Abstract
The title compound, N-[1-(2,5-dimethyl-3-thienyl)ethylidene]-1,3-benzothiazol-2-amine has been synthesized by condensation of 2-aminobenzothiazole and 3-acetyl-2,5-dimethylthiophene in ethanol. The structure of this new compound was confirmed by elemental analysis, IR, 1H-NMR, 13C-NMR and EI-MS spectral analysis.
Heterocyclic compounds containing nitrogen and sulphur exhibit a wide variety of biological activities such as antibacterial [1], antifungal [2], antitumor [3], or anti-HIV activity [4]. The thiazole ring dramatically increases the diversity of certain biological properties such as antibacterial [5], antiviral [6], and antitubercular [7] activities. In this paper, we report the synthesis of a novel compound by condensation of 2-aminobenzothiazole and 3-acetyl-2,5-dimethyl thiophene.
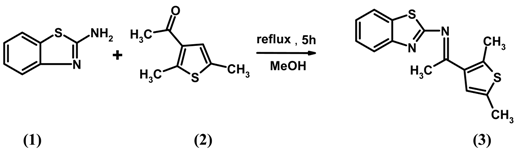
A mixture of 2-aminobenzothiazole (0.50 g, 0.0033 mol) and 3-acetyl-2,5-dimethylthiophene (0.475 mL, 0.0033 mol) in methanol (15 mL) was refluxed for 5 h with stirring to give a yellow precipitate. This material was filtered and recrystallized from a mixture of methanol and chloroform to give the pure compound 3.
Yield: 72%; m.p. 86 °C.
EI-MS m/z (rel. int. %): [M+1]+ 287 (68).
IR (KBr) vmax cm–1: 3271 (C-H), 3054 (C-H aromatic), 1637 (C=N), 1104 (C-N).
1H-NMR (600 MHz, CDCl3) δ: 7.59 (CH, dd, J = 7.2 Hz), 7.54 (CH, dd, J = 4.2 Hz), 7.32 (CH, d, J = 3.6 Hz), 7.12 (CH, d, J = 4.2 Hz), 6.98 (thienyl-4H, s), 2.66 (CH3, s), 2.55 (CH3, s), 2.44 (CH3, s).
13C-NMR (150 MHz, CDCl3) δ: 194.26, 166.04, 151.98, 147.31, 135.78, 135.01, 131.51, 126.61, 125.97, 122.26, 120.91, 119.12, 29.86, 16.07, 14.55.
Anal. calc. for C15H14N2S2: C, 62.97, H, 4.89, N, 9.79. Found: C, 62.95, H, 4.85, N, 9.75.
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgements
The authors would like to thank the Chemistry Department, King Abdul Aziz University, Jeddah, Saudi Arabia for providing the research facilities.
References and Notes
- Przybylski, P.; Pyta, K.; Stefanska, J.; Ratajczak-Sitarz, M.; Katrusiak, A.; Huczynski, A.; Brzezinski, B. Synthesis, crystal structures and antibacterial activity studies of aza-derivatives of phytoalexin from cotton plant–gossypol. Eur. J. Med. Chem. 2009, 44, 4393–4403. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, R.; Maheswaran, S. Synthesis, spectra, dioxygen affinity and antifungal activity of Ru(III) Schiff base complexes. J. Inorg. Biochem. 2003, 96, 457–462. [Google Scholar] [CrossRef]
- Silveira, V.C.; Luz, J.S.; Oliveira, C.C.; Graziani, I.; Ciriolo, M.R.; Ferreira, A.M.C. Double-strand DNA cleavage induced by oxindole-Schiff base copper(II) complexes with potential antitumor activity. J. Inorg. Biochem. 2008, 102, 1090–1103. [Google Scholar] [CrossRef] [PubMed]
- Pandeya, S.N.; Sriram, D.; Nath, G.; Clercq, E. Synthesis, antibacterial, antifungal and anti-HIV activities of Schiff and Mannich bases derived from isatin derivatives and N-[4-(4′-chlorophenyl)thiazol-2-yl]thiosemicarbazide. Eur. J. Pharma. Scie. 1999, 9, 25–31. [Google Scholar] [CrossRef]
- Holla, B.S.; Malini, K.V.; Rao, V.S.; Sarojini, B.K.; Kumari, N.S. Synthesis of some new 2,4-disubstituted thiazoles as possible antibacterial and anti-inflammatory agents. Eur. J. Med. Chem. 2003, 38, 313–318. [Google Scholar] [CrossRef]
- El-Sabbagh, O.I.; Baraka, M.M.; Ibrahim, S.M.; Pannecouque, C.; Graciela, Andrei; Snoeck, R.; Balzarini, J.; Rashad, A.A. Synthesis and antiviral activity of new pyrazole and thiazole derivatives. Eur. J. Med. Chem. 2009, 44, 3746–3753. [Google Scholar] [CrossRef] [PubMed]
- Mallikarjuna, B.P.; Sastry, B.S.; Kumar, G.V.S.; Rajendraprasad, Y.; Chandrashekar, S.M.; Sathisha, K. Synthesis of new 4-isopropylthiazole hydrazide analogs and some derived clubbed triazole, oxadiazole ring systems–A novel class of potential antibacterial, antifungal and antitubercular agents. Eur. J. Med. Chem. 2009, 44, 4739–4746. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
