Abstract
In recent years, the introduction of alien and invasive marine species in the Mediterranean Sea has rapidly increased. Laonome triangularis is a sabellid worm, native to the Australian coasts. Its first record in the Mediterranean Sea dates back to 2009 and, in this area, it is currently labeled as an allochthonous species. In the present work, we report the fìrst record of this polycheate in Italian waters and we provide some diagnostic features that are useful to identify the species.
Alien species can disrupt the functioning of marine ecosystems, trigger alterations in aquatics food webs and modify the structures of coastal habitats [1,2,3,4]. The Mediterranean basin is one of the areas most affected by biological invasions and the number of reported alien species has steadily increased in recent years [2,4].
Laonome triangularis, Hutchings and Murray, 1984 is a polychaete belonging to the family Sabellidae and was originally described in Australia and redescribed by Capa in 2007 [5]. This polychaete is currently considered an invasive alien species established in the Eastern Mediterranean areas [1,2,4,5]. The first record of L. triangularis in the Mediterranean Sea was made by Çinar in 2009, in an area close to the Port of İskenderun and the Mersin Bays (Turkey) [1]. To date, this species has never been reported in the Western Mediterranean basin, including Italian waters.
On 9 May 2023, during a sampling campaign carried out within the project SeAlien, a total of nine specimens of L. triangularis (Figure 1a) were collected from a soft bottom in an area adjacent to the entrance channel of the commercial Port of Civitavecchia (Central Tyrrhenian Sea, Latium, Italy). Samples were collected on muddy sand at a depth of 22 m (42.118045 N, 11.749808 E) with an 18-litre Van Veen grapple: three replicates were taken at the sampling point and the sampled sediment was then sieved with 1 mm mesh. The retained sediment fraction was preserved in 75% ethanol and all collected organisms were sorted in the labatory, identified at the lowest possible taxonomic level and stored in separate containers with 75% ethanol.

Figure 1.
Laonome triangularis: (a) Entire specimen. Scale bar: 3 mm; (b) radioles with transverse rows of pigment. Scale bar: 500 µm; (c) last chaetigers and triangular pygidium. Scale bar: 500 µm. Photo credit: Andrea Bonifazi.
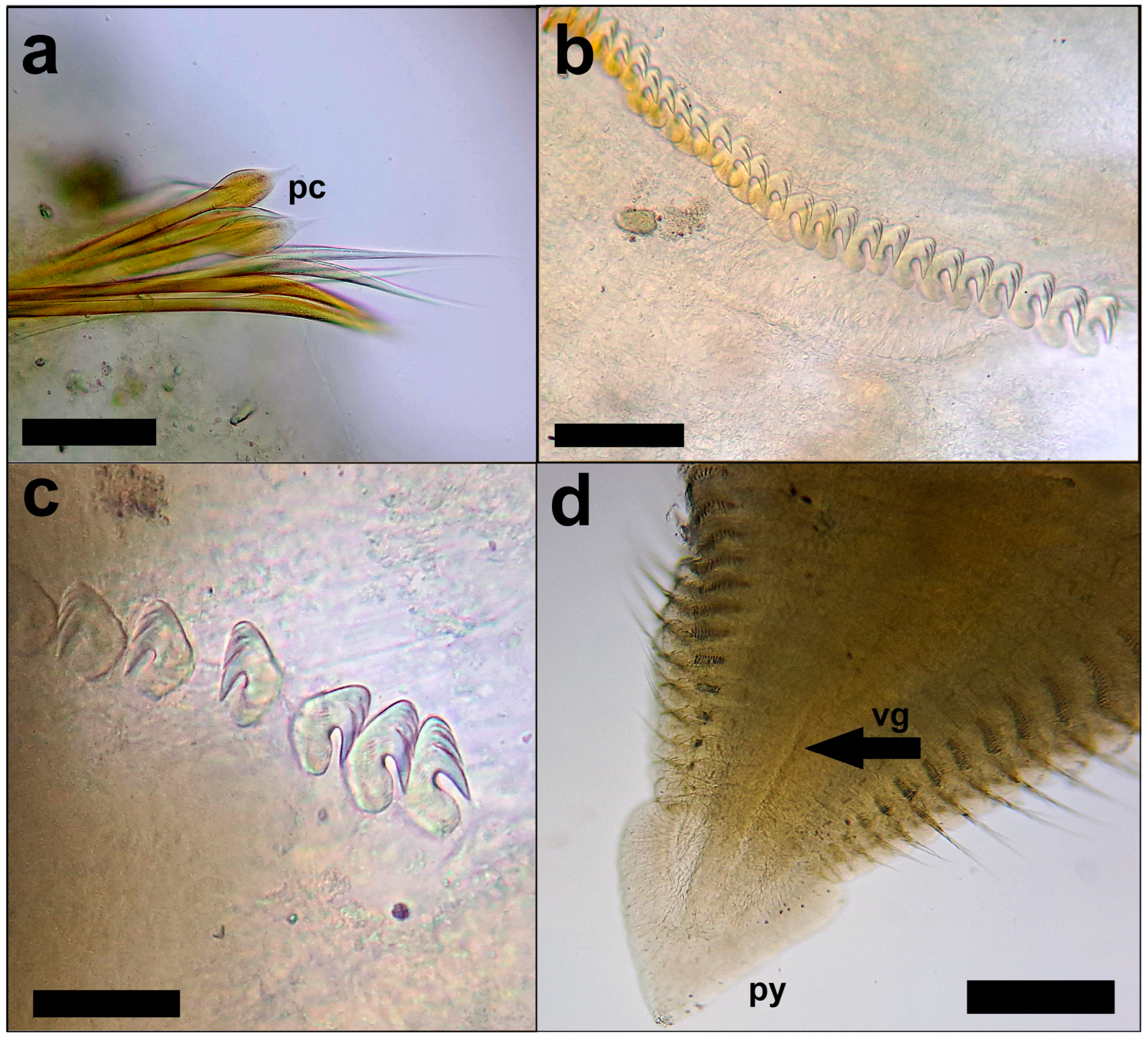
The diagnostic features and the morphological traits of L. triangularis specimens were examined following the method of Hutchings and Murray (1984), Capa (2007) and Çinar (2009). L. triangularis can be distinguished from the other congeneric Mediterranean species by the combination of the following diagnostic features: the crown is characterized by the presence of transverse rows of pigment localized at the base of some pinnules (Figure 1b); the pygidium is triangular (Figure 1c); the collar lobes are triangular in shape (Figure 2a); the paleate chaetae starti from the first chaetiger (Figure 3a); the thoracic hooks with three or four rows of teeth above the main fang (Figure 3b); and the abdominal hooks show, along with two or three rows of teeth above the main fang (sensu Çinar, 2009) (Figure 3c).

Figure 2.
Laonome triangularis: (a) Posterior peristomial ring collar (prc) with triangular lobes in ventral view; branchial crown removed. Scale bar: 500 µm; (b) peristomial ring (pr) in dorsal view. Scale bar: 200 µm; (c) dorsal lips tapered (dL). Scale bar: 500 µm; (d) radioles with axial skeleton. Scale bar: 200 µm. Photo credit: Andrea Bonifazi.
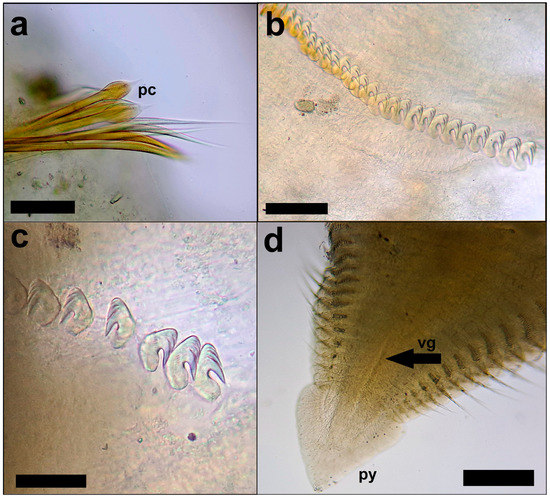
Figure 3.
Laonome triangularis: (a) First chaetiger with paleate chetae (pc). Scale bar: 100 µm; (b) thoracic neuropodial uncini. Scale bar: 100 µm; (c) abdominal notopodial uncini. Scale bar: 50 µm; (d) pygidium triangular (py) with a ventral grove (vg). Scale bar: 300 µm. Photo credit: Andrea Bonifazi.
All the individuals we analyzed coincided with the descriptions of the other authors and showed the following morphological characteristics: the nine collected specimens were 16.2–18.1 mm long, including crown, and 1.2 mm wide, with 8 thoracic and 47–50 abdominal chaetigers; body was pale brownish after preservation with a cylindrical shape and tapering posteriorly (Figure 1a); branchial crown with 14–15 pairs of radioles and characterized by the presence of four transverse rows of pigment (Figure 1b); posterior peristomial ring collar was well-developed and higher than the lateral and anterior margins with triangular lobes (Figure 2a); anterior peristomial ring was poorly developed, with the same height throughout its extension (Figure 2b); lateral collar margin entire; dorsal lips tapered, radiolar appendages present (Figure 2c); radioles quadrangular with smooth surface; axial skeleton with two rows of cells (Figure 2d); small pinnules arranged in two rows; basal palmate membrane present, but underdeveloped; ventral notch present and about half the length of the total extension of the collar; a thin transverse ridge present between the collar and the first chaetiger; the first chaetiger has a superior row of elongated narrowly-hooded chaetae and an inferior row of paleate chetae (Figure 3a); the other thoracic chaetigers have a superior row of narrowly hooded chaetae and two inferior rows of paleate chetae; thoracic neuropodial uncini with long tori and three of four rows of teeth above the main fang (Figure 3b); abdominal notopodial uncini with two or three rows of teeth above the main fang (Figure 3c); posterior abdominal segments with a ventral anal depression, which develops over the last 17–19 chaetigers; triangular pygidium with a ventral grove (Figure 3d). The tubes were large, fragile and composed of agglutinated mud.
The analysis of the macrobenthic fauna associated with L. triangularis revealed that polychaetes were the dominant taxon, mainly represented by the capitellid Pseudoleiocapitella fauveli (Harmelin, 1964) and the lumbrinereid Lumbrineris pinaster (Martins, Carrera-Parra, Quintino and Rodrigues, 2012). Another well-represented species was the ophiuroid Amphiura chiajei, (Forbes, 1843).
Regarding the ecology of L. triangularis, previous work has reported a similar habitat for this species [1,5,6]. Indeed, in both Australian and Turkish waters, specimens of L. triangularis have been sampled on muddy and sandy substrates at depths between 7 and 30 m.
Çinar (2009) speculates that the records of this species in areas adjacent to a commercial port may be due to ballast water. Furthermore, he specified that this euryhaline polychaete can easily adapt to new environments due to its ecological characteristics [1]. In recent years, many alien and invasive species have been reported in the Port of Civitavecchia [4,7] and our specimens were sampled in an area adjacent to this port, supporting the hypothesis proposed by Cinar. For this reason, commercial ports are considered a bioinvasion hotspot, constituting the initial area of settlement of alien and invasive species [4,8,9].
This work represents the first record of the sabellid L. triangularis both in the western part of the Mediterranean Sea and in Italian waters. It further emphasises the need for constant biological monitoring in port environments in order to prevent the spread of invasive alien species in the Mediterranean basin.
Author Contributions
A.B. analyzed and described the specimens, photographed (stereo and optical microscope) the specimens and wrote the manuscript. M.F.L. coordinated the project. S.D.B. conceived the project and carried out the sampling campaign. R.C. carried out the sampling campaign. M.F. sorted the benthic macroinvertebrates sampled in the campaign and contributed to the identification. S.M. conceived the project and carried out the sampling campaign. M.P. conceived the project and carried out the sampling campaign. E.M. analyzed and described the specimens and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the project SeAlien, grant number Reg (UE) 508/2014.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Çinar, M.E. Alien polychaete species (Annelida: Polychaeta) on the southern coast of Turkey (Levantine Sea, eastern Mediterranean), with 13 new records for the Mediterranean Sea. J. Nat. Hist. 2009, 43, 2283–2328. [Google Scholar] [CrossRef]
- Zenetos, A.; Gofas, S.; Verlaque, M.; Cinar, M.; Garcia Raso, J.; Bianchi, C.; Morri, C.; Azzurro, E.; Bilecenoglu, M.; Froglia, C.; et al. Alien species in the Mediterranean Sea by 2010. A contribution to the application of European Union’s Marine Strategy Framework Directive (MSFD). Part I. Spatial distribution. Mediterr. Mar. Sci. 2010, 11, 381–493. [Google Scholar] [CrossRef]
- Seebens, H.; Blackburn, T.M.; Dyer, E.E.; Genovesi, P.; Hulme, P.E.; Jeschke, J.M.; Essl, F. No saturation in the accumulation of alien species worldwide. Nat. Commun. 2017, 8, 14435. [Google Scholar] [CrossRef] [PubMed]
- Ragkousis, M.; Zenetos, A.; Ben Souissi, J.; Tsiamis, K.; Ferrario, J.; Marchini, A.; Edelist, D.; Crocetta, F.; Bariche, M.; Deidun, A.; et al. Unpublished Mediterranean and Black Sea records of marine alien, cryptogenic, and neonative species. BioInvasions Rec. 2023, 12, 339–369. [Google Scholar] [CrossRef]
- Capa, M. Taxonomic revision and phylogenetic relationships of apomorphic sabellids (Polychaeta) from Australia. Invertebr. Syst. 2007, 21, 537–567. [Google Scholar] [CrossRef]
- Hutchings, P.A.; Murray, A. Taxonomy of polychaetes from the Hawkesbury River and the southern estuaries of New South Wales, Australia. Rec. Aust. Mus. 1984, 3, 1–118. [Google Scholar] [CrossRef]
- Bonifazi, A.; Mancini, E.; Ventura, D. First record of the invasive and cryptogenic species Jassa slatteryi (Crustacea: Amphipoda) in Italian coastal waters. J. Sea Res. 2017, 136, 37–41. [Google Scholar] [CrossRef]
- Tempesti, J.; Mangano, M.C.; Langeneck, J.; Lardicci, C.; Maltagliati, F.; Castelli, A. Non-indigenous species in Mediterranean ports: A knowledge baseline. Mar. Environ. Res. 2020, 161, 105056. [Google Scholar] [CrossRef] [PubMed]
- Tempesti, J.; Langeneck, J.; Maltagliati, F.; Castelli, A. Macrobenthic fouling assemblages and NIS success in a Mediterranean port: The role of use destination. Mar. Pollut. Bull. 2020, 150, 110768. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).