Mechanisms of Action of Dorsal Root Ganglion Stimulation
Abstract
:1. Introduction
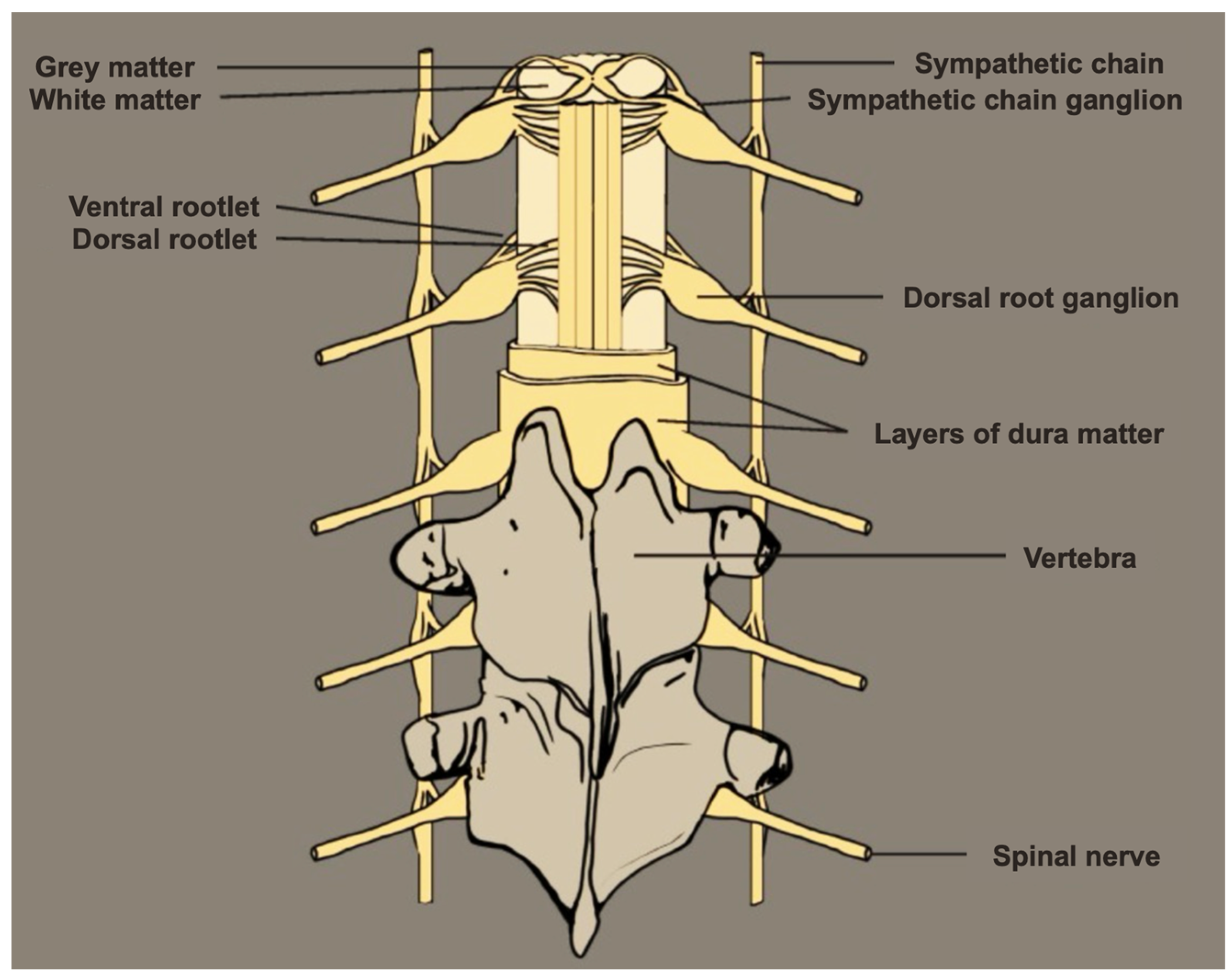
2. DRG Anatomy
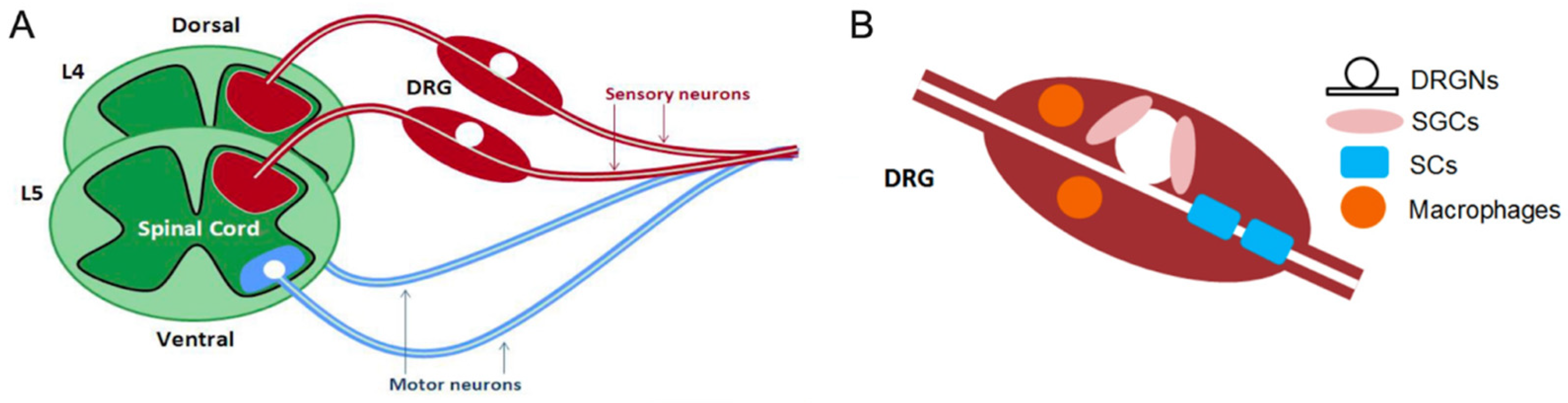
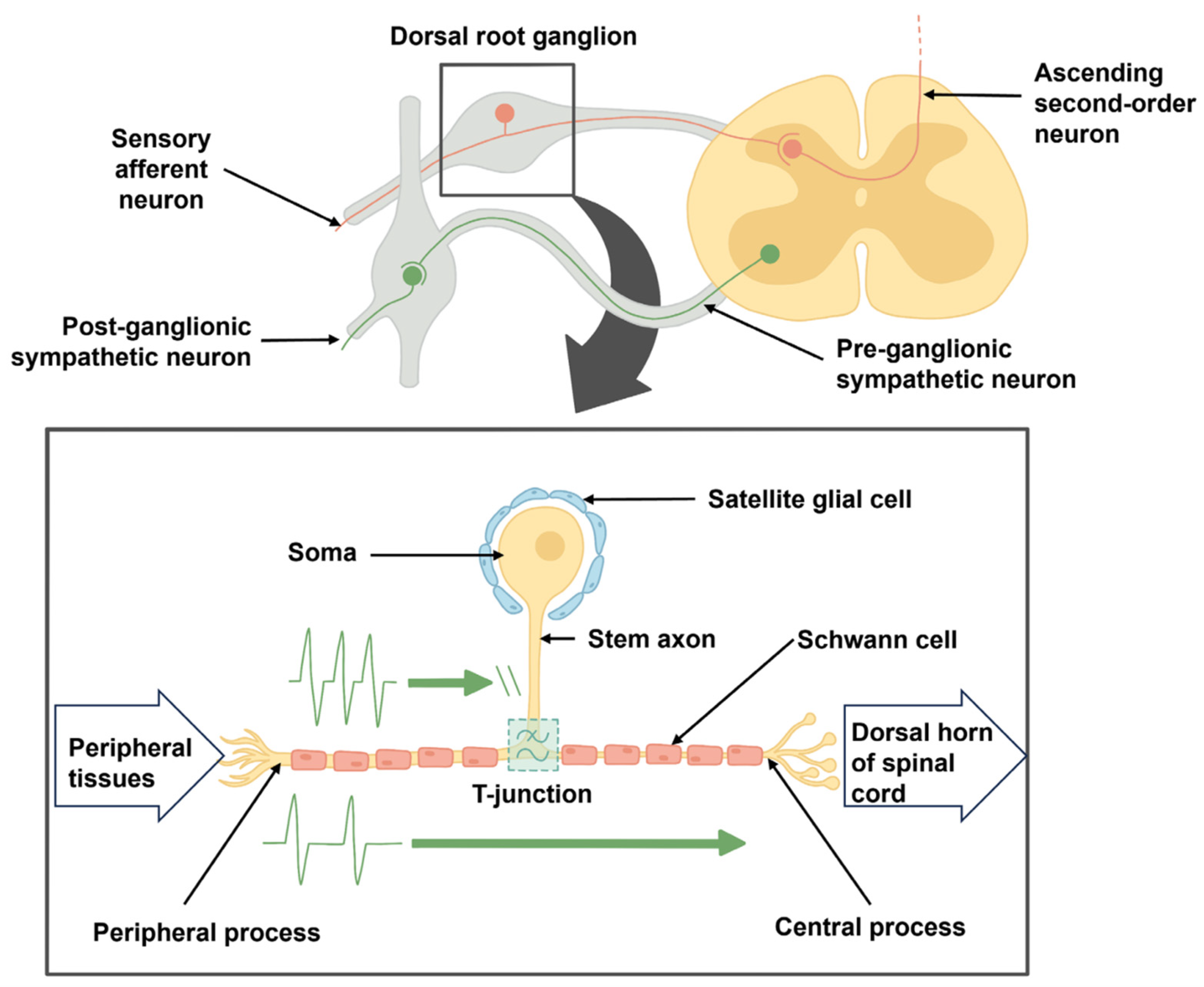
2.1. DRG Neurons Are Pseudo-Unipolar
2.2. DRG Size
2.3. DRG Soma Size
2.4. DRG Position in the Foramina
2.5. DRG Vasculature
2.6. DRG Stimulation (DRG-S) versus Spinal Cord Stimulation (SCS)
2.7. Clinical Applications of DRG-S
3. DRG Mechanisms
3.1. Effects at T-Junction
3.2. Effect on Nociceptive Neurons: Suppressing PSN Hyperexcitability
3.3. Effect on Glial Cells
3.4. Orthodromic and Antidromic Effects
3.5. Effects on GABA
3.6. Supraspinal Effects
3.7. Effects on Ion Channels
3.8. Reversal of Cytokine Release
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AP | Action potentials |
| BOLD | Blood oxygen-level dependent |
| CGRP | Calcitonin gene-related peptide |
| CNS | Central nervous system |
| CRPS | Complex regional pain syndrome |
| CSF | Cerebrospinal fluid |
| DBS | Deep brain stimulation |
| DRG | Dorsal root ganglion |
| DRG-S | Dorsal root ganglion stimulation |
| DRR | Dorsal root reflex |
| FBSS | Failed back surgery syndrome |
| FDA | Food and Drug Administration |
| fMRI | Functional magnetic resonance imaging |
| GABA | Gamma-aminobutyric acid |
| GFAP | Glial fibrillary acidic protein |
| LEP | Laser-evoked potential |
| LTMR | Low-threshold mechanoreceptor |
| NSLBP | Non-surgical low back pain |
| PDPN | Painful diabetic polyneuropathy |
| PNS | Peripheral nervous system |
| PSN | Primary sensory neurons |
| RCT | Randomized controlled trial |
| SCS | Spinal cord stimulation |
| SGC | Satellite glial cells |
| SMPO | Subthreshold membrane potential oscillations |
References
- Goldberg, D.S.; McGee, S. Pain as a Global Public Health Priority. BMC Public Health 2011, 11, 770. [Google Scholar] [CrossRef]
- Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research; National Academies Press: Washington, DC, USA, 2011. [Google Scholar] [CrossRef]
- Gaskin, D.J.; Richard, P. The Economic Costs of Pain in the United States. J. Pain 2012, 13, 715–724. [Google Scholar] [CrossRef]
- Manchikanti, L.; Helm, S.; Fellows, B.; Janata, J.W.; Pampati, V.; Grider, J.S.; Boswell, M.V. Opioid epidemic in the United States. Pain Physician 2012, 15 (Suppl. S3), ES9–ES38. [Google Scholar] [CrossRef]
- Shealy, C.N.; Taslitz, N.; Mortimer, J.T.; Becker, D.P. Electrical inhibition of pain: Experimental evaluation. Anesth. Analg. 1967, 46, 299–305. [Google Scholar] [CrossRef]
- Berger, A.A.; Liu, Y.; Possoit, H.E.; Rogers, A.C.; Moore, W.; Gress, K.; Cornett, E.M.; Kaye, A.D.; Imani, F.; Sadegi, K.; et al. Dorsal Root Ganglion (DRG) and Chronic Pain. Anesthesiol. Pain Med. 2021, 11, e113020. [Google Scholar] [CrossRef] [PubMed]
- Krames, E.S. The Dorsal Root Ganglion in Chronic Pain and as a Target for Neuromodulation: A Review. Neuromodulation 2015, 18, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Vancamp, T.; Levy, R.M.; Peña, I.; Pajuelo, A.G. Relevant Anatomy, Morphology, and Implantation Techniques of the Dorsal Root Ganglia at the Lumbar Levels. Neuromodulation 2017, 20, 690–702. [Google Scholar] [CrossRef] [PubMed]
- Deer, T.R.; Levy, R.M.; Kramer, J.M.; Poree, L.; Amirdelfan, K.; Grigsby, E.; Staats, P.S.; Burton, A.W.; Burgher, A.H.; Obray, J.B.; et al. Dorsal Root Ganglion Stimulation Yielded Higher Treatment Success Rate for Complex Regional Pain Syndrome and Causalgia at 3 and 12 Months: A Randomized Comparative Trial. Pain 2016, 158, 669–681. [Google Scholar] [CrossRef] [PubMed]
- Mol, F.M.U.; Roumen, R.M.H. DRG Spinal Cord Stimulation as Solution for Patients with Severe Pain Due to Anterior Cutaneous Nerve Entrapment Syndrome: A Case Series. Neuromodulation 2018, 21, 317–319. [Google Scholar] [CrossRef] [PubMed]
- Deer, T.R.; Grigsby, E.; Weiner, R.L.; Wilcosky, B.; Kramer, J.M. A prospective study of dorsal root ganglion stimulation for the relief of chronic pain. Neuromodulation 2013, 16, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Huygen, F.; Liem, L.; Nijhuis, H.; Cusack, W.F.; Kramer, J.M. Evaluating Dorsal Root Ganglion Stimulation in a Prospective Dutch Cohort. Neuromodulation 2019, 22, 80–86. [Google Scholar] [CrossRef]
- Esposito, M.; Malayil, R.; Hanes, M.; Deer, T.R. Unique Characteristics of the Dorsal Root Ganglion as a Target for Neuromodulation. Pain Med. 2019, 20, S23–S30. [Google Scholar] [CrossRef]
- Kostelic, J.K.; Haughton, V.M.; Sether, L.A. Lumbar Spinal Nerves in the Neural Foramen: MR Appearance. Radiology 1991, 178, 837–839. [Google Scholar] [CrossRef]
- Grandhi, R.K.; Abd-Elsayed, A. Neuroanatomy. In Textbook of Neuroanesthesia and Neurocritical Care; Springer: Berlin/Heidelberg, Germany, 2019; pp. 3–16. [Google Scholar] [CrossRef]
- Ahimsadasan, N.; Reddy, V.; Khan Suheb, M.Z.; Kumar, A. Neuroanatomy, Dorsal Root Ganglion. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Lemes, J.B.P.; De Campos Lima, T.; Santos, D.O.; Neves, A.F.; De Oliveira, F.S.; Parada, C.A.; Da Cruz Lotufo, C.M. Participation of Satellite Glial Cells of the Dorsal Root Ganglia in Acute Nociception. Neurosci. Lett. 2018, 676, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, S.; Kobayashi, N.; Terashita, T.; Shimokawa, T.; Shigemoto, K.; Mominoki, K.; Wakisaka, H.; Saito, S.; Miyawaki, K.; Saito, K.; et al. Phylogenetic Investigation of Dogiel’s Pericellular Nests and Cajal’s Initial Glomeruli in the Dorsal Root Ganglion. J. Comp. Neurol. 2005, 491, 234–245. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, A.I.; Mar, F.M.; Sousa, M.M. The Intriguing Nature of Dorsal Root Ganglion Neurons: Linking Structure with Polarity and Function. Prog. Neurobiol. 2018, 168, 86–103. [Google Scholar] [CrossRef]
- Martin, S.; Reid, A.J.; Verkhratsky, A.; Magnaghi, V.; Faroni, A. Gene Expression Changes in Dorsal Root Ganglia Following Peripheral Nerve Injury: Roles in Inflammation, Cell Death and Nociception. Neural Regen. Res. 2019, 14, 939–947. [Google Scholar] [CrossRef] [PubMed]
- Middleton, S.J.; Pérez-Sánchez, J.; Dawes, J.D.K. The Structure of Sensory Afferent Compartments in Health and Disease. J. Anat. 2021, 241, 1186–1210. [Google Scholar] [CrossRef] [PubMed]
- Devor, M. Unexplained Peculiarities of the Dorsal Root Ganglion. Pain 1999, 82 (Suppl. S6), S27–S35. [Google Scholar] [CrossRef] [PubMed]
- West, C.A.; Hart, A.; Terenghi, G.; Wiberg, M. Sensory Neurons of the Human Brachial Plexus. Neurosurgery 2012, 70, 1183–1194. [Google Scholar] [CrossRef] [PubMed]
- Tubbs, R.S.; Loukas, M.; Slappey, J.B.; Shoja, M.M.; Oakes, W.J.; Salter, E.G. Clinical Anatomy of the C1 Dorsal Root, Ganglion, and Ramus: A Review and Anatomical Study. Clin. Anat. 2007, 20, 624–627. [Google Scholar] [CrossRef]
- Ebraheim, N.A.; Lu, J. Morphometric evaluation of the sacral dorsal root ganglia. A cadaveric study. Surg. Radiol. Anat. 1998, 20, 105–108. [Google Scholar] [CrossRef]
- Braz, J.; Solorzano, C.; Wang, X.; Basbaum, A.I. Transmitting pain and itch messages: A contemporary view of the spinal cord circuits that generate gate control. Neuron 2014, 82, 522–536. [Google Scholar] [CrossRef] [PubMed]
- Mandell, J.; Czuczman, G.J.; Gaviola, G.C.; Ghazikhanian, V.; Cho, C.H. The Lumbar Neural Foramen and Transforaminal Epidural Steroid Injections: An Anatomic Review with Key Safety Considerations in Planning the Percutaneous Approach. Am. J. Roentgenol. 2017, 209, W26–W35. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.S.; Kim, Y.-D.; Song, B.H.; Deog, Y.; Song, J.H.; Lee, M.H. Position of Dorsal Root Ganglia in the Lumbosacral Region in Patients with Radiculopathy. Korean J. Anesthesiol. 2010, 59, 398. [Google Scholar] [CrossRef] [PubMed]
- Vialle, E.; Vialle, L.R.; Contreras, W.; Jacob, C. Anatomical Study on the Relationship between the Dorsal Root Ganglion and the Intervertebral Disc in the Lumbar Spine. Rev. Bras. Ortop. 2015, 50, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, R.V.; Slipman, C.W.; Bhagia, S.M. Anatomy of the Intervertebral Foramen. Pain Physician 2002, 4, 372–378. [Google Scholar] [CrossRef]
- Jiménez-Andrade, J.M.; Herrera, M.; Ghilardi, J.R.; Vardanyan, M.; Melemedjian, O.K.; Mantyh, P.W. Vascularization of the Dorsal Root Ganglia and Peripheral Nerve of the Mouse: Implications for Chemical-Induced Peripheral Sensory Neuropathies. Mol. Pain 2008, 4, 1744–1810. [Google Scholar] [CrossRef] [PubMed]
- Kiernan, J.A. Vascular permeability in the peripheral autonomic and somatic nervous systems: Controversial aspects and comparisons with the blood-brain barrier. Microsc. Res. Tech. 1996, 35, 122–136. [Google Scholar] [CrossRef]
- Kobayashi, T.; Storrie, B.; Simons, K.; Dotti, C.G. A Functional Barrier to Movement of Lipids in Polarized Neurons. Nature 1992, 359, 647–650. [Google Scholar] [CrossRef]
- Bevan, S.; Yeats, J.C. Protons Activate a Cation Conductance in a Sub-population of Rat Dorsal Root Ganglion Neurones. J. Physiol. 1991, 433, 145–161. [Google Scholar] [CrossRef]
- Devor, M.; Wall, P.D. Cross-Excitation in Dorsal Root Ganglia of Nerve-Injured and Intact Rats. J. Neurophysiol. 1990, 64, 1733–1746. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Imran, M.A. Leland Stanford Junior University and Spinal Modulation LLC. Method and System for Stimulating a Dorsal Root Ganglion. U.S. Patent 7,580,753, 25 August 2009. [Google Scholar]
- Koetsier, E.; Franken, G.; Debets, J.; Heijmans, L.; Van Kuijk, S.M.J.; Linderoth, B.; Joosten, E.A.; Maino, P. Mechanism of Dorsal Root Ganglion Stimulation for Pain Relief in Painful Diabetic Polyneuropathy Is Not Dependent on GABA Release in the Dorsal Horn of the Spinal Cord. CNS Neurosci. Ther. 2019, 26, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Franken, G.; Douven, P.; Debets, J.; Joosten, E.A. Conventional Dorsal Root Ganglion Stimulation in an Experimental Model of Painful Diabetic Peripheral Neuropathy: A Quantitative Immunocytochemical Analysis of Intracellular Γ-Aminobutyric Acid in Dorsal Root Ganglion Neurons. Neuromodul. Technol. Neural Interface 2021, 24, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Holsheimer, J.; Den Boer, J.A.; Struijk, J.J.; Rozeboom, A.R. MR assessment of the normal position of the spinal cord in the spinal canal. Am. J. Neuroradiol. 1994, 15, 951–959. [Google Scholar] [PubMed]
- Figley, C.R.; Stroman, P.W. Investigation of human cervical and upper thoracic spinal cord motion: Implications for imaging spinal cord structure and function. Magn. Reson. Med. 2007, 58, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Kramer, J.; Liem, L.; Russo, M.; Smet, I.; Van Buyten, J.P.; Huygen, F. Lack of body positional effects on paresthesias when stimulating the dorsal root ganglion (DRG) in the treatment of chronic pain. Neuromodulation 2015, 18, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Lam, C.K.; Rosenow, J.M. Patient perspectives on the efficacy and ergonomics of rechargeable spinal cord stimulators. Neuromodulation 2010, 13, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.A.; Jensen, M.P.; Romano, J.M. Do beliefs, coping, and catastrophizing independently predict functioning in patients with chronic pain? Pain 2000, 85, 115–125. [Google Scholar] [CrossRef]
- Piedade, G.S.; Gillner, S.; McPhillips, P.S.; Vesper, J.; Slotty, P.J. Frequency dependency of therapeutic efficacy in dorsal root ganglion stimulation for neuropathic pain. Acta Neurochir. 2022, 164, 1193–1199. [Google Scholar] [CrossRef]
- Chapman, K.B.; Yousef, T.A.; Foster, A.; D Stanton-Hicks, M.; van Helmond, N. Mechanisms for the Clinical Utility of Low-Frequency Stimulation in Neuromodulation of the Dorsal Root Ganglion. Neuromodulation 2021, 24, 738–745. [Google Scholar] [CrossRef]
- Chapman, K.B.; Yousef, T.A.; Vissers, K.C.; van Helmond, N.; D Stanton-Hicks, M. Very Low Frequencies Maintain Pain Relief from Dorsal Root Ganglion Stimulation: An Evaluation of Dorsal Root Ganglion Neurostimulation Frequency Tapering. Neuromodulation 2021, 24, 746–752. [Google Scholar] [CrossRef] [PubMed]
- Piedade, G.S.; Gillner, S.; McPhillips, P.S.; Vesper, J.; Slotty, P.J. Effect of low-frequency dorsal root ganglion stimulation in the treatment of chronic pain. Acta Neurochir. 2023, 165, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Chapman, K.B.; Spiegel, M.A.; van Helmond, N.; Patel, K.V.; Yang, A.; Yousef, T.A.; Mandelberg, N.; Deer, T.; Mogilner, A.Y. Dorsal Root Ganglion Stimulation as a Salvage Therapy Following Failed Spinal Cord Stimulation. Neuromodulation 2022, 25, 1024–1032. [Google Scholar] [CrossRef]
- Deer, T.R.; Pope, J.E. Dorsal root ganglion stimulation approval by the Food and Drug Administration: Advice on evolving the process. Expert Rev. Neurother. 2016, 16, 1123–1125. [Google Scholar] [CrossRef] [PubMed]
- Ghorayeb, J.H.; Chitneni, A.; Rupp, A.; Parkash, A.; Abd-Elsayed, A. Dorsal Root Ganglion Stimulation for the Treatment of Chronic Pelvic Pain: A Systematic Review. Pain Pract. 2023, 23, 838–846. [Google Scholar] [CrossRef]
- D’Souza, R.S.; Langford, B.; Dombovy-Johnson, M.; Abd-Elsayed, A. Neuromodulation Interventions for the Treatment of Painful Diabetic Neuropathy: A Systematic Review. Curr. Pain Headache Rep. 2022, 26, 365–377. [Google Scholar] [CrossRef]
- Elsayed, A.A.; Moghim, R.; Reffat, N. Dorsal Root Ganglion Stimulation (DRG-s) for Potential Resolution of Restless Leg Syndrome Symptoms and Increased Cost Savings for Patients: A Case Study. Pain Pract. 2024; Epub ahead of print. [Google Scholar] [CrossRef]
- Abd-Elsayed, A.; Pope, J.E.; Mundey, D.A.; Slavin, K.V.; Falowski, S.; Chitneni, A.; Popielarski, S.R.; John, J.; Grodofsky, S.; Vanetesse, T.; et al. Diagnosis, Treatment, and Management of Painful Scar: A Narrative Review. J. Pain Res. 2022, 15, 925–937. [Google Scholar] [CrossRef]
- Weiner, R.L.; Yeung, A.; Montes Garcia, C.; Tyler Perryman, L.; Speck, B. Treatment of FBSS low back pain with a novel percutaneous DRG wireless stimulator: Pilot and feasibility study. Pain Med. 2016, 17, 1911–1916. [Google Scholar] [CrossRef]
- Huygen, F.; Liem, L.; Cusack, W.; Kramer, J. Stimulation of the L2-L3 dorsal root ganglia induces effective pain relief in the low back. Pain Pract. 2018, 18, 205–213. [Google Scholar] [CrossRef]
- Krames, E.S. The Role of the Dorsal Root Ganglion in the Development of Neuropathic Pain. Pain Med. 2014, 15, 1669–1685. [Google Scholar] [CrossRef]
- Spencer, P.S.; Raine, C.S.; Wiśniewski, H.M. Axon Diameter and Myelin Thickness—Unusual Relationships in Dorsal Root Ganglia. Anat. Rec. 1973, 176, 225–243. [Google Scholar] [CrossRef] [PubMed]
- Sørenstua, M.; Leonardsen, A.L.; Chin, K.J. Dorsal Root Ganglion: A Key to Understanding the Therapeutic Effects of the Erector Spinae Plane (ESP) and Other Intertransverse Process Blocks? Reg. Anesthesia Pain Med. 2023, 49, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Gemes, G.; Koopmeiners, A.S.; Rigaud, M.; Lirk, P.; Sapunar, D.; Bangaru, M.L.Y.; Vilceanu, D.; Garrison, S.R.; Ljubković, M.; Mueller, S.M.; et al. Failure of Action Potential Propagation in Sensory Neurons: Mechanisms and Loss of Afferent Filtering in C-Type Units after Painful Nerve Injury. J. Physiol. 2013, 591, 1111–1131. [Google Scholar] [CrossRef]
- Amir, R.; Devor, M. Electrical Excitability of the SOMA of Sensory Neurons Is Required for Spike Invasion of the SOMA, but Not for Through-Conduction. Biophys. J. 2003, 84, 2181–2191. [Google Scholar] [CrossRef]
- Sundt, D.; Gamper, N.; Jaffe, D.B. Spike Propagation through the Dorsal Root Ganglia in an Unmyelinated Sensory Neuron: A Modeling Study. J. Neurophysiol. 2015, 114, 3140–3153. [Google Scholar] [CrossRef] [PubMed]
- Corder, G.; Castro, D.C.; Bruchas, M.R.; Scherrer, G. Endogenous and Exogenous Opioids in Pain. Annu. Rev. Neurosci. 2018, 41, 453–473. [Google Scholar] [CrossRef]
- Chapman, K.B.; Groenen, P.S.; Vissers, K.C.; van Helmond, N.; Stanton-Hicks, M.D. The Pathways and Processes Underlying Spinal Transmission of Low Back Pain: Observations From Dorsal Root Ganglion Stimulation Treatment. Neuromodulation 2021, 24, 610–621. [Google Scholar] [CrossRef]
- D’Souza, R.S.; Kubrova, E.; Her, Y.F.; Barman, R.; Smith, B.J.; Alvarez, G.M.; West, T.; Abd-Elsayed, A. Dorsal Root Ganglion Stimulation for Lower Extremity Neuropathic Pain Syndromes: An Evidence-Based Literature Review. Adv. Ther. 2022, 39, 4440–4473. [Google Scholar] [CrossRef]
- Graham, R.; Sankarasubramanian, V.; Lempka, S.F. Dorsal Root Ganglion Stimulation for Chronic Pain: Hypothesized Mechanisms of Action. J. Pain 2022, 23, 196–211. [Google Scholar] [CrossRef]
- Koopmeiners, A.S.; Mueller, S.M.; Kramer, J.; Hogan, Q.H. Effect of Electrical Field Stimulation on Dorsal Root Ganglion Neuronal Function. Neuromodul. Technol. Neural Interface 2013, 16, 304–311. [Google Scholar] [CrossRef]
- Suh, Y.S.; Chung, K.; Coggeshall, R.E. A Study of Axonal Diameters and Areas in Lumbosacral Roots and Nerves in the Rat. J. Comp. Neurol. 1984, 222, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Chiu, S.Y. Computer Model for Action Potential Propagation through Branch Point in Myelinated Nerves. J. Neurophysiol. 2001, 85, 197–210. [Google Scholar] [CrossRef]
- Cummins, T.R.; Dib-Hajj, S.D.; Black, J.A.; Akopian, A.N.; Wood, J.N.; Waxman, S.G. A Novel Persistent Tetrodotoxin-Resistant Sodium Current in SNS-Null and Wild-Type Small Primary Sensory Neurons. J. Neurosci. 1999, 19, RC43. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Hu, S.-J.; Greenquist, K.W.; Zhang, J.; LaMotte, R.H. Mechanical and Thermal Hyperalgesia and Ectopic Neuronal Discharge after Chronic Compression of Dorsal Root Ganglia. J. Neurophysiol. 1999, 82, 3347–3358. [Google Scholar] [CrossRef] [PubMed]
- Amir, R.; Michaelis, M.; Devor, M. Burst Discharge in Primary Sensory Neurons: Triggered by Subthreshold Oscillations, Maintained by Depolarizing Afterpotentials. J. Neurosci. 2002, 22, 1187–1198. [Google Scholar] [CrossRef] [PubMed]
- Amir, R.; Michaelis, M.; Devor, M. Membrane Potential Oscillations in Dorsal Root Ganglion Neurons: Role in Normal Electrogenesis and Neuropathic Pain. J. Neurosci. 1999, 19, 8589–8596. [Google Scholar] [CrossRef] [PubMed]
- Kovalsky, Y.; Amir, R.; Devor, M. Simulation in Sensory Neurons Reveals a Key Role for Delayed NA+ Current in Subthreshold Oscillations and Ectopic Discharge: Implications for Neuropathic Pain. J. Neurophysiol. 2009, 102, 1430–1442. [Google Scholar] [CrossRef] [PubMed]
- Amir, R.; Argoff, C.E.; Bennett, G.J.; Cummins, T.R.; Durieux, M.E.; Gerner, P.; Gold, M.S.; Porreca, F.; Strichartz, G.R. The Role of Sodium Channels in Chronic Inflammatory and Neuropathic Pain. J. Pain 2006, 7, S1–S29. [Google Scholar] [CrossRef]
- Amir, R.; Kocsis, J.D.; Devor, M. Multiple Interacting Sites of Ectopic Spike Electrogenesis in Primary Sensory Neurons. J. Neurosci. 2005, 25, 2576–2585. [Google Scholar] [CrossRef]
- Ma, C.; LaMotte, R.H. Multiple Sites for Generation of Ectopic Spontaneous Activity in Neurons of the Chronically Compressed Dorsal Root Ganglion. J. Neurosci. 2007, 27, 14059–14068. [Google Scholar] [CrossRef]
- Amir, R.; Liu, C.; Kocsis, J.D.; Devor, M. Oscillatory Mechanism in Primary Sensory Neurones. Brain 2002, 125, 421–435. [Google Scholar] [CrossRef]
- Campbell, J.N.; Raja, S.N.; Meyer, R.A.; Mackinnon, S.E. Myelinated Afferents Signal the Hyperalgesia Associated with Nerve Injury. Pain 1988, 32, 89–94. [Google Scholar] [CrossRef]
- Bethea, J.R.; Fischer, R. Role of Peripheral Immune Cells for Development and Recovery of Chronic Pain. Front. Immunol. 2021, 12, 641588. [Google Scholar] [CrossRef]
- Znaor, L.; Lovrić, S.; Hogan, Q.; Sapunar, D. Association of neural inflammation with hyperalgesia following spinal nerve ligation. Croat. Med. J. 2007, 48, 35–42. [Google Scholar] [PubMed]
- Hanani, M.; Spray, D.C. Emerging Importance of Satellite Glia in Nervous System Function and Dysfunction. Nat. Rev. Neurosci. 2020, 21, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Utzschneider, D.A.; Kocsis, J.D.; Devor, M. Mutual Excitation among Dorsal Root Ganglion Neurons in the Rat. Neurosci. Lett. 1992, 146, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Mohr, K.M.; Pallesen, L.T.; Richner, M.; Vægter, C.B. Discrepancy in the Usage of GFAP as a Marker of Satellite Glial Cell Reactivity. Biomedicines 2021, 9, 1022. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.-Y.; Belzer, V.; Hanani, M. Gap Junctions in Dorsal Root Ganglia: Possible Contribution to Visceral Pain. Eur. J. Pain 2010, 14, 49.e1–49.e9. [Google Scholar] [CrossRef] [PubMed]
- Dubový, P.; Klusáková, I.; Svíženská, I.H.; Brázda, V. Satellite Glial Cells Express IL-6 and Corresponding Signal-Transducing Receptors in the Dorsal Root Ganglia of Rat Neuropathic Pain Model. Neuron Glia Biol. 2010, 6, 73–83. [Google Scholar] [CrossRef] [PubMed]
- De Souza, G.E.; Talbot, J.; Da Cruz Lotufo, C.M.; Cunha, F.Q.; Cunha, T.M.; Ferreira, S.H. Fractalkine Mediates Inflammatory Pain through Activation of Satellite Glial Cells. Proc. Natl. Acad. Sci. USA 2013, 110, 11193–11198. [Google Scholar] [CrossRef]
- Mitterreiter, J.G.; Ouwendijk, W.J.D.; Van Velzen, M.; Van Nierop, G.P.; Osterhaus, A.D.M.E.; Verjans, G.M.G.M. Satellite Glial Cells in Human Trigeminal Ganglia Have a Broad Expression of Functional Toll-like Receptors. Eur. J. Immunol. 2017, 47, 1181–1187. [Google Scholar] [CrossRef] [PubMed]
- Estivill-Torrús, G.; Martínez-Padilla, A.B.; Sánchez-Salido, L.; Evercooren, A.B.-V.; Garcia-Diaz, B. The Dorsal Root Ganglion as a Target for Neurorestoration in Neuropathic Pain. Neural Regen. Res. 2023, 19, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Sorkin, L.S.; Eddinger, K.A.; Woller, S.A.; Yaksh, T.L. Origins of Antidromic Activity in Sensory Afferent Fibers and Neurogenic Inflammation. Semin. Immunopathol. 2018, 40, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, F.J.; Kavookjian, A.M.; Light, A.R. Synaptic interactions between GABA- immunoreactive profiles and the terminals of functionally defined myelinated nociceptors in the monkey and cat spinal cord. J. Neurosci. 1992, 12, 2901–2917. [Google Scholar] [CrossRef] [PubMed]
- Todd, A.J. Plasticity of inhibition in the spinal cord. In Pain Control. Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2015; Volume 227, pp. 171–190. [Google Scholar] [CrossRef]
- Toennies, J.F. Reflex discharge from the spinal cord over the dorsal roots. J. Neurophysiol. 1938, 1, 378–390. [Google Scholar] [CrossRef]
- Lobanov, O.V.; Peng, Y.B. Differential Contribution of Electrically Evoked Dorsal Root Reflexes to Peripheral Vasodilatation and Plasma Extravasation. J. Neuroinflamm. 2011, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Willis, W.D., Jr. Dorsal root potentials and dorsal root reflexes: A double-edged sword. Exp. Brain Res. 1999, 124, 395–421. [Google Scholar] [CrossRef]
- Chapman, K.B.; Sayed, D.; Lamer, T.J.; Hunter, C.; Weisbein, J.; Patel, K.V.; Dickerson, D.; Hagedorn, J.M.; Lee, D.W.; Amirdelfan, K.; et al. Best Practices for Dorsal Root Ganglion Stimulation for Chronic Pain: Guidelines from the American Society of Pain and Neuroscience. J. Pain Res. 2023, 16, 839–879. [Google Scholar] [CrossRef]
- Du, X.; Hao, H.; Yang, Y.; Huang, S.C.; Wang, C.; Gigout, S.; Ramli, R.; Li, X.; Jaworska, E.; Edwards, I.; et al. Local GABAergic Signaling within Sensory Ganglia Controls Peripheral Nociceptive Transmission. J. Clin. Investig. 2017, 127, 1741–1756. [Google Scholar] [CrossRef]
- Guan, Y. Spinal Cord Stimulation: Neurophysiological and Neurochemical Mechanisms of Action. Curr. Pain Headache Rep. 2012, 16, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Stiller, C.; Cui, J.; O’Connor, W.T.; Brodin, E.; Meyerson, B.A.; Linderoth, B. Release of Gamma-Aminobutyric Acid in the Dorsal Horn and Suppression of Tactile Allodynia by Spinal Cord Stimulation in Mononeuropathic Rats. Neurosurgery 1996, 39, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Janssen, S.; Truin, M.; Van Kleef, M.; Joosten, E.A. Differential GABAergic Disinhibition during the Development of Painful Peripheral Neuropathy. Neuroscience 2011, 184, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Janssen, S.; Gerard, S.; Raijmakers, M.; Truin, M.; Van Kleef, M.; Joosten, E.A. Decreased Intracellular GABA Levels Contribute to Spinal Cord Stimulation-Induced Analgesia in Rats Suffering from Painful Peripheral Neuropathy: The Role of KCC2 and GABAA Receptor-Mediated Inhibition. Neurochem. Int. 2012, 60, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Smits, H.; Van Kleef, M.; Holsheimer, J.; Joosten, E.A. Experimental Spinal Cord Stimulation and Neuropathic Pain: Mechanism of Action, Technical Aspects, and Effectiveness. Pain Pract. 2012, 13, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.A.; Kohno, T.; Karchewski, L.A.; Scholz, J.; Baba, H.; Woolf, C.J. Partial Peripheral Nerve Injury Promotes a Selective Loss of GABAergic Inhibition in the Superficial Dorsal Horn of the Spinal Cord. J. Neurosci. 2002, 22, 6724–6731. [Google Scholar] [CrossRef]
- Patel, S.; Naeem, S.; Kesingland, A.; Froestl, W.; Capogna, M.; Urbán, L.; Fox, A. The Effects of GABAB Agonists and Gabapentin on Mechanical Hyperalgesia in Models of Neuropathic and Inflammatory Pain in the Rat. Pain 2001, 90, 217–226. [Google Scholar] [CrossRef]
- Somers, D.L.; Clemente, F.R. Dorsal Horn Synaptosomal Content of Aspartate, Glutamate, Glycine and GABA Are Differentially Altered Following Chronic Constriction Injury to the Rat Sciatic Nerve. Neurosci. Lett. 2002, 323, 171–174. [Google Scholar] [CrossRef]
- Drew, G.M.; Siddall, P.J.; Duggan, A.W. Mechanical Allodynia Following Contusion Injury of the Rat Spinal Cord Is Associated with Loss of GABAergic Inhibition in the Dorsal Horn. Pain 2004, 109, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Pawela, C.P.; Kramer, J.M.; Hogan, Q.H. Dorsal Root Ganglion Stimulation Attenuates the BOLD Signal Response to Noxious Sensory Input in Specific Brain Regions: Insights into a Possible Mechanism for Analgesia. NeuroImage 2017, 147, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Mehta, V.; Bouchareb, Y.; Ramaswamy, S.; Ahmad, A.; Wodehouse, T.; Haroon, A. Metabolic Imaging of Pain Matrix Using 18F Fluoro-Deoxyglucose Positron Emission Tomography/Computed Tomography for Patients Undergoing L2 Dorsal Root Ganglion Stimulation for Low Back Pain. Neuromodulation 2020, 23, 222–233. [Google Scholar] [CrossRef]
- Morgalla, M.; De Barros Filho, M.F.; Chander, B.S.; Soekadar, S.R.; Tatagiba, M.; Lepski, G. Neurophysiological Effects of Dorsal Root Ganglion Stimulation (DRGS) in Pain Processing at the Cortical Level. Neuromodul. Technol. Neural Interface 2019, 22, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Goudman, L.; De Groote, S.; Linderoth, B.; De Smedt, A.; Eldabe, S.; Duarte, R.; Moens, M. Exploration of the Supraspinal Hypotheses about Spinal Cord Stimulation and Dorsal Root Ganglion Stimulation: A Systematic Review. J. Clin. Med. 2021, 10, 2766. [Google Scholar] [CrossRef]
- Verrills, P.; Mitchell, B.; Vivian, D.; Cusack, W.F.; Kramer, J.M. Dorsal Root Ganglion Stimulation Is Paresthesia-Independent: A Retrospective Study. Neuromodulation 2019, 22, 937–942. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.S.; Kayani, K.; Whyte-Oshodi, D.; Whyte-Oshodi, A.; Nachiappan, N.; Gnanarajah, S.; Mohammed, R. Voltage Gated Sodium Channels as Therapeutic Targets for Chronic Pain. J. Pain Res. 2019, 12, 2709–2722. [Google Scholar] [CrossRef]
- Smith, P.A. K+ Channels in Primary Afferents and Their Role in Nerve Injury-Induced Pain. Front. Cell. Neurosci. 2020, 14, 566418. [Google Scholar] [CrossRef]
- Theile, J.W.; Cummins, T.R. Recent Developments Regarding Voltage-Gated Sodium Channel Blockers for the Treatment of Inherited and Acquired Neuropathic Pain Syndromes. Front. Pharmacol. 2011, 2, 54. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chung, K.; Chung, J.M. Ectopic Discharges and Adrenergic Sensitivity of Sensory Neurons after Spinal Nerve Injury. Brain Res. 1999, 849, 244–247. [Google Scholar] [CrossRef]
- Mendell, L.M. The Path to Discovery of Windup and Central Sensitization. Front. Pain Res. 2022, 3, 833104. [Google Scholar] [CrossRef]
- Kajander, K.C.; Wakisaka, S.; Bennett, G.J. Spontaneous Discharge Originates in the Dorsal Root Ganglion at the Onset of a Painful Peripheral Neuropathy in the Rat. Neurosci. Lett. 1992, 138, 225–228. [Google Scholar] [CrossRef]
- Kent, A.R.; Min, X.; Hogan, Q.H.; Kramer, J.M. Mechanisms of Dorsal Root Ganglion Stimulation in Pain Suppression: A Computational Modeling Analysis. Neuromodulation 2018, 21, 234–246. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, C.C.; Savasta, M.; Goff, L.K.; Vitek, J.L. Uncovering the Mechanism(s) of Action of Deep Brain Stimulation: Activation, Inhibition, or Both. Clin. Neurophysiol. 2004, 115, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd-Elsayed, A.; Vardhan, S.; Aggarwal, A.; Vardhan, M.; Diwan, S.A. Mechanisms of Action of Dorsal Root Ganglion Stimulation. Int. J. Mol. Sci. 2024, 25, 3591. https://doi.org/10.3390/ijms25073591
Abd-Elsayed A, Vardhan S, Aggarwal A, Vardhan M, Diwan SA. Mechanisms of Action of Dorsal Root Ganglion Stimulation. International Journal of Molecular Sciences. 2024; 25(7):3591. https://doi.org/10.3390/ijms25073591
Chicago/Turabian StyleAbd-Elsayed, Alaa, Swarnima Vardhan, Abhinav Aggarwal, Madhurima Vardhan, and Sudhir A. Diwan. 2024. "Mechanisms of Action of Dorsal Root Ganglion Stimulation" International Journal of Molecular Sciences 25, no. 7: 3591. https://doi.org/10.3390/ijms25073591




