathways and is considered a programmed form of cell death []. One of the major pathways that induces apoptosis is the mitochondria-dependent pathway. Studies have shown that the Bcl-2 family of proteins, located at the outer membranes of mitochondria, plays a crucial role in regulating this intrinsic pathway. These proteins can either inhibit or promote changes in mitochondrial membrane permeability, which are necessary for the release of cytochrome c and other proteins involved in apoptosis. The released proteins then form apoptosomes, which activate caspases, leading to cell death [,,,].
Apoptosis, the process of cell death, is initiated by caspases, a type of protease that specifically target cysteine aspartyl residues. These caspases cleave various essential cellular proteins, disrupting the nuclear scaffold and cytoskeleton necessary for normal cell function [,]. Tumor cells can develop resistance to apoptosis through the expression of anti-apoptotic proteins like Bcl-2 or by reducing or mutating pro-apoptotic proteins such as Bax [,]. It seems that the regulation of both Bcl-2 and Bax expression is controlled by the p53 tumor suppressor gene, since this gene can either suppress or promote changes in mitochondrial membrane permeability, which is required for the release of cytochrome c and other apoptogenic proteins [,].
It has been demonstrated that the cysteine-aspartate-specific protease caspase-8 is involved in several cellular processes, including necroptosis, autophagy and pyroptosis, anoikis, cell apoptosis, and T cell differentiation []. Enzymes called proteases, which control programmed cell death, are members of the caspase protein family. Caspase-9 is an essential component of the mitochondrial apoptotic pathway and a member of the intrinsic pathway. When cells are under stress, the release of cytochrome c from mitochondria starts the intrinsic route. Cytochrome c forms a multiprotein complex known as an apoptosome through interactions with pro-caspase-9, apoptotic protease activating factor 1, and deoxyadenosine triphosphate. The apoptosome activates caspase-9, which triggers a cascade of effector caspases [].
Apoptosis is regulated by the Bcl-2 family, which includes the anti-apoptotic and pro-apoptotic proteins (Bax, Bok, Bak, etc.). These proteins often interact in dimers and act as apoptotic switches. Anti-apoptotic proteins, such as Bcl-2, block the functions of these pro-apoptotic proteins. The pro-apoptotic and anti-apoptotic protein–protein interactions must be inhibited to prevent tumor cells from escaping apoptosis []. Bcl-2 family proteins are tightly involved in the regulation of intrinsic apoptosis [].
NFκB, a transcription factor, plays a crucial role in gene expression related to cell survival. It promotes the upregulation of anti-apoptotic and pro-survival genes, including those from the Bcl-2 family and IAP proteins. In breast cancer cell lines, NFκB increased the expression of anti-apoptotic genes and proteins while decreasing pro-apoptotic ones [,,]. The translocation of NFκB to the nucleus is preceded by phosphorylation, ubiquitination, and the proteolytic degradation of IκBα, an inhibitor of NFκB [,,,]. Previous studies have demonstrated higher levels of NFκB polypeptides (both p100 and p52) in mammary carcinoma cell lines and primary tumors compared to normal breast cells [,,].
A single genetic change will rarely lead to the development of a malignant tumor []. If the cancer cells can evade apoptotic stimuli, they can survive and acquire drug resistance []. The tumors may achieve similar ends by increasing the expression of anti-apoptotic regulators (Bcl-2, Bcl-xL) or of survival signals (Igf1/2), by downregulating pro-apoptotic factors (Bax, Bim, Puma), or by short-circuiting the extrinsic ligand-induced death pathway [].
This review analyzed the role of noscapine and its anti-tumor activity demonstrated by triggering apoptosis on various cancer cell lines from different tissues.
2. Noscapine
Noscapine, a phthalide isoquinoline alkaloid derived from opium obtained from Papaverum somniferum, has been extensively researched and used as an oral anti-tussive agent [,,]. It has demonstrated anti-tumor properties against various cancer types, including lung, cervical, prostate, ovarian, and breast cancer, both in vitro and in vivo, while exerting minimal adverse side effects [,,,,].
For a long time, noscapine has been used as an effective oral medication to treat cough, and it has been recognized as a highly beneficial drug with minimal side effects. Some of its valuable advantages over other microtubule drugs are its low toxicity, water solubility, and suitability for oral administration [,,,]. When taken orally, noscapine has demonstrated a significant decrease in tumor size, while also showing minimal to no toxic effects on the body [,].
Noscapine has demonstrated minimal or negligible toxicity towards various organs, including the kidney, heart, liver, bone marrow, spleen, and small intestine. Furthermore, it does not inhibit primary humoral immune responses in mice. In addition to these benefits, noscapine exhibits good oral tolerance and has a low risk of addiction [,,]. Other advantages of noscapine are its water solubility and suitability for oral administration, making it superior to many other anti-microtubule drugs []. The present review is focused on showing that noscapine has anti-tumor activity in different cell lines, as well as its molecular mechanism of action and its comparison with other drugs.
A sub-therapeutic dose of noscapine (300 mg/kg/day) was administered orally to nude mice with implanted tumors []. The results revealed that various organs, including the liver, kidney, spleen, lung, heart, brain, gut, and sciatic nerve, showed no significant differences or pathological abnormalities when compared to the control group [].
One group of drugs that target microtubules, such as colchicine, nocodazole, and the vinca alkaloids, inhibit the formation of microtubules. Another group, including toxoids and epothilones, promotes the formation of microtubules and stabilizes them. However, these drugs disrupt the normal dynamics of microtubules, leading to cell cycle arrest, usually during prometaphase, blocking the progression of mitosis and ultimately causing cell death [,,,]. Although microtubule-targeting drugs like vinca alkaloids and taxanes have been proven to be effective in treating different types of cancer in humans, their clinical success has been limited due to the development of drug resistance and the associated toxicities, such as leukocytopenia, diarrhea, alopecia, and peripheral neuropathies caused by the blockage of axonal transport [,].
Furthermore, the discovery of new tubulin ligands with antimitotic properties and the identification of their binding sites and modes of action hold promise regarding the developing a new generation of structure-based drugs with improved potential to treat cancer [,,]. Microtubules, which consist of repeating α/β-tubulin heterodimers, are crucial cytoskeletal polymers found in all eukaryotes; these highly dynamic polymers, composed of tubulin subunits, play a vital role in various cellular processes, including cell division, cell motility, and cytoplasmic organization, both in vivo and in vitro [,,].
The dynamic instability of microtubules is driven by the binding and hydrolysis of GTP by tubulin subunits [,,]. Each tubulin monomer binds to one GTP molecule. The binding to α-tubulin at the N-site is permanent, while the binding to β-tubulin at the E-site is replaceable. Polymerization can only occur with dimers containing GTP in their E-site, but, once polymerized, this nucleotide is hydrolyzed and becomes permanent [,,]. The GTP cap model is the most widely accepted hypothesis to explain dynamic instability. According to this model, the microtubule structure is supported by a layer of tubulin subunits at the ends that still contain GTP, while the body of the microtubule consists of GDP–tubulin subunits. When this cap is randomly lost, the microtubule undergoes rapid depolymerization. The assembly and stability of microtubules are regulated by the nucleotide state of tubulin and can be influenced by cellular factors that either stabilize or destabilize microtubules at different locations in the cell or stages in the cell cycle. Disruption of microtubule dynamics can result in the formation of abnormally stable or unstable microtubules, which hinders the normal rearrangement needed for cell division [,,].
Various anti-tubulin agents have been categorized into three main groups based on their binding sites: the colchicine-binding site, those binding at the vinblastine site, and the taxol-binding site. Functionally, these antimitotic ligands can be divided into two classes: those that inhibit microtubule assembly and those that promote microtubule assembly and stabilization. However, regardless of their differences, these agents primarily induce mitotic arrest by inhibiting normal dynamic instability at low concentrations [].
As described in [], noscapine has been tested in phase I and II clinical trials for various human cancers, although its exact mechanism of action as a stabilizer or destabilizer of microtubules has not been determined yet. The drug demonstrated the ability to change the dynamics of microtubule assembly, leading to cell cycle arrest during mitosis and apoptosis in multiple mammalian tumor cell lines [,]. However, unlike other microtubule-targeting anti-cancer drugs, noscapine did not have an impact on microtubule polymerization or the overall polymer mass of tubulin, even at high concentrations. Furthermore, when applied to tissue culture cells, noscapine did not cause significant deformation in cellular microtubules. Instead, it specifically altered the steady-state dynamics of microtubule assembly, which effectively halted the progression of mitosis. This unique characteristic of noscapine suggests that it did not interfere with other microtubule-dependent cellular processes such as organelle distribution and axonal transport, which has been a major concern with many other microtubule-targeting anti-cancer drugs [,,].
A study found that noscapine did not bind to the same site on tubulin as paclitaxel []. Additionally, fluorescence experiments showed that noscapine did not compete with colchicine [,,]. Nonetheless, an in silico investigation revealed a potential binding site for noscapine at the α/β-tubulin interface near the colchicine-binding site. This finding was supported by a study that employed molecular docking and molecular dynamic simulations to identify the predicted binding site of noscapine at the intradomain region of α- and β-tubulin []. Upon the binding of noscapine, there was an observed increase in the stability of the tubulin elements at the E-site components and a decrease in the dynamical motions of certain parts of tubulin located alongside the protofilament. These effects interfered with the longitudinal interactions in microtubules, suggesting a positive impact on microtubule polymerization [,].
Analogs of Noscapine
Analogs of noscapine have been extensively studied. A recent study [] reported compounds derived from noscapine, known as noscapine analogs (Figure 1A–E), which show great potential in cancer treatment.
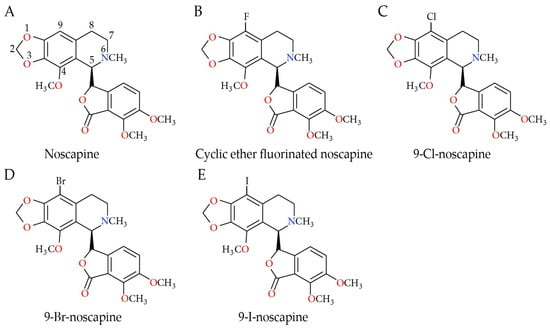
Figure 1.
Structures of (A) noscapine, (B) cyclic ether fluorinated noscapine analog (CEFNA), (C) 9-Cl-noscapine (EM015), (D) 9-Br-noscapine, and (E) 9-I-noscapine.
Among these analogs, the cyclic ether fluorinated noscapine analog (CEFNA) has demonstrated anti-cancer properties []. It has been observed that CEFNA inhibits the growth of, specifically, the MCF-7 and MDA-MB-231 breast cancer cell lines. When these cells were treated with CEFNA at various doses (5, 10, and 25 µM), it caused the formation of multipolar spindles and condensed chromosomes, indicating a halt in the cell cycle at the G2/M phase. Moreover, CEFNA-treated MCF-7 cells exhibited apoptosis, as evidenced by the presence of fragmented micronuclei and apoptotic bodies after 72 h of treatment with 25 µM CEFNA [].
Another analog, 9-Cl-noscapine (EM015), stood out as it contained a chlorine atom in position 9 of the isoquinoline ring system (9-Cl-noscapine). Compared to noscapine, 9-Cl-noscapine exhibited higher affinity for binding to tubulin, resulting in the inhibition of cellular proliferation in various breast cancer cell lines, including MCF-7, MDA-MB-231, ER-MDA-MB-231, BT474, SK-Br3, and T47D. The IC50 value of 9-Cl-noscapine ranged from 2 to 10 µM, which was 15- to 20-fold lower than that of noscapine. Moreover, 9-Cl-noscapine, similar to noscapine, disrupted the cell cycle of breast cancer cells by inducing spindle abnormalities, leading to apoptosis specifically in the G2/M cell cycle phase. Notably, 9-Cl-noscapine required fewer doses than noscapine to achieve these effects []. Furthermore, this study demonstrated that 9-Cl-noscapine effectively inhibited the growth of human breast xenografts compared to the noscapine and control groups. It not only prevented tumor growth but also significantly prolonged the survival of mice almost three-fold. Additionally, the use of 9-Cl-noscapine did not lead to any metastatic lesions or disruption of the hydro-electrolytic acid–base balance. The immune system, specifically the B and T cell lineages, remained unaffected, with minimal or no side effects [].
In a study [], the compound 9-Br-noscapine was found to inhibit cell growth in the MDA-MB-231 (estrogen, progesterone, and ERB2 receptor negative) cell line. Through Western blot analysis, it was observed that treating MDA-MB-231 cells with 9-Br-noscapine at various concentrations (ranging from 0.01 µM to 1000 µM) led to an increase in Bax protein levels and a decrease in Bcl-2 levels. This resulted in an increase in the Bax/Bcl-2 ratio in a time-dependent manner. Additionally, the activation of caspase-3 and PARP cleavage, along with mitochondrial damage and cytochrome c release, induced apoptosis in MDA-MB-231 cells []. The oral administration of 9-Br-noscapine to mice with implanted tumors derived from MDA-MB-231 cells resulted in a reduction in tumor volume on days 16, 22, and 30. The tumor volume reduction was 45%, 59%, and 74%, respectively, compared to mice that received only the vehicle solution. Immunohistochemical analysis revealed the widespread expression of cleaved caspase-3, cleaved PARP, and TUNEL-positive cells in the remaining small regressed tumor sections of the 9-Br-noscapine treatment groups. In conclusion, the study demonstrated that the regression of tumor xenografts was a result of apoptosis induced by 9-Br-noscapine [].
Furthermore, 9-Br-noscapine was administered to nude mice with implanted tumors derived from MDA-MB-231 cells. Tissue sections of the liver, kidney, spleen, brain, heart, lung, gut, and sciatic nerve were examined using H&E staining. The results indicated that 9-Br-noscapine did not cause any detectable pathological abnormalities or metastatic lesions in these organs. Furthermore, a complete blood count analysis revealed that 9-Br-noscapine treatment did not alter the counts of red blood cells or white blood cells, hemoglobin concentrations, or hematocrit in mice with hormone-refractory xenograft tumors [].
Halogenated noscapine analogs (9-F-noscapine, 9-Cl-noscapine, 9-Br-noscapine, and 9-I-noscapine) have greater tubulin-binding activity compared to noscapine. These medications have a more pronounced impact on the cell cycle profile, causing heightened arrest at the G2/M phase when compared to noscapine. At concentrations of 5 and 10 µM, the effects of these halogenated compounds on the cell cycle differ in terms of the extent of their detrimental effects, increasing the percentage of sub-G1 cells with hypodiploid DNA content, which is indicative of apoptosis [,]. The authors discovered that when the MCF-7 (estrogen and progesterone receptor positive, ErbB2 receptor negative) cell line was treated with halogenated noscapine, there was a noticeable presence of spindles and condensed chromosomes that were not properly organized at the metaphase plate, indicating the onset of mitotic arrest as early as 12 h and maximizing at 24 h of drug treatment. These findings demonstrated that the insertion of halogens into noscapine enhanced its tubulin-binding activity and influenced its potential as a therapeutic agent for various types of cancer cells, particularly the MCF-7 cell line [].
3. Noscapine’s Effect in Several Types of Cancer
3.1. Breast Cancer
Several studies have demonstrated that noscapine decreased the levels of the NFκB, P-IκBα, Bcl-2, and survivin proteins, all known as anti-apoptotic proteins. On the other hand, the expression of the cleaved IκBα, PARP, Bax, caspase-8, caspase-9, and caspase-3 proteins, which are pro-apoptotic proteins, were increased compared to the control group. The ratio of Bax/Bcl2 observed in the control group was lower than that observed when treated with noscapine [,,].
A previous study [] demonstrated that noscapine, a natural opium alkaloid with purity of 97%, induced apoptosis in breast cancer cell lines, specifically MDA-MB-231 and MCF-7. This study compared the effects of noscapine on MCF-10F, a normal breast cell line, used as a control. Such results showed that noscapine exhibited lower toxicity in normal cells while effectively functioning as an anti-cancer agent by triggering apoptosis in breast cancer cells. This was supported by the findings of increased Bax gene and protein expression in all three cell lines, as well as a decrease in the Bcl-xL gene and Bcl-2 protein expression. Noscapine increased Bax protein expression in the MCF-10F cell line, but there was no significant effect on Bcl-2 expression. However, noscapine increased Bax protein expression and decreased Bcl-2 expression in the MCF-7 cell line. Similarly, noscapine increased Bax protein expression and decreased Bcl-2 expression in the MDA-MB-231 breast cancer cell line; there was also an increase in the Bax/Bcl-2 ratio in all three types of cells. The amount of 53 µM noscapine induced an increase in the Bax/Bcl-2 protein expression ratio from 0.03 to 0.70 in the MCF-10F cell line This ratio represents the balance between the pro-apoptotic protein Bax and the anti-apoptotic protein Bcl-2, suggesting a shift towards apoptosis. Similarly, in the MCF-7 cell line treated with 30 µM noscapine, the Bax/Bcl-2 ratio increased from 0.71 to 1.08. Furthermore, treatment with 20 µM noscapine resulted in an increase in the Bax/Bcl-2 ratio from 0.99 to 3.64 in the MDA-MB-231 cell line. Noscapine not only upregulated the expression levels of the caspase-8 and caspase-9 genes but also facilitated the cleavage of caspase-8. Additionally, noscapine downregulated the expression of anti-apoptotic genes and proteins while increasing the expression of pro-apoptotic genes and proteins. These effects may be attributed to the downregulation of the NFκB gene and protein expression. NFκB is a transcription factor associated with breast cancer initiation and progression. Furthermore, noscapine was found to enhance the expression of the IκBα gene, NFKBIA []. Our previous work [] also indicated that noscapine-treated MCF-10F cells increased Bax, caspase-8, and IκBα gene expression. However, there was no change in Bcl-xL, caspase-9, or NFκB under the effect of this drug. Noscapine increased Bax, caspase-9, and IκBα gene expression while decreasing the levels of Bcl-xL, and NFκB in MCF-7. Caspase-8 showed no significant effect of noscapine. Noscapine-treated MDA-MB-231 cells showed increased Bax, caspase-9, and Caspase-8 gene expression and decreased levels of Bcl-xL, and NFκB. IκBα showed no significant effect of noscapine.
Additionally, some authors [] indicated that noscapine upregulated the caspase-8 and caspase-9 gene expression levels in the MCF-10F and MDA-MB-231 breast cancer cell lines. It also promoted the cleavage of caspase-8, suggesting the involvement of both extrinsic and intrinsic apoptosis pathways in noscapine-induced apoptosis.
On the other hand, noscapine can be used in combination with certain drugs to treat various types of cancer. For example, doxorubicin is commonly used as a chemotherapy agent for patients with metastatic breast cancer []. When noscapine and doxorubicin were used together (30 µM and 0.4 µg/mL, respectively), it was observed that the expression of certain proteins involved in apoptosis, such as Bax, caspase-8, caspase-9, caspase-3, and cleaved caspase-3, increased. Additionally, the expression of NFκB, IκBα, P-IκBα, and Bcl-2 decreased when compared to the control group. When noscapine and doxorubicin were used in combination, the expression of the VEGF protein decreased compared to each substance alone, and there was a decrease in survivin protein expression compared to the single-drug treatment and the control group []. Furthermore, the combined treatment induced apoptosis in 65% of the tumor cells, whereas noscapine and doxorubicin alone induced apoptosis in 20% and 32% of the tumor cells, respectively [].
Noscapine, either alone or in combination with doxorubicin, demonstrated efficacy against triple-negative breast cancer cell lines and enhanced the anti-cancer effects of doxorubicin synergistically []. This effect was achieved by deactivating the NFκB and anti-angiogenic pathways and promoting apoptosis. As a result, these findings suggest that a combination of orally administered noscapine and doxorubicin could be a potential therapy for aggressive triple-negative breast cancer.
A previous study indicated that the oral administration of noscapine at doses ranging from 150 to 550 mg/kg/day resulted in a significant reduction in tumor volume in MDA-MB-231 xenografts. However, the combined treatment proved to be the most effective in inhibiting tumor growth compared to individual treatments with either doxorubicin or noscapine [].
Cell proliferation was evaluated by observing the effects of docetaxel, tamoxifen, and noscapine in the MCF-7 and MDA-MB-231 cell lines; although noscapine showed cytotoxic effects in a time- and dose-dependent manner, MDA-MB-231 cells were more susceptible to its effects; however, noscapine inhibited MCF-7 and MDA-MB-231 cells’ proliferation in vitro, which was comparable to the effects of tamoxifen and docetaxel []. The combination of N-3-Br-benzyl-noscapine (Br-Bn-Nos), a derivative of noscapine, and docetaxel was demonstrated to have improved anti-cancer potential compared to the single regimen []. In drug-resistant xenografts, noscapine at low concentrations with docetaxel decreased the tumor volume in comparison with each substance alone and downregulated the expression of anti-apoptotic factors and multidrug resistance proteins [].
3.2. Clinical Relevance Analyzed by Bioinformatics in Breast Cancer Patients
Identifying genes linked to specific tissues has proven valuable in elucidating their biological roles and understanding diseases such as breast cancer (BRCA) and its various subtypes, including BRCA-Basal, BRCA-Her2, BRCA-Lum-A, and BRCA-Lum-B [].
Hence, genes such as BAX, the BCL2-associated X gene; BCL2L1, the BCL2-like 1 gene (Bcl-xL); CASP8, the caspase-8 gene; CASP9, the caspase-9 gene; RELA, the RELA proto-oncogene (NF-kB subunit); and NFKBIA, the NFKB inhibitor alpha gene (IkBα) were extracted from the Tumor Immune Estimation Resource database v 2.0 (TIMER2.0, http://timer.cistrome.org, accessed on 14 September 2023) []. The results showed whether such genes could have therapeutic target potential, to discover the co-expression patterns of genes across TCGA cancer types such as BRCA. The ER status raw data were extracted from the University of California, Santa Cruz (https://xena.ucsc.edu, accessed on 14 September 2023) UCSC Xena functional genomics explorer [].
3.2.1. Gene Correlation in Breast Cancer Patients
Correlations were found between TP53 gene expression and BAX, BCL2L1, CASP8, CASP9, NFKBIA, and RELA (Figure 2).
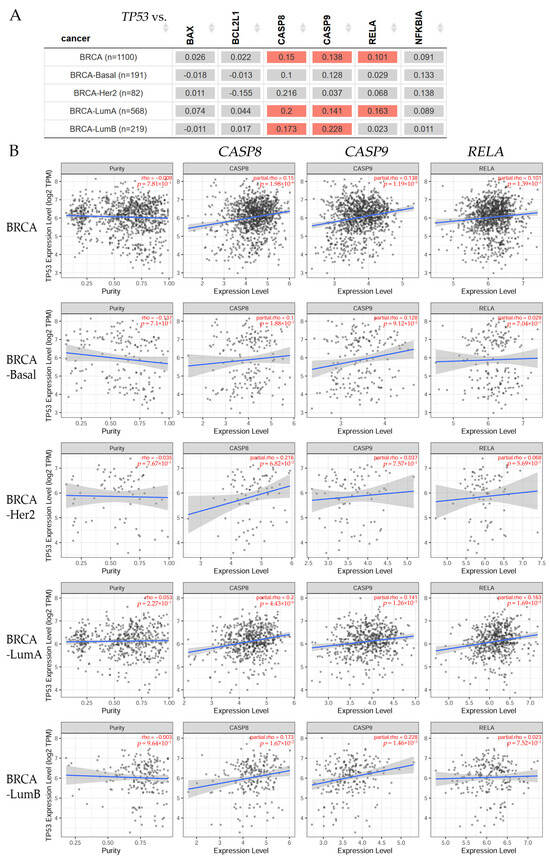
Figure 2.
(A) The heatmap table shows the correlations between TP53, the tumor protein p53 gene, and BAX, the BCL2-associated X gene; BCL2L1, the BCL2-like 1 gene; CASP8, the caspase-8 gene; CASP9, the caspase-9 gene; NFKBIA, the NFKB inhibitor alpha gene; and RELA, the RELA proto-oncogene (NFkB, Rel A) in breast invasive carcinoma (BRCA) subtypes. The red color indicates a statistically significant positive correlation (Spearman’s, p < 0.05) and gray denotes a non-significant result. (B) Box plots show significant correlations between TP53 expression with purity adjustment (left) and CASP8, CASP9, and RELA gene expression levels (right) in BRCA subtypes. Correlation values for each analysis are stated in red on the right (Spearman’s, p < 0.05). Raw data were extracted from TIMER2.0 (http://timer.cistrome.org), accessed on 14 September 2023 [].
The results in Figure 2A show that there was no correlation between TP53 and BAX, BCL2L1, and NFKBIA gene expression levels; however, there was a significant (p < 0.05) difference between TP53 gene expression and CASP8 and CASP9 for BRCA-LumA and BRCA-LumB patients. The correlation between TP53 and RELA was significant (p < 0.05) for BRCA-LumA patients, corroborated by box plots (Figure 2B) showing significant (p < 0.05) correlations between TP53 expression with purity adjustment (left) and CASP8, CASP9, and RELA gene expression levels (right).
3.2.2. Differential Gene Expression Levels between Tumor and Normal Tissues across Various Breast Cancer Subtypes
Studies analyzed the differential gene expression levels between tumor and normal tissue across various breast cancer subtypes, as shown in Figure 3.
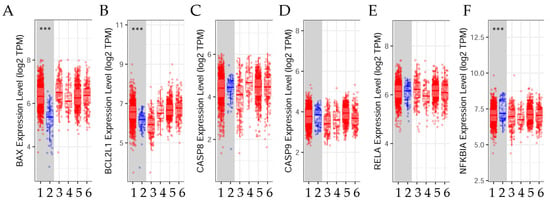
Figure 3.
Differential gene expression levels between tumor and normal tissue in breast invasive carcinoma (BRCA). The box plots show the distribution of gene expression levels of (A) BAX, the BCL2-associated X gene; (B) BCL2L1, the BCL2-like 1 gene; (C) CASP8, the caspase-8 gene; (D) CASP9, the caspase-9 gene; (E) RELA, the RELA proto-oncogene; and (F) NFKBIA, the NFKB inhibitor alpha gene in tumor versus normal tissue (Wilcoxon rank-sum test, ***: p < 0.001). Raw data were extracted from the Tumor Immune Estimation Resource database v 2.0 (TIMER2.0, http://timer.cistrome.org), accessed on 14 September 2023 []. (1) BRCA. Tumor (n = 1093). (2) BRCA. Normal (n = 112). (3) BRCA-Basal. Tumor (n = 190). (4) BRCA-Her2. Tumor (n = 82). (5) BRCA-LumA. Tumor (n = 564). (6) BRCA-LumB. Tumor (n = 217).
The results show that BAX and BCL2L1 were significantly (p < 0.001) higher in the tumor tissue than in the normal tissue when comparisons were made, whereas NFKBIA was significantly (p < 0.001) higher in the normal than in the cancer tissue. There was no significant relationship between tumor and normal tissue in terms of CASP8, CASP9, and RELA gene expression.
3.2.3. Estrogen Receptor Status
Bioinformatic studies analyzed BAX, BCL2L1, CASP8, CASP 9, RELA, and NFKBIA, gene expression, and estrogen receptor status, as seen in Figure 4.
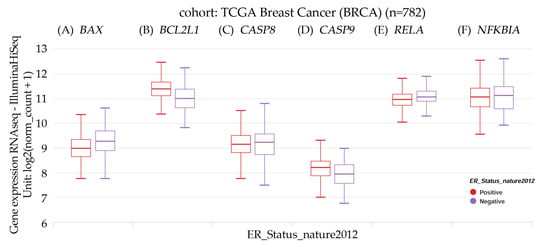
Figure 4.
Xena Chart View showing box plot transcript expression of (A) BAX, the BCL2-associated X gene; (B) BCL2L1, the BCL2-like 1 gene; (C) CASP8, the caspase-8 gene; (D) CASP9, the caspase-9 gene; (E) RELA, the RELA proto-oncogene; and (F) NFKBIA, the NFKB inhibitor alpha gene in breast invasive carcinoma (BRCA). Cohort: TCGA Breast Cancer (BRCA), n = 782, stratified by nature2012 for estrogen receptor status (ER) (one-way ANOVA, p < 0.05). Raw data were extracted from the University of California, Santa Cruz (ena.ucsc.edu) UCSC Xena functional genomics explorer, accessed on 14 September 2023 [].
The results indicated that those BRCA patients characterized by BAX and RELA gene expression had a significant (p = 1.097 × 10−8 and p = 0.0001753, respectively) negative ER status, whereas those with BCL2L1 and CASP9 had a significant (p = 0.000 and p = 6.778 × 10−7, respectively) positive ER status. There was no significant difference in those patients having CASP8 and NFKBIA gene expression.
3.2.4. Disease Stage Factor in Several Breast Cancer Subtypes
Gene expression concerning disease stage factors across various breast cancer subtypes is analyzed in Table 1. The clinical relevance of the gene expression associated with the disease stage is important.

Table 1.
Clinical relevance of genes across various cancer types analyzed by the disease stage factor.
The analysis of the clinical stages of patients with breast invasive carcinoma indicated that the BAX, BCL2L1, CASP8, CASP9, RELA, and NFKBIA gene expression levels were significantly (p < 0.001) higher in stages 3 and 4 for all BRCA patients, and in stage 4 for BRCA-LumA patients, than in other clinical stages. Additionally, stage 4 showed a significant (either p < 0.05 or p < 0.01) difference in BRCA-Her2 and BRCA-LumB patients. These genes showed no significance at any stage in BRCA-Basal patients.
3.3. Lung Cancer
Gemcitabine, a pyrimidine nucleoside–antimetabolite agent, has shown effectiveness against various types of cancer in humans. When combined with noscapine, the anti-cancer activity of gemcitabine against non-small-cell lung cancer increased in an additive to synergistic manner. This synergism resulted in higher levels of apoptosis compared to treatment with either drug alone. Additionally, in mice with implanted tumors, the combination of noscapine and gemcitabine led to a reduction in tumor volume. This reduction decreased the expression of anti-apoptotic and angiogenic proteins, as well as increasing the expression of pro-apoptotic proteins, within the tumor tissue [,].
3.4. Ovarian Cancer
Apoptosis was observed in human ovarian carcinoma cells when exposed to noscapine (97% purity) at a concentration of 20 µM, with the extent of apoptosis increasing as the duration of exposure to the drug increased. These findings were evaluated using techniques such as TUNEL and Annexin V []. Additionally, noscapine at a concentration of 40 µM exhibited cytotoxic effects on paclitaxel-resistant human ovarian carcinoma cell lines [].
A recent study showed that noscapine-induced apoptosis in ovarian cancer cell lines was associated with the JNK pathway. The study found that treating the cell lines with noscapine led to increased levels of c-Jun protein and the phosphorylation of c-Jun by JNK; this phosphorylation influenced the expression of apoptotic genes and proteins. Like other microtubule drugs, noscapine also inhibited the microtubule dynamics, caused mitotic arrest, induced apoptosis, and exhibited strong anti-tumor activity [,,]. However, the exact molecular mechanisms that underlie the apoptosis and mitotic arrest induced by anti-microtubule agents, as well as the relationship between these two events, remain unclear.
Cisplatin, a primary chemotherapy drug for ovarian cancer, is known for its high toxicity and the development of resistance in cancer cells. In contrast, the use of noscapine enhanced the sensitivity to cisplatin in ovarian cancer cells resistant to drugs []. By combining 2.5 µM noscapine with cisplatin at different concentrations (0, 2, 4, and 8 µg/mL), the proliferation of ovarian cisplatin-resistant cancer cells was reduced, the expression levels of genes and anti-apoptotic proteins were decreased, and the expression of genes and pro-apoptotic proteins was increased compared to using either drug alone. Moreover, the combination of cisplatin and noscapine was found to effectively reduce tumor growth in nude mice [].
3.5. Endometrium
The effect of noscapine was examined on the expression of apoptotic genes, growth scores, angiogenesis, and nitric oxide secretion in the eutopic endometrium in endometriosis patients and normal endometrium patients. Results indicated that the expression of apoptotic genes increased, while the levels of Bcl-2 and Sirt1 decreased [,].
3.6. Colon Cancer
In a recent study, it was observed that the use of noscapine (97% purity) resulted in a reduction in cell proliferation in human colon cancer cells. The effectiveness of noscapine varied based on the dosage (001, 01, 1, 10, 100, or 1000 µM) and duration of treatment (0, 12, 24, 36, 48, or 72 h). After 72 h, the IC50 value for noscapine was found to be 75 µM. Additionally, the study revealed that the cells treated with noscapine experienced cell cycle arrest at the G2/M stage, leading to an increase in apoptosis, as confirmed by flow cytometry analysis. Furthermore, it was observed that at 75 µM, noscapine-induced apoptosis resulted in the upregulation of Bax expression and downregulation of Bcl-2 expression. This apoptotic response was accompanied by an increase in the protein expression of caspase-3 and caspase-9, as well as a decrease in survivin expression. These findings suggest that the induction of apoptosis by noscapine occurs through the mitochondrial pathway [].
Another study showed that the inhibition of p38 mitogen-activated protein kinase (MAPK) increased the sensitivity of the 5-fluorouracil-resistant SW480 human colon cancer cells to noscapine (at a dose of 25 µM). This was achieved by suppressing proliferation, the induction of cell cycle arrest and apoptosis, and the reversal of multidrug resistance in the SW480 cells treated with 20 µg/mL 5-fluorouracil [].
3.7. Stomach Cancer
In a research study, it was found that noscapine exhibited cytotoxic effects on gastric cancer cell lines (BGC823, SGC7901, MGC803, and HGC27). The cytotoxicity was observed in a dose-dependent and time-dependent manner, with varying concentrations of 0, 50, 100, or 150 µM and time intervals of 0, 12, 24, or 36 h. The cytotoxic activity was attributed to apoptosis, as evidenced by chromatin condensation observed through the DAPI method and quantified using flow cytometry. The apoptotic effect was also found to be dependent on the dosage of noscapine. Furthermore, the expression of the Bax and Bcl-2 proteins was examined, revealing an increase in Bax and a decrease in Bcl-2 levels after treatment with noscapine. Additionally, the activity of caspase-9 and caspase-3 was enhanced following noscapine treatment [].
3.8. Neuroblastoma
The drug noscapine was found to effectively inhibit cellular proliferation in various neuroblastoma cell lines (SK-SY5Y, SH-EP1, SK-N-MC, SK-N-AS, LA1-55N, LA1-5S, NB1643, NB1691, SK-N-SH, and IMR32). This inhibition was dose-dependent, with IC50 values ranging from 21 to 101 µM for most of these cell lines. Noscapine induced mitotic arrest at the G2/M phase of the cell cycle. Additionally, treatment with noscapine led to the activation of caspase-3 and the cleavage of PARP in the treated cells. When SK-SY5Y and LA1-5S cells were exposed to noscapine, there was a significant reduction in the levels of survivin mRNA and protein as early as 12 h after treatment. Importantly, the ectopic expression of survivin provided significant protection against noscapine-induced cytoplasmic histone-associated apoptotic DNA fragmentation [].
3.9. Clinical Relevance Analyzed by Bioinformatics in Several Types of Cancer
3.9.1. Gene Correlation in Different Cancer Patients
Correlations were found between TP53 gene expression and BAX, BCL2L1, CASP8, CASP9, RELA, and NFKBIA in lung adenocarcinoma (LUAD), lung squamous cell carcinoma (LUSC), ovarian serous (OV), uterine corpus endometrial carcinoma (UCEC), colon adenocarcinoma (COAD), stomach adenocarcinoma (STAD), and glioblastoma multiforme (GBM), as seen in Figure 5.
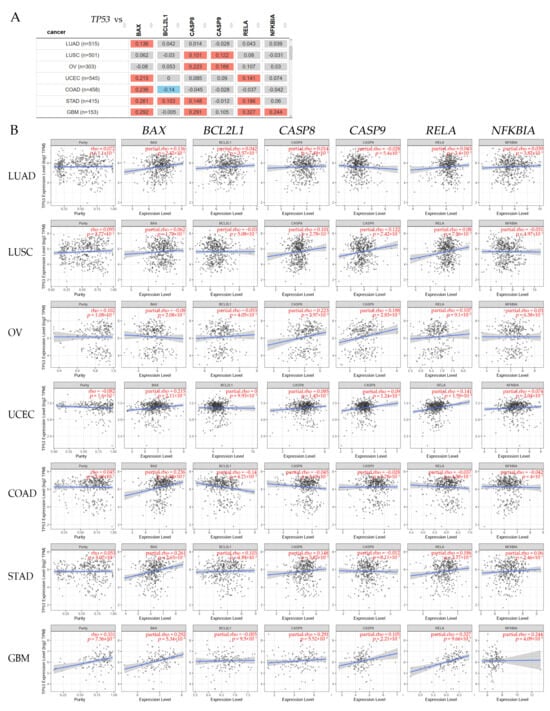
Figure 5.
(A) Correlations between TP53, the tumor protein p53 gene, and BAX, the BCL2-associated X gene; BCL2L1, the BCL2-like 1 gene; CASP8, the caspase-8 gene; CASP9, the caspase-9 gene; NFKBIA, the NFKB inhibitor alpha gene; and RELA, the RELA proto-oncogene (NFkB, Rel A) in different types of cancer. The red color indicates a statistically significant positive correlation (Spearman’s, p < 0.05), blue indicates a statistically significant negative correlation (Spearman’s, p < 0.05), and gray denotes a non-significant result. (B). Box plots show significant correlations between TP53 expression with purity adjustment (left) and BAX, BCL2L1, CASP8, CASP9, RELA, and NFKBIA gene expression levels (right) in different types of cancer. Correlation values for each analysis are stated in red on the right (Spearman’s, p < 0.05). Raw data were extracted from TIMER2.0 (http://timer.cistrome.org), accessed on 10 January 2024 []. TCGA abbreviations—LUAD: lung adenocarcinoma; LUSC: lung squamous cell carcinoma; OV: ovarian serous; UCEC: uterine corpus endometrial carcinoma; COAD: colon adenocarcinoma; STAD: stomach adenocarcinoma; GBM: glioblastoma multiforme.
The results in Figure 5A indicate that TP53 had a significantly (p < 0.05) positive correlation with BAX expression in LUAD, UCEC, COAD, STAD, and GBM patients, being also positively and significantly correlated (p < 0.05) with BCL2L1 expression levels in STAD patients. Meanwhile, it was negatively correlated with BCL2L1 expression in COAD patients. There was a significant (p < 0.05) positive correlation with CASP8 expression levels in LUSC, OV, STAD, and GBM. The TP53 and CASP9 expression levels showed a significant (p < 0.05) positive correlation in LUSC and OV patients. NFKBIA showed a significant (p < 0.05) positive correlation with TP53 expression in GBM. Additionally, TP53 presented a significant correlation with the RELA expression levels in COAD, STAD, and GBM patients. Representative box plots (Figure 5B) corroborated these significant (p < 0.05) correlations between TP53 expression with purity adjustment (left) and the BAX, BCL2L1, CASP8, CASP9, RELA, and NFKBIA gene expression levels (right).
3.9.2. Differential Gene Expression Levels between Tumor and Normal Tissue across Different Types of Cancer
Studies analyzed the differential gene expression levels between tumor and normal tissue across different types of cancer, as shown in Figure 6.
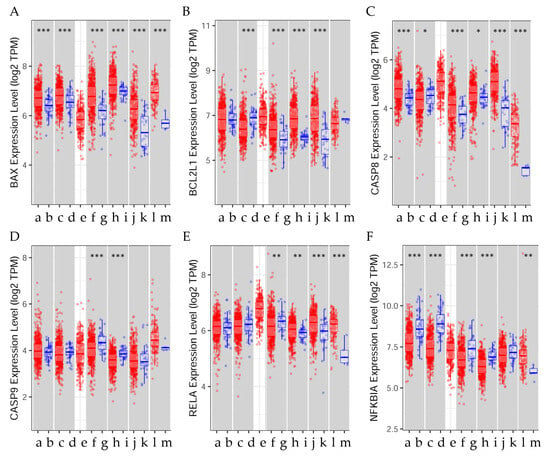
Figure 6.
Differential gene expression levels between tumor and normal tissue in different types of cancer. The box plots show the distribution of the gene expression levels of (A) BAX, the BCL2-associated X gene; (B) BCL2L1, the BCL2-like 1 gene; (C) CASP8, the caspase-8 gene; (D) CASP9, the caspase-9 gene; (E) RELA, the RELA proto-oncogene; and (F) NFKBIA, the NFKB inhibitor alpha gene in tumor versus normal tissue (Wilcoxon rank-sum test, *: p < 0.05, **: p < 0.01, ***: p < 0.001). Raw data were extracted from the Tumor Immune Estimation Resource database v 2.0 (TIMER2.0, http://timer.cistrome.org), accessed on 14 September 2023 []. (a) LUAD. Tumor (n = 515). (b) LUAD. Normal (n = 59). (c) LUSC. Tumor (n = 501). (d) LUSC. Normal (n = 51). (e) OV. Tumor (n = 303). (f) UCEC. Tumor (n = 545). (g) UCEC. Normal (n = 35). (h) COAD. Tumor (n = 457). (i) COAD. Normal (n = 41). (j) STAD. Tumor (n = 415). (k) STAD. Normal (n = 35). (l) GBM. Tumor (n = 153). (m) GBM. Normal (n = 5).
Figure 6 shows that the BAX expression levels were significantly (p < 0.001) higher in the tumor tissue than in the normal tissue when comparisons were made in LUAD, LUSC, OV, UCEC, COAD, STAD, and GBM patients. Similarly, BCL2L1 expression was significantly (p < 0.001) higher in the tumor than in the normal tissue in UCEC, COAD, and STAD patients, whereas CASP8 was significantly (p < 0.001) higher in the normal tissue than in the tumor tissue (either p < 0.05 or p < 0.001) in LUSC. CASP8 was higher in the tumor tissue than in the normal tissue in LUAD, UCEC, COAD, STAD, and GBM; however, it was significantly (p < 0.05) higher in the normal than in the tumor tissue in LUSC patients. The CASP9 expression level was higher in the normal tissue than the tumor tissue in UCEC and COAD patients. The RELA expression level was significantly (p < 0.01) higher in the normal tissue than in the tumor tissue in UCEC patients, whereas it was significantly (either p < 0.01 or p < 0.001) higher in the tumor than the normal tissue in COAD, STAD, and GBM patients. The NFKBIA expression level was significantly (p < 0.001) higher in the normal than in the cancer tissue in LUAD, LUSC, UCEC, and COAD patients; however, it was significantly (p < 0.01) higher in the tumor than the normal tissue in GBM patients.
Scheme 1 shows a comparison between normal and malignant tissue, indicating that the BAX expression levels were higher in the tumor tissue than in the normal tissue in LUAD, LUSC, OV, UCEC, COAD, STAD, and GBM patients. BCL2L1 expression was higher in the tumor than in the normal tissue in UCEC, COAD, and STAD patients, whereas it was higher in the normal tissue than in the tumor tissue in LUSC. The CASP8 gene expression levels were higher in the tumor tissue than in the normal tissue in LUAD, UCEC, COAD, STAD, and GBM; however, it was higher in the normal than in the tumor tissue in LUSC patients. The CASP9 gene expression level was higher in the normal tissue than the tumor tissue in UCEC and COAD patients. The RELA gene expression level was higher in the normal tissue than in the tumor tissue in UCEC patients, whereas it was higher in the tumor than the normal tissue in COAD, STAD, and GBM patients. The NFKBIA gene expression level was higher in the normal than in the cancer tissue in LUAD, LUSC, UCEC, and COAD patients; however, it was higher in the tumor than in the normal tissue in GBM patients. The expression level of NFKBIA was considerably higher in normal tissue than in tumors, suggesting that it may act as a tumor suppressor.
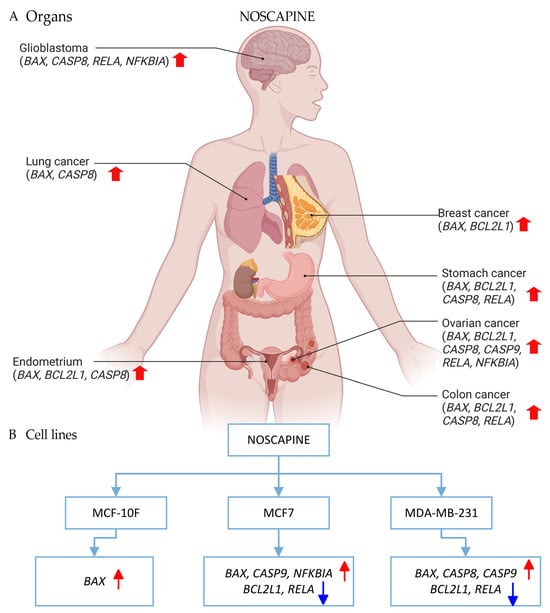
Scheme 1.
(A) Noscapine causes changes in apoptotic genes in cancer in human patients, such as glioblastoma, lung, endometrium, breast, stomach, ovary, and colon. The red arrow indicates an increase and the blue arrow indicates a decrease in gene expression. (B) It also causes changes in apoptotic genes and protein expression in breast cancer cell lines.
The studies analyzed the effect of noscapine on apoptosis in various cancer cell lines. For example, noscapine increased the gene expression levels of BAX in the MCF-10F cell line; it increased BAX, CASP9, and NFKBIA in MCF-7; and it increased BAX, CASP9, and CASP8 in the MDA-MB-231 cell line. The results indicated that those BRCA patients characterized by BAX and RELA gene expression had a negative ER status, whereas those with BCL2L1 and CASP9 had a positive ER status. There was no significant difference in those patients having CASP8 and NFKBIA gene expression. The analysis of the clinical stages of patients with breast invasive carcinoma indicated that the BAX, BCL2L1, CASP8, CASP9, RELA, and NFKBIA gene expression levels were higher in stages 3 and 4 for all BRCA patients, and in stage 4 for BRCA-LumA patients, than in other clinical stages. Additionally, stage 4 showed a difference in BRCA-Her2 and BRCA-LumB patients but not at any stage in BRCA-Basal patients.
4. Conclusions
It can be concluded that noscapine affected genes and proteins related to apoptosis in cancer cell lines and several types of cancer patients.
Author Contributions
Formal analysis, G.M.C., E.O.Q.-V. and L.A.C.; Writing—original draft, E.O.Q.-V.; Writing—review and editing, G.M.C. and L.A.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Fondo Nacional de Desarrollo Científico y Tecnológico (FONDECYT), grant numbers FONDECYT 1200656 (G.M.C.) and FONDECYT 1231537 (G.M.C.).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
TIMER20 is freely available at http://timercistromeorg (accessed on 6 August 2021); the UCSC Xena online exploration tools are freely available at http://xenaucscedu/ (accessed on 20 August 2021). The data generated in the present study may be requested from the corresponding author.
Acknowledgments
The authors wish to thank Agencia Nacional de Investigación y Desarrollo (ANID) of the Government of Chile for their financial support.
Conflicts of Interest
The authors declare no conflicts of interest The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- IARC. Cancer Today: Estimated Age-Standardized Incidence and Mortality Rates (World) in 2020. International Agency for Research on Cancer 2023. Available online: https://gco.iarc.fr/today/ (accessed on 22 August 2023).
- Millimouno, F.M.; Dong, J.; Yang, L.; Li, J.; Li, X. Targeting apoptosis pathways in cancer and perspectives with natural compounds from mother nature. Cancer Prev. Res. 2014, 7, 1081–1107. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2015. CA Cancer J. Clin. 2015, 65, 5–29. [Google Scholar] [CrossRef] [PubMed]
- NIH. Risk Factors for Cancer National Cancer Institute. Available online: https://www.cancer.gov/about-cancer/causes-prevention/risk (accessed on 6 February 2024).
- Wong, R.S. Apoptosis in cancer: From pathogenesis to treatment. J. Exp. Clin. Cancer Res. 2011, 30, 87. [Google Scholar] [CrossRef]
- Hata, A.N.; Engelman, J.A.; Faber, A.C. The BCL2 Family: Key Mediators of the Apoptotic Response to Targeted Anticancer Therapeutics. Cancer Discov. 2015, 5, 475–487. [Google Scholar] [CrossRef]
- Zong, W.X.; Rabinowitz, J.D.; White, E. Mitochondria and Cancer. Mol. Cell 2016, 61, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.; Watari, H.; AbuAlmaaty, A.; Ohba, Y.; Sakuragi, N. Apoptosis and molecular targeting therapy in cancer. Biomed. Res. Int. 2014, 2014, 150845. [Google Scholar] [CrossRef]
- Li, M.X.; Dewson, G. Mitochondria and apoptosis: Emerging concepts. F1000Prime Rep. 2015, 7, 42. [Google Scholar] [CrossRef]
- Ionov, Y.; Yamamoto, H.; Krajewski, S.; Reed, J.C.; Perucho, M. Mutational inactivation of the proapoptotic gene BAX confers selective advantage during tumor clonal evolution. Proc. Natl. Acad. Sci. USA 2000, 97, 10872–10877. [Google Scholar] [CrossRef]
- Shalini, S.; Dorstyn, L.; Dawar, S.; Kumar, S. Old, new and emerging functions of caspases. Cell Death Differ. 2015, 22, 526–539. [Google Scholar] [CrossRef]
- Yilmaz, S.G.; Yencilek, F.; Yildirim, A.; Yencilek, E.; Isbir, T. Effects of Caspase 9 Gene Polymorphism in Patients with Prostate Cancer. In Vivo 2017, 31, 205–208. [Google Scholar] [CrossRef][Green Version]
- Afzal, M.; Alarifi, A.; Abduh, N.A.Y.; Ayub, A.; Muddassir, M. Identification of anti-cancer organometallic compounds by inhibition of BCL-2/Bax interactions. Comput. Biol. Med. 2023, 167, 107657. [Google Scholar] [CrossRef]
- Shi, Z.E.; Zhang, M.Y.; Liu, J.Y.; Zhang, W.D.; Hu, D.M.; Wang, Q.X.; Ji, X.L.; Jiang, Y.Y.; Qu, Y.Q. Autophagy Induced by BCL2-Related ceRNA Network Participates in the Occurrence of COPD. Int. J. Chronic Obstr. Pulm. Dis. 2022, 17, 791–808. [Google Scholar] [CrossRef] [PubMed]
- Barham, W.; Chen, L.; Tikhomirov, O.; Onishko, H.; Gleaves, L.; Stricker, T.P.; Blackwell, T.S.; Yull, F.E. Aberrant activation of NF-kappaB signaling in mammary epithelium leads to abnormal growth and ductal carcinoma in situ. BMC Cancer 2015, 15, 647. [Google Scholar] [CrossRef]
- Li, F.; Zhang, J.; Arfuso, F.; Chinnathambi, A.; Zayed, M.E.; Alharbi, S.A.; Kumar, A.P.; Ahn, K.S.; Sethi, G. NF-kappaB in cancer therapy. Arch. Toxicol. 2015, 89, 711–731. [Google Scholar] [CrossRef] [PubMed]
- Sethi, G.; Ahn, K.S.; Aggarwal, B.B. Targeting nuclear factor-kappa B activation pathway by thymoquinone: Role in suppression of antiapoptotic gene products and enhancement of apoptosis. Mol. Cancer Res. 2008, 6, 1059–1070. [Google Scholar] [CrossRef] [PubMed]
- Sung, B.; Ahn, K.S.; Aggarwal, B.B. Noscapine, a benzylisoquinoline alkaloid, sensitizes leukemic cells to chemotherapeutic agents and cytokines by modulating the NF-kappaB signaling pathway. Cancer Res. 2010, 70, 3259–3268. [Google Scholar] [CrossRef]
- Khan, S.; Lopez-Dee, Z.; Kumar, R.; Ling, J. Activation of NFkB is a novel mechanism of pro-survival activity of glucocorticoids in breast cancer cells. Cancer Lett. 2013, 337, 90–95. [Google Scholar] [CrossRef]
- Cao, Y.; Karin, M. NF-kappaB in mammary gland development and breast cancer. J. Mammary Gland. Biol. Neoplasia 2003, 8, 215–223. [Google Scholar] [CrossRef]
- Cogswell, P.C.; Guttridge, D.C.; Funkhouser, W.K.; Baldwin, A.S., Jr. Selective activation of NF-kappa B subunits in human breast cancer: Potential roles for NF-kappa B2/p52 and for Bcl-3. Oncogene 2000, 19, 1123–1131. [Google Scholar] [CrossRef]
- Wang, W.; Nag, S.A.; Zhang, R. Targeting the NFkappaB signaling pathways for breast cancer prevention and therapy. Curr. Med. Chem. 2015, 22, 264–289. [Google Scholar] [CrossRef]
- Wertz, I.E. TNFR1-activated NF-kappaB signal transduction: Regulation by the ubiquitin/proteasome system. Curr. Opin. Chem. Biol. 2014, 23, 71–77. [Google Scholar] [CrossRef]
- Croce, C.M. Oncogenes and cancer. N. Engl. J. Med. 2008, 358, 502–511. [Google Scholar] [CrossRef]
- Kassam, F.; Enright, K.; Dent, R.; Dranitsaris, G.; Myers, J.; Flynn, C.; Fralick, M.; Kumar, R.; Clemons, M. Survival outcomes for patients with metastatic triple-negative breast cancer: Implications for clinical practice and trial design. Clin. Breast Cancer 2009, 9, 29–33. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Afzali, M.; Ghaeli, P.; Khanavi, M.; Parsa, M.; Montazeri, H.; Ghahremani, M.H.; Ostad, S.N. Non-addictive opium alkaloids selectively induce apoptosis in cancer cells compared to normal cells. Daru 2015, 23, 16. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, S.M.; Mackenzie, A.J.; Spina, D.; Page, C.P. The pharmacology of cough. Trends Pharmacol. Sci. 2004, 25, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Takahama, K.; Shirasaki, T. Central and peripheral mechanisms of narcotic antitussives: Codeine-sensitive and -resistant coughs. Cough 2007, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Gupta, K.; Yao, J.; Ye, K.; Panda, D.; Giannakakou, P.; Joshi, H.C. Paclitaxel-resistant human ovarian cancer cells undergo c-Jun NH2-terminal kinase-mediated apoptosis in response to noscapine. J. Biol. Chem. 2002, 277, 39777–39785. [Google Scholar] [CrossRef] [PubMed]
- Aneja, R.; Lopus, M.; Zhou, J.; Vangapandu, S.N.; Ghaleb, A.; Yao, J.; Nettles, J.H.; Zhou, B.; Gupta, M.; Panda, D.; et al. Rational design of the microtubule-targeting anti-breast cancer drug EM015. Cancer Res. 2006, 66, 3782–3791. [Google Scholar] [CrossRef] [PubMed]
- DeBono, A.J.; Xie, J.H.; Ventura, S.; Pouton, C.W.; Capuano, B.; Scammells, P.J. Synthesis and biological evaluation of N-substituted noscapine analogues. ChemMedChem 2012, 7, 2122–2133. [Google Scholar] [CrossRef] [PubMed]
- Manchukonda, N.K.; Naik, P.K.; Santoshi, S.; Lopus, M.; Joseph, S.; Sridhar, B.; Kantevari, S. Rational design, synthesis, and biological evaluation of third generation alpha-noscapine analogues as potent tubulin binding anti-cancer agents. PLoS ONE 2013, 8, e77970. [Google Scholar] [CrossRef]
- Aneja, R.; Zhou, J.; Zhou, B.; Chandra, R.; Joshi, H.C. Treatment of hormone-refractory breast cancer: Apoptosis and regression of human tumors implanted in mice. Mol. Cancer Ther. 2006, 5, 2366–2377. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, M.O.; Dahlstrom, B.; Eckernas, S.A.; Johansson, M.; Alm, A.T. Pharmacokinetics of oral noscapine. Eur. J. Clin. Pharmacol. 1990, 39, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Dahlstrom, B.; Mellstrand, T.; Lofdahl, C.G.; Johansson, M. Pharmacokinetic properties of noscapine. Eur. J. Clin. Pharmacol. 1982, 22, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Chougule, M.B.; Patel, A.R.; Jackson, T.; Singh, M. Antitumor activity of Noscapine in combination with Doxorubicin in triple negative breast cancer. PLoS ONE 2011, 6, e17733. [Google Scholar] [CrossRef] [PubMed]
- Gajewski, M.M.; Alisaraie, L.; Tuszynski, J.A. Peloruside, laulimalide, and noscapine interactions with beta-tubulin. Pharm. Res. 2012, 29, 2985–2993. [Google Scholar] [CrossRef]
- Zhou, J.; Panda, D.; Landen, J.W.; Wilson, L.; Joshi, H.C. Minor alteration of microtubule dynamics causes loss of tension across kinetochore pairs and activates the spindle checkpoint. J. Biol. Chem. 2002, 277, 17200–17208. [Google Scholar] [CrossRef]
- Gali-Muhtasib, H.; Hmadi, R.; Kareh, M.; Tohme, R.; Darwiche, N. Cell death mechanisms of plant-derived anticancer drugs: Beyond apoptosis. Apoptosis 2015, 20, 1531–1562. [Google Scholar] [CrossRef]
- Crown, J.; O’Leary, M. The taxanes: An update. Lancet 2000, 355, 1176–1178. [Google Scholar] [CrossRef]
- Rahmanian-Devin, P.; Baradaran Rahimi, V.; Jaafari, M.R.; Golmohammadzadeh, S.; Sanei-Far, Z.; Askari, V.R. Noscapine, an Emerging Medication for Different Diseases: A Mechanistic Review. Evid. Based Complement. Alternat. Med. 2021, 2021, 8402517. [Google Scholar] [CrossRef]
- Aneja, R.; Vangapandu, S.N.; Lopus, M.; Viswesarappa, V.G.; Dhiman, N.; Verma, A.; Chandra, R.; Panda, D.; Joshi, H.C. Synthesis of microtubule-interfering halogenated noscapine analogs that perturb mitosis in cancer cells followed by cell death. Biochem. Pharmacol. 2006, 72, 415–426. [Google Scholar] [CrossRef]
- Nogales, E. Structural insights into microtubule function. Annu. Rev. Biochem. 2000, 69, 277–302. [Google Scholar] [CrossRef]
- Ye, K.; Zhou, J.; Landen, J.W.; Bradbury, E.M.; Joshi, H.C. Sustained activation of p34(cdc2) is required for noscapine-induced apoptosis. J. Biol. Chem. 2001, 276, 46697–46700. [Google Scholar] [CrossRef]
- Saxton, W.M.; Stemple, D.L.; Leslie, R.J.; Salmon, E.D.; Zavortink, M.; McIntosh, J.R. Tubulin dynamics in cultured mammalian cells. J. Cell Biol. 1984, 99, 2175–2186. [Google Scholar] [CrossRef]
- Stanton, R.A.; Gernert, K.M.; Nettles, J.H.; Aneja, R. Drugs that target dynamic microtubules: A new molecular perspective. Med. Res. Rev. 2011, 31, 443–481. [Google Scholar] [CrossRef]
- Lopus, M.; Naik, P.K. Taking aim at a dynamic target: Noscapinoids as microtubule-targeted cancer therapeutics. Pharmacol. Rep. 2015, 67, 56–62. [Google Scholar] [CrossRef]
- Sajadian, S.; Vatankhah, M.; Majdzadeh, M.; Kouhsari, S.M.; Ghahremani, M.H.; Ostad, S.N. Cell cycle arrest and apoptogenic properties of opium alkaloids noscapine and papaverine on breast cancer stem cells. Toxicol. Mech. Methods 2015, 25, 388–395. [Google Scholar] [CrossRef]
- Ye, K.; Ke, Y.; Keshava, N.; Shanks, J.; Kapp, J.A.; Tekmal, R.R.; Petros, J.; Joshi, H.C. Opium alkaloid noscapine is an antitumor agent that arrests metaphase and induces apoptosis in dividing cells. Proc. Natl. Acad. Sci. USA 1998, 95, 1601–1606. [Google Scholar] [CrossRef] [PubMed]
- Alisaraie, L.; Tuszynski, J.A. Determination of noscapine’s localization and interaction with the tubulin-alpha/beta heterodimer. Chem. Biol. Drug Des. 2011, 78, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Aneja, R.; Vangapandu, S.N.; Joshi, H.C. Synthesis and biological evaluation of a cyclic ether fluorinated noscapine analog. Bioorg Med. Chem. 2006, 14, 8352–8358. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudian, M.; Rahimi-Moghaddam, P. The anti-cancer activity of noscapine: A review. Recent Pat. Anti-Cancer Drug Discov. 2009, 4, 92–97. [Google Scholar] [CrossRef]
- Quisbert-Valenzuela, E.O.; Calaf, G.M. Apoptotic effect of noscapine in breast cancer cell lines. Int. J. Oncol. 2016, 48, 2666–2674. [Google Scholar] [CrossRef]
- Kocak, C.; Kocak, F.E.; Ozturk, B.; Tekin, G.; Vatansev, H. Cytotoxic, anti-proliferative and apoptotic effects of noscapine on human estrogen receptor positive (MCF-7) and negative (MDA-MB-231) breast cancer cell lines. Bratisl. Lek. Listy 2020, 121, 43–50. [Google Scholar] [CrossRef]
- Dash, S.G.; Kantevari, S.; Pandey, S.K.; Naik, P.K. Synergistic interaction of N-3-Br-benzyl-noscapine and docetaxel abrogates oncogenic potential of breast cancer cells. Chem. Biol. Drug Des. 2021, 98, 466–479. [Google Scholar] [CrossRef]
- Doddapaneni, R.; Patel, K.; Chowdhury, N.; Singh, M. Reversal of drug-resistance by noscapine chemo-sensitization in docetaxel resistant triple negative breast cancer. Sci. Rep. 2017, 7, 15824. [Google Scholar] [CrossRef]
- Toda, S.; Frankel, N.W.; Lim, W.A. Engineering cell-cell communication networks: Programming multicellular behaviors. Curr. Opin. Chem. Biol. 2019, 52, 31–38. [Google Scholar] [CrossRef]
- Li, T.; Fu, J.; Zeng, Z.; Cohen, D.; Li, J.; Chen, Q.; Li, B.; Liu, X.S. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020, 48, W509–W514. [Google Scholar] [CrossRef]
- Goldman, M.J.; Craft, B.; Hastie, M.; Repecka, K.; McDade, F.; Kamath, A.; Baberjee, A.; Luo, Y.; Rogers, D.; Brooks, A.N.; et al. Visualizing and interpreting cancer genomics data via the Xena platform. Nat. Biotechnol. 2020, 38, 675–678. [Google Scholar] [CrossRef]
- Chougule, M.B.; Patel, A.; Sachdeva, P.; Jackson, T.; Singh, M. Enhanced anticancer activity of gemcitabine in combination with noscapine via antiangiogenic and apoptotic pathway against non-small cell lung cancer. PLoS ONE 2011, 6, e27394. [Google Scholar] [CrossRef]
- Shen, W.; Liang, B.; Yin, J.; Li, X.; Cheng, J. Noscapine Increases the Sensitivity of Drug-Resistant Ovarian Cancer Cell Line SKOV3/DDP to Cisplatin by Regulating Cell Cycle and Activating Apoptotic Pathways. Cell Biochem. Biophys. 2015, 72, 203–213. [Google Scholar] [CrossRef]
- Hsin, Y.H.; Chen, C.F.; Huang, S.; Shih, T.S.; Lai, P.S.; Chueh, P.J. The apoptotic effect of nanosilver is mediated by a ROS- and JNK-dependent mechanism involving the mitochondrial pathway in NIH3T3 cells. Toxicol. Lett. 2008, 179, 130–139. [Google Scholar] [CrossRef]
- Martin, L.T.P.; Nachtigal, M.W.; Selman, T.; Nguyen, E.; Salsman, J.; Dellaire, G.; Dupre, D.J. Bitter taste receptors are expressed in human epithelial ovarian and prostate cancers cells and noscapine stimulation impacts cell survival. Mol. Cell Biochem. 2019, 454, 203–214. [Google Scholar] [CrossRef]
- Khazaei, M.R.; Nasr-Esfahani, M.H.; Chobsaz, F.; Khazaei, M. Noscapine Inhibiting the Growth and Angiogenesis of Human Eutopic Endometrium of Endometriosis Patients through Expression of Apoptotic Genes and Nitric Oxide Reduction in Three-Dimensional Culture Model. Iran. J. Pharm. Res. 2019, 18, 836–845. [Google Scholar] [CrossRef]
- Yang, Z.R.; Liu, M.; Peng, X.L.; Lei, X.F.; Zhang, J.X.; Dong, W.G. Noscapine induces mitochondria-mediated apoptosis in human colon cancer cells in vivo and in vitro. Biochem. Biophys. Res. Commun. 2012, 421, 627–633. [Google Scholar] [CrossRef]
- Han, Z.; Meng, L.; Huang, X.; Tan, J.; Liu, W.; Chen, W.; Zou, Y.; Cai, Y.; Huang, S.; Chen, A.; et al. Inhibition of p38 MAPK increases the sensitivity of 5-fluorouracil-resistant SW480 human colon cancer cells to noscapine. Oncol. Lett. 2022, 23, 52. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Luo, X.J.; Liao, F.; Lei, X.F.; Dong, W.G. Noscapine induces mitochondria-mediated apoptosis in gastric cancer cells in vitro and in vivo. Cancer Chemother. Pharmacol. 2011, 67, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; He, J.; Li, S.; Cao, G.; Tang, S.; Tong, Q.; Joshi, H.C. Noscapine induced apoptosis via downregulation of survivin in human neuroblastoma cells having wild type or null p53. PLoS ONE 2012, 7, e40076. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).