Abstract
The reaction of 2,4-difluoronitrobenzene with an excess of aniline at 175 °C led to the isolation of an unexpected brown quinone substituted with two phenylamino groups and one phenylimino group. This product was easily distinguished from other expected derivatives because it is brown rather than yellow. The UV/Vis has a weak long wavelength absorption at 480–600 nm accounting for the colour.
1. Introduction
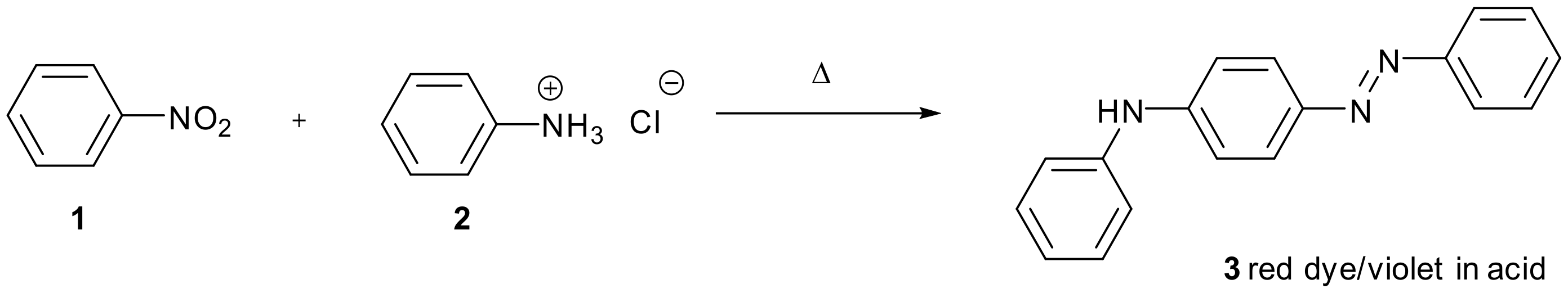
The condensation of amines or carbanions with nitro-substituted aromatic compounds where hydrogen is substituted is comparatively rare. Figure 1 shows an early example from the 19th century in which the reaction of aniline with nitrobenzene is catalysed by acid [1]. A total of 56 pounds of aniline hydrochloride 2 was reacted with 44 pounds of nitrobenzene 2 to produce red dye 3.
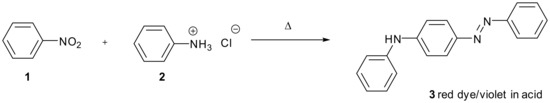
Figure 1.
Acid catalysed reaction of nitrobenzene 1 with aniline hydrochloride 2 forming red dye 3 which is violet in acid.
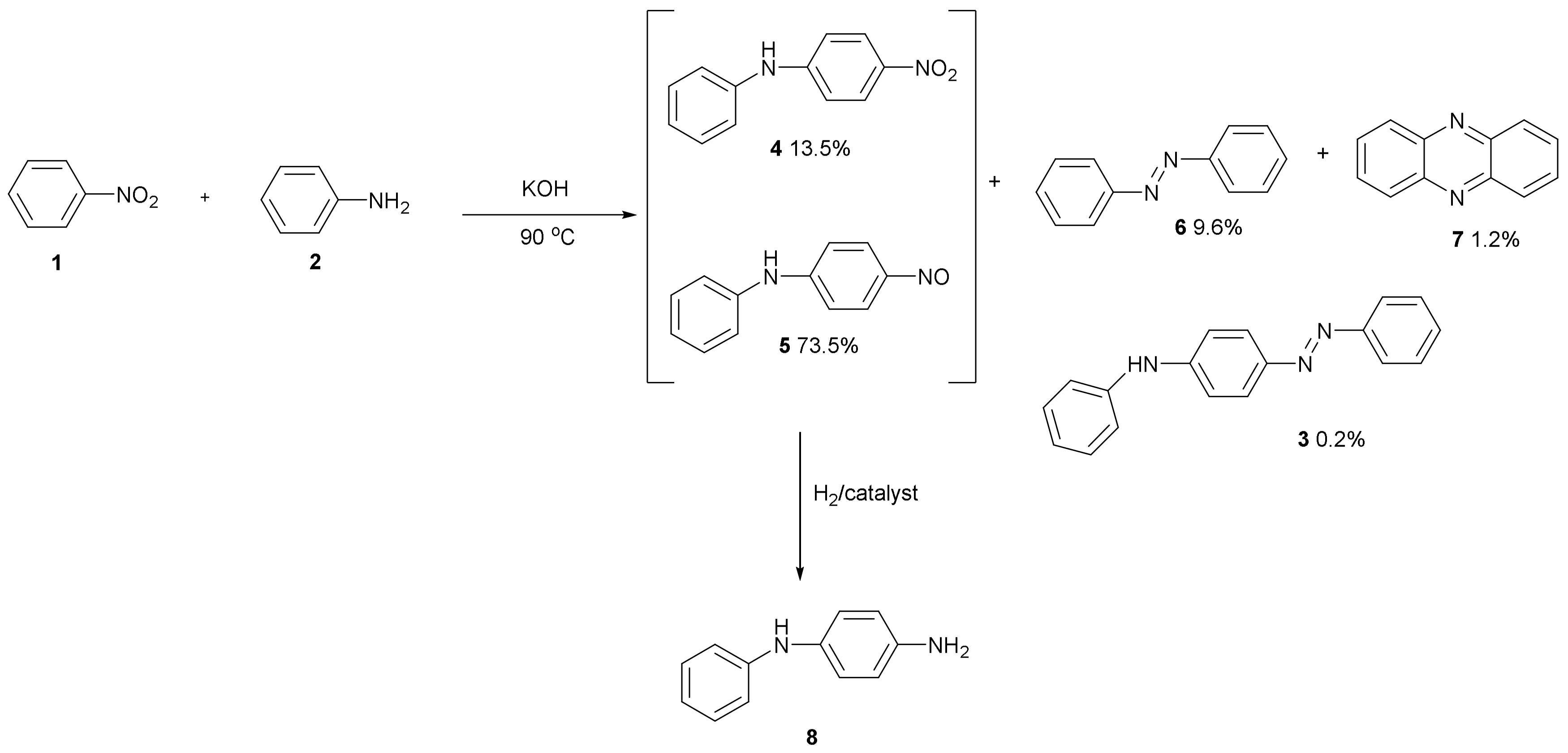
Figure 2 shows the products which form—4-nitrodiphenylamine 4 and 4-nitrosodiphenylamine 5—from the KOH base-catalysed reaction of nitrobenzene 1 with aniline 2 along with azobenzene 6, phenazine 7 and (phenylamino)azobenzene 3 [2]. The catalytic hydrogenation of products 4 and 5 produces anti-oxidant 8. This has been used for making pseudo-mauveine which is mauveine lacking any methyl groups [3].
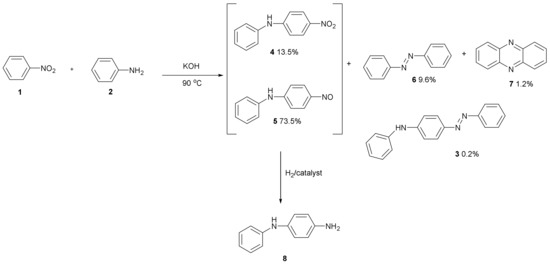
Figure 2.
Anti-oxidant 8 formed from the KOH catalysed reaction of aniline with nitrobenzene followed by catalytic hydrogenation of products 4 and 5.
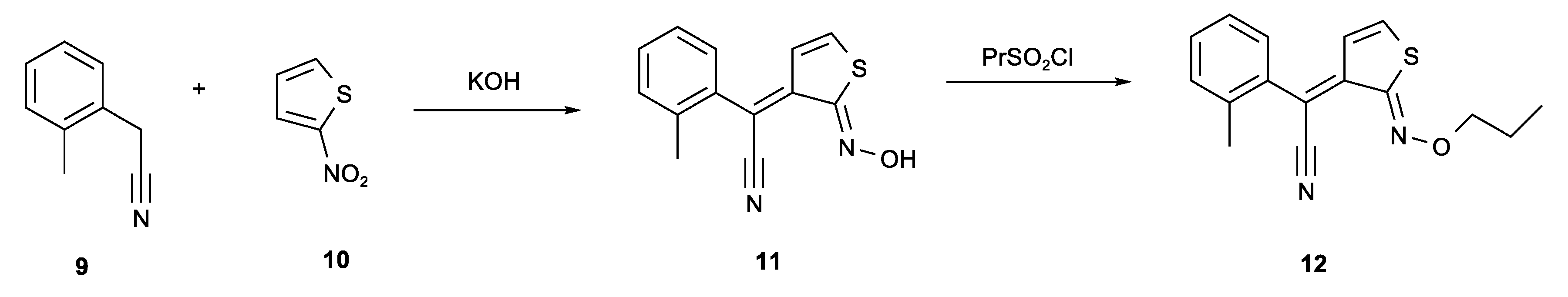
Figure 3 shows a synthesis of a photoacid generator called Irgacure PAG 103 [4,5]. The reaction of o-xylyl cyanide 9 and 2-nitrothiophene 10 with KOH gives adduct 11 which can be sulfonated to produce photoacid generator 12. The photochemical decomposition, which involves an unusual 6π photocyclisation of the C=N of an oxime leading to an acridine derivative and acid, has been explored [6,7]. Heterocyclic examples with a similar synthesis to compound 11 are also known [8]. When the carbanion of compound 9 reacts with the 2-nitrothiophene 10, the elimination of the ring hydrogen of the tetrahedral carbon leads to the reduction of the nitro group to a nitroso group.
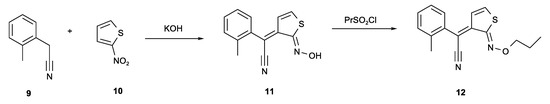
Figure 3.
KOH catalysed condensation of o-xylyl cyanide 9 with 2-nitrothiophene 10 on route to photoacid generator PAG 103 12.
2. Discussion
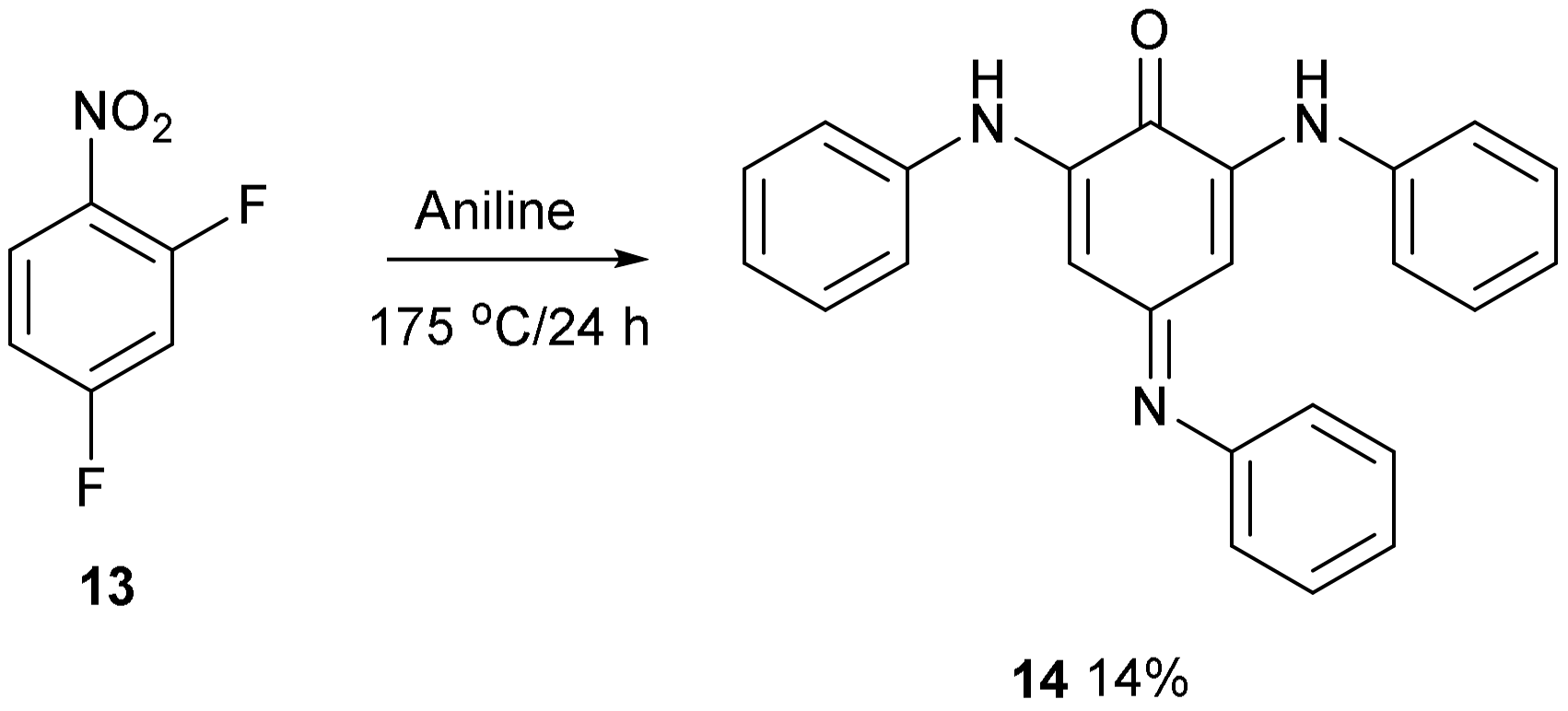
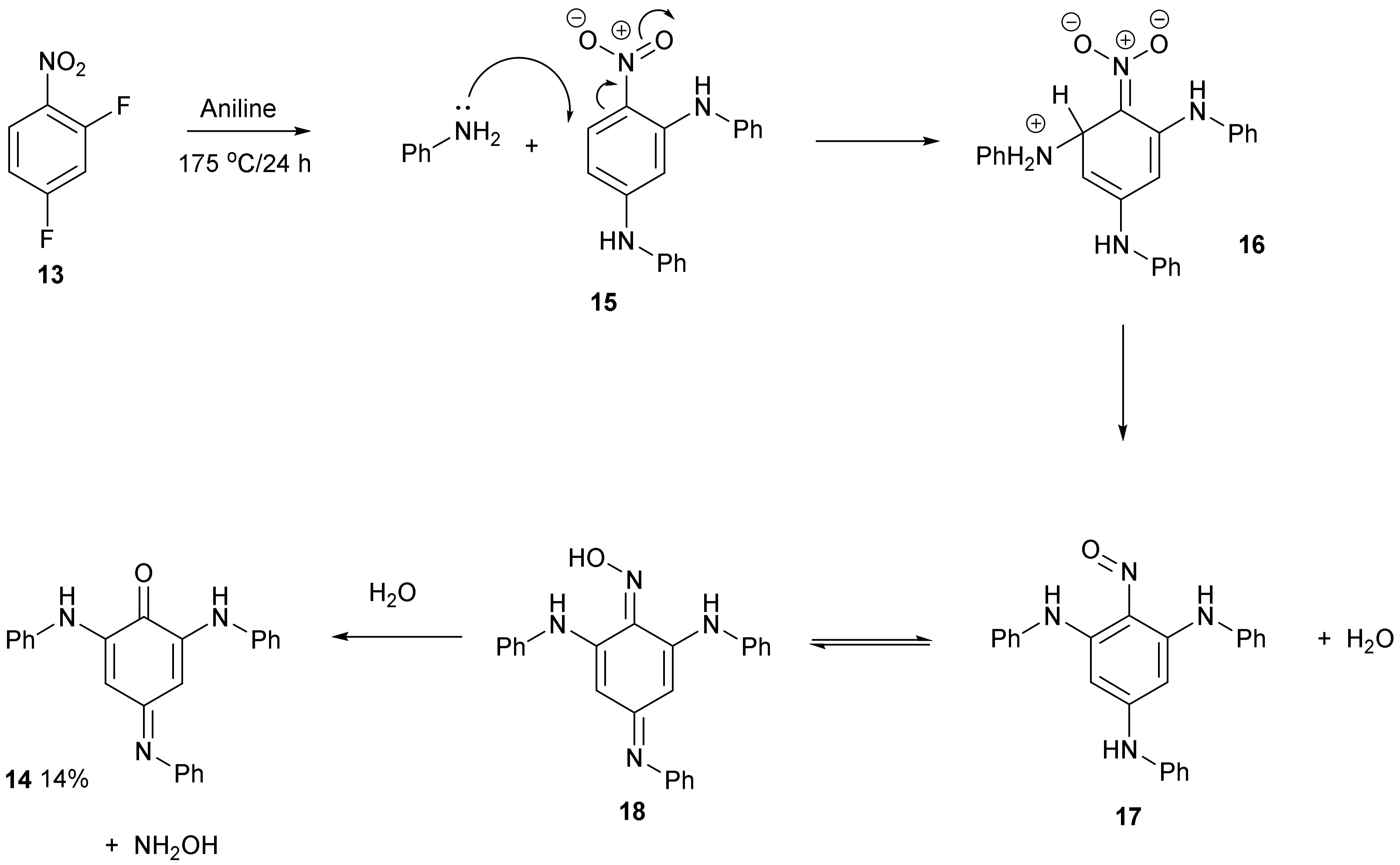
Treatment of 2,4-difluoronitrobenzene 13 with aniline at 175 °C followed by purification on a silica column produced compound 14 as a brown solid in 14% yield. Nitrobenzenes substituted with amino groups are usually yellow coloured [9,10], so this compound was a different chromophore. The UV/Vis spectrum showed a strong absorption at 383 nm and a weaker broad absorption at 480–600 nm which accounts for the stronger colour. It had 18 carbon resonances in the 13C NMR spectrum with one carbonyl group at 181 ppm and the 1H NMR spectrum had 17 aromatic protons. Two broader resonances were observed further downfield at 7.68 and 8.69 ppm and were assigned to the two NH hydrogens. There were two sharp resonances at 6.21 and 6.22 ppm which were assigned to the two quinone hydrogen atoms. The accurate mass gave a formulae of C24H19N3O. The supplementary materials (Figures S1–S5) have the proton and carbon spectra and the UV/Vis spectrum (Figure S6).
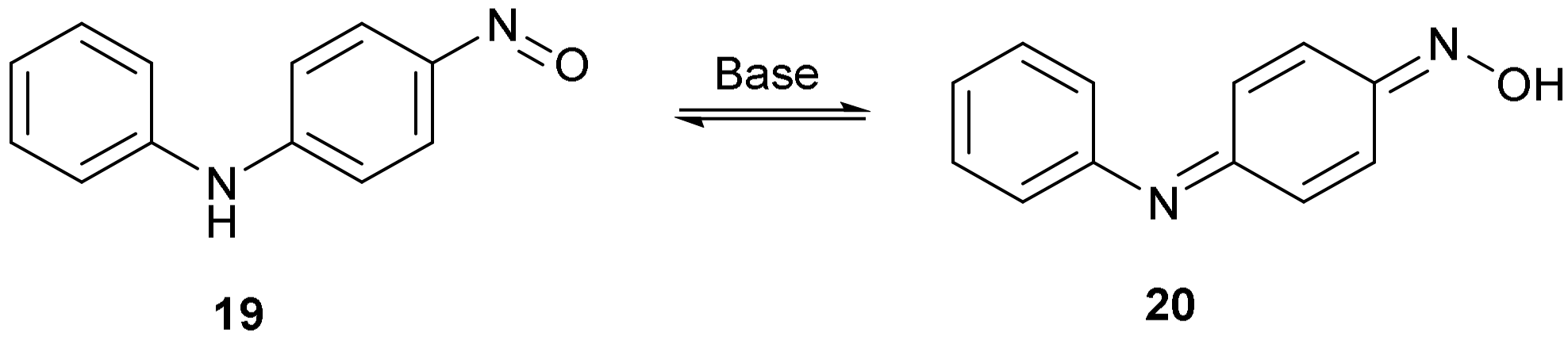
The structure of compound 14 (Figure 4) was deduced from the spectroscopic data and the anticipated chemistry which is shown in Figure 5. Initially, aniline was expected to displace the two fluorine atoms to give 2,4-bis(phenylamino)nitrobenzene 15. Forceful conditions are required because aniline is not a strong nucleophile for displacing the second fluorine of 2,4-difluoronitrobenzene 13. Only one site ortho to the nitro group remained unsubstituted. In line with the previous literature reactions of nitrobenzene 1 and aniline 2 [1,2], aniline may attack the unsubstituted site, thereby producing intermediate 16, followed by the elimination of hydrogen from the tetrahedral carbon which reduces the nitro group to a nitroso group forming intermediate 17 and water. The tautomerism of compound 17 to compound 18 followed by the hydrolysis of the oxime will produce a final product of 14. 4-Nitrosodiphenylamine 19 undergoes a similar type of base-catalysed isomerism to oxime 20 (Figure 6) [11].
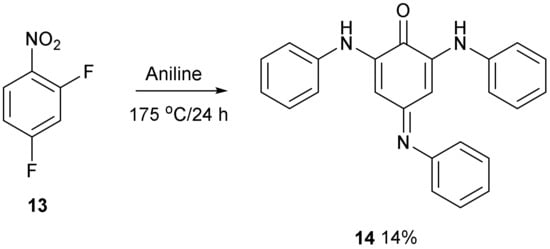
Figure 4.
Synthesis of compound 14.
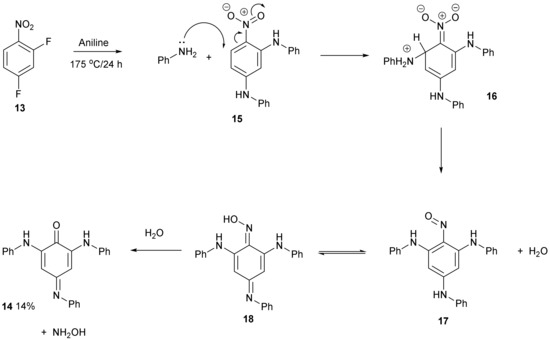
Figure 5.
Proposed mechanistic scheme for the formation of compound 14.
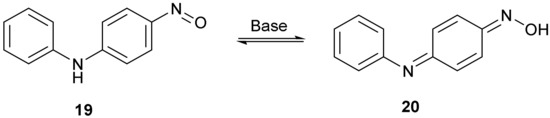
Figure 6.
The base-catalysed tautomerism of 4-nitrosodiphenylamine to an oxime.
3. Materials and Methods
IR spectra were recorded on a Diamond Anvil Fourier transform infrared (FTIR) spectrometer (ThermoScientific, Hemel Hempstead, UK). Ultraviolet (UV) spectra were recorded using a PerkinElmer Lambda 25 UV-Vis spectrometer (PerkinElmer, Coventry, UK) with EtOH as the solvent. The term sh means shoulder. 1H and 13C nuclear magnetic resonance (NMR) spectra were recorded at 400 and 100.5 MHz, respectively, using a Varian 400 spectrometer. Chemical shifts, δ, are given in ppm and were measured by performing a comparison with the residual solvent. Coupling constants, J, are given in Hz. High-resolution mass spectra were obtained at the University of Wales, Swansea, using an Atmospheric Solids Analysis Probe (ASAP) (Positive mode) Instrument: Xevo G2-S ASAP (SpectraLab, Markham, ON, Canada). Melting points were determined on a Kofler hot-stage microscope.
- Synthesis of 2,6-Bis(phenylamino)-4-(iminophenyl)benzoquinone (14)
2,4-Difluoronitrobenzene 13 (500 mg, 3.15 mmol) in aniline 2 (20 mL) was heated at 170 °C for 24 h. The reaction mixture was dark and complex. It was scraped up with a spatula into DCM and applied to a long 12 inch silica column. Elution with DCM gave the title compound (165 mg, 14%) as a brown powder, mp 197–198 °C (from dichloromethane:light petroleum ether). It runs closely with another yellow product. λmax (EtOH)/nm 537sh (log ε 3.9), 383 (4.0) and 276 (4.0); λmax (Diamond)(cm−1) 3259(s), 1632(s), 1573(vs), 1499(vs), 1442(vs), 1292(vs), 1174(s), 847(w), 739(s), 693(vs) and 531(s); δH (400 MHz; CDCl3) 6.21 (1H, s), 6.22 (1H, s), 6.99 (2H, d, J = 4.0), 7.06 (1H, t, J = 8.0 and 8.0), 7.09 (2H, d, J = 8.0), 7.18 (1H, t, J = 8.0 and 8.0), 7.24 (1H, t, J = 8.0 and 8.0), 7.29 (2H, m), 7.36 (2H, d, J = 8.0), 7.42 (2H, t, J = 8.0 and 8.0), 7.45 (2H, J = 8.0 and 8.0), 7.68 (1H, s, NH) and 8.69 (1H, s, NH); δC (100.1 MHz; CDCl3) 90.5, 97.1, 120.8, 120.9, 123.1, 124.0, 124.8, 125.4, 129.0, 129.4, 129.6, 137.9, 138.6, 140.7, 149.4, 149.6, 153.8 and 181.1 (C = 0); m/z (Orbitrap ASAP) 366.1607 (M + H+, 100%) C24H19N3O + H+ requires 366.1606.
4. Conclusions
We previously demonstrated that a low 4% yield of 2,4-bis(phenylamino)nitrobenzene 15 can be obtained from the reaction of an excess of aniline with 2,4-difluoronitrobenzene 13 [12]. Efforts to improve this reaction have not improved the yield but instead led to the isolation of an unexpected brown solid characterised by spectroscopic methods as a quinone substituted with three phenylamino groups. There is only one similar compound reported in the literature which is an isomer of compound 14 and can be formed by an enzyme-catalysed condensation of aniline with substituted catechols or by other methods [13].
Supplementary Materials
The following supporting information can be downloaded online: Molfile of Compound 14; Figure S1: 400 MHz Proton NMR of compound 14 in CDCl3; Figure S2: 400 MHz Proton NMR of compound 14 in CDCl3 Expansion (15 protons); Figure S3: 400 MHz Proton NMR of compound 14 in CDCl3 Scale 0–12 ppm; Figure S4: 400 MHz 13 Carbon NMR of compound 14 in CDCl3 (18 peaks); Figure S5: 400 MHz 13 Carbon NMR of compound 14 in CDCl3 Scale 0–200 ppm; Figure S6: UV/Vis spectrum of compound 14 in EtOH.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The University of Aberdeen Development Trust for funding.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of the compounds are not available from the authors.
References
- Holliday, J. Preparing Colouring Matters. GB Patent 2564, 6 October 1865. [Google Scholar]
- Beska, E.; Toman, P.; Fiedler, K.; Hronec, M.; Pinter, J. Method of Preparation of 4-Aminodiphenylamine. U.S. Patent 6,388,136 B1, 14 May 2002. [Google Scholar]
- Plater, M.J. A synthesis of pseudo-mauveine and a homologue. J. Chem. Res. 2011, 35, 304–309. [Google Scholar] [CrossRef]
- Ohsawa, Y.; Takemura, K.; Seki, A. Photoacid Generators, Chemically Amplified Resist Compositions, and Patterning Process. U.S. Patent 7494760 B2, 24 February 2009. [Google Scholar]
- Ohsawa, Y.; Maeda, K.; Watanabe, S. Photoacid Generators, Chemically Amplified Resist Compositions, and Patterning Process. U.S. Patent 0292768 A1, 20 December 2007. [Google Scholar]
- Plater, M.J.; Williamson, W.T.A.; Raab, A. Photochemical fragmentation of Irgacure PAG 103. ACS Omega 2019, 4, 19875–19879. [Google Scholar] [CrossRef] [PubMed]
- Asakura, T.; Yamato, H.; Tanaka, K.; Takahashi, R.; Kura, H.; Nakano, T. Studies on photodecomposition of an oxime sulfonate. J. Photopolym. Sci. Technol. 2014, 27, 227–230. [Google Scholar] [CrossRef][Green Version]
- Rad, N.I.; Teslenko, Y.O.; Obushak, M.D.; Matiychuk, V.S.; Lytvyn, R.Z.J. Oximes as products in the reactions of 5-substituted 2-nitrothiophenes with arylacetonitriles. J. Heterocycl. Chem. 2011, 48, 1371–1374. [Google Scholar] [CrossRef]
- Chuckowree, I.; Syed, M.A.; Getti, G.; Patel, A.P.; Garner, H.; Tizzard, G.J.; Coles, S.J.; Spencer, J. Synthesis of a 1,3,5-benzotriazepine-2,4-dione based library. Tetrahedron Lett. 2012, 53, 3607–3611. [Google Scholar] [CrossRef]
- Mortzfeld, F.B.; Pietruszka, J.; Baxendale, I.R. A Simple and Efficient Flow Preparation of Pyocyanin a Virulence Factor of Pseudomonas aeruginosa. Eur. J. Org. Chem. 2019, 2019, 5424–5433. [Google Scholar] [CrossRef]
- Lord MFG CO ± (Lord Manufacturing Company). Reacting Oximes with Nitric Oxide and the Products of the Reaction. GB991620A, 12 May 1965. [Google Scholar]
- Plater, M.J.; Harrison, W.T.A. A paddle-wheel motif versus an extended network: Two crystalline forms of 2,4-bis(phenylamino)nitrobenzene. J. Chem. Res. 2015, 39, 98–104. [Google Scholar] [CrossRef]
- Pyne, S.G.; Truscott, R.J.W. Model studies for insect protein sclerotisation: Oxidative loss of the side chain from 4-substituted catechols. Tetrahedron 1990, 46, 661–670. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).