Abstract
Littoraria melanostoma (Gray, 1839) is one of the most common species of gastropods in mangroves. They quickly respond during the early stage of mangrove restoration and usually form a dominant community within a certain period. We characterized the complete mitochondrial genome of this species. The whole mitogenome of L. melanostoma was 16,149 bp in length and its nucleotide composition showed a high AT content of 64.16%. It had 37 genes, including 13 protein-coding genes, 2 ribosomal RNA genes, and 22 transfer RNA genes, and 1 control region between tRNA-Phe and COX3. The A/T composition in the control region was 74.7%, and is much higher than the overall A/T composition of the mitochondrial genomes. The amino acid composition and codon usage of the mitochondrial genomes from seven superfamilies of Littorinimorpha were analyzed, and the results showed that CUU (Leu), GCU (Ala), AUU (Ile), UCU (Ser), UUA (Leu), GUU (Gly), and UUU (Phe) are the commonly used codons. The maximum likelihood phylogenetic tree reconstructed using 62 species of Littorinimorpha presented consistency between the molecular and morphological classifications, which provide a basis to understand the phylogeny and evolution of this order. In the phylogenetic tree, L. melanostoma is located within Littorinoidea and is closely related to L. sinensis, a rock-dwelling species that is widespread in the coastal intertidal zone of China.
1. Introduction
Mangroves are woody plant communities that become established on the intertidal flats of tropical and subtropical coasts. The harsh natural conditions of the coastal intertidal zone and the unique advantages of the transition zone between the land and sea have produced the unique biome of the mangrove ecosystem. In addition, the resulting extremely rich biodiversity also plays a crucial role in the ecosystem. As one mollusk group in mangroves, gastropods play an important role in the detritus cycle and consume a large amount of plant tissue and humus [1].
Littorinidae (Children, 1834), one of the major groups of arboreal gastropods in mangrove forests, adapt to the special environment of the coastal intertidal zone through numerous physiological and ecological manners, including their multiple reproductive means, which are known as a variable reproductive strategy [2]; complex food composition [3]; vertical climbing ability [4,5,6]; and variable shell color and shape [7]. As gastropods have a limited ability to move, they only generally move on the same tree without external interference [8], and it is difficult for them to migrate from the intertidal zone to land. Consequently, they have a relatively fixed pattern of spatial distribution [9,10]. Simultaneous studies have shown that species of Littorinidae respond quickly to the restoration of mangrove vegetation, and that they can form a dominant community in the early stage of vegetation restoration, as exemplified by Littoraria melanostoma [11]. Most of the Littorinidae in mangroves live on mangrove and salt marsh plants, driftwood, and stakes.
The family Littorinidae comprises more than 200 species that are common members of marine intertidal communities around the world, and most of them only live on mangrove plants [12]. Species of Littorinidae living on mangroves that have been reported in China include L. melanostoma, L. ardouiniana, L. intermedia, L. pallescens, L. articulata, and L. scabra, etc. [13,14,15]. L. melanostoma is often found in the branches and leaves of mangroves (Figure 1).

Figure 1.
(a) Specimen image of L. melanostoma. (b) L. melanostoma inhabited on the leaves of Avicennia marina (a common species of mangrove in China).
Morphological differences make the identification of gastropods confusing, and the classification information is constantly adjusted. In recent years, molecular systematic studies were used to analyze the evolution and phylogeny of gastropods. Reid et al. used nuclear 28S rRNA, mitochondrial 12S rRNA, and COI to construct the phylogeny of the family Littorinidae [12] and the genus Littoraria [16]. Li et al. used complete mitochondrial genome sequences to analyze the phylogenic relationship between the genera Littorina and Littoraria, but only four species were used in this study [17]. The phylogeny of Stromboidea, a superfamily in Littorinimorpha, was studied based on 13 mitochondrial protein-coding genes [18]. A phylogenetic study based on the mt genomes of deep-sea gastropods from Abyssochrysoidea indicated that this superfamily belonged to Littorinimorpha [19,20], but up to now, in the MolluscaBase (www.molluscabase.org), (accessed on 5 September 2023) their taxonomic status of order level remains uncertain [21].
Through searching in Genbank, we found that the number of gastropods with available mitogenomes has increased. Up to August 2023, the complete or partial mitochondrial genome of ~100 Littorinimorpha species were available in the National Center of Biotechnology Information (NCBI) (https://www.ncbi.nlm.nih.gov), but there were few studies on the phylogenetic analyses that investigate relationships across the Littorinimorpha.
In this study, L. melanostoma, one of the most common Gastropoda species in the mangrove wetlands of China, was studied for its molecular evolution and phylogeny. The mitochondrial genomes of L. melanostoma was sequenced, and the genome structure, base composition, codon usage, intergenic region, and codon preference of the mitochondrial genomes were analyzed. In addition, a phylogenetic tree of 62 species from 9 superfamilies of Littorinimorpha based on 13 protein-coding genes (PCGs) was constructed using the maximum likelihood (ML) method. This study may increase our understanding of the phylogeny and evolution of Littorinimorpha.
2. Materials and Methods
2.1. Sample Collection and DNA Extraction
Specimens of L. melanostoma was obtained from mangrove wetlands in Beihai, Guangxi, China (21.57° N, 109.16° E), and vouchered in the specimen room of the Guangxi Mangrove Research Center (Accession numbers LM#11-20, respectively). Since this species consists of unprotected invertebrates, no specific permission was required to collect samples from these locations. Total genomic DNA was obtained from the muscles of individuals using a QIAamp DNA Micro Kit (Qiagen, Hilden, Germany).
2.2. Sequencing, Assembling and Analysis
The cox1 gene was amplified using the universal primers LCO1490 and HCO2198 via the standard PCR method [22]. The extracted DNA was sequenced using a NovaSeq 6000 platform (Illumina, San Diego, CA, USA), and the mitogenome was assembled with NOVOPlasty v2.7.0 [23] and annotated with MitoZ v2.4 [24]. PCGs were determined through determining the open reading frames (ORFs) based on the invertebrate mitochondrial genetic code, and rRNAs and tRNAs were identified using the MITOS Web Server (http://mitos2.bioinf.uni-leipzig.de/index.py) [25]. The codon usage was calculated using MEGA 7.0 [26]. Strand bias was calculated using the following formulae: AT skew = (A − T)/(A + T), and GC skew = (G − C)/(G + C) [27]. The circular maps of the mitochondrial genomes were drawn using the online mitochondrial visualization tool Organellar Genome DRAW [28]. The nucleotide composition, codon usage, and comparative mitogenomic architecture tables for the mitogenomes and data that were used to plot the relative synonymous codon usage (RSCU) figures were all calculated/created using PhyloSuite [29].
2.3. Phylogenetic Analysis
The phylogenetic analyses were based on the concatenated nucleotide and amino acid alignments of 13 PCGs in 62 complete mt genomes, including L. melanostoma, and 61 species from 9 superfamilies of Littorinimorpha (Table 1). The nucleotide sequences of the complete mt genomes were downloaded from GenBank. The total quantity of complete mt genomes and amino acid sequences of 13 PCGs were aligned using MAFFT v.7.215 [30]. Following this, MACSE (multiple alignment of coding sequences) was used to account for frameshifts and stop codons with the score matrix ‘BLOSUM62’, which were then trimmed with Gblocks [31], with the minimum length of a block being ‘10’ and the maximum number of contiguous non-conserved positions being ‘8’, respectively. The phylogenetic tree was reconstructed using IQ-TREE v2 [32] based on ML with the partitioning method [33], and the parameter used was Bootstrap: ultrafast, num of bootstrap: 10,000, max of iteration: 1000, and minimum correlation coefficient: 0.9. The phylogenetic tree was visualized using the Interactive Tree Of Life (iTOL) v5 (https://itol.embl.de) [34].

Table 1.
Classification and origins of the mitogenomic sequences used in this study.
3. Results and Discussion
3.1. Genome Structure and Organization
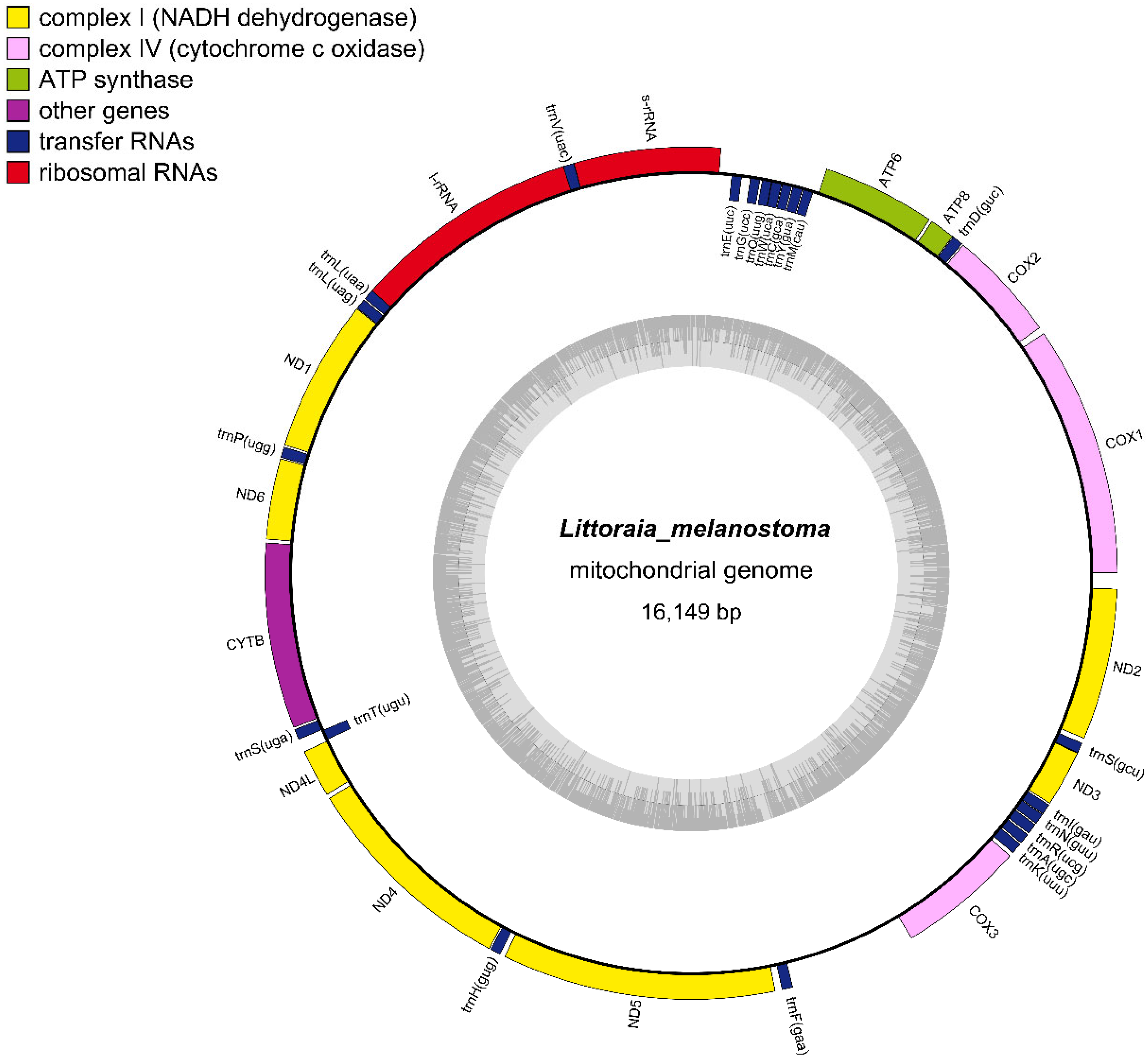
The complete L. melanostoma mitochondrial genome was 16,149 bp long, and it was uploaded to GenBank after annotation (ACCESSION ID: NC064398). The gene compositions of L. melanostoma were the same as most of the mitochondrial genomes of gastropods that have been published, and contained 37 genes, including 13 protein-coding, 2 rRNA, and 22 tRNA genes [24,35] (Figure 2). According to the difference in G+T content, the two strands of mitochondrial DNA could be separated into a heavy strand (H strand) and a light strand (L strand). The 13 protein-coding genes in the mitochondrial genomes of L. melanostoma are located on the H strand, which is consistent with the findings of previous studies that showed that the mitochondrial genomes of Littorinidae, such as Littorina fabalis, Littorina obtusata, and Littorinasaxatilis, harbor protein-encoding genes on the H strand [35]. Most genes were located on the H strand, except for the eight tRNAs that were located on the L strand, and include trnM (CAU), trnY (GUA), trnC (GCA), trnW (UCA), trnQ (UUG), trnG (UCC), trnE (UUC), and trnT (UGU). The thirteen PCGs include seven NADH dehydrogenase genes (complex I)—ND1, ND2, ND3, ND4, ND4L, ND5, and ND6; three cytochrome c oxidase genes (complex IV)—COX1, COX2, and COX3; two ATPase subunits (ATP6 and ATP8); and one cytochrome b gene.
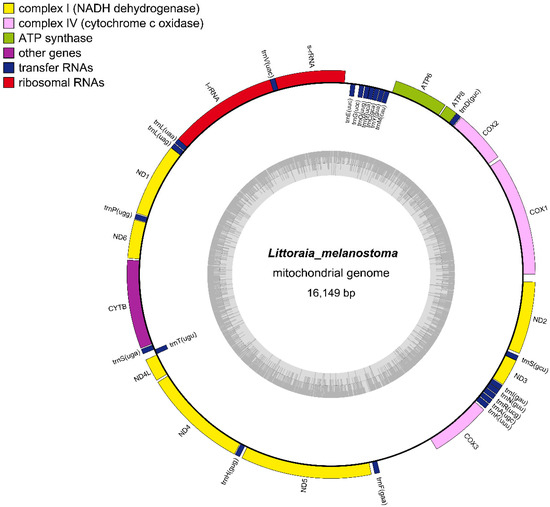
Figure 2.
Circular maps of the mitogenomes of L. melanostoma.
Table 2 summarizes the proportions of gene bases and protein-coding gene sequence bases in the complete mitochondrial genome sequences of L. melanostoma. The base composition of the L. melanostoma mitochondrial genome was 29.79% A, 34.37% T, 14.66% G, and 21.18% C. The A+T content (64.16%) of its mitochondrial genes was higher than the G+C content (35.84%), and the A+T content of the protein-coding genes was 62.39%. These results indicate that L. melanostoma genomes display an obvious nucleotide composition that is biased to A+T, which is consistent with the other genomes of Littorinidae species that have been reported [35]. The base composition bias is usually reflected by the AT skew and GC skew. The calculated AT skew and GC skew of the L. melanostoma mitochondrial genome were 0.071 and −0.182, respectively. These data indicate that the bases T and C appear more frequently than A and G in the mitochondrial genomes of L. melanostoma.

Table 2.
Composition and base content of the L. melanostoma protein-coding genes.
3.2. PCGs and Codon Usage
The nucleotide lengths of the 13 PCGs of L. melanostoma were 11,034 bp, which encode 3678 amino acid residues in total. Most of the PCGs that were examined began with the ATN codon and ended with either the TAA or TAG codons (Table 3).

Table 3.
Mitogenomic organization of L. melanostoma.
The codon usage of 13 PCGs in the L. melanostoma mitochondrial genomes is shown in Table 3. In the L. melanostoma genome, only one gene (ND3) used ATA as its start codon, while two used ATT as their start codon, namely ND4 and ND5. The remaining 10 genes (COX1, COX2, ATP8, ATP6, ND1, ND6, CYTB, ND4L, COX3, and ND2) all used ATG as their start codon. ND4L, ND5, and ND2 used TAG, CTT, and AAT as their stop codon, respectively, and these codons were each used by only one gene. There were 10 genes (COX1, COX2, ATP8, ATP6, ND1, ND6, CYTB, ND4, COX3, and ND3) that used TAA as their stop codon.
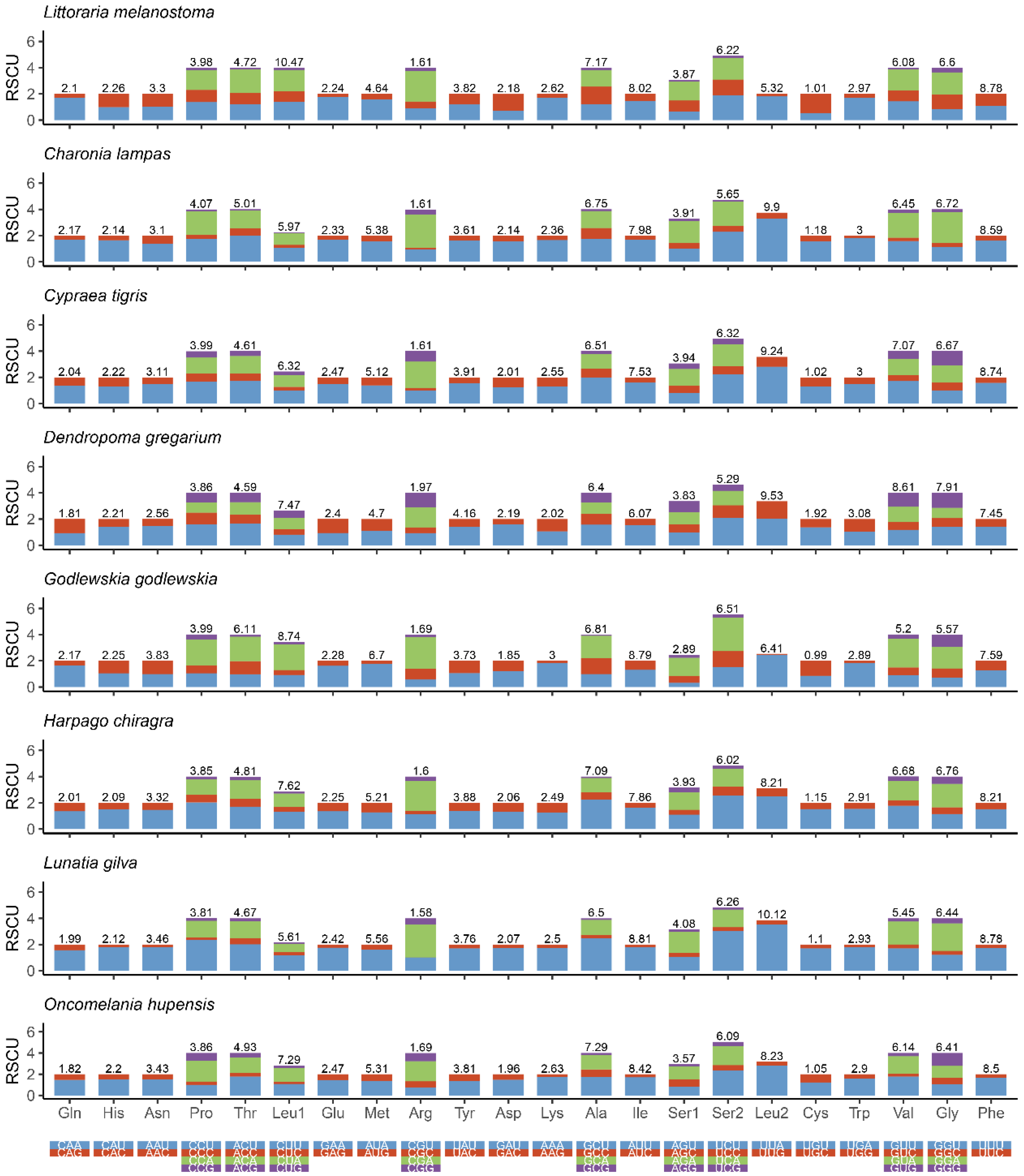
Figure 3 shows the amino acid composition and codon usage of the mitochondrial genomes of eight species from seven superfamilies. The results showed that CUU (Leu), GCU (Ala), AUU (Ile), UCU (Ser), UUA (Leu), GUU (Gly), and UUU (Phe) were the mostly commonly used codons. These observations suggest that there is a strong AT bias for protein-coding genes in the mitochondrial genomes of Littorinidae animals.
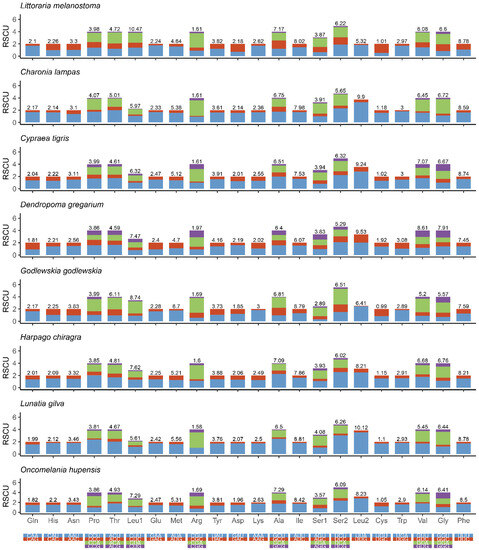
Figure 3.
Relative synonymous codon usage (RSCU) in the mitogenomes of eight species of Littorinimorpha.
3.3. Ribosomal and Transfer RNA Genes
Two rRNA genes, l-rRNA and s-rRNA, are located between trnL (UAA) and trnV (UAA), and between trnV (UAA) and trE (UUC), respectively.
In the mitochondrial genome of L. melanostoma, l-rRNA was 1419 bp, and s-rRNA was 901 bp (Table 3). A total of 22 tRNA genes were found in L. melanostoma, and its cloverleaf structures was 65–72 bp.
3.4. Intergenic Spaces and Overlapping Sequences
There were five overlapping gene regions in the mitochondrial genome of L. melanostoma, which ranged from 1 to 22 bp in length, and 30 intergenic regions, which ranged from 1 to 773 bp long. The longest intergenic region was located between trnF (GAA) and COX3 (Table 3).
3.5. Control Regions
The control region (CR) of mitochondrial DNA is the primary non-coding region of the mitochondrial genome of animals, also known as the D-loop region, which is a key part for the replication and transcription of the mitochondrial genome and regulates the replication and transcription of the mitochondrial genome. During the process of evolution, since the selection pressure that acts on this region is relatively non-intrusive, the CR usually displays the largest sequence and variation in length, the highest rate of evolution, and is the most polymorphic in the mitochondrial genome [36]. However, since the UTR sequences of invertebrates are poorly conserved, there is no defined CR in their mitochondrial genomes [37]. For example, Marques studied the genomes of L. fabalis, L. obtusata, and L. saxatilis, and found a region that contained some unique features, such as a non-coding region with a hairpin structure and a tandem repeat sequence, located between tRNA-Phe (trnF [GAA]) and COX3, and an AT content that was higher than the overall AT content in the mitochondrial genome. This region was then predicted as the CR. Similarly, we found a non-coding sequence that contained some unique features in the L. melanostoma genomes. It was between tRNA-Phe and COX3, and the AT content was 74.7%. This is much higher than the AT content of the mitochondrial genomes (64.16%). Thus, we consider that this region is a unique non-coding region of the Littorinoidea superfamily, which may play a regulatory role in the replication and transcription of the mtDNA of this genus.
3.6. Phylogenetic Analyses
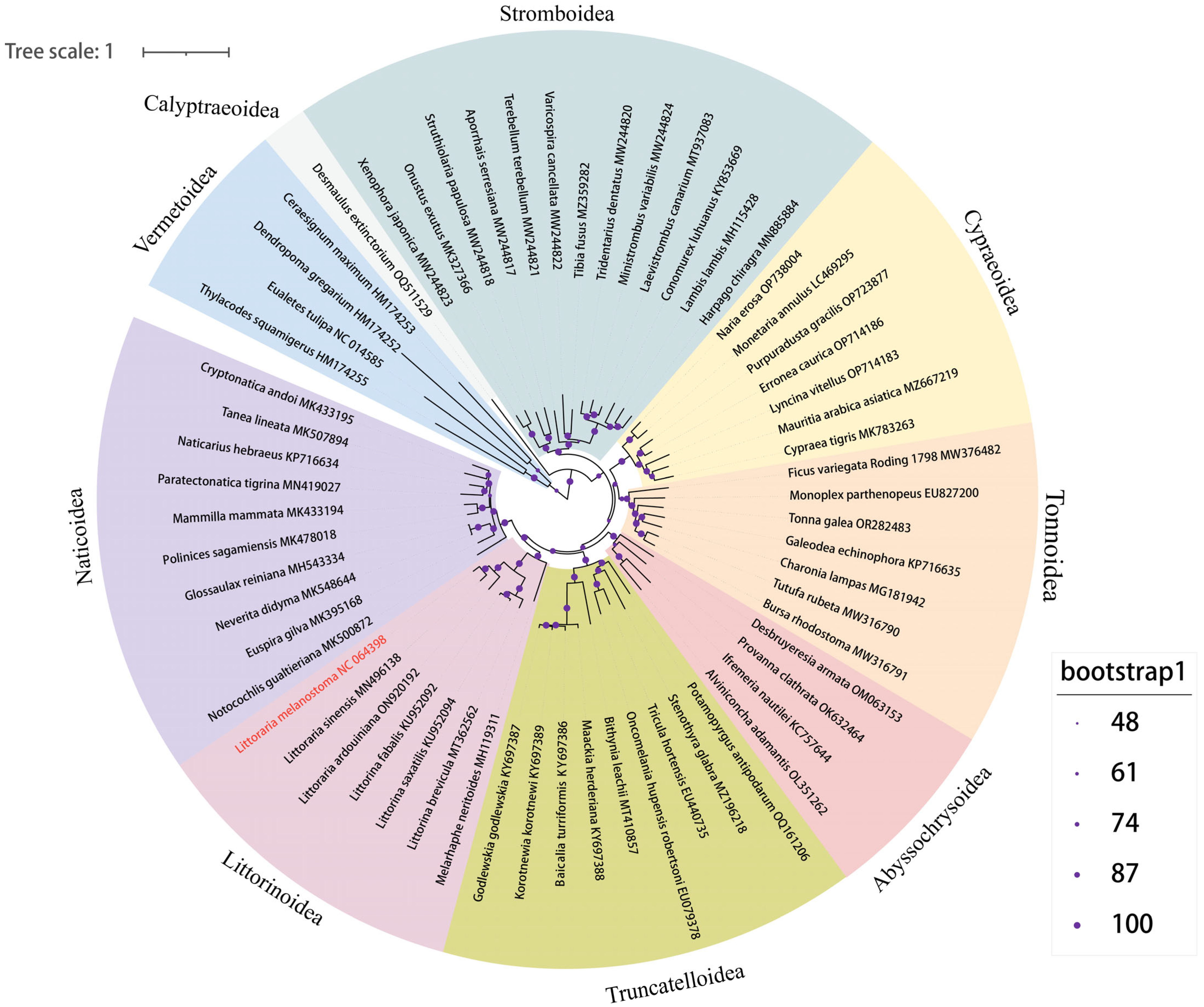
To further study the genetic background and taxonomic relationship of Littorinimorpha, the complete mitochondrial genome sequences of L. melanostoma were compared with the complete mitochondrial genome sequences of sixty-one other species from nine superfamilies of Littorinimorpha. A phylogenetic tree was constructed based on 13 PCGs using IQ-TREE v2 with the ML method (Figure 4). The phylogenetic relationship was found to be significantly consistent with the results using the traditional classification method. The protagonist of this study, L. melanostoma, was included in the Littorinoidea clade, and is closely related to L. sinensis, which is a rock-dwelling species that is widespread in the coastal intertidal zone of China, just as in the previous study [16]. Littorinoidea and Naticoidea were grouped into one clade, which is consistent with a previously published study [38]. A further previous study confirmed that Vermetoidea is placed at the basal position within Littorinimorpha, and our study obtained a consistent conclusion in which the four Vermetoidea species formed a sister clade of the other superfamilies [39]. The other eight superfamilies were grouped into one clade, while Calyptraeoidea presented a sister branch of the other superfamilies. The mt genome of D. extinctorium is the only species of Calyptraeoidea superfamily, which was uploaded in Genbank, which indicates that this superfamily lacks attention. The position of four deep-sea snails from Abyssochrysoidea supported the placement of this superfamily belonging to the order Littorinimorpha, just as in a previously published study.
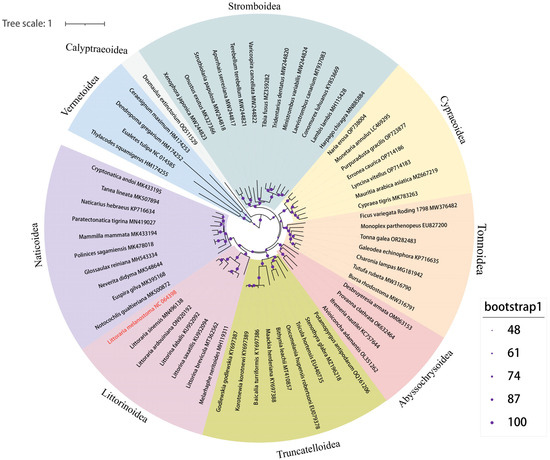
Figure 4.
Maximum likelihood phylogenetic tree inferred from the 13 PCGs of Littorinimorpha. Different background colors mark the 9 superfamilies. Bootstraps values were indicated by different sizes of the purple symbol, as shown in the legend. The species which sequenced the mt genome in this study was colored in red.
4. Conclusions
In this study, the mitogenomes of L. melanostoma were sequenced, and 37 genes (13 PCGs, 22 tRNA genes, and 2 rRNA genes) and 1 control region were located in positions considered typical of a Littorinoidea mitogenome. The ML phylogenetic relationships based on the 13 PCGs of the order Littorinimorpha were analyzed, indicating that the basis for the relationship based on a molecular analysis is consistent with that of the traditional morphological method.
Author Contributions
K.C. drafted the manuscript and performed data analysis. M.Y. collected and processed animal samples. X.L. designed and conceived the experiment and performed the data analysis. H.D. and X.L. edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by [National Natural Science Foundation of China] grant number [32060282], [Special funding for Science and Technology bases and talents of Guangxi Province] grant number [AD20159032], [Open Research Fund Program of Guangxi Key Lab of Mangrove Conservation and Utilization] grant number [GKLMC-202102], and [Guangxi Key Research and Development Program funding] grant number [AB19245045]. The APC was funded by [Guangxi Key Research and Development Program funding] grant number [AB19245045].
Institutional Review Board Statement
Since this species consists of unprotected invertebrates, no specific permission was required to collect samples from these locations.
Data Availability Statement
All relevant data are within the paper.
Acknowledgments
We thank Mengling Liu from the Marine Environment Monitoring Central Station of Guangxi for identifying animal samples and thank Yunqing Liu from the Luoyang Institute of Science and Technology for his guidance of the bioinformatics.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Torres, P.; Alfiado, A.; Glassom, D.; Jiddawi, N.; Macia, A.; Reid, D.G.; Paula, J. Species composition, comparative size and abundance of the genus Littoraria (Gastropoda: Littorinidae) from different mangrove strata along the East African coast. Hydrobiologia 2008, 614, 339–351. [Google Scholar] [CrossRef]
- Berry, A.; Chew, E. Reproductive systems and cyclic release of eggs in Littorina melanostoma from Malayan mangrove swamps (Mollusca: Gastropoda). J. Zool. 1973, 171, 333–344. [Google Scholar] [CrossRef]
- Lee, O.H.; Williams, G.A.; Hyde, K.D. The diets of Littoraria ardouiniana and L. melanostoma in Hong Kong mangroves. J. Mar. Biol. Assoc. UK 2001, 81, 967–973. [Google Scholar] [CrossRef]
- Hamilton, P. Intertidal distribution and long-term movements of Littorina irrorata (Mollusca: Gastropoda). Mar. Biol. 1978, 46, 49–58. [Google Scholar] [CrossRef]
- Alfaro, A.C. Diet of Littoraria scabra, while vertically migrating on mangrove trees: Gut content, fatty acid, and stable isotope analyses. Estuar. Coast. Shelf Sci. 2008, 79, 718–726. [Google Scholar] [CrossRef]
- Little, C.; Stirling, P. Activation of a mangrove snail, Littorina scabra scabra (L.) (Gastropoda: Prosobranchia). Mar. Freshwater Res. 1984, 35, 607–610. [Google Scholar] [CrossRef]
- Cook, L. Systematic effects on morph frequency in the polymorphic mangrove snail Littoraria pallescens. Heredity 1990, 65, 423–427. [Google Scholar] [CrossRef]
- Lee, O.H.; Williams, G.A. Spatial distribution patterns of Littoraria species in Hong Kong mangroves. Hydrobiologia 2002, 481, 137–145. [Google Scholar] [CrossRef]
- Blanco, J.F.; Cantera, J.R. The vertical distribution of mangrove gastropods and environmental factors relative to tide level at Buenaventura Bay, Pacific Coast of Colombia. Bull. Mar. Sci. 1999, 65, 617–630. [Google Scholar]
- Reid, D.G. Habitat and zonation patterns of Littoraria species (Gastropoda: Littorinidae) in Indo-Pacific mangrove forests. Biol. J. Linn. Soc. 1985, 26, 39–68. [Google Scholar] [CrossRef]
- Chen, S.Y.; Chen, B.; Liao, J.J.; Chen, G.L.; Huang, Y.; Chen, G.C. Composition and distribution pattern of Littorinid snails in young rehabilitated mangroves. Chin. J. Ecol. 2017, 36, 460. [Google Scholar]
- Reid, D.G.; Dyal, P.; Williams, S.T. A global molecular phylogeny of 147 periwinkle species (Gastropoda, Littorininae). Zool. Scr. 2012, 41, 125–136. [Google Scholar] [CrossRef]
- Lai, T.H.; He, B.Y. Studies on the macrobenthos species diversity for Guangxi mangrove areas. Guangxi Sci. 1998, 5, 166–172. [Google Scholar]
- Chen, G.; Ye, Y. Restoration of Aegiceras corniculatum mangroves in Jiulongjiang Estuary changed macro-benthic faunal community. Ecol. Eng. 2011, 37, 224–228. [Google Scholar] [CrossRef]
- Tang, C.Y. Vertical Distribution Patterns of Mangrove Arboreal Mollusks and Its Influencing Factors. Master’s Thesis, Xiamen University, Xiamen, China, 2019. [Google Scholar]
- Reid, D.G.; Dyal, P.; Williams, S. Global diversification of mangrove fauna: A molecular phylogeny of Littoraria (Gastropoda: Littorinidae). Mol. Phylogenet Evol. 2010, 55, 185–201. [Google Scholar] [CrossRef] [PubMed]
- Li, M.Y.; Li, Y.L.; Xing, T.F.; Liu, J.X. First mitochondrial genome of a periwinkle from the genus Littoraria: Littoraria sinensis. Mitochondr. DNA 2019, 4, 4124–4125. [Google Scholar] [CrossRef] [PubMed]
- Irwin, A.R.; Strong, E.E.; Kano, Y.; Harper, E.M.; Williams, S.T. Eight new mitogenomes clarify the phylogenetic relationships of Stromboidea within the caenogastropod phylogenetic framework. Mol. Phylogenet Evol. 2021, 158, 107081. [Google Scholar] [CrossRef]
- Lee, W.K.; Hou, B.K.; Ju, S.J.; Kim, S.J. Complete mitochondrial genome of the hydrothermal vent provannid snail Alviniconcha boucheti (Gastropoda: Abyssochrysoidea) from the North Fiji Basin. Mitochondr. DNA 2020, 5, 1848–1849. [Google Scholar] [CrossRef]
- Xu, T.; Sun, J.; Chen, C.; Qian, P.Y.; Qiu, J.W. The mitochondrial genome of the deep-sea snail Provanna sp. (Gastropoda: Provannidae). Mitochondr. DNA 2016, 27, 4026–4027. [Google Scholar] [CrossRef]
- MolluscaBase. 2023. Available online: https://www.molluscabase.org (accessed on 5 September 2023). [CrossRef]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotech. 1994, 3, 294–299. [Google Scholar]
- Dierckxsens, N.; Mardulyn, P.; Smits, G. NOVOPlasty: De novo assembly of organelle genomes from whole genome data. Nucleic Acids Res. 2017, 45, e18. [Google Scholar]
- Meng, G.; Li, Y.; Yang, C.; Liu, S. MitoZ: A toolkit for animal mitochondrial genome assembly, annotation and visualization. Nucleic Acids Res. 2019, 47, e63. [Google Scholar] [CrossRef] [PubMed]
- Bernt, M.; Donath, A.; Jühling, F.; Externbrink, F.; Florentz, C.; Fritzsch, G.; Pütz, J.; Middendorf, M.; Stadler, P.F. MITOS: Improved de novo metazoan mitochondrial genome annotation. Mol. Phylogenet Evol. 2013, 69, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Lohse, M.; Drechsel, O.; Kahlau, S.; Bock, R. OrganellarGenomeDRAW—A suite of tools for generating physical maps of plastid and mitochondrial genomes and visualizing expression data sets. Nucleic Acids Res. 2013, 41, W575–W581. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Gao, F.; Jakovlić, I.; Zou, H.; Zhang, J.; Li, W.X.; Wang, G.T. PhyloSuite: An integrated and scalable desktop platform for streamlined molecular sequence data management and evolutionary phylogenetics studies. Mol. Ecol. Resour. 2020, 20, 348–355. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Talavera, G.; Castresana, J. Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst. Biol. 2007, 56, 564–577. [Google Scholar] [CrossRef]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; Von Haeseler, A.; Lanfear, R. IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.; Von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL) v5: An online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021, 49, W293–W296. [Google Scholar] [CrossRef] [PubMed]
- Marques, J.P.; Sotelo, G.; Larsson, T.; Johannesson, K.; Panova, M.; Faria, R. Comparative mitogenomic analysis of three species of periwinkles: Littorina fabalis, L. obtusata and L. saxatilis. Mar. Genom. 2017, 32, 41–47. [Google Scholar] [CrossRef]
- Song, W.T.; Gao, X.G.; Li, Y.F.; Liu, W.D.; Liu, Y.; He, C.B. Comparison of mitochondrial genomes of bivalves. Hereditas 2009, 31, 1127–1134. [Google Scholar] [CrossRef]
- Hoffmann, R.J.; Boore, J.; Brown, W. A novel mitochondrial genome organization for the blue mussel, Mytilus Edulis. Genetics 1992, 131, 397–412. [Google Scholar] [CrossRef]
- Jiang, D.; Zheng, X.; Zeng, X.; Kong, L.; Li, Q. The complete mitochondrial genome of Harpago chiragra and Lambis lambis (Gastropoda: Stromboidea): Implications on the Littorinimorpha phylogeny. Sci. Rep. 2019, 9, 17683. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.H.; Hwang, U.W. The complete mitochondrial genome of an endangered triton snail Charonia lampas (Littorinimorpha: Charoniidae) from South Korea. Mitochondr. DNA 2021, 6, 956–958. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).