Abstract
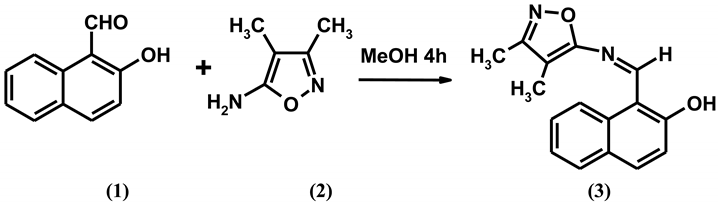
The title compound, 1-{[(3,4-dimethylisoxazol-5-yl)imino]methyl}-2-naphthol has been synthesized by condensation of 5-amino-3,4-dimethylisoxazole and 2-hydroxy-1-naphthaledhyde in ethanol. The structure of this new compound was confirmed by elemental analysis, IR, 1H-NMR, 13C-NMR and EI-MS spectral analysis.
Compounds with the structure of -C=N- (azomethine group) are known as Schiff bases, which are usually synthesized by condensation of primary amines and active carbonyl groups. Schiff bases are an important class of compounds in the medicinal and pharmaceutical field, including antibacterial [1,2], antifungal [3,4] and antitumor activity [5,6]. Heterocycle-containing Schiff bases can show dramatically increased biological activities. As evident from the literature, it was noted that a lot of research has been carried out on Schiff bases, but no work has been done on this particular type of Schiff base. In this paper we report the synthesis of a novel Schiff base from 5-amino-3,4-dimethylisoxazole and 2-hydroxy-1-naphthaledhyde.
A mixture of 5-amino-3,4-dimethylisoxazole (0.50 g, 0.0025 mol) and 2-hydroxy-1-naphthaledhyde (0.43 g, 0.0025 mol) in methanol (15 mL) was refluxed at 80 °C for 5 h with continuous stirring. Progress of the reaction was monitored by TLC. After completion of the reaction, the solution was cooled. The heavy precipitate thus obtained was collected by filtration and purified by recrystallization from methanol and chloroform to give the title compound (3).

Yield: 72%; m.p. 160 °C
EI-MS m/z (rel. int.%): 267 (75) [M + 1]+
IR (KBr) vmax cm-1: 2933 (C-H), 1626 (C=C), 1585 (HC=N), 1123 (C-N).
1H-NMR (600 MHz, CDCl3) δ: 14.46 (s, OH), 8.30 (d, J = 12.7 Hz, 1H, H-3), 7.99 (d, J = 13.5 Hz, 1H, H-4), 7.87 (d, J = 11.8 Hz, 1H, H-5), 7.70 (dd, J = 8.6 Hz, 1H, H-6), 7.51 (dd, J = 10.8 Hz, 1H, H-7), 7.28 (d, J = 13.6 Hz, 1H, H-8), 7.34 (s, 1H, CHolefinic), 2.36 (s, CH3), 2.15 (s, CH3).
13C-NMR (150 MHz, CDCl3) δ: 163.69, 162.76, 162.21, 158.27, 136.55, 132.85, 129.33, 128.28, 128.00, 124.19, 121.14, 119.78, 119.44, 105.57, 10.77, 6.71.
Anal. calc. for C16H14O2N2: C, 72.17, H, 5.30, N, 10.52. Found: C, 72.13, H, 5.26, N, 10.48.
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgements
This research was supported by Dean Ship of Scientific Research at King Abdul Aziz University, Jeddah, Saudi Arabia (Grant No. 171/428) and is gratefully acknowledge.
References and Notes
- Panneerselvam, P.; Rather, B.A.; Reddy, D.R.S.; Kumar, N.R. Synthesis and anti-microbial screening of some Schiff bases of 3-amino-6,8-dibromo-2-phenylquinazolin-4(3H)-ones. Eur. J. Med. Chem. 2009, 44, 2328–2333. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Wang, J.; Bai, J. Synthesis and antimicrobial activity of the Schiff base from chitosan a; nd citral. Carbohyd. Res. 2009, 344, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Bharti, S.K.; Nath, G.; Tilak, R.; Singh, S.K. Synthesis, anti-bacterial and anti-fungal activities of some novel Schiff bases containing 2,4-disubstituted thiazole ring. Eur. J. Med. Chem. 2010, 45, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Bagihalli, G.B.; Avaji, P.G.; Patil, S.A.; Badami, P.S. Synthesis, spectral characterization, in vitro antibacterial, antifungal and cytotoxic activities of Co(II), Ni(II) and Cu(II) complexes with 1,2,4-triazole Schiff bases. Eur. J. Med. Chem. 2008, 43, 2639–2649. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, Z. Crystal structures, antioxidation and DNA binding properties of Eu(III) complexes with Schiff-base ligands derived from 8-hydroxyquinoline-2-carboxyaldehyde and three aroylhydrazines. J. Inorg. Biochem. 2009, 103, 1014–1022. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, H.; Akhter, Z.; Yameen, S.; Siddiqi, H.M.; Mirza, B.; Rifat, A. Synthesis and biological evaluations of some Schiff-base esters of ferrocenyl aniline and simple aniline. J. Orgnometa. Chem. 2009, 694, 2198–2203. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
