Molecular Insights into the Centaurea Calocephala Complex (Compositae) from the Balkans—Does Phylogeny Match Systematics?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. DNA Extraction, Amplification, Cloning, and Sequencing
2.3. Phylogenetic Analyses
2.4. Chromosome Number
3. Results
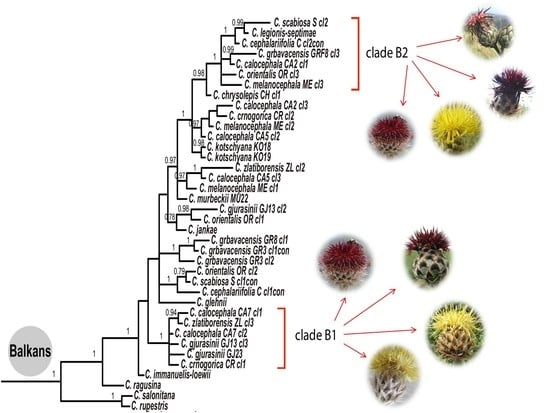
3.1. Phylogenetic Analysis
3.2. Network Analyses
3.3. Chromosome Numbers
4. Discussion
4.1. Utility of Nuclear-Ribosomal ETS and Low-Copy AGT1 in Acrocentron
4.2. Phylogeny of the Balkan Clade: Introgression and Gene Flow
5. Conclusions
- (a)
- The Balkan–Eurasian clade of sect. Acrocentron is a natural group.
- (b)
- The C. calocephala complex as currently defined is not a monophyletic group, given that C. cephalariifolia, C. glehnii, C. legionis-septimae, and C. scabiosa are nested between other species of the complex. All these species should be included in any future study of the complex. Species are well-delimited from a morphological point of view, and they are the result of allopatric speciation in the Balkan Peninsula. In these morphologically well-defined species, successive hybridization events accompanied by introgression and gene flow caused by latitudinal and altitudinal shifts in the transits from glacial to interglacial periods were superimposed.
- (c)
- As a result of this introgression, any reconstruction of the species’ boundaries using sequence data is problematic within the group, and the use of other, more resolving markers should be considered.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Turrill, W.B. The Plant-Life of the Balkan Peninsula: A Phytogeographical Study; Clarendon: Oxford, UK, 1929. [Google Scholar]
- Stevanović, V. Exploration of Balkan Flora after Turrill’s time—the current situation and future challenges. In Proceedings of the 5th Balkan Botanical Congress: Book of Abstracts; Faculty of Biology, University of Belgrade & Serbian Academy of Sciences and Arts: Belgrade, Serbia, 2009; p. 9. [Google Scholar]
- Stevanović, V. Analysis of the central European and Mediterranean orophytic element on the mountains of the w. and central Balkan Peninsula, with special reference to endemics. Bocconea 1996, 5, 77–97. [Google Scholar]
- Savić, I.R. Diversification of the Balkan fauna: Its origin, historical development and present status. In Advances in Arachnology and Developmental Biology; Makarov, E., Dimitrijević, R.E., Eds.; SASA Belgrade UNESCO MAB Serbia: Belgrade, Serbia, 2008; pp. 57–78. [Google Scholar]
- Hewitt, G.M. Post-glacial re-colonization of European biota. Biol. J. Linn. Soc. 1999, 68, 87–112. [Google Scholar] [CrossRef]
- Hewitt, G. The genetic legacy of the Quaternary Ice Ages. Nature 2000, 405, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Petit, R.J.; Aguinagalde, I.; de Beaulieu, J.-L.; Bittkau, C.; Brewer, S.; Cheddadi, R.; Ennos, R.; Fineschi, S.; Grivet, D.; Lascoux, M. Glacial Refugia: Hotspots but not melting pots of genetic diversity. Science 2003, 300, 1563–1565. [Google Scholar] [CrossRef] [Green Version]
- Tzedakis, P.C. The Balkans as prime glacial refugial territory of European temperate trees. In Balkan Biodiversity; Griffiths, H.I., Kryštufek, B., Reed, J.M., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 49–68. [Google Scholar] [CrossRef]
- Médail, F.; Diadema, K. Glacial refugia influence plant diversity patterns in the Mediterranean Basin. J. Biogeogr. 2009, 36, 1333–1345. [Google Scholar] [CrossRef]
- Surina, B.; Schönswetter, P.; Schneeweiss, G.M. Quaternary range dynamics of ecologically divergent species (Edraianthus serpyllifolius and E. tenuifolius, Campanulaceae) within the Balkan refugium. J. Biogeogr. 2011, 38, 1381–1393. [Google Scholar] [CrossRef]
- Lakušić, D.; Liber, Z.; Nikolić, T.; Surina, B.; Kovačić, S.; Bogdanović, S.; Stefanović, S. Molecular phylogeny of the Campanula pyramidalis species complex (Campanulaceae) inferred from chloroplast and nuclear non-coding sequences and its taxonomic implications. Taxon 2013, 62, 505–524. [Google Scholar] [CrossRef] [Green Version]
- Janković, I.; Šatović, Z.; Liber, Z.; Kuzmanović, N.; Radosavljević, I.; Lakušić, D. Genetic diversity and morphological variability in the Balkan endemic Campanula secundiflora s.l. (Campanulaceae). Bot. J. Linn. Soc. 2016, 180, 64–88. [Google Scholar] [CrossRef] [Green Version]
- Janković, I.; Satovic, Z.; Liber, Z.; Kuzmanović, N.; Di Pietro, R.; Radosavljević, I.; Nikolov, Z.; Lakušić, D. Genetic and morphological data reveal new insights into the taxonomy of Campanula versicolor s.l. (Campanulaceae). Taxon 2019, 68, 340–369. [Google Scholar] [CrossRef]
- Ronikier, M.; Zalewska-Gałosz, J. Independent evolutionary history between the Balkan Ranges and more northerly mountains in Campanula alpina s.l. (Campanulaceae): Genetic divergence and morphological segregation of taxa. Taxon 2014, 63, 116–131. [Google Scholar] [CrossRef]
- Bogdanović, S.; Rešetnik, I.; Brullo, S.; Shuka, L. Campanula aureliana (Campanulaceae), a new species from Albania. Plant Syst. Evol. 2015, 301, 1555–1567. [Google Scholar] [CrossRef]
- Aleksić, J.M.; Škondrić, S.; Lakušić, D. Comparative phylogeography of capitulate Campanula species from the Balkans, with description of a new species, C. daucoides. Plant Syst. Evol. 2018, 304, 549–575. [Google Scholar] [CrossRef]
- Bogdanović, S.; Brullo, S.; Rešetnik, I.; Lakušić, D.; Satovic, Z.; Liber, Z. Campanula skanderbegii: Molecular and morphological evidence of a new Campanula species (Campanulaceae) endemic to Albania. Syst. Bot. 2014, 39, 1250–1260. [Google Scholar] [CrossRef]
- Grdiša, M.; Liber, Z.; Radosavljević, I.; Carović-Stanko, K.; Kolak, I.; Satovic, Z. Genetic diversity and structure of Dalmatian Pyrethrum (Tanacetum cinerariifolium Trevir./Sch./Bip., Asteraceae) within the Balkan refugium. PLoS ONE 2014, 9, e105265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hardion, L.; Baumel, A.; Verlaque, R.; Vila, B. Distinct evolutionary histories of lowland biota on Italian and Balkan Peninsulas revealed by the phylogeography of Arundo plinii (Poaceae). J. Biogeogr. 2014, 41, 2150–2161. [Google Scholar] [CrossRef]
- Perný, M.; Tribsch, A.; Anchev, M.E. Infraspecific differentiation in the Balkan diploid Cardamine acris (Brassicaceae): Molecular and morphological evidence. Folia Geobot. 2004, 39, 405–429. [Google Scholar] [CrossRef]
- Lakušić, D.; Novčić, R.; Kučera, J.; Marhold, K. Cardamine pancicii Hayek (Brassicaceae), a neglected species of the Balkan Peninsula: Morphological and molecular evidence. Willdenowia 2006, 36, 177–191. [Google Scholar] [CrossRef]
- Kučera, J.; Tremetsberger, K.; Vojta, J.; Marhold, K. Molecular study of the Cardamine maritima group (Brassicaceae) from the Balkan and Apennine Peninsulas based on amplified fragment length polymorphism. Plant Syst. Evol. 2008, 275, 193–207. [Google Scholar] [CrossRef]
- Kutnjak, D.; Kuttner, M.; Niketić, M.; Dullinger, S.; Schönswetter, P.; Frajman, B. Escaping to the summits: Phylogeography and predicted range dynamics of Cerastium dinaricum, an endangered high mountain plant endemic to the western Balkan Peninsula. Mol. Phylogenetics Evol. 2014, 78, 365–374. [Google Scholar] [CrossRef]
- Đurović, S.Z.; Temunović, M.; Niketić, M.; Tomović, G.; Schönswetter, P.; Frajman, B.; Lavergne, S. Impact of Quaternary climatic oscillations on phylogeographic patterns of three habitat-segregated Cerastium taxa endemic to the Dinaric Alps. J. Biogeogr. 2021, 48, 2022–2036. [Google Scholar] [CrossRef]
- Stefanović, S.; Lakušić, D.; Kuzmina, M.; Međedović, S.; Tan, K.; Stevanović, V. Molecular phylogeny of Edraianthus (Grassy Bells; Campanulaceae) based on non-coding plastid DNA sequences. Taxon 2008, 57, 452–475. [Google Scholar]
- Lakušić, D.; Rakić, T.; Stefanović, S.; Surina, B.; Stevanović, V. Edraianthus × lakusicii (Campanulaceae) a new intersectional natural hybrid: Morphological and molecular evidence. Plant Syst. Evol. 2009, 280, 77–88. [Google Scholar] [CrossRef]
- Lakušić, D.; Stefanović, S.; Siljak-Yakovlev, S.; Rakić, T.; Kuzmanović, N.; Surina, B. Edraianthus stankovicii (Campanulaceae), an overlooked taxon from the Balkan Peninsula—Evidence from morphometric, molecular and genome size studies. Phytotaxa 2016, 269, 69. [Google Scholar] [CrossRef]
- Surina, B.; Rakić, T.; Stefanović, S.; Stevanović, V.; Lakušić, D. One new species of the genus Edraianthus, and a change in taxonomic status for Edraianthus serpyllifolius f. pilosulus (Campanulaceae) from the Balkan Peninsula. Syst. Bot. 2009, 34, 602–608. [Google Scholar] [CrossRef]
- Jug-Dujaković, M.; Ninčević, T.; Liber, Z.; Grdiša, M.; Šatović, Z. Salvia officinalis survived in situ Pleistocene glaciation in ‘refugia within refugia’ as inferred from AFLP markers. Plant Syst. Evol. 2020, 306, 38. [Google Scholar] [CrossRef]
- Stojanović, D.; Aleksić, J.M.; Jančić, I.; Jančić, R. A Mediterranean medicinal plant in the Continental Balkans: A plastid DNA-based phylogeographic survey of Salvia officinalis (Lamiaceae) and its conservation implications. Willdenowia 2015, 45, 103–118. [Google Scholar] [CrossRef]
- Buzurović, U.; Tomović, G.; Niketić, M.; Bogdanović, S.; Aleksić, J.M. Phylogeographic and taxonomic considerations on Goniolimon tataricum (Plumbaginaceae) and its relatives from south-eastern Europe and the Apennine Peninsula. Plant Syst. Evol. 2020, 306, 29. [Google Scholar] [CrossRef]
- Kuzmanović, N.; Comanescu, P.; Frajman, B.; Lazarević, M.; Paun, O.; Schönswetter, P.; Lakušić, D. Genetic, cytological and morphological differentiation within the Balkan-Carpathian Sesleria rigida sensu Fl. Eur. (Poaceae): A taxonomically intricate tetraploid-octoploid complex. Taxon 2013, 62, 458–472. [Google Scholar] [CrossRef] [Green Version]
- Kuzmanović, N.; Lakušić, D.; Frajman, B.; Alegro, A.; Schönswetter, P. Phylogenetic relationships in Seslerieae (Poaceae) including resurrection of Psilathera and Sesleriella, two monotypic genera endemic to the Alps. Taxon 2017, 66, 1349–1370. [Google Scholar] [CrossRef]
- López-González, N.; Bobo-Pinilla, J.; Padilla-García, N.; Loureiro, J.; Castro, S.; Rojas-Andrés, B.M.; Martínez-Ortega, M.M. Genetic similarities versus morphological resemblance: Unraveling a polyploid complex in a Mediterranean biodiversity hotspot. Mol. Phylogenetics Evol. 2021, 155, 107006. [Google Scholar] [CrossRef]
- Rundel, P.W.; Arroyo, M.T.K.; Cowling, R.M.; Keeley, J.E.; Lamont, B.B.; Vargas, P. Mediterranean biomes: Evolution of their vegetation, floras, and climate. Annu. Rev. Ecol. Evol. Syst. 2016, 47, 383–407. [Google Scholar] [CrossRef]
- Susanna, A.; Garcia-Jacas, N. Tribe Cardueae Cass. (1819). In Flowering Plants. Eudicots: Asterales; Kadereit, J.W., Jeffrey, C., Eds.; Springer: New York, NY, USA, 2007; Volume 8, pp. 123–146. ISBN 3-540-31051-7. [Google Scholar]
- Gardou, C. Quelques Vues Synthétiques sur les Centaurées de la Section Acrocentron (Cass.) O. Hoffm. Dans la Flore Méditerranéenne; La flore du bassin méditerranéen; Centre National de la Recherche Scientifique: Paris, France, 1975; pp. 537–547.
- Wagenitz, G. Centaurea L. In Flora of Turkey and the East Aegean Islands; Davis, P.H., Ed.; Edinburgh University Press: Edinburgh, UK, 1975; Volume 5, pp. 465–585. [Google Scholar]
- Font, M.; Garnatje, T.; Garcia-Jacas, N.; Susanna, A. Delineation and phylogeny of Centaurea sect. Acrocentron based on DNA sequences: A restoration of the genus Crocodylium and indirect evidence of introgression. Plant Syst. Evol. 2002, 234, 15–26. [Google Scholar] [CrossRef]
- Cassini, H. Dictionaire de Sciences Naturelles, Paris, 1819. In Cassini on Compositae; King, R., Dawson, H.W., Eds.; Oriole Editions: New York, NY, USA, 1975. [Google Scholar]
- Wagenitz, G. Pollenmorphologie und Systematik in der Gattung Centaurea L. s. 1. Flora Allg. Bot. 1955, 142, 213–279. [Google Scholar] [CrossRef]
- Garcia-Jacas, N.; Susanna, A. Karyological notes on Centaurea sect. Acrocentron (Asteraceae). Plant Syst. Evol. 1992, 179, 1–18. [Google Scholar] [CrossRef]
- Garcia-Jacas, N.; Susanna, A.; Mozaffarian, V. New chromosome counts in the subtribe Centaureinae (Asteraceae, Cardueae) from West Asia, III. Bot. J. Linn. Soc. 1998, 128, 413–422. [Google Scholar] [CrossRef]
- Ghaffari, S.M. Chromosome studies of some species of Centaurea section Acrocentron (Asteraceae) from Iran. Pak. J. Bot. 1999, 31, 301–305. [Google Scholar]
- Martin, E.; Dinc, M.; Duran, A. Karyomorphological study of eight Centaurea L. taxa (Asteraceae) from Turkey. Turk. J. Bot. 2009, 33, 97–104. [Google Scholar]
- Font, M.; Garcia-Jacas, N.; Vilatersana, R.; Roquet, C.; Susanna, A. Evolution and biogeography of Centaurea section Acrocentron inferred from nuclear and plastid DNA sequence analyses. Ann. Bot. 2009, 103, 985–997. [Google Scholar] [CrossRef]
- Novaković, J. Morphological, Phytochemical and Molecular Studies of Carpathian-Balkan Complex Centaurea atropurpurea (Asteraceae)—Phylogenetic and Taxonomic Implications. Ph.D. Thesis, Faculty of Biology, University of Belgrade, Belgrade, Serbia, 2019. [Google Scholar]
- Novaković, J.; Zlatković, B.; Lazarević, M.; Garcia-Jacas, N.; Susanna, A.; Marin, P.D.; Lakušić, D.; Janaćković, P. Centaurea zlatiborensis (Asteraceae, Cardueae−Centaureinae), a new endemic species from Zlatibor mountain range, Serbia. Nord. J. Bot. 2018, 36, 1–8. [Google Scholar] [CrossRef]
- Garcia-Jacas, N. Centaurea kunkelii (Asteraceae, Cardueae), a new hybridogenic endecaploid species of sect. Acrocentron from Spain. Ann. Bot. Fenn. 1998, 159–167. [Google Scholar]
- Garcia-Jacas, N.; Susanna, A. Centaurea × polymorpha Lagasca: Los problemas de un híbrido. Fontqueria 1993, 36, 65–66. [Google Scholar]
- Garcia-Jacas, N.; Soltis, P.S.; Font, M.; Soltis, D.E.; Vilatersana, R.; Susanna, A. The polyploid series of Centaurea toletana: Glacial migrations and introgression revealed by nrDNA and cpDNA sequence analyzes. Mol. Phylogenetics Evol. 2009, 52, 377–394. [Google Scholar] [CrossRef] [PubMed]
- Hilpold, A.; Vilatersana, R.; Susanna, A.; Meseguer, A.S.; Boršić, I.; Constantinidis, T.; Filigheddu, R.; Romaschenko, K.; Suárez-Santiago, V.N.; Tugay, O.; et al. Phylogeny of the Centaurea group (Centaurea, Compositae)—Geography is a better predictor than morphology. Mol. Phylogenetics Evol. 2014, 77, 195–215. [Google Scholar] [CrossRef] [PubMed]
- Rieseberg, L.H. Hybrid origins of plant species. Annu. Rev. Ecol. Syst. 1997, 28, 359–389. [Google Scholar] [CrossRef] [Green Version]
- Mallet, J. Hybrid speciation. Nature 2007, 446, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Soltis, P.S.; Soltis, D.E. The role of hybridization in plant speciation. Annu. Rev. Plant Biol. 2009, 60, 561–588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chapman, M.A.; Burke, J.M. Genetic divergence and hybrid speciation. Evolution 2007, 61, 1773–1780. [Google Scholar] [CrossRef]
- Paun, O.; Forest, F.; Fay, M.F.; Chase, M.W. Hybrid speciation in angiosperms: Parental divergence drives ploidy. New Phytol. 2009, 182, 507–518. [Google Scholar] [CrossRef] [Green Version]
- Brennan, A.C.; Barker, D.; Hiscock, S.J.; Abbott, R.J. Molecular genetic and quantitative trait divergence associated with recent homoploid hybrid speciation: A study of Senecio squalidus (Asteraceae). Heredity 2012, 108, 87–95. [Google Scholar] [CrossRef] [Green Version]
- Koutecký, P.; Badurová, T.; Štech, M.; Košnar, J.; Karásek, J. Hybridization between diploid Centaurea pseudophrygia and tetraploid C. jacea (Asteraceae): The role of mixed pollination, unreduced gametes, and mentor effects. Biol. J. Linn. Soc. 2011, 104, 93–106. [Google Scholar] [CrossRef] [Green Version]
- Mameli, G.; López-Alvarado, J.; Farris, E.; Susanna, A.; Filigheddu, R.; Garcia-Jacas, N. The role of parental and hybrid species in multiple introgression events: Evidence of homoploid hybrid speciation in Centaurea (Cardueae, Asteraceae). Bot. J. Linn. Soc. 2014, 175, 453–467. [Google Scholar] [CrossRef] [Green Version]
- Pisanu, S.; Mameli, G.; Farris, E.; Binelli, G.; Filigheddu, R. A natural homoploid hybrid between Centaurea horrida and Centaurea filiformis (Asteraceae) as revealed by morphological and genetic traits. Folia Geobot. 2011, 46, 69–86. [Google Scholar] [CrossRef]
- Bancheva, S.; Geraci, A.; Raimondo, F.M. Genetic diversity in the Centaurea cineraria group (Compositae) in Sicily using isozymes. Plant Biosyst. 2006, 140, 10–16. [Google Scholar] [CrossRef]
- Albrecht, M.; Duelli, P.; Obrist, M.K.; Kleijn, D.; Schmid, B. Effective long-distance pollen dispersal in Centaurea jacea. PLoS ONE 2009, 4, e6751. [Google Scholar] [CrossRef] [PubMed]
- Moreyra, L.D.; Márquez, F.; Susanna, A.; Garcia-Jacas, N.; Vázquez, F.M.; López-Pujol, J. Genesis, evolution, and genetic diversity of the hexaploid, narrow endemic Centaurea tentudaica. Diversity 2021, 13, 72. [Google Scholar] [CrossRef]
- Small, R.L.; Cronn, R.C.; Wendel, J.F. Use of nuclear genes for phylogeny reconstruction in plants. Aust. Syst. Bot. 2004, 17, 145. [Google Scholar] [CrossRef]
- Slovák, M.; Kučera, J.; Záveská, E.; Vd’ačný, P. Dealing with discordant genetic signal caused by hybridisation, incomplete lineage sorting and paucity of primary nucleotide homologies: A case study of closely related members of the genus Picris Subsection Hieracioides (Compositae). PLoS ONE 2014, 9, e104929. [Google Scholar] [CrossRef] [Green Version]
- Wan, D.; Sun, Y.; Zhang, X.; Bai, X.; Wang, J.; Wang, A.; Milne, R. Multiple ITS copies reveal extensive hybridization within Rheum (Polygonaceae), a genus that has undergone rapid radiation. PLoS ONE 2014, 9, e89769. [Google Scholar] [CrossRef] [Green Version]
- Oberprieler, C.; Greiner, R.; Konowalik, K.; Vogt, R. The reticulate evolutionary history of the polyploid NW Iberian Leucanthemum pluriflorum clan (Compositae, Anthemideae) as inferred from NrDNA ETS sequence diversity and eco-climatological niche-modelling. Mol. Phylogenetics Evol. 2014, 70, 478–491. [Google Scholar] [CrossRef]
- Doyle, J.J.; Dickson, E.E. Preservation of plant samples for DNA restriction endonuclease analysis. Taxon 1987, 36, 715–722. [Google Scholar] [CrossRef]
- Cullings, K.W. Design and testing of a plant-specific PCR primer for ecological and evolutionary studies. Mol. Ecol. 1992, 1, 233–240. [Google Scholar] [CrossRef]
- Tel-Zur, N.; Abbo, S.; Myslabodski, D.; Mizrahi, Y. Modified CTAB procedure for DNA isolation from epiphytic cacti of the genera Hylocereus and Selenicereus (Cactaceae). Plant Mol. Biol. Report. 1999, 17, 249–254. [Google Scholar] [CrossRef]
- Linder, C.R.; Goertzen, L.R.; Heuvel, B.V.; Francisco-Ortega, J.; Jansen, R.K. The complete External Transcribed Spacer of 18S–26S RDNA: Amplification and phylogenetic utility at low taxonomic levels in Asteraceae and closely allied families. Mol. Phylogenetics Evol. 2000, 14, 285–303. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, B.G.; Markos, S. Phylogenetic utility of the External Transcribed Spacer (ETS) of 18S–26S RDNA: Congruence of ETS and ITS Trees of Calycadenia (Compositae). Mol. Phylogenetics Evol. 1998, 10, 449–463. [Google Scholar] [CrossRef]
- Susanna, A.; Galbany-Casals, M.; Romaschenko, K.; Barres, L.; Martin, J.; Garcia-Jacas, N. Lessons from Plectocephalus (Compositae, Cardueae-Centaureinae): ITS Disorientation in Annuals and Beringian dispersal as revealed by molecular analyses. Ann. Bot. 2011, 108, 263–277. [Google Scholar] [CrossRef]
- López-Pujol, J.; Garcia-Jacas, N.; Susanna, A.; Vilatersana, R. Should we conserve pure species or hybrid species? Delimiting hybridization and introgression in the Iberian endemic Centaurea podospermifolia. Biol. Conserv. 2012, 152, 271–279. [Google Scholar] [CrossRef]
- Li, M.; Wunder, J.; Bissoli, G.; Scarponi, E.; Gazzani, S.; Barbaro, E.; Saedler, H.; Varotto, C. Development of COS genes as universally amplifiable markers for phylogenetic reconstructions of closely related plant species. Cladistics 2008, 24, 727–745. [Google Scholar] [CrossRef] [Green Version]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Cline, J.; Braman, J.C.; Hogrefe, H.H. PCR Fidelity of pfu DNA polymerase and other thermostable DNA polymerases. Nucleic Acids Res. 1996, 24, 3546–3551. [Google Scholar] [CrossRef] [Green Version]
- Popp, M.; Oxelman, B. Inferring the history of the polyploid Silene aegaea (Caryophyllaceae) using plastid and homoeologous nuclear DNA sequences. Mol. Phylogenetics Evol. 2001, 20, 474–481. [Google Scholar] [CrossRef]
- Martin, D.P.; Murrell, B.; Golden, M.; Khoosal, A.; Muhire, B. RDP4: Detection and analysis of recombination patterns in virus genomes. Virus Evol. 2015, 1, vev003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Posada, D.J. ModelTest: Phylogenetic Model Averaging. Mol. Biol. Evol. 2008, 25, 1253–1256. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; Van Der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [PubMed] [Green Version]
- Bryant, D.; Moulton, V. Neighbor-Net: An agglomerative method for the construction of phylogenetic networks. Mol. Biol. Evol. 2004, 21, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Huson, D.H.; Bryant, D. Application of phylogenetic networks in evolutionary studies. Mol. Biol. Evol. 2006, 23, 254–267. [Google Scholar] [CrossRef] [PubMed]
- Feulgen, R.; Rossenbeck, H. Mikroskopisch-chemischer Nachweis einer Nucleinsäure vom Typus der Thymonucleinsäure und die-darauf beruhende elektive Färbung von Zellkernen in mikroskopischen Präparaten. Biol. Chem. 1924, 135, 203–248. [Google Scholar] [CrossRef]
- Siljak-Yakovlev, S. Etude Cytogénétique et Palynologique de Compositae Endémiques ou Reliques de la Flore Yougoslave. Ph.D. Thesis, Centre d’Orsay, Université Paris-Sud, Orsay, France, 1986. [Google Scholar]
- Kay, K.M.; Reeves, P.A.; Olmstead, R.G.; Schemske, D.W. Rapid speciation and the evolution of hummingbird pollination in neotropical Costus subgenus Costus (Costaceae): Evidence from NrDNA ITS and ETS sequences. Am. J. Bot. 2005, 92, 1899–1910. [Google Scholar] [CrossRef] [Green Version]
- Frajman, B.; Rešetnik, I.; Niketić, M.; Ehrendorfer, F.; Schönswetter, P. Patterns of rapid diversification in heteroploid Knautia sect. Trichera (Caprifoliaceae, Dipsacoideae), one of the most intricate taxa of the European flora. BMC Evol. Biol. 2016, 16, 204. [Google Scholar] [CrossRef] [Green Version]
- Frajman, B.; Eggens, F.; Oxelman, B. Hybrid origins and homoploid reticulate evolution within Heliosperma (Sileneae, Caryophyllaceae)—A multigene phylogenetic approach with relative dating. Syst. Biol. 2009, 58, 328–345. [Google Scholar] [CrossRef] [Green Version]
- Klein, J.T.; Kadereit, J.W. Allopatric hybrids as evidence for past range dynamics in Sempervivum (Crassulaceae), a western Eurasian high mountain oreophyte. Alp. Bot. 2016, 126, 119–133. [Google Scholar] [CrossRef]
- Barres, L.; Sanmartín, I.; Anderson, C.L.; Susanna, A.; Buerki, S.; Galbany-Casals, M.; Vilatersana, R. Reconstructing the evolution and biogeographic history of tribe Cardueae (Compositae). Am. J. Bot. 2013, 100, 867–882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrando-Moraira, S.; Calleja, J.A.; Galbany-Casals, M.; Garcia-Jacas, N.; Liu, J.-Q.; López-Alvarado, J.; López-Pujol, J.; Mandel, J.R.; Massó, S.; Montes-Moreno, N.; et al. Nuclear and plastid DNA phylogeny of tribe Cardueae (Compositae) with Hyb-Seq data: A new subtribal classification and a temporal diversification framework. Mol. Phylogenetics Evol. 2019, 137, 313–332. [Google Scholar] [CrossRef] [PubMed]
- Folk, R.A.; Soltis, P.S.; Soltis, D.E.; Guralnick, R. New prospects in the detection and comparative analysis of hybridization in the Tree of Life. Am. J. Bot. 2018, 105, 364–375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kučera, J.; Marhold, K.; Lihová, J. Cardamine maritima group (Brassicaceae) in the Amphi-Adriatic area: A hotspot of species diversity revealed by DNA sequences and morphological variation. Taxon 2010, 59, 148–164. [Google Scholar] [CrossRef]
- Heuertz, M.; Carnevale, S.; Fineschi, S.; Sebastiani, F.; Hausman, J.F.; Paule, L.; Vendramin, G.G. Chloroplast DNA phylogeography of European ashes, Fraxinus sp. (Oleaceae): Roles of hybridization and life history traits. Mol. Ecol. 2006, 15, 2131–2140. [Google Scholar] [CrossRef] [PubMed]
- Stebbins, G.L. The role of hybridization in evolution. Proc. Am. Philos. Soc. 1959, 103, 231–251. [Google Scholar]
- Mayr, E. Systematics and the Origin of Species; Columbia University Press: New York, NY, USA, 1942. [Google Scholar]
- Harrison, R.G. Hybrids and hybrid zones: Historical perspective. In Hybrid Zones and the Evolutionary Process; Oxford University Press: London, UK, 1993; pp. 3–12. [Google Scholar]
- Wilkins, J.S. Species: A History of the Idea; University of California Press: Berkeley, CA, USA, 2009; Volume 1, ISBN 0-520-94507-7. [Google Scholar]
- De Queiroz, K. The general lineage concept of species, species chteria, and the process of speciation. In Endless Forms: Species and Speciation; Oxford University Press: London, UK, 1998; pp. 57–75. [Google Scholar]
- De Queiroz, K. Species concepts and species delimitation. Syst. Biol. 2007, 56, 879–886. [Google Scholar] [CrossRef] [Green Version]
- Fernández Casas, F.J.; Susanna, A. Monografía de la sección Chamaecyanus Willk. del género Centaurea L. Treb. Inst. Bot. Barc. 1985, 10, 5–174. [Google Scholar]
- Tonian, T.R. Relation between chromosome number and some morphological features of Centaureinae Less representatives. Rev. Biol. 1980, 33, 552–554. [Google Scholar]
- Garcia-Jacas, N.; Susanna, A.; Vilatersana, R.; Guara, M. New chromosome counts in the subtribe Centaureinae (Asteraceae, Cardueae) from West Asia, II. Bot. J. Linn. Soc. 1998, 128, 403–412. [Google Scholar] [CrossRef]
- Stefureac, T.; Tacina, A. Cariological and chorological investigations on two endemic taxa in the Romanian flora. Acta Bot. Hort. Bucur. 1982, 111–116. [Google Scholar]
- Bancheva, S.; Greilhuber, J. Genome size in Bulgarian Centaurea s.l. (Asteraceae). Plant Syst. Evol. 2006, 257, 95–117. [Google Scholar] [CrossRef]
- Rice, A.; Glick, L.; Abadi, S.; Einhorn, M.; Kopelman, N.M.; Salman-Minkov, A.; Mayzel, J.; Chay, O.; Mayrose, I. The Chromosome Counts Database (CCDB)—A community resource of plant chromosome numbers. New Phytol. 2015, 206, 19–26. [Google Scholar] [CrossRef]
- Garcia-Jacas, N.; Susanna, A.; Ilarslan, R.; Ilarslan, H. New chromosome counts in the subtribe Centaureinae (Asteraceae, Cardueae) from West Asia. Bot. J. Linn. Soc. 1997, 125, 343–349. [Google Scholar] [CrossRef]
- Uysal, T.; Ertuğrul, K.; Susanna, A.; Garcia-Jacas, N. New chromosome counts in the genus Centaurea (Asteraceae) from Turkey. Bot. J. Linn. Soc. 2009, 159, 280–286. [Google Scholar] [CrossRef]
- Lovrić, A. IOPB Chromosome Number Reports 77. Taxon 1982, 31, 761–777. [Google Scholar]
- Routsi, E.; Georgiadis, T. Cytogeographical study of Centaurea L. sect. Acrocentron (Cass.) DC. (Asteraceae) in Greece. Bot. Helv. 1999, 109, 139–151. [Google Scholar]
- Devesa, J.A.; Valdés, B.; Ottonello, D. IOPB chromosome number reports 100. Taxon 1988, 37, 920. [Google Scholar]
- Garcia-Jacas, N. Estudi Taxonòmic i Biosistemàtic de les Espècies Ibèriques i Nord-Africanes del Gènere Centaurea sect. Acrocentron. Ph.D. Thesis, Universitat de Barcelona, Barcelona, Spain, 1992. [Google Scholar]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novaković, J.; Janaćković, P.; Susanna, A.; Lazarević, M.; Boršić, I.; Milanovici, S.; Lakušić, D.; Zlatković, B.; Marin, P.D.; Garcia-Jacas, N. Molecular Insights into the Centaurea Calocephala Complex (Compositae) from the Balkans—Does Phylogeny Match Systematics? Diversity 2022, 14, 394. https://doi.org/10.3390/d14050394
Novaković J, Janaćković P, Susanna A, Lazarević M, Boršić I, Milanovici S, Lakušić D, Zlatković B, Marin PD, Garcia-Jacas N. Molecular Insights into the Centaurea Calocephala Complex (Compositae) from the Balkans—Does Phylogeny Match Systematics? Diversity. 2022; 14(5):394. https://doi.org/10.3390/d14050394
Chicago/Turabian StyleNovaković, Jelica, Pedja Janaćković, Alfonso Susanna, Maja Lazarević, Igor Boršić, Sretco Milanovici, Dmitar Lakušić, Bojan Zlatković, Petar D. Marin, and Núria Garcia-Jacas. 2022. "Molecular Insights into the Centaurea Calocephala Complex (Compositae) from the Balkans—Does Phylogeny Match Systematics?" Diversity 14, no. 5: 394. https://doi.org/10.3390/d14050394
APA StyleNovaković, J., Janaćković, P., Susanna, A., Lazarević, M., Boršić, I., Milanovici, S., Lakušić, D., Zlatković, B., Marin, P. D., & Garcia-Jacas, N. (2022). Molecular Insights into the Centaurea Calocephala Complex (Compositae) from the Balkans—Does Phylogeny Match Systematics? Diversity, 14(5), 394. https://doi.org/10.3390/d14050394