Abstract
Background/Objectives: The risk of fractures associated with immune checkpoint inhibitors (ICIs) is increasing; however, the relationship between fracture risk and potential factors, such as osteoporosis and hyperthyroidism, remains unclear. Methods: Using VigiBase, the World Health Organization’s global pharmacovigilance database, we investigated the signals for osteoporosis, hyperthyroidism, and fractures associated with ICIs (nivolumab, pembrolizumab, atezolizumab, durvalumab, ipilimumab, and tremelimumab) by calculating information components (ICs) and their 95% confidence intervals (CIs). Furthermore, we estimated the association between the occurrence of fractures in patients receiving ICIs and osteoporosis or hyperthyroidism. Results: Signals of hyperthyroidism (IC = 4.66, 95% CI: 4.58–4.73), but not osteoporosis (IC = −1.79, 95% CI: −2.22 to −1.36) or fractures (IC = −0.21, 95% CI: −0.36 to −0.06), were detected in patients using ICIs. Osteoporosis (odds ratio: 118.00, 95% CI: 61.00–230.00) was associated with an increased reporting frequency of fractures related to ICIs, whereas hyperthyroidism (odds ratio: 0.60, 95% CI: 0.19–1.87) was not associated with such an increase. Conclusions: The VigiBase analysis indicates that the use of ICIs does not increase the reporting frequency of osteoporosis or fractures. Additionally, hyperthyroidism did not increase the reporting frequency of fractures associated with ICIs.
1. Introduction
Advances in cancer immunotherapy using immune checkpoint inhibitors (ICIs) have resulted in a paradigm shift in cancer treatment. ICIs inhibit immune checkpoint proteins, such as programmed cell death protein 1 (PD-1), programmed death-ligand 1 (PD-L1), and cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4), thereby activating immunity through T lymphocyte activation and exerting anticancer effects. By contrast, ICIs can induce immune-related adverse events (irAEs) that are specific to these therapies. IrAEs are off-target toxic effects resulting from an excessively activated immune system. IrAEs can occur in any organ system, including the respiratory, cardiovascular, central nervous, gastrointestinal, hematological, and musculoskeletal systems, and skin [1].
A recent report proposed that osteoporotic fractures may be an unrecognized adverse event [2]. It suggests that T cells activated by ICIs may increase the expression of the receptor activator of nuclear factor-κB ligand (RANKL) and other cytokines, including tumor necrosis factor (TNF)-α and interleukin (IL)-18, IL-17, and IL-12, potentially leading to the development of osteoporosis. Indeed, the activation of T cells is known to induce the production of RANKL and TNF-α [3,4,5]. Immune cells have been suggested to play a crucial role in bone remodeling [6,7]. Bone-marrow-derived macrophages are known to polarize into M1 macrophages under sustained RANKL stimulation, subsequently differentiating into osteoclasts [8]. Macrophages also enhance the release of TNF-α, IL-6, IL-1, IL-12, IL-18, IL-23, and reactive oxygen species [9,10]. In addition, myeloid-derived suppressor cells have been shown to differentiate into osteoclasts in the presence of RANKL and macrophage colony stimulating factor [11,12,13]. Thus, the enhancement of immune responses by ICIs and associated effects on bone remodeling may increase the risk of fractures; however, clinical evidence remains limited.
A case series in humans reported six patients treated with ICIs who exhibited fractures or resorptive bone lesions [14]. Vertebral compression fractures and resorptive lesions—including those of the shoulder, hand, and clavicle—were observed. Elevated bone resorption markers were noted in five of the six patients. Another case series reported that three patients treated with ICIs presented with vertebral fractures; one patient exhibited a calcaneal fracture [15]. In a retrospective administrative database study, the incidence rate ratio of sustaining a major fracture in the year after versus prior to ICI initiation was reported to be 2.43 (95% confidence interval [CI]: 1.34–4.27) among patients treated using ICIs [16]. Based on these reports, ICIs may promote osteoporosis and increase the risk of fractures.
In contrast, ICIs can also cause endocrinopathies, such as hyperthyroidism and hypothyroidism, as one of the irAEs [17,18,19]. Among them, hyperthyroidism has been associated with an increased risk of osteoporosis and fractures [20,21]. It is a representative endocrine abnormality caused by ICIs, with a relatively high incidence of 3.9–8.5% and 10.2–16.4% in patients receiving PD-1/PD-L1 and combination therapy with CTLA-4 inhibitors, respectively [22,23]. Clarifying the relationships between ICI fracture risk, osteoporosis, and hyperthyroidism will provide important evidence that will contribute to the proper use of ICIs.
Recently, pharmacovigilance signal detection studies have increasingly utilized large-scale databases of adverse drug reactions (ADRs) collected through spontaneous reporting systems [24,25]. Spontaneous reporting systems and pharmacovigilance databases play a crucial role in identifying previously unrecognized AEs that were not detected in clinical trials and in evaluating drug safety based on real-world use in specific populations and clinical settings [26,27]. In pharmacovigilance studies, signal detection methods such as disproportionality analysis using reporting odds ratios and information components (ICs) are employed to determine the incidence of AEs [28,29,30]. VigiBase, provided by the World Health Organization (WHO), is a global pharmacovigilance database containing spontaneous ADR reports. Since its inception in 1964, VigiBase has accumulated data on approximately 36.6 million patients and 92.6 million ADRs. This extensive database is particularly valuable for identifying signals of rare ADRs, such as fractures in patients treated with ICIs.
Therefore, this study analyzed VigiBase to investigate the following: first, the presence of signals for osteoporosis and hyperthyroidism, which are risk factors for ICI-associated fractures; second, the presence of signals for fractures; and third, the association between fractures and osteoporosis or hyperthyroidism in patients using ICIs.
2. Results
2.1. Signals of Osteoporosis and Hyperthyroidism
The ICs and their 95% CIs were calculated using a three-by-three contingency table (Table 1). The ICs for osteoporosis and hyperthyroidism, reported as the ADRs of ICIs, are shown in Table 2. Signals were not detected for osteoporosis for all ICIs (patients reported to use at least one ICI, IC [95% CI]: −1.79 [−2.22 to −1.36]) or each ICI (IC [95% CI]: nivolumab, −1.46 [−2.04 to −0.89]; pembrolizumab, −1.87 [−2.61 to −1.12]; atezolizumab, −2.23 [−3.67 to −0.79]; durvalumab, −1.92 [−3.96 to 0.12]; ipilimumab, −2.07 [−3.16 to −0.98]; tremelimumab, not applicable [N.A.]). When hyperthyroidism, a typical irAE of ICIs, was evaluated, signals were detected for all ICIs (IC [95% CI]: 4.66 [4.58–4.73]) and each ICI (IC [95% CI]: nivolumab, 4.87 [4.77–4.98]; pembrolizumab, 4.33 [4.18–4.47]; atezolizumab, 4.47 [4.25–6.49]; durvalumab, 4.65 [4.34–4.96]; ipilimumab, 4.89 [4.73–5.05]), excluding tremelimumab (IC [95% CI]: 1.25 [−0.42 to 2.92]).

Table 1.
Three-by-three contingency table.

Table 2.
Information components of immune checkpoint inhibitors for osteoporosis and hyperthyroidism.
2.2. Signals of Fractures
The IC values of the fractures, reported as ADRs of the ICIs, are shown in Table 3. Signals were not detected for fractures for all ICIs (IC [95% CI]: −0.21 [−0.36 to −0.06]) or each ICI (ICs [95% CIs]: nivolumab, −0.09 [−3.08 to 0.12]; pembrolizumab, −0.41 [−0.68 to −0.14]; atezolizumab, −0.03 [−0.37 to 0.43]; durvalumab, −0.38 [−1.12 to 0.37]; ipilimumab, −0.02 [−0.34 to 0.31]; tremelimumab, −0.55 [−2.59 to 1.50]).

Table 3.
Information components of immune checkpoint inhibitors for fractures.
2.3. Association Between Fractures and Osteoporosis or Hyperthyroidism in Patients with ICIs
To investigate the association between fractures and osteoporosis or hyperthyroidism in patients treated with ICIs, multivariable logistic regression analysis was performed. Covariates included age (≥65 years) and gender (female), which generally affect the occurrence of fractures. Multivariable analysis revealed that an age ≥ 65 years, female sex, and osteoporosis (but not hyperthyroidism) were associated with an increased reporting frequency of fractures in patients treated using ICIs (odds ratio [95% CI]: age ≥ 65 years, 1.67 [1.35–2.07]; female sex, 1.56 [1.27–1.91]; osteoporosis, 118.00 [61.00–230.00]; hyperthyroidism, 0.60 [0.19–1.87]) (Table 4).

Table 4.
Multivariable logistic analysis for the occurrence of fractures in patients with immune checkpoint inhibitors.
3. Discussion
The acceleration of osteoporosis and the associated risk of fractures by ICIs has been proposed; yet, unexpectedly, our study did not detect any signals for either osteoporosis or fractures associated with ICI use using VigiBase. Previous reports have evaluated the fracture signals associated with ICIs in the FDA Adverse Event Reporting System (FAERS), a worldwide pharmacovigilance database with a focus on the U.S. [15,31]. Filippini et al. [15] reported that ICIs—classified as PD-1 (nivolumab, pembrolizumab, cemiplimab), PD-L1 (atezolizumab, durvarmab, avelumab), and CTLA-4 (ipilimumab, tremelimumab) inhibitors—showed several types of fracture signals among the preferred terms (PTs) defined by the Medical Dictionary for Regulatory Activities (MedDRA) high-level group term (HLGT) “Bone and Joint Injuries”. This included pathological fracture, spinal compression fractures, and femoral neck fractures. Liu et al. [31] reported that no ICIs (nivolumab, pembrolizumab, atezolizumab, durvalumab, or ipilimumab) were associated with signals of overall fracture; however, signals for certain PTs of fractures, such as spinal compression fracture, thoracic vertebral fracture, and osteoporotic fracture, were detected for several ICIs. This report also indicated that no osteoporotic signals were detected in each ICI. Based on these reports, ICIs may partially increase the risk of certain fractures (PT levels); however, they are unlikely to significantly increase the overall risk of fracture. Our results also support this finding, as no signals were detected in the 78 fracture PT groups encompassed under the HLGT “Fractures”. Given that ICIs activate T cells, which, in turn, stimulate osteoclasts by increasing RANKL and cytokine levels, the risk of osteoporosis and fractures associated with ICIs is considered to occur uniformly, regardless of the target of action of the ICIs (i.e., PD-1, PD-L1, or CTLA-4). In the aforementioned studies, fracture and osteoporosis signals were evaluated for each ICI, or for groups of ICI targets of action. However, in this study, no fractures or osteoporosis signals were detected using VigiBase, even when all six ICIs were evaluated as a group. Therefore, further evidence is needed, and more cautious discussions are required regarding the potential increased risk of osteoporosis and fractures associated with ICIs.
Additionally, fracture risk may be affected by both osteoporosis and hyperthyroidism. Although we detected a signal for hyperthyroidism associated with ICIs, hyperthyroidism did not affect the reporting frequency of ICI-related fractures. Based on our analysis using VigiBase, it is unlikely that hyperthyroidism (an irAE) increases the risk of ICI-associated fractures. A meta-analysis reported that hyperthyroidism (thyroid-stimulating hormone [TSH] < 0.45 mIU/L) increases the risk of fractures, with an even higher risk observed when TSH levels are <0.10 mIU/L [32]. One proposed mechanism is the expression of TSH receptors in osteoblasts and osteoclasts, where changes in TSH activity may accelerate bone turnover, thereby increasing fracture risk [33]. In contrast, a recent study investigating the association between thyroid dysfunction and osteoporosis using publicly available databases reported that hyperthyroidism, FT3, FT4, and TSH levels were not risk factors for osteoporosis [34]. Based on these findings, the fracture risk in hyperthyroidism may vary depending on factors such as the underlying mechanism, severity, duration of the condition, and TSH levels. In our study, hyperthyroidism did not affect the reporting frequency of ICI-associated fractures, suggesting that the pathological changes in hyperthyroidism caused by ICIs do not lead to the functional alterations that induce fractures. Factors associated with fractures induced by ICI were elderly age, female sex, and osteoporosis, which are commonly known to affect fractures. Furthermore, while the risk of developing osteoporosis due to ICIs does not appear to be elevated, patients at risk of osteoporosis should be closely monitored for potential fractures.
This study has some limitations; first, it should be noted that VigiBase—comprising individual case safety reports corrected from spontaneous reporting systems—is a passive reporting database with many biases, including under-reporting, over-reporting, and confounding by comorbidities. Second, the diagnosis of osteoporosis requires the measurement of bone turnover markers, such as bone-specific alkaline phosphatase and bone mineral density [35,36]. However, not all patients receiving ICIs undergo regular bone density measurements, raising the possibility of osteoporosis under-reporting. Third, the influence of risk factors for fractures, such as glucocorticoid therapy associated with ICI therapy, bone metastasis, and fall risk, was not excluded. Nevertheless, this study suggests results that differ from the newly proposed hypothesis of increased osteoporosis and fracture risk associated with ICIs; thus, the hypothesis presented is crucial and warrants further investigation.
4. Materials and Methods
4.1. Data Source
The WHO VigiBase database is one of the most extensive global pharmacovigilance databases with over 36 million reports collected from >130 countries worldwide, based on post-marketing pharmacovigilance activity. In this study, data from December 1964–2023 were downloaded and processed.
4.2. Dataset Creation
A flowchart illustrating the creation of the disproportionality analysis dataset is shown in Figure 1. First, suspected duplicates included in the “SUSPECTEDDUPLICATES” table—made by statistical algorithms developed by the World Health Organization Collaborating Centre—were excluded from the “DEMO” table. Additionally, after excluding patients of unknown sex and age, 26,019,605 patients were included in the analysis. Drug_name from the “MP” table, Substance_Id and Substance_name from the “SUN” table, and ATC_code from the “ATC” table were added to the “DRUG” table (Modified DRUG Table). The corresponding cases were extracted from the “Modified DRUG” and “ADR” tables according to the definitions of ICIs and ADRs, respectively, and linked to the analysis population to create an analysis dataset for the disproportionality analysis. VigiBase was processed using Navicat 16 in SQLite ver. 16.3.3 (PremiumSoft, Osaka, Japan).
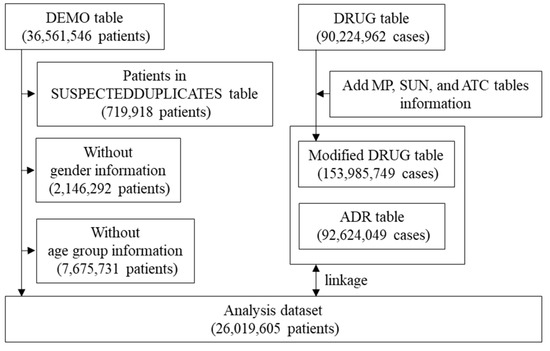
Figure 1.
Flow chart for the creation of the disproportionality analysis dataset.
4.3. Definition of Immune Checkpoint Inhibitors
Six ICIs (Substance Id) were included in the study: PD-1 inhibitors: nivolumab (27193) and pembrolizumab (28886); PD-L1 inhibitors: atezolizumab (32021) and durvalumab (31669); CTLA-4 inhibitors: ipilimumab (17640) and tremelimumab (18514, 38501). Nivolumab, pembrolizumab, atezolizumab, durvalumab, and ipilimumab were selected as representative ICIs, as they each had more than 3000 reported cases in the analysis dataset. Additionally, tremelimumab was included in the analysis since it is used in combination with durvalumab.
4.4. Definition of Adverse Drug Reactions
Osteoporosis, hyperthyroidism, and fractures were defined according to the PTs in MedDRA 26.0 J. Eleven PTs were associated with osteoporosis, as defined by Standardized MedDRA Queries (SMQs) for Osteoporosis/Osteopenia (SMQ code 20000178), and nineteen PTs were associated with hyperthyroidism, as defined by SMQs for Hyperthyroidism (SMQ code 20000161; Tables S1 and S2). Seventy-eight PTs were identified as fractures from PTs grouped under six high-level terms (HLTs) within the Fractures HLGT (HLGT code 10017322): seven PTs in Skull and Face Fractures (HLT code 10040958), four PTs in Thoracic Cage Fractures Non-spinal (HLT code 10072987), ten PTs in Spinal Column Fractures (HLT code 10072985), four PTs in Pelvic Fractures (HLT code 10034246), twenty-nine PTs in Limb Fractures (HLT code 10075885), and twenty-four PTs in Fractures NEC (HLT code 10072986; Table S3).
4.5. Statistical Analysis
In the disproportionality analysis, ICs were used to evaluate the signal detection of ADRs. The IC is an ADR signal index of the Bayesian Confidence Propagation Neural Network (BCPNN) analysis, calculated based on the Bayesian statistical approach developed by the WHO Uppsala Monitoring Center [29,37]. It can be used to detect ADR signals even in small samples [38,39]. The ICs and their 95% CIs were calculated using a three-by-three contingency table (Table 1) and equations as below. ADR signals were considered positive if the lower limit of the 95% CI for the IC was >0 [40].
- IC equations:
Multivariate logistic regression analyses were performed on data from patients using ICIs that were extracted from the analysis dataset. Statistical significance was set at p < 0.05. Statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), a graphical user interface for R version 4.2.2 (The R Foundation for Statistical Computing, Vienna, Austria) based on a modified version of R Commander (version 1.61) designed to add statistical functions frequently used in biostatistics [41].
5. Conclusions
This study using VigiBase suggests that the ICI-associated osteoporosis incidence and consequent increase in fracture incidence are unlikely. Although ICIs may increase the incidence of hyperthyroidism, it is unlikely to lead to a higher incidence of fractures. Commonly known fracture risk factors such as elderly age, female sex, and osteoporosis also elevate the risk of fractures in patients using ICIs, emphasizing the need for caution irrespective of ICI use. Therefore, further investigation, including the use of other real-world databases, is needed to better understand the impact of ICIs on osteoporosis and fracture incidence.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ph18030333/s1, Table S1: Definition of osteoporosis; Table S2: Definition of hyperthyroidism; Table S3: Definition of fractures.
Author Contributions
Conceptualization, T.K.; Data curation, H.H.; Formal analysis, T.K.; Methodology, T.K. and M.H.; Writing—original draft, T.K.; Writing—review and editing, H.H., M.H., T.T., R.I., T.N., Y.Z. and S.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Because VigiBase contains anonymized data, ethics committee approval was not required for this study.
Informed Consent Statement
No applicable.
Data Availability Statement
Data are available upon reasonable request.
Acknowledgments
The study results and conclusions do not represent the opinions of the Uppsala Monitoring Centre, National Centers, or the WHO.
Conflicts of Interest
The authors report no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ADRs | Adverse drug reactions |
| ICIs | Immune checkpoint inhibitors |
| BCPNN | Bayesian Confidence Propagation Neural Network |
| WHO | World Health Organization |
| HLTs | High-level terms |
| SMQs | Standardized MedDRA Queries |
| TSH | Thyroid-stimulating hormone |
| MedDRA | Medical Dictionary for Regulatory Activities |
| HLGT | High-level group term |
| FAERS | FDA Adverse Event Reporting System |
| CIs | Confidence intervals |
| ICs | Information components |
| TNF-α | Tumor necrosis factor-alpha |
| IL | Interleukin |
| RANKL | Receptor activator of nuclear factor-κB ligand |
| irAEs | Immune-related adverse events |
| CTLA-4 | Cytotoxic T-lymphocyte-associated antigen 4 |
| PD-L1 | Programmed death-ligand 1 |
| PD-1 | Programmed cell death protein 1 |
References
- Martins, F.; Sofiya, L.; Sykiotis, G.P.; Lamine, F.; Maillard, M.; Fraga, M.; Shabafrouz, K.; Ribi, C.; Cairoli, A.; Guex-Crosier, Y.; et al. Adverse effects of immune-checkpoint inhibitors: Epidemiology, management and surveillance. Nat. Rev. Clin. Oncol. 2019, 16, 563–580. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, M.; Ye, C. Osteoporotic fractures: An unrecognized adverse event of immune checkpoint inhibitors? J. Immunother. Cancer 2024, 12, e009309. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Li, F.; Li, X.; Wang, Z.G.; Zhang, B. TNF-α and RANKL promote osteoclastogenesis by upregulating RANK via the NF-κB pathway. Mol. Med. Rep. 2018, 17, 6605–6611. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Matsuyama, T.; Hosokawa, Y.; Makihira, S.; Seki, M.; Karimbux, N.Y.; Goncalves, R.B.; Valverde, P.; Dibart, S.; Li, Y.P.; et al. B and T lymphocytes are the primary sources of RANKL in the bone resorptive lesion of periodontal disease. Am. J. Pathol. 2006, 169, 987–998. [Google Scholar] [CrossRef]
- Yao, Z.; Li, P.; Zhang, Q.; Schwarz, E.M.; Keng, P.; Arbini, A.; Boyce, B.F.; Xing, L. Tumor necrosis factor-alpha increases circulating osteoclast precursor numbers by promoting their proliferation and differentiation in the bone marrow through up-regulation of c-Fms expression. J. Biol. Chem. 2006, 281, 11846–11855. [Google Scholar] [CrossRef]
- Joseph, G.J.; Johnson, D.B.; Johnson, R.W. Immune checkpoint inhibitors in bone metastasis: Clinical challenges, toxicities, and mechanisms. J. Bone Oncol. 2023, 43, 100505. [Google Scholar] [CrossRef]
- Yang, N.; Liu, Y. The Role of the Immune Microenvironment in Bone Regeneration. Int. J. Med. Sci. 2021, 18, 3697–3707. [Google Scholar] [CrossRef]
- Huang, R.; Wang, X.; Zhou, Y.; Xiao, Y. RANKL-induced M1 macrophages are involved in bone formation. Bone Res. 2017, 5, 17019. [Google Scholar] [CrossRef]
- Canton, M.; Sánchez-Rodríguez, R.; Spera, I.; Venegas, F.C.; Favia, M.; Viola, A.; Castegna, A. Reactive Oxygen Species in Macrophages: Sources and Targets. Front. Immunol. 2021, 12, 734229. [Google Scholar] [CrossRef]
- Sun, Y.; Li, J.; Xie, X.; Gu, F.; Sui, Z.; Zhang, K.; Yu, T. Macrophage-Osteoclast Associations: Origin, Polarization, and Subgroups. Front. Immunol. 2021, 12, 778078. [Google Scholar] [CrossRef]
- Kirkwood, K.L.; Zhang, L.; Thiyagarajan, R.; Seldeen, K.L.; Troen, B.R. Myeloid-Derived Suppressor Cells at the Intersection of Inflammaging and Bone Fragility. Immunol. Investig. 2018, 47, 844–854. [Google Scholar] [CrossRef] [PubMed]
- Sawant, A.; Ponnazhagan, S. Myeloid-derived suppressor cells as osteoclast progenitors: A novel target for controlling osteolytic bone metastasis. Cancer Res. 2013, 73, 4606–4610. [Google Scholar] [CrossRef] [PubMed]
- Danilin, S.; Merkel, A.R.; Johnson, J.R.; Johnson, R.W.; Edwards, J.R.; Sterling, J.A. Myeloid-derived suppressor cells expand during breast cancer progression and promote tumor-induced bone destruction. Oncoimmunology 2012, 1, 1484–1494. [Google Scholar] [CrossRef] [PubMed]
- Moseley, K.F.; Naidoo, J.; Bingham, C.O.; Carducci, M.A.; Forde, P.M.; Gibney, G.T.; Lipson, E.J.; Shah, A.A.; Sharfman, W.H.; Cappelli, L.C. Immune-related adverse events with immune checkpoint inhibitors affecting the skeleton: A seminal case series. J. Immunother. Cancer 2018, 6, 104. [Google Scholar] [CrossRef]
- Filippini, D.M.; Gatti, M.; Di Martino, V.; Cavalieri, S.; Fusaroli, M.; Ardizzoni, A.; Raschi, E.; Licitra, L. Bone fracture as a novel immune-related adverse event with immune checkpoint inhibitors: Case series and large-scale pharmacovigilance analysis. Int. J. Cancer 2021, 149, 675–683. [Google Scholar] [CrossRef]
- Ye, C.; Lee, K.; Leslie, W.D.; Lin, M.; Walker, J.; Kolinsky, M. Fracture rate increases after immune checkpoint inhibitor treatment: A potential new immune related adverse event. Osteoporos. Int. 2023, 34, 735–740. [Google Scholar] [CrossRef]
- Dökmetaş, M.; Muğlu, H.; Özcan, E.; Bayram Kuvvet, B.; Helvacı, K.; Kalacı, E.; Kahraman, S.; Aykan, M.B.; Çiçin, İ.; Selçukbiricik, F.; et al. Endocrine Adverse Events in Patients Treated with Immune Checkpoint Inhibitors: A Comprehensive Analysis. Medicina 2025, 61, 123. [Google Scholar] [CrossRef]
- Yamauchi, I.; Yabe, D. Best practices in the management of thyroid dysfunction induced by immune checkpoint inhibitors. Eur. Thyroid J. 2025, 14, e240328. [Google Scholar] [CrossRef]
- Elshafie, O.; Khalil, A.B.; Salman, B.; Atabani, A.; Al-Sayegh, H. Immune Checkpoint Inhibitors-Induced Endocrinopathies: Assessment, Management and Monitoring in a Comprehensive Cancer Centre. Endocrinol. Diabetes Metab. 2024, 7, e00505. [Google Scholar] [CrossRef]
- Lee, S.Y.; Pearce, E.N. Hyperthyroidism: A Review. JAMA 2023, 330, 1472–1483. [Google Scholar] [CrossRef]
- Nicholls, J.J.; Brassill, M.J.; Williams, G.R.; Bassett, J.H. The skeletal consequences of thyrotoxicosis. J. Endocrinol. 2012, 213, 209–221. [Google Scholar] [CrossRef] [PubMed]
- González-Rodríguez, E.; Rodríguez-Abreu, D. Immune Checkpoint Inhibitors: Review and Management of Endocrine Adverse Events. Oncologist 2016, 21, 804–816. [Google Scholar] [CrossRef] [PubMed]
- Stelmachowska-Banaś, M.; Czajka-Oraniec, I. Management of endocrine immune-related adverse events of immune checkpoint inhibitors: An updated review. Endocr. Connect. 2020, 9, R207–R228. [Google Scholar] [CrossRef]
- Patadia, V.K.; Coloma, P.; Schuemie, M.J.; Herings, R.; Gini, R.; Mazzaglia, G.; Picelli, G.; Fornari, C.; Pedersen, L.; van der Lei, J.; et al. Using real-world healthcare data for pharmacovigilance signal detection—The experience of the EU-ADR project. Expert. Rev. Clin. Pharmacol. 2015, 8, 95–102. [Google Scholar] [CrossRef]
- Raschi, E.; Gatti, M.; Gelsomino, F.; Ardizzoni, A.; Poluzzi, E.; De Ponti, F. Lessons to be Learnt from Real-World Studies on Immune-Related Adverse Events with Checkpoint Inhibitors: A Clinical Perspective from Pharmacovigilance. Target. Oncol. 2020, 15, 449–466. [Google Scholar] [CrossRef]
- Noguchi, Y.; Tachi, T.; Teramachi, H. Detection algorithms and attentive points of safety signal using spontaneous reporting systems as a clinical data source. Brief Bioinform. 2021, 22, bbab347. [Google Scholar] [CrossRef]
- Montastruc, J.L.; Lafaurie, M.; de Canecaude, C.; Durrieu, G.; Sommet, A.; Montastruc, F.; Bagheri, H. Fatal adverse drug reactions: A worldwide perspective in the World Health Organization pharmacovigilance database. Br. J. Clin. Pharmacol. 2021, 87, 4334–4340. [Google Scholar] [CrossRef]
- van Puijenbroek, E.P.; Bate, A.; Leufkens, H.G.; Lindquist, M.; Orre, R.; Egberts, A.C. A comparison of measures of disproportionality for signal detection in spontaneous reporting systems for adverse drug reactions. Pharmacoepidemiol. Drug Saf. 2002, 11, 3–10. [Google Scholar] [CrossRef]
- Bate, A.; Lindquist, M.; Edwards, I.R.; Olsson, S.; Orre, R.; Lansner, A.; De Freitas, R.M. A Bayesian neural network method for adverse drug reaction signal generation. Eur. J. Clin. Pharmacol. 1998, 54, 315–321. [Google Scholar] [CrossRef]
- Lindquist, M.; Ståhl, M.; Bate, A.; Edwards, I.R.; Meyboom, R.H. A retrospective evaluation of a data mining approach to aid finding new adverse drug reaction signals in the WHO international database. Drug Saf. 2000, 23, 533–542. [Google Scholar] [CrossRef]
- Liu, H.; Li, Y.; Li, J.; Zhang, Q.; Wu, J.; Li, X.; Meng, L.; Cao, S.; Li, H. Musculoskeletal adverse events induced by immune checkpoint inhibitors: A large-scale pharmacovigilance study. Front. Pharmacol. 2023, 14, 1199031. [Google Scholar] [CrossRef] [PubMed]
- Blum, M.R.; Bauer, D.C.; Collet, T.H.; Fink, H.A.; Cappola, A.R.; da Costa, B.R.; Wirth, C.D.; Peeters, R.P.; Åsvold, B.O.; den Elzen, W.P.; et al. Subclinical thyroid dysfunction and fracture risk: A meta-analysis. JAMA 2015, 313, 2055–2065. [Google Scholar] [CrossRef] [PubMed]
- Abe, E.; Marians, R.C.; Yu, W.; Wu, X.B.; Ando, T.; Li, Y.; Iqbal, J.; Eldeiry, L.; Rajendren, G.; Blair, H.C.; et al. TSH is a negative regulator of skeletal remodeling. Cell 2003, 115, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Leng, P.; Qiu, Y.; Zhou, M.; Zhu, Y.; Yin, N.; Zhou, M.; Wu, W.; Liu, M. Hypothyroidism correlates with osteoporosis: Potential involvement of lipid mediators. Front. Med. 2024, 11, 1453502. [Google Scholar] [CrossRef]
- Cabral, H.W.; Andolphi, B.F.; Ferreira, B.V.; Alves, D.C.; Morelato, R.L.; Chambo, A.F.; Borges, L.S. The use of biomarkers in clinical osteoporosis. Rev. Assoc. Med. Bras. 2016, 62, 368–376. [Google Scholar] [CrossRef]
- Messina, C.; Fusco, S.; Gazzotti, S.; Albano, D.; Bonaccorsi, G.; Guglielmi, G.; Bazzocchi, A. DXA beyond bone mineral density and the REMS technique: New insights for current radiologists practice. Radiol. Med. 2024, 129, 1224–1240. [Google Scholar] [CrossRef]
- Weinstein, L.; Radano, T.A.; Jack, T.; Kalina, P.; Eberhardt, J.S., 3rd. Application of multivariate probabilistic (Bayesian) networks to substance use disorder risk stratification and cost estimation. Perspect. Health Inf. Manag. 2009, 6, 1b. [Google Scholar]
- Bate, A.; Evans, S.J. Quantitative signal detection using spontaneous ADR reporting. Pharmacoepidemiol. Drug Saf. 2009, 18, 427–436. [Google Scholar] [CrossRef]
- Tanaka, J.; Koseki, T.; Kondo, M.; Ito, Y.; Yamada, S. Analyses of Ocular Adverse Reactions Associated With Anticancer Drugs Based on the Japanese Pharmacovigilance Database. Anticancer Res. 2022, 42, 4439–4451. [Google Scholar] [CrossRef]
- Noguchi, Y.; Murayama, A.; Esaki, H.; Sugioka, M.; Koyama, A.; Tachi, T.; Teramachi, H. Angioedema Caused by Drugs That Prevent the Degradation of Vasoactive Peptides: A Pharmacovigilance Database Study. J. Clin. Med. 2021, 10, 5507. [Google Scholar] [CrossRef]
- Kanda, Y. Investigation of the freely available easy-to-use software ’EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).