Polyhexamethylene Biguanide Reduces High-Risk Human Papilloma Virus Viral Load in Cervical Cell Samples Derived from ThinPrep Pap Test
Abstract
:1. Introduction
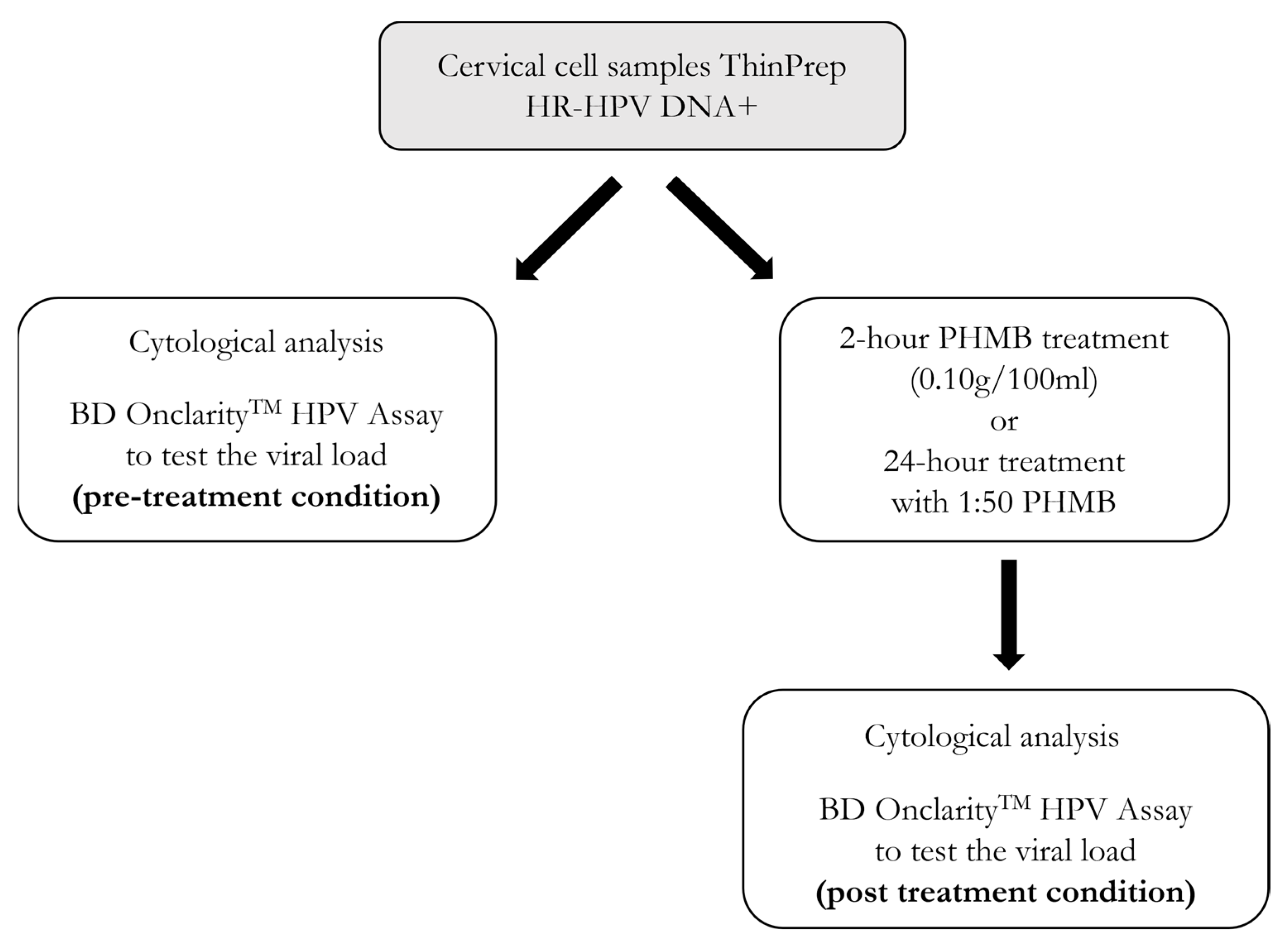
2. Materials and Methods
2.1. The ThinPrep® Pap Test
2.2. PHMB Solution and Treatment
2.3. BD OnclarityTM HPV Assay
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Collected Samples
3.2. HR-HPV Prevalence and Genotype Distribution
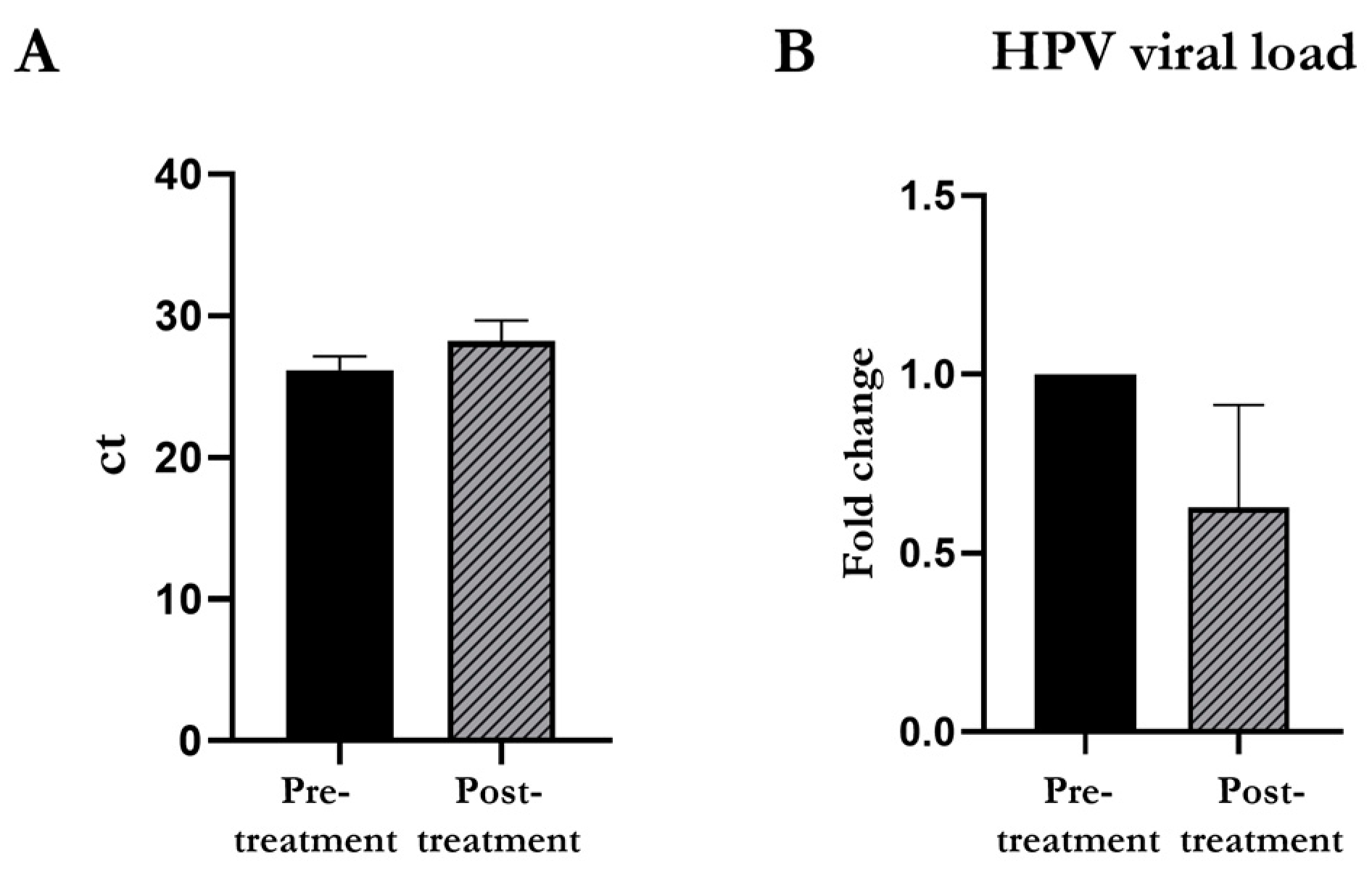
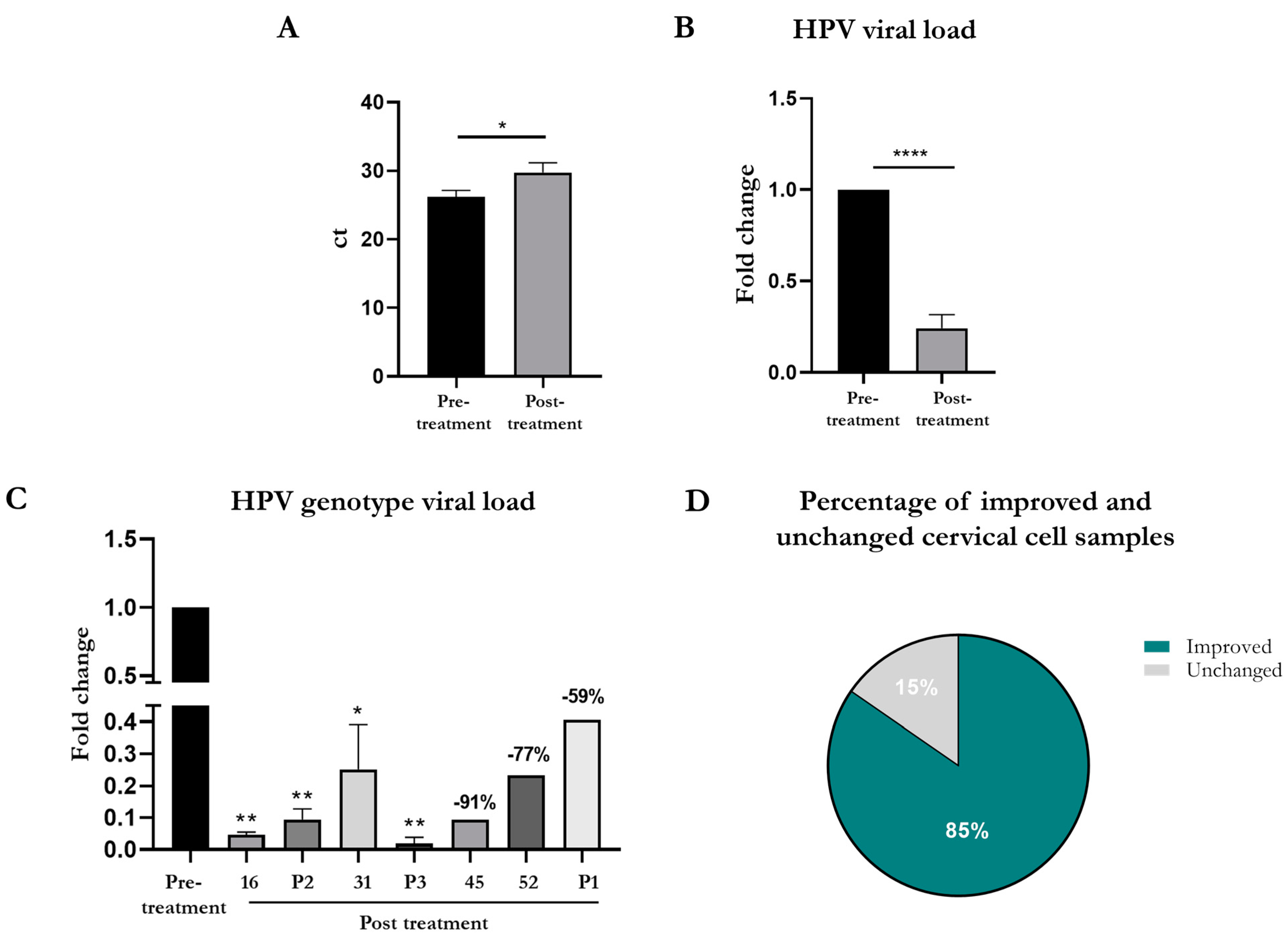
3.3. HPV Viral Load Detected through the BD OnclarityTM HPV Assay
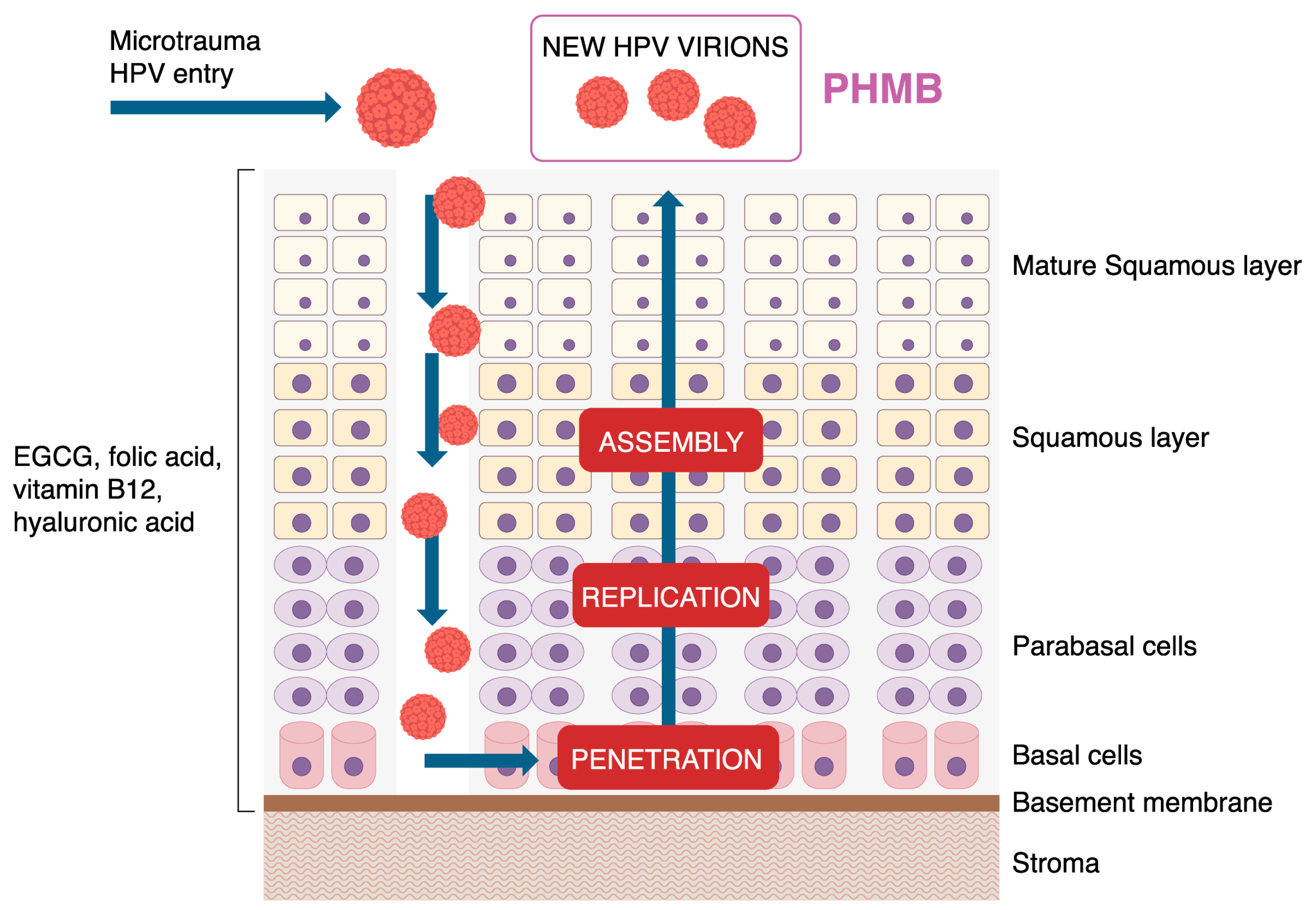
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Liverani, C.A. The four steps in the prevention of human papillomavirus-associated neoplasia: Considerations for preventive measures, screening, disease impact, and potential overtreatments in HPV-related pathology. Arch. Gynecol. Obstet. 2013, 288, 979–988. [Google Scholar] [CrossRef]
- Fehrmann, F.; Laimins, L.A. Human papillomaviruses: Targeting differentiating epithelial cells for malignant transformation. Oncogene 2003, 22, 5201–5207. [Google Scholar] [CrossRef]
- Egawa, N.; Doorbar, J. The low-risk papillomaviruses. Virus Res. 2017, 231, 119–127. [Google Scholar] [CrossRef]
- Ma, X.; Yang, M. The correlation between high-risk HPV infection and precancerous lesions and cervical cancer. Am. J. Transl. Res. 2021, 13, 10830–10836. [Google Scholar]
- Bruni, L.A.G.; Serrano, B.; Mena, M.; Collado, J.J.; Gómez, D.; Muñoz, J.; Bosch, F.X.; de Sanjosé, S. Human Papillomavirus and Related Diseases in the World. 2023. Available online: https://hpvcentre.net/statistics/reports/USA.pdf (accessed on 5 May 2024).
- Nayar, R.; Wilbur, D.C. The Pap test and Bethesda 2014. Cancer Cytopathol. 2015, 123, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Perkins, R.B.; Guido, R.S.; Castle, P.E.; Chelmow, D.; Einstein, M.H.; Garcia, F.; Huh, W.K.; Kim, J.J.; Moscicki, A.B.; Nayar, R.; et al. 2019 ASCCP Risk-Based Management Consensus Guidelines for Abnormal Cervical Cancer Screening Tests and Cancer Precursors. J. Low. Genit. Tract Dis. 2020, 24, 102–131. [Google Scholar] [CrossRef] [PubMed]
- Zanotti, K.M.; Belinson, J. Update on the diagnosis and treatment of human papillomavirus infection. Cleve Clin. J. Med. 2002, 69, 948–963. [Google Scholar] [CrossRef]
- Xie, J.; Wang, S.; Li, Z.; Ao, C.; Wang, J.; Wang, L.; Peng, X.; Zeng, K. 5-aminolevulinic acid photodynamic therapy reduces HPV viral load via autophagy and apoptosis by modulating Ras/Raf/MEK/ERK and PI3K/AKT pathways in HeLa cells. J. Photochem. Photobiol. B 2019, 194, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Kore, V.B.; Anjankar, A. A Comprehensive Review of Treatment Approaches for Cutaneous and Genital Warts. Cureus 2023, 15, e47685. [Google Scholar] [CrossRef]
- Claesson, U.; Lassus, A.; Happonen, H.; Hogstrom, L.; Siboulet, A. Topical treatment of venereal warts: A comparative open study of podophyllotoxin cream versus solution. Int. J. STD AIDS 1996, 7, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Jiang, Y.; Zhao, R. Advances in ablative treatment for human papillomavirus related cervical pre-cancer lesions. Gynecol. Obstet. Clin. Med. 2023, 3, 213–219. [Google Scholar] [CrossRef]
- Hübner, N.O.; Kramer, A. Review on the efficacy, safety and clinical applications of polihexanide, a modern wound antiseptic. Skin. Pharmacol. Physiol. 2010, 23 (Suppl. S1), 17–27. [Google Scholar] [CrossRef] [PubMed]
- Niro, A.; Pignatelli, F.; Fallico, M.; Sborgia, A.; Passidomo, F.; Gigliola, S.; Nacucchi, A.; Sborgia, G.; Boscia, G.; Alessio, G.; et al. Polyhexamethylene biguanide hydrochloride (PHMB)-properties and application of an antiseptic agent. A narrative review. Eur. J. Ophthalmol. 2022, 33, 655–666. [Google Scholar] [CrossRef] [PubMed]
- Chindera, K.; Mahato, M.; Sharma, A.K.; Horsley, H.; Kloc-Muniak, K.; Kamaruzzaman, N.F.; Kumar, S.; McFarlane, A.; Stach, J.; Bentin, T.; et al. The antimicrobial polymer PHMB enters cells and selectively condenses bacterial chromosomes. Sci. Rep. 2016, 6, 23121. [Google Scholar] [CrossRef] [PubMed]
- Rippon, M.G.; Rogers, A.A.; Ousey, K. Polyhexamethylene biguanide and its antimicrobial role in wound healing: A narrative review. J. Wound Care 2023, 32, 5–20. [Google Scholar] [CrossRef] [PubMed]
- Minozzi, M.; Gerli, S.; Di Renzo, G.C.; Papaleo, E.; Ferrari, A. The efficacy and safety of a single dose of polyhexamethylene biguanide gynaecologic solution versus a seven-dose regimen of vaginal clindamycin cream in patients with bacterial vaginosis. Eur. Rev. Med. Pharmacol. Sci. 2008, 12, 59–65. [Google Scholar] [PubMed]
- Punjataewakupt, A.; Napavichayanun, S.; Aramwit, P. The downside of antimicrobial agents for wound healing. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 39–54. [Google Scholar] [CrossRef]
- Babalska, Z.L.; Korbecka-Paczkowska, M.; Karpinski, T.M. Wound Antiseptics and European Guidelines for Antiseptic Application in Wound Treatment. Pharmaceuticals 2021, 14, 1253. [Google Scholar] [CrossRef]
- Krebs, F.C.; Miller, S.R.; Ferguson, M.L.; Labib, M.; Rando, R.F.; Wigdahl, B. Polybiguanides, particularly polyethylene hexamethylene biguanide, have activity against human immunodeficiency virus type 1. Biomed. Pharmacother. 2005, 59, 438–445. [Google Scholar] [CrossRef]
- Valluri, S.; Fleming, T.P.; Laycock, K.A.; Tarle, I.S.; Goldberg, M.A.; Garcia-Ferrer, F.J.; Essary, L.R.; Pepose, J.S. In vitro and in vivo effects of polyhexamethylene biguanide against herpes simplex virus infection. Cornea 1997, 16, 556–559. [Google Scholar] [CrossRef] [PubMed]
- Pinto, F. Mechanisms of Action of Polyhexamethylene Biguanide-Based Biocides against Non-Enveloped Virus. Ph.D. Thesis, Cardiff University, Cardiff, UK, 2010. [Google Scholar]
- Gentile, A.; Gerli, S.; Di Renzo, G.C. A new non-invasive approach based on polyhexamethylene biguanide increases the regression rate of HPV infection. BMC Clin. Pathol. 2012, 12, 17. [Google Scholar] [CrossRef]
- Marelli, G.; Papaleo, E.; Origoni, M.; Caputo, L.; Ferrari, A. Polyhexamethylene biguanide for treatment of external genital warts: A prospective, double-blind, randomized study. Eur. Rev. Med. Pharmacol. Sci. 2005, 9, 369–372. [Google Scholar]
- Solomon, D.; Davey, D.; Kurman, R.; Moriarty, A.; O’Connor, D.; Prey, M.; Raab, S.; Sherman, M.; Wilbur, D.; Wright, T., Jr.; et al. The 2001 Bethesda System: Terminology for reporting results of cervical cytology. JAMA 2002, 287, 2114–2119. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, M.; Giubbi, C.; Sechi, I.; Bottari, F.; Iacobone, A.D.; Musumeci, R.; Perdoni, F.; Muresu, N.; Piana, A.; Fruscio, R.; et al. Evaluation of BD Onclarity™ HPV Assay on Self-Collected Vaginal and First-Void Urine Samples as Compared to Clinician-Collected Cervical Samples: A Pilot Study. Diagnostics 2022, 12, 3075. [Google Scholar] [CrossRef] [PubMed]
- Ejegod, D.M.; Junge, J.; Franzmann, M.; Kirschner, B.; Bottari, F.; Sideri, M.; Sandri, M.T.; Bonde, J. Clinical and analytical performance of the BD Onclarity™ HPV assay for detection of CIN2+ lesions on SurePath samples. Papillomavirus Res. 2016, 2, 31–37. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Chiantera, V.; Gerli, S.; Proietti, S.; Lepore, E.; Unfer, V.; Carugno, J.; Favilli, A. Preventing Persistence of HPV Infection with Natural Molecules. Pathogens 2023, 12, 416. [Google Scholar] [CrossRef] [PubMed]
- Muller, G.; Kramer, A. Biocompatibility index of antiseptic agents by parallel assessment of antimicrobial activity and cellular cytotoxicity. J. Antimicrob. Chemother. 2008, 61, 1281–1287. [Google Scholar] [CrossRef]
- Kramer, A.; Eberlein, T.; Muller, G.; Dissemond, J.; Assadian, O. Re-evaluation of polihexanide use in wound antisepsis in order to clarify ambiguities of two animal studies. J. Wound Care 2019, 28, 246–255. [Google Scholar] [CrossRef]
- Leslie, S.W.; Sajjad, H.; Kumar, S. Genital Warts; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Grandi, G.; Botticelli, L.; Fraia, P.D.; Babalini, C.; Masini, M.; Unfer, V. The Association of Four Natural Molecules-EGCG, Folic Acid, Vitamin B12, and HA-To Counteract HPV Cervical Lesions: A Case Report. J. Pers. Med. 2023, 13, 567. [Google Scholar] [CrossRef] [PubMed]
- Aragona, C.; Bezerra Espinola, M.S.; Bilotta, G.; Porcaro, G.; Calcagno, M. Evaluating the Efficacy of Pervistop((R)), a New Combination Based on EGCG, Folic Acid, Vitamin B12 and Hyaluronic Acid on Patients with Human Papilloma Virus (HPV) Persistent Infections and Cervical Lesions: A Pilot Study. J. Clin. Med. 2023, 12, 2171. [Google Scholar] [CrossRef] [PubMed]
| ThinPrep PAP Test Cytological Results | No. Samples = 13 | % |
|---|---|---|
| LSIL | 8 | 61.5% |
| ASCUS | 4 | 30.8% |
| NILM (BV) | 1 | 7.7% |
| HR-HPV Genotype | No. Samples = 13 | % |
|---|---|---|
| HPV 16 | 2 | 15.4% |
| HPV 31 | 4 | 30.8% |
| HPV 45 | 1 | 7.7% |
| HPV 52 | 1 | 7.7% |
| Multiple HPV infections | 10 | 76.9% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Fraia, L.; Babalini, C.; Calcagno, M.; Proietti, S.; Lepore, E.; Di Fraia, P. Polyhexamethylene Biguanide Reduces High-Risk Human Papilloma Virus Viral Load in Cervical Cell Samples Derived from ThinPrep Pap Test. Curr. Issues Mol. Biol. 2024, 46, 4874-4884. https://doi.org/10.3390/cimb46050293
Di Fraia L, Babalini C, Calcagno M, Proietti S, Lepore E, Di Fraia P. Polyhexamethylene Biguanide Reduces High-Risk Human Papilloma Virus Viral Load in Cervical Cell Samples Derived from ThinPrep Pap Test. Current Issues in Molecular Biology. 2024; 46(5):4874-4884. https://doi.org/10.3390/cimb46050293
Chicago/Turabian StyleDi Fraia, Ludovica, Carla Babalini, Marco Calcagno, Sara Proietti, Elisa Lepore, and Pietro Di Fraia. 2024. "Polyhexamethylene Biguanide Reduces High-Risk Human Papilloma Virus Viral Load in Cervical Cell Samples Derived from ThinPrep Pap Test" Current Issues in Molecular Biology 46, no. 5: 4874-4884. https://doi.org/10.3390/cimb46050293


_Kim.png)



