Efficient Production of 9,22-Dihydroxy-23,24-bisnorchol-4-ene-3-one from Phytosterols by Modifying Multiple Genes in Mycobacterium fortuitum
Abstract
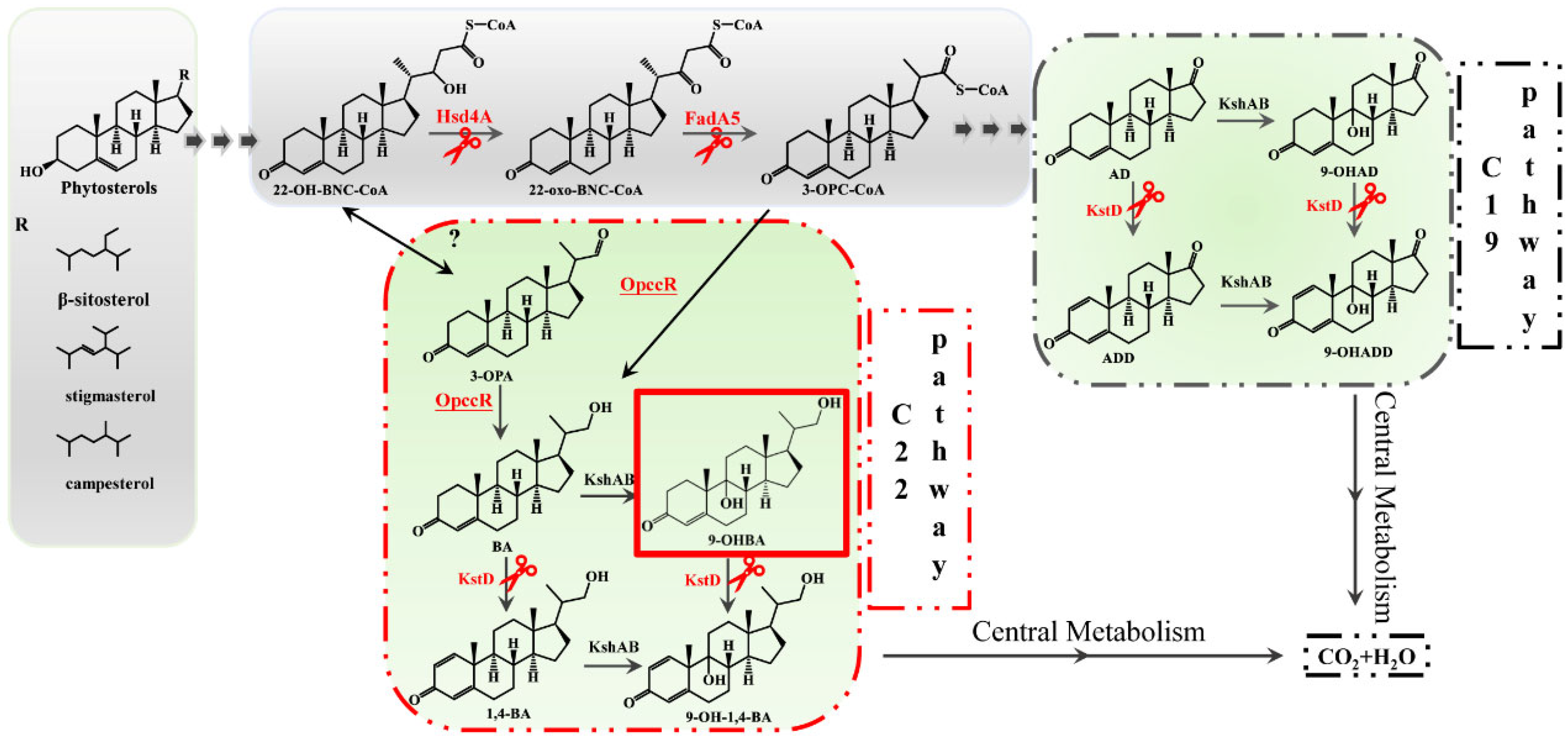
:1. Introduction
2. Results
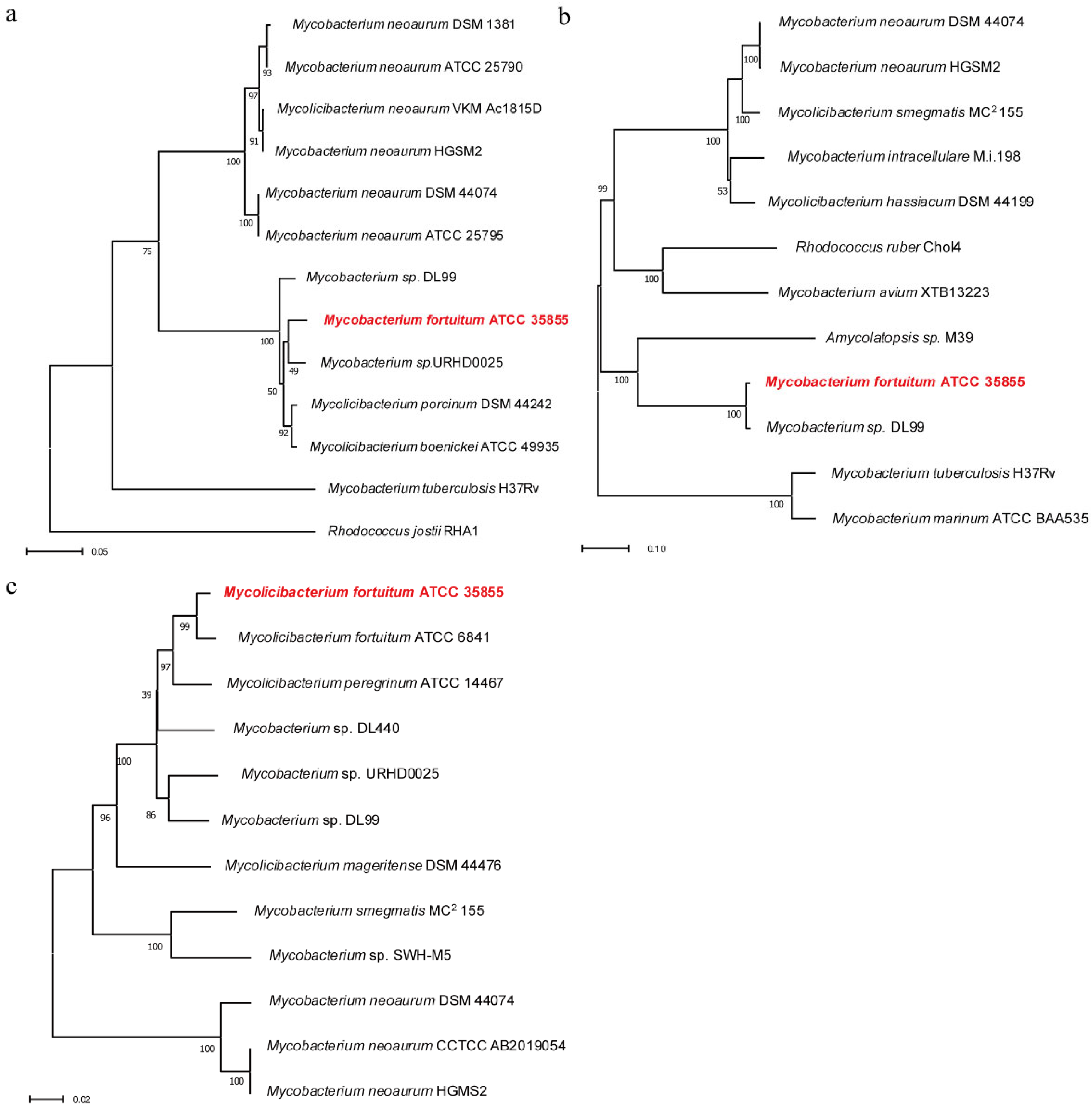
2.1. Construction of Phylogenetic Tree of Hsd4A, FadA5, and OpccR
2.2. Hsd4A—The Key Enzyme in the C19 Pathway
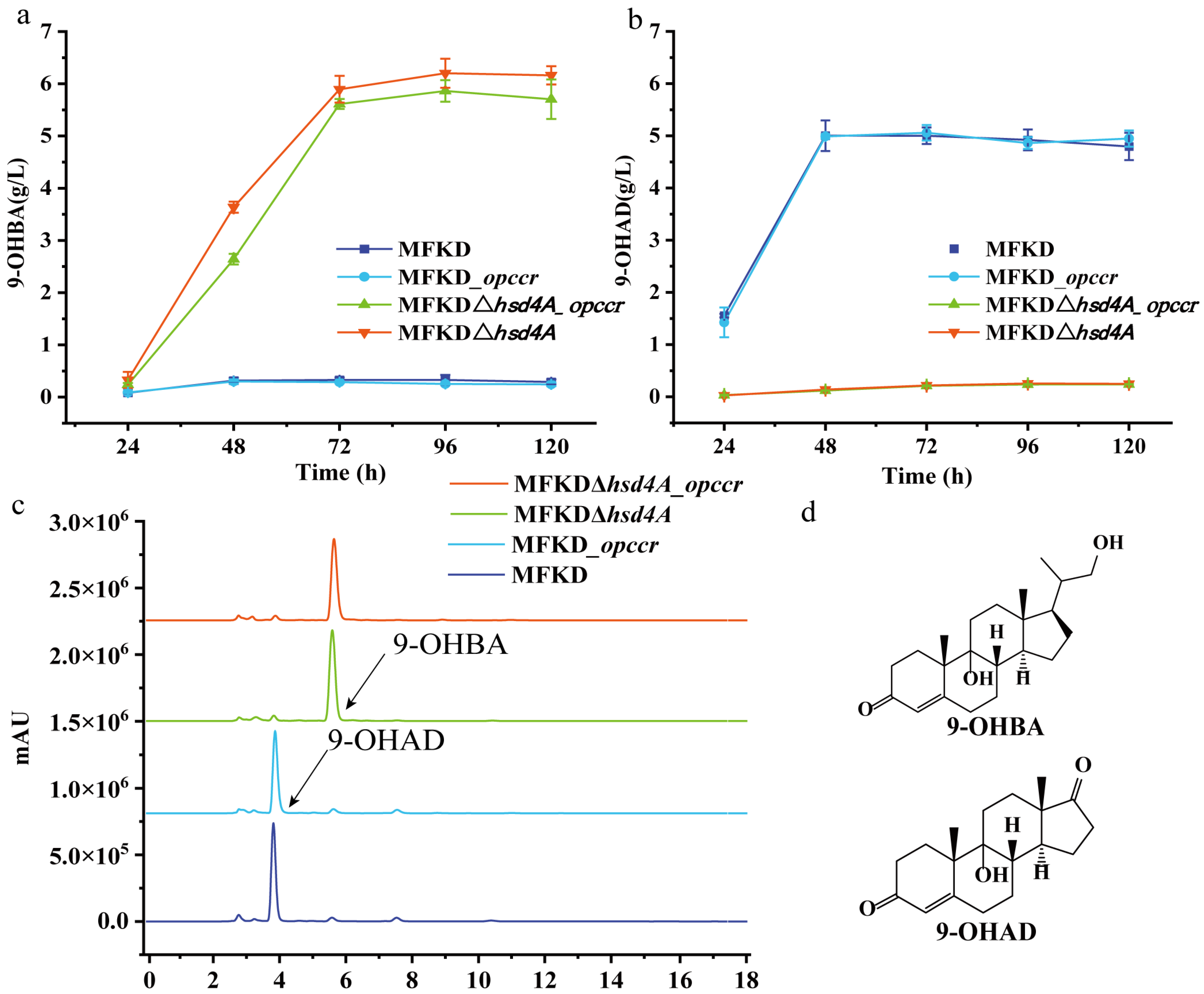
2.3. Deletion of fadA5 Improves the Selectivity of 9-OHBA by Eliminating 9-OHAD
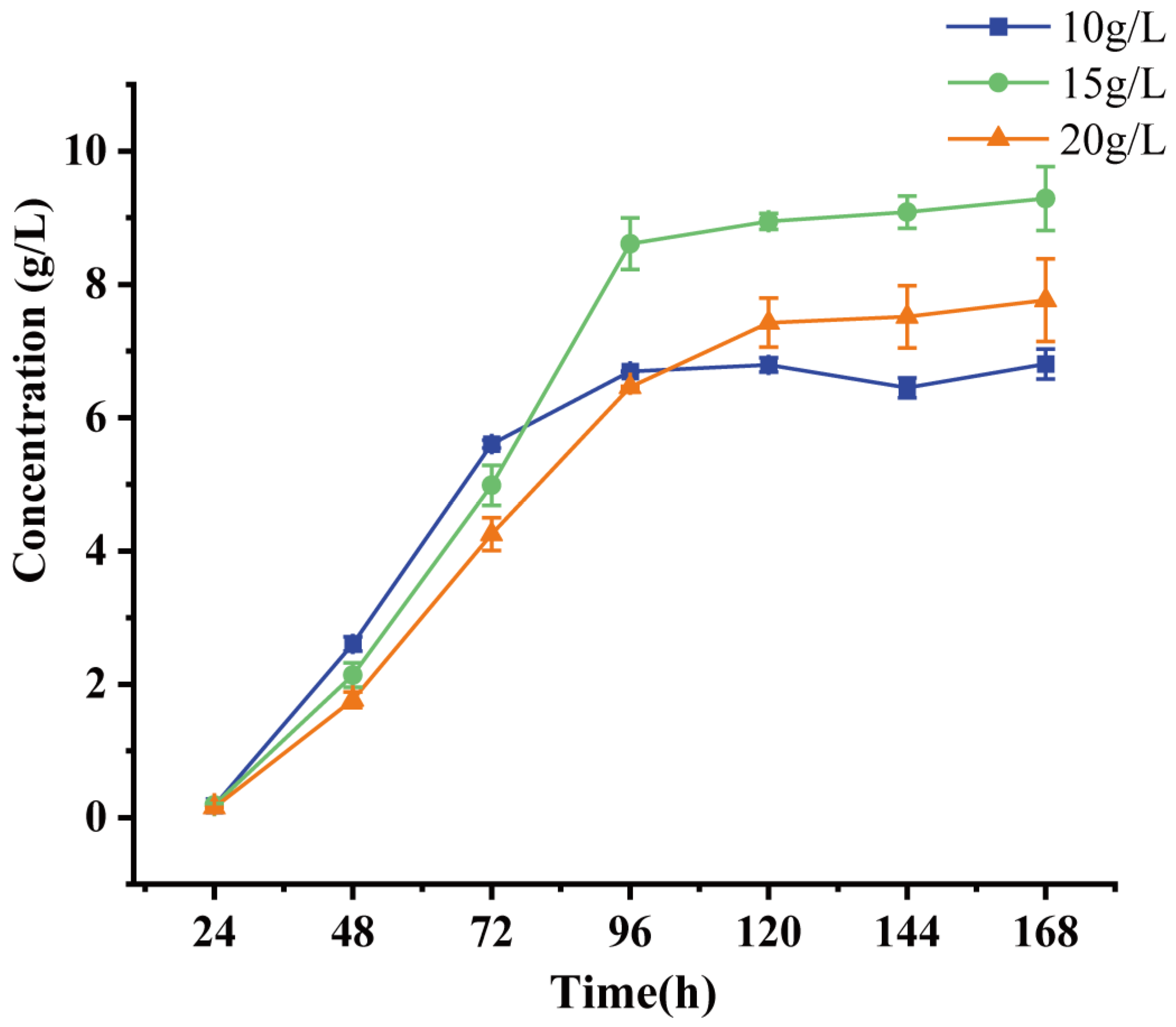
2.4. Evaluation of the 9-OHBA-Producing Strain
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains, Plasmids, Reagents, and Culture Conditions
4.2. Bioinformatic Analysis
4.3. Construction of Mutant Strains
| Name | Description | Source |
|---|---|---|
| Strains | ||
| Escherichia coli | E. coli DH5α | Vazyme Biotech Co., Ltd., Nanjing, China |
| MFKD | 9-OHAD producer, kstD1&2&3&4&5 deletion mutant of ATCC 35855 | Our lab |
| MFKD_opccR | ATCC 35855 opccR overexpression in MFKD via p40-opccR | This study |
| MFKDΔhsd4A | hsd4A deletion mutant of MFKD | This study |
| MFKDΔhsd4A_ opccR | ATCC 35855 opccR overexpression in MFKDΔhsd4A via p40-opccR | This study |
| MFKDΔhsd4AΔfadA5 | hsd4A and fadA5 double-deletion mutant of MFKD | This study |
| Plasmids | ||
| pKADel | Plasmid for allelic exchange, Pag85-lacZ Phsp60-sacB, AprR, KanR | [19] |
| pKADelΔhsd4A | pKADel carrying two homologous arms of hsd4A | This study |
| pKADelΔfadA5 | pKADel carrying two homologous arms of fadA5 | This study |
| p40 | pMV306 with Psmyc promoter, KanR | [19] |
| p40-opccR | p40 possessing opccR from M. fortuitum ATCC 35855 | This study |
4.4. Bioconversion and Analytical Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Donova, M.V. Steroid Bioconversions. Methods Mol. Biol. 2017, 1645, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rugutt, J.K.; Rugutt, K.J. Antimycobacterial activity of steroids, long-chain alcohols and lytic peptides. Nat. Prod. Res. 2012, 26, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Seth, D.; Kamat, D. Intranasal Steroid Therapy for Allergic Rhinitis. Pediatr. Ann. 2019, 48, e43–e48. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J.; Ji, W.T.; Song, L.; Tao, X.Y.; Zhao, M.; Gao, B.; Meng, H.; Wang, F.Q.; Wei, D.Z. Transformation of phytosterols into pregnatetraenedione by a combined microbial and chemical process. Green Chem. 2022, 24, 3759–3771. [Google Scholar] [CrossRef]
- Peng, H.; Wang, Y.; Jiang, K.; Chen, X.; Zhang, W.; Zhang, Y.; Deng, Z.; Qu, X. A Dual Role Reductase from Phytosterols Catabolism Enables the Efficient Production of Valuable Steroid Precursors. Angew. Chem. Int. Ed. Engl. 2021, 60, 5414–5420. [Google Scholar] [CrossRef] [PubMed]
- Zhao, A.; Zhang, X.; Li, Y.; Wang, Z.; Lv, Y.; Liu, J.; Alam, M.A.; Xiong, W.; Xu, J. Mycolicibacterium cell factory for the production of steroid-based drug intermediates. Biotechnol. Adv. 2021, 53, 107860. [Google Scholar] [CrossRef] [PubMed]
- Donova, M.V. Transformation of steroids by actinobacteria: A review. Appl. Biochem. Microbiol. 2007, 43, 1–14. [Google Scholar] [CrossRef]
- Almeida, C.A.S.; Baggio, S.R.; Mariutti, L.R.B.; Bragagnolo, N. One-step rapid extraction of phytosterols from vegetable oils. Food Res. Int. 2020, 130, 108891. [Google Scholar] [CrossRef]
- Andryushina, V.A.; Rodina, N.V.; Stytsenko, T.S.; Huy, L.D.; Druzhinina, A.V.; Yaderetz, V.V.; Voishvillo, N.E. Conversion of soybean sterols into 3,17-diketosteroids using actinobacteria Mycobacterium neoaurum, Pimelobacter simplex, and Rhodococcus erythropolis. Appl. Biochem. Microbiol. 2011, 47, 270–273. [Google Scholar] [CrossRef]
- Fernandez-Cabezon, L.; Galan, B.; Garcia, J.L. New Insights on Steroid Biotechnology. Front. Microbiol. 2018, 9, 958. [Google Scholar] [CrossRef]
- McLeod, M.P.; Warren, R.L.; Hsiao, W.W.L.; Araki, N.; Myhre, M.; Fernandes, C.; Miyazawa, D.; Wong, W.; Lillquist, A.L.; Wang, D.; et al. The complete genome of Rhodococcus sp RHA1 provides insights into a catabolic powerhouse. Proc. Natl. Acad. Sci. USA 2006, 103, 15582–15587. [Google Scholar] [CrossRef]
- Schaefer, C.M.; Lu, R.; Nesbitt, N.M.; Schiebel, J.; Sampson, N.S.; Kisker, C. FadA5 a thiolase from Mycobacterium tuberculosis: A steroid-binding pocket reveals the potential for drug development against tuberculosis. Structure 2015, 23, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Wipperman, M.F.; Sampson, N.S.; Thomas, S.T. Pathogen roid rage: Cholesterol utilization by Mycobacterium tuberculosis. Crit. Rev. Biochem. Mol. 2014, 49, 269–293. [Google Scholar] [CrossRef] [PubMed]
- Yao, K.; Wang, F.Q.; Zhang, H.C.; Wei, D.Z. Identification and engineering of cholesterol oxidases involved in the initial step of sterols catabolism in Mycobacterium neoaurum. Metab. Eng. 2013, 15, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Donova, M.V.; Egorova, O.V. Microbial steroid transformations: Current state and prospects. Appl. Microbiol. Biotechnol. 2012, 94, 1423–1447. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.C.; He, B.R.; Zhang, J.X.; Yuan, C.Y.; Han, S.W.; Du, G.L.; Shi, J.P.; Sun, J.S.; Zhang, B.G. Phytosterol conversion into C9 non-hydroxylated derivatives through gene regulation in. Appl. Microbiol. Biotechnol. 2023, 107, 7635–7646. [Google Scholar] [CrossRef]
- Xu, L.Q.; Liu, Y.J.; Yao, K.; Liu, H.H.; Tao, X.Y.; Wang, F.Q.; Wei, D.Z. Unraveling and engineering the production of 23,24-bisnorcholenic steroids in sterol metabolism. Sci. Rep. 2016, 6, 21928. [Google Scholar] [CrossRef]
- Yuan, C.Y.; Ma, Z.G.; Zhang, J.X.; Liu, X.C.; Du, G.L.; Sun, J.S.; Shi, J.P.; Zhang, B.G. Production of 9,21-dihydroxy-20-methyl-pregna-4-en-3-one from phytosterols in Mycobacterium neoaurum by modifying multiple genes and improving the intracellular environment. Microb. Cell Fact. 2021, 20, 229. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, J.; Yuan, C.; Du, G.; Han, S.; Shi, J.; Sun, J.; Zhang, B. Improving the production of 9alpha-hydroxy-4-androstene-3,17-dione from phytosterols by 3-ketosteroid-Delta(1)-dehydrogenase deletions and multiple genetic modifications in Mycobacterium fortuitum. Microb. Cell Fact. 2023, 22, 53. [Google Scholar] [CrossRef]
- Song, S.; He, J.; Gao, M.; Huang, Y.; Cheng, X.; Su, Z. Loop pathways are responsible for tuning the accumulation of C19- and C22-sterol intermediates in the mycobacterial phytosterol degradation pathway. Microb. Cell Fact. 2023, 22, 19. [Google Scholar] [CrossRef]
- Zhang, J.X.; Zhang, R.J.; Song, S.K.; Su, Z.D.; Shi, J.P.; Cao, H.J.; Zhang, B.G. Whole-Genome Analysis of Mycobacterium neoaurum DSM 1381 and the Validation of Two Key Enzymes Affecting C22 Steroid Intermediates in Sterol Metabolism. Int. J. Mol. Sci. 2023, 24, 6148. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Schaefer, C.M.; Neshitt, N.M.; Kuper, J.; Kisker, C.; Sampson, N.S. Catabolism of the Cholesterol Side Chain in Mycobacterium tuberculosis Is Controlled by a Redox-Sensitive Thiol Switch. ACS Infect. Dis. 2017, 3, 666–675. [Google Scholar] [CrossRef]
- Turk, M.; Lietzow, R. Stabilized nanoparticles of phytosterol by rapid expansion from supercritical solution into aqueous solution. AAPS PharmSciTech 2004, 5, e56. [Google Scholar] [CrossRef] [PubMed]
- Mancilla, R.A.; Little, C.; Amoroso, A. Efficient Bioconversion of High Concentration Phytosterol Microdispersion to 4-Androstene-3,17-Dione (AD) by Mycobacterium sp B3805. Appl. Biochem. Biotechnol. 2018, 185, 494–506. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.F.; Huang, Y.L.; Rathman, J.F.; Yang, S.T. Lecithin-enhanced biotransforation of cholesterol to androsta-1,4-diene-3,17-dione and androsta-4-ene-3,1 7-dione. J. Chem. Technol. Biotechnol. 2002, 77, 1349–1357. [Google Scholar] [CrossRef]
- Zhou, L.F.; Li, H.; Xu, Y.A.; Liu, W.; Zhang, X.M.; Gong, J.S.; Xu, Z.H.; Shi, J.S. Effects of a nonionic surfactant TX-40 on 9 alpha-hydroxyandrost-4-ene-3,17-dione biosynthesis and physiological properties of Mycobacterium sp. LY-1. Process Biochem. 2019, 87, 89–94. [Google Scholar] [CrossRef]
- Su, L.Q.; Xu, S.P.; Shen, Y.B.; Xia, M.L.; Ren, X.X.; Wang, L.F.; Shang, Z.H.; Wang, M. The Sterol Carrier Hydroxypropyl-beta-Cyclodextrin Enhances the Metabolism of Phytosterols by Mycobacterium neoaurum. Appl. Environ. Microbiol. 2020, 86, e00441-20. [Google Scholar] [CrossRef]
- Parish, T. Use of a flexible cassette method to generate a double unmarked Mycobacterium tuberculosis tlyA plcABC mutant by gene replacement. Microbiology 2000, 146, 1969–1975. [Google Scholar] [CrossRef]

| Strain | Relative Selectivity (%) | |||
|---|---|---|---|---|
| 9-OHAD | 9-OHBA | AD | BA | |
| MFKD | 78.09 ± 0.21 | 4.21 ± 0.08 | 0.82 ± 0.01 | 0.09 ± 0.01 |
| MFKD_opccR | 77.98 ± 2.06 | 4.76 ± 0.31 | 0.87 ± 0.16 | 0.12 ± 0.03 |
| MFKDΔhsd4A | 4.07 ± 0.12 | 81.47 ± 0.04 | 0.35 ± 0.01 | 0.21 ± 0.01 |
| MFKDΔhsd4A_ opccR | 4,67 ± 0.13 | 81.22 ± 0.37 | 0.63 ± 0.22 | 0.19 ± 0.01 |
| MFKDΔhsd4AΔfadA5 | 0.90 ± 0.08 | 95.13 ± 0.46 | 0.37 ± 0.08 | 0.19 ± 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, S.; Liu, X.; He, B.; Zhai, X.; Yuan, C.; Li, Y.; Lin, W.; Wang, H.; Zhang, B. Efficient Production of 9,22-Dihydroxy-23,24-bisnorchol-4-ene-3-one from Phytosterols by Modifying Multiple Genes in Mycobacterium fortuitum. Int. J. Mol. Sci. 2024, 25, 3579. https://doi.org/10.3390/ijms25073579
Han S, Liu X, He B, Zhai X, Yuan C, Li Y, Lin W, Wang H, Zhang B. Efficient Production of 9,22-Dihydroxy-23,24-bisnorchol-4-ene-3-one from Phytosterols by Modifying Multiple Genes in Mycobacterium fortuitum. International Journal of Molecular Sciences. 2024; 25(7):3579. https://doi.org/10.3390/ijms25073579
Chicago/Turabian StyleHan, Suwan, Xiangcen Liu, Beiru He, Xinghui Zhai, Chenyang Yuan, Yixin Li, Weichao Lin, Haoyu Wang, and Baoguo Zhang. 2024. "Efficient Production of 9,22-Dihydroxy-23,24-bisnorchol-4-ene-3-one from Phytosterols by Modifying Multiple Genes in Mycobacterium fortuitum" International Journal of Molecular Sciences 25, no. 7: 3579. https://doi.org/10.3390/ijms25073579






