The Effect of Muscle Exercise on Perforators Flow: A Prospective Cohort Study
Abstract
:1. Introduction
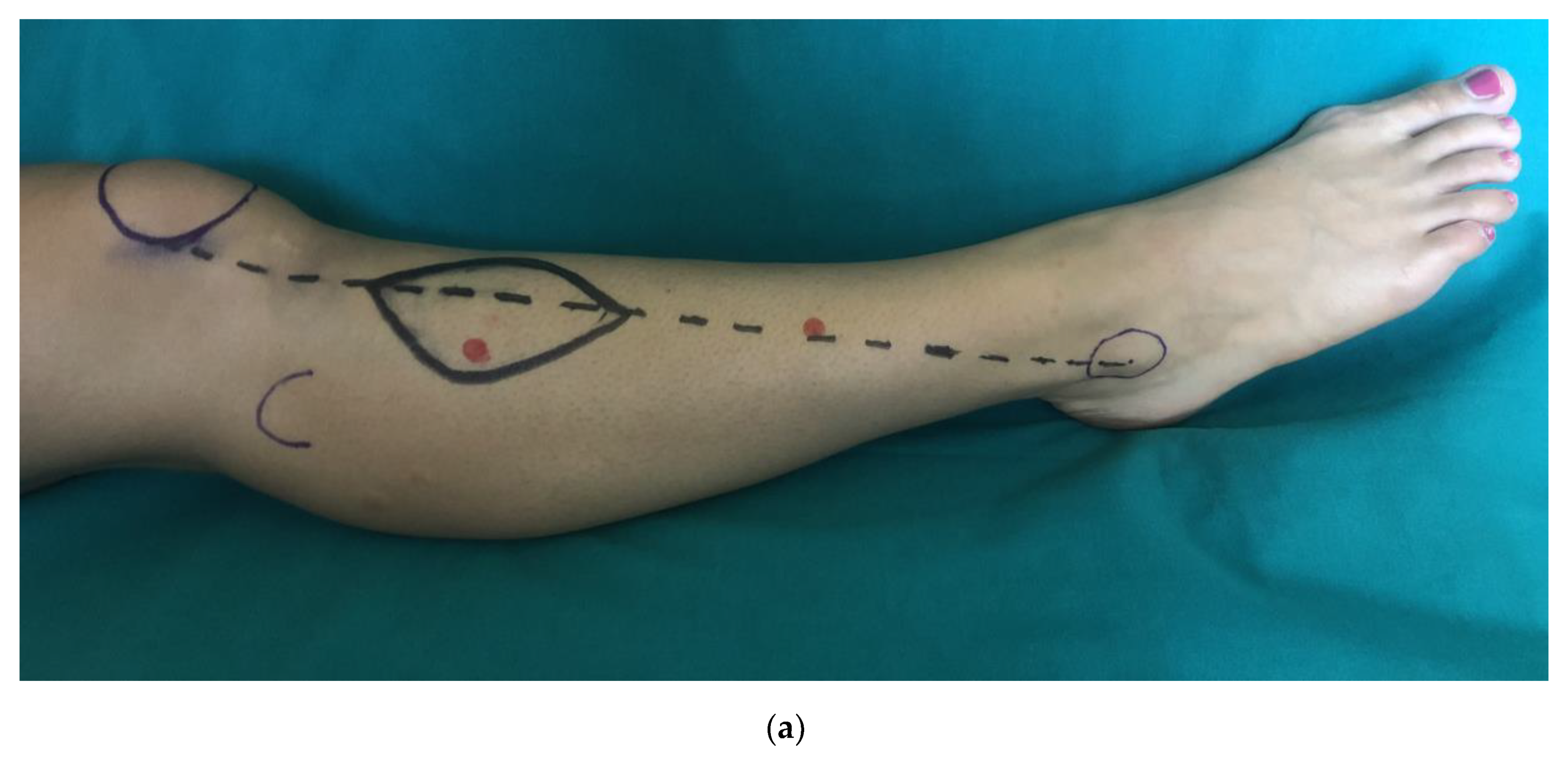
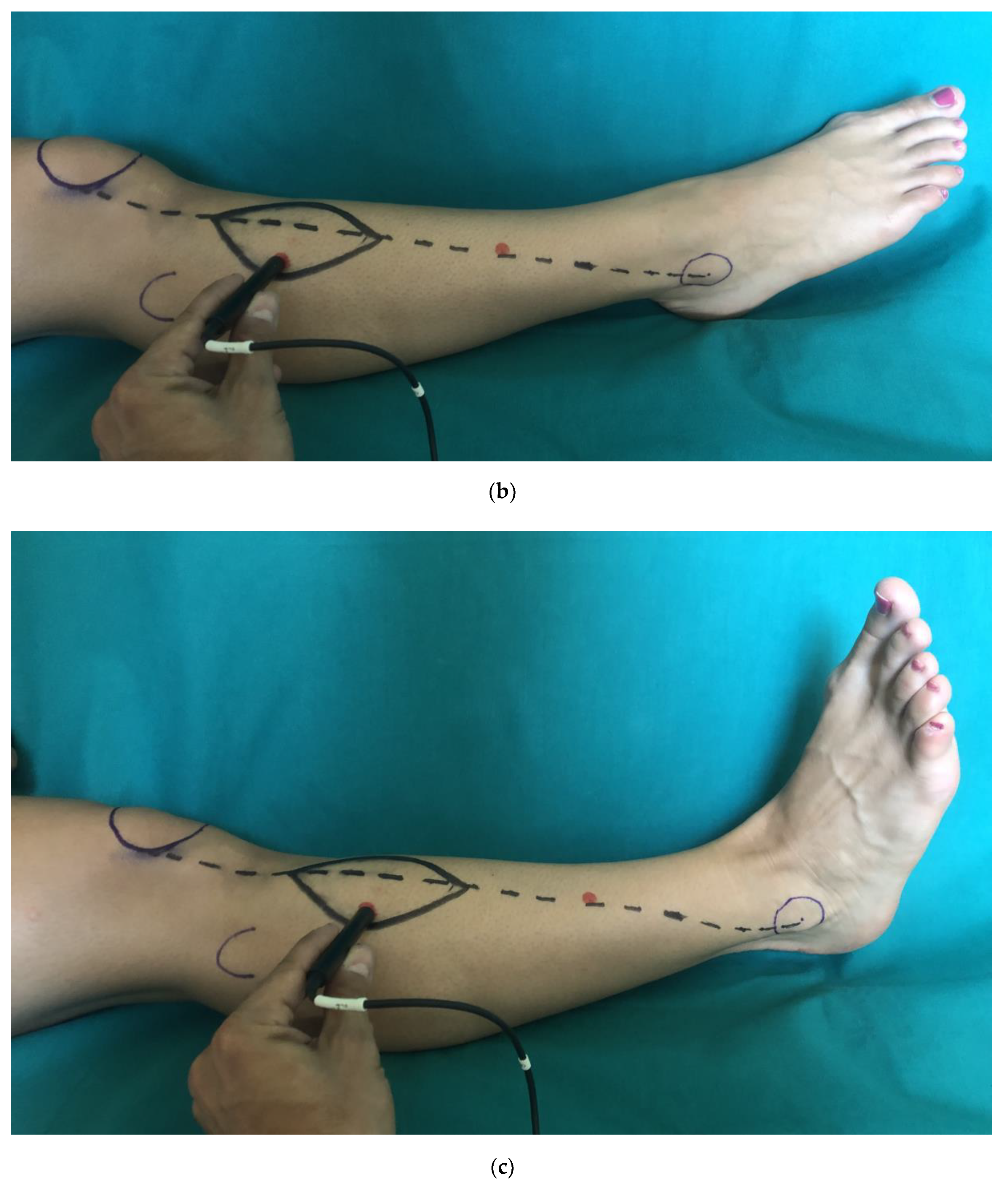
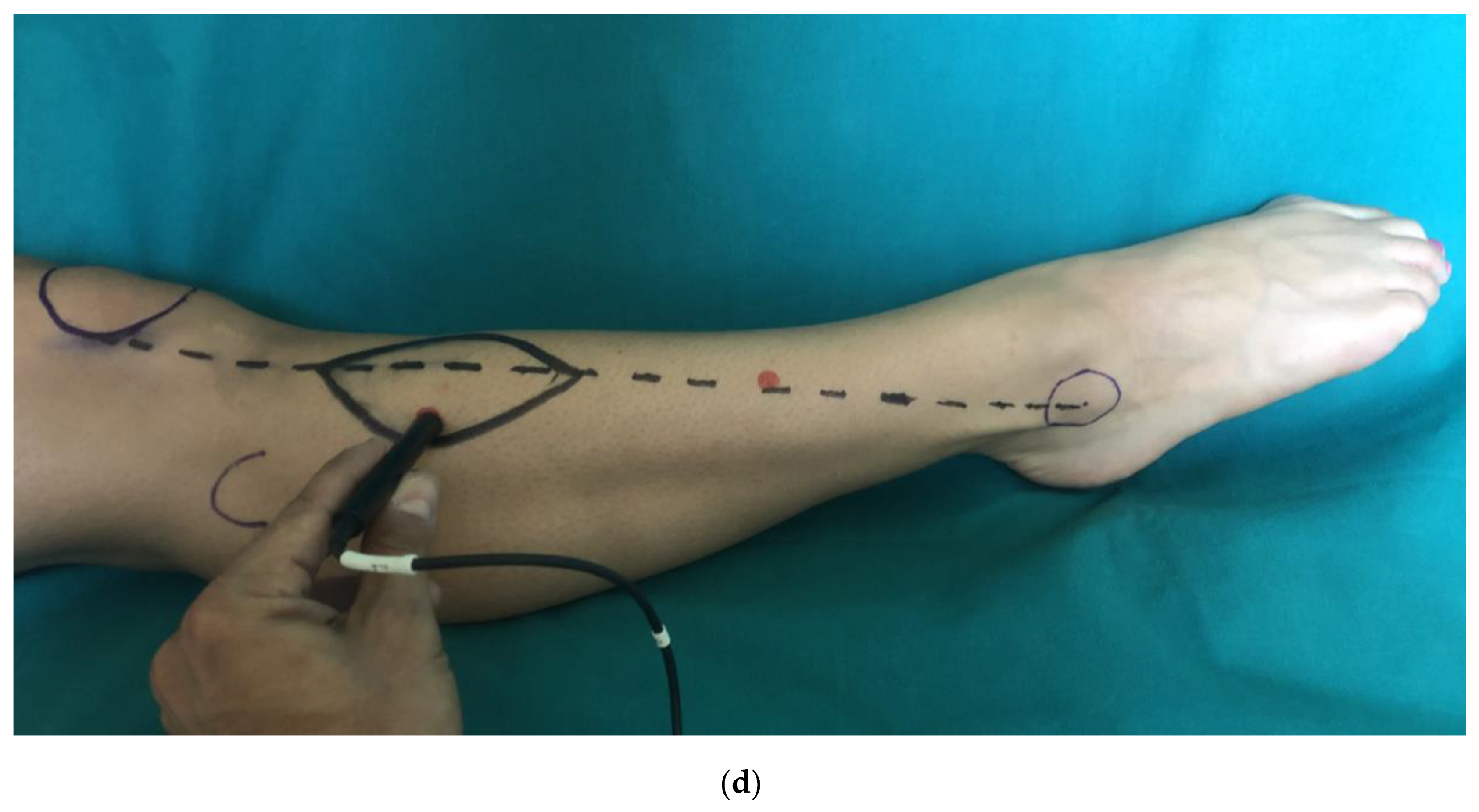
2. Materials and Methods
2.1. Clinical Examination
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Soltanian, H.; Garcia, R.M.; Hollenbeck, S.T. Current concepts in lower extremities reconstruction. Plast. Reconstr. Surg. 2015, 136, 815e–829e. [Google Scholar] [CrossRef]
- Bekara, F.; Herlin, C.; Somda, S.; de Runz, A.; Grolleau, J.L.; Chaput, B. Free versus perforator-pedicled propeller flaps in lower extremity reconstruction: What is the safest coverage? A meta analysis. Microsurgery 2018, 38, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Engel, H.; Lin, C.-H.; Wei, F.-C. Role of microsurgery in lower extrimities reconstruction. Plast. Reconstr. Surg. 2011, 127, 228S–238S. [Google Scholar] [CrossRef] [PubMed]
- Bekara, F.; Herlin, C.; Mojallal, A.; Sinna, R.; Ayestaray, B.; Letois, F.; Chavoin, J.P.; Garrido, I.; Grolleau, J.L.; Chaput, B. A systematic review and meta-analysis of perforator-pedicled propeller flaps in lower extremitiy defects. Plast. Reconstr. Surg. 2016, 137, 314–331. [Google Scholar] [CrossRef] [PubMed]
- Blondeel, P.N.; Van Landuyt, K.H.I.; Monstrey, S.J.M.; Hamdi, M.; Matton, G.E.; Allen, R.J.; Dupin, C.; Feller, A.; Koshima, I.; Kostakoglu, N.; et al. The “Gent” Consensus on perforator flap terminology: Preliminary definitions. Plast. Reconstr. Surg. 2003, 112, 1378–1383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sinna, R.; Boloorchi, A.; Mahajan, A.L.; Qassemyar, Q.; Robbe, M. What should define a “perforator flap”? Plast. Reconstr. Surg. 2010, 126, 2258–2263. [Google Scholar] [CrossRef] [PubMed]
- Tapadar, A.; Palit, S.; Kundu, R.; Sarkar, A.; Banetjee, A. A study of the perforating arteries of the leg derived from the anterior tibial, posterior tibial and peroneal arteries. J. Anat. Soc. India 2014, 63, 43–47. [Google Scholar] [CrossRef]
- Vaienti, L.; Cottone, G.; De Francesco, F.; Borelli, F.; Zaccaria, G.; Amendola, F. The suprafascial course of the lower leg perforators: An anatomical study. Arch. Plast. Surg. 2020, 47, 165–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morrison, W.A.; Shen, T.Y. Anterior tibial artery flap: Anatomy and case report. Br. J. Plast. Surg. 1987, 40, 230–235. [Google Scholar] [CrossRef]
- Vinet, A.; Karpoff, L.; Walther, G.; Startun, A.; Obert, P.; Goret, L.; Dauzat, M.; Perez-Martin, A. Vascular reactivity at rest and during excercise in middle-aged obese men: Effects of short-term, low-intensity, exercise training. Int. J. Obes. (Lond.) 2011, 35, 820–828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hearon, C.M.; Dinenno, F.A. Regulation of skeletal muscle blood flow during exercise in ageing humans. J. Physiol. 2016, 594, 2261–2273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clifford, P.S.; Hellsten, Y. Vasodilatatory mechanism in contracting skeletal muscle. J. Appl. Physiol. 2004, 97, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Riccio, M.; Zingaretti, N.; Verdini, F.; Marchesini, A.; de Francesco, F.; Parodi, P.C. Functional donor-site morbidity after soleus muscle-flap procedure in the treatment of the lower limb severe injuries. Handchir. Mikrochir. Plast. Chir. 2019, 51, 453–463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olivier, J.D.; Deloughery, E.P.; Gupta, N.; Boczar, D.; Sisti, A.; Huayllani, M.T.; Restrepo, D.J.; Hu, M.S.; Forte, A.J. Local pro- and anti-coagulation therapy in the plastic surgical patient: A literature review of the evidence and clinical applications. Medicina 2019, 55, 208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, J.E. Guyton and Hall Textbook of Medical Physiology, 13th ed.; Saunders: Philadelphia, PA, USA, 2015. [Google Scholar]
- Gifford, J.R.; Richardson, R.S. CORP: Ultrasound assessment of vascular function with the passive leg movement technique. J. Appl. Physiol. 2017, 123, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Trinity, J.D.; Richardson, R.S. Physiological impact and clinical relevance of passive exercise/movement. Sport Med. 2019, 49, 1365–1381. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Min, P.; Grassetti, L.; Lazzeri, D.; Sadigh, P.; Nicoli, F.; Torresetti, M.; Gao, W.; di Benedetto, G.; Zhang, W.; et al. A prospective head-to-head comparison of color doppler ultrasound and computed tomographic angiography in the preoperative planning of lower extremity perforator flaps. Plast. Reconstr. Surg. 2016, 137, 335–347. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Jeong, H.H.; Lee, Y.; Powers, H.; Suh, Y.C.; Christoffi, P.; Suh, H.P.; Hong, J.P. Direction of flap rotation in propeller flaps: Does it really matter? J. Reconstr. Microsurg. 2019, 35, 549–556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gliemann, L.; Vestergaard Hansen, C.; Rytter, N.; Hellsten, Y. Regulation of skeletal muscle blood flow during exercise. Curr. Opin. Physiol. 2019, 10, 146–155. [Google Scholar] [CrossRef]
| Patient | Sex | Age (y) | Physical Activity | Smoking |
|---|---|---|---|---|
| 1 | M | 28 | low | no |
| 2 | F | 27 | high | yes |
| 3 | M | 35 | low | no |
| 4 | M | 30 | high | no |
| 5 | M | 27 | high | no |
| 6 | F | 31 | low | no |
| 7 | M | 25 | none | no |
| 8 | M | 25 | none | no |
| 9 | F | 25 | none | no |
| 10 | M | 26 | high | no |
| 11 | M | 27 | high | no |
| 12 | M | 27 | low | no |
| 13 | M | 23 | low | no |
| 14 | M | 24 | low | no |
| 15 | M | 23 | low | no |
| 16 | M | 23 | low | yes |
| 17 | M | 24 | low | no |
| 18 | M | 27 | high | no |
| 19 | F | 21 | low | no |
| 20 | F | 28 | low | no |
| 21 | F | 24 | none | no |
| 22 | M | 24 | none | yes |
| 23 | F | 25 | low | no |
| 24 | F | 38 | high | yes |
| 25 | F | 24 | low | yes |
| 26 | F | 24 | none | no |
| 27 | M | 37 | low | yes |
| 28 | F | 24 | low | no |
| 29 | F | 25 | low | no |
| 30 | F | 40 | high | no |
| 31 | M | 32 | low | no |
| 32 | M | 21 | none | yes |
| 33 | F | 37 | low | no |
| 34 | M | 24 | none | yes |
| Proximal Perforator | Distal Perforator | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Systolic Velocity | Diastolic Velocity | Median Velocity | Systolic Velocity | Diastolic Velocity | Median Velocity | |||||||
| Pt | Pre | Post | Pre | Post | Pre | Post | Pre | Post | Pre | Post | Pre | Post |
| 1 | 15 | 18 | 1 | 14 | 2 | 17 | 18 | 30 | 1 | 11 | 3 | 12 |
| 2 | 12 | 33 | 1 | 18 | 2 | 17 | 13 | 28 | 1 | 16 | 1 | 19 |
| 3 | 14 | 70 | 2 | 30 | 3 | 30 | 20 | 25 | 1 | 12 | 1 | 12 |
| 4 | 14 | 50 | 1 | 25 | 2 | 30 | 20 | 44 | 1 | 15 | 2 | 20 |
| 5 | 20 | 50 | 1 | 28 | 3 | 23 | 12 | 18 | 5 | 16 | 3 | 12 |
| 6 | 15 | 32 | 1 | 22 | 3 | 19 | 12 | 17 | 1 | 9 | 1 | 7 |
| 7 | 23 | 31 | 9 | 16 | 10 | 15 | 30 | 34 | 6 | 5 | 6 | 4 |
| 8 | 11 | 19 | 1 | 12 | 1 | 12 | 30 | 36 | 1 | 11 | 5 | 10 |
| 9 | 16 | 56 | 1 | 17 | 2 | 26 | 15 | 23 | 1 | 7 | 2 | 9 |
| 10 | 13 | 29 | 1 | 10 | 1 | 12 | 25 | 40 | 1 | 0 | 2 | 5 |
| 11 | 8 | 36 | 3 | 18 | 1 | 14 | 23 | 17 | 1 | 3 | 2 | 1 |
| 12 | 13 | 38 | 3 | 28 | 3 | 20 | 22 | 23 | 3 | 13 | 2 | 12 |
| 13 | 22 | 35 | 1 | 15 | 3 | 14 | 19 | 19 | 1 | 2 | 2 | 3 |
| 14 | 17 | 52 | 1 | 16 | 3 | 18 | 21 | 28 | 1 | 4 | 2 | 8 |
| 15 | 19 | 42 | 1 | 16 | 3 | 18 | 16 | 16 | 1 | 7 | 2 | 7 |
| 16 | 21 | 26 | 1 | 7 | 4 | 10 | 14 | 23 | 1 | 8 | 1 | 4 |
| 17 | 13 | 19 | 1 | 7 | 2 | 7 | 16 | 65 | 1 | 10 | 2 | 21 |
| 18 | 12 | 29 | 1 | 17 | 3 | 18 | 16 | 30 | 1 | 14 | 2 | 15 |
| 19 | 23 | 42 | 8 | 54 | 8 | 33 | 11 | 41 | 1 | 21 | 2 | 19 |
| 20 | 33 | 44 | 6 | 34 | 8 | 32 | 17 | 21 | 1 | 9 | 3 | 7 |
| 21 | 20 | 36 | 7 | 19 | 8 | 5 | 10 | 20 | 1 | 9 | 3 | 7 |
| 22 | 28 | 34 | 1 | 4 | 6 | 9 | 18 | 42 | 3 | 8 | 4 | 9 |
| 23 | 12 | 25 | 2 | 13 | 4 | 8 | 16 | 23 | 1 | 10 | 2 | 11 |
| 24 | 16 | 29 | 1 | 18 | 3 | 16 | 11 | 22 | 1 | 9 | 2 | 10 |
| 25 | 17 | 27 | 1 | 14 | 3 | 16 | 19 | 39 | 1 | 16 | 3 | 19 |
| 26 | 16 | 18 | 1 | 6 | 4 | 8 | 11 | 22 | 1 | 6 | 2 | 10 |
| 27 | 11 | 28 | 1 | 14 | 1 | 11 | 16 | 24 | 3 | 9 | 3 | 10 |
| 28 | 18 | 33 | 1 | 7 | 3 | 8 | 16 | 18 | 1 | 9 | 2 | 7 |
| 29 | 12 | 41 | 1 | 14 | 2 | 18 | 21 | 32 | 1 | 5 | 2 | 7 |
| 30 | 15 | 47 | 2 | 17 | 3 | 19 | 17 | 29 | 1 | 11 | 2 | 12 |
| 31 | 17 | 25 | 1 | 16 | 5 | 24 | 14 | 34 | 1 | 12 | 2 | 11 |
| 32 | 16 | 31 | 1 | 15 | 5 | 23 | 13 | 39 | 2 | 13 | 3 | 8 |
| 33 | 16 | 28 | 2 | 15 | 2 | 13 | 13 | 23 | 1 | 9 | 3 | 12 |
| 34 | 21 | 38 | 1 | 20 | 3 | 16 | 14 | 31 | 1 | 13 | 3 | 11 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amendola, F.; Vaienti, L.; Cottone, G.; Zaccaria, G.; Steinberger, Z.; Dereatti, N.; Riccio, M.; Caputo, G.G.; De Francesco, F.; Zingaretti, N. The Effect of Muscle Exercise on Perforators Flow: A Prospective Cohort Study. Medicina 2020, 56, 338. https://doi.org/10.3390/medicina56070338
Amendola F, Vaienti L, Cottone G, Zaccaria G, Steinberger Z, Dereatti N, Riccio M, Caputo GG, De Francesco F, Zingaretti N. The Effect of Muscle Exercise on Perforators Flow: A Prospective Cohort Study. Medicina. 2020; 56(7):338. https://doi.org/10.3390/medicina56070338
Chicago/Turabian StyleAmendola, Francesco, Luca Vaienti, Giuseppe Cottone, Giovanna Zaccaria, Zvi Steinberger, Nicole Dereatti, Michele Riccio, Glenda Giorgia Caputo, Francesco De Francesco, and Nicola Zingaretti. 2020. "The Effect of Muscle Exercise on Perforators Flow: A Prospective Cohort Study" Medicina 56, no. 7: 338. https://doi.org/10.3390/medicina56070338







