Abstract
Fertility preservation and restoration using cryo-banked prepubertal testicular tissue is a pivotal part of the childhood hematological cancer care pathway. Estimations indicate that one in 900–1400 young adults is a childhood cancer survivor, underlying the urge to develop fertility restoration protocols as some of the patients have reached the age to father their own genetic child. While it has been reported that 39% of patients present cancer cells in their testes, no efficient decontamination technique has been identified to circumvent cancer reintroduction after autologous testicular cell transplantation. Obtaining single-cell suspensions and selecting only testicular cells might be an option. In this review, mechanical dissociation/enzymatic digestion protocols applied to human testicular tissue, as well as selection and enrichment strategies, and their outcome will be presented and discussed. While the literature revealed a plethora of mechanical dissociation/enzymatic digestion protocols, testicular tissue characteristics are often missing, precluding the comparison of protocols and their outcomes. Downstream selection and enrichment strategies showed promising results with flow cytometry reaching fractions with the highest purity. Future studies should focus on investigating digestion outcomes to elucidate potential influences on both the cell type-specific viability and the cell-to-cell interactions necessary for cell proliferation and differentiation of selected or enriched testicular cell types. Such research outputs will then also be crucial for further progress in in vitro spermatogenesis from testicular cell suspensions as another option for patients that banked testicular tissue at the time of a hematological cancer.
1. Introduction
Thanks to improvements in cancer therapies over the last years, pediatric cancer survival rates have increased to more than 80% [1]. Estimations predict one in 900 to 1400 young adults to be a childhood cancer survivor [2]. Unfortunately, these cancer therapies may lead to impaired fertility in adulthood. Extending indications of drugs affecting fertility to benign disease increases even more the population concerned by this matter. Indeed, while it was shown that the majority of patients exposed to gonadotoxic therapies only experience transient fertility impairment and spermatogenesis recovery 1–5 years post-treatment [3], still 42.2% to 66.4% of the patients has germ cell (GC) failure with resulting infertility whether being prepubertal, peripubertal or postpubertal at the time of treatment [4,5,6,7,8]. For prepubertal boys, unlike adolescent boys and adult men who can benefit from conventional semen freezing, cryopreservation of immature testicular tissue (ITT) containing spermatogonial stem cells (SSCs) is the only experimental fertility preservation option which is ethically accepted to offer these patients a chance of fathering their own genetic child [9,10,11,12,13]. Survey data show that this approach is increasingly proposed worldwide [14,15].
Despite the urge, as some of the patients have reached the age to conceive [16], still no fertilizing spermatozoa have been obtained from ITT. Several methods have been investigated to reproduce spermatogenesis with cryopreserved ITT, i.e., in vitro maturation (IVM) and transplantation of single-cell suspensions or ITT fragments [13]. While autotransplantation appears to be the most promising technique with the generation of mature sperm and offspring in monkeys [17], a major concern is the possibility of cancer cell contamination of cryo-stored ITT [18,19], especially in the case of hematological malignancies showing a mean contamination rate in pre- and peripubertal boys between 37 and 39% [18,19], carrying thereby the risk of causing disease relapse if autotransplanted [20]. Indeed, it was shown that as few as 20 leukemic cells are sufficient to reinduce cancer in rats [21]. Therefore, autotransplantation of human ITT grafts is currently excluded when there is a risk of cancer cell contamination of the tissue. Xenotransplantation of human ITT and retrieval of spermatozoa to be used for in vitro fertilization with intracytoplasmic sperm injection (IVF-ICSI) would circumvent this risk but is clinically unaccepted due to xeno-microbiological agents [22]. IVM of human ITT or testicular cell suspensions aimed at differentiating SSCs in spermatozoa to be further used for IVF-ICSI would be the optimal solution to overcome the risk of cancer cell contamination and disease relapse. However, it is still at the research stage and limited to the generation of haploid spermatids from IVM of tissue fragments [23] and of cells phenotypically resembling spermatozoa after 3D-testicular cell culture [24]. Autotransplantation of properly selected spermatogonia including SSCs into the original testicular niche could be more promising based on the development of preimplantation embryos in non-human primates [25]. In addition, very recently a protocol for the autotransplantation of testicular cells in humans has been reported, pointing to the fast evolution in available methods [26] and the urgent clinical need of proper cell selection techniques. Alternatively, in vitro cancer-free-organoid formation in which selected testicular cells will colonize into an extracellular matrix (ECM)-based hydrogel/scaffold prior to transplantation could be considered [27,28]. Therefore, cell selection seems a relevant technique to obtain cancer-free single-cell suspension and guarantee safe autotransplantation. Currently, clinical implementation of the various cell selection techniques is hampered by inefficient cancer cell decontamination, risk of toxic components, requirements for post-sorting applications, low cell recovery, and limited standardization (Table 1).

Table 1.
Testicular cell selection techniques with main outcomes and unaddressed research gaps before clinical application. FACS: Fluorescent-activated cell sorting; GC: Germ cell; LC: Leydig cell; MACS: Magnetic-activated cell sorting; PTMC: Peritubular Myoid cell; SC: Sertoli cell; SSC: spermatogonial stem cell.
To obtain single-cell suspensions, demonstrating high viability while maintaining cellular diversity and preserving rare subsets of testicular cells, a plethora of methods and markers are currently being used by different teams worldwide (Figure 1). Notably, detailed information on tissue digestion protocols with reporting of cell yields and numbers of viable cells is scarce, challenging protocol standardization, study comparison, and the interpretation of results. This systematic review aims to summarize the current literature on testicular tissue dissociation/digestion protocols; the cell selection and enrichment strategies, including the post-selection characterization of the major testicular subtypes by morphology and cell-specific markers at both the gene and protein levels (Figure 2).
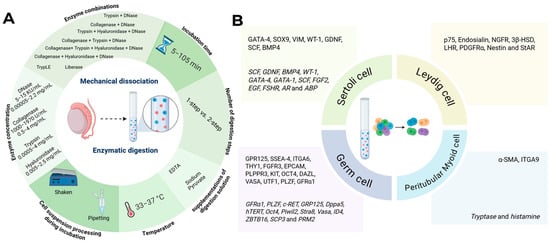
Figure 1.
Graphical overview of the methods and markers used in the included papers. (A) Mechanical dissociation/enzymatic digestion variables including incubation time, supplementations of digestion solution, temperature, enzyme combinations and concentrations, number of digestion steps and cell suspension processing during incubation. Variables are presented in ranges if applicable. (B) Markers for testicular cell positivity, at protein and gene-level (italics) used during cell selection and downstream verification. Created in BioRender, https://BioRender.com/2c3f9cg.
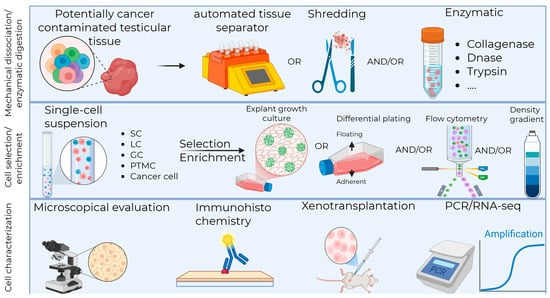
Figure 2.
Testicular tissue mechanical dissociation/enzymatic digestion protocols and cell selection/enrichment strategies for downstream fertility restoration strategies. SC, Sertoli cell; LC, Leydig cell; GC, germ cell; PTMC, Peritubular Myoid cell; PCR, polymerase chain reaction; RNA-seq, ribonucleic acid sequencing. Created in BioRender, https://BioRender.com/50ykhfj.
2. Materials and Methods
2.1. Study Design
This is a scoping review written via the PRISMA guidelines and published protocol in the Open Science Framework network (https://osf.io/82mp4/ (accessed on 9 September 2025)) [31].
2.2. Literature Search
Medline and its electronic database PubMed were used to search articles related to the topic of the review, using the queries listed in Table 2. All research publications published until 31 May 2025 were included. Forward and backward snowballing was applied to identify extra articles of interest in references and citations, respectively.

Table 2.
Overview of queries used for the systematic review.
2.3. Eligibility Criteria
Original research papers, written in English on experiments conducted on human testicular tissue by using the built-in search engine criterium were included. All reports containing testicular tissue dissociation, enzymatic digestion, selection or enrichment techniques, regardless of the final aim of the papers, were considered for inclusion. Exclusion criteria were articles published in languages other than English, reviews, guidelines, books, protocols without reporting results, research articles, and scientific video protocols. Papers reporting only outcomes on the selection/enrichment of differentiated GCs, being already standard of care in medically assisted reproduction, were not included. Any additional references found in original articles or review papers that were found relevant and missing from the primary search were added.
2.4. Study Selection
Title and abstract screening were performed by S.D.W. and N.N.A. in a blinded, independent, and separate way. These two searches were compared and discussed between S.D.W. and N.N.A. and in all cases, consensus was reached. The PRISMA flowchart illustrating the selection process of references is illustrated in Figure 3.
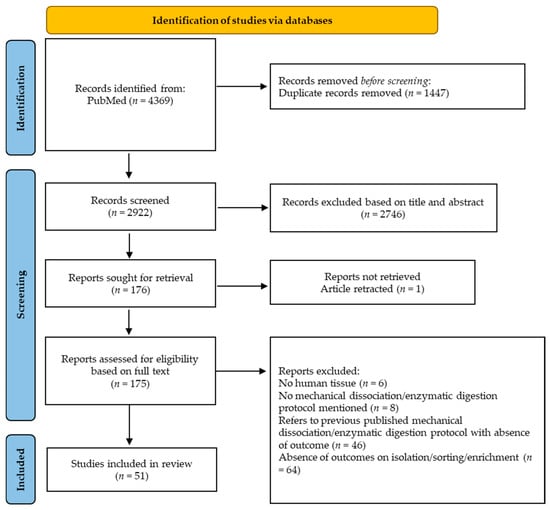
Figure 3.
General flowchart of article selection process.
2.5. Data Extraction and Interpretation
Full text screening was performed by S.D.W. and N.N.A. in a blinded and independent manner. EndNote (V.21.0.1.) and Excel (Microsoft, V2311 Build 16.0.17029.20177) were used for the full text screening process. The results were, again, discussed among S.D.W. and N.N.A., and in all cases, consensus was reached. The data that was extracted consisted of the mechanical dissociation/enzymatic digestion protocols and corresponding cellular yield and/or viability. For cell selection, the data extracted consisted of the selection strategy, markers, and purity outcomes with corresponding verification techniques. When cell viability or yield were not reported, those outcomes were calculated provided viable and death cell quantification numbers and weight of digested testicular tissue were mentioned.
3. Results and Discussion
In total, 4369 articles were identified from PubMed. After the removal of duplicate records, 2922 were screened based on title and abstract. A total of 51 studies fulfilled the eligibility criteria and were included in this scoping review. The papers are classified based on dissociation/digestion protocol applied and the cell type to sort/enrich. For each paper, the first author, year of publication, age of the donor, medical diagnosis/tissue source/anatomopathology, number of donors, tissue weight/size, storage of testicular tissue, dissociation/digestion protocol, and/or outcomes are mentioned.
3.1. Testicular Tissue Dissociation/Digestion to Obtain Single-Cell Suspensions
Different protocols of dissociation/digestion have been reported. While most publications used enzymatic digestion to obtain single-cell suspensions, some, mostly older publications, used mechanical dissociation. Mechanical dissociation can be described as dissociating tissue fragments by applying physical force ranging from simple shredding [32] to (automated) tissue separators [33,34,35]. Enzymatic digestion uses digestive enzymes to break down the ECM to obtain isolated seminiferous tubules followed by cleavage of cell–cell junctions, often carried out in multi-step protocols [36,37]. Both strategies have the objective to create a single-cell suspension with high viable cell numbers, minimal cell debris or aggregates, while simultaneously preserving cellular integrity including membrane receptors for downstream selection applications and cell functioning [36]. Single-cell suspensions can then either be used as a complete cellular mix or as suspensions of selected or enriched specific cell types.
A complete overview of reported mechanical dissociation/enzymatic digestion protocols with their main outcomes regarding cell yield and viability is provided in Table 3.
As numerous protocols are reported in publications that do not aim to focus on single-cell suspension outcomes, dissociation or digestion protocols are often vaguely described without specific information on start material characteristics and data on total or cell-type-specific viability and yield.
We identified only one publication in which mechanical dissociation and enzymatic digestion were compared while reporting specific outcomes such as overall cell yield, viability, and influence on testicular cell types [33]. An automated tissue separator called “the Medimachine” to mechanically dissociate fresh human adult testicular tissue was compared to a commonly applied two-step enzymatic digestion protocol using collagenase I, followed by trypsin in combination with DNase. Although the Medimachine approach provided considerable advantages regarding lab time and costs, the overall viability was higher for enzymatic digestion compared to mechanical dissociation, resulting in 76% and 38% viability when using fresh testicular tissue, and 74% and 16% when starting with cryopreserved testicular tissue, respectively. Interestingly, investigating the proportion of undifferentiated GCs demonstrated an increase in SALL4+ undifferentiated GCs when cryopreserved tissue and fresh tissue were mechanical dissociated compared to enzymatic digested with 26.92 ± 6.52% versus 27.33 ± 9.49% and 7.95 ± 4.38% versus 19.58 ± 7.88 SALL4+ cells, respectively [33].
Only few studies used mechanical dissociation to obtain single-cell suspensions from testicular tissue [33,35]. Kurpisz et al., 1988 [35], used a previously published mechanical isolation protocol [38] based on multiple chilled razor blades to dissociate and mince adult tissue. While the authors reported the suspension to contain 20–25% of Sertoli cells (SCs), they observed extensive SC damage. Somatic cells’ vulnerability to mechanical dissociation was also observed by Schneider et al., 2015 [33], using the “Medimachine” automated tissue separator as significant lower vimentin (VIM) expression was observed after mechanical dissociation compared to enzymatic digestion.
Enzymatic digestion was applied following a plethora of protocols including various types and concentrations of enzymes, one-step vs. multistep strategies, different incubation times and conditions considering the maturation stage of the tissue (fetal, pre- or peripubertal, adult), some other tissue specificities (diseased or healthy, less or more fibrotic) and the downstream applications, e.g., IVM, auto-transplantation of single-cell suspension. Different subtypes of the same enzyme also influence outcomes. Collagenase IV, a protease secreted by SCs [39] has less tryptic activity leading to a less harmful effect on membrane receptors compared to the less pure, more crude, collagenase I. Although a direct comparison on human ITT is still awaited, a study conducted on rat hepatocytes already identified collagenase IV to better preserve the activity of specific membrane receptors [40]. The superiority of collagenase IV (versus collagenase I) was also shown when used to extract round spermatids and spermatozoa from adult testicular tissue leading to a higher proportion of vital cells and less debris [41]. Despite these observations, collagenase I was used in recent studies, but outcomes of the enzymatic digestion were unfortunately not available [42].
3.1.1. Testicular Tissue Characteristics Influencing Testicular Dissociation/Digestion Outcomes
One of the most important characteristics of testicular tissue to be dissociated or digested is its prior cryopreservation or not, as cryopreservation can be seen as a first step of tissue dissociation. Cryopreservation can lead to membrane damage [43] especially for ITT proven to be twice as susceptible to this damage compared to adult tissue [43,44]. Indeed, significant differences between fresh and frozen/thawed testicular biopsies have been found both after mechanical dissociation and enzymatic digestion [33,45,46]. Zheng et al., 2014 [45], obtained significant lower overall cell yield when frozen/thawed tissue was enzymatically digested, though it is of note that all fresh tissue fragments were derived from the same patient, limiting the interpretation of these results. In line with their observation, lower viability (74.0% viability and 1.59 × 104 viable cells/mg) was also observed by Pacchiarotti et al., 2013 [46], after enzymatic digestion of frozen/thawed versus fresh tissue (90.1% viability and 4.25 × 104 cells/mg) from adult sexual reassignment biopsies. However, in his comparative study, Schneider et al., 2015 [33], obtained 16% and 38% living cells when mechanically dissociating adult human frozen/thawed and fresh testicular fragments, respectively, while no significant difference was observed for enzymatic digestion (74% and 76% viability, respectively, for adult human frozen/thawed and fresh testicular tissue).
3.1.2. Protocol-Related Factors Influencing Testicular Dissociation/Digestion Outcomes
While the vast majority of publications clearly report enzymatic digestion protocols with fixed incubation timings, only one evaluated the progress of digestion by microscopy and the physical presence of remaining testicular tissue and continued until all remaining tissue was digested. This study stopped the enzymatic digestion protocol when the contorted seminiferous tubules became soft and loose and released a “great number” of cells into the solution without specification about exact cell numbers [47]. This strategy resulted in 4.86 ± 1.19 × 106 cell yield after digestion with 91.07 ± 2.16% viability. Unfortunately, start material weight was not reported, thus hampering comparison with protocols using fixed incubation times [47].

Table 3.
Mechanical dissociation/enzymatic digestion protocols applied on human testicular tissue.
Table 3.
Mechanical dissociation/enzymatic digestion protocols applied on human testicular tissue.
| Mechanical Dissociation Applied to Human Testicular Tissue | |||||
| Reference | Age of Donor | Medical Diagnosis/Tissue Source/Anatomopathology Number of Donors (n) Tissue Weight/Size | Fresh/Frozen | Dissociation Protocol | Main Outcomes |
| Kurpisz et al., 1988 [35] | 32–38 years | Azoospermia n = NA | NA | Battery of chilled razor blades (automated) | Single-cell suspension containing spermatocytes, spermatids and 20–25% extensively damaged Sertoli cells |
| Schneider et al., 2015 [33] | NA | Sex reassignment surgery n = 76 (500 mg) | Fresh n = 6 Frozen n = 7 | Medimachine dissociation (automated) for 15 min | Mechanical dissociation of cryopreserved tissue resulted in significantly higher total cell yield compared to enzymatic digestion 38.04% vs. 15.72% viability after mechanical dissociation using fresh or cryopreserved tissue, respectively 75.93% vs. 73.99% viability after enzymatic digestion using fresh or cryopreserved tissue, respectively 2-fold lower spermatogonia (UTF1), 1.5-fold lower germ cell (MAGEA4), 2-fold lower Sertoli cell (VIM), and 5-fold lower Peritubular Myoid cell (ACTA2) expression when Medimachine dissociation was applied on fresh tissue compared to enzymatic digestion 7.95 ± 4.38% and 26.92 ± 6.52% SALL4+ cells when cryopreserved tissue was enzymatically digested compared to mechanical dissociation 19.58 ± 7.88% and 27.33 ± 9.49% SALL4+ cells when fresh tissue was enzymatically digested compared to mechanical dissociation |
| Enzymatic Digestion Applied to Human Testicular Tissue | |||||
| Reference | Age of Donor | Medical Diagnosis/Tissue Source/Anatomopathology Number of Donors (n) Tissue Weight/Size | Fresh/Frozen | Digestion Protocol | Main Outcomes |
| Berensztein et al., 1992 [48] | Prepubertal | Cadaver testis n = 7 | Fresh | Step 1: 5 KU/mL DNase + 1.182 × 103 U/mL collagenase at 37 °C for 20 min Sedimentation for 5 min Centrifugation Step 2: 15 KU/mL DNase + 1.970 × 103 U/mL collagenase at 37 °C for 20 min Filter 0.4 mm | 1.13 ± 0.40 × 108 cell yield/g after 1st step of digestion 2.45 ± 0.32 × 108 cell yield/g at the end of digestion |
| Brook et al., 2001 [49] | 22–35 years | Biopsy with normal spermatogenesis n = 11 Orchiectomy whole testes n = 8 | Fresh | Mincing (3 × 3 × 3 mm3) Step 1: 1 mg/mL collagenase I at 37 °C for 12–20 min Filter 120 µM Step 2: 6 µg/mL bovine pancreatic trypsin + 2 mM EDTA + 16 µg/mL ovine hyaluronidase + 0.4 µg/mL DNase I + 0.2 mM sodium pyruvate at 37 °C for 12–15 min 500 µg/mL Soybean trypsin inhibitor Centrifugation 500× g at 4 °C for 10 min | 66% viability after digestion |
| Liu et al., 2011 [47] | Fetal 6–7 months | Miscarriage n = Not available (NA) | Fresh | Mincing Step 1: 1 mg/mL collagenase I at 37 °C for 10 min Centrifugation Supernatant removed Step 2: 0.25% Trypsin at 37 °C for 10–15 min Centrifugation Filter 200 mesh | 4.86 ± 1.19 × 106 cell yield after digestion 91.07 ± 2.16% viability after digestion |
| Nowroozi et al., 2011 [50] | 25–52 years | Non-obstructive azoospermia (NOA) n = 47 (100–200 mg/fragment) | Fresh | 0.5 mg/L collagenase + 0.5 mg/L Trypsin + 0.5 mg/L hyaluronidase + 0.05 mg/L DNase for 20 min at 37 °C with agitation | 93.40 ± 5.04% viability after digestion |
| Izadyar et al., 2011 [51] | NA | Obstructive azoospermia (OA) testicular sperm extraction (TESE) n = 29 Orchiectomy with normal spermatogenesis n = 2 | Fresh | Step 1: 1 mg/mL collagenase A + 10 U/mL DNase at 37 °C for 15 min Gravity sedimentation and discard of supernatant Step 2: 1.5 mg/mL collagenase A + 1.5 mg/mL hyaluronidase V + 0.5 mg/mL Trypsin + 10 U/mL DNase at 37 °C for 20 min Filter Centrifuge 400× g for 10 min | 0.5 × 106 cell yield after digestion 87% viability after digestion |
| Mirzapour et al., 2011 [52] | 28–50 years | Azoospermia (maturation arrest) n = 20 (100–200 mg/patient) | Fresh | Step 1: 0.5 mg/mL Trypsin + 0.5 mg/mL Hyaluronidase + 0.05 mg/mL DNase at 37 °C for 20 min Centrifugation 112 Relative centrifugal force (RCF) for 4 min Wash Dulbecco’s modified eagle medium (DMEM) Step 2: 0.5 mg/mL Trypsin + 0.5 mg/mL Hyaluronidase + 0.05 mg/mL DNase at 37 °C for 5 min Centrifugation 542 RCF for 4 min at 37 °C Filter 70 µM nylon | 93.40 ± 5.04% viability after digestion |
| Koruji et al., 2012 [53] | 32–50 years | NOA TESE n = 20 | Fresh | Mincing Step 1: 1 mg/mL collagenase I + 1 mg/mL Hyaluronidase + 1 mg/mL Trypsin + 0.05 mg/mL DNase at 37 °C for 30 min with shaking and pipetting Centrifugation 2 min 1100 revolutions per minute (rpm) 3 × wash in DMEM Step 2: repetition step 1 for 30–45 min Filter 40 µM | ≥92% viability after 24 h differential plating |
| Riboldi et al., 2012 [54] | NA | OA n = 9 NOA TESE n = 11 | Fresh | Mincing (1 mm3) Step 1: 1000 IU/mL collagenase IA for 20 min at 37 °C on shaker Step 2: TrypLE select for 10 min at 37 °C on shaker Filter 50 µM Centrifugation 1000 rpm for 5 min | 75% viability after digestion OA: 4.16 ± 4.90 × 106 cell yield after digestion NOA: 1.99 ± 1.97 × 106 cell yield after digestion |
| Zohni et al., 2012 [55] | 39–50 years | OA n = 18 (mean biopsy weight 67.1 ± 8.3 mg/patient) | Fresh | Step 1: 1 mg/mL collagenase I + 1 mg/mL collagenase IV + 1 mg/mL Hyaluronidase + 1 mg/mL DNase I at 33 °C for 15 min with periodic shaking Centrifugation 500× g for 5 min Step 2: 0.5 mg/mL Trypsin + 1 mg/mL DNase I at 33 °C for 5 min | 8.6 ± 0.4 × 104 cell yield/mg after digestion 5.8 × 106 cell yield/patient 95.5 ± 1.7% viability after digestion |
| Pacchiarotti et al., 2013 [46] | 25–40 years | Sex reassignment surgery n = 5 | Fresh Frozen | Mincing Liberase (0.3 U/mL collagenase I and II + 1000 U/mL Thermolysin) at 37 °C shaking 110 RPM for 1.75 h Filter 100 µm centrifugation 400 × g for 5 min at 4 °C | Fresh: 42.5 ± 9.3 × 106 cell yield/g with 90.1 ± 1.3% viability after digestion Fresh: 0.6 ± 0.1 × 106 spermatogonia stem cell (SSEA-4+), 1.6 ± 0.5 × 106 Leydig cell (LHR+) and 16.6 ± 4.1 × 106 germ cell (VASA+) cell yield/g after digestion Frozen 15.9 ± 4.4 × 106 cell yield/g with 74.0 ± 2.2% viability after digestion Frozen: 0.3 ± 0.1 × 106 Spermatogonial stem cell (SSEA-4+), 22 ± 0.9 × 106 Leydig cell (LHR+) and 10.9 ± 3.3 × 106 germ cell (VASA+) cell yield/g after digestion |
| Kossack et al., 2013 [56] | NA | Biopsy with normal spermatogenesis n = 4 Klinefelter patients n = 3 | Fresh | Step 1: 1 mg/mL collagenase IA at 37 °C for 30 min Centrifugation 438 × g for 5 min Remove supernatant Step 2: 4 mg/mL Trypsin + 2.2 mg/mL DNase I for 10 min at 37 °C | 2.91 ± 1.21 × 106 cell yield after digestion of normal spermatogenesis samples 2.87 ± 2.03 × 106 cell yield after digestion of Klinefelter patient samples |
| Zheng et al., 2014 [45] | 13–40 years | Cadaver testis n = NA (0.5–2 g/experiment) | Fresh Frozen | Mincing Step 1: 1 mg/mL collagenase IV + 0.7 mg/mL DNase in HBSS at 37 °C for 15 min Step 2: 0.25% Trypsin/EDTA + 0.7 mg/mL DNase in HBSS at 37 °C with periodic rocking for 5 min Filter 40 µM | Fresh: 29 ± 16 × 106 cell yield/g after digestion Frozen: significant lower yield after digestion, exact cellular yield not reported |
| Guo et al., 2015 [57] | 22–35 years | OA n = 50 | Fresh | Step 1: 2 mg/mL collagenase IV + 1 µg/mL DNase I at 34 °C for 15 min Step 2: 4 mg/mL collagenase IV + 2.5 mg/mL hyaluronidase + 2 mg/mL trypsin + 1 µg/mL DNase I | ≥98% viability after overnight differential plating |
| Jabari et al., 2023 [58] | 15, 21, and 26 years old | Cadaver testis n = 3 | Fresh | Mincing Step 1: 1 mg/mL collagenase I + 1 mg/mL hyaluronidase + 1 mg/mL Trypsin + 0.05 mg/mL DNase at 37 °C with 150 cycles/min shaker for 30 min Centrifugation 1100 rpm for 4 min Wash in DMEM Step 2: Repetition step 1 for 25 min Filter 40 µm | >91% viability after digestion |
| Nikmahzar et al., 2023 [59] | 28, 32, and 44 years | Cadaver testis n = 3 | Fresh | Step 1: 1 mg/mL collagenase IV + 1 mg/mL hyaluronidase at 37 °C for 10 min at 150 cycles/min shaken Centrifugation at 1100 rpm for 10 min Step 2: 1 mg/mL collagenase + 0.5 mg/mL DNase I + 1 mg/mL hyaluronidase at 37 °C for 10 min Filter 100 µM and 40 µM | 70% viability after digestion |
ACTA2, actin alpha 2; DMEM, Dulbecco’s modified eagle medium; EDTA, ethylenediaminetetraacetic acid; HBSS, Hanks’ balanced salt solution; IU, international unit; KU, Kunitz unit; LHR, luteinizing hormone receptor; MAGEA4, melanoma-associated antigen 4; NA, not available; NOA, non-obstructive azoospermia; OA, obstructive azoospermia; RCF, relative centrifugal force; RPM, revolutions per minute; SALL4, Sal-like protein 4; SSEA-4, stage-specific embryonic antigen-4; TESE, testicular sperm extraction; UTF1, undifferentiated embryonic cell transcription factor 1; VIM, vimentin.
3.2. Testicular Cell Selection/Enrichment, and Characterization
Selection or enrichment of testicular cells is essential for research purposes, as the human testis contains a highly heterogenous population of SCs, GCs, Leydig cells (LCs), and Peritubular Myoid cells (PTMCs) each at different developmental stages. As some cell types are scarce, and can be masked by more abundant cell subpopulations, obtaining the purest fraction of a certain cell of interest is pivotal for downstream applications. In particular, the cells of focus for fertility restoration approaches, the undifferentiated GCs, are present in very low proportions ranging from 1% undifferentiated GCs in human ITT from newborns of 2–7 days old [60] to 3–4% in the biopsy of a 1-year- and 7-year-old boy [61].
Table 4 summarizes selection/enrichment strategies and their outcomes for all major testicular cell types.
3.2.1. Sertoli Cells
Many studies have tried to isolate the SC, a known key player in both the architecture and the function of the testes. Among these, SCs are known to regulate the formation of the blood–testis barrier through functional and dynamic tight junctions creating an exclusive immunogenic environment to protect GCs from own immune cell attacks [62]. Apart from aiming to isolate SCs to study their morphology or transcriptome, this is also performed to use them as feeder cells, as spermatogonia require feeder-cell-based substrates to promote their survival and proliferation in vitro [47,52,63]. Methods identified in the literature to isolate SCs are based on density gradients, differential plating, flow cytometry or a combination of techniques to further purify the obtained SC fraction.
Selection/Enrichment of SCs by Density Gradient Separation
Silica-based gradient separation preparations such as Percoll are commonly used in cell selection applications. Percoll uses silica particles coated with polyvinylpyrrolidone to create a low viscosity density gradient medium in which cells can be separated after centrifugation [47,64]. SCs, identified morphologically, were isolated from 6-to-7-month-old fetal ITT by recuperating the bands of 19–27% and 35–43% Percoll. Unfortunately, the efficiency of the technique remains unknown as neither cellular yield nor viability were reported [47].
Selection/Enrichment of SCs by Differential Plating
SCs are well characterized by their ability to adhere to (un)-coated plastics, a characteristic that GCs possess with much less affinity [65]. Therefore, most studies aiming to isolate or enrich high-purity SC fractions perform differential plating strategies by separating adherent and floating cell fractions. An attachment rate of 63.33 ± 8.76% [65] and a mean number of adherent cells of 1.93 ± 1.16 × 105 somatic cells were obtained with adult tissue [66]. Enzymatic digestion of adult obstructive azoospermia (OA) testicular biopsies followed by 24 h incubation onto plastic in standard medium-separated floating GCs from adherent SCs. This approach allowed the enrichment of SCs by 95% purity and 98% viability [57]. Another study incubating the single-cell suspension for 48 h in standard medium characterized the SCs morphologically and concluded 90% SC purity with over 95% viability [67]. Mirzapour et al., 2011 [52], used coated plastics to even further facilitate the adherence of SCs by using Datura Stramonium Agglutinin (DSA)-coated plastics and while purity reached 95%, it did not differ significantly from uncoated plastic differential plating.
Selection/Enrichment of SCs by Combinatory Strategies
Adult human SCs could be successfully isolated after multiple enzymatic digestion steps, centrifugations, multiple gravity sedimentation steps with supernatant discarded in each step and following long-term culture. The fractions were analyzed based on morphology [68,69] or based on SC-specific marker identification resulting in a pure SC fraction of 95% SCs [70] with viability ranging from 75 to 95%.
3.2.2. Leydig Cells
LCs are found adjacent to the seminiferous tubules and act to stimulate spermatogenesis by producing androgens [71]. Only one study examined the theoretical yield of LCs after enzymatic digestion. The average theoretical number of LCs per adult testis was calculated to be 139.12 ± 81.89 × 106, with minimum and maximum values of 43.20 × 106 and 351.28 × 106, respectively [72], which account for 2–4% of the total testicular cell population [73].
Selection/Enrichment of LCs by Density Gradient Separation
Discontinuous Percoll gradients are commonly used to isolate and enrich LCs from enzymatically digested single-cell suspensions [72,74,75,76]. Only one study investigated the purity of LCs in all 3 layers formed between 25 and 70% and showed a 3β-HSD+ LC enrichment of 12–28%, 48–70%, and 30–56% in band 1 (25–45% Percoll), 2 (45–60% Percoll), and 3 (60–70% Percoll), respectively [74]. Despite having the highest enrichment of LCs in band 2, it only recovered 4–18% of all LCs, as 77–95% of all LCs are found in band 1 [74]. Another study collected the 34% and 40% bands, yielding a 95% enriched 3β-HSD+ viable LC suspension. Although the exact purity was not reported, they claimed to have obtained 500.000 LCs/mL [75]. When collecting all cells between 34 and 60%, a highly viable (±94%) LC-enriched fraction was obtained ranging from 80 to 83% 3β-HSD+ LCs. Of note, they identified a difference in staining intensity among the same LC-enriched suspensions indicating a heterogeneity among the LC populations [76]. Another study recovered LCs in the interphase of 35–60% Percoll and yielded 60–77% pure 3β-HSD+ LCs [72]. Furthermore, one study identified a recovery of only 6–12% of the total LC population by collecting all bands formed between 25 and 70% Percoll [74].
Selection/Enrichment of LCs by Flow Cytometry
To obtain LCs by sorting, some promising membrane markers have been identified. In their study, Xia et al., 2020 [77] successfully isolated stem Leydig cells (sLCs) by fluorescent-activated cell sorting (FACS), using the presence of endosialin receptors. The endosialin+ sorted cells account for 0.31 ± 0.03% of the total cell population, though purity was not reported. After one week of culture, 99.30 ± 0.32% of the FACS-sorted cells were still endosialin+. Characterization of the obtained endosialin+ fraction after one week of culture revealed a purity of ≥98% using well-established LC markers such as PDGFR-α, NGFR, and Nestin. Furthermore, these endosialin+ cells were xenotransplanted for 4 weeks and were exclusively found in the interstitial area keeping their PDGFR-α expression, while a small proportion of the grafted cells expressed adult LC markers (3β-HSD, CYP11A1, CYP17A1, and StAR), indicating their differentiation potential into LCs.
3.2.3. Peritubular Myoid Cells
Testicular PTMCs are smooth, muscle-like cells that surround the seminiferous tubules forming the blood–testis barrier together with SCs and will support sperm movement through contractile activity [78]. Only a few studies attempting to isolate and obtain highly pure PTMC fractions are reported. Thanks to their migration capacities, a unique selection method, not reported for other testicular cell types, the explant growth culture or tubule crawling method, has been applied to isolate PTMCs from testicular tissue [79,80,81,82].
Selection/Enrichment of PTMCs by Explant Growth Culture
In this approach, testicular fragments are cultured for 1 to 3 weeks in defined medium containing recalcified human serum to promote cell migration allowing PTMCs to grow out of the biopsies and attach to the culture plate [81,82,83,84]. Results suggested that the PTMC fraction host putative sLCs as 79–90% of all isolated cells expressed sLC marker PDGFRα and pluripotency marker Nanog of which almost all had positive PTMC marker α-SMA and LC marker StAR staining; this finding was further confirmed by reverse transcription polymerase chain reaction (RT-PCR) in four independent human adult donors using sLC and PTMC markers [82]. This indicates that explant growth culture inevitably leads to contamination with other somatic cells and thus is unable to obtain highly pure PTMCs [79].
Selection/Enrichment of PTMCs by Flow Cytometry
To overcome somatic cell contamination, Han et al., 2025 [79], identified ITGA9 as being only expressed on PTMCs while NGFR is a commonly expressed membrane receptor for both adult LCs and PTMCs, opening the way for flow cytometry. Indeed, by sorting the ITGA9+/NGFR+ fraction, this study successfully isolated ≥95% pure PTMC fractions being positive for well-characterized PTMC markers in combination with the absence of LC and SC markers.
3.2.4. Germ Cells
Different approaches such as magnetic-activated cell sorting (MACS), differential plating, flow cytometry, and Percoll gradient have been applied to isolate and select SSCs and undifferentiated GCs. To date, no SSC-specific marker has been identified for any species, but the combination of multiple markers is able to identify spermatogonial cell types in the human testis of different maturation statuses. Thanks to improvement in sequencing assays, nowadays, analysis at the single-cell level revealed some promising new markers for putative SSCs [85,86,87].
Selection/Enrichment of SSCs by MACS
To date, SSCs can only be identified using the xenotransplantation assay with downstream verification of colonization capacities of SSCs. A proof of principle can be found in the study of Nickkholgh et al., 2014 [88], in which they tested SSC enrichment after a 50 day culture and MACS by analyzing colony formation efficiency after xenotransplantation. They showed that ITGA6+ sorted cells had the highest number of colony formations compared to Human Leukocyte Antigen class I (HLA)−/GPR125+, GPR125+, and HLA−/ITGA6+ with 7.1; 3.9; 3.9 and 2-fold SSCs enrichment, respectively. Using cell number (not number of colonies formed) as an outcome, other studies have shown that MACS for the cell-surface protein SSEA-4, resulting in 90.2% SSEA-4+ cells, enriches human adult testicular cells for SSC activity by ≥40-fold [51].
Selection/Enrichment of SSCs by Flow Cytometry
Recently, a comparative RNA-sequencing revealed a 38-fold SSC activity in PLPPR3+ FACS-sorted fractions compared to unsorted testicular fractions. However, PLPPR3 was identified to be a transmembrane protein with half of the PLPPR3+ cells having PLPPR3 present on the membrane which limits the use of PLPPR3 to sort SSCs [89].
Selection/Enrichment of GCs by Differential Plating
Separation of GCs by their weak ability to attach to uncoated plastic is a common approach to obtain GC-enriched fractions [52,58,65,90,91]. In their study, Medrano et al., 2016 [65], investigated the number of adult spermatogonia per cm2 in unsorted and 24 h-incubated differential plated fractions. They found an enrichment of GCs in the unattached differential plated fraction compared to the attached differential plated fraction with 50 and 8 VASA+/UTF1+ undifferentiated GCs/cm2, respectively. Other approaches used protein-coated plastic dishes, such as the commonly used DSA-coating, to facilitate the adherence of somatic testicular cells leading theoretically to higher enriched floating GC fractions. As incubating single-cell suspensions for 2–3 h led to a 95% pure GC fraction by recovering the floating cell fraction, this indicates that protein-coated dishes did not result in better outcomes [52].
Selection/Enrichment of GCs by Flow Cytometry
To achieve undifferentiated GC selection/enrichment, several cell-specific markers have been used. Among those, epithelial cell adhesion molecule (EPCAM) and HLA resulted in the promising enrichment of putative SSCs. In the study of Medrano et al., 2016 [65], cryopreserved adult testicular cells were enzymatically digested and a FACS-sorted GC enriched fraction was compared with an unsorted single-cell suspension. The comparison revealed that GC enrichment by HLA negative/EPCAM positive resulted in an enrichment of VASA+/UTF1+ human putative SSCs by 14%; unfortunately, no xenotransplantation assay was performed to further characterize the obtained cells.
Selection/Enrichment of GCs by MACS
Alternatively to FACS, sorting cells by magnetic bead pre-labeling of the cell of interest proved to be effective. He et al., 2010 [92], isolated GPR125+ putative SSCs from adult donors by MACS and obtained 95% pure GPR125+ fractions which showed after 2-week culture a 5-fold increase in cell numbers retaining ITGA6+, GFRA1+, and THY1+ expression and phenotypical characteristics of putative human SSCs.
Selection/Enrichment of GCs by Combinatory Approaches
As all these flow cytometric sorting strategies did not lead to optimal outcomes, further effort was put into obtaining GC-sorted fractions. Von Kopylow et al., 2016 [93], sorted patient-derived single-cell suspensions from adult tissue containing normal spermatogenesis and maturation arrest by MACS followed by micromanipulation cell-picking of only the FGFR3+ fraction. A 100% pure putative SSC population could be obtained with 100% of the picked cells expressing UTF1 and 95% viability, though stem cell characteristics have not been tested and not all UTF1+ cells were recovered as the negative sorted fraction still contained 1% UTF1+ cells. Furthermore, human fetal undifferentiated GCs could be isolated using Percoll density gradient separation by recovering all cells in layers formed between 27 and 35% in combination with 24 h culture and followed by flow cytometry. This combinatory approach resulted in 86.7% purity confirmed by the presence of transcription factor membrane marker OCT4 [47]. Alternatively, STA-PUT enrichment based on sedimentation and microscopical evaluation was found to lead to promising viability and purity of enriched testicular cell suspensions. Testes from OA patients were enzymatically digested and GCs were first enriched by differential plating. This enriched single-cell suspension was further loaded on the STA-PUT gravity apparatus to separate and enrich all GC types. In brief, the GC-enriched single-cell suspension was mixed with multiple bovine serum albumin concentrations to form a gradient. At the end, 45 fractions each containing cells of similar size were formed and fractions containing cells with similar morphology were pooled manually to separate different GC types. In general, spermatogonia could be enriched with a purity of 90% based on immunohistochemistry and RT-PCR using well-characterized specific undifferentiated GC markers and led to a viability ≥98%. Spermatogonia, microscopically identified as cells with spherical, large round or ovoid nuclei, diameter 9–12µM and high ratio of nucleus to cytoplasm, were found in fraction 10–15 [94].

Table 4.
Selection/enrichment strategies applied on human testicular tissue.
Table 4.
Selection/enrichment strategies applied on human testicular tissue.
| Sertoli Cell Selection/Enrichment Strategies Applied on Human Testicular Tissue | |||||
| Reference | Age of Donor | Medical Diagnosis/Tissue Source/Anatomopathology Number of Donors (n) Tissue Weight/Size | Fresh/Frozen | Selection/Enrichment Strategy After Enzymatic Digestion + Verification Technique | Main Outcomes |
| Lipshultz et al., 1982 [69] | Adult | Sex reassignment surgery n = NA | Fresh | Differential plating and culture for 45 days Morphological identification | >95% pure Sertoli cell culture after 45 days 75–85% cell viability after 45 days |
| Teng et al., 2005 [67] | 28–42 years | Cadaver testis n = NA | Fresh | Differential plating and culture for 28 days Morphological identification | >90% pure Sertoli cell culture after 28 days >95% cell viability after 28 days |
| Chui et al., 2011 [68] | 12–36 years | Cadaver testis n = 7 | Fresh | Differential plating and culture for 20 days Morphological identification + Flow cytometry (GATA-4/SOX9) + RT-PCR (SCF, GDNF and BMP4) | ≥95% pure Sertoli cells expressing Sertoli cell (SCF, GDNF, and BMP4) markers with 90% viability after 20 days culture |
| Mirzapour et al., 2011 [52] | 28–50 years | Azoospermia (maturation arrest) n = 20 (100–200 mg/sample) | Fresh | Differential plating to (un)-DSA coated dishes and culture for 72 h ICC (VIM) | >95% pure Sertoli cells after 72 h culture No statistically significant difference in Sertoli cell purity when using DSA-coated dishes compared to uncoated dishes |
| Riboldi et al., 2012 [54] | NA | Obstructive azoospermia (OA) n = 9 Non-obstructive azoospermia (NOA) TESE n = 11 | Fresh | Differential plating and culture | 95% pure Sertoli cell after differential plating |
| Guo et al., 2015 [57] | 22–35 years | OA n = 50 | Fresh | Differential plating and overnight culture RT-PCR (WT-1, GATA-4, GATA-1, GDNF, BMP4, SCF, FGF2, EGF, FSHR, AR and ABP) + ICC (WT-1, GDNF, SCF, BMP4, VIM, PCNA, and GATA-4) | 98% Sertoli cell viability after overnight culture 95% pure Sertoli cell culture <5% of the enriched cells expressed Peritubular Myoid cells (α-SMA) or Leydig cell (CYP11A1) markers |
| Gaur et al., 2018 [70] | 26–56 years | Cadaver testis n = 5 | Fresh | Multiple gravity sedimentation steps ICC (GATA-4) | >95% pure Sertoli cell culture |
| Leydig Cell Selection/Enrichment Strategies Applied on Human Testicular Tissue | |||||
| Reference | Age of Donor | Medical Diagnosis/Tissue Source/Anatomopathology Number of Donors (n) Tissue Weight/Size | Fresh/Frozen | Selection/Enrichment Strategy after Enzymatic Digestion + Verification Technique | Main Outcomes |
| Simpson et al., 1987 [74] | 57–85 years | Orchiectomy for prostatic carcinoma n = 10 (Testis fragment weight used 5–8 g/patient) | NA | Discontinuous Percoll gradient Immunohistochemistry (3β-HSD) | After DPG 3 bands were obtained Band 1: 95–97% of all testicular cells, 12–28% pure Leydig cells accounting for 77–95% of all Leydig cells, 10.6–16.6 × 106 Leydig cell yield Band 2: 2–4% of all testicular cells, 48–70% pure Leydig cells accounting for 4–18% of all Leydig cells, 0.7–5.7 × 106 Leydig cell yield Band 3: 0.7–1.5% of all testicular cells, 30–56% pure Leydig cell yield accounting for 1–6% of all Leydig cells, 0.3–1.9 × 106 Leydig cell yield Overall, 11.6–24.2 × 106 Leydig cell yield |
| Qureshi et al., 1993 [72] | 54–89 years | Orchiectomy for prostatic carcinoma n = 27 (Testes paired weight 6.6–59.48 g) | Fresh | Discontinuous Percoll Gradient Immunohistochemistry (3β-HSD) | 60–77% pure Leydig cells obtained after DPG 139.12 ± 81.89 × 106 Leydig cell yield/testis |
| Sivakumar et al., 2006 [75] | 60–70 years | Orchiectomy for prostatic carcinoma n = NA | Fresh | Discontinuous Percoll Gradient Immunohistochemistry (3β-HSD) | 95% cell viability after DPG 0.5 × 106 Leydig cell yield/mL after DPG |
| Bilinska et al., 2009 [76] | 60–67 years | Orchiectomy for prostatic carcinoma n = 4 | Fresh | Discontinuous Percoll Gradient Morphological identification ICC (3β-HSD/LHR) | DPG 34–60%: 80–83% pure Leydig cells 94% Leydig cell viability |
| Zhang et al., 2017 [73] | 18 and 19 years 23, 25, 28, 32 years | Cadaver testis n = 2 OA n = 4 | NA | FACS (p75) | p75 sorted stem Leydig cells account for 1.79% of the total cell population. |
| Xia et al., 2020 [77] | 56–60 years 57–67 years | Cadaver testis n = 2 Orchiectomy for prostatic carcinoma n = 2 | NA | FACS (Endosialin) ICC (PDGFRα, NGFR, and Nestin) | Endosialin+ sorted stem Leydig cells accounted for 0.31 ± 0.03% of the entire cell population with >98% expressing Leydig cell (PDGFRα, NGFR, and Nestin) markers |
| Han et al., 2025 [79] | NA | OA TESE n = 3 | Fresh | FACS (ITGA9/NGFR) ICC (3β-HSD, α-SMA, StAR) | ITGA9+/NGFR−sorted cells account for 0.2% of the total cell population, ITGA9+/NGFR+ sorted cells account for 0.68% of the total cell population and 95% expressed Peritubular Myoid cell (α-SMA) marker ITGA9−/NGFR + sorted cells were Leydig cell (3β-HSD, StAR) marker positive |
| Peritubular Myoid Cell Selection/Enrichment Strategies Applied on Human Testicular Tissue | |||||
| Reference | Age of Donor | Medical Diagnosis/Tissue Source/Anatomopathology Number of Donors (n) Tissue Weight/Size | Fresh/Frozen | Selection/Enrichment Strategy + Verification Technique | Main Outcomes |
| Albrecht et al., 2006 [81] | 29, 32, 32, 34, 35, 36, 40, 41, 46, 47 years | OA with normal spermatogenesis n = 8 Varicocele with slightly reduced spermatogenesis n = 2 | Fresh | Explant growth culture Morphological identification + immunohistochemistry (FSH, LH-r, α-SMA, THY1) + RT-PCR (Tryptase, Histamine) | Peritubular Myoid cells become visible after 1–2 weeks and expressed Peritubular Myoid cell (α-SMA, Tryptase and Histamine) and germ cell (THY1) markers while not expressing Sertoli cell (FSH) and Leydig cell (LH-r) markers |
| Landreh et al., 2014 [82] | 31–52 years | OA NOA n = NA | NA | Explant growth culture Immunohistochemistry (PDGFR-α, α-SMA, StAR) | Almost all outgrowth cells expressed Peritubular Myoid marker (α-SMA) and Leydig cell (StAR) marker and 79–90% of them were Leydig cell (PDGFR-α) marker positive |
| Rolland et al., 2019 [84] | Average 51 years Average 80 years | Cadaver testis n = 8 Orchiectomy n = 9 | NA | Explant growth culture | Peritubular Myoid cells grow out after 2–3 weeks |
| Han et al., 2025 [79] | NA | OA TESE normal spermatogenesis n = 3 | Fresh | FACS (ITGA9/NGFR) ICC (3β-HSD, α-SMA, StAR) | ITGA9+/NGFR−sorted cells account for 0.2% of the total cell population, ITGA9+/NGFR+ sorted cells account for 0.68% of the total cell population and 95% expressed Peritubular Myoid cell (α-SMA) marker ITGA9−/NGFR+ sorted cells were Leydig cell (3β-HSD, StAR) marker positive |
| Germ Cell Selection/Enrichment Strategies Applied on Human Testicular Tissue | |||||
| Reference | Age of Donor | Medical Diagnosis/Tissue Source/Anatomopathology Number of Donors (n) Tissue Weight/Size | Fresh/Frozen | Selection/Enrichment Strategy after Enzymatic Digestion + Verification Technique | Main Outcomes |
| He et al., 2010 [92] | 16–58 years | Cadaver testis n = 5 | Fresh | Differential plating + MACS (GPR125) ICC (GPR125) | 6 × 106 germ cell yield/g after differential plating 3 × 104 germ cell yield after MACS >95% pure germ cell after MACS cells could be proliferated 5-fold during 14-day culture while retaining phenotypical characteristics of SSCs |
| Liu et al., 2011 [47] | Fetal 6–7 months | Miscarriage n = NA | Fresh | Discontinuous Percoll gradient followed by 3 h-differential plating ICC (SSEA-4, OCT4) | 86.7% pure OCT4+ germ cell after DPG and differential plating of which the majority expressed germ cell marker SSEA-4 |
| Izadyar et al., 2011 [51] | NA | OA TESE n = 29 Orchiectomy with normal spermatogenesis n = 2 | Fresh | MACS (SSEA-4) RT-PCR (c-KIT, GFRα1, PLZF, c-RET, GPR125, Dppa5, and hTERT) + Xenotransplantation | SSEA-4+ sorted germ cells account for 13.3 ± 1.4% of the entire single-cell suspension and had significantly higher expression of SSC-specific genes compared to SSEA-4- sorted cells SSEA-4+ sorted cells showed 40–50-fold HNP+ cells after xenotransplantation compared to unsorted single-cell suspension xenotransplantation |
| Mirzapour et al., 2011 [52] | 28–50 years | Azoospermia (maturation arrest) n = 20 (100–200 mg/patient) | Fresh | Differential plating to DSA-coated dishes for 2–3 h and culture ICC (OCT4) + RT-PCR (Oct4, Nanog, Piwil2, Stra8, Vasa, Bax, and DMC1) + Xenotransplantation | 95% pure germ cell after differential plating and SSC colonies expressed key germ cell markers after 2 weeks of culture |
| Nickkholgh et al., 2014 [88] | NA | Orchiectomy (prostate cancer) n = 2 | Frozen | Culture for 50 days followed by MACS (GPR125 or ITGA6 in combination with HLA) qRT-PCR (ID4) + Xenotransplantation | 5.3 ± 3.8% ITGA6+, 1.99 ± 1.5% HLA−/ITGA6+, 1.89 ± 0.9% GPR125+, and 2.33 ± 0.7% HLA−/GPR125+ after MACS higher expression of undifferentiated germ cells (ID4) in ITGA6+ and HLA−/GPR125+ sorted fraction Xenotransplantation: No significant difference among GPR125+/HLA− or ITGA6+/HLA− or GPR125+ or ITGA6+ MACS sorted fractions regarding SSC colony formation |
| Smith et al., 2014 [95] | Adult | Normal spermatogenesis n = 13 | NA | FACS (SSEA-4, THY1) RT-PCR (ZBTB16, GFRa1), ICC (DAZL, VASA) | SSEA-4+ sorted germ cells expressed undifferentiated germ cell (DAZL, VASA) markers and had significantly higher undifferentiated germ cell (1.9-fold ZBTB16, 10-fold GFRa1, and 3-fold GPR125) marker expression compared to THY1+ sorted germ cell fractions |
| Von Kopylow et al., 2016 [93] | Adult | OA with normal spermatogenesis n = 37 Meiotic arrest n = 3 (30 mg/testis) | Fresh | MACS (FGFR3) + Micromanipulation cell-picking Morphological identification + ICC (UTF1) | 54–138 and 220–280 UTF1+ undifferentiated germ cell yield after micromanipulation in normal spermatogenesis and meiotic arrest patients, respectively 100% pure UTF1+ undifferentiated germ cells compared to 1–2% UTF1+ cells in unsorted control with viability 95% |
| Medrano et al., 2016 [65] | Adult | Bilateral orchiectomy (prostate cancer, normal spermatogenesis) n = 3 | Frozen | Differential plating 24 h or FACS (HLA−/EPCAM+) ICC (VASA/UTF1) | 27% pure VASA+/UTF1+ undifferentiated germ cells in sorted compared to 13% unsorted fraction 112 undifferentiated germ cell/cm2 yield compared to unsorted cells and differentially plated cells 61 and 49 undifferentiated germ cells/cm2, respectively 50 and 8 VASA+/UTF1+ undifferentiated germ cells/cm2, in floating and adherent fraction after 24 h differential plating, respectively |
| Tan et al., 2020 [89] | 30–50 years | vasectomy reversal n = 29 | Frozen | FACS (PLPPR3 or KIT) Xenotransplantation | Xenotransplantation: 38-fold SSC-activity enrichment of PLPPR3+ SSCs compared to unsorted cells |
| Salem et al., 2023 [91] | 22, 25, and 28 years | Cadaver testis n = 3 | Fresh | Differential plating Morphological identification + ICC (PLZF, GFRA1) + RT-PCR (GFRA1, PLZF, SCP3, and PRM2) + xenotransplantation | GFRA1+ and PLZF+ SSC colonies were visible after 2 weeks of culture Xenotransplantation: SSC colonies were positive for undifferentiated germ cell markers (PLZF and GFRA1), and differentiated germ cell markers (SCP3 and PRM2) after 8 weeks |
3β-HSD, 3 beta-hydroxysteroid dehydrogenase; ABP, androgen binding protein; AR, androgen receptor; α-SMA, alpha-smooth muscle actin; BAX, BCL2 associated X; BMP4, bone morphogenetic protein 4; CYP11A1, cytochrome P450 family 11 subfamily A member 1; DAZL, deleted in azoospermia-like; DMC1, DNA Meiotic recombinase 1; DPG, discontinuous Percoll gradient; DPPA5, developmental pluripotency associated 5; DSA, Datura stramonium agglutinin; EGF, epidermal growth factor; EPCAM, epithelial cell adhesion molecule; FACS, fluorescent-activated cell sorting; FGF2, fibroblast growth factor 2; FGFR3, fibroblast growth factor receptor 3; FSH, follicle stimulating hormone; GATA-1, GATA binding protein 1; GATA-4, GATA binding protein 4; GDNF, glial cell line-derived neurotrophic factor; GPR125, G-protein coupled receptor 125; HLA, human leukocyte antigen; HNP, human nuclear protein; hTERT, human telomerase reverse transcriptase; ICC, immunocytochemistry; ITGA6, integrin subunit alpha 6; ITGA9, integrin subunit alpha 9; LHR, luteinizing hormone receptor; MACS, magnetic-activated cell sorting; NA, not available; NGFR, nerve growth factor receptor; NOA, non-obstructive azoospermia; OA, obstructive azoospermia; OCT4, octamer-binding transcription factor 4; PCNA, proliferating cell nuclear antigen; PDGFR-α, platelet-derived growth factor receptor alpha; PIWIL2, piwi-like RNA-mediated gene silencing 2; PLPPR3, phospholipid phosphatase related 3; PLZF, promyelocytic leukemia zinc finger; PRM2, protamine 2; RT-PCR, reverse transcription polymerase chain reaction; SCF, stem cell factor; SCP3, synaptonemal complex protein 3; SOX9, SRY-Box 9; SSC, spermatogonial stem cell; SSEA-4, stage-specific embryonic antigen-4; StAR, steroidogenic acute regulatory protein; STRA8, stimulated by retinoic acid gene 8; TESE, testicular sperm extraction; THY1, thymocyte differentiation antigen 1; UTF1, undifferentiated embryonic cell transcription factor 1; VIM, vimentin; WT-1, Wilms’ tumor gene 1; ZBTB16, zinc finger and BTB domain-containing protein 16.
3.3. Discussion
Research on human testicular tissue or cells is often hampered by low cellular starting numbers in combination with cell death due to dissociation/digestion-induced damage and suboptimal selection and/or enrichment strategies. The current literature, comparing mechanical dissociation protocols or enzymatic digestion protocols is inconclusive as most crucial start material characteristics are not reported. Review results indicate that testicular tissue cryopreservation influences digestion/dissociation outcomes which may hamper comparisons between studies. We may assume that other start material characteristics such as maturity and disease-related tissue alterations may also potentially influence digestion/dissociation outcomes. This further challenges the interpretation of outcomes of the few studies including a control group due to the different tissue characteristics before digestion, such as accumulated ECM products resulting in fibrotic remodeling [96], increased innervation density and numbers of mast cells and macrophages [97,98,99] and lower numbers of SCs, GCs, and, in particular, SSCs [100] compared to normozoospermia age-matched controls.
Furthermore, for similar tissue characteristics, digestion protocols differ in the types of enzymes, concentrations, incubation times, and conditions, thus further precluding the identification of the best enzymatic digestion protocol. When there are no predetermined fixed incubation times, the decision to stop an enzymatic reaction can also have an influence on the resulting single-cell suspension as incomplete digestion may enrich certain cell types in the final suspension at the cost of others that are more difficult to liberate.
Enzymatic digestion demonstrated its superiority compared to mechanical dissociation, in particular for cryopreserved tissue. This is especially important to consider as fertility restoration is based on cryo-stored ITT available in fertility centers worldwide, and as a better preservation of the integrity of the somatic cell fraction was observed for enzymatic digestion compared to mechanical dissociation [33]. The question of which enzymatic digestion protocol should preferably be used and in which condition is still unaddressed. Besides concerns related to differences in start material content or its prior cryopreservation or not, this gap of knowledge in today’s literature is mainly caused by the fact that only few studies report the start volume and/or weight of the tissue in combination with both cell viability and cellular yield, making it impossible to calculate the efficacy of each protocol. Indeed, an example can be found in the study of Sivakumar et al., 2006, [75] in which they reported a LC yield of 500.00 cells/mL; without knowing the start material weight, no normalization of cellular yield per mg of digested tissue can be calculated, thus hampering comparisons with other protocols.
Once single-cell suspensions are obtained, different strategies have been identified to select or enrich specific testicular cell types for downstream reproductive applications such as a safe single-cell transplantation to achieve recolonization of the human testicular niche. So far, attempts to decontaminate testicular tissue by cancer cell sorting resulted in disappointing results with insufficient decontamination while cell culture seemed to be more effective to eliminate cancer cells (for review see [18]). However, final proof of the efficacy of the culture approach is needed based on the possibility that cancer-specific antigen receptor gene targets used for minimal residual disease (MRD)-PCR could be modified during culture and because leukemic cells added to the culture could behave differently from leukemic cells obtained from contaminated testicular tissue. Alternatively, testicular cell selection or a combination strategy with a negative selection of cancer cells could be a promising strategy. Today, to the best of our knowledge, such a strategy with verification of the presence or not of cancer cells, has not been investigated yet. Therefore, this review tried to summarize options to sort testicular cells and report their outcomes to verify if pure single-cell suspensions, indicating absence of cancer cells, could be obtained.
Techniques such as gravity sedimentation, Percoll gradient, and differential plating allowed the selection of highly pure specific cell types, but studies reporting the obtained purity after selection/enrichment in combination with the total recovery number are rarely found. Such data is pivotal in the case of fertility restoration as not only pure single-cell fractions, but also sufficient cell numbers are required for fertility restoration. This is crucial in the case of SSC selection, as these stem cells maintain spermatogenesis and the colonization rate after transplantation is dependent on the number of transplanted SSCs [101]. Before research gaps are addressed, flow cytometry appears the most promising selection technique that led to the highest enrichment in SSCs in studies on cancer cell decontamination of human testicular cell suspensions. Of note, xenotransplantation of FACS-sorted human GC fractions did not result in tumor formation, but the invasive capacity of the cancer cell line that was used is questionable, as in controls where pure cancer cells were xenotransplanted, only a 43% tumor development rate was observed [29]. This highlights the need for further validation of this decontamination strategy. These results indicate that, currently, no testicular tissue digestion and cell selection is able to guarantee the safe autotransplantation of human SSCs when cancer cells are present in testicular tissue. However, this technique was successful in mice based on the absence of tumor development after allotransplantation of FACS-sorted fractions and histological assessment of testes recovered 8 weeks post-allotransplantation [102]. Therefore, further progress may be expected for human samples with combinations of cell markers to better target the cells of interest as single cell sequencing already identified heterogeneity among spermatogonial cell populations [85,86,87,103].
Since this scoping review focused on reproductive purposes starting from ITT, articles only reporting outcomes of dissociation/digestion and/or cell selection/enrichment for differentiated GCs were not included. These papers could contain interesting information; however, the usage of these papers is out of scope for the purpose of this review.
4. Conclusions
At present, testicular tissue dissociation/digestion is performed using a plethora of protocols, with no superiority of a particular protocol. The literature indicated that some testicular cell types, in particular the somatic SCs, are more sensitive to dissociation-induced damage. Furthermore, start material characteristics are important, and protocols might need to be adapted according to them. Currently, comparing protocols and their outcomes is hampered by the missing systematic reporting of start material characteristics and corresponding outcomes both at overall and cell-type specific levels. Summarizing tables in this review might help researchers to identify the highest achievements with digestion protocols and downstream selection/enrichment strategies applied to human testicular tissue, and to further cover research gaps towards the clinical application of fertility restoration techniques in cancer patients.
Author Contributions
Conceptualization, S.D.W. and C.W.; writing—original draft preparation, S.D.W., N.N.A. and C.W.; writing—review and editing, S.D.W. and C.W.; visualization, S.D.W. and C.W. All authors have read and agreed to the published version of the manuscript.
Funding
The research work of S.D.W. was funded by the Foundation against Cancer, grant number C/2020/1448.
Data Availability Statement
Data are contained within the article.
Acknowledgments
During the preparation of this manuscript/study, the authors have not used artificial intelligence.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| 3β-HSD | 3β-Hydroxysteroid Dehydrogenase |
| α-SMA | Alpha Smooth Muscle Actin |
| ABP | Androgen binding protein |
| AR | Androgen receptor |
| ACTA2 | Actin alpha 2 |
| BAX | BCL2 associated X |
| BMP4 | Bone morphogenetic protein 4 |
| CYP11A1 | Cytochrome P450 family 11 subfamily A member 1 |
| DAZL | Deleted in Azoospermia-like |
| DMC1 | DNA meiotic recombinase 1 |
| DMEM | Dulbecco’s modified eagle medium |
| DPG | Discontinuous Percoll gradient |
| DPPA5 | Developmental pluripotency associated 5 |
| DSA | Datura Stramonium Agglutinin |
| ECM | Extracellular Matrix |
| EDTA | Ethylenediaminetetraacetic acid |
| EGF | Epidermal growth factor |
| EPCAM | Epithelial Cell Adhesion Molecule |
| FACS | Fluorescent-Activated Cell Sorting |
| FGF2 | Fibroblast growth factor 2 |
| FGFR3 | Fibroblast Growth Factor Receptor 3 |
| FSH | Follicle stimulating hormone |
| GATA-1 | GATA Binding Protein 1 |
| GATA-4 | GATA Binding Protein 4 |
| GC | Germ Cell |
| GDNF | Glial cell line-derived neurotrophic factor |
| GFRA | Growth Factor Receptor A |
| GPR125 | G-Protein Coupled Receptor 125 |
| HBSS | Hanks’ balanced salt solution |
| HLA | Human Leukocyte Antigen |
| HNP | Human nuclear protein |
| hTERT | Human telomerase reverse transcriptase |
| ICC | Immunocytochemistry |
| ITGA6 | Integrin Subunit Alpha 6 |
| ITGA9 | Integrin Subunit Alpha 9 |
| ITT | Immature Testicular Tissue |
| IU | International unit |
| IVF-ICSI | In Vitro Fertilization–Intracytoplasmatic Sperm Injection |
| IVM | In Vitro Maturation |
| KU | Kunitz unit |
| LC | Leydig Cell |
| LHR | Luteinizing Hormone Receptor |
| MACS | Magnetic-Activated Cell Sorting |
| MAGEA4 | Melanoma-associated antigen 4 |
| MRD-PCR | Minimal Residual Disease Polymerase Chain Reaction |
| NA | Not available |
| NGFR | Nerve Growth Factor Receptor |
| NOA | Non-obstructive azoospermia |
| OA | Obstructive azoospermia |
| OCT4 | Octamer-Binding Transcription Factor 4 |
| PCNA | Proliferating cell nuclear antigen |
| PDGFR-α | Platelet-Derived Growth Factor Receptor Alpha |
| PIWIL2 | Piwi-like RNA-mediated gene silencing 2 |
| PLPPR3 | Phospholipid Phosphatase Related 3 |
| PLZF | Promyelocytic leukemia zinc finger |
| PRM2 | Protamine 2 |
| PTMC | Peritubular Myoid Cell |
| RCF | Relative centrifugal force |
| RPM | Revolutions per minute |
| RT-PCR | Reverse Transcription Polymerase Chain Reaction |
| SALL4 | Sal-like protein 4 |
| SC | Sertoli Cell |
| SCF | Stem cell factor |
| SCP3 | Synaptonemal complex protein 3 |
| sLC | Stem Leydig Cell |
| SOX9 | SRY-box 9 |
| SSC | Spermatogonial Stem Cell |
| SSEA-4 | Stage-Specific Embryonic Antigen-4 |
| StAR | Steroidogenic Acute Regulatory Protein |
| STRA8 | Stimulated By Retinoic Acid 8 |
| TESE | Testicular sperm extraction |
| THY1 | Thymocyte Antigen 1 |
| UTF1 | Undifferentiated Embryonic Cell Transcription Factor 1 |
| VIM | Vimentin |
| WT-1 | Wilms’ tumor gene 1 |
| ZBTB16 | Zinc finger and BTB domain-containing protein 16 |
References
- Botta, L.; Gatta, G.; Capocaccia, R.; Stiller, C.; Cañete, A.; Maso, L.D.; Innos, K.; Mihor, A.; Erdmann, F.; Spix, C.; et al. Long-term survival and cure fraction estimates for childhood cancer in Europe (EUROCARE-6): Results from a population-based study. Lancet Oncol. 2022, 23, 1525–1536. [Google Scholar] [CrossRef]
- Silva, N.d.P.; Gini, A.; Dolya, A.; Colombet, M.; Soerjomataram, I.; Youlden, D.; Stiller, C.; Steliarova-Foucher, E.; Aitken, J.; Bray, F.; et al. Prevalence of childhood cancer survivors in Europe: A scoping review. EJC Paediatr. Oncol. 2024, 3, 100155. [Google Scholar] [CrossRef] [PubMed]
- Howell, S.J.; Shalet, S.M. Spermatogenesis After Cancer Treatment: Damage and Recovery. JNCI Monogr. 2005, 2005, 12–17. [Google Scholar] [CrossRef]
- Rivkees, S.A.; Crawford, J.D. The Relationship of Gonadal Activity and Chemotherapy-Induced Gonadal Damage. JAMA 1988, 259, 2123–2125. [Google Scholar] [CrossRef] [PubMed]
- Green, D.M.; Kawashima, T.; Stovall, M.; Leisenring, W.; Sklar, C.A.; Mertens, A.C.; Donaldson, S.S.; Byrne, J.; Robison, L.L. Fertility of Male Survivors of Childhood Cancer: A Report From the Childhood Cancer Survivor Study. J. Clin. Oncol. 2010, 28, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Green, D.M.; Liu, W.; Kutteh, W.H.; Ke, R.W.; Shelton, K.C.; Sklar, C.A.; Chemaitilly, W.; Pui, C.-H.; Klosky, J.L.; Spunt, S.L.; et al. Cumulative alkylating agent exposure and semen parameters in adult survivors of childhood cancer: A report from the St Jude Lifetime Cohort Study. Lancet Oncol. 2014, 15, 1215–1223. [Google Scholar] [CrossRef]
- Hudson, M.M.; Ness, K.K.; Gurney, J.G.; Mulrooney, D.A.; Chemaitilly, W.; Krull, K.R.; Green, D.M.; Armstrong, G.T.; Nottage, K.A.; Jones, K.E.; et al. Clinical Ascertainment of Health Outcomes Among Adults Treated for Childhood Cancer. JAMA 2013, 309, 2371–2381. [Google Scholar] [CrossRef]
- Brignardello, E.; Felicetti, F.; Castiglione, A.; Chiabotto, P.; Corrias, A.; Fagioli, F.; Ciccone, G.; Boccuzzi, G. Endocrine health conditions in adult survivors of childhood cancer: The need for specialized adult-focused follow-up clinics. Eur. J. Endocrinol. 2013, 168, 465–472. [Google Scholar] [CrossRef]
- Wyns, C.; Curaba, M.; Petit, S.; Vanabelle, B.; Laurent, P.; Wese, J.-F.; Donnez, J. Management of fertility preservation in prepubertal patients: 5 years’ experience at the Catholic University of Louvain. Hum. Reprod. 2011, 26, 737–747. [Google Scholar] [CrossRef]
- Wyns, C.; Kanbar, M.; Giudice, M.G.; Poels, J. Fertility preservation for prepubertal boys: Lessons learned from the past and update on remaining challenges towards clinical translation. Hum. Reprod. Updat. 2020, 27, 433–459. [Google Scholar] [CrossRef]
- Stukenborg, J.-B.; Wyns, C. Fertility sparing strategies for pre- and peripubertal male cancer patients. Ecancermedicalscience 2020, 14, 1016. [Google Scholar] [CrossRef]
- Jensen, C.F.S.; Dong, L.; Gul, M.; Fode, M.; Hildorf, S.; Thorup, J.; Hoffmann, E.; Cortes, D.; Fedder, J.; Andersen, C.Y.; et al. Fertility preservation in boys facing gonadotoxic cancer therapy. Nat. Rev. Urol. 2021, 19, 71–83. [Google Scholar] [CrossRef]
- Wyns, C.; Curaba, M.; Vanabelle, B.; Van Langendonckt, A.; Donnez, J. Options for fertility preservation in prepubertal boys. Hum. Reprod. Updat. 2010, 16, 312–328. [Google Scholar] [CrossRef]
- Duffin, K.; Neuhaus, N.; Andersen, C.Y.; Barraud-Lange, V.; Braye, A.; Eguizabal, C.; Feraille, A.; Ginsberg, J.P.; Gook, D.; Goossens, E.; et al. A 20-year overview of fertility preservation in boys: New insights gained through a comprehensive international survey. Hum. Reprod. Open 2024, 2024, hoae010. [Google Scholar] [CrossRef] [PubMed]
- Goossens, E.; Jahnukainen, K.; Mitchell, R.; van Pelt, A.; Pennings, G.; Rives, N.; Poels, J.; Wyns, C.; Lane, S.; Rodriguez-Wallberg, K.; et al. Fertility preservation in boys: Recent developments and new insights. Hum. Reprod. Open 2020, 2020, hoaa016. [Google Scholar] [CrossRef] [PubMed]
- Kanbar, M.; de Michele, F.; Giudice, M.G.; Desmet, L.; Poels, J.; Wyns, C. Long-term follow-up of boys who have undergone a testicular biopsy for fertility preservation. Hum. Reprod. 2020, 36, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Fayomi, A.P.; Peters, K.; Sukhwani, M.; Valli-Pulaski, H.; Shetty, G.; Meistrich, M.L.; Houser, L.; Robertson, N.; Roberts, V.; Ramsey, C.; et al. Autologous grafting of cryopreserved prepubertal rhesus testis produces sperm and offspring. Science 2019, 363, 1314–1319, Erratum in Science 2019, 364, eaax4999. [Google Scholar] [CrossRef]
- Kourta, D.; Kanbar, M.; Amorim, C.A.; Wyns, C. Cancer cell contamination and decontamination methods for ovaries and testes: Special focus on prepubertal gonads with a view to safe fertility restoration. Hum. Reprod. 2023, 38, 780–798. [Google Scholar] [CrossRef]
- Kourta, D.; Camboni, A.; Saussoy, P.; Kanbar, M.; Poels, J.; Wyns, C. Evaluating testicular tissue for future autotransplantation: Focus on cancer cell contamination and presence of spermatogonia in tissue cryobanked for boys diagnosed with a hematological malignancy. Hum. Reprod. 2024, 39, 486–495. [Google Scholar] [CrossRef]
- Hou, M.; Andersson, M.; Eksborg, S.; Soder, O.; Jahnukainen, K. Xenotransplantation of testicular tissue into nude mice can be used for detecting leukemic cell contamination. Hum. Reprod. 2007, 22, 1899–1906. [Google Scholar] [CrossRef]
- Jahnukainen, K.; Hou, M.; Petersen, C.; Setchell, B.; Söder, O. Intratesticular transplantation of testicular cells from leukemic rats causes transmission of leukemia. Cancer Res. 2001, 61, 706–710. [Google Scholar]
- Patience, C.; YTakeuchi, R.A. Weiss, Zoonosis in xenotransplantation. Curr. Opin. Immunol. 1998, 10, 539–542. [Google Scholar] [CrossRef]
- de Michele, F.; Poels, J.; Vermeulen, M.; Ambroise, J.; Gruson, D.; Guiot, Y.; Wyns, C. Haploid Germ Cells Generated in Organotypic Culture of Testicular Tissue From Prepubertal Boys. Front. Physiol. 2018, 9, 1413. [Google Scholar] [CrossRef]
- Abofoul-Azab, M.; AbuMadighem, A.; Lunenfeld, E.; Kapelushnik, J.; Shi, Q.; Pinkas, H.; Huleihel, M. Development of Postmeiotic Cells In Vitro from Spermatogonial Cells of Prepubertal Cancer Patients. Stem Cells Dev. 2018, 15, 1007–1020. [Google Scholar] [CrossRef]
- Hermann, B.P.; Sukhwani, M.; Winkler, F.; Pascarella, J.N.; Peters, K.A.; Sheng, Y.; Valli, H.; Rodriguez, M.; Ezzelarab, M.; Dargo, G.; et al. Spermatogonial Stem Cell Transplantation into Rhesus Testes Regenerates Spermatogenesis Producing Functional Sperm. Cell Stem Cell 2012, 11, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Zielen, A.C.; Peters, K.A.; Shetty, G.; Gross, D.A.; Hanna, C.B.; Dovey, S.L.; Wecht, A.; Cannon, G.M.; Meistrich, M.L.; Hsieh, M.; et al. Ultrasound-Guided Rete Testis Approach to Sperm Aspiration and Spermatogonial Stem Cell Transplantation in Patients with Azoospermia. medRxiv 2025. [Google Scholar] [CrossRef] [PubMed]
- Kanbar, M.; Vermeulen, M.; Wyns, C. Organoids as tools to investigate the molecular mechanisms of male infertility and its treatments. Reproduction 2021, 161, R103–R112. [Google Scholar] [CrossRef] [PubMed]
- Baert, Y.; Rombaut, C.; Goossens, E. Scaffold-Based and Scaffold-Free Testicular Organoids from Primary Human Testicular Cells. Methods Mol Biol 2019, 1576, 283–290. [Google Scholar]
- Dovey, S.L.; Valli, H.; Hermann, B.P.; Sukhwani, M.; Donohue, J.; Castro, C.A.; Chu, T.; Sanfilippo, J.S.; Orwig, K.E. Eliminating malignant contamination from therapeutic human spermatogonial stem cells. J. Clin. Investig. 2013, 4, 1833–1843. [Google Scholar] [CrossRef]
- Geens, M.; Goossens, E.; Tournay, H. Cell selection by selective matrix adhesion is not sufficiently efficient for complete malignant cell depletion from contaminated human testicular cell suspensions. Fertil. Steril. 2011, 95, 787–791. [Google Scholar] [CrossRef]
- Human Testicular Tissue Digestion, Testicular Cells Selection and Downstream Characterization for Reproductive Purposes: A Scoping Review. Available online: https://osf.io/82mp4/ (accessed on 9 September 2025).
- Verheyen, G.; Popovic-Todorovic, B.; Tournaye, H. Processing and selection of surgically-retrieved sperm for ICSI: A review. Basic Clin. Androl. 2017, 27, 6. [Google Scholar] [CrossRef]
- Schneider, F.; Redmann, K.; Wistuba, J.; Schlatt, S.; Kliesch, S.; Neuhaus, N. Comparison of enzymatic digestion and mechanical dissociation of human testicular tissues. Fertil. Steril. 2015, 104, 302–311.e3. [Google Scholar] [CrossRef]
- Harichandan, A.; Sivasubramaniyan, K.; Hennenlotter, J.; Poths, S.; Bedke, J.; Kruck, S.; Stenzl, A.; Bühring, H.-J. Molecular Signatures of Primary Human Spermatogonial Progenitors and Its Neighboring Peritubular Stromal Compartment. Stem Cells Dev. 2017, 26, 263–273. [Google Scholar] [CrossRef]
- Kurpisz, M.; Mapp, P.; Łukaszyk, A.; Ogilvie, J.; Festenstein, H.; Sachs, J. Characterization of two monoclonal antibodies raised against human testicular cells. Andrologia 1988, 20, 304–310. [Google Scholar] [CrossRef]
- Reichard, A.; Asosingh, K. Best Practices for Preparing a Single Cell Suspension from Solid Tissues for Flow Cytometry. Cytom. Part A 2018, 95, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Liu, M.; Rong, M.; Zhang, X.; Wang, G.; Liu, Y.; Li, H.; Duan, S. The pros and cons of mechanical dissociation and enzymatic digestion in patient-derived organoid cultures for solid tumor. Cell Organoid 2024, 1, 9410009. [Google Scholar] [CrossRef]
- Lam, D.M.K.; Furrer, R.; Bruce, W.R. The Separation, Physical Characterization, and Differentiation Kinetics of Spermatogonial Cells of the Mouse. Proc. Natl. Acad. Sci. USA 1970, 65, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Griswold, M.D. Protein secretions of Sertoli cells. Int. Rev. Cytol. 1988, 110, 133–156. [Google Scholar]
- Berardo, C.; Ferrigno, A.; Siciliano, V.; Richelmi, P.; Vairetti, M.; Di Pasqua, L.G. Isolation of rat hepatocytes for pharmacological studies on metabotropic glutamate receptor (mGluR) subtype 5: A comparison between collagenase I versus collagenase IV. Eur. J. Histochem. 2020, 64, 3123. [Google Scholar] [CrossRef]
- Crabbé, E.; Verheyen, G.; Tournaye, H.; Van Steirteghem, A. The use of enzymatic procedures to recover testicular germ cells. Hum. Reprod. 1997, 12, 1682–1687. [Google Scholar] [CrossRef][Green Version]
- Dong, L.; Gul, M.; Hildorf, S.; Pors, S.E.; Kristensen, S.G.; Hoffmann, E.R.; Cortes, D.; Thorup, J.; Andersen, C.Y. Xeno-Free Propagation of Spermatogonial Stem Cells from Infant Boys. Int. J. Mol. Sci. 2019, 20, 5390. [Google Scholar] [CrossRef]
- Pakhomov, O.; Posokhov, Y. Detecting changes of testicular interstitial cell membranes with a fluorescent probe after incubation and cryopreservation with cryoprotective agents. Cryobiology 2025, 118, 105194. [Google Scholar] [CrossRef] [PubMed]
- Unni, S.; Kasiviswanathan, S.; D’sOuza, S.; Khavale, S.; Mukherjee, S.; Patwardhan, S.; Bhartiya, D. Efficient cryopreservation of testicular tissue: Effect of age, sample state, and concentration of cryoprotectant. Fertil. Steril. 2012, 97, 200–208.e1. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Thomas, A.; Schmidt, C.; Dann, C. Quantitative detection of human spermatogonia for optimization of spermatogonial stem cell culture. Hum. Reprod. 2014, 29, 2497–2511. [Google Scholar] [CrossRef] [PubMed]
- Pacchiarotti, J.; Ramos, T.; Howerton, K.; Greilach, S.; Zaragoza, K.; Olmstead, M.; Izadyar, F. Developing a Clinical-Grade Cryopreservation Protocol for Human Testicular Tissue and Cells. BioMed Res. Int. 2013, 2013, 930962. [Google Scholar] [CrossRef]
- Liu, S.; Tang, Z.; Xiong, T.; Tang, W. Isolation and characterization of human spermatogonial stem cells. Reprod. Biol. Endocrinol. 2011, 9, 141. [Google Scholar] [CrossRef]
- Berensztein, E.B.; Belgorosky, A.; Rivarola, M.A. Primary culture of prepubertal human testicular cells isolated from testes collected at necropsy. Eur. J. Endocrinol. 1992, 127, 66–71. [Google Scholar] [CrossRef]
- Brook, P.F.; Radford, J.A.; Shalet, S.M.; Joyce, A.D.; Gosden, R.G. Isolation of germ cells from human testicular tissue for low temperature storage and autotransplantation. Fertil. Steril. 2001, 75, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Nowroozi, M.R.; Ahmadi, H.; Rafiian, S.; Mirzapour, T.; Movahedin, M. In Vitro Colonization of Human Spermatogonia Stem Cells: Effect of Patient’s Clinical Characteristics and Testicular Histologic Findings. Urology 2011, 78, 1075–1081. [Google Scholar] [CrossRef]
- Izadyar, F.; Wong, J.; Maki, C.; Pacchiarotti, J.; Ramos, T.; Howerton, K.; Yuen, C.; Greilach, S.; Zhao, H.H.; Chow, M.; et al. Identification and characterization of repopulating spermatogonial stem cells from the adult human testis. Hum. Reprod. 2011, 26, 1296–1306. [Google Scholar] [CrossRef]
- Mirzapour, T.; Movahedin, M.; Ibrahim, T.A.T.; Koruji, M.; Haron, A.W.; Nowroozi, M.R.; Rafieian, S.H. Effects of basic fibroblast growth factor and leukaemia inhibitory factor on proliferation and short-term culture of human spermatogonial stem cells. Andrologia 2011, 44, 41–55. [Google Scholar] [CrossRef]
- Koruji, M.; Shahverdi, A.; Janan, A.; Piryaei, A.; Lakpour, M.R.; Sedighi, M.A.G. Proliferation of small number of human spermatogonial stem cells obtained from azoospermic patients. J. Assist. Reprod. Genet. 2012, 29, 957–967. [Google Scholar] [CrossRef]
- Riboldi, M.; Rubio, C.; Pellicer, A.; Gil-Salom, M.; Simón, C. In vitro production of haploid cells after coculture of CD49f+ with Sertoli cells from testicular sperm extraction in nonobstructive azoospermic patients. Fertil. Steril. 2012, 98, 580–590.e4. [Google Scholar] [CrossRef] [PubMed]
- Zohni, K.; Zhang, X.; Tan, S.L.; Chan, P.; Nagano, M. CD9 Is Expressed on Human Male Germ Cells That Have a Long-Term Repopulation Potential after Transplantation into Mouse Testes1. Biol. Reprod. 2012, 87, 27. [Google Scholar] [CrossRef] [PubMed]
- Kossack, N.; Terwort, N.; Wistuba, J.; Ehmcke, J.; Schlatt, S.; Schöler, H.; Kliesch, S.; Gromoll, J. A combined approach facilitates the reliable detection of human spermatogonia in vitro. Hum. Reprod. 2013, 28, 3012–3025. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Hai, Y.; Yao, C.; Chen, Z.; Hou, J.; Li, Z.; He, Z. Long-term culture and significant expansion of human Sertoli cells whilst maintaining stable global phenotype and AKT and SMAD1/5 activation. Cell Commun. Signal. 2015, 13, 20. [Google Scholar] [CrossRef]
- Jabari, A.; Gholami, K.; Khadivi, F.; Koruji, M.; Amidi, F.; Gilani, M.A.S.; Mahabadi, V.P.; Nikmahzar, A.; Salem, M.; Movassagh, S.A.; et al. In vitro complete differentiation of human spermatogonial stem cells to morphologic spermatozoa using a hybrid hydrogel of agarose and laminin. Int. J. Biol. Macromol. 2023, 235, 123801. [Google Scholar] [CrossRef]
- Nikmahzar, A.; Khadivi, F.; Koruji, M.; Jahanshahi, M.; Tarazjani, M.D.; Shabani, M.; Abbasi, Y.; Abbasi, M. Evaluation of Apoptosis-related Genes and Hormone Secretion Profiles Using Three Dimensional Culture System of Human Testicular Organoids. Galen. Med. J. 2023, 12, e2805. [Google Scholar]
- Sohni, A.; Tan, K.; Song, H.-W.; Burow, D.; de Rooij, D.G.; Laurent, L.; Hsieh, T.-C.; Rabah, R.; Hammoud, S.S.; Vicini, E.; et al. The Neonatal and Adult Human Testis Defined at the Single-Cell Level. Cell Rep. 2019, 26, 1501–1517.e4. [Google Scholar] [CrossRef]
- Guo, J.; Nie, X.; Giebler, M.; Mlcochova, H.; Wang, Y.; Grow, E.J.; DonorConnect; Kim, R.; Tharmalingam, M.; Matilionyte, G.; et al. The Dynamic Transcriptional Cell Atlas of Testis Development during Human Puberty. Cell Stem Cell 2020, 26, 262–276.e4. [Google Scholar] [CrossRef]
- Kaur, G.; Thompson, L.A.; Dufour, J.M. Sertoli cells–Immunological sentinels of spermatogenesis. Semin. Cell Dev. Biol. 2014, 30, 36–44. [Google Scholar] [CrossRef]
- David, S.; Orwig, K.E. Spermatogonial Stem Cell Culture in Oncofertility. Urol. Clin. N. Am. 2020, 47, 227–244. [Google Scholar] [CrossRef]
- Kato, Y.; Nagao, Y. Effect of polyvinylpyrrolidone on sperm function and early embryonic development following intracytoplasmic sperm injection in human assisted reproduction. Reprod. Med. Biol. 2012, 11, 165–176. [Google Scholar] [CrossRef]
- Medrano, J.V.; Rombaut, C.; Simon, C.; Pellicer, A.; Goossens, E. Human spermatogonial stem cells display limited proliferation in vitro under mouse spermatogonial stem cell culture conditions. Fertil. Steril. 2016, 106, 1539–1549.e8. [Google Scholar] [CrossRef]
- Gat, I.; Maghen, L.; Filice, M.; Wyse, B.; Zohni, K.; Jarvi, K.; Lo, K.C.; Fisher, A.G.; Librach, C. Optimal culture conditions are critical for efficient expansion of human testicular somatic and germ cells in vitro. Fertil. Steril. 2017, 107, 595–605.e7. [Google Scholar] [CrossRef] [PubMed]
- Teng, Y.; Xue, W.-J.; Ding, X.-M.; Feng, X.-S.; Xiang, H.-L.; Jiang, Y.-Z.; Tian, P.-X. Isolation and culture of adult Sertoli cells and their effects on the function of co-cultured allogeneic islets in vitro. Chin. Med. J. 2005, 118, 1857–1862. [Google Scholar] [PubMed]
- Chui, K.; Trivedi, A.; Cheng, C.Y.; Cherbavaz, D.B.; Dazin, P.F.; Huynh, A.L.T.; Mitchell, J.B.; Rabinovich, G.A.; Noble-Haeusslein, L.J.; John, C.M. Characterization and Functionality of Proliferative Human Sertoli Cells. Cell Transplant. 2011, 20, 619–635. [Google Scholar] [CrossRef] [PubMed]
- Lipshultz, L.I.; Murthy, L.; Tindall, D.J. Characterization of Human Sertoli Cells in Vitro*. J. Clin. Endocrinol. Metab. 1982, 55, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Gaur, M.; Ramathal, C.; Pera, R.A.R.; Turek, P.J.; John, C.M. Isolation of human testicular cells and co-culture with embryonic stem cells. Reproduction 2018, 155, 151–164. [Google Scholar] [CrossRef]
- Zirkin, B.R.; Papadopoulos, V. Leydig cells: Formation, function, and regulation. Biol. Reprod. 2018, 99, 101–111. [Google Scholar] [CrossRef]
- Qureshi, S.J.; Sharpe, R.M. Evaluation of possible determinants and consequences of Leydig cell heterogeneity in man. Int. J. Androl. 1993, 16, 293–305. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, J.; Deng, C.; Jiang, M.H.; Feng, X.; Xia, K.; Li, W.; Lai, X.; Xiao, H.; Ge, R.-S.; et al. Transplanted human p75-positive stem Leydig cells replace disrupted Leydig cells for testosterone production. Cell Death Dis. 2017, 8, e3123. [Google Scholar] [CrossRef]
- Simpson, B.J.B.; Wu, F.C.W.; Sharpe, R.M. Isolation of Human Leydig Cells Which Are Highly Responsive to Human Chorionic Gonadotropin. J. Clin. Endocrinol. Metab. 1987, 65, 415–422. [Google Scholar] [CrossRef]
- Sivakumar, R.; Sivaraman, P.B.; Mohan-Babu, N.; Jainul-Abideen, I.M.; Kalliyappan, P.; Balasubramanian, K. Radiation Exposure Impairs Luteinizing Hormone Signal Transduction and Steroidogenesis in Cultured Human Leydig Cells. Toxicol. Sci. 2006, 91, 550–556. [Google Scholar] [CrossRef]
- Bilinska, B.; Kotula-Balak, M.; Sadowska, J. Morphology and function of human Leydig cells in vitro. Immunocytochemical and radioimmunological analyses. Eur. J. Histochem. 2009, 53, 35–42. [Google Scholar] [CrossRef]
- Xia, K.; Ma, Y.; Feng, X.; Deng, R.; Ke, Q.; Xiang, A.P.; Deng, C. Endosialin defines human stem Leydig cells with regenerative potential. Hum. Reprod. 2020, 35, 2197–2212. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.B.; Walker, W.H. The regulation of spermatogenesis by androgens. Semin. Cell Dev. Biol. 2014, 30, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Zhao, L.; Luo, J.; Shi, C.; Yao, C.; Ji, Z.; Xu, J.; Xu, S.; Tian, R.; Zhi, E.; et al. Identification and isolation of human testicular peritubular myoid cells and Leydig cells by a combination of ITGA9 and NGFR. Reprod. Biol. Endocrinol. 2025, 23, 82. [Google Scholar] [CrossRef] [PubMed]
- Mayerhofer, A. Human testicular peritubular cells: More than meets the eye. Reproduction 2013, 145, R107–R116. [Google Scholar] [CrossRef]
- Albrecht, M.; RämSch, R.; KöhN, F.M.; Schwarzer, J.U.; Mayerhofer, A. Isolation and Cultivation of Human Testicular Peritubular Cells: A New Model for the Investigation of Fibrotic Processes in the Human Testis and Male Infertility. J. Clin. Endocrinol. Metab. 2006, 91, 1956–1960. [Google Scholar] [CrossRef]
- Landreh, L.; Spinnler, K.; Schubert, K.; Häkkinen, M.R.; Auriola, S.; Poutanen, M.; Söder, O.; Svechnikov, K.; Mayerhofer, A. Human Testicular Peritubular Cells Host Putative Stem Leydig Cells With Steroidogenic Capacity. J. Clin. Endocrinol. Metab. 2014, 99, E1227–E1235. [Google Scholar] [CrossRef] [PubMed]
- Liebich, A.; Schmid, N.; Koupourtidou, C.; Herrmann, C.; Dietrich, K.-G.; Welter, H.; Ninkovic, J.; Mayerhofer, A. The Molecular Signature of Human Testicular Peritubular Cells Revealed by Single-Cell Analysis. Cells 2022, 11, 3685. [Google Scholar] [CrossRef]
- Rolland, A.D.; Evrard, B.; Darde, T.A.; Le Béguec, C.; Le Bras, Y.; Bensalah, K.; Lavoué, S.; Jost, B.; Primig, M.; Dejucq-Rainsford, N.; et al. RNA profiling of human testicular cells identifies syntenic lncRNAs associated with spermatogenesis. Hum. Reprod. 2019, 34, 1278–1290. [Google Scholar] [CrossRef] [PubMed]
- Gille, A.-S.; Lapoujade, C.; Wolf, J.-P.; Fouchet, P.; Barraud-Lange, V. Contribution of Single-Cell Transcriptomics to the Characterization of Human Spermatogonial Stem Cells: Toward an Application in Male Fertility Regenerative Medicine? Int. J. Mol. Sci. 2019, 20, 5773. [Google Scholar] [CrossRef]
- Givelet, M.; Firlej, V.; Lassalle, B.; Gille, A.S.; Lapoujade, C.; Holtzman, I.; Jarysta, A.; Haghighirad, F.; Dumont, F.; Jacques, S.; et al. Transcriptional profiling of β-2M(-)SPα-6(+)THY1(+) spermatogonial stem cells in human spermatogenesis. Stem Cell Rep. 2022, 17, 936–952. [Google Scholar] [CrossRef] [PubMed]
- Lapoujade, C.; Blanco, M.; Givelet, M.; Gille, A.S.; Allemand, I.; Lenez, L.; Thiounn, N.; Roux, S.; Wolf, J.P.; Patrat, C.; et al. Characterisation and hierarchy of the spermatogonial stem cell compartment in human spermatogenesis by spectral cytometry using a 16-colors panel. Cell. Mol. Life Sci. 2024, 82, 15. [Google Scholar] [CrossRef]
- Nickkholgh, B.; Mizrak, S.C.; Korver, C.M.; van Daalen, S.K.; Meissner, A.; Repping, S.; van Pelt, A.M. Enrichment of spermatogonial stem cells from long-term cultured human testicular cells. Fertil. Steril. 2014, 102, 558–565.e5. [Google Scholar] [CrossRef]
- Tan, K.; Song, H.-W.; Thompson, M.; Munyoki, S.; Sukhwani, M.; Hsieh, T.-C.; Orwig, K.E.; Wilkinson, M.F. Transcriptome profiling reveals signaling conditions dictating human spermatogonia fate in vitro. Proc. Natl. Acad. Sci. USA 2020, 117, 17832–17841. [Google Scholar] [CrossRef]
- Movassagh, S.A.; Dehkordi, M.B.; Pourmand, G.; Gholami, K.; Talebi, A.; Esfandyari, S.; Jabari, A.; Samadian, A.; Abbasi, M. Isolation, identification and differentiation of human spermatogonial cells on three-dimensional decellularized sheep testis. Acta Histochem. 2020, 122, 151623. [Google Scholar] [CrossRef]
- Salem, M.; Khadivi, F.; Feizollahi, N.; Khodarahmian, M.; Marghmaleki, M.S.; Ayub, S.; Chegini, R.; Bashiri, Z.; Abbasi, Y.; Naji, M.; et al. Melatonin Promotes Differentiation of Human Spermatogonial Stem Cells Cultured on Three-Dimensional Decellularized Human Testis Matrix. Urol. J. 2023, 21, 250–264. [Google Scholar] [CrossRef]
- He, Z.; Kokkinaki, M.; Jiang, J.; Dobrinski, I.; Dym, M. Isolation, Characterization, and Culture of Human Spermatogonia1. Biol. Reprod. 2010, 82, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Von Kopylow, K.; Schulze, W.; Salzbrunn, A.; Spiess, A.-N. Isolation and gene expression analysis of single potential human spermatogonial stem cells. Mol. Hum. Reprod. 2016, 22, 229–239. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, Y.; Niu, M.; Yao, C.; Hai, Y.; Yuan, Q.; Liu, Y.; Guo, Y.; Li, Z. Fractionation of human spermatogenic cells using STA-PUT gravity sedimentation and their miRNA profiling. Sci. Rep. 2015, 5, 8084. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.F.; Yango, P.; Altman, E.; Choudhry, S.; Poelzl, A.; Zamah, A.M.; Rosen, M.; Klatsky, P.C.; Tran, N.D. Testicular Niche Required for Human Spermatogonial Stem Cell Expansion. STEM CELLS Transl. Med. 2014, 3, 1043–1054. [Google Scholar] [CrossRef]
- Davidoff, M.S.; Breucker, H.; Holstein, A.F.; Seidl, K. Cellular architecture of the lamina propria of human seminiferous tubules. Cell Tissue Res. 1990, 262, 253–261. [Google Scholar] [CrossRef]
- Frungieri, M.B.; Calandra, R.S.; Lustig, L.; Meineke, V.; Köhn, F.M.; Vogt, H.-J.; Mayerhofer, A. Number, distribution pattern, and identification of macrophages in the testes of infertile men. Fertil. Steril. 2002, 78, 298–306. [Google Scholar] [CrossRef]
- Meineke, V.; Frungieri, M.B.; Jessberger, B.; Vogt, H.-J.; Mayerhofer, A. Human testicular mast cells contain tryptase: Increased mast cell number and altered distribution in the testes of infertile men. Fertil. Steril. 2000, 74, 239–244. [Google Scholar] [CrossRef]
- Hussein, M.R.; Abou-Deif, E.S.; Bedaiwy, M.A.; Said, T.M.; Mustafa, M.G.; Nada, E.; Ezat, A.; Agarwal, A. Phenotypic characterization of the immune and mast cell infiltrates in the human testis shows normal and abnormal spermatogenesis. Fertil. Steril. 2005, 83, 1447–1453. [Google Scholar] [CrossRef]
- Hentrich, A.; Wolter, M.; Szardening-Kirchner, C.; Lüers, G.H.; Bergmann, M.; Kliesch, S.; Konrad, L. Reduced numbers of Sertoli, germ, and spermatogonial stem cells in impaired spermatogenesis. Mod. Pathol. 2011, 24, 1380–1389, Erratum in Mod. Pathol. 2017, 30, 311. [Google Scholar] [CrossRef]
- Dobrinski, I.; Ogawa, T.; Avarbock, M.R.; Brinster, R.L. Computer assisted image analysis to assess colonization of recipient seminiferous tubules by spermatogonial stem cells from transgenic donor mice. Mol. Reprod Dev. 1999, 53, 142–148. [Google Scholar] [CrossRef]
- Fujita, K.; Ohta, H.; Tsujimura, A.; Takao, T.; Miyagawa, Y.; Takada, S.; Matsumiya, K.; Wakayama, T.; Okuyama, A. Transplantation of spermatogonial stem cells isolated from leukemic mice restores fertility without inducing leukemia. J. Clin. Investig. 2005, 115, 1855–1861. [Google Scholar] [CrossRef] [PubMed]
- Kwaspen, L.; Kanbar, M.; Wyns, C. Mapping the Development of Human Spermatogenesis Using Transcriptomics-Based Data: A Scoping Review. Int. J. Mol. Sci. 2024, 25, 6925. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



