Abstract
Bovine breeding began in Peru more than five centuries ago; since then, adaptation has started. Nowadays, Creole cattle are part of economic activities of Peruvian farmers, as they supply food and profits, among other things, for families in Andean region. Creole cattle have many strengths such as resistance to diseases, low nutrient requirements and easy adaptation to different levels of altitude and environmental conditions. However, even with all these attributes, they are not valued within production systems. Moreover, these valuable genetic resources are disappearing gradually in Perú. For this reason, this study aims to characterize the zoometry of Creole cows from the Southern Amazonas region of Peru. Biotype classification was performed by hierarchical cluster and multivariate factors analysis. Qualitative traits were analyzed with descriptive statistics and Duncan’s test (α = 0.05) was used to compare means among groups. Associations between qualitative traits were determined with Chi-square. We distinguished three biotypes of Creole cows with characteristics for meat and milk production. This information is helpful for future conservation programs for Creole cattle.
1. Introduction
The origin of the Creole bovine in Peru started in 1493 when Christopher Columbus introduced the bovine species in America. Since then, adaptation to diverse Peruvian ecosystems has started [1]. Over the centuries, Creole bovine went through numerous random crosses generating not only diversity in phenotypes but also multiple adaptive advantages compared to specialized bovine breeds (Holstein, Brown Swiss, Angus, Simmental), such as lower nutrient requirements in their diet, lower disease susceptibility, higher fertility rates, and greater longevity under adverse environmental conditions [2]. However, creole bovine is undergoing genetic erosion due to the introduction of specialized breeds [3,4]. The identification of Creole bovines based on their diversity is conceptualized as phenotypes [5]. Their natural environment influences specific zoometric traits and gives evidence of diverse phenotypes such as coat coloring pattern, head characteristics, horns present or absent, body frame, among others [6]. Therefore, given such advantages and environmental variation in the American continent, the Creole bovine has been zoometrically characterized in countries such as Mexico [7,8], Chile [9], Argentina [10], Colombia [11], Ecuador [12], Uruguay [13,14], and Venezuela [2]. All these data constitute valuable information for identifying biotypes and their zootechnic indices.
In Peru, Creole bovine are of great interest for the rural farmers’ economy in Andean region, supplying food (meat and milk), profits, among others [15,16]. Although, introduction of specialized breeds such as Holstein, Jersey, Brown Swiss, Angus, Brangus, Simmental, intends to increase productivity, they are more susceptible to harsh environmental conditions. Therefore, multi-functionality, genetic value, and socio-economic relevance of Creole bovine, are advantages that should be preserved. In Amazonas region, one study was carried out in six dairy basins: Molinopampa and Olleros (Chachapoyas province), Florida, and Progreso (Bongará province) and Huambo and Limabamba (Rodríguez de Mendoza province), where the Creole bovine’s population was higher in quantity than specialized breeds such as Simmental, Holstein, and their crosses [17]. However, Creole bovine population is decreasing in Amazonas region, despite the fact that they provide economic resources for rural families, they require less investment in sanitary treatments than specialized breeds [18]. Also, they are very heterogeneous, represented by many morphotypes that have been scarcely studied [19]. For this reason, we aim to identify the zoometric characteristics of the southern Amazonas region’s Creole cows.
2. Materials and Methods
2.1. Location
This study was carried out in three provinces of the southern Amazonas region where Creole cows are raised: Chachapoyas, Luya, and Bongará (Table S1; Figure S1). In these districts, cows graze only Dactylis glomerata L, Trifolium repens, Pennisetum clandestinum, and Philoglossa mimuloides natural grassland.
2.2. Zoometric Indices
Zoometric indices from local Creole cows from Chachapoyas, Luya and Bongará (29, 37 and 29 cows, respectively) were registered. Measurements were recorded early in the morning at 7:00 to fasting animals following the guidelines of ARRIVE 2.0 (Animal Research: Reporting of In Vivo Experiments) available at https://arriveguidelines.org/ (accessed on 14 January 2021).
The cows were classified according to dental age (six teeth, eight teeth and full mouth) [20] and lactations number (first, fourth or more lactations) provided by the owners.
2.2.1. Coat Coloring Pattern
The coat coloring pattern was classified as simple (one color), compound (more than two defined colors, including spots), or mixed (gradient color scheme with no defined limits). The coat color was registered as black (A), reddish bay (B), light brown (C), dark brown or muddy (D, E), white moor (F), brindle (G), black overo “callejón” (H), smoky “cardeno” (I), mulatto (J), pinto (K) [21,22]. In addition to these, local color denominations were registered, such as nevado rosillo (L), jalmada (M), frijol (N), casullo (O) or bay (P) (Figure 1). Also, we distinguish the hoof (dark or light), and horns tip (black, dark, or amber) colors, and the presence or absence of horns [21,22].

Figure 1.
Coat coloring patterns of Creole cows from the southern Amazonas region.
2.2.2. Quantitative and Qualitative Traits
Local farmers use Creole bovine for mixed purposes (tractive force, milking, and meat) and they manage the selection criteria. Therefore, Creole bovines do not have a standard linear breeding description. So, we used the morphometric traits from the Fleckvieh bovine linear breeding description (Fleckscore) available on the web https://www.fleckscore.com/ (accessed on 18 November 2020) and [23,24]. Defects in udder, frame, and feet and leg were also registered (Table S3). Moreover, measurements of structure (hight at cross, body depth, hip width, back length, and rump length), udder (teats length, teats thickness, fore udder length, udder depth, central ligament), musculature (concave, slightly concave, straight, slightly convex/normal, convex/thin) and feet and legs (hock angularity, hock development, and pasterns) were considered (Table S2). Finally, a body condition score was used to assess the nutritional status (Table S4) [24].
2.3. Statistical Analysis
Creole cows were classified into biotypes in a dendrogram by hierarchical grouping of qualitative and quantitative traits using cluster analysis. Ward’s algorithm and Mahalanobis distance were used to construct the tree, and then a cut at 60% of the largest distance was made to establish the clusters. Through a multivariate factor analysis, we got the correlations between the analyzed traits. The Kaiser-Meyer-Olkin assumption (KMO = 0.4) was evaluated, indicating a relative sub structuration in the study population, and partial correlation between the identified variables. According to Bartlett sphericity test (p < 0.001), it is suitable to apply the factorial model in multivariate analysis. To compare biotypes, an ANOVA (p < 0.05) and Duncan’s multiple comparisons test was used to check differences between clusters. Associations among traits were determined with Chi-square test (95% confidence interval). In addition, quantitative and qualitative traits according to age and lactation were analyzed with descriptive statistics, ANOVA, Duncan’s test (α = 0.05), and Chi-square test. All statistical analyzes were carried out in SPSS v.15 program.
3. Results and Discussion
3.1. Byotipe Classification
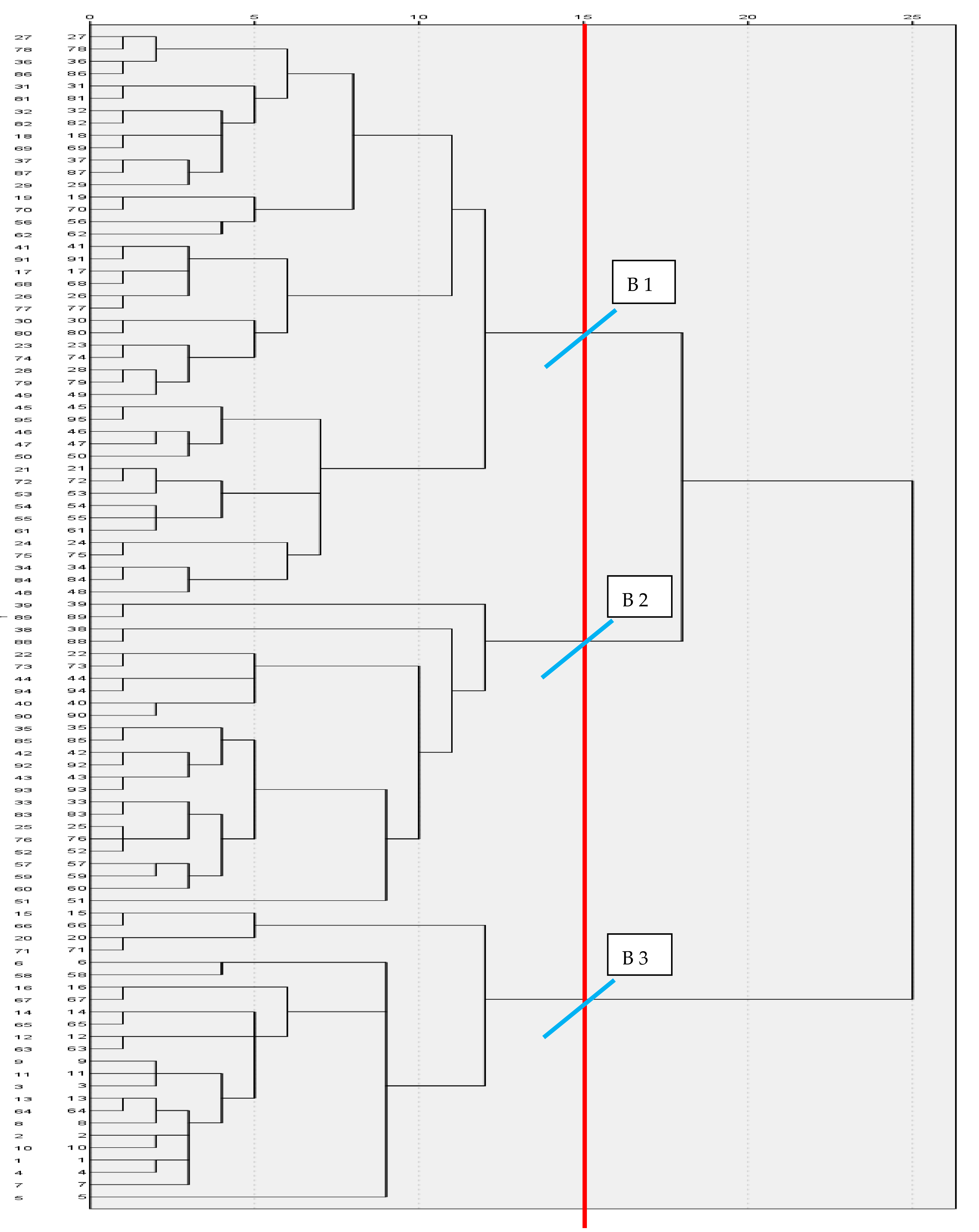
In this study, three biotypes of Creole cows were differentiated: biotypes I, II, and III grouped 46, 20, and 29 cows, respectively (Figure 2). The validation of biotypes differences by analysis of variance of hypothesis test, was highly significant (p < 0.001), which showed that there were differences among biotypes (Table S5). South American creole bovine has evolved by natural selection and adapted to different conditions such as humid tropical forests, subtropical dry forests, and mountainous and Patagonian steppe. Nowadays, most South and Central American countries have Creole bovine with specific characteristics (milk, meat, or dual purpose) with broad genetic diversity and phenotypic variability. Unfortunately, in recent years, there has been a drastic reduction in their population [25,26]. Similarly, four biotypes of Creole bovine were identified in the Patagonian (Northeast of Argentina), differentiating northern animals from the southern ones [27].
Figure 2.
Dendrogram that uses the Ward method. Re-scaled distance cluster combination.
Moreover, in another research Chilean Patagonian Creole bovines were grouped according to their meat or milk production characteristics [28]. In our case, we found Creole cows from the Amazonas region that have a slight inclination for meat and dual purpose (meat and milk). Same as ours, three Creole bovine biotypes from Argentina showed differences in head width, head length, thoracic perimeter, total length, anterior rump width, and rump length [29].
Highly significant differences were found between biotypes found for quantitative traits. Biotype I has greater teats length, fore udder length, central ligament, and heels. Biotype III is characterized by its superiority in body depth, back length, teats thickness, and pelvic tilt with respect to other biotypes (Table 1). Udder depth and body condition did not influence the clusters formation, so the values of these traits were not significantly different among groups.

Table 1.
Quantitative traits of Creole cow biotypes from Amazonas region, Peru 1.
We found four significant and highly significant associations of qualitative traits according to identified biotypes (Table 2). A greater number of biotype I cows have a simple coloration pattern, while in biotype III there is a greater presence of compound color. Creole bovines are characterized by peculiar morphometry and morphology, such as coat color diversity and large horns [30]. Although it is true in this study, the horns’ length was not measured, they were observed in 100% of the population, which agrees with the reports for Uruguayan bovine [30].

Table 2.
Qualitative traits of Creole cow biotypes from Amazonas region, Peru (%) 1.
Front teat placement was significantly associated with biotypes (p < 0.01), biotype II is characterized by slightly protruding teats, biotype III by centered teats, and biotype I by slightly inwardly tucked teats. Rear teat orientation is also significantly associated with biotypes, biotype II presents inside teats and biotype I presents slightly tucked teats. Hock cleanliness was significantly associated with biotypes, where biotype II presents slightly undefined hock cleanliness and biotype III presents clear hock (Table 2).
The description of defects associated with biotypes is detailed in Table 3. Backline impressed, kidney impressed, rump arched, and rolled hoof are associated with biotype III bovines; and chest narrow is associated with biotype II bovines.

Table 3.
Presence of defects in Creole cows biotypes from Amazonas region, Peru (%) 1.
Based on Table 4 resulting from multivariate analysis, the variables can be reduced to six factors or components and explained total accumulative variance of 92.1%. The first component explained 49%, 18% for the second component, 9% for the third component, 6.5% for the fourth component, 5% for the fifth component, and 4% for the sixth component.

Table 4.
Total accumulated variance according to multivariate factor analysis.
Correlation matrix of principal components is detailed in Table 5. The first component highly correlated with fore udder length and udder depth. For the second component, traits with the highest correlation are hip width and rump length. The third component correlates highly with raised to sacrum and rump arched, but body depth shows negative and high correlation. Fourth component correlates highly with hock angle and pelvic tilt, back length, and udder depth, and fifth component correlates highly with coat color and coloring pattern. Lastly, sixth component is highly correlated with back length and pasterns.

Table 5.
Correlation matrix of initial traits according to selected components.
According to Table 5, biotype III can be explained up to 67% by traits of components 1 and 3, biotype II is explained up to 66% by traits of components 1, 2, and 6 and the biotype I can be explained up to 67% by traits of components 1, 4. and 5.
3.2. Age and Lactation of Creole Cow
According to https://www.fleckscore.com/ (accessed on 18 November 2020), the age of Creole cows was significantly associated (p < 0.01) with muscularity, fore udder attachment, and front teats placement (Table 6). As expected, more than 60% of six- and eight- teeth cows and more than 40% of full-mouth cows had straight muscularity. More than 65% of six-teeth cows and more than 50% of eight-teeth cows had a fore udder attachment of 30–40°. Furthermore, more than 40% of full-mouth cows had a fore udder attachment of 40–50°. Finally, 38.7% and 48.8% of eight-teeth and full-mouth cows had centered teats, respectively.

Table 6.
Significant qualitative traits of Creole cows (N = 95) according to age and lactation (%) 1.
Table 4 shows associations of qualitative zoometric traits according to number of lactations. A more significant number of animals with slightly protruding, centered and inward teats were found in second lactation Creole cows (p < 0.01), and more than 60% of third-lactating cows had front teats placement slightly inward. Among the regions of western hemisphere, the characteristics of milking ease, stature, and body condition of the Creole cows differ significantly. Compared to animals initially imported, these differences are due to time, natural selection, and breeding preferences of local breeders [31]. Non-significant associations according to age and lactation are detailed in Table S6).
The Creole cows’ age is significantly associated with absence of defects such as chest narrow, loosely shoulders and front legs distorted (p < 0.05) (Table 7 and Table S7). More than 50% of six-teeth cows did not have chest narrow defect. In the same way, more than 70% and 85% of eight-teeth animals and full-mouth animals did not have chest narrow defect. Moreover, more than 60% of six-teeth animals and full-mouth and more than 85% of eight-teeth cows did not present loosely shoulder defect. Finally, 100% of eight-teeth and a full-mouth and more than 90% of Creole cows do not have front legs distorted defect (p < 0.05). On the other hand, all defects were seen in all animals in different percentages.

Table 7.
Presence of significant defects in structural traits of Creole cows (N = 95) according to age (%) 1.
The presence of defects in Creole cows could be due to absence of selection programs, and also because these animals are displaced to harsh areas with predomination of woody plants, cacti, and native pastures with low levels of nutrients [32,33]. Such is the case of creole cattle within Copper Canyon in Chihuahua, Mexico, isolated by the unlimited road infrastructure. Isolation caused groups with similar characteristics within groups, but different among groups, originating an adaptation process. It was reported that Creole cows show ease of adaptation and survival to harsh conditions, and they can survive with high stress and low nutrients levels [34]. Moreover, all these environmental characteristics cause defects such as short stature or irregular conformation, among others [35]. Lactation number was not associated (p > 0.05) with any of structural defect traits (Table S7).
From fourteen quantitative traits evaluated, only rear udder length was significantly different among ages (p < 0.05), being higher in six-teeth cows than eight-teeth and full-mouth cows (Table 8 and Table S8). The height at cross (125 cm), body depth (70 cm), hip width (45.6), back length (89 cm), and rump length (45 cm) were similar among ages. Research about zoometric evaluation of Creole bovine from Áncash region reported 115 cm in height at withers [36], similar to Creole bovine from Puno region [37] with height 118 cm. Creole cows from Amazonas region has height at cross 125.2 cm, similar to six-teeth and full-mouth Creole cows (122 cm and 124 cm; respectively) from Puno region [37]. Hip width (45.6 cm) and back length (89.1 cm) of Creole cows are higher than hip width and height at withers of Creole bulls of Mixtec region of Mexico (32.8 cm and 66.1 cm, respectively) [7]. Rump length was similar to Chinampo Creole bovine from Mexico (45 cm) [8], and six-teeth and full-mouth (43 cm) in Creole bovine from the Puno region [37]. Quantitative traits are necessary for biotype classification, such as hip width and length, which facilitate easy calving and potential winning of muscle mass [38].

Table 8.
Significant quantitative traits of Creole cows (N = 95) according to age and lactation 1.
Differences between Creole cows of Amazonas region and Creole bovines from other origins could be due to agroclimatic and nutritional factors. Moreover, phenotypic differences between bovines of different latitudes and altitudes could be due to environmental conditions, breeding purpose, functionality, distances among groups, and adaptation to the agroclimatic factors [5,39].
Regarding the mammary system, Creole cows from Amazonas region have averages of 5.7, 2.7, 14.1, 18.4, and 2.2 cm for teat length, teat thickness, fore udder length, udder depth, and central ligament, respectively. Hoof height varied from 2.6 to 2.8 cm, rump angle was 5 cm, and body condition of 2.7 points (BC2). This poor body condition was due to limited technical management and food quality intake, leading to low daily weight gains [12]. A body condition score from 1 to 2.5 is associated with parasites accumulators; meanwhile, animals with body condition score higher than 3 points are considered resilient to diseases [40].
According to lactations number, differences between hip width and rump length were significant (p < 0.05). Cows with more than two lactations have 2 cm wider hip than first-lactating cows (Table 8). Similar phenomenon happens when evaluating rump length. Cows with more than two lactations have greater rump length than first-lactating cows (p < 0.05). Regarding the hip width, Mixtec bovine has average hip width of 32.82 cm [7], lower than Creole cows from Amazonas region; this is possible because of different phenotypes and genetic variations [41]. Differences could also be due to natural selection that bovines have undergone; natural selection of bovine with more environmental adaptation skills to wild areas and rough conditions is essential for survival [42,43].
After evaluating the effect of age and lactations number of Creole cows on qualitative and quantitative traits, we determined their association with the classified biotypes through cluster analysis. We found no significant association between age and biotype or lactations number with biotype (Table 9), which supports the hypothesis that our classification of Creole cow biotypes from the Amazon region does not necessarily correspond to these two factors.

Table 9.
Association of age and lactation with Creole cows biotypes (frequency values, %) 1.
4. Conclusions
Three biotypes of Creole cows from the southern Amazonas region were found. Biotype I grouped Creole cows with greater teat length, fore udder length, central ligament length, and heel length, while biotype III grouped Creole cows with superiority in body depth, back length, teats thickness and inclination tilt. However, biotype II animals are associated with a simple coloration pattern, slightly protruding teats, and slightly indefinite hock cleanliness. Multivariate factor analysis contributed to reducing the variables in six components, where biotype III can be explained by traits of components 1 and 3 (67%), biotype II by traits of components 1, 2, and 6 (66%) and biotype I by traits of components 1, 4 and 5 (67%).
Six-tooth Creole cows had greater rear udder length and first-lactation cows had lower hip width and rump length. In addition, age was associated with muscularity, fore udder attachment and front teat placement, and lactation was associated with front teat placement. However, age and lactation number groups were not associated with biotype groups conformed.
This research allowed us to differentiate the Creole cows of the southern Amazonas region. We observed cows with meat potential (with greater body depth and back length), dairy potential (animals without udder defects, suitable udder attachment, and well positioned teats), and dual purpose. The phenotypic variability observed in this study could be related to polymorphisms associated with production parameters; therefore, molecular analyses will be needed to reinforce this characterization. Moreover, this work contributes to generating information for future conservation programs and using this valuable genetic resource.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/d13110510/s1, Figure S1: Geographical location of Chachapoyas, Luya and Bongará province in Amazonas. Table S1: Creole bovine location. Table S2: Qualitative and quantitative traits of creole bovine. Table S3: Defects of female Creole cows. Table S4: Body Condition [24]. Table S5: Validation of the clusters or cow biotypes by ANOVA, KMO and Bartlett test. Table S6: Qualitative traits of creole cows (N = 95) according to age and lactation (%). Table S7: Presence of defect in structure traits of Creole cow (N = 95) according to age and lactation (%). Table S8: Quantitative traits of Creole cows (N = 95) according to age and lactation.
Author Contributions
Conceptualization, R.E.R. and I.S.C.-C., S.M.P.-V. and R.E.R., data analysis H.A.Q.-C. and J.A.S.-U., writing the original draft, J.A.S.-U. and R.E.R., Final drafting, revision and editing, I.S.C.-C. and J.A.S.-U., Funding Acquisition, I.S.C.-C., Proyect Administration, I.S.C.-C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was written in the context of the project “Caracterización molecular de genes asociados con la terneza de la carne de bovinos criollos de la región Amazonas” funded by Programa Nacional de Investigación Científica y Estudios Avanzados (PROCIENCIA), grant number 109-2018-FONDECYT.
Institutional Review Board Statement
Our study did not carry out activities that generate stress to the animals, so it did not apply to make a request to the ethics committee of our institution, even so, we adhere to the ethics guidelines of ARRIVE 2.0 (Animal Research: Reporting of In Vivo Experiments) available at https://arriveguidelines.org/ (accessed on 17 October 2021).
Informed Consent Statement
Not applicable.
Data Availability Statement
Data sharing not applicable.
Acknowledgments
The authors are deeply grateful to all the Creole cattle producers from the different provinces involved in this research.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Primo, A.T. The Iberic cattle in the Americas: 500 years later. Arch. Zootec. 1992, 41, 421–432. [Google Scholar]
- Contreras, G.; Chirinos, Z.; Zambrano, S.; Molero, E.; Paéz, A. Caracterización morfológica e índices zoométricos de vacas Criollo Limonero de Venezuela. Rev. Fac. Agron. Univ. Zulia. 2011, 28, 91–103. [Google Scholar]
- Felius, M.; Beerling, M.L.; Buchanan, D.S.; Theunissen, B.; Koolmees, P.A.; Lenstra, J.A. On the history of cattle genetic resources. Diversity 2014, 6, 705–750. [Google Scholar] [CrossRef]
- Mapiye, C.; Chikwanha, O.C.; Chimonyo, M.; Dzama, K. Strategies for sustainable use of indigenous cattle genetic resources in Southern Africa. Diversity 2019, 11, 214. [Google Scholar] [CrossRef]
- Eding, J.H.; Laval, G. Measuring Genetic Uniqueness in Livestock. Genebanks and the Conservation of Farm Animal Genetic Resources; Institute for Animal Science and Health: Lelystad, The Netherlands, 1999; pp. 33–58. [Google Scholar]
- Alderson, L. The categorisation of types and breeds of cattle in Europe. Arch. Zootec. 1992, 41, 325–334. [Google Scholar]
- Méndez, M.; Serrano, J.S.; Ávila, R.; Rosas, M.; Méndez, N. Caracterización morfométrica del bovino criollo mixteco. Arch. Zootec. 2002, 51, 217–221. [Google Scholar]
- Espinoza, J.L.; Guevara, J.A.; Palacios, E.A. Caracterización morfométrica y faneróptica del bovino Criollo Chinampo de México. Arch. Zootec. 2009, 58, 277–279. [Google Scholar] [CrossRef]
- Aracena, M.; Mujica, F. Caracterización del bovino criollo patagónico chileno. un estudio de caso. Agro Sur. 2011, 39, 106–115. [Google Scholar] [CrossRef]
- Martínez, R.D.; Fernández, E.N.; Género, E.R.; Rumiano, F.J. El ganado bovino criollo en Argentina. Arch. Zootec. 2000, 49, 353–361. [Google Scholar]
- Mahecha, L.; Angulo, J.; Manrique, L.P. Estudio bovinométrico y relaciones entre medidas corporales y el peso vivo en la raza Lucerna. Rev. Colomb. Cienc. Pecu. 2002, 15, 80–87. [Google Scholar]
- Aguirre, L.; Bermeo, A.; Maza, D.; Merino, L. Estudio fenotípico y zoométrico del bovino criollo de la sierra media y alta de la región sur del Ecuador (RSE). Actas Iberoam. Conserv. Anim. 2011, 1, 392–397. [Google Scholar]
- Rodríguez, M.; Fernández, G.; Silveira, C.; Delgado, J.V. Estudio étnico de los bovinos criollos del Uruguay: I. Análisis biométrico. Arch. Zootec. 2001, 50, 113–118. [Google Scholar]
- Fernández, G.; Rodríguez, M.; Silveira, C.; Barba, C. Estudio étnico de los bovinos criollos del Uruguay: II. Análisis de las faneras. Arch. Zootec. 2001, 50, 119–124. [Google Scholar]
- FAO. Food and Agriculture Organization of the United Nations. Segundo Informe Sobre la Situación de los Recursos Zoogenéticos Mundiales para la Alimentación y Agricultura. 2015. Available online: http://www.fao.org/3/i5077s/i5077s.pdf (accessed on 1 April 2021).
- Aguirre, L.A.; Apolo, G.; Chalco, L.; Martínez, A. Caracterización genética de la población bovina criolla de la Región Sur del Ecuador y su relación genética con otras razas bovinas. Anim. Genet. Resour. Inf. 2014, 54, 93–101. [Google Scholar] [CrossRef]
- Murga, L.; Vasquez, H.; Bardales, J. Caracterización de los sistemas de producción de ganado bovino en las cuencas ganaderas de Ventilla, Florida y Leyva-región Amazonas. Rev. Investig. Científica UNTRM Cienc. Nat. Ing. 2018, 1, 28–37. [Google Scholar]
- Julon, D.; Puicón, V.; Chávez, A.; Bardales, W.; Conzales, J.; Vasquez, H.; Maicelo, J. Prevalencia de Fasciola hepatica y parásitos gastrointestinales en bovinos de la Región Amazonas, Perú. Rev. Inv. Vet. Perú 2020, 31, e17560. [Google Scholar] [CrossRef]
- Rivas, E.; Veli, E.; Aquino, Y.; Rivas, V.; Pastor, S.; Estrada, R. Acciones para la caracterización y conservación del bovino criollo Peruano (Bos taurus). Anim. Genet. Resour. 2007, 40, 33–42. [Google Scholar] [CrossRef]
- Luengo, L.J.; Aros, I.C.; Gómez, R.L. Determinación de la edad del bovino según las características morfológicas de los dientes incisivos. Contribución a la aplicación de la norma chilena 1423 Of. 84. Terminología y clasificación. Av. Cienc. Vet. 1990, 5. [Google Scholar] [CrossRef]
- Prieto, J.L.; Del Pino, J. Guía de Campo del Toro de Lidia. Pintas, Particularidades y Encornaduras, 1st ed.; Almuzara: Córdoba, Spain, 2013. [Google Scholar]
- FAO. Food and Agriculture Organization of the United Nations. La Situación de los Recursos Zoogenéticos Mundiales para la Alimentación y la Agricultura. Rischkowsky, B., Pilling, D., Eds.; 2010. Available online: http://www.fao.org/3/a1250s/a1250s.pdf (accessed on 14 March 2021).
- FAO. Food and Agriculture Organization of the United Nations. Draft Guidelines for the In Vivo Conservation of Animal Genetic Resources. 2012. Available online: http://www.fao.org/3/me879e/me879e.pdf (accessed on 9 April 2021).
- Sánchez, L.; Iglesias, A. Valoración morfológica en bovinos de aptitud cárnica y razas rústica. In Valoración Morfológica de los Animales Domésticos; Ministerio de Medio Ambiente y Medio Rural y Marino: Madrid, Spain, 2009; pp. 275–304. [Google Scholar]
- De Luca, J.C.; Zufriategui, L.; Picco, S.J.; Ripoli, M.V.; Giovambattista, G.; Rojas, F.V.; Dulout, F.N. Incidence of 1/29 translocation in Bolivian Creole and Brahman Yacumeño cattle. Theriogenology 2002, 58, 1273–1281. [Google Scholar] [CrossRef]
- Giovambattista, G.; Takeshima, S.N.; Ripoli, M.V.; Matsumoto, Y.; Franco, L.A.; Saito, H.; Onuma, M.; Aida, Y. Characterization of bovine MHC DRB3 diversity in Latin American Creole cattle breeds. Gene 2013, 519, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Martínez, R.; Fernández, E.; Abbiati, N.; Broccoli, A. Caracterización zoométrica de bovinos criollos: Patagónicos vs. noroeste argentino. Rev. MVZ Córdoba 2007, 12, 1042–1049. [Google Scholar] [CrossRef]
- Piñeira, J.; Mujica, F.; Felmer, R.; Ortiz, M.; Pizarro, G.; Aracena, M. Caracterización genética de un rebaño de bovino criollo patagónico chileno. Agro Sur. 2011, 39, 46–56. [Google Scholar] [CrossRef]
- Martínez, R.D.; Fernández, E.N.; Género, E.; Broccoli, A. Avances en la Caracterización Genética y Morfológica del Bovino Criollo de Origen Patagónico. 2006, pp. 1–6. Available online: https://www.produccion-animal.com.ar/informacion_tecnica/raza_criolla/30-caracterizacion.pdf (accessed on 10 January 2021).
- Rincón, G.; D’angelo, M.; Gagliardi, R.; Kelly, L.; Llambí, S.; Postiglioni, A. Genomic polymorphism in Uruguayan Creole cattle using RAPD and microsatellite markers. Res. Vet. Sci. 2000, 69, 171–174. [Google Scholar] [CrossRef]
- Rouse, J.E. The Criollo: Spanish Cattle in the Americas; University of Oklahoma Press: Norman, OK, USA, 1977. [Google Scholar]
- Ortega-Ochoa, C.; Villalobos, C.; Martínez-Nevárez, J.; Britton, C.M.; Sosebee, R.E. Chihuahua’s cattle industry and a decade of drought: Economical and ecological implications. Rangelands 2008, 30, 2–7. [Google Scholar] [CrossRef]
- Anderson, D.M.; Estell, R.E.; Gonzalez, A.L.; Cibils, A.F.; Torell, L.A. Criollo cattle: Heritage genetics for arid landscapes. Rangelands 2015, 37, 62–67. [Google Scholar] [CrossRef]
- Ulloa-Arvizu, R.; Gayosso-Vázquez, A.; Ramos-Kuri, M.; Estrada, F.J.; Montano, M.; Alonso, R.A. Genetic analysis of Mexican Criollo cattle populations. J. Anim. Breed. Genet. 2008, 125, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Peel, D.S.; Johnson, R.J.; Matthews, K.H., Jr. Cow-Calf Beef Production in Mexico; Economic Research Service Report LDP-M-196-01; United States Department of Agriculture: Washington, DC, USA, 2010.
- Delgado, A.; García, C.; Allcahuamán, D.; Aguilar, C.; Estrada, P.; Vega, H. Caracterización fenotípica del ganado criollo en el Parque Nacional Huascarán-Ancash, Perú. Rev. Inv. Vet. Perú 2019, 30, 1143–1149. [Google Scholar] [CrossRef]
- Rojas, R.; Gómez, N. Biometría y constantes clínicas del bovino Criollo en el centro de investigación y producción Chuquibambilla de Puno (Peru). Arch. Zootec. 2005, 54, 233–236. [Google Scholar]
- Aguirre-Riofrio, E.L.; Abad-Guamán, R.M.; Uchuari-Pauta, M.D. Morphometric Evaluation of Phenotypic Groups of Creole Cattle of Southern Ecuador. Diversity 2019, 11, 221. [Google Scholar] [CrossRef]
- Van, H.T. Drowning in the Genepool: Managing Genetic Diversity in Genebank Collections. Ph.D. Thesis, Swedish University of Agricultural Scienses, Svalov, Sweden, 1994. [Google Scholar]
- Morales, G.; Pino, L.A.; Sandoval, E.; Florio, J.; Jiménez, D. Niveles de infestación parasitaria, condición corporal y valores de hematocrito en bovinos resistentes, resilientes y acumuladores de parásitos en un rebaño Criollo Río Limón. Zootec. Trop. 2006, 24, 333–346. [Google Scholar]
- Xu, Y.; Jiang, Y.; Shi, T.; Cai, H.; Lan, X.; Zhao, X.; Plath, M.; Chen, H. Whole-genome sequencing reveals mutational landscape underlying phenotypic differences between two widespread Chinese cattle breeds. PLoS ONE 2017, 12, e0183921. [Google Scholar] [CrossRef] [PubMed]
- Carvalheiro, R.; Costilla, R.; Neves, H.H.; Albuquerque, L.G.; Moore, S.; Hayes, B.J. Unraveling genetic sensitivity of beef cattle to environmental variation under tropical conditions. Genet. Sel. Evol. 2019, 51, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Pierce, M.D.; Dzama, K.; Muchadeyi, F.C. Genetic diversity of seven cattle breeds inferred using copy number variations. Front. Gene 2018, 9, 163. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).