CC Chemokine Family Members’ Modulation as a Novel Approach for Treating Central Nervous System and Peripheral Nervous System Injury—A Review of Clinical and Experimental Findings
Abstract
:1. Introduction
| Chemokine | Chemokine Receptors | Central Nervous System Injury | Peripheral Nervous System Injury |
|---|---|---|---|
| CCL1 | CCR8 | ↑ IRBI [51] ↑ ICH [52] | ↑ PSNL [53] ↑ CCI [54] |
| CCL2 | CCR1, CCR2, CCR4 | ↑ TBI [55,56,57,58] ↑ CIBI [59,60] ↑ AIBI [61] ↑ ICH [62] ↑ SCI [63] ↑ CPSP [64] | ↑ CCI [48,54,65,66,67] |
| CCL3 | CCR1, CCR5 | ↑TBI [58,68,69] ↑ AIBI [61] | ↑ CCI [66,70,71,72] ↑ PSNL [73] |
| CCL4 | CCR1, CCR5 | ↑ TBI [68] | ↑ CCI [65,66,72] |
| CCL5 | CCR1, CCR3, CCR5 | ↑ TBI [74] ↑ IRBI [51] ↑ ICH [75] ↑ MCAO [76] | ↑ CCI [65,77] |
| CCL6 | CCR1 | ND | ↑ CCI [65,66,78] |
| CCL7 | CCR1, CCR2, CCR3, CCR5 | ↑ TBI [55] | ↑ CCI [65,66,78,79,80] ↑ SNL [81] |
| CCL8 | CCR1, CCR2, CCR3, CCR5 | ND | ↑ CCI [65] |
| CCL9/10 | CCR1 | ↑ TBI [68] ↑ IRBI [51] | ↑ CCI [65,66,82] |
| CCL11 | CCR3, CCR5 | ↑ TBI [58] ↑ NHI [83] | ↑ CCI [80] |
| CCL12 | CCR2 | ↑ TBI [55] ↑ ICH [84] | ↑ CCI [85,86] |
| CCL13 | CCR1, CCR2, CCR3, CCR5 | NOT PRESENT IN MICE/RATS | |
| CCL14 | CCR1 | NOT PRESENT IN MICE/RATS | |
| CCL15 | CCR1, CCR3 | NOT PRESENT IN MICE/RATS | |
| CCL16 | CCR1 | NOT PRESENT IN MICE/RATS | |
| CCL17 | CCR4 | ↑ TBI [87,88] ↑ IRBI [51] ↑ ICH [89,90,91] | ↑ CCI [92] |
| CCL18 | CCR8 | NOT PRESENT IN MICE/RATS | |
| CCL19 | CCR7 | ↑ TBI [56] | ND |
| CCL20 | CCR6 | ↑ TBI [93,94,95,96] ↑ ICH [97] ↑ MCAO [98] ↑ SCI [99] | ND |
| CCL21 | CCR7 | ND | ↑ CCI [100] ↑ SNL [101] |
| CCL22 | CCR4 | ↑ TBI [56,87,102] ↑ CPSP [103] ↑ NHI [104] | ↑ CCI [92] |
| CCL23 | CCR1 | NOT PRESENT IN MICE/RATS | |
| CCL24 | CCR3 | ND | – CCI [65,80] |
| CCL25 | CCR9, CCR10 | ND | ND |
| CCL26 | CCR3, CCR10 | ND | – CCI [80] |
| CCL27 | CCR10 | ND | ND |
| CCL28 | CCR3, CCR10 | ND | – CCI [80] |
2. CCR1—Ligands and Pharmacological Modulation
2.1. CCR1—Endogenous Ligands

2.2. CCR1—Pharmacological Modulation
| Receptor(s) | Antagonist | Beneficial Effect of Antagonist after CNS Injury | Beneficial Effect of Antagonist after PNS Injury |
|---|---|---|---|
| SINGLE ANTAGONISTS OF CC-FAMILY RECEPTORS | |||
| CCR1 | J113863 | ND | CCI [65,66] |
| BX513 | ND | SSNL [164] | |
| CCR2 | RS504393 | TBI [222,223]; LDH [197] | CCI [48,224]; LAMNT [225] |
| CCX872 | TBI [226] | ND | |
| INCB3344 | CIBI [227] | CCI [228] | |
| AZ889 | ND | CCI [229] | |
| CCR3 | SB328437 | ND | CCI [65,80] |
| SB 297006 | ND | ND | |
| CCR4 | C021 | ICH [89,90] | CCI [92,230] |
| AZD-2098 | SAH [231] | ND | |
| CCR5 | Maraviroc | TBI [232,233]; FCS [234]; CIBI [235]; ICH [236] | CCI [71,85,237] |
| AZD-5672 | ND | CCI [72] | |
| TAK-220 | FCI [238] | CCI [72] | |
| CCR6 | PF-07054894 | ND | ND |
| CCR7 | - | ND | ND |
| CCR8 | AZ084 | ND | ND |
| R243 | ND | ND | |
| CCR9 | Vercirnon | ND | ND |
| MLN3126 | ND | ND | |
| CCR10 | BI-6901 | ND | ND |
| DOUBLE ANTAGONISTS OF CC-FAMILY RECEPTORS | |||
| CCR1/CCR3 | UCB 35625 | ND | CCI [65] |
| CCR2/CCR5 | Cenicriviroc | TBI [203] | CCI [70,85] |
| BMS-813160 | ND | ND | |
| PF-04634817 | ND | ND | |
3. CCR2—Ligands and Pharmacological Modulation
3.1. CCR2—Endogenous Ligands
3.2. CCR2—Pharmacological Modulation
4. CCR3—Ligands and Pharmacological Modulation
4.1. CCR3—Endogenous Ligands
4.2. CCR3—Pharmacological Modulation
5. CCR4—Ligands and Pharmacological Modulation
5.1. CCR4—Endogenous Ligands
5.2. CCR4—Pharmacological Modulation
6. CCR5—Ligands and Pharmacological Modulation
6.1. CCR5—Endogenous Ligands
6.2. CCR5—Pharmacological Modulation
7. CCR6—Ligands and Pharmacological Modulation
7.1. CCR6—Endogenous Ligands
7.2. CCR6—Pharmacological Modulation
8. CCR7—Ligands and Pharmacological Modulation
8.1. CCR7—Endogenous Ligands
8.2. CCR7—Pharmacological Modulation
9. CCR8—Ligands and Pharmacological Modulation
9.1. CCR8—Endogenous Ligands
9.2. CCR8—Pharmacological Modulation
10. CCR9—Ligands and Pharmacological Modulation
10.1. CCR9—Endogenous Ligands
10.2. CCR9—Pharmacological Modulation
11. CCR10—Ligands and Pharmacological Modulation
11.1. CCR10—Endogenous Ligands
11.2. CCR10—Pharmacological Modulation
12. Analgesic Potential of Dual Chemokine CC Receptor Antagonists
13. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- James, S.L.; Bannick, M.S.; Montjoy-Venning, W.C.; Lucchesi, L.R.; Dandona, L.; Dandona, R.; Hawley, C.; Hay, S.I.; Jakovljevic, M.; Khalil, I.; et al. Global, Regional, and National Burden of Traumatic Brain Injury and Spinal Cord Injury, 1990-2016: A Systematic Analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 56–87. [Google Scholar] [CrossRef] [PubMed]
- Dewan, M.C.; Rattani, A.; Gupta, S.; Baticulon, R.E.; Hung, Y.C.; Punchak, M.; Agrawal, A.; Adeleye, A.O.; Shrime, M.G.; Rubiano, A.M.; et al. Estimating the Global Incidence of Traumatic Brain Injury. J. Neurosurg. 2019, 130, 1080–1097. [Google Scholar] [CrossRef] [PubMed]
- NIH (National Institutes of Health). Traumatic Brain Injury Information. Available online: https://www.nih.gov/about-nih/what-we-do/nih-turning-discovery-into-health/healthy-mind/traumatic-brain-injury-tbi (accessed on 16 November 2023).
- Bouras, M.; Asehnoune, K.; Roquilly, A. Immune Modulation after Traumatic Brain Injury. Front. Med. 2022, 9, 995044. [Google Scholar] [CrossRef] [PubMed]
- Mira, R.G.; Lira, M.; Cerpa, W. Traumatic Brain Injury: Mechanisms of Glial Response. Front. Physiol. 2021, 12, 740939. [Google Scholar] [CrossRef] [PubMed]
- Johnson, V.E.; Stewart, W.; Smith, D.H. Widespread Tau and Amyloid-Beta Pathology Many Years after a Single Traumatic Brain Injury in Humans. Brain Pathol. 2012, 22, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Gentleman, S.M.; Nash, M.J.; Sweeting, C.J.; Graham, D.I.; Roberts, G.W. β-Amyloid Precursor Protein (ΒAPP) as a Marker for Axonal Injury after Head Injury. Neurosci. Lett. 1993, 160, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Graham, N.S.N.; Sharp, D.J. Understanding Neurodegeneration after Traumatic Brain Injury: From Mechanisms to Clinical Trials in Dementia. J. Neurol. Neurosurg. Psychiatry 2019, 90, 1221–1233. [Google Scholar] [CrossRef] [PubMed]
- Scheid, R.; Walther, K.; Guthke, T.; Preul, C.; Von Cramon, D.Y. Cognitive Sequelae of Diffuse Axonal Injury. Arch. Neurol. 2006, 63, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Sariaslan, A.; Sharp, D.J.; D’Onofrio, B.M.; Larsson, H.; Fazel, S. Long-Term Outcomes Associated with Traumatic Brain Injury in Childhood and Adolescence: A Nationwide Swedish Cohort Study of a Wide Range of Medical and Social Outcomes. PLoS Med. 2016, 13, 15–19. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Li, X.; Zhang, S.; Zhao, J.; Zhu, X.; Tian, G. Head Injury as a Risk Factor for Dementia and Alzheimer’s Disease: A Systematic Review and Meta-Analysis of 32 Observational Studies. PLoS ONE 2017, 12, e0169650. [Google Scholar] [CrossRef]
- Nordström, A.; Nordström, P. Traumatic Brain Injury and the Risk of Dementia Diagnosis: A Nationwide Cohort Study. PLoS Med. 2018, 15, e1002496. [Google Scholar] [CrossRef] [PubMed]
- Fann, J.R.; Ribe, A.R.; Pedersen, H.S.; Fenger-Grøn, M.; Christensen, J.; Benros, M.E.; Vestergaard, M. Long-Term Risk of Dementia among People with Traumatic Brain Injury in Denmark: A Population-Based Observational Cohort Study. Lancet Psychiatry 2018, 5, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Irvine, K.A.; David Clark, J. Chronic Pain after Traumatic Brain Injury: Pathophysiology and Pain Mechanisms. Pain Med. 2018, 19, 1315–1333. [Google Scholar] [CrossRef] [PubMed]
- Smith-Seemiller, L.; Fow, N.R.; Kant, R.; Franzen, M.D. Presence of Post-Concussion Syndrome Symptoms in Patients with Chronic Pain vs Mild Traumatic Brain Injury. Brain Inj. 2003, 17, 199–206. [Google Scholar] [CrossRef]
- Dobscha, S.K.; Clark, M.E.; Morasco, B.J.; Freeman, M.; Campbell, R.; Helfand, M. Systematic Review of the Literature on Pain in Patients with Polytrauma Including Traumatic Brain Injury. Pain Med. 2009, 10, 1200–1217. [Google Scholar] [CrossRef] [PubMed]
- Lucas, S. Posttraumatic Headache: Clinical Characterization and Management. Curr. Pain Headache Rep. 2015, 19, 48. [Google Scholar] [CrossRef] [PubMed]
- Bose, P.; Hou, J.; Nelson, R.; Nissim, N.; Parmer, R.; Keener, J.; Wacnik, P.W.; Thompson, F.J. Effects of Acute Intrathecal Baclofen in an Animal Model of Tbi-Induced Spasticity, Cognitive, and Balance Disabilities. J. Neurotrauma 2013, 30, 1177–1191. [Google Scholar] [CrossRef] [PubMed]
- Ofek, H.; Defrin, R. The Characteristics of Chronic Central Pain after Traumatic Brain Injury. Pain 2007, 131, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Wang, H.L.; Pu, H.J.; Shi, Y.J.; Zhang, J.; Zhang, W.T.; Wang, G.H.; Hu, X.M.; Leak, R.K.; Chen, J.; et al. Ethyl Pyruvate Protects against Blood-Brain Barrier Damage and Improves Long-Term Neurological Outcomes in a Rat Model of Traumatic Brain Injury. CNS Neurosci. Ther. 2015, 21, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Costantini, T.W.; D’Mello, R.; Eliceiri, B.P.; Coimbra, R.; Bansal, V. Altering Leukocyte Recruitment Following Traumatic Brain Injury with Ghrelin Therapy. J. Trauma Acute Care Surg. 2014, 77, 709–715. [Google Scholar] [CrossRef]
- Noble, J.; Munro, C.A.; Prasad, V.S.S.V.; Midha, R. Analysis of Upper and Lower Extremity Peripheral Nerve Injuries in a Population of Patients with Multiple Injuries. J. Trauma-Inj. Infect. Crit. Care 1998, 45, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Campbell, W.W. Evaluation and Management of Peripheral Nerve Injury. Clin. Neurophysiol. 2008, 119, 1951–1965. [Google Scholar] [CrossRef] [PubMed]
- Kretschmer, T.; Antoniadis, G.; Braun, V.; Rath, S.A.; Richter, H.P. Evaluation of Iatrogenic Lesions in 722 Surgically Treated Cases of Peripheral Nerve Trauma. J. Neurosurg. 2001, 94, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Althagafi, A.; Nadi, M. Acute Nerve Injury; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Selecki, B.; Ring, I.; Simpson, D.; Vanderfield, G.; Sewell, M. Trauma to the Central and Peripheral Nervous Systems. Part II: A Statistical Profile of Surgical Treatment New South Wales 1977. Aust. N. Z. J. Surg. 1982, 52, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Stone, L.; Keenan, M.A. Peripheral Nerve Injuries in the Adult with Traumatic Brain Injury. Clin. Orthop. Relat. Res. 1988, 233, 136–144. [Google Scholar] [CrossRef]
- Wordliczek, J.; Dobrogowski, J. Leczenie Bólu; PZWL: Warszawa, Poland, 2007. [Google Scholar]
- Latremoliere, A.; Woolf, C.J. Central Sensitization: A Generator of Pain Hypersensitivity by Central Neural Plasticity. J. Pain 2009, 10, 895–926. [Google Scholar] [CrossRef] [PubMed]
- Finnerup, N.B.; Kuner, R.; Jensen, T.S. Neuropathic Pain: From Mechanisms to Treatment. Physiol. Rev. 2021, 101, 259–301. [Google Scholar] [CrossRef] [PubMed]
- Muley, M.M.; Krustev, E.; Reid, A.R.; McDougall, J.J. Prophylactic Inhibition of Neutrophil Elastase Prevents the Development of Chronic Neuropathic Pain in Osteoarthritic Mice. J. Neuroinflamm. 2017, 14, 168. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.R.; Lopes, A.H.; Talbot, J.; Cecilio, N.T.; Rossato, M.F.; Silva, R.L.; Souza, G.R.; Silva, C.R.; Lucas, G.; Fonseca, B.A.; et al. Neuroimmune–Glia Interactions in the Sensory Ganglia Account for the Development of Acute Herpetic Neuralgia. J. Neurosci. 2017, 37, 6408–6422. [Google Scholar] [CrossRef]
- Lim, H.; Lee, H.; Noh, K.; Lee, S.J. IKK/NF-B-Dependent Satellite Glia Activation Induces Spinal Cord Microglia Activation and Neuropathic Pain after Nerve Injury. Pain 2017, 158, 1666–1677. [Google Scholar] [CrossRef]
- Purwata, T.E. High TNF-Alpha Plasma Levels and Macrophages INOS and TNF-Alpha Expression as Risk Factors for Painful Diabetic Neuropathy. J. Pain Res. 2011, 4, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Kambrun, C.; Roca-Lapirot, O.; Salio, C.; Landry, M.; Moqrich, A.; Le Feuvre, Y. TAFA4 Reverses Mechanical Allodynia through Activation of GABAergic Transmission and Microglial Process Retraction. Cell Rep. 2018, 22, 2886–2897. [Google Scholar] [CrossRef] [PubMed]
- White, F.A.; Jung, H.; Miller, R.J. Chemokines and the Pathophysiology of Neuropathic Pain. Proc. Natl. Acad. Sci. USA 2007, 104, 20151–20158. [Google Scholar] [CrossRef]
- Kwiatkowski, K.; Mika, J. The Importance of Chemokines in Neuropathic Pain Development and Opioid Analgesic Potency. Pharmacol. Rep. 2018, 70, 821–830. [Google Scholar] [CrossRef]
- Savarin-Vuaillat, C.; Ransohoff, R.M. Chemokines and Chemokine Receptors in Neurological Disease: Raise, Retain, or Reduce? Neurotherapeutics 2007, 4, 590–601. [Google Scholar] [CrossRef]
- Hughes, C.E.; Nibbs, R.J.B. A Guide to Chemokines and Their Receptors. FEBS J. 2018, 285, 2944–2971. [Google Scholar] [CrossRef] [PubMed]
- Ciechanowska, A.; Rojewska, E.; Piotrowska, A.; Barut, J.; Pawlik, K.; Ciapała, K.; Kreiner, G.; Mika, J. New Insights into the Analgesic Properties of the XCL1/XCR1 and XCL1/ITGA9 Axes Modulation under Neuropathic Pain Conditions-Evidence from Animal Studies. Front. Immunol. 2022, 13, 1058204. [Google Scholar] [CrossRef] [PubMed]
- Rojewska, E.; Zychowska, M.; Piotrowska, A.; Kreiner, G.; Nalepa, I.; Mika, J. Involvement of Macrophage Inflammatory Protein-1 Family Members in the Development of Diabetic Neuropathy and Their Contribution to Effectiveness of Morphine. Front. Immunol. 2018, 9, 494. [Google Scholar] [CrossRef]
- Gleissner, C.A.; Von Hundelshausen, P.; Ley, K. Platelet Chemokines in Vascular Disease. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1920–1927. [Google Scholar] [CrossRef]
- Steen, A.; Larsen, O.; Thiele, S.; Rosenkilde, M.M. Biased and g Protein-Independent Signaling of Chemokine Receptors. Front. Immunol. 2014, 5, 277. [Google Scholar] [CrossRef]
- Feng, Z.; Sun, R.; Cong, Y.; Liu, Z. Critical Roles of G Protein-Coupled Receptors in Regulating Intestinal Homeostasis and Inflammatory Bowel Disease. Mucosal Immunol. 2022, 15, 819–828. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.K.; Poluri, K.M. Molecular Insights into Kinase Mediated Signaling Pathways of Chemokines and Their Cognate G Protein Coupled Receptors. Front. Biosci. 2020, 25, 1361–1385. [Google Scholar] [CrossRef]
- Henrot, P.; Prevel, R.; Berger, P.; Dupin, I. Chemokines in COPD: From Implication to Therapeutic Use. Int. J. Mol. Sci. 2019, 20, 2785. [Google Scholar] [CrossRef] [PubMed]
- Rizeq, B.; Malki, M.I. The Role of CCL21/CCR7 Chemokine Axis in Breast Cancer Progression. Cancers 2020, 12, 1036. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, K.; Piotrowska, A.; Rojewska, E.; Makuch, W.; Mika, J. The RS504393 Influences the Level of Nociceptive Factors and Enhances Opioid Analgesic Potency in Neuropathic Rats. J. Neuroimmune Pharmacol. 2017, 12, 402–419. [Google Scholar] [CrossRef] [PubMed]
- Cocchi, F.; DeVico, A.L.; Garzino-Demo, A.; Arya, S.K.; Gallo, R.C.; Lusso, P. Identification of RANTES, MIP-1 Alpha, and MIP-1 Beta as the Major HIV-Suppressive Factors Produced by CD8+ T Cells. Science 1995, 270, 1811–1815. [Google Scholar] [CrossRef] [PubMed]
- Menten, P.; Wuyts, A.; Van Damme, J. Macrophage Inflammatory Protein-1. Cytokine Growth Factor Rev. 2002, 13, 455–481. [Google Scholar] [CrossRef] [PubMed]
- Baba, N.; Wang, F.; Iizuka, M.; Shen, Y.; Yamashita, T.; Takaishi, K.; Tsuru, E.; Matsushima, S.; Miyamura, M.; Fujieda, M.; et al. Induction of Regional Chemokine Expression in Response to Human Umbilical Cord Blood Cell Infusion in the Neonatal Mouse Ischemia-Reperfusion Brain Injury Model. PLoS ONE 2019, 14, e0221111. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, M.; Kai, T.; Shimizu, Y.; Yano, Y.; Urabe, Y.; Tasaka, S.; Akagi, M.; Yamaguchi, Y.; Inoue, A. Gadolinium Causes M1 and M2 Microglial Apoptosis after Intracerebral Haemorrhage and Exerts Acute Neuroprotective Effects. J. Pharm. Pharmacol. 2020, 72, 709–718. [Google Scholar] [CrossRef]
- Akimoto, N.; Honda, K.; Uta, D.; Beppu, K.; Ushijima, Y.; Matsuzaki, Y.; Nakashima, S.; Kido, M.A.; Imoto, K.; Takano, Y.; et al. CCL-1 in the Spinal Cord Contributes to Neuropathic Pain Induced by Nerve Injury. Cell Death Dis. 2013, 4, e679. [Google Scholar] [CrossRef]
- Zajaczkowska, R.; Kwiatkowski, K.; Pawlik, K.; Piotrowska, A.; Rojewska, E.; Makuch, W.; Wordliczek, J.; Mika, J. Metamizole Relieves Pain by Influencing Cytokine Levels in Dorsal Root Ganglia in a Rat Model of Neuropathic Pain. Pharmacol. Rep. 2020, 72, 1310–1322. [Google Scholar] [CrossRef] [PubMed]
- Popiolek-Barczyk, K.; Ciechanowska, A.; Ciapała, K.; Pawlik, K.; Oggioni, M.; Mercurio, D.; De Simoni, M.G.; Mika, J. The CCL2/CCL7/CCL12/CCR2 Pathway Is Substantially and Persistently Upregulated in Mice after Traumatic Brain Injury, and CCL2 Modulates the Complement System in Microglia. Mol. Cell. Probes 2020, 54, 101671. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.H.; Lin, W.; Su, Y.C.; Cheng-Yo Hsuan, Y.; Chen, Y.C.; Chang, C.P.; Chou, W.; Lin, K.C. Modulation of Parietal Cytokine and Chemokine Gene Profiles by Mesenchymal Stem Cell as a Basis for Neurotrauma Recovery. J. Formos. Med. Assoc. 2019, 118, 1661–1673. [Google Scholar] [CrossRef] [PubMed]
- Semple, B.D.; Bye, N.; Rancan, M.; Ziebell, J.M.; Morganti-Kossmann, M.C. Role of CCL2 (MCP-1) in Traumatic Brain Injury (TBI): Evidence from Severe TBI Patients and CCL2−/− Mice. J. Cereb. Blood Flow Metab. 2010, 30, 769–782. [Google Scholar] [CrossRef] [PubMed]
- Clausen, F.; Marklund, N.; Hillered, L. Acute Inflammatory Biomarker Responses to Diffuse Traumatic Brain Injury in the Rat Monitored by a Novel Microdialysis Technique. J. Neurotrauma 2019, 36, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Geng, H.; Chen, L.; Tang, J.; Chen, Y.; Wang, L. The Role of CCL2/CCR2 Axis in Cerebral Ischemia-Reperfusion Injury and Treatment: From Animal Experiments to Clinical Trials. Int. J. Mol. Sci. 2022, 23, 3485. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Wang, X.; Ma, C.; Ji, J.; Xu, W.; Shao, Q.; Liao, X.; Li, Y.; Cheng, F.; Wang, Q. Identification of Potential Regulating Effect of Baicalin on NFκB/CCL2/CCR2 Signaling Pathway in Rats with Cerebral Ischemia by Antibody-Based Array and Bioinformatics Analysis. J. Ethnopharmacol. 2022, 284, 114773. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Ma, Q.; Li, Y.; Li, B.; Zhang, L. Inhibition of MicroRNA-210 Suppresses pro-Inflammatory Response and Reduces Acute Brain Injury of Ischemic Stroke in Mice. Exp. Neurol. 2018, 300, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Tsirka, S.E. The CCL2-CCR2 System Affects the Progression and Clearance of Intracerebral Hemorrhage. Glia 2012, 60, 908–918. [Google Scholar] [CrossRef]
- Stammers, A.T.; Liu, J.; Kwon, B.K. Expression of Inflammatory Cytokines Following Acute Spinal Cord Injury in a Rodent Model. J. Neurosci. Res. 2012, 90, 782–790. [Google Scholar] [CrossRef]
- Yang, F.; Jing, J.J.; Fu, S.Y.; Su, X.Z.; Zhong, Y.L.; Chen, D.S.; Wu, X.Z.; Zou, Y.Q. Spinal MCP-1 Contributes to Central Post-Stroke Pain by Inducing Central Sensitization in Rats. Mol. Neurobiol. 2023, 60, 2086–2098. [Google Scholar] [CrossRef] [PubMed]
- Pawlik, K.; Ciapała, K.; Ciechanowska, A.; Kwiatkowski, K.; Mika, J. Pharmacological Evidence of the Important Roles of CCR1 and CCR3 and Their Endogenous Ligands CCL2/7/8 in Hypersensitivity Based on a Murine Model of Neuropathic Pain. Cells 2023, 12, 98. [Google Scholar] [CrossRef] [PubMed]
- Pawlik, K.; Piotrowska, A.; Kwiatkowski, K.; Ciapała, K.; Popiolek-Barczyk, K.; Makuch, W.; Mika, J. The Blockade of CC Chemokine Receptor Type 1 Influences the Level of Nociceptive Factors and Enhances Opioid Analgesic Potency in a Rat Model of Neuropathic Pain. Immunology 2020, 159, 413–428. [Google Scholar] [CrossRef] [PubMed]
- Ciapała, K.; Rojewska, E.; Pawlik, K.; Ciechanowska, A.; Mika, J. Analgesic Effects of Fisetin, Peimine, Astaxanthin, Artemisinin, Bardoxolone Methyl and 740 Y-P and Their Influence on Opioid Analgesia in a Mouse Model of Neuropathic Pain. Int. J. Mol. Sci. 2023, 24, 9000. [Google Scholar] [CrossRef] [PubMed]
- Ciechanowska, A.; Popiolek-Barczyk, K.; Pawlik, K.; Ciapała, K.; Oggioni, M.; Mercurio, D.; De Simoni, M.G.; Mika, J. Changes in Macrophage Inflammatory Protein-1 (MIP-1) Family Members Expression Induced by Traumatic Brain Injury in Mice. Immunobiology 2020, 225, 151911. [Google Scholar] [CrossRef] [PubMed]
- Förstner, P.; Rehman, R.; Anastasiadou, S.; Haffner-Luntzer, M.; Sinske, D.; Ignatius, A.; Roselli, F.; Knöll, B. Neuroinflammation after Traumatic Brain Injury Is Enhanced in Activating Transcription Factor 3 Mutant Mice. J. Neurotrauma 2018, 35, 2317–2329. [Google Scholar] [CrossRef]
- Kwiatkowski, K.; Pawlik, K.; Ciapała, K.; Piotrowska, A.; Makuch, W.; Mika, J. Bidirectional Action of Cenicriviroc, a CCR2/CCR5 Antagonist, Results in Alleviation of Pain-Related Behaviors and Potentiation of Opioid Analgesia in Rats With Peripheral Neuropathy. Front. Immunol. 2020, 11, 615327. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, K.; Piotrowska, A.; Rojewska, E.; Makuch, W.; Jurga, A.; Slusarczyk, J.; Trojan, E.; Basta-Kaim, A.; Mika, J. Beneficial Properties of Maraviroc on Neuropathic Pain Development and Opioid Effectiveness in Rats. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2016, 64, 68–78. [Google Scholar] [CrossRef]
- Ciechanowska, A.; Pawlik, K.; Ciapała, K.; Mika, J. Pharmacological Modulation of the MIP-1 Family and Their Receptors Reduces Neuropathic Pain Symptoms and Influences Morphine Analgesia: Evidence from a Mouse Model. Brain Sci. 2023, 13, 579. [Google Scholar] [CrossRef]
- Kiguchi, N.; Kobayashi, Y.; Maeda, T.; Saika, F.; Kishioka, S. CC-Chemokine MIP-1α in the Spinal Cord Contributes to Nerve Injury-Induced Neuropathic Pain. Neurosci. Lett. 2010, 484, 17–21. [Google Scholar] [CrossRef]
- Sewell, D.L.; Nacewicz, B.; Liu, F.; Macvilay, S.; Erdei, A.; Lambris, J.D.; Sandor, M.; Fabry, Z. Complement C3 and C5 Play Critical Roles in Traumatic Brain Cryoinjury: Blocking Effects on Neutrophil Extravasation by C5a Receptor Antagonist. J. Neuroimmunol. 2004, 155, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Xu, Y.; Guo, P.; Chen, Y.J.; Zhou, J.; Xia, M.; Tan, B.; Liu, X.; Feng, H.; Chen, Y. CCL5/CCR5-Mediated Peripheral Inflammation Exacerbates Blood–brain Barrier Disruption after Intracerebral Hemorrhage in Mice. J. Transl. Med. 2023, 21, 196. [Google Scholar] [CrossRef] [PubMed]
- Offner, H.; Subramanian, S.; Parker, S.M.; Afentoulis, M.E.; Vandenbark, A.A.; Hurn, P.D. Experimental Stroke Induces Massive, Rapid Activation of the Peripheral Immune System. J. Cereb. Blood Flow Metab. 2006, 26, 654–665. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.; Fan, Q.; Zhao, Y.; Cheng, M.-Y.; Liu, H.; Li, J.; Lu, F.-F.; Jia, J.-T.; Cheng, W.; Yan, C.-D. Spinal NF-ΚB and Chemokine Ligand 5 Expression during Spinal Glial Cell Activation in a Neuropathic Pain Model. PLoS ONE 2015, 10, e0115120. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, A.; Ciapała, K.; Pawlik, K.; Kwiatkowski, K.; Rojewska, E.; Mika, J. Comparison of the Effects of Chemokine Receptors CXCR2 and CXCR3 Pharmacological Modulation in Neuropathic Pain Model—In Vivo and In Vitro Study. Int. J. Mol. Sci. 2021, 22, 11074. [Google Scholar] [CrossRef] [PubMed]
- Imai, S.; Narita, M.M.; Ikegami, D.; Yamashita, A.; Shimizu, T.; Narita, M.M.; Niikura, K.; Furuya, M.; Kobayashi, Y.; Miyashita, K.; et al. Epigenetic Transcriptional Activation of Monocyte Chemotactic Protein 3 Contributes to Long-Lasting Neuropathic Pain. Brain 2013, 136, 828–843. [Google Scholar] [CrossRef] [PubMed]
- Pawlik, K.; Ciechanowska, A.; Ciapała, K.; Rojewska, E. Blockade of CC Chemokine Receptor Type 3 Diminishes Pain and Enhances Opioid Analgesic Potency in a Model of Neuropathic Pain. Front. Immunol 2021, 12, 781310. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Deng, G.; Wang, H.; Yang, M.; Yang, R.; Li, X.; Zhang, X.; Yuan, H. Interleukin-1β Pre-Treated Bone Marrow Stromal Cells Alleviate Neuropathic Pain through CCL7-Mediated Inhibition of Microglial Activation in the Spinal Cord. Sci. Rep. 2017, 7, 42260. [Google Scholar] [CrossRef] [PubMed]
- Bogacka, J.; Pawlik, K.; Ciapała, K.; Ciechanowska, A.; Mika, J. CC Chemokine Receptor 4 (CCR4) as a Possible New Target for Therapy. Int. J. Mol. Sci. 2022, 23, 15638. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Baba, N.; Shen, Y.; Yamashita, T.; Tsuru, E.; Tsuda, M.; Maeda, N.; Sagara, Y. CCL11 Promotes Migration and Proliferation of Mouse Neural Progenitor Cells. Stem Cell Res. Ther. 2017, 8, 26. [Google Scholar] [CrossRef]
- Huang, J.; Yang, G.; Xiong, X.; Wang, M.; Yuan, J.; Zhang, Q.; Gong, C.; Qiu, Z.; Meng, Z.; Xu, R.; et al. Age-Related CCL12 Aggravates Intracerebral Hemorrhage-Induced Brain Injury via Recruitment of Macrophages and T Lymphocytes. Aging Dis. 2020, 11, 1103–1115. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, K.; Ciapała, K.; Rojewska, E.; Makuch, W.; Mika, J. Comparison of the Beneficial Effects of RS504393, Maraviroc and Cenicriviroc on Neuropathic Pain-Related Symptoms in Rodents: Behavioral and Biochemical Analyses. Int. Immunopharmacol. 2020, 84, 106540. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, K.; Popiolek-Barczyk, K.; Piotrowska, A.; Rojewska, E.; Ciapała, K.; Makuch, W.; Mika, J. Chemokines CCL2 and CCL7, but Not CCL12, Play a Significant Role in the Development of Pain-Related Behavior and Opioid-Induced Analgesia. Cytokine 2019, 119, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Chio, C.C.; Lin, H.J.; Tian, Y.F.; Chen, Y.C.; Lin, M.T.; Lin, C.H.; Chang, C.P.; Hsu, C.C. Exercise Attenuates Neurological Deficits by Stimulating a Critical HSP70/NF-ΚB/IL-6/Synapsin I Axis in Traumatic Brain Injury Rats. J. Neuroinflamm. 2017, 14, 90. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Hylin, M.J.; Kobori, N.; Hood, K.N.; Moore, A.N.; Dash, P.K. Post-Injury Administration of Galantamine Reduces Traumatic Brain Injury Pathology and Improves Outcome. J. Neurotrauma 2018, 35, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Jin, P.; Sherchan, P.; Liu, S.; Cui, Y.; Huang, L.; Zhang, J.H.; Gong, Y.; Tang, J. Recombinant CCL17-Dependent CCR4 Activation Alleviates Neuroinflammation and Neuronal Apoptosis through the PI3K/AKT/Foxo1 Signaling Pathway after ICH in Mice. J. Neuroinflamm. 2021, 18, 62. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Sherchan, P.; Jin, P.; Huang, L.; Travis, Z.; Zhang, J.H.; Gong, Y.; Tang, J. Recombinant CCL17 Enhances Hematoma Resolution and Activation of CCR4/ERK/Nrf2/CD163 Signaling Pathway After Intracerebral Hemorrhage in Mice. Neurotherapeutics 2020, 17, 1940–1953. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Bian, L.; Du, Y.; Wang, D.; Jiang, R.; Lu, J.; Zhao, X. The Roles of Chemokines Following Intracerebral Hemorrhage in Animal Models and Humans. Front. Mol. Neurosci. 2023, 15, 1091498. [Google Scholar] [CrossRef] [PubMed]
- Bogacka, J.; Popiolek-Barczyk, K.; Pawlik, K.; Ciechanowska, A.; Makuch, W.; Rojewska, E.; Dobrogowski, J.; Przeklasa-Muszynska, A.; Mika, J. CCR4 Antagonist (C021) Influences the Level of Nociceptive Factors and Enhances the Analgesic Potency of Morphine in a Rat Model of Neuropathic Pain. Eur. J. Pharmacol. 2020, 880, 173166. [Google Scholar] [CrossRef]
- Das, M.; Tang, X.; Han, J.Y.; Mayilsamy, K.; Foran, E.; Biswal, M.R.; Tzekov, R.; Mohapatra, S.S.; Mohapatra, S. CCL20-CCR6 Axis Modulated Traumatic Brain Injury-Induced Visual Pathologies. J. Neuroinflamm. 2019, 16, 115. [Google Scholar] [CrossRef]
- Dalgard, C.L.; Cole, J.T.; Kean, W.S.; Lucky, J.J.; Sukumar, G.; McMullen, D.C.; Pollard, H.B.; Watson, W.D. The Cytokine Temporal Profile in Rat Cortex after Controlled Cortical Impact. Front. Mol. Neurosci. 2012, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- Leonardo, C.C.; Musso, J.; Das, M.; Rowe, D.D.; Collier, L.A.; Mohapatra, S.; Pennypacker, K.R. CCL20 Is Associated with Neurodegeneration Following Experimental Traumatic Brain Injury and Promotes Cellular Toxicity In Vitro. Transl. Stroke Res. 2012, 3, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Mayilsamy, K.; Markoutsa, E.; Das, M.; Chopade, P.; Puro, D.; Kumar, A.; Gulick, D.; Willing, A.E.; Mohapatra, S.S.; Mohapatra, S. Treatment with ShCCL20-CCR6 Nanodendriplexes and Human Mesenchymal Stem Cell Therapy Improves Pathology in Mice with Repeated Traumatic Brain Injury. Nanomedicine 2020, 29, 102247. [Google Scholar] [CrossRef] [PubMed]
- Yao, N.; Li, Y.; Han, J.; Wu, S.; Liu, X.; Wang, Q.; Li, Z.; Shi, F.D. Microglia-Derived CCL20 Deteriorates Neurogenesis Following Intraventricular Hemorrhage. Exp. Neurol. 2023, 370, 114561. [Google Scholar] [CrossRef] [PubMed]
- Terao, Y.; Ohta, H.; Oda, A.; Nakagaito, Y.; Kiyota, Y.; Shintani, Y. Macrophage Inflammatory Protein-3alpha Plays a Key Role in the Inflammatory Cascade in Rat Focal Cerebral Ischemia. Neurosci. Res. 2009, 64, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Yang, Z.; Li, X.; Lu, H. C-C Motif Chemokine Ligand 20 Regulates Neuroinflammation Following Spinal Cord Injury via Th17 Cell Recruitment. J. Neuroinflamm. 2016, 13, 162. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, A.; Rojewska, E.; Pawlik, K.; Kreiner, G.; Ciechanowska, A.; Makuch, W.; Zychowska, M.; Mika, J. Pharmacological Blockade of CXCR3 by (±)-NBI-74330 Reduces Neuropathic Pain and Enhances Opioid Effectiveness-Evidence from in Vivo and in Vitro Studies. BBA-Mol. Basis Dis. 2018, 1864, 3418–3437. [Google Scholar] [CrossRef] [PubMed]
- Biber, K.; Tsuda, M.; Tozaki-Saitoh, H.; Tsukamoto, K.; Toyomitsu, E.; Masuda, T.; Boddeke, H.; Inoue, K. Neuronal CCL21 Up-Regulates Microglia P2X4 Expression and Initiates Neuropathic Pain Development. EMBO J. 2011, 30, 1864–1873. [Google Scholar] [CrossRef] [PubMed]
- Madathil, S.K.; Wilfred, B.S.; Urankar, S.E.; Yang, W.; Leung, L.Y.; Gilsdorf, J.S.; Shear, D.A. Early Microglial Activation Following Closed-Head Concussive Injury Is Dominated by Pro-Inflammatory M-1 Type. Front. Neurol. 2018, 9, 964. [Google Scholar] [CrossRef]
- Pu, Z.; Bao, X.; Xia, S.; Shao, P.; Xu, Y. Serpine1 Regulates Peripheral Neutrophil Recruitment and Acts as Potential Target in Ischemic Stroke. J. Inflamm. Res. 2022, 15, 2649–2663. [Google Scholar] [CrossRef]
- Zhang, B.; Ran, Y.; Wu, S.; Zhang, F.; Huang, H.; Zhu, C.; Zhang, S.; Zhang, X. Inhibition of Colony Stimulating Factor 1 Receptor Suppresses Neuroinflammation and Neonatal Hypoxic-Ischemic Brain Injury. Front. Neurol. 2021, 12, 607370. [Google Scholar] [CrossRef]
- Murdoch, C.; Finn, A. Chemokine Receptors and Their Role in Inflammation and Infectious Diseases. Blood 2000, 95, 3032–3043. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, R.; Nagira, M.; Kitaura, M.; Imagawa, N.; Imai, T.; Yoshie, O. Secondary Lymphoid-Tissue Chemokine Is a Functional Ligand for the CC Chemokine Receptor CCR7. J. Biol. Chem. 1998, 273, 7118–7122. [Google Scholar] [CrossRef] [PubMed]
- Kohout, T.A.; Nicholas, S.L.; Perry, S.J.; Reinhart, G.; Junger, S.; Struthers, R.S. Differential Desensitization, Receptor Phosphorylation, Beta-Arrestin Recruitment, and ERK1/2 Activation by the Two Endogenous Ligands for the CC Chemokine Receptor 7. J. Biol. Chem. 2004, 279, 23214–23222. [Google Scholar] [CrossRef] [PubMed]
- Scheu, S.; Ali, S.; Ruland, C.; Arolt, V.; Alferink, J. The C-C Chemokines CCL17 and CCL22 and Their Receptor CCR4 in CNS Autoimmunity. Int. J. Mol. Sci. 2017, 18, 2306. [Google Scholar] [CrossRef] [PubMed]
- N’Diaye, M.; Le Ferrec, E.; Lagadic-Gossmann, D.; Corre, S.; Gilot, D.; Lecureur, V.; Monteiro, P.; Rauch, C.; Galibert, M.D.; Fardel, O. Aryl Hydrocarbon Receptor- and Calcium-Dependent Induction of the Chemokine CCL1 by the Environmental Contaminant Benzo[a]Pyrene. J. Biol. Chem. 2006, 281, 19906–19915. [Google Scholar] [CrossRef]
- Arai, H.; Tsou, C.L.; Charo, I.F. Chemotaxis in a Lymphocyte Cell Line Transfected with C-C Chemokine Receptor 2B: Evidence That Directed Migration Is Mediated by Βγ Dimers Released by Activation of Gαi-Coupled Receptors. Proc. Natl. Acad. Sci. USA 1997, 94, 14495. [Google Scholar] [CrossRef]
- Neptune, E.R.; Bourne, H.R. Receptors Induce Chemotaxis by Releasing the Betagamma Subunit of Gi, Not by Activating Gq or Gs. Proc. Natl. Acad. Sci. USA 1997, 94, 14489–14494. [Google Scholar] [CrossRef]
- Hirsch, E.; Katanaev, V.L.; Garlanda, C.; Azzolino, O.; Pirola, L.; Silengo, L.; Sozzani, S.; Mantovani, A.; Altruda, F.; Wymann, M.P. Central Role for G Protein-Coupled Phosphoinositide 3-Kinase Gamma in Inflammation. Science 2000, 287, 1049–1052. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, E.; Aloisi, F. Chemokines and Glial Cells: A Complex Network in the Central Nervous System. Neurochem. Res. 2004, 29, 1017–1038. [Google Scholar] [CrossRef] [PubMed]
- Huber, A.K.; Giles, D.A.; Segal, B.M.; Irani, D.N. An Emerging Role for Eotaxins in Neurodegenerative Disease. Clin. Immunol. 2018, 189, 29–33. [Google Scholar] [CrossRef]
- Popiolek-Barczyk, K.; Mika, J. Targeting the Microglial Signaling Pathways: New Insights in the Modulation of Neuropathic Pain. Curr. Med. Chem. 2016, 23, 2908–2928. [Google Scholar] [CrossRef]
- Futosi, K.; Fodor, S.; Mócsai, A. Neutrophil Cell Surface Receptors and Their Intracellular Signal Transduction Pathways. Int. Immunopharmacol. 2013, 17, 638–650. [Google Scholar] [CrossRef]
- Henc, I.; Bryl, E. Chemokiny Jako Ważne Mediatory Stanu Zapalnego. Forum Med. Rodz. 2013, 7, 251–262. [Google Scholar]
- Strazza, M.; Mor, A. Consider the Chemokines: A Review of the Interplay between Chemokines and T Cell Subset Function. Discov. Med. 2017, 24, 31–39. [Google Scholar] [PubMed]
- Cowell, R.M.; Xu, H.; Galasso, J.M.; Silverstein, F.S. Hypoxic-Ischemic Injury Induces Macrophage Inflammatory Protein-1alpha Expression in Immature Rat Brain. Stroke 2002, 33, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Zuo, G.; Sherchan, P.; Huang, L.; Ocak, U.; Xu, W.; Travis, Z.D.; Wang, W.; Zhang, J.H.; Tang, J. CCR1 Activation Promotes Neuroinflammation Through CCR1/TPR1/ERK1/2 Signaling Pathway After Intracerebral Hemorrhage in Mice. Neurotherapeutics 2020, 17, 1170–1183. [Google Scholar] [CrossRef] [PubMed]
- Halks-Miller, M.; Schroeder, M.L.; Haroutunian, V.; Moenning, U.; Rossi, M.; Achim, C.; Purohit, D.; Mahmoudi, M.; Horuk, R. CCR1 Is an Early and Specific Marker of Alzheimer’s Disease. Ann. Neurol. 2003, 54, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Sunnemark, D.; Eltayeb, S.; Wallström, E.; Appelsved, L.; Malmberg, Å.; Lassmann, H.; Ericsson-Dahlstrand, A.; Piehl, F.; Olsson, T. Differential Expression of the Chemokine Receptors CX3CR1 and CCR1 by Microglia and Macrophages in Myelin-Oligodendrocyte-Glycoprotein-Induced Experimental Autoimmune Encephalomyelitis. Brain Pathol. 2003, 13, 617–629. [Google Scholar] [CrossRef]
- Shi, C.; Jin, J.; Xu, H.; Ma, J.; Li, T.; Xie, Y.; Li, Z. CCR1 Enhances SUMOylation of DGCR8 by Up-Regulating ERK Phosphorylation to Promote Spinal Nerve Ligation-Induced Neuropathic Pain. Gene Ther. 2021, 29, 379–389. [Google Scholar] [CrossRef]
- Lloyd, E.; Somera-Molina, K.; Van Eldik, L.J.; Watterson, D.M.; Wainwright, M.S. Suppression of Acute Proinflammatory Cytokine and Chemokine Upregulation by Post-Injury Administration of a Novel Small Molecule Improves Long-Term Neurologic Outcome in a Mouse Model of Traumatic Brain Injury. J. Neuroinflamm. 2008, 5, 959–974. [Google Scholar] [CrossRef] [PubMed]
- Israelsson, C.; Bengtsson, H.; Kylberg, A.; Kullander, K.; Lewén, A.; Hillered, L.; Ebendal, T. Distinct Cellular Patterns of Upregulated Chemokine Expression Supporting a Prominent Inflammatory Role in Traumatic Brain Injury. J. Neurotrauma 2008, 25, 959–974. [Google Scholar] [CrossRef] [PubMed]
- Thacker, M.A.; Clark, A.K.; Bishop, T.; Grist, J.; Yip, P.K.; Moon, L.D.F.; Thompson, S.W.N.; Marchand, F.; McMahon, S.B. CCL2 Is a Key Mediator of Microglia Activation in Neuropathic Pain States. Eur. J. Pain 2009, 13, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.J.; Shin, H.Y.; Cui, Y.; Kim, H.; Le Thi, A.H.; Choi, J.Y.; Kim, E.Y.; Hwang, D.H.; Kim, B.G. CCL2 Mediates Neuron–Macrophage Interactions to Drive Proregenerative Macrophage Activation Following Preconditioning Injury. J. Neurosci. 2015, 35, 15934–15947. [Google Scholar] [CrossRef] [PubMed]
- Kiguchi, N.; Kobayashi, Y.; Saika, F.; Kishioka, S. Epigenetic Upregulation of CCL2 and CCL3 via Histone Modifications in Infiltrating Macrophages after Peripheral Nerve Injury. Cytokine 2013, 64, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, S.; Kotani, T.; Kuwabara, H.; Suzuka, T.; Kiboshi, T.; Fukui, K.; Ishida, T.; Fujiki, Y.; Shiba, H.; Hata, K.; et al. CCL2 Produced by CD68+/CD163+ Macrophages as a Promising Clinical Biomarker of Microscopic Polyangiitis-Interstitial Lung Disease. Rheumatology 2021, 60, 4643–4653. [Google Scholar] [CrossRef] [PubMed]
- Tecchio, C.; Cassatella, M.A. Neutrophil-Derived Chemokines on the Road to Immunity. Semin. Immunol. 2016, 28, 119. [Google Scholar] [CrossRef] [PubMed]
- Errede, M.; Annese, T.; Petrosino, V.; Longo, G.; Girolamo, F.; de Trizio, I.; d’Amati, A.; Uccelli, A.; Kerlero de Rosbo, N.; Virgintino, D. Microglia-Derived CCL2 Has a Prime Role in Neocortex Neuroinflammation. Fluids Barriers CNS 2022, 19, 68. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.J.; Zhang, L.; Samad, O.A.; Suter, M.R.; Yasuhiko, K.; Xu, Z.-Z.Z.; Park, J.-Y.Y.; Lind, A.-L.L.; Ma, Q.; Ji, R.-R.R. JNK-Induced MCP-1 Production in Spinal Cord Astrocytes Contributes to Central Sensitization and Neuropathic Pain. J. Neurosci. 2009, 29, 4096–4108. [Google Scholar] [CrossRef]
- Ajuebor, M.N.; Flower, R.J.; Hannon, R.; Christie, M.; Bowers, K.; Verity, A.; Perretti, M. Endogenous Monocyte Chemoattractant Protein-1 Recruits Monocytes in the Zymosan Peritonitis Model. J. Leukoc. Biol. 1998, 63, 108–116. [Google Scholar] [CrossRef]
- Rotterman, T.M.; Haley-Johnson, Z.; Pottorf, T.S.; Chopra, T.; Chang, E.; Zhang, S.; McCallum, W.M.; Fisher, S.; Franklin, H.; Alvarez, M.; et al. Modulation of Central Synapse Remodeling after Remote Peripheral Injuries by the CCL2-CCR2 Axis and Microglia. Cell Rep. 2024, 43, 113776. [Google Scholar] [CrossRef] [PubMed]
- Di Battista, A.P.; Rhind, S.G.; Hutchison, M.G.; Hassan, S.; Shiu, M.Y.; Inaba, K.; Topolovec-vranic, J.; Neto, A.C.; Rizoli, S.B.; Baker, A.J. Inflammatory Cytokine and Chemokine Profiles Are Associated with Patient Outcome and the Hyperadrenergic State Following Acute Brain Injury. J. Neuroinflamm. 2016, 13, 40. [Google Scholar] [CrossRef] [PubMed]
- Zuurman, M.W.; Heeroma, J.; Brouwer, N.; Boddeke, H.W.G.M.; Biber, K. LPS-Induced Expression of a Novel Chemokine Receptor (L-CCR) in Mouse Glial Cells in Vitro and in Vivo. Glia 2003, 41, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Xie, W.; Zhang, J.; Strong, J.A.; Zhang, J.M. Sympathectomy Decreases Pain Behaviors and Nerve Regeneration by Downregulating Monocyte Chemokine CCL2 in Dorsal Root Ganglia in the Rat Tibial Nerve Crush Model. Pain 2022, 163, E106–E120. [Google Scholar] [CrossRef] [PubMed]
- Semple, B.D.; Frugier, T.; Morganti-Kossmann, M.C. CCL2 Modulates Cytokine Production in Cultured Mouse Astrocytes. J. Neuroinflamm. 2010, 7, 67. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.R.; Iftinca, M.; Gomes, F.I.F.; Segal, J.P.; Smith, O.M.A.; Bannerman, C.A.; Mendes, A.S.; Defaye, M.; Robinson, M.E.C.; Gilron, I.; et al. Skin-Resident Dendritic Cells Mediate Postoperative Pain via CCR4 on Sensory Neurons. Proc. Natl. Acad. Sci. USA 2022, 119, e2118238119. [Google Scholar] [CrossRef]
- Piotrowska, A.; Rojewska, E.; Pawlik, K.; Kreiner, G.; Ciechanowska, A.; Makuch, W.; Nalepa, I.; Mika, J. Pharmacological Blockade of Spinal CXCL3/CXCR2 Signaling by NVP CXCR2 20, a Selective CXCR2 Antagonist, Reduces Neuropathic Pain Following Peripheral Nerve Injury. Front. Immunol. 2019, 10, 2198. [Google Scholar] [CrossRef] [PubMed]
- Zychowska, M.; Rojewska, E.; Pilat, D.; Mika, J. The Role of Some Chemokines from the CXC Subfamily in a Mouse Model of Diabetic Neuropathy. J. Diabetes Res. 2015, 2015, 750182. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.C.; Cao, D.L.; Zhang, X.; Zhang, Z.J.; He, L.N.; Li, C.H.; Zhang, W.W.; Wu, X.B.; Berta, T.; Ji, R.R.; et al. CXCL13 Drives Spinal Astrocyte Activation and Neuropathic Pain via CXCR5. J. Clin. Investig. 2016, 126, 745–761. [Google Scholar] [CrossRef]
- Rojewska, E.; Ciapała, K.; Mika, J. Kynurenic Acid and Zaprinast Diminished CXCL17-Evoked Pain-Related Behaviour and Enhanced Morphine Analgesia in a Mouse Neuropathic Pain Model. Pharmacol. Rep. 2019, 71, 139–148. [Google Scholar] [CrossRef]
- Zychowska, M.; Rojewska, E.; Piotrowska, A.; Kreiner, G.; Mika, J. Microglial Inhibition Influences XCL1/XCR1 Expression and Causes Analgesic Effects in a Mouse Model of Diabetic Neuropathy. Anesthesiology 2016, 125, 573–589. [Google Scholar] [CrossRef] [PubMed]
- Staniland, A.A.; Clark, A.K.; Wodarski, R.; Sasso, O.; Maione, F.; D’Acquisto, F.; Malcangio, M. Reduced Inflammatory and Neuropathic Pain and Decreased Spinal Microglial Response in Fractalkine Receptor (CX3CR1) Knockout Mice. J. Neurochem. 2010, 114, 1143–1157. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.; Li, J.; Wang, J.; Zhou, L.; Li, J.; Liang, J.; Yin, G.; Li, X.; Cheng, Y.; Zhang, K. ITGA9 Inhibits Proliferation and Migration of Dermal Microvascular Endothelial Cells in Psoriasis. Clin. Cosmet. Investig. Dermatol. 2022, 15, 2795–2806. [Google Scholar] [CrossRef] [PubMed]
- Reyes, N.; Benedetti, I.; Rebollo, J.; Correa, O.; Geliebter, J. Atypical Chemokine Receptor CCRL2 Is Overexpressed in Prostate Cancer Cells. J. Biomed. Res. 2019, 33, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Salvi, V.; Sozio, F.; Sozzani, S.; Prete, A. Del Role of Atypical Chemokine Receptors in Microglial Activation and Polarization. Front. Aging Neurosci. 2017, 9, 148. [Google Scholar] [CrossRef] [PubMed]
- Pruenster, M.; Mudde, L.; Bombosi, P.; Dimitrova, S.; Zsak, M.; Middleton, J.; Richmond, A.; Graham, G.J.; Segerer, S.; Nibbs, R.J.B.; et al. The Duffy Antigen Receptor for Chemokines Transports Chemokines and Supports Their Promigratory Activity. Nat. Immunol. 2009, 10, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Hansell, C.A.; Maclellan, L.M.; Oldham, R.S.; Doonan, J.; Chapple, K.J.; Anderson, E.J.R.; Linington, C.; Mcinnes, I.B.; Nibbs, R.J.B.; Goodyear, C.S. The Atypical Chemokine Receptor ACKR2 Suppresses Th17 Responses to Protein Autoantigens. Immunol. Cell Biol. 2015, 93, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Banisadr, G.; Podojil, J.R.; Miller, S.D.; Miller, R.J. Pattern of CXCR7 Gene Expression in Mouse Brain Under Normal and Inflammatory Conditions. J. Neuroimmune Pharmacol. 2016, 11, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Friess, M.C.; Kritikos, I.; Schineis, P.; Medina-Sanchez, J.D.; Gkountidi, A.O.; Vallone, A.; Sigmund, E.C.; Schwitter, C.; Vranova, M.; Matti, C.; et al. Mechanosensitive ACKR4 Scavenges CCR7 Chemokines to Facilitate T Cell De-Adhesion and Passive Transport by Flow in Inflamed Afferent Lymphatics. Cell Rep. 2022, 38, 110334. [Google Scholar] [CrossRef]
- Zychowska, M.; Rojewska, E.; Piotrowska, A.; Kreiner, G.; Nalepa, I.; Mika, J. Spinal CCL1/CCR8 Signaling Interplay as a Potential Therapeutic Target–Evidence from a Mouse Diabetic Neuropathy Model. Int. Immunopharmacol. 2017, 52, 261–271. [Google Scholar] [CrossRef]
- Zhu, X.; Liu, J.; Chen, O.; Xue, J.; Huang, S.; Zhu, W.; Wang, Y. Neuroprotective and Anti-Inflammatory Effects of Isoliquiritigenin in Kainic Acid-Induced Epileptic Rats via the TLR4/MYD88 Signaling Pathway. Inflammopharmacology 2019, 27, 1143–1153. [Google Scholar] [CrossRef]
- Zhu, X.; Wei, D.; Chen, O.; Zhang, Z.; Xue, J.; Huang, S.; Zhu, W.; Wang, Y. Upregulation of CCL3/MIP-1alpha Regulated by MAPKs and NF-KappaB Mediates Microglial Inflammatory Response in LPS-Induced Brain Injury. Acta Neurobiol. Exp. 2016, 76, 304–317. [Google Scholar] [CrossRef] [PubMed]
- Stefini, R.; Catenacci, E.; Piva, S.; Sozzani, S.; Valerio, A.; Bergomi, R.; Cenzato, M.; Mortini, P.; Latronico, N. Chemokine Detection in the Cerebral Tissue of Patients with Posttraumatic Brain Contusions. J. Neurosurg. 2008, 108, 958–962. [Google Scholar] [CrossRef] [PubMed]
- Karpus, W.J.; Kennedy, K.J. MIP-1alpha and MCP-1 Differentially Regulate Acute and Relapsing Autoimmune Encephalomyelitis as Well as Th1/Th2 Lymphocyte Differentiation. J. Leukoc. Biol. 1997, 62, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Karpus, W.J.; Lukacs, N.W.; McRae, B.L.; Strieter, R.M.; Kunkel, S.L.; Miller, S.D. An Important Role for the Chemokine Macrophage Inflammatory Protein-1 Alpha in the Pathogenesis of the T Cell-Mediated Autoimmune Disease, Experimental Autoimmune Encephalomyelitis. J. Immunol. 1995, 155, 5003–5010. [Google Scholar] [CrossRef]
- Kennedy, K.J.; Strieter, R.M.; Kunkel, S.L.; Lukacs, N.W.; Karpus, W.J. Acute and Relapsing Experimental Autoimmune Encephalomyelitis Are Regulated by Differential Expression of the CC Chemokines Macrophage Inflammatory Protein-1α and Monocyte Chemotactic Protein-1. J. Neuroimmunol. 1998, 92, 98–108. [Google Scholar] [CrossRef] [PubMed]
- De Haas, A.H.; Van Weering, H.R.J.; De Jong, E.K.; Boddeke, H.W.G.M.; Biber, K.P.H. Neuronal Chemokines: Versatile Messengers in Central Nervous System Cell Interaction. Mol. Neurobiol. 2007, 36, 137–151. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, U.K. Microglia as a Source and Target of Cytokines. Glia 2002, 40, 140–155. [Google Scholar] [CrossRef] [PubMed]
- Wallace, C.A.; Moir, G.; Malone, D.F.G.; Duncan, L.; Devarajan, G.; Crane, I.J. Regulation of T-Lymphocyte CCL3 and CCL4 Production by Retinal Pigment Epithelial Cells. Investig. Ophthalmol. Vis. Sci. 2013, 54, 722–730. [Google Scholar] [CrossRef]
- Blidberg, K.; Palmberg, L.; Dahlén, B.; Lantz, A.S.; Larsson, K. Chemokine Release by Neutrophils in Chronic Obstructive Pulmonary Disease. Innate Immun. 2012, 18, 503–510. [Google Scholar] [CrossRef]
- Matsushita, K.; Tozaki-Saitoh, H.; Kojima, C.; Masuda, T.; Tsuda, M.; Inoue, K.; Hoka, S. Chemokine (C-C Motif) Receptor 5 Is an Important Pathological Regulator in the Development and Maintenance of Neuropathic Pain. Anesthesiology 2014, 120, 1491–1503. [Google Scholar] [CrossRef] [PubMed]
- Marciniak, E.; Faivre, E.; Dutar, P.; Alves Pires, C.; Demeyer, D.; Caillierez, R.; Laloux, C.; Buée, L.; Blum, D.; Humez, S. The Chemokine MIP-1α/CCL3 Impairs Mouse Hippocampal Synaptic Transmission, Plasticity and Memory. Sci. Rep. 2015, 5, 15862. [Google Scholar] [CrossRef] [PubMed]
- Meucci, O.; Fatatis, A.; Simen, A.A.; Bushell, T.J.; Gray, P.W.; Miller, R.J. Chemokines Regulate Hippocampal Neuronal Signaling and Gp120 Neurotoxicity. Proc. Natl. Acad. Sci. USA 1998, 95, 14500–14505. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Cui, G.; Zhu, M.; Kang, X.; Guo, H. Neuroinflammation in Alzheimer’s Disease: Chemokines Produced by Astrocytes and Chemokine Receptors. Int. J. Clin. Exp. Pathol. 2014, 7, 8342–8355. [Google Scholar] [PubMed]
- Xia, M.Q.; Qin, S.X.; Wu, L.J.; Mackay, C.R.; Hyman, B.T. Immunohistochemical Study of the Beta-Chemokine Receptors CCR3 and CCR5 and Their Ligands in Normal and Alzheimer’s Disease Brains. Am. J. Pathol. 1998, 153, 31–37. [Google Scholar] [CrossRef] [PubMed]
- MacNair, L.; Xiao, S.; Miletic, D.; Ghani, M.; Julien, J.-P.; Keith, J.; Zinman, L.; Rogaeva, E.; Robertson, J. MTHFSD and DDX58 Are Novel RNA-Binding Proteins Abnormally Regulated in Amyotrophic Lateral Sclerosis. Brain 2016, 139, 86–100. [Google Scholar] [CrossRef] [PubMed]
- Bennouna, S.; Bliss, S.K.; Curiel, T.J.; Denkers, E.Y. Cross-Talk in the Innate Immune System: Neutrophils Instruct Recruitment and Activation of Dendritic Cells during Microbial Infection. J. Immunol. 2003, 171, 6052–6058. [Google Scholar] [CrossRef] [PubMed]
- Charmoy, M.; Brunner-Agten, S.; Aebischer, D.; Auderset, F.; Launois, P.; Milon, G.; Proudfoot, A.E.I.; Tacchini-Cottier, F. Neutrophil-Derived CCL3 Is Essential for the Rapid Recruitment of Dendritic Cells to the Site of Leishmania Major Inoculation in Resistant Mice. PLoS Pathog. 2010, 6, e1000755. [Google Scholar] [CrossRef] [PubMed]
- Ghirnikar, R.S.; Lee, Y.L.; He, T.R.; Eng, L.F. Chemokine Expression in Rat Stab Wound Brain Injury. J. Neurosci. Res. 1996, 46, 727–733. [Google Scholar] [CrossRef]
- Makker, P.G.S.; Duffy, S.S.; Lees, J.G.; Perera, C.J.; Tonkin, R.S.; Butovsky, O.; Park, S.B.; Goldstein, D.; Moalem-Taylor, G. Characterisation of Immune and Neuroinflammatory Changes Associated with Chemotherapy-Induced Peripheral Neuropathy. PLoS ONE 2017, 12, e0170814. [Google Scholar] [CrossRef]
- Yang, T.; Liu, Y.W.; Zhao, L.; Wang, H.; Yang, N.; Dai, S.S.; He, F. Metabotropic Glutamate Receptor 5 Deficiency Inhibits Neutrophil Infiltration after Traumatic Brain Injury in Mice. Sci. Rep. 2017, 7, 9998. [Google Scholar] [CrossRef] [PubMed]
- Koch, A.E.; Kunkel, S.L.; Shah, M.R.; Fu, R.; Mazarakis, D.D.; Haines, G.K.; Burdick, M.D.; Pope, R.M.; Strieter, R.M. Macrophage Inflammatory Protein-Iβ: A C-C Chemokine in Osteoarthritis. Clin. Immunol. Immunopathol. 1995, 77, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Hanifi-Moghaddam, P.; Kappler, S.; Seissler, J.; Müller-Scholze, S.; Martin, S.; Roep, B.O.; Strassburger, K.; Kolb, H.; Schloot, N.C. Altered Chemokine Levels in Individuals at Risk of Type 1 Diabetes Mellitus. Diabet. Med. 2006, 23, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Capelli, A.; Di Stefano, A.; Gnemmi, I.; Balbo, P.; Cerutti, C.G.; Balbi, B.; Lusuardi, M.; Donner, C.F. Increased MCP-1 and MIP-1β in Bronchoalveolar Lavage Fluid of Chronic Bronchitics. Eur. Respir. J. 1999, 14, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Ajuebor, M.N.; Hogaboam, C.M.; Kunkel, S.L.; Proudfoot, A.E.I.; Wallace, J.L. The Chemokine RANTES Is a Crucial Mediator of the Progression from Acute to Chronic Colitis in the Rat. J. Immunol. 2001, 166, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Schall, T.J.; Bacon, K.; Toy, K.J.; Goeddel, D.V. Selective Attraction of Monocytes and T Lymphocytes of the Memory Phenotype by Cytokine RANTES. Nature 1990, 347, 669–671. [Google Scholar] [CrossRef] [PubMed]
- Sandhir, R.; Puri, V.; Klein, R.M.; Berman, N.E.J. Differential Expression of Cytokines and Chemokines during Secondary Neuron Death Following Brain Injury in Old and Young Mice. Neurosci. Lett. 2004, 369, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Helmy, A.; Carpenter, K.L.H.; Menon, D.K.; Pickard, J.D.; Hutchinson, P.J.A. The Cytokine Response to Human Traumatic Brain Injury: Temporal Profiles and Evidence for Cerebral Parenchymal Production. J. Cereb. Blood Flow Metab. 2011, 31, 658–670. [Google Scholar] [CrossRef] [PubMed]
- Lumpkins, K.; Bochicchio, G.V.; Zagol, B.; Ulloa, K.; Simard, J.M.; Schaub, S.; Meyer, W.; Scalea, T. Plasma Levels of the Beta Chemokine Regulated upon Activation, Normal T Cell Expressed, and Secreted (RANTES) Correlate with Severe Brain Injury. J. Trauma-Inj. Infect. Crit. Care 2008, 64, 358–361. [Google Scholar] [CrossRef]
- Ajoy, R.; Lo, Y.C.; Ho, M.H.; Chen, Y.Y.; Wang, Y.; Chen, Y.H.; Jing-Yuan, C.; Changou, C.A.; Hsiung, Y.C.; Chen, H.M.; et al. CCL5 Promotion of Bioenergy Metabolism Is Crucial for Hippocampal Synapse Complex and Memory Formation. Mol. Psychiatry 2021, 26, 6451–6468. [Google Scholar] [CrossRef]
- Tokami, H.; Ago, T.; Sugimori, H.; Kuroda, J.; Awano, H.; Suzuki, K.; Kiyohara, Y.; Kamouchi, M.; Kitazono, T. RANTES Has a Potential to Play a Neuroprotective Role in an Autocrine/Paracrine Manner after Ischemic Stroke. Brain Res. 2013, 1517, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Liou, J.; Mao, C.-C.; Ching-Wah Sum, D.; Liu, F.-C.; Lai, Y.-S.; Li, J.-C.; Day, Y.-J. Peritoneal Administration of Met-RANTES Attenuates Inflammatory and Nociceptive Responses in a Murine Neuropathic Pain Model. J. Pain 2013, 14, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.B.; Tran, P.B.; Gillard, S.E.; Hurley, R.W.; Hammond, D.L.; Miller, R.J. Chemokines and Glycoprotein 120 Produce Pain Hypersensitivity by Directly Exciting Primary Nociceptive Neurons. J. Neurosci. 2001, 21, 5027–5035. [Google Scholar] [CrossRef] [PubMed]
- Liou, J.T.; Yuan, H.B.; Mao, C.C.; Lai, Y.S.; Day, Y.J. Absence of C-C Motif Chemokine Ligand 5 in Mice Leads to Decreased Local Macrophage Recruitment and Behavioral Hypersensitivity in a Murine Neuropathic Pain Model. Pain 2012, 153, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Malon, J.T.; Cao, L. Calcitonin Gene-Related Peptide Contributes to Peripheral Nerve Injury-Induced Mechanical Hypersensitivity through CCL5 and P38 Pathways. J. Neuroimmunol. 2016, 297, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Hang, L.; Shao, D.-H.; Chen, Z.; Chen, Y.-F.; Shu, W.-W.; Zhao, Z.-G. Involvement of Spinal CC Chemokine Ligand 5 in the Development of Bone Cancer Pain in Rats. Basic Clin. Pharmacol. Toxicol. 2013, 113, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Du, X.; Lan, F.; Li, N.; Zhang, C.; Zhu, C.; Wang, X.; He, Y.; Shao, Z.; Chen, H.; et al. Eosinophilic Inflammation Promotes CCL6-Dependent Metastatic Tumor Growth. Sci. Adv. 2021, 7, 5943–5969. [Google Scholar] [CrossRef] [PubMed]
- Kanno, M.; Suzuki, S.; Fujiwara, T.; Yokoyama, A.; Sakamoto, A.; Takahashi, H.; Imai, Y.; Tanaka, J. Functional Expression of CCL6 by Rat Microglia: A Possible Role of CCL6 in Cell–Cell Communication. J. Neuroimmunol. 2005, 167, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Li, F.; Zhang, C.; Li, N.; Huang, H.; Shao, Z.; Zhang, M.; Zhan, X.; He, Y.; Ju, Z.; et al. Eosinophil-Derived Chemokine (HCCL15/23, MCCL6) Interacts with CCR1 to Promote Eosinophilic Airway Inflammation. Signal Transduct. Target. Ther. 2021 61 2021, 6, 91. [Google Scholar] [CrossRef]
- Kim, K.-S.; Rajarathnam, K.; Clark-Lewis, I.; Sykes, B.D. Structural Characterization of a Monomeric Chemokine: Monocyte Chemoattractant Protein-3. FEBS Lett. 1996, 395, 24–25. [Google Scholar] [CrossRef]
- Renner, N.A.; Ivey, N.S.; Redmann, R.K.; Lackner, A.A.; MacLean, A.G. MCP-3/CCL7 Production by Astrocytes: Implications for SIV Neuroinvasion and AIDS Encephalitis. J. Neurovirol. 2011, 17, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Thompson, W.L.; Van Eldik, L.J. Inflammatory Cytokines Stimulate the Chemokines CCL2/MCP-1 and CCL7/MCP-7 through NFκB and MAPK Dependent Pathways in Rat Astrocytes. Brain Res. 2009, 1287, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Palma, J.A.P.; Kim, B.S. The Scope and Activation Mechanisms of Chemokine Gene Expression in Primary Astrocytes Following Infection with Theiler’s Virus. J. Neuroimmunol. 2004, 149, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.W.; Sadeghi, Z.; Levine, A.D.; Penn, M.S.; von Recum, H.A.; Caplan, A.I.; Hijaz, A. The Role of CXCL12 and CCL7 Chemokines in Immune Regulation, Embryonic Development, and Tissue Regeneration. Cytokine 2014, 69, 277–283. [Google Scholar] [CrossRef]
- Ke, B.; Huang, X.X.; Li, Y.; Li, L.Y.; Xu, Q.X.; Gao, Y.; Liu, Y.; Luo, J. Neuronal-Derived Ccl7 Drives Neuropathic Pain by Promoting Astrocyte Proliferation. Neuroreport 2016, 27, 849–857. [Google Scholar] [CrossRef]
- Thirion, S.; Nys, G.; Fiten, P.; Masure, S.; Van Damme, J.; Opdenakker, G. Mouse Macrophage Derived Monocyte Chemotactic Protein-3: CDNA Cloning and Identification as MARC/FIC. Biochem. Biophys. Res. Commun. 1994, 201, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Zhang, Y.; Zhang, J.; Zhu, Z.; Lv, Q.; Su, J. Astrocyte-Derived CCL7 Promotes Microglia-Mediated Inflammation Following Traumatic Brain Injury. Int. Immunopharmacol. 2021, 99, 107975. [Google Scholar] [CrossRef]
- Ali, S.; Robertson, H.; Wain, J.H.; Isaacs, J.D.; Malik, G.; Kirby, J.A. A Non-Glycosaminoglycan-Binding Variant of CC Chemokine Ligand 7 (Monocyte Chemoattractant Protein-3) Antagonizes Chemokine-Mediated Inflammation. J. Immunol. 2005, 175, 1257–1266. [Google Scholar] [CrossRef]
- Xuan, W.; Qu, Q.; Zheng, B.; Xiong, S.; Fan, G.-H. The Chemotaxis of M1 and M2 Macrophages Is Regulated by Different Chemokines. J. Leukoc. Biol. 2015, 97, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Morganti, J.M.; Riparip, L.K.; Chou, A.; Liu, S.; Gupta, N.; Rosi, S. Age Exacerbates the CCR2/5-Mediated Neuroinflammatory Response to Traumatic Brain Injury. J. Neuroinflamm. 2016, 13, 80. [Google Scholar] [CrossRef]
- Chou, A.; Krukowski, K.; Morganti, J.M.; Riparip, L.K.; Rosi, S. Persistent Infiltration and Impaired Response of Peripherally-Derived Monocytes after Traumatic Brain Injury in the Aged Brain. Int. J. Mol. Sci. 2018, 19, 1616. [Google Scholar] [CrossRef] [PubMed]
- Almeida-Suhett, C.P.; Li, Z.; Marini, A.M.; Braga, M.F.M.; Eiden, L.E. Temporal Course of Changes in Gene Expression Suggests a Cytokine-Related Mechanism for Long-Term Hippocampal Alteration after Controlled Cortical Impact. J. Neurotrauma 2014, 31, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Dyhrfort, P.; Shen, Q.; Clausen, F.; Thulin, M.; Enblad, P.; Kamali-Moghaddam, M.; Lewén, A.; Hillered, L. Monitoring of Protein Biomarkers of Inflammation in Human Traumatic Brain Injury Using Microdialysis and Proximity Extension Assay Technology in Neurointensive Care. J. Neurotrauma 2019, 36, 2872–2885. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Jiang, B.C.; Cao, D.L.; Zhao, L.X.; Zhang, Y.L. Chemokine CCL8 and Its Receptor CCR5 in the Spinal Cord Are Involved in Visceral Pain Induced by Experimental Colitis in Mice. Brain Res. Bull. 2017, 135, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Chen, W.; Xu, H.; Yang, J.; Jiang, J.; Jiang, Y.; Xu, G. Correlation of CCL8 Expression with Immune Cell Infiltration of Skin Cutaneous Melanoma: Potential as a Prognostic Indicator and Therapeutic Pathway. Cancer Cell Int. 2021, 21, 635. [Google Scholar] [CrossRef] [PubMed]
- Bäckryd, E.; Lind, A.-L.; Thulin, M.; Larsson, A.; Gerdle, B.; Gordh, T. High Levels of Cerebrospinal Fluid Chemokines Point to the Presence of Neuroinflammation in Peripheral Neuropathic Pain: A Cross-Sectional Study of 2 Cohorts of Patients Compared with Healthy Controls. Pain 2017, 158, 2487–2495. [Google Scholar] [CrossRef] [PubMed]
- Sadik, C.D.; Kim, N.D.; Luster, A.D. Neutrophils Cascading Their Way to Inflammation. Trends Immunol. 2011, 32, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Simats, A.; García-Berrocoso, T.; Penalba, A.; Giralt, D.; Llovera, G.; Jiang, Y.; Ramiro, L.; Bustamante, A.; Martinez-Saez, E.; Canals, F.; et al. CCL23: A New CC Chemokine Involved in Human Brain Damage. J. Intern. Med. 2018, 283, 461–475. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, A.; Montecucco, F. CCL23 Is a Promising Biomarker of Injury in Patients with Ischaemic Stroke. J. Intern. Med. 2018, 283, 476–478. [Google Scholar] [CrossRef]
- Lin, H.; Shen, J.; Zhu, Y.; Zhou, L.; Zhang, S.; Liu, Z.; Wu, F.; Zhan, R. Serum CCL23 Emerges as a Biomarker for Poor Prognosis in Patients with Intracerebral Hemorrhage. Clin. Chim. Acta 2022, 537, 188–193. [Google Scholar] [CrossRef]
- Rottman, J.; Slavin, A.; Silva, R.; Weiner, H.; Gerard, C.; Hancock, W. Leukocyte Recruitment during Onset of Experimental Allergic Encephalomyelitis Is CCR1 Dependent. Eur. J. Immunol. 2000, 30, 2372–2377. [Google Scholar] [CrossRef]
- Pilat, D.; Piotrowska, A.; Rojewska, E.; Jurga, A.; Ślusarczyk, J.; Makuch, W.; Basta-Kaim, A.; Przewlocka, B.; Mika, J. Blockade of IL-18 Signaling Diminished Neuropathic Pain and Enhanced the Efficacy of Morphine and Buprenorphine. Mol. Cell. Neurosci. 2016, 71, 114–124. [Google Scholar] [CrossRef]
- Pilat, D.; Rojewska, E.; Jurga, A.M.; Piotrowska, A.; Makuch, W.; Przewlocka, B.; Mika, J. IL-1 Receptor Antagonist Improves Morphine and Buprenorphine Efficacy in a Rat Neuropathic Pain Model. Eur. J. Pharmacol. 2015, 764, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Mika, J.; Zychowska, M.; Popiolek-Barczyk, K.; Rojewska, E.; Przewlocka, B. Importance of Glial Activation in Neuropathic Pain. Eur. J. Pharmacol. 2013, 716, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Wordliczek, J.; Dobrogowski, J.; Zajączkowska, R. Zastosowanie Leków Opioidowych w Leczeniu Bólu. Wszechświat 2013, 114, 10–12. [Google Scholar]
- Szczudlik, A.; Dobrogowski, J.; Wordliczek, J.; Stępień, A.; Krajnik, M.; Leppert, W.; Woroń, J.; Przeklasa-Muszyńska, A.; Kocot-Kępska, M.; Zajączkowska, R.; et al. Diagnosis and Management of Neuropathic Pain: Review of Literature and Recommendations of the Polish Society for the Study of Pain and the Neurological Society-Part One. Ból 2014, 15, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Gladue, R.P.; Brown, M.F.; Zwillich, S.H. CCR1 Antagonists: What Have We Learned from Clinical Trials. Curr. Top. Med. Chem. 2010, 10, 1268–1277. [Google Scholar] [CrossRef] [PubMed]
- Trummer, D.; Walzer, A.; Groettrup-Wolfers, E.; Schmitz, H. Efficacy, Safety and Tolerability of the CCR1 Antagonist BAY 86-5047 for the Treatment of Endometriosis-Associated Pelvic Pain: A Randomized Controlled Trial. Acta Obstet. Gynecol. Scand. 2017, 96, 694–701. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, L.; Wu, Q.; Wang, T. Chemokine CCL2 Induces Apoptosis in Cortex Following Traumatic Brain Injury. J. Mol. Neurosci. 2013, 51, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Dong, H.; Qian, Q.; Zhang, X.; Wang, Y.; Jin, W.; Qian, Y. Astrocyte-Derived CCL2 Participates in Surgery-Induced Cognitive Dysfunction and Neuroinflammation via Evoking Microglia Activation. Behav. Brain Res. 2017, 332, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, A.; Kwiatkowski, K.; Rojewska, E.; Slusarczyk, J.; Makuch, W.; Basta-Kaim, A.; Przewlocka, B.; Mika, J. Direct and Indirect Pharmacological Modulation of CCL2/CCR2 Pathway Results in Attenuation of Neuropathic Pain-In Vivo and in Vitro Evidence. J. Neuroimmunol. 2016, 297, 9–19. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Dong, Y.L.; Lu, Y.; Cao, S.; Zhao, Z.Q.; Gao, Y.J. Chemokine CCL2 and Its Receptor CCR2 in the Medullary Dorsal Horn Are Involved in Trigeminal Neuropathic Pain. J. Neuroinflamm. 2012, 9, 136. [Google Scholar] [CrossRef]
- Morganti, J.M.; Jopson, T.D.; Liu, S.; Riparip, L.-K.; Guandique, C.K.; Gupta, N.; Ferguson, A.R.; Rosi, S. CCR2 Antagonism Alters Brain Macrophage Polarization and Ameliorates Cognitive Dysfunction Induced by Traumatic Brain Injury. J. Neurosci. 2015, 35, 748–760. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.X.; Broughton, B.R.S.; Ah Kim, H.; Lee, S.; Drummond, G.R.; Sobey, C.G. Evidence That Ly6C(Hi) Monocytes Are Protective in Acute Ischemic Stroke by Promoting M2 Macrophage Polarization. Stroke 2015, 46, 1929–1937. [Google Scholar] [CrossRef] [PubMed]
- Van Steenwinckel, J.; Goazigo, A.R.L.; Pommier, B.; Mauborgne, A.; Dansereau, M.A.; Kitabgi, P.; Sarret, P.; Pohl, M.; Parsadaniantz, S.M. CCL2 Released from Neuronal Synaptic Vesicles in the Spinal Cord Is a Major Mediator of Local Inflammation and Pain after Peripheral Nerve Injury. J. Neurosci. 2011, 31, 5865–5875. [Google Scholar] [CrossRef] [PubMed]
- Serrano, A.; Paré, M.; Mcintosh, F.; Jr Elmes, S.; Martino, G.; Jomphe, C.; Lessard, E.; Lembo, P.M.; Vaillancourt, F.; Perkins, M.N.; et al. Blocking Spinal CCR2 with AZ889 Reversed Hyperalgesia in a Model of Neuropathic Pain. Mol. Pain 2010, 6, 1744–8069. [Google Scholar] [CrossRef]
- Bogacka, J.; Ciapała, K.; Pawlik, K.; Kwiatkowski, K.; Dobrogowski, J.; Przeklasa-Muszynska, A.; Mika, J. CCR4 Antagonist (C021) Administration Diminishes Hypersensitivity and Enhances the Analgesic Potency of Morphine and Buprenorphine in a Mouse Model of Neuropathic Pain. Front. Immunol. 2020, 11, 1241. [Google Scholar] [CrossRef]
- Zhang, A.; Liu, Y.; Xu, H.; Zhang, Z.; Wang, X.; Yuan, L.; Lenahan, C.; Zhang, C.; Jiang, J.; Fang, C.; et al. CCL17 Exerts Neuroprotection through Activation of CCR4/MTORC2 Axis in Microglia after Subarachnoid Haemorrhage in Rats. Stroke Vasc. Neurol. 2022, 8, 4–16. [Google Scholar] [CrossRef]
- Liu, X.L.; Sun, D.D.; Zheng, M.T.; Li, X.T.; Niu, H.H.; Zhang, L.; Zhou, Z.W.; Rong, H.T.; Wang, Y.; Wang, J.W.; et al. Maraviroc Promotes Recovery from Traumatic Brain Injury in Mice by Suppression of Neuroinflammation and Activation of Neurotoxic Reactive Astrocytes. Neural Regen. Res. 2023, 18, 141–149. [Google Scholar] [CrossRef]
- Friedman-Levi, Y.; Liraz-Zaltsman, S.; Shemesh, C.; Rosenblatt, K.; Kesner, E.L.; Gincberg, G.; Carmichael, S.T.; Silva, A.J.; Shohami, E. Pharmacological Blockers of CCR5 and CXCR4 Improve Recovery after Traumatic Brain Injury. Exp. Neurol. 2021, 338, 113604. [Google Scholar] [CrossRef]
- Wu, Q.L.; Cui, L.Y.; Ma, W.Y.; Wang, S.S.; Zhang, Z.; Feng, Z.P.; Sun, H.S.; Chu, S.F.; He, W.B.; Chen, N.H. A Novel Small-Molecular CCR5 Antagonist Promotes Neural Repair after Stroke. Acta Pharmacol. Sin. 2023, 44, 1935–1947. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Cao, P.; Guo, X.; Yin, M.; Li, X.; Jiang, L.; Shao, J.; Chen, X.; Jiang, C.; Tao, L.; et al. Maraviroc, an Inhibitor of Chemokine Receptor Type 5, Alleviates Neuroinflammatory Response after Cerebral Ischemia/Reperfusion Injury via Regulating MAPK/NF-ΚB Signaling. Int. Immunopharmacol. 2022, 108, 108755. [Google Scholar] [CrossRef]
- Tang, L.; Wang, L.; Jin, F.; Hao, Y.; Zhao, T.; Zheng, W.; He, Z. Inflammatory Regulation by Restraining M2 Microglial Polarization: Neurodestructive Effects of Kallikrein-Related Peptidase 8 Activation in Intracerebral Hemorrhage. Int. Immunopharmacol. 2023, 124, 110855. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, A.; Kwiatkowski, K.; Rojewska, E.; Makuch, W.; Mika, J. Maraviroc Reduces Neuropathic Pain through Polarization of Microglia and Astroglia—Evidence from in Vivo and in Vitro Studies. Neuropharmacology 2016, 108, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Takami, S.; Minami, M.; Katayama, T.; Nagata, I.; Namura, S.; Satoh, M. TAK-779, a Nonpeptide CC Chemokine Receptor Antagonist, Protects the Brain against Focal Cerebral Ischemia in Mice. J. Cereb. Blood Flow Metab. 2002, 22, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte Chemoattractant Protein-1 (MCP-1): An Overview. J. Interf. Cytokine Res. 2009, 29, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, T.; Bravo, R. Cloning and Functional Expression of MCCR2, a Murine Receptor for the C-C Chemokines JE and FIC. J. Biol. Chem. 1996, 271, 11603–11606. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.; Bhangoo, S.; Banisadr, G.; Freitag, C.; Ren, D.; White, F.A.; Miller, R.J. Visualization of Chemokine Receptor Activation in Transgenic Mice Reveals Peripheral Activation of CCR2 Receptors in States of Neuropathic Pain. J. Neurosci. 2009, 29, 8051–8062. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xiang, Q.S.; Echeverry, S.; Mogil, J.S.; De Koninck, Y.; Rivest, S. Expression of CCR2 in Both Resident and Bone Marrow-Derived Microglia Plays a Critical Role in Neuropathic Pain. J. Neurosci. 2007, 27, 12396–12406. [Google Scholar] [CrossRef]
- Mizutani, M.; Pino, P.A.; Saederup, N.; Charo, I.F.; Ransohoff, R.M.; Cardona, A.E. The Fractalkine Receptor but Not CCR2 Is Present on Microglia from Embryonic Development throughout Adulthood. J. Immunol. 2012, 188, 29–36. [Google Scholar] [CrossRef]
- Saederup, N.; Cardona, A.E.; Croft, K.; Mizutani, M.; Cotleur, A.C.; Tsou, C.L.; Ransohoff, R.M.; Charo, I.F. Selective Chemokine Receptor Usage by Central Nervous System Myeloid Cells in CCR2-Red Fluorescent Protein Knock-in Mice. PLoS ONE 2010, 5, e13693. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.J.; Ji, R.R. Chemokines, Neuronal-Glial Interactions, and Central Processing of Neuropathic Pain. Pharmacol. Ther. 2010, 126, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Pawlik, K.; Mika, J. Targeting Members of the Chemokine Family as a Novel Approach to Treating Neuropathic Pain. Molecules 2023, 28, 5766. [Google Scholar] [CrossRef] [PubMed]
- Willenborg, S.; Lucas, T.; Van Loo, G.; Knipper, J.A.; Krieg, T.; Haase, I.; Brachvogel, B.; Hammerschmidt, M.; Nagy, A.; Ferrara, N.; et al. CCR2 Recruits an Inflammatory Macrophage Subpopulation Critical for Angiogenesis in Tissue Repair. Blood 2012, 120, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Xing, Z.; Lu, C.; Hu, D.; Yu, Y.Y.; Wang, X.; Colnot, C.; Nakamura, M.; Wu, Y.; Miclau, T.; Marcucio, R.S. Multiple Roles for CCR2 during Fracture Healing. DMM Dis. Model. Mech. 2010, 3, 451–458. [Google Scholar] [CrossRef] [PubMed]
- El Khoury, J.; Toft, M.; Hickman, S.E.; Means, T.K.; Terada, K.; Geula, C.; Luster, A.D. Ccr2 Deficiency Impairs Microglial Accumulation and Accelerates Progression of Alzheimer-like Disease. Nat. Med. 2007, 13, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Naert, G.; Rivest, S. CC Chemokine Receptor 2 Deficiency Aggravates Cognitive Impairments and Amyloid Pathology in a Transgenic Mouse Model of Alzheimer’s Disease. J. Neurosci. 2011, 31, 6208–6220. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.L.; Kim, C.C.; Ryba, B.E.; Niemi, E.C.; Bando, J.K.; Locksley, R.M.; Liu, J.; Nakamura, M.C.; Seaman, W.E. Traumatic Brain Injury Induces Macrophage Subsets in the Brain. Eur. J. Immunol. 2013, 43, 2010–2022. [Google Scholar] [CrossRef] [PubMed]
- Dimitrijevic, O.B.; Stamatovic, S.M.; Keep, R.F.; Andjelkovic, A.V. Absence of the Chemokine Receptor CCR2 Protects against Cerebral Ischemia/Reperfusion Injury in Mice. Stroke 2007, 38, 1345–1353. [Google Scholar] [CrossRef]
- Mahad, D.J.; Ransohoff, R.M. The Role of MCP-1 (CCL2) and CCR2 in Multiple Sclerosis and Experimental Autoimmune Encephalomyelitis (EAE). Semin. Immunol. 2003, 15, 23–32. [Google Scholar] [CrossRef]
- Saadane, A.; Veenstra, A.A.; Minns, M.S.; Tang, J.; Du, Y.; Abubakr Elghazali, F.; Lessieur, E.M.; Pearlman, E.; Kern, T.S. CCR2-Positive Monocytes Contribute to the Pathogenesis of Early Diabetic Retinopathy in Mice. Diabetologia 2023, 66, 590–602. [Google Scholar] [CrossRef]
- Belarbi, K.; Jopson, T.; Arellano, C.; Fike, J.R.; Rosi, S. CCR2 Deficiency Prevents Neuronal Dysfunction and Cognitive Impairments Induced by Cranial Irradiation. Cancer Res. 2013, 73, 1201–1210. [Google Scholar] [CrossRef] [PubMed]
- Quinones, M.P.; Estrada, C.A.; Kalkonde, Y.; Ahuja, S.K.; Kuziel, W.A.; Mack, M.; Ahuja, S.S. The Complex Role of the Chemokine Receptor CCR2 in Collagen-Induced Arthritis: Implications for Therapeutic Targeting of CCR2 in Rheumatoid Arthritis. J. Mol. Med. 2005, 83, 672–681. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, K.; Mika, J. Chemokines under Neuropathic Pain. Ból 2014, 15, 19–35. [Google Scholar] [CrossRef]
- Hsieh, C.L.; Niemi, E.C.; Wang, S.H.; Lee, C.C.; Bingham, D.; Zhang, J.; Cozen, M.L.; Charo, I.; Huang, E.J.; Liu, J.; et al. CCR2 Deficiency Impairs Macrophage Infiltration and Improves Cognitive Function after Traumatic Brain Injury. J. Neurotrauma 2014, 31, 1677–1688. [Google Scholar] [CrossRef]
- Abbadie, C.; Lindia, J.A.; Cumiskey, A.M.; Peterson, L.B.; Mudgett, J.S.; Bayne, E.K.; DeMartino, J.A.; MacIntyre, D.E.; Forrest, M.J. Impaired Neuropathic Pain Responses in Mice Lacking the Chemokine Receptor CCR2. Proc. Natl. Acad. Sci. USA 2003, 100, 7947–7952. [Google Scholar] [CrossRef] [PubMed]
- Charo, I.F.; Myers, S.J.; Herman, A.; Franci, C.; Connolly, A.J.; Coughlin, S.R. Molecular Cloning and Functional Expression of Two Monocyte Chemoattractant Protein 1 Receptors Reveals Alternative Splicing of the Carboxyl-Terminal Tails. Proc. Natl. Acad. Sci. USA 1994, 91, 2752–2756. [Google Scholar] [CrossRef] [PubMed]
- Wong, L.M.; Myers, S.J.; Tsou, C.L.; Gosling, J.; Arai, H.; Charo, I.F. Organization and Differential Expression of the Human Monocyte Chemoattractant Protein 1 Receptor Gene. Evidence for the Role of the Carboxyl-Terminal Tail in Receptor Trafficking. J. Biol. Chem. 1997, 272, 1038–1045. [Google Scholar] [CrossRef] [PubMed]
- Bartoli, C.; Civatte, M.; Pellissier, J.; Figarella-Branger, D. CCR2A and CCR2B, the Two Isoforms of the Monocyte Chemoattractant Protein-1 Receptor Are up-Regulated and Expressed by Different Cell Subsets in Idiopathic Inflammatory Myopathies. Acta Neuropathol. 2001, 102, 385–392. [Google Scholar] [CrossRef]
- Weber, K.S.C.; Nelson, P.J.; Gröne, H.J.; Weber, C. Expression of CCR2 by Endothelial Cells: Implications for MCP-1 Mediated Wound Injury Repair and In Vivo Inflammatory Activation of Endothelium. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 2085–2093. [Google Scholar] [CrossRef]
- Spinetti, G.; Wang, M.; Monticone, R.; Zhang, J.; Zhao, D.; Lakatta, E.G. Rat Aortic MCP-1 and Its Receptor CCR2 Increase with Age and Alter Vascular Smooth Muscle Cell Function. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Stamatovic, S.M.; Keep, R.F.; Kunkel, S.L.; Andjelkovic, A.V. Potential Role of MCP-1 in Endothelial Cell Tight Junction “Opening”: Signaling via Rho and Rho Kinase. J. Cell Sci. 2003, 116, 4615–4628. [Google Scholar] [CrossRef] [PubMed]
- Park, H.K.; Na, Y.H.; Nguyen, H.T.; Nguyen, L.P.; Hurh, S.; Seong, J.Y.; Lee, C.S.; Ham, B.J.; Hwang, J.I. Analysis of CCR2 Splice Variant Expression Patterns and Functional Properties. Cell Biosci. 2022, 12, 59. [Google Scholar] [CrossRef] [PubMed]
- Denk, F.; Crow, M.; Didangelos, A.; Lopes, D.M.; McMahon, S.B. Persistent Alterations in Microglial Enhancers in a Model of Chronic Pain. Cell Rep. 2016, 15, 1771–1781. [Google Scholar] [CrossRef] [PubMed]
- Dawes, J.M.; Kiesewetter, H.; Perkins, J.R.; Bennett, D.L.H.; McMahon, S.B. Chemokine Expression in Peripheral Tissues from the Monosodium Iodoacetate Model of Chronic Joint Pain. Mol. Pain 2013, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Longobardi, L.; Temple, J.D.; Tagliafierro, L.; Willcockson, H.; Esposito, A.; D’Onofrio, N.; Stein, E.; Li, T.; Myers, T.J.; Ozkan, H.; et al. Role of the C-C Chemokine Receptor-2 in a Murine Model of Injury-Induced Osteoarthritis. Osteoarthr. Cartil. 2017, 25, 914–925. [Google Scholar] [CrossRef]
- Yamagami, S.; Tamura, M.; Hayashi, M.; Endo, N.; Tanabe, H.; Katsuura, Y.; Komoriya, K. Differential Production of MCP-1 and Cytokine-Induced Neutrophil Chemoattractant in the Ischemic Brain after Transient Focal Ischemia in Rats. J. Leukoc. Biol. 1999, 65, 744–749. [Google Scholar] [CrossRef]
- Singhal, G.; Baune, B.T. Microglia: An Interface between the Loss of Neuroplasticity and Depression. Front. Cell. Neurosci. 2017, 11, 270. [Google Scholar] [CrossRef]
- Somebang, K.; Rudolph, J.; Imhof, I.; Li, L.; Niemi, E.C.; Shigenaga, J.; Tran, H.; Gill, T.M.; Lo, I.; Zabel, B.A.; et al. CCR2 Deficiency Alters Activation of Microglia Subsets in Traumatic Brain Injury. Cell Rep. 2021, 36, 109727. [Google Scholar] [CrossRef]
- Flores-Toro, J.A.; Luo, D.; Gopinath, A.; Sarkisian, M.R.; Campbell, J.J.; Charo, I.F.; Singh, R.; Schall, T.J.; Datta, M.; Jain, R.K.; et al. CCR2 Inhibition Reduces Tumor Myeloid Cells and Unmasks a Checkpoint Inhibitor Effect to Slow Progression of Resistant Murine Gliomas. Proc. Natl. Acad. Sci. USA 2020, 117, 1129–1138. [Google Scholar] [CrossRef]
- Zhu, X.; Cao, S.; Zhu, M.D.; Liu, J.Q.; Chen, J.J.; Gao, Y.J. Contribution of Chemokine CCL2/CCR2 Signaling in the Dorsal Root Ganglion and Spinal Cord to the Maintenance of Neuropathic Pain in a Rat Model of Lumbar Disc Herniation. J. Pain 2014, 15, 516–526. [Google Scholar] [CrossRef] [PubMed]
- Johnston, I.; Milligan, E.; Wieseler-Frank, J.; Frank, M.G.; Zapata, V.; Campisi, J.; Langer, S.; Martin, D.; Green, P.; Fleshner, M.; et al. A Role for Proinflammatory Cytokines and Fractalkine in Analgesia, Tolerance, and Subsequent Pain Facilitation Induced by Chronic Intrathecal Morphine. J. Neurosci. 2004, 24, 7353–7365. [Google Scholar] [CrossRef]
- Zhao, C.; Guo, R.; Hu, F.; Chen, P.; Cui, Y.; Feng, J.; Meng, J.; Mo, L.; Liao, X. Spinal MCP-1 Contributes to the Development of Morphine Antinociceptive Tolerance in Rats. Am. J. Med. Sci. 2012, 344, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Wetzel, M.A.; Steele, A.D.; Eisenstein, T.K.; Adler, M.W.; Henderson, E.E.; Rogers, T.J. Mu-Opioid Induction of Monocyte Chemoattractant Protein-1, RANTES, and IFN-Gamma-Inducible Protein-10 Expression in Human Peripheral Blood Mononuclear Cells. J. Immunol. 2000, 165, 6519–6524. [Google Scholar] [CrossRef]
- Szabo, I.; Chen, X.H.; Xin, L.; Adler, M.W.; Howard, O.M.Z.; Oppenheim, J.J.; Rogers, T.J. Heterologous Desensitization of Opioid Receptors by Chemokines Inhibits Chemotaxis and Enhances the Perception of Pain. Proc. Natl. Acad. Sci. USA 2002, 99, 10276–10281. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.P.; Jun, Y.C.; Choi, J.K.; Kim, J.I.; Carp, R.I.; Kim, Y.S. The Expression of RANTES and Chemokine Receptors in the Brains of Scrapie-Infected Mice. J. Neuroimmunol. 2005, 158, 26–33. [Google Scholar] [CrossRef]
- Chang, X.; Shen, J.; Yang, H.; Xu, Y.; Gao, W.; Wang, J.; Zhang, H.; He, S. Upregulated Expression of CCR3 in Osteoarthritis and CCR3 Mediated Activation of Fibroblast-like Synoviocytes. Cytokine 2016, 77, 211–219. [Google Scholar] [CrossRef]
- Van der Meer, P.; Ulrich, A.M.; Gonalez-Scarano, F.; Lavi, E. Immunohistochemical Analysis of CCR2, CCR3, CCR5, and CXCR4 in the Human Brain: Potential Mechanisms for HIV Dementia. Exp. Mol. Pathol. 2000, 69, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, H.; Sherbini, O.; Pai, E.L.L.; Kang, S.U.; Kwon, J.S.; Yang, J.; He, W.; Wang, H.; Eacker, S.M.; et al. High-Content Genome-Wide RNAi Screen Reveals CCR3 as a Key Mediator of Neuronal Cell Death. eNeuro 2016, 3, ENEURO.0185-16.2016. [Google Scholar] [CrossRef] [PubMed]
- Martin, E.; Delarasse, C. Complex Role of Chemokine Mediators in Animal Models of Alzheimer’s Disease. Biomed. J. 2018, 41, 34–40. [Google Scholar] [CrossRef]
- Albright, A.V.; Shieh, J.T.C.; Itoh, T.; Lee, B.; Pleasure, D.; O’Connor, M.J.; Doms, R.W.; González-Scarano, F. Microglia Express CCR5, CXCR4, and CCR3, but of These, CCR5 Is the Principal Coreceptor for Human Immunodeficiency Virus Type 1 Dementia Isolates. J. Virol. 1999, 73, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Francis, J.N.; Lloyd, C.M.; Sabroe, I.; Durham, S.R.; Till, S.J. T Lymphocytes Expressing CCR3 Are Increased in Allergic Rhinitis Compared with Non-Allergic Controls and Following Allergen Immunotherapy. Allergy Eur. J. Allergy Clin. Immunol. 2007, 62, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Flynn, G.; Maru, S.; Loughlin, J.; Romero, I.A.; Male, D. Regulation of Chemokine Receptor Expression in Human Microglia and Astrocytes. J. Neuroimmunol. 2003, 136, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Huaux, F.; Gharaee-Kermani, M.; Liu, T.; Morel, V.; McGarry, B.; Ullenbruch, M.; Kunkel, S.L.; Wang, J.; Xing, Z.; Phan, S.H. Role of Eotaxin-1 (CCL11) and CC Chemokine Receptor 3 (CCR3) in Bleomycin-Induced Lung Injury and Fibrosis. Am. J. Pathol. 2005, 167, 1485–1496. [Google Scholar] [CrossRef] [PubMed]
- Humbles, A.A.; Lu, B.; Friend, D.S.; Okinaga, S.; Lora, J.; Al-garawi, A.; Martin, T.R.; Gerard, N.P.; Gerard, C. The Murine CCR3 Receptor Regulates Both the Role of Eosinophils and Mast Cells in Allergen-Induced Airway Inflammation and Hyperresponsiveness. Proc. Natl. Acad. Sci. USA 2002, 99, 1479–1484. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, C.P.; Ponath, P.D. CCR3 Blockade as a New Therapy for Asthma. Expert Opin. Investig. Drugs 2000, 9, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Kim, S.Y.; Song, S.J.; Hong, H.K.; Lee, Y.; Oh, B.Y.; Lee, W.Y.; Cho, Y.B. Crosstalk between CCL7 and CCR3 Promotes Metastasis of Colon Cancer Cells via ERK-JNK Signaling Pathways. Oncotarget 2016, 7, 36842–36853. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, K.; Kukova, G.; Bunemann, E.; Buhren, B.A.; Sonkoly, E.; Szollosi, A.G.; Muller, A.; Savinko, T.; Lauerma, A.I.; Alenius, H.; et al. The Chemokine Receptor CCR3 Participates in Tissue Remodeling during Atopic Skin Inflammation. J. Dermatol. Sci. 2013, 71, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, H.; Honda, Y.; Tanaka, S.; Miyagawa, T.; Honda, M.; Honda, K.; Tokunaga, K.; Kodama, T. Narcolepsy Susceptibility Gene CCR3 Modulates Sleep-Wake Patterns in Mice. PLoS ONE 2017, 12, e0187888. [Google Scholar] [CrossRef]
- Kindstedt, E.; Holm, C.K.; Sulniute, R.; Martinez-Carrasco, I.; Lundmark, R.; Lundberg, P. CCL11, a Novel Mediator of Inflammatory Bone Resorption. Sci. Rep. 2017, 7, 5334. [Google Scholar] [CrossRef]
- Zhu, L.-P.; Xu, M.-L.; Yuan, B.-T.; Ma, L.-J.; Gao, Y.-J. Chemokine CCL7 Mediates Trigeminal Neuropathic Pain via CCR2/CCR3-ERK Pathway in the Trigeminal Ganglion of Mice. Mol. Pain 2023, 19, 174480692311693. [Google Scholar] [CrossRef] [PubMed]
- Nazarinia, D.; Behzadifard, M.; Gholampour, J.; Karimi, R.; Gholampour, M. Eotaxin-1 (CCL11) in Neuroinflammatory Disorders and Possible Role in COVID-19 Neurologic Complications. Acta Neurol. Belg. 2022, 122, 865–869. [Google Scholar] [CrossRef]
- Provost, V.; Larose, M.-C.; Langlois, A.; Rola-Pleszczynski, M.; Flamand, N.; Laviolette, M. CCL26/Eotaxin-3 Is More Effective to Induce the Migration of Eosinophils of Asthmatics than CCL11/Eotaxin-1 and CCL24/Eotaxin-2. J. Leukoc. Biol. 2013, 94, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Menzies-Gow, A.; Ying, S.; Sabroe, I.; Stubbs, V.L.; Soler, D.; Williams, T.J.; Kay, A.B. Eotaxin (CCL11) and Eotaxin-2 (CCL24) Induce Recruitment of Eosinophils, Basophils, Neutrophils, and Macrophages as Well as Features of Early- and Late-Phase Allergic Reactions Following Cutaneous Injection in Human Atopic and Nonatopic Volunteers. J. Immunol. 2002, 169, 2712–2718. [Google Scholar] [CrossRef]
- García, J.J.; Cidoncha, A.; Bote, M.E.; Hinchado, M.D.; Ortega, E. Altered Profile of Chemokines in Fibromyalgia Patients. Ann. Clin. Biochem. 2014, 51, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, Y.L.; Yu, S.Y. Synovial Fluid Eotaxin-1 Levels May Reflect Disease Progression in Primary Knee Osteoarthritis Among Elderly Han Chinese: A Cross-Sectional Study. Cartilage 2019, 10, 408. [Google Scholar] [CrossRef] [PubMed]
- Izumi, K.; Bieber, K.; Ludwig, R.J. Current Clinical Trials in Pemphigus and Pemphigoid. Front. Immunol. 2019, 10, 978. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, J.P.R.; Mukhtar, F. Passive Monoclonal and Polyclonal Antibody Therapies. Immunol. Concepts Transfus. Med. 2020, 251–348. [Google Scholar] [CrossRef]
- Ugur, M.; Derouiche, L.; Massotte, D. Heteromerization Modulates Mu Opioid Receptor Functional Properties in Vivo. Front. Pharmacol. 2018, 9, 1240. [Google Scholar] [CrossRef]
- Rogers, T.J. Bidirectional Regulation of Opioid and Chemokine Function. Front. Immunol. 2020, 11, 94. [Google Scholar] [CrossRef]
- Lee, Y.K.; Choi, D.Y.; Jung, Y.Y.; Yun, Y.W.; Lee, B.J.; Han, S.B.; Hong, J.T. Decreased Pain Responses of C-C Chemokine Receptor 5 Knockout Mice to Chemical or Inflammatory Stimuli. Neuropharmacology 2013, 67, 57–65. [Google Scholar] [CrossRef]
- Shah, S.A.; Kanabar, V.; Riffo-Vasquez, Y.; Mohamed, Z.; Cleary, S.J.; Corrigan, C.; James, A.L.; Elliot, J.G.; Shute, J.K.; Page, C.P.; et al. Platelets Independently Recruit into Asthmatic Lungs and Models of Allergic Inflammation via CCR3. Am. J. Respir. Cell Mol. Biol. 2021, 64, 557–568. [Google Scholar] [CrossRef]
- Wang, C.; Wang, Y.; Hong, T.; Cheng, B.; Gan, S.; Chen, L.; Zhang, J.; Zuo, L.; Li, J.; Cui, X. Blocking the Autocrine Regulatory Loop of Gankyrin/STAT3/CCL24/CCR3 Impairs the Progression and Pazopanib Resistance of Clear Cell Renal Cell Carcinoma. Cell Death Dis. 2020, 11, 117. [Google Scholar] [CrossRef]
- Yoshie, O.; Matsushima, K. CCR4 and Its Ligands: From Bench to Bedside. Int. Immunol. 2015, 27, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Salanga, C.L.; Handel, T.M. Chemokine Oligomerization and Interactions with Receptors and Glycosaminoglycans: The Role of Structural Dynamics in Function. Exp. Cell Res. 2011, 317, 590–601. [Google Scholar] [CrossRef]
- Cadosch, D.; Al-Mushaiqri, M.S.; Gautschi, O.P.; Chan, E.; Jung, F.J.; Skirving, A.P.; Filgueira, L. Immune Response Deviation and Enhanced Expression of Chemokine Receptor CCR4 in TBI Patients Due to Unknown Serum Factors. Injury 2010, 41, e4–e9. [Google Scholar] [CrossRef] [PubMed]
- Yoshie, O. CCR4 as a Therapeutic Target for Cancer Immunotherapy. Cancers 2021, 13, 5542. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Kim, J.; Huang, M.; Kosonen, R.; Lee, J.E. CCR4 and CCR5 Involvement in Monocyte-Derived Macrophage Migration in Neuroinflammation. Front. Immunol. 2022, 13, 876033. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chu, S.F.; Ai, Q.D.; Zhang, Z.; Guan, F.F.; Wang, S.S.; Dong, Y.X.; Zhu, J.; Jian, W.X.; Chen, N.H. CKLF1 Aggravates Focal Cerebral Ischemia Injury at Early Stage Partly by Modulating Microglia/Macrophage Toward M1 Polarization Through CCR4. Cell. Mol. Neurobiol. 2019, 39, 651–669. [Google Scholar] [CrossRef]
- Bai, M.; Sun, R.; Cao, B.; Feng, J.; Wang, J. Monocyte-Related Cytokines/Chemokines in Cerebral Ischemic Stroke. CNS Neurosci. Ther. 2023, 29, 3693–3712. [Google Scholar] [CrossRef]
- Kiguchi, N.; Ding, H.; Peters, C.M.; Kock, N.D.; Kishioka, S.; Cline, J.M.; Wagner, J.D.; Ko, M.C. Altered Expression of Glial Markers, Chemokines, and Opioid Receptors in the Spinal Cord of Type 2 Diabetic Monkeys. Biochim. Biophys. Acta 2017, 1863, 274. [Google Scholar] [CrossRef] [PubMed]
- Bogacka, J.; Ciapała, K.; Pawlik, K.; Dobrogowski, J.; Przeklasa-Muszynska, A.; Mika, J. Blockade of CCR4 Diminishes Hypersensitivity and Enhances Opioid Analgesia–Evidence from a Mouse Model of Diabetic Neuropathy. Neuroscience 2020, 441, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Kurashima, K.; Fujimura, M.; Myou, S.; Ishiura, Y.; Onai, N.; Matsushima, K. Asthma Severity Is Associated with an Increase in Both Blood CXCR3+ and CCR4+ T Cells. Respirology 2006, 11, 152–157. [Google Scholar] [CrossRef] [PubMed]
- El-Husseini, Z.W.; Vonk, J.M.; van den Berge, M.; Gosens, R.; Koppelman, G.H. Association of Asthma Genetic Variants with Asthma-Associated Traits Reveals Molecular Pathways of Eosinophilic Asthma. Clin. Transl. Allergy 2023, 13, e12239. [Google Scholar] [CrossRef] [PubMed]
- Kakinuma, T.; Nakamura, K.; Wakugawa, M.; Mitsui, H.; Tada, Y.; Saeki, H.; Torii, H.; Asahina, A.; Onai, N.; Matsushima, K.; et al. Thymus and Activation-Regulated Chemokine in Atopic Dermatitis: Serum Thymus and Activation-Regulated Chemokine Level Is Closely Related with Disease Activity. J. Allergy Clin. Immunol. 2001, 107, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Wu, H.; Jin, Z.J.; Ma, D.; Yuen, S.; Jing, X.Q.; Shi, M.M.; Shen, B.Y.; Peng, C.H.; Zhao, R.; et al. Up-Regulation of Chemokine Receptor CCR4 Is Associated with Human Hepatocellular Carcinoma Malignant Behavior. Sci. Rep. 2017, 7, 12362. [Google Scholar] [CrossRef] [PubMed]
- Jafarzadeh, A.; Arabi, Z.; Ahangar-Parvin, R.; Mohammadi-Kordkhayli, M.; Nemati, M. Ginger Extract Modulates the Expression of Chemokines CCL20 and CCL22 and Their Receptors (CCR6 and CCR4) in the Central Nervous System of Mice with Experimental Autoimmune Encephalomyelitis. Drug Res. 2017, 67, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Bajetto, A.; Bonavia, R.; Barbero, S.; Schettini, G. Characterization of Chemokines and Their Receptors in the Central Nervous System: Physiopathological Implications. J. Neurochem. 2002, 82, 1311–1329. [Google Scholar] [CrossRef] [PubMed]
- Nomiyama, H.; Imai, T.; Kusuda, J.; Miura, R.; Callen, D.F.; Yoshie, O. Assignment of the Human CC Chemokine Gene TARC (SCYA17) to Chromosome 16q13. Genomics 1997, 40, 211–213. [Google Scholar] [CrossRef]
- Fülle, L.; Offermann, N.; Niklas, J.; Belen, A.; Oliver, E.; Luca, S.; Fabian, R.; Knöpper, K.; Alferink, J.; Abdullah, Z.; et al. CCL17 Exerts a Neuroimmune Modulatory Function and Is Expressed in Hippocampal Neurons. Glia 2018, 66, 2246–2261. [Google Scholar] [CrossRef]
- Dai, Y.; Wu, Z.; Wang, F.; Zhang, Z.; Yu, M. Identification of Chemokines and Growth Factors in Proliferative Diabetic Retinopathy Vitreous. BioMed. Res. Int. 2014, 2014, 486386. [Google Scholar] [CrossRef] [PubMed]
- Vulcano, M.; Albanesi, C.; Stoppacciaro, A.; Bagnati, R.; D’Amico, G.; Struyf, S.; Transidico, P.; Bonecchi, R.; Del Prete, A.; Allavena, P.; et al. Dendritic Cells as a Major Source of Macrophage-Derived Chemokine/CCL22 in Vitro and in Vivo. Eur. J. Immunol. 2001, 31, 812–822. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, U.; Kuroda, E. Regulation of Macrophage-Derived Chemokine (MDC, CCL22) Production. Crit. Rev. Immunol. 2002, 22, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Rapp, M.; Wintergerst, M.W.M.; Kunz, W.G.; Vetter, V.K.; Knott, M.M.L.; Lisowski, D.; Haubner, S.; Moder, S.; Thaler, R.; Eiber, S.; et al. CCL22 Controls Immunity by Promoting Regulatory T Cell Communication with Dendritic Cells in Lymph Nodes. J. Exp. Med. 2019, 216, 1170. [Google Scholar] [CrossRef] [PubMed]
- Imai, T.; Chantry, D.; Raport, C.J.; Wood, C.L.; Nishimura, M.; Godiska, R.; Yoshie, O.; Gray, P.W. Macrophage-Derived Chemokine Is a Functional Ligand for the CC Chemokine Receptor 4. J. Biol. Chem. 1998, 273, 1764–1768. [Google Scholar] [CrossRef] [PubMed]
- Mariani, M.; Lang, R.; Binda, E.; Panina-Bordignon, P.; D’Ambrosio, D. Dominance of CCL22 over CCL17 in Induction of Chemokine Receptor CCR4 Desensitization and Internalization on Human Th2 Cells. Eur. J. Immunol. 2004, 34, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhang, Q.; Chen, Q.H.; Ran, F.Y.; Yu, L.M.; Liu, X.; Fu, Q.; Song, G.Y.; Tang, J.M.; Zhang, T. Combination of G-CSF and AMD3100 Improves the Anti-Inflammatory Effect of Mesenchymal Stem Cells on Inducing M2 Polarization of Macrophages through NF-ΚB-IL1RA Signaling Pathway. Front. Pharmacol. 2019, 10, 579. [Google Scholar] [CrossRef] [PubMed]
- Abboud, D.; Daubeuf, F.; Do, Q.T.; Utard, V.; Villa, P.; Haiech, J.; Bonnet, D.; Hibert, M.; Bernard, P.; Galzi, J.L.; et al. A Strategy to Discover Decoy Chemokine Ligands with an Anti-Inflammatory Activity. Sci. Rep. 2015, 5, 14746. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.B.; Xie, F.; Chang, K.K.; Shang, W.Q.; Meng, Y.H.; Yu, J.J.; Li, H.; Sun, Q.; Yuan, M.M.; Jin, L.P.; et al. Chemokine CCL17 Induced by Hypoxia Promotes the Proliferation of Cervical Cancer Cell. Am. J. Cancer Res. 2015, 5, 3072–3084. [Google Scholar]
- Dogan, R.-N.E.; Long, N.; Forde, E.; Dennis, K.; Kohm, A.P.; Miller, S.D.; Karpus, W.J. CCL22 Regulates Experimental Autoimmune Encephalomyelitis by Controlling Inflammatory Macrophage Accumulation and Effector Function. J. Leukoc. Biol. 2011, 89, 93. [Google Scholar] [CrossRef]
- Purandare, A.; Somerville, J. Antagonists of CCR4 as Immunomodulatory Agents. Curr. Top. Med. Chem. 2012, 6, 1335–1344. [Google Scholar] [CrossRef]
- Purandare, A.V.; Gao, A.; Wan, H.; Somerville, J.; Burke, C.; Seachord, C.; Vaccaro, W.; Wityak, J.; Poss, M.A. Identification of Chemokine Receptor CCR4 Antagonist. Bioorganic Med. Chem. Lett. 2005, 15, 2669–2672. [Google Scholar] [CrossRef] [PubMed]
- Cahn, A.; Hodgson, S.; Wilson, R.; Robertson, J.; Watson, J.; Beerahee, M.; Hughes, S.C.; Young, G.; Graves, R.; Hall, D.; et al. Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of GSK2239633, a CC-Chemokine Receptor 4 Antagonist, in Healthy Male Subjects: Results from an Open-Label and from a Randomised Study. BMC Pharmacol. Toxicol. 2013, 14, 14. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, K.; Ishikawa, N.; Igarashi, S.; Kawano, N.; Masuda, N.; Hattori, K.; Miyazaki, T.; Ogino, S.I.; Orita, M.; Matsumoto, Y.; et al. Potent CCR4 Antagonists: Synthesis, Evaluation, and Docking Study of 2,4-Diaminoquinazolines. Bioorganic Med. Chem. 2008, 16, 7968–7974. [Google Scholar] [CrossRef]
- Yokoyama, K.; Ishikawa, N.; Igarashi, S.; Kawano, N.; Masuda, N.; Hamaguchi, W.; Yamasaki, S.; Koganemaru, Y.; Hattori, K.; Miyazaki, T.; et al. Potent and Orally Bioavailable CCR4 Antagonists: Synthesis and Structure-Activity Relationship Study of 2-Aminoquinazolines. Bioorganic Med. Chem. 2009, 17, 64–73. [Google Scholar] [CrossRef]
- McMillin, M.; Frampton, G.; Thompson, M.; Galindo, C.; Standeford, H.; Whittington, E.; Alpini, G.; DeMorrow, S. Neuronal CCL2 Is Upregulated during Hepatic Encephalopathy and Contributes to Microglia Activation and Neurological Decline. J. Neuroinflamm. 2014, 11, 121. [Google Scholar] [CrossRef] [PubMed]
- Remer, M.; Al-Shamkhani, A.; Glennie, M.; Johnson, P. Mogamulizumab and the Treatment of CCR4-Positive T-Cell Lymphomas. Immunotherapy 2014, 6, 1187–1206. [Google Scholar] [CrossRef] [PubMed]
- Doi, T.; Muro, K.; Ishii, H.; Kato, T.; Tsushima, T.; Takenoyama, M.; Oizumi, S.; Gemmoto, K.; Suna, H.; Enokitani, K.; et al. A Phase I Study of the Anti-CC Chemokine Receptor 4 Antibody, Mogamulizumab, in Combination with Nivolumab in Patients with Advanced or Metastatic Solid Tumors. Clin. Cancer Res. 2019, 25, 6614–6622. [Google Scholar] [CrossRef] [PubMed]
- Kaul, M.; Ma, Q.; Medders, K.E.; Desai, M.K.; Lipton, S.A. HIV-1 Coreceptors CCR5 and CXCR4 Both Mediate Neuronal Cell Death but CCR5 Paradoxically Can Also Contribute to Protection. Cell Death Differ. 2007, 14, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Rottman, J.B.; Ganley, K.P.; Williams, K.; Wu, L.; Mackay, C.R.; Ringler, D.J. Cellular Localization of the Chemokine Receptor CCR5: Correlation to Cellular Targets of HIV-1 Infection. Am. J. Pathol. 1997, 151, 1341–1351. [Google Scholar]
- Zhou, M.; Greenhill, S.; Huang, S.; Silva, T.K.; Sano, Y.; Wu, S.; Cai, Y.; Nagaoka, Y.; Sehgal, M.; Cai, D.J.; et al. CCR5 Is a Suppressor for Cortical Plasticity and Hippocampal Learning and Memory. Elife 2016, 5, e20985. [Google Scholar] [CrossRef] [PubMed]
- Joy, M.T.; Ben Assayag, E.; Shabashov-Stone, D.; Liraz-Zaltsman, S.; Mazzitelli, J.; Arenas, M.; Abduljawad, N.; Kliper, E.; Korczyn, A.D.; Thareja, N.S.; et al. CCR5 Is a Therapeutic Target for Recovery after Stroke and Traumatic Brain Injury. Cell 2019, 176, 1143–1157. [Google Scholar] [CrossRef] [PubMed]
- Carbonell, W.S.; Murase, S.I.; Horwitz, A.F.; Mandell, J.W. Migration of Perilesional Microglia after Focal Brain Injury and Modulation by CC Chemokine Receptor 5: An in Situ Time-Lapse Confocal Imaging Study. J. Neurosci. 2005, 25, 7040–7047. [Google Scholar] [CrossRef] [PubMed]
- Marella, M.; Chabry, J. Neurons and Astrocytes Respond to Prion Infection by Inducing Microglia Recruitment. J. Neurosci. 2004, 24, 620–627. [Google Scholar] [CrossRef] [PubMed]
- Miyagi, T.; Chuang, L.F.; Doi, R.H.; Carlos, M.P.; Torres, J.V.; Chuang, R.Y. Morphine Induces Gene Expression of CCR5 in Human CEM X174 Lymphocytes. J. Biol. Chem. 2000, 275, 31305–31310. [Google Scholar] [CrossRef] [PubMed]
- Ochi-Ishi, R.; Nagata, K.; Inoue, T.; Tozaki-Saitoh, H.; Tsuda, M.; Inoue, K. Involvement of the Chemokine CCL3 and the Purinoceptor P2×7 in the Spinal Cord in Paclitaxel-Induced Mechanical Allodynia. Mol. Pain 2014, 10, 1744–8069-10-53. [Google Scholar] [CrossRef] [PubMed]
- Bidlack, J.M. Detection and Function of Opioid Receptors on Cells from the Immune System. Clin. Diagn. Lab. Immunol. 2000, 7, 719–723. [Google Scholar] [CrossRef] [PubMed]
- Mika, J.; Popiolek-Barczyk, K.; Rojewska, E.; Makuch, W.; Starowicz, K.; Przewlocka, B. Delta-Opioid Receptor Analgesia Is Independent of Microglial Activation in a Rat Model of Neuropathic Pain. PLoS ONE 2014, 9, e104420. [Google Scholar] [CrossRef] [PubMed]
- Lisi, L.; Tramutola, A.; De Luca, A.; Navarra, P.; Dello Russo, C. Modulatory Effects of the CCR5 Antagonist Maraviroc on Microglial Pro-Inflammatory Activation Elicited by Gp120. J. Neurochem. 2012, 120, 106–114. [Google Scholar] [CrossRef]
- Pease, J.E.; Horuk, R. Chemokine Receptor Antagonists: Part 2. Expert Opin. Ther. Pat. 2009, 19, 199–221. [Google Scholar] [CrossRef]
- Schutyser, E.; Struyf, S.; Van Damme, J. The CC Chemokine CCL20 and Its Receptor CCR6. Cytokine Growth Factor Rev. 2003, 14, 409–426. [Google Scholar] [CrossRef]
- Lee, A.Y.S.; Reimer, D.; Zehrer, A.; Lu, M.; Mielenz, D.; Körner, H. Expression of Membrane-Bound CC Chemokine Ligand 20 on Follicular T Helper Cells in T-B-Cell Conjugates. Front. Immunol. 2017, 8, 1871. [Google Scholar] [CrossRef]
- Liao, L.S.; Zhang, M.W.; Gu, Y.J.; Sun, X.C. Targeting CCL20 Inhibits Subarachnoid Hemorrhage-Related Neuroinflammation in Mice. Aging 2020, 12, 14849–14862. [Google Scholar] [CrossRef]
- Reboldi, A.; Coisne, C.; Baumjohann, D.; Benvenuto, F.; Bottinelli, D.; Lira, S.; Uccelli, A.; Lanzavecchia, A.; Engelhardt, B.; Sallusto, F. C-C Chemokine Receptor 6-Regulated Entry of TH-17 Cells into the CNS through the Choroid Plexus Is Required for the Initiation of EAE. Nat. Immunol. 2009, 10, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Sachi, N.; Kamiyama, N.; Saechue, B.; Ozaka, S.; Dewayani, A.; Ariki, S.; Chalalai, T.; Soga, Y.; Fukuda, C.; Kagoshima, Y.; et al. CCL20/CCR6 Chemokine Signaling Is Not Essential for Pathogenesis in an Experimental Autoimmune Encephalomyelitis Mouse Model of Multiple Sclerosis. Biochem. Biophys. Res. Commun. 2023, 641, 123–131. [Google Scholar] [CrossRef]
- Coursey, T.G.; Gandhi, N.B.; Volpe, E.A.; Pflugfelder, S.C.; De Paiva, C.S. Chemokine Receptors CCR6 and CXCR3 Are Necessary for CD4(+) T Cell Mediated Ocular Surface Disease in Experimental Dry Eye Disease. PLoS ONE 2013, 8, e78508. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.Q.; Yuan, G.Q.; Zhang, G.G.; Chen, Y.T.; Nie, Q.Q.; Wang, Z. Identification of CCL20 as a Key Biomarker of Inflammatory Responses in the Pathogenesis of Intracerebral Hemorrhage. Inflammation 2023, 46, 1290–1304. [Google Scholar] [CrossRef] [PubMed]
- Lötsch, J.; Mustonen, L.; Harno, H.; Kalso, E. Machine-Learning Analysis of Serum Proteomics in Neuropathic Pain after Nerve Injury in Breast Cancer Surgery Points at Chemokine Signaling via SIRT2 Regulation. Int. J. Mol. Sci. 2022, 23, 3488. [Google Scholar] [CrossRef]
- Miclescu, A.A.; Granlund, P.; Butler, S.; Gordh, T. Association between Systemic Inflammation and Experimental Pain Sensitivity in Subjects with Pain and Painless Neuropathy after Traumatic Nerve Injuries. Scand. J. Pain 2023, 23, 184–199. [Google Scholar] [CrossRef]
- Alrumaihi, F. The Multi-Functional Roles of CCR7 in Human Immunology and as a Promising Therapeutic Target for Cancer Therapeutics. Front. Mol. Biosci. 2022, 9, 834149. [Google Scholar] [CrossRef]
- Gomez-Nicola, D.; Pallas-Bazarra, N.; Valle-Argos, B.; Nieto-Sampedro, M. CCR7 Is Expressed in Astrocytes and Upregulated after an Inflammatory Injury. J. Neuroimmunol. 2010, 227, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.X.; Cao, X.; Tang, Y.C.; Liu, Y.; Tang, F.R. CCR7, CCR8, CCR9 and CCR10 in the Mouse Hippocampal CA1 Area and the Dentate Gyrus during and after Pilocarpine-Induced Status Epilepticus. J. Neurochem. 2007, 100, 1072–1088. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, M.; Gerdle, B.; Ghafouri, B.; Bäckryd, E. The Inflammatory Profile of Cerebrospinal Fluid, Plasma, and Saliva from Patients with Severe Neuropathic Pain and Healthy Controls-a Pilot Study. BMC Neurosci. 2021, 22, 6. [Google Scholar] [CrossRef]
- Guo, R.; Chen, Y.; Liu, L.; Wen, J.; Yang, H.; Zhu, Y.; Gao, M.; Liang, H.; Lai, W.; Long, H. Nerve Growth Factor Enhances Tooth Mechanical Hyperalgesia Through C-C Chemokine Ligand 19 in Rats. Front. Neurol. 2021, 12, 596. [Google Scholar] [CrossRef]
- Zhao, P.; Waxman, S.G.; Hains, B.C. Modulation of Thalamic Nociceptive Processing after Spinal Cord Injury through Remote Activation of Thalamic Microglia by Cysteine–Cysteine Chemokine Ligand 21. J. Neurosci. 2007, 27, 8893–8902. [Google Scholar] [CrossRef]
- Hauser, M.A.; Legler, D.F. Common and Biased Signaling Pathways of the Chemokine Receptor CCR7 Elicited by Its Ligands CCL19 and CCL21 in Leukocytes. J. Leukoc. Biol. 2016, 99, 869–882. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liang, L.; Cao, S.; Hou, G.; Zhang, Q.; Ma, H.; Shi, B. Serum CCL21 as a Potential Biomarker for Cognitive Impairment in Spinal Cord Injury. BioMed Res. Int. 2020, 2020, 6692802. [Google Scholar] [CrossRef]
- Rappert, A.; Biber, K.; Nolte, C.; Lipp, M.; Schubel, A.; Lu, B.; Gerard, N.P.; Gerard, C.; Boddeke, H.W.G.M.; Kettenmann, H. Secondary Lymphoid Tissue Chemokine (CCL21) Activates CXCR3 to Trigger a Cl− Current and Chemotaxis in Murine Microglia. J. Immunol. 2002, 168, 3221–3226. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, K.; Pickert, G.; Wijnvoord, N.; Häussler, A.; Tegeder, I. Dichotomy of CCL21 and CXCR3 in Nerve Injury-Evoked and Autoimmunity-Evoked Hyperalgesia. Brain. Behav. Immun. 2013, 32, 186–200. [Google Scholar] [CrossRef]
- Schweickart, V.L.; Raport, C.J.; Godiska, R.; Byers, M.G.; Eddy, R.L.; Shows, T.B.; Gray, P.W. Cloning of Human and Mouse EBI1, a Lymphoid-Specific G-Protein-Coupled Receptor Encoded on Human Chromosome 17q12-Q21.2. Genomics 1994, 23, 643–650. [Google Scholar] [CrossRef]
- Jaeger, K.; Bruenle, S.; Weinert, T.; Guba, W.; Muehle, J.; Miyazaki, T.; Weber, M.; Furrer, A.; Haenggi, N.; Tetaz, T.; et al. Structural Basis for Allosteric Ligand Recognition in the Human CC Chemokine Receptor 7. Cell 2019, 178, 1222–1230.e10. [Google Scholar] [CrossRef]
- Saha, S.; Shukla, A.K. The Inside Story: Crystal Structure of the Chemokine Receptor CCR7 with an Intracellular Allosteric Antagonist. Biochemistry 2020, 59, 12–14. [Google Scholar] [CrossRef] [PubMed]
- Hull-Ryde, E.A.; Porter, M.A.; Fowler, K.A.; Kireev, D.; Li, K.; Simpson, C.D.; Sassano, M.F.; Suto, M.J.; Pearce, K.H.; Janzen, W.; et al. Identification of Cosalane as an Inhibitor of Human and Murine CC-Chemokine Receptor 7 Signaling via a High-Throughput Screen. SLAS Discov. Adv. Life Sci. Drug Discov. 2018, 23, 1083–1091. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Hasegawa, H.; Kohno, M.; Inoue, A.; Ito, M.R.; Fujita, S. Antagonist of Secondary Lymphoid-Tissue Chemokine (CCR Ligand 21) Prevents the Development of Chronic Graft-versus-Host Disease in Mice. J. Immunol. 2003, 170, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Pilkington, K.R.; Clark-Lewis, I.; McColl, S.R. Inhibition of Generation of Cytotoxic T Lymphocyte Activity by a CCL19/Macrophage Inflammatory Protein (MIP)-3beta Antagonist. J. Biol. Chem. 2004, 279, 40276–40282. [Google Scholar] [CrossRef] [PubMed]
- Salem, A.; Alotaibi, M.; Mroueh, R.; Basheer, H.A.; Afarinkia, K. CCR7 as a Therapeutic Target in Cancer. Biochim. Biophys. Acta Rev. Cancer 2021, 1875, 188499. [Google Scholar] [CrossRef] [PubMed]
- Förster, R.; Schubel, A.; Breitfeld, D.; Kremmer, E.; Renner-Müller, I.; Wolf, E.; Lipp, M. CCR7 Coordinates the Primary Immune Response by Establishing Functional Microenvironments in Secondary Lymphoid Organs. Cell 1999, 99, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, A.; Ravens, S.; Fleige, H.; Haas, J.D.; Oberdörfer, L.; Łyszkiewicz, M.; Förster, R.; Prinz, I. CCR7-Mediated Migration in the Thymus Controls Γδ T-Cell Development. Eur. J. Immunol. 2014, 44, 1320–1329. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.A.; Meingassner, J.G.; Lipp, M.; Moore, H.D.; Rot, A. CCR7 Is Required for the in Vivo Function of CD4+ CD25+ Regulatory T Cells. J. Exp. Med. 2007, 204, 735–745. [Google Scholar] [CrossRef]
- Tiffany, H.L.; Lautens, L.L.; Gao, J.L.; Pease, J.; Locati, M.; Combadiere, C.; Modi, W.; Bonner, T.I.; Murphy, P.M. Identification of CCR8: A Human Monocyte and Thymus Receptor for the CC Chemokine I-309. J. Exp. Med. 1997, 186, 165–170. [Google Scholar] [CrossRef]
- Miller, M.D.; Hata, S.; De Waal Malefyt, R.; Krangel, M.S. A Novel Polypeptide Secreted by Activated Human T Lymphocytes. J. Immunol. 1989, 143, 2907–2916. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.A.; Ling, M.F.; Leung, J.; Shreffler, W.G.; Luster, A.D. Identification of Human CCR8 as a CCL18 Receptor. J. Exp. Med. 2013, 210, 1889–1898. [Google Scholar] [CrossRef] [PubMed]
- Moser, B. Chemokine Receptor-Targeted Therapies: Special Case for CCR8. Cancers 2022, 14, 511. [Google Scholar] [CrossRef] [PubMed]
- Gombert, M.; Dieu-Nosjean, M.-C.; Winterberg, F.; Bünemann, E.; Kubitza, R.C.; Da Cunha, L.; Haahtela, A.; Lehtimäki, S.; Müller, A.; Rieker, J.; et al. CCL1-CCR8 Interactions: An Axis Mediating the Recruitment of T Cells and Langerhans-Type Dendritic Cells to Sites of Atopic Skin Inflammation. J. Immunol. 2005, 174, 5082–5091. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Pérez, F.; Kato, Y.; Gonzalez-Menendez, I.; Laiño, J.; Ohbayashi, M.; Burggraf, M.; Krause, M.; Kirberg, J.; Iwakura, Y.; Martella, M.; et al. CCR8 Leads to Eosinophil Migration and Regulates Neutrophil Migration in Murine Allergic Enteritis. Sci. Rep. 2019, 9, 9608. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Zhong, S.; Du, H.; Han, S.; Wei, X.; Gong, Q. MiR-184-5p Represses Neuropathic Pain by Regulating CCL1/CCR8 Signaling Interplay in the Spinal Cord in Diabetic Mice. Neurol. Res. 2024, 46, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Jun, K.J.; Lee, M.J.; Shin, D.C.; Woo, M.Y.; Kim, K.; Park, S. Identification of CCL1 as a Gene Differentially Expressed in CD4+ T Cells Expressing TIM-3. Immune Netw. 2011, 11, 203. [Google Scholar] [CrossRef]
- Akimoto, N.; Ifuku, M.; Mori, Y.; Noda, M. Effects of Chemokine (C-C Motif) Ligand 1 on Microglial Function. Biochem. Biophys. Res. Commun. 2013, 436, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Xiao, W.; Wang, F.; Liu, J.; Zhi, L.J. MiR-21-5p Inhibits Neuropathic Pain Development via Directly Targeting C-C Motif Ligand 1 and Tissue Inhibitor of Metalloproteinase-3. J. Cell. Biochem. 2019, 120, 16614–16623. [Google Scholar] [CrossRef]
- Zaballos, Á.; Gutiérrez, J.; Varona, R.; Ardavín, C.; Márquez, G. Cutting Edge: Identification of the Orphan Chemokine Receptor GPR-9-6 as CCR9, the Receptor for the Chemokine TECK. J. Immunol. 1999, 162, 5671–5675. [Google Scholar] [CrossRef]
- Igaki, K.; Komoike, Y.; Nakamura, Y.; Watanabe, T.; Yamasaki, M.; Fleming, P.; Yang, L.; Soler, D.; Fedyk, E.; Tsuchimori, N. MLN3126, an Antagonist of the Chemokine Receptor CCR9, Ameliorates Inflammation in a T Cell Mediated Mouse Colitis Model. Int. Immunopharmacol. 2018, 60, 160–169. [Google Scholar] [CrossRef]
- Wermers, J.D.; McNamee, E.N.; Wurbel, M.A.; Jedlicka, P.; Riveranieves, J. The Chemokine Receptor CCR9 Is Required for the T-Cell-Mediated Regulation of Chronic Ileitis in Mice. Gastroenterology 2011, 140, 1526–1535.e3. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Doan, N.; Said, J.; Karunasiri, D.; Pullarkat, S.T. Strong Expression of Chemokine Receptor CCR9 in Diffuse Large B-Cell Lymphoma and Follicular Lymphoma Strongly Correlates with Gastrointestinal Involvement. Hum. Pathol. 2014, 45, 1451–1458. [Google Scholar] [CrossRef] [PubMed]
- Tu, Z.; Xiao, R.; Xiong, J.; Tembo, K.M.; Deng, X.; Xiong, M.; Liu, P.; Wang, M.; Zhang, Q. CCR9 in Cancer: Oncogenic Role and Therapeutic Targeting. J. Hematol. Oncol. 2016, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-R.; Peden, K.W.C.; Zaitseva, M.B.; Golding, H.; Farber, J.M. CCR9A and CCR9B: Two Receptors for the Chemokine CCL25/TECK/Ck Beta-15 That Differ in Their Sensitivities to Ligand. J. Immunol. 2000, 164, 1293–1305. [Google Scholar] [CrossRef] [PubMed]
- Spencer, D.M.; Wandless, T.J.; Schreiber, S.L.; Crabtree, G.R. Controlling Signal Transduction with Synthetic Ligands. Science 1993, 262, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Walters, M.J.; Wang, Y.; Lai, N.; Baumgart, T.; Zhao, B.N.; Dairaghi, D.J.; Bekker, P.; Ertl, L.S.; Penfold, M.E.T.; Jaen, J.C.; et al. Characterization of CCX282-B, an Orally Bioavailable Antagonist of the CCR9 Chemokine Receptor, for Treatment of Inflammatory Bowel Disease. J. Pharmacol. Exp. Ther. 2010, 335, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Sun, M.; Yang, Z.; Lu, C.; Wang, Q.; Wang, H.; Deng, C.; Liu, Y.; Yang, Y. The Roles of CCR9/CCL25 in Inflammation and Inflammation-Associated Diseases. Front. Cell Dev. Biol. 2021, 9, 686548. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Han, J.; Wu, M.; Xu, L.; Wang, Y.; Yuan, W.; Hua, F.; Fan, H.; Dong, F.; Qu, X.; et al. Toll-Like Receptor 4 Promotes Th17 Lymphocyte Infiltration Via CCL25/CCR9 in Pathogenesis of Experimental Autoimmune Encephalomyelitis. J. Neuroimmune Pharmacol. 2019, 14, 493–502. [Google Scholar] [CrossRef]
- Zhou, R.; Wang, L.; Chen, L.; Feng, X.; Zhou, R.; Xiang, P.; Wen, J.; Huang, Y.; Zhou, H. Bone Marrow-Derived GCA+ Immune Cells Drive Alzheimer’s Disease Progression. Adv. Sci. 2023, 10, e2303402. [Google Scholar] [CrossRef]
- Chen, L.; Liu, X.; Zhang, H.Y.; Du, W.; Qin, Z.; Yao, Y.; Mao, Y.; Zhou, L. Upregulation of Chemokine Receptor CCR10 Is Essential for Glioma Proliferation, Invasion and Patient Survival. Oncotarget 2014, 5, 6576–6583. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Chen, J.X.; Chen, Y.; Cai, L.L.; Wang, X.Z.; Guo, W.H.; Zheng, J.F. The Chemokine Receptor CCR10 Promotes Inflammation-Driven Hepatocarcinogenesis via PI3K/Akt Pathway Activation. Cell Death Dis. 2018, 9, 232. [Google Scholar] [CrossRef] [PubMed]
- Homey, B.; Alenius, H.; Müller, A.; Soto, H.; Bowman, E.P.; Yuan, W.; McEvoy, L.; Lauerma, A.I.; Assmann, T.; Bünemann, E.; et al. CCL27-CCR10 Interactions Regulate T Cell-Mediated Skin Inflammation. Nat. Med. 2002, 8, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Mirshahpanah, P.; Li, Y.Y.Y.; Burkhardt, N.; Asadullah, K.; Zollner, T.M. CCR4 and CCR10 Ligands Play Additive Roles in Mouse Contact Hypersensitivity. Exp. Dermatol. 2008, 17, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Wise, E.L.; Duchesnes, C.; Da Fonseca, P.C.A.; Allen, R.A.; Williams, T.J.; Pease, J.E. Small Molecule Receptor Agonists and Antagonists of CCR3 Provide Insight into Mechanisms of Chemokine Receptor Activation. J. Biol. Chem. 2007, 282, 27935–27943. [Google Scholar] [CrossRef] [PubMed]
- Salanga, C.L.; O’Hayre, M.; Handel, T. Modulation of Chemokine Receptor Activity through Dimerization and Crosstalk. Cell. Mol. Life Sci. 2009, 66, 1370–1386. [Google Scholar] [CrossRef] [PubMed]
- Cherney, R.J.; Anjanappa, P.; Selvakumar, K.; Batt, D.G.; Brown, G.D.; Rose, A.V.; Vuppugalla, R.; Chen, J.; Pang, J.; Xu, S.; et al. BMS-813160: A Potent CCR2 and CCR5 Dual Antagonist Selected as a Clinical Candidate. ACS Med. Chem. Lett. 2021, 12, 1753–1758. [Google Scholar] [CrossRef] [PubMed]
- Gale, J.D.; Gilbert, S.; Blumenthal, S.; Elliott, T.; Pergola, P.E.; Goteti, K.; Scheele, W.; Perros-Huguet, C. Effect of PF-04634817, an Oral CCR2/5 Chemokine Receptor Antagonist, on Albuminuria in Adults with Overt Diabetic Nephropathy. Kidney Int. Rep. 2018, 3, 1316–1327. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.; Saag, M.; Dejesus, E.; Gathe, J.; Lalezari, J.; Landay, A.L.; Cade, J.; Enejosa, J.; Lefebvre, E.; Feinberg, J. A 48-Week Randomized Phase 2b Study Evaluating Cenicriviroc versus Efavirenz in Treatment-Naive HIV-Infected Adults with C-C Chemokine Receptor Type 5-Tropic Virus. AIDS 2016, 30, 869–878. [Google Scholar] [CrossRef]
- Fadel, H.; Temesgen, Z. Maraviroc. Drugs Today 2007, 43, 749–758. [Google Scholar] [CrossRef]
- Szpakowska, M.; Chevigné, A. VCCL2/VMIP-II, the Viral Master KEYmokine. J. Leukoc. Biol. 2016, 99, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Lindow, M.; Nansen, A.; Bartholdy, C.; Stryhn, A.; Hansen, N.J.V.; Boesen, T.P.; Wells, T.N.C.; Schwartz, T.W.; Thomsen, A.R. The Virus-Encoded Chemokine VMIP-II Inhibits Virus-Induced Tc1-Driven Inflammation. J. Virol. 2003, 77, 7393–7400. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liu, S.; Jiang, Z.; Feng, L.; Gao, Y.; Chen, Y.; Xu, A.; Huang, W.; Zhang, N.; Sun, H. Study on the Promotion of Lymphocytes in Patients with COVID-19 by Broad-Spectrum Chemokine Receptor Inhibitor VMIP-II and Its Mechanism of Signal Transmission in Vitro. Signal Transduct. Target. Ther. 2021, 6, 104. [Google Scholar] [CrossRef] [PubMed]
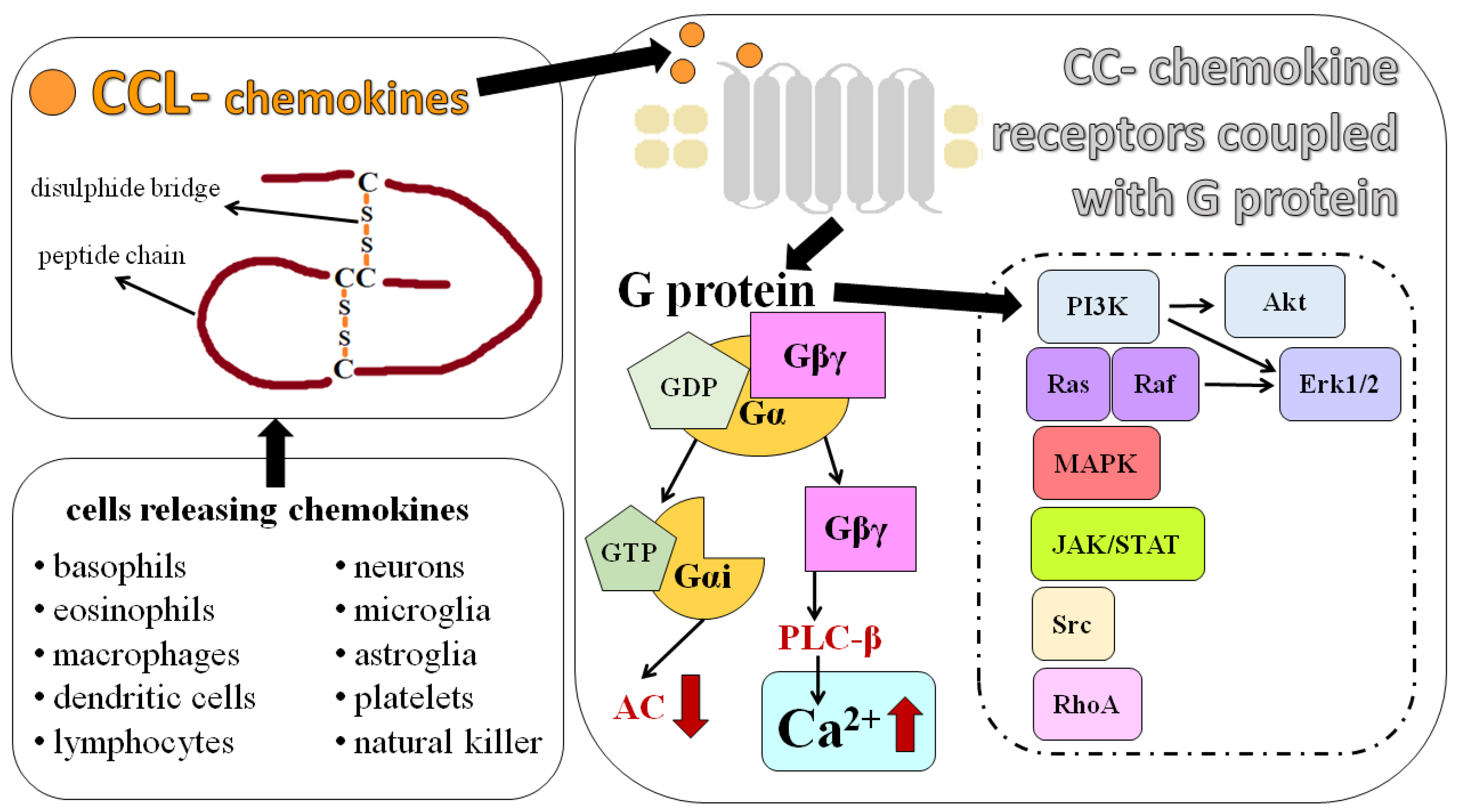
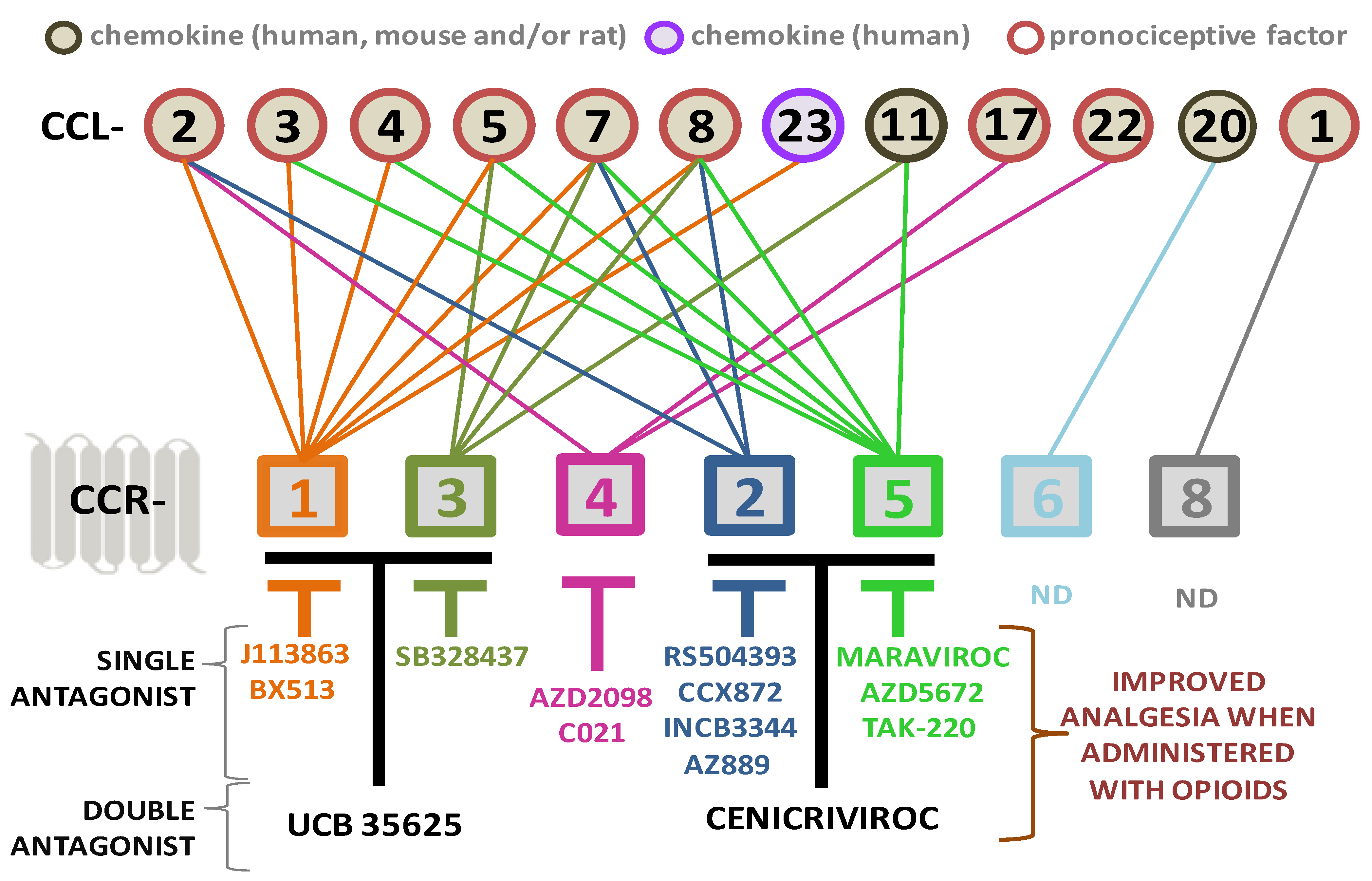
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciechanowska, A.; Mika, J. CC Chemokine Family Members’ Modulation as a Novel Approach for Treating Central Nervous System and Peripheral Nervous System Injury—A Review of Clinical and Experimental Findings. Int. J. Mol. Sci. 2024, 25, 3788. https://doi.org/10.3390/ijms25073788
Ciechanowska A, Mika J. CC Chemokine Family Members’ Modulation as a Novel Approach for Treating Central Nervous System and Peripheral Nervous System Injury—A Review of Clinical and Experimental Findings. International Journal of Molecular Sciences. 2024; 25(7):3788. https://doi.org/10.3390/ijms25073788
Chicago/Turabian StyleCiechanowska, Agata, and Joanna Mika. 2024. "CC Chemokine Family Members’ Modulation as a Novel Approach for Treating Central Nervous System and Peripheral Nervous System Injury—A Review of Clinical and Experimental Findings" International Journal of Molecular Sciences 25, no. 7: 3788. https://doi.org/10.3390/ijms25073788







