A Review of Research on the Mustard Hill Coral, Porites astreoides
Abstract
:1. Introduction
2. Taxonomy
| Kingdom: Animalia |
| Phylum: Cnidaria |
| Class: Anthozoa |
| Subclass: Hexacorallia |
| Order: Scleractinia |
| Family: Poritidae |
| Genus: Porites |
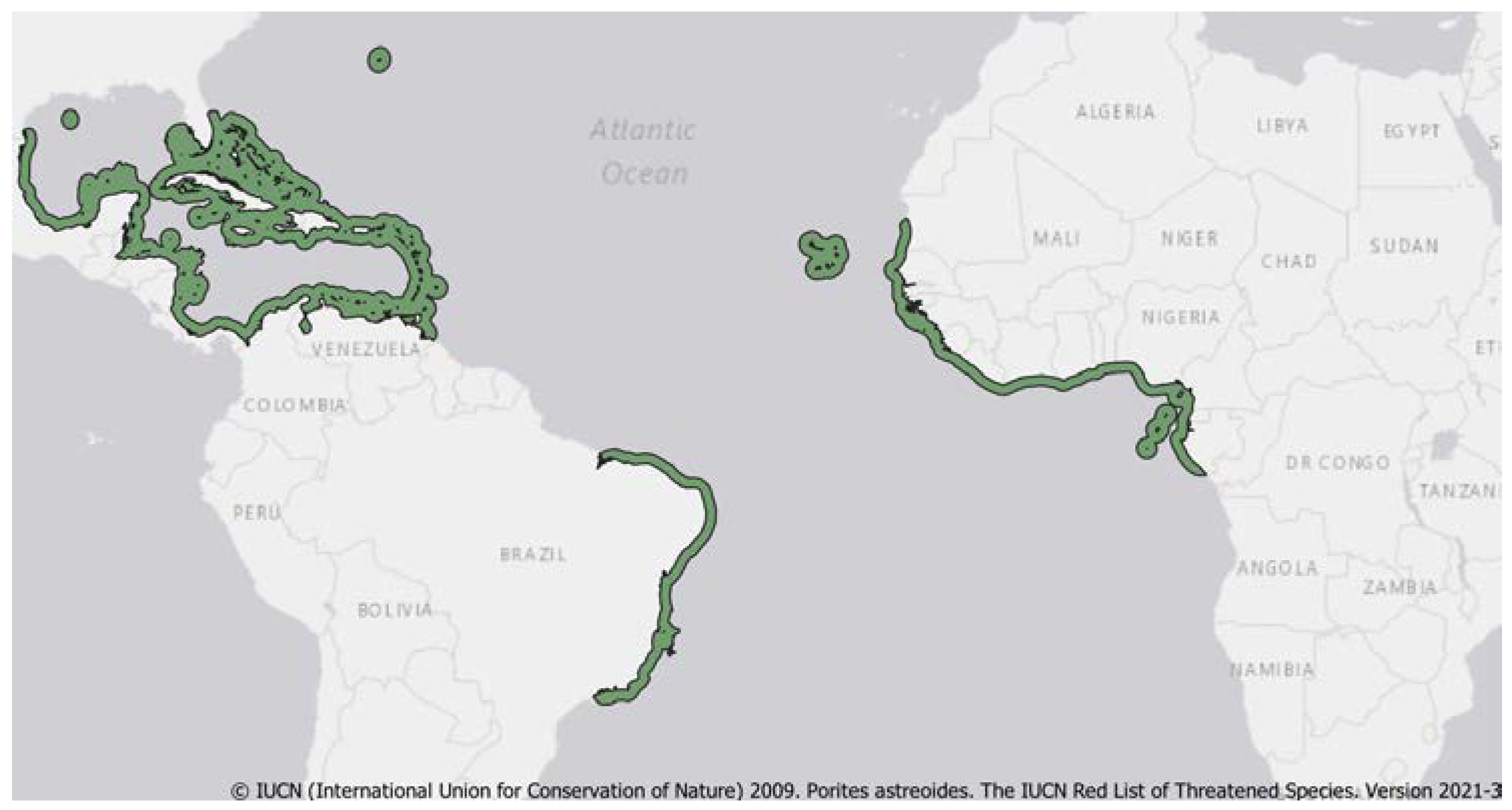
3. Distribution
4. Physiology & Life-History
5. Resilience
6. Ecology
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Woodhead, A.J.; Hicks, C.C.; Norström, A.V.; Williams, G.J.; Graham, N.A.J. Coral reef ecosystem services in the Anthropocene. Funct. Ecol. 2019, 33, 1023–1034. [Google Scholar] [CrossRef] [Green Version]
- Wilkinson, C. Status of Coral Reefs of the World: 2008; Global Coral Reef Monitoring Network and Reef and Rainforest Research Centre: Townsville, Australia, 2008. [Google Scholar]
- Okazaki, R.R.; Towle, E.K.; van Hooidonk, R.; Mor, C.; Winter, R.N.; Piggot, A.M.; Cunning, R.; Baker, A.C.; Klaus, J.S.; Swart, P.K.; et al. Species-specific responses to climate change and community composition determine future calcification rates of Florida Keys reefs. Glob. Chang. Biol. 2016, 23, 1023–1035. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.T.; Steneck, R.S.; Murphy, G.N.; Kench, P.S.; Edinger, E.N.; Smithers, S.G.; Mumby, P.J. Regional-scale dominance of non-framework building corals on Caribbean reefs affects carbonate production and future reef growth. Glob. Chang. Biol. 2015, 21, 1153–1164. [Google Scholar] [CrossRef]
- Cramer, K.L.; Donovan, M.K.; Jackson, J.B.C.; Greenstein, B.J.; Korpanty, C.A.; Cook, G.M.; Pandolfi, J.M. The transformation of Caribbean coral communities since humans. Ecol. Evol. 2021, 11, 10098–10118. [Google Scholar] [CrossRef]
- Darling, E.S.; McClanahan, T.R.; Côté, I.M. Life histories predict coral community disassembly under multiple stressors. Glob. Chang. Biol. 2013, 19, 1930–1940. [Google Scholar] [CrossRef]
- Baumann, J.H.; Townsend, J.E.; Courtney, T.A.; Aichelman, H.E.; Davies, S.W.; Lima, F.P.; Castillo, K.D. Temperature regimes impact coral assemblages along environmental gradients on lagoonal reefs in Belize. PLoS ONE 2016, 11, e0162098. [Google Scholar] [CrossRef] [Green Version]
- Eagleson, R.G.; Lumsden, J.S.; Álvarez-Filip, L.; Herbinger, C.M.; Horricks, R.A. Coverage increases of Porites astreoides in Grenada determined by shifts in size-frequency distribution. Diversity 2021, 13, 288. [Google Scholar] [CrossRef]
- Lange, I.D.; Perry, C.T.; Alvarez-Filip, L. Carbonate budgets as indicators of functional reef “health”: A critical review of data underpinning census-based methods and current knowledge gaps. Ecol. Indic. 2020, 110, 105857. [Google Scholar] [CrossRef]
- Green, D.H.; Edmunds, P.J.; Carpenter, R.C. Increasing relative abundance of Porites astreoides on Caribbean reefs mediated by an overall decline in coral cover. Mar. Ecol. Prog. Ser. 2008, 359, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Lima, L.F.O.; Bursch, H.; Dinsdale, E.A. Win some, lose some: The ecophysiology of Porites astreoides as a key coral species to Caribbean reefs. Front. Mar. Sci. 2022, 9, 1475. [Google Scholar] [CrossRef]
- Grottoli, A.G.; Warner, M.E.; Levas, S.J.; Aschaffenburg, M.D.; Schoepf, V.; Mcginley, M.; Baumann, J.; Matsui, Y. The cumulative impact of annual coral bleaching can turn some coral species winners into losers. Glob. Chang. Biol. 2014, 20, 3823–3833. [Google Scholar] [CrossRef] [PubMed]
- Edmunds, P.J.; Didden, C.; Frank, K. Over three decades, a classic winner starts to lose in a Caribbean coral community. Ecosphere 2021, 12, e03517. [Google Scholar] [CrossRef]
- WoRMS—World Register of Marine Species—Porites astreoides Lamarck. 1816. Available online: https://www.marinespecies.org/aphia.php?p=taxdetails&id=288889 (accessed on 14 March 2023).
- Madin, J.S.; Anderson, K.D.; Andreasen, M.H.; Bridge, T.C.L.; Cairns, S.D.; Connolly, S.R.; Darling, E.S.; Diaz, M.; Falster, D.S.; Franklin, E.C.; et al. The Coral Trait Database, a curated database of trait information for coral species from the global oceans. Sci. Data 2016, 3, 160017. [Google Scholar] [CrossRef] [Green Version]
- Shearer, T.L.; Coffroth, M.A. Barcoding corals: Limited by interspecific divergence, not intraspecific variation. Mol. Ecol. Resour. 2008, 8, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Kitahara, M.V.; Cairns, S.D.; Stolarski, J.; Blair, D.; Miller, D.J. A comprehensive phylogenetic analysis of Scleractinia (Cnidaria, Anthozoa) based on mitochondrial CO1 sequence data. PLoS ONE 2010, 5, e11490. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.H.; Putnam, H.M. The genome of the mustard hill coral, Porites astreoides. GigaByte 2022, 2022, gigabyte65. [Google Scholar] [CrossRef]
- Serrano, X.M.; Baums, I.B.; Smith, T.B.; Jones, R.J.; Shearer, T.L.; Baker, A.C. Long distance dispersal and vertical gene flow in the Caribbean brooding coral Porites astreoides. Sci. Rep. 2016, 6, 21619. [Google Scholar] [CrossRef]
- International Union for Conservation of Nature and Natural Resources. The IUCN Red List of Threatened Species; IUCN Global Species Programme Red List Unit: Cambridge, UK, 2000. [Google Scholar]
- Goreau, T. The ecology of Jamaican coral reefs. Species composition and zonation. Ecology 1959, 40, 67–90. [Google Scholar] [CrossRef]
- Baumann, J.H. Elevated maximum temperatures and high-magnitude thermal variability drive low coral diversity on nearshore lagoonal reefs in Belize. bioRxiv 2016, 036400. [Google Scholar] [CrossRef] [Green Version]
- Soong, K. Colony size as a species character in massive reef corals. Coral Reefs 1993, 12, 77–83. [Google Scholar] [CrossRef]
- Elizalde-Rendón, E.M.; Horta-Puga, G.; González-Diaz, P.; Carricart-Ganivet, J.P. Growth characteristics of the reef-building coral Porites astreoides under different environmental conditions in the Western Atlantic. Coral Reefs 2010, 29, 607–614. [Google Scholar] [CrossRef]
- Pereira, N.S.; Sial, A.N.; Frei, R.; Ullmann, C.V.; Korte, C.; Kikuchi, R.K.P.; Ferreira, V.P.; Kilbourne, K.H. The potential of the coral species Porites astreoides as a paleoclimate archive for the tropical South Atlantic Ocean. J. S. Am. Earth Sci. 2017, 77, 276–285. [Google Scholar] [CrossRef]
- Edmunds, P.J.; Lasker, H.R. Cryptic regime shift in benthic community structure on shallow reefs in St. John, US Virgin Islands. Mar. Ecol. Prog. Ser. 2016, 559, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Edmunds, P. Population biology of Porites astreoides and Diploria strigosa on a shallow Caribbean reef. Mar. Ecol. Prog. Ser. 2010, 418, 87–104. [Google Scholar] [CrossRef]
- Nagelkerken, I.; Bak, R. Differential regeneration of artificial lesions among sympatric morphs of the Caribbean corals Porites astreoides and Stephanocoenia michelinii. Mar. Ecol. Prog. Ser. 1998, 163, 279–283. [Google Scholar] [CrossRef] [Green Version]
- Gleason, D.F. Differential effects of ultraviolet radiation on green and brown morphs of the Caribbean coral Porites astreoides. Limnol. Oceanogr. 1993, 38, 1452–1463. [Google Scholar] [CrossRef] [Green Version]
- Lord, K.S.; Barcala, A.; Aichelman, H.E.; Kriefall, N.G.; Brown, C.; Knasin, L.; Secor, R.; Tone, C.; Tsang, L.; Finnerty, J.R. Distinct phenotypes associated with mangrove and lagoon habitats in two widespread Caribbean corals, Porites astreoides and Porites divaricata. Biol. Bull. 2021, 240, 169–190. [Google Scholar] [CrossRef]
- Rodriguez-Lanetty, M.; Granados-Cifuentes, C.; Barberan, A.; Bellantuono, A.J.; Bastidas, C. Ecological inferences from a deep screening of the complex bacterial consortia associated with the coral, Porites astreoides. Mol. Ecol. 2013, 22, 4349–4362. [Google Scholar] [CrossRef]
- Sunagawa, S.; Woodley, C.M.; Medina, M.; Whitman, W.; Coleman, D.; Wiebe, W.; Pedros-Alio, C.; Sogin, M.; Morrison, H.; Huber, J.; et al. Threatened corals provide underexplored microbial habitats. PLoS ONE 2010, 5, e9554. [Google Scholar] [CrossRef] [Green Version]
- Glasl, B.; Herndl, G.J.; Frade, P.R. The microbiome of coral surface mucus has a key role in mediating holobiont health and survival upon disturbance. ISME J. 2016, 10, 2280–2292. [Google Scholar] [CrossRef] [Green Version]
- Meyer, J.L.; Paul, V.J.; Teplitski, M.; Riegl, B.; Bruckner, A.; Coles, S.; Renaud, P.; Dodge, R.; Loya, Y.; Sakai, K.; et al. Community shifts in the surface microbiomes of the coral Porites astreoides with unusual lesions. PLoS ONE 2014, 9, e100316. [Google Scholar] [CrossRef] [PubMed]
- Vega Thurber, R.; Burkepile, D.E.; Correa, A.M.S.; Thurber, A.R.; Shantz, A.A.; Welsh, R.; Pritchard, C.; Rosales, S. Macroalgae decrease growth and alter microbial community structure of the reef-building coral, Porites astreoides. PLoS ONE 2012, 7, e44246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morrow, K.M.; Liles, M.R.; Paul, V.J.; Moss, A.G.; Chadwick, N.E. Bacterial shifts associated with coral-macroalgal competition in the Caribbean Sea. Mar. Ecol. Prog. Ser. 2013, 488, 103–117. [Google Scholar] [CrossRef]
- Hauff, B.; Haslun, J.A.; Strychar, K.B.; Ostrom, P.H.; Cervino, J.M. Symbiont diversity of zooxanthellae (Symbiodinium Spp.) in Porites astreoides and Montastraea cavernosa from a reciprocal transplant in the Lower Florida Keys. Int. J. Biol. 2016, 8, 9. [Google Scholar] [CrossRef]
- Kenkel, C.D.; Goodbody-Gringley, G.; Caillaud, D.; Davies, S.W.; Bartels, E.; Matz, M.V. Evidence for a host role in thermotolerance divergence between populations of the mustard hill coral (Porites astreoides) from different reef environments. Mol. Ecol. 2013, 22, 4335–4348. [Google Scholar] [CrossRef] [PubMed]
- Haslun, J.A.; Hauff-Salas, B.; Strychar, K.B.; Ostrom, N.E.; Cervino, J.M. Biotic stress contributes to seawater temperature induced stress in a site-specific manner for Porites astreoides. Mar. Biol. 2018, 165, 160. [Google Scholar] [CrossRef]
- García-Arredondo, A.; Rojas-Molina, A.; Ibarra-Alvarado, C.; Lazcano-Pérez, F.; Arreguín-Espinosa, R.; Sánchez-Rodríguez, J. Composition and biological activities of the aqueous extracts of three Scleractinian Corals from the Mexican Caribbean: Pseudodiploria strigosa, Porites astreoides and Siderastrea siderea. J. Venom. Anim. Toxins Incl. Trop. Dis. 2016, 22, 32. [Google Scholar] [CrossRef] [Green Version]
- Goodbody-Gringley, G.; de Putron, S.J. Brooding corals: Planulation patterns, larval behavior, and recruitment dynamics in the face of environmental change. In The Cnidaria, Past, Present and Future; Springer International Publishing: Cham, Switzerland, 2016; pp. 279–289. [Google Scholar]
- Vollmer, A. Rare Parthenogenic Reproduction in a Common Reef Coral, Porites astreoides. Master’s Thesis, Nova Southeastern University, Davies, FL, USA, 2018. Available online: https://nsuworks.nova.edu/occ_stuetd/464/ (accessed on 20 March 2023).
- Darling, E.S.; Alvarez-Filip, L.; Oliver, T.A.; McClanahan, T.R.; Côté, I.M. Evaluating life-history strategies of reef corals from species traits. Ecol. Lett. 2012, 15, 1378–1386. [Google Scholar] [CrossRef]
- Edinger, E.N.; Risk, M.J. Preferential survivorship of brooding corals in a regional extinction. Paleobiology 1995, 21, 200–219. [Google Scholar] [CrossRef]
- Williams, S.M.; Sánchez-Godínez, C.; Newman, S.P.; Cortés, J. Ecological assessments of the coral reef communities in the Eastern Caribbean and the effects of herbivory in influencing coral juvenile density and algal cover. Mar. Ecol. 2016, 38, e12395. [Google Scholar] [CrossRef]
- Chornesky, E.; Peters, E. Sexual reproduction and colony growth in the Scleractinian coral Porites astreoides. Biol. Bull. 1987, 172, 161–177. [Google Scholar] [CrossRef]
- de Putron, S.; Smith, S. Planula release and reproductive seasonality of the Scleractinian coral Porites astreoides in Bermuda, a high-latitude reef. Bull. Mar. Sci. 2011, 87, 75–90. [Google Scholar] [CrossRef] [Green Version]
- McGuire, M.P. Timing of larval release by Porites astreoides in the Northern Florida Keys. Coral Reefs 1998, 17, 369–375. [Google Scholar] [CrossRef]
- Holstein, D.M.; Smith, T.B.; Paris, C.B. Depth-independent reproduction in the reef coral Porites astreoides from shallow to mesophotic zones. PLoS ONE 2016, 11, e0146068. [Google Scholar] [CrossRef] [PubMed]
- Riquet, F.; Japaud, A.; Nunes, F.L.D.; Serrano, X.M.; Baker, A.C.; Bezault, E.; Bouchon, C.; Fauvelot, C. Complex spatial patterns of genetic differentiation in the Caribbean mustard hill coral Porites astreoides. Coral Reefs 2022, 41, 813–828. [Google Scholar] [CrossRef]
- White, K.Q. A Correlation between Larval Size and Spat Growth and Development in Coral Species. Honours Thesis, Saint Mary’s University, Halifax, NS, Canada, 2014. Available online: https://library2.smu.ca/handle/01/25781 (accessed on 20 March 2023).
- Olsen, K.; Sneed, J.M.; Paul, V.J. Differential larval settlement responses of Porites astreoides and Acropora palmata in the presence of the green alga Halimeda opuntia. Coral Reefs 2016, 35, 521–525. [Google Scholar] [CrossRef]
- Rushmore, M.E.; Ross, C.; Fogarty, N.D. Physiological responses to short-term sediment exposure in adults of the Caribbean coral Montastraea cavernosa and adults and recruits of Porites astreoides. Coral Reefs 2021, 40, 1579–1591. [Google Scholar] [CrossRef]
- Gleason, D.F.; Edmunds, P.J.; Gates, R.D. Ultraviolet radiation effects on the behavior and recruitment of larvae from the reef coral Porites astreoides. Mar. Biol. 2006, 148, 503–512. [Google Scholar] [CrossRef]
- Lillis, A.; Apprill, A.; Suca, J.J.; Becker, C.; Llopiz, J.K.; Mooney, T.A. Soundscapes influence the settlement of the common Caribbean coral Porites astreoides irrespective of light conditions. R. Soc. Open Sci. 2018, 5, 181358. [Google Scholar] [CrossRef] [Green Version]
- Goodbody-Gringley, G.; Wong, K.H.; Becker, D.M.; Glennon, K.; de Putron, S.J. Reproductive ecology and early life history traits of the brooding coral, Porites astreoides, from shallow to mesophotic zones. Coral Reefs 2018, 37, 483–494. [Google Scholar] [CrossRef]
- Ritson-Williams, R.; Arnold, S.; Paul, V. Patterns of larval settlement preferences and post-settlement survival for seven Caribbean corals. Mar. Ecol. Prog. Ser. 2016, 548, 127–138. [Google Scholar] [CrossRef]
- Ross, C.; Ritson-Williams, R.; Olsen, K.; Paul, V.J. Short-term and latent post-settlement effects associated with elevated temperature and oxidative stress on larvae from the coral Porites astreoides. Coral Reefs 2013, 32, 71–79. [Google Scholar] [CrossRef]
- Porto-Hannes, I.; Zubillaga, A.L.; Shearer, T.L.; Bastidas, C.; Salazar, C.; Coffroth, M.A.; Szmant, A.M. Population structure of the corals Orbicella faveolata and Acropora palmata in the Mesoamerican Barrier Reef system with comparisons over Caribbean basin-wide spatial scale. Mar. Biol. 2015, 162, 81–98. [Google Scholar] [CrossRef]
- Kenkel, C.D.; Meyer, E.; Matz, M. Gene expression under chronic heat stress in populations of the mustard hill coral (Porites astreoides) from different thermal environments. Mol. Ecol. 2013, 22, 4322–4334. [Google Scholar] [CrossRef] [PubMed]
- Kenkel, C.D.; Setta, S.P.; Matz, M. Heritable differences in fitness-related traits among populations of the mustard hill coral, Porites astreoides. Heredity 2015, 115, 509–516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castillo, K.D.; Ries, J.B.; Weiss, J.M.; Lima, F.P. Decline of fore reef corals in response to recent warming linked to history of thermal exposure. Nat. Clim. Chang. 2012, 2, 756–760. [Google Scholar] [CrossRef]
- Hall, E.R.; DeGroot, B.C.; Fine, M. Lesion recovery of two Scleractinian corals under low pH conditions: Implications for restoration efforts. Mar. Pollut. Bull. 2015, 100, 321–326. [Google Scholar] [CrossRef]
- Kenkel, C.D.; Matz, M. Gene expression plasticity as a mechanism of coral adaptation to a variable environment. Nat. Ecol. Evol. 2016, 1, 0014. [Google Scholar] [CrossRef]
- Manzello, D.P.; Enochs, I.C.; Kolodziej, G.; Carlton, R. Coral growth patterns of Montastraea cavernosa and Porites astreoides in the Florida Keys: The importance of thermal stress and inimical waters. J. Exp. Mar. Biol. Ecol. 2015, 471, 198–207. [Google Scholar] [CrossRef] [Green Version]
- Claquin, P.; Rene-Trouillefou, M.; Lopez, P.J.; Japaud, A.; Bouchon-Navaro, Y.; Cordonnier, S.; Bouchon, C. Singular physiological behavior of the Scleractinian coral Porites astreoides in the dark phase. Coral Reefs 2020, 40, 139–150. [Google Scholar] [CrossRef]
- Levas, S.; Grottoli, A.G.; Schoepf, V.; Aschaffenburg, M.; Baumann, J.; Bauer, J.E.; Warner, M.E. Can heterotrophic uptake of dissolved organic carbon and zooplankton mitigate carbon budget deficits in annually bleached corals? Coral Reefs 2016, 35, 495–506. [Google Scholar] [CrossRef] [Green Version]
- Gintert, B.E.; Manzello, D.P.; Enochs, I.C.; Kolodziej, G.; Carlton, R.; Gleason, A.C.R.; Gracias, N. Marked annual coral bleaching resilience of an inshore patch reef in the Florida Keys: A nugget of hope, aberrance, or last man standing? Coral Reefs 2018, 37, 533–547. [Google Scholar] [CrossRef] [Green Version]
- Mallela, J.; Crabbe, M.J.C. Hurricanes and coral bleaching linked to changes in coral recruitment in Tobago. Mar. Environ. Res. 2009, 68, 158–162. [Google Scholar] [CrossRef] [Green Version]
- Ritson-Williams, R.; Ross, C.; Paul, V.J.; Hoegh-Guldberg, O.; Pandolfi, J.; Bradbury, R.; Sala, E.; Hughes, T.; Bjorndal, K.; Cooke, R.; et al. Elevated temperature and allelopathy impact coral recruitment. PLoS ONE 2016, 11, e0166581. [Google Scholar] [CrossRef] [Green Version]
- Gallery, D.N.; Green, M.L.; Kuffner, I.B.; Lenz, E.A.; Toth, L.T. Genetic structure and diversity of the mustard hill coral Porites astreoides along the Florida Keys reef tract. Mar. Biodivers. 2021, 51, 63. [Google Scholar] [CrossRef]
- Lenz, E.A.; Bartlett, L.A.; Stathakopoulos, A.; Kuffner, I.B. Physiological differences in bleaching response of the coral Porites astreoides along the Florida Keys reef tract during high-temperature stress. Front. Mar. Sci. 2021, 8, 615795. [Google Scholar] [CrossRef]
- González-Barrios, F.J.; Cabral-Tena, R.A.; Alvarez-Filip, L. Recovery disparity between coral cover and the physical functionality of reefs with impaired coral assemblages. Glob. Chang. Biol. 2021, 27, 640–651. [Google Scholar] [CrossRef] [PubMed]
- Buglass, S.; Donner, S.D.; Alemu I, J.B. A study on the recovery of Tobago’s coral reefs following the 2010 mass bleaching event. Mar. Pollut. Bull. 2016, 104, 198–206. [Google Scholar] [CrossRef] [Green Version]
- Ennis, R.S.; Brandt, M.E.; Grimes, K.R.W.; Smith, T.B. Coral reef health response to chronic and acute changes in water quality in St. Thomas, United States Virgin Islands. Mar. Pollut. Bull. 2016, 111, 418–427. [Google Scholar] [CrossRef] [Green Version]
- Barranco, L.M.; Carriquiry, J.D.; Rodríguez-Zaragoza, F.A.; Cupul-Magaña, A.L.; Villaescusa, J.A.; Calderón-Aguilera, L.E.; Calderón-Aguilera, L.E. Spatiotemporal variations of live coral cover in the northern Mesoamerican Reef system, Yucatan Peninsula, Mexico. Sci. Mar. 2016, 80, 143–150. [Google Scholar] [CrossRef] [Green Version]
- Mumby, P.J.; Harborne, A.R. Marine reserves enhance the recovery of corals on Caribbean reefs. PLoS ONE 2010, 5, e8657. [Google Scholar] [CrossRef] [PubMed]
- Holstein, D.M.; Paris, C.B.; Mumby, P.J. Consistency and inconsistency in multispecies population network dynamics of coral reef ecosystems. Mar. Ecol. Prog. Ser. 2014, 499, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Alvarez-Filip, L.; Carricart-Ganivet, J.P.; Horta-Puga, G.; Iglesias-Prieto, R. Shifts in coral-assemblage composition do not ensure persistence of reef functionality. Sci. Rep. 2013, 3, 3486. [Google Scholar] [CrossRef] [Green Version]
- Rotjan, R.D.; Lewis, S.M. Selective predation by parrotfishes on the reef coral Porites astreoides. Mar. Ecol. Prog. Ser. 2005, 305, 193–201. [Google Scholar] [CrossRef] [Green Version]
- Muller, E.; de Gier, W.; ten Hove, H.A.; van Moorsel, G.W.N.M.; Hoeksema, B.W. Nocturnal predation of Christmas tree worms by a batwing coral crab at Bonaire (Southern Caribbean). Diversity 2020, 12, 455. [Google Scholar] [CrossRef]
- Hoeksema, B.W.; Timmerman, R.F.; Spaargaren, R.; Smith-Moorhouse, A.; van der Schoot, R.J.; Langdon-Down, S.J.; Harper, C.E. Morphological modifications and injuries of corals caused by symbiotic feather duster worms (Sabellidae) in the Caribbean. Diversity 2022, 14, 332. [Google Scholar] [CrossRef]
- Goreau, T.F.; Hartman, W.D. Sponge: Effect on the form of reef corals. Science 1966, 151, 343–344. [Google Scholar] [CrossRef] [PubMed]
- van der Schoot, R.J.; Hoeksema, B.W. Abundance of coral-associated fauna in relation to depth and eutrophication along the leeward side of Curaçao, Southern Caribbean. Mar. Environ. Res. 2022, 181, 105738. [Google Scholar] [CrossRef] [PubMed]
- Hoeksema, B.W.; Wels, D.; van der Schoot, R.J.; ten Hove, H.A. Coral injuries caused by Spirobranchus opercula with and without epibiotic turf algae at Curaçao. Mar. Biol. 2019, 166, 60. [Google Scholar] [CrossRef] [Green Version]
- Alvarez-Filip, L.; Dulvy, N.K.; Côté, I.M.; Watkinson, A.R.; Gill, J.A. Coral identity underpins architectural complexity on Caribbean reefs. Ecol. Appl. 2011, 21, 2223–2231. [Google Scholar] [CrossRef]
- Alvarez-Filip, L.; González-Barrios, F.J.; Pérez-Cervantes, E.; Molina-Hernández, A.; Estrada-Saldívar, N. Stony coral tissue loss disease decimated Caribbean coral populations and reshaped reef functionality. Commun. Biol. 2022, 5, 440. [Google Scholar] [CrossRef]
- Perry, C.T.; Alvarez-Filip, L.; Graham, N.A.J.; Mumby, P.J.; Wilson, S.K.; Kench, P.S.; Manzello, D.P.; Morgan, K.M.; Slangen, A.B.A.; Thomson, D.P.; et al. Loss of coral reef growth capacity to track future increases in sea level. Nature 2018, 558, 396–400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bove, C.B.; Davies, S.W.; Ries, J.B.; Umbanhowar, J.; Thomasson, B.C.; Farquhar, E.B.; McCoppin, J.A.; Castillo, K.D. Global change differentially modulates Caribbean coral physiology. PLoS ONE 2022, 17, e0273897. [Google Scholar] [CrossRef] [PubMed]
- Albright, R.; Langdon, C. Ocean acidification impacts multiple early life history processes of the Caribbean coral Porites astreoides. Glob. Chang. Biol. 2011, 17, 2478–2487. [Google Scholar] [CrossRef]
- Miller, M.; Qian, P.-Y.; Williams, D.E.; Huntington, B.E.; Piniak, G.A.; Vermeij, M.J.A. Decadal comparison of a diminishing coral community: A study using demographics to advance inferences of community status. PeerJ 2016, 4, e1643. [Google Scholar] [CrossRef] [Green Version]
- Johnston, N.K.; Campbell, J.E.; Paul, V.J.; Hay, M.E. Effects of future climate on coral-coral competition. PLoS ONE 2020, 15, e0235465. [Google Scholar] [CrossRef]
- Horwitz, R.; Hoogenboom, M.O.; Fine, M. Spatial competition dynamics between reef corals under ocean acidification. Sci. Rep. 2017, 7, 40288. [Google Scholar] [CrossRef]
- Camp, E.F.; Schoepf, V.; Mumby, P.J.; Hardtke, L.A.; Rodolfo-Metalpa, R.; Smith, D.J.; Suggett, D.J. The future of coral reefs subject to rapid climate change: Lessons from natural extreme environments. Front. Mar. Sci. 2018, 5, 4. [Google Scholar] [CrossRef] [Green Version]


| Porites astreoides | 2050 | 2100 |
|---|---|---|
| Larval metabolism | −27% | −63% |
| Settlement success | −43.50% | −57.50% |
| Post-settlement growth | −16% | −35% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eagleson, R.G.; Álvarez-Filip, L.; Lumsden, J.S. A Review of Research on the Mustard Hill Coral, Porites astreoides. Diversity 2023, 15, 462. https://doi.org/10.3390/d15030462
Eagleson RG, Álvarez-Filip L, Lumsden JS. A Review of Research on the Mustard Hill Coral, Porites astreoides. Diversity. 2023; 15(3):462. https://doi.org/10.3390/d15030462
Chicago/Turabian StyleEagleson, Ryan G., Lorenzo Álvarez-Filip, and John S. Lumsden. 2023. "A Review of Research on the Mustard Hill Coral, Porites astreoides" Diversity 15, no. 3: 462. https://doi.org/10.3390/d15030462







