Abstract
An 80-year-old man was admitted with an L5 compression fracture, L4/5 spondylolisthesis, and L5 radiculopathy and underwent a TLIF procedure. Refractory hypotension occurred, though it indicated a possible great vessel injury with vasopressor and fluid infusion. Emergent intraoperative angiography was performed, which showed extravasation at the right common iliac artery. Resuscitative endovascular balloon occlusion of the aorta followed by right common iliac artery stenting was successfully performed to arrest the bleeding. The iatrogenic right common iliac artery laceration was complicated with abdomen compartment syndrome and acute kidney injury. The patient received supportive care, including continuous venovenous hemofiltration (CVVH) for a week, after which the patient’s condition improved. The patient did not have any residual complications at the one-month follow-up. Great vessel injury during the TLIF procedure is rare but fatal. Refractory hypotension is indicative of a great vessel injury. Endovascular intervention is a fast and promising method to diagnose and treat arterial injury.
1. Introduction
Transforaminal lumbar interbody fusion (TLIF) is a common, effective and safe treatment to manage spondylolisthesis with neurological deficit. Iatrogenic vessel injury during a fusion bed preparation is rare but a potentially fatal complication. Here, we introduce a case where an iatrogenic right common iliac artery injury occurred during TLIF. The vessel was resuscitated by endovascular balloon occlusion in the aorta to repair the vascular lesion.
2. Illustrative Case
An 80-year-old man was admitted to our hospital due to low back pain with radiating numbness and soreness in the right buttock and lateral leg. He had been complaining about these symptoms for two years, and they worsened after a falling episode. The patient was brought to the orthopedic department, where a physical examination showed positive lumbar (L) 4/5 knocking tenderness with right L5 radiculopathy. Lumbar spine X-ray and magnetic resonance imaging (Figure 1A–C) showed an L5 body compression fracture, L4/5 spondylolisthesis, and right L4 lateral recess stenosis and severe central stenosis. Based on the diagnosis of an L5 compression fracture with L4/5 instability and neural compression, L4/5 posterior instrumentation fusion and TLIF were performed. The surgery was performed by the open surgical approach. During the preparation of the L4/5 fusion bed, accidental penetration with a rasp occurred while removing a soft callus from the L5 upper endplate. The surgical site was clean, and no active bleeding was observed. However, progressive tachycardia and hypotension were noted five minutes later. The gauze was packed into the posterior wound, and the patient was rapidly moved to the supine position. A distended abdomen was noted. Thereafter, pulseless electric activity was noted, and cardiopulmonary resuscitation was performed. Additionally, retroperitoneum fluid accumulation was observed on sonography. An emergency angiogram showed extravasation of contrast from the right common iliac artery (Figure 2A). An endovascular procedure was performed instead of an exploration laparotomy due to the lesion site’s proximity to the vertebral body and difficulty in achieving a direct approach for repair.
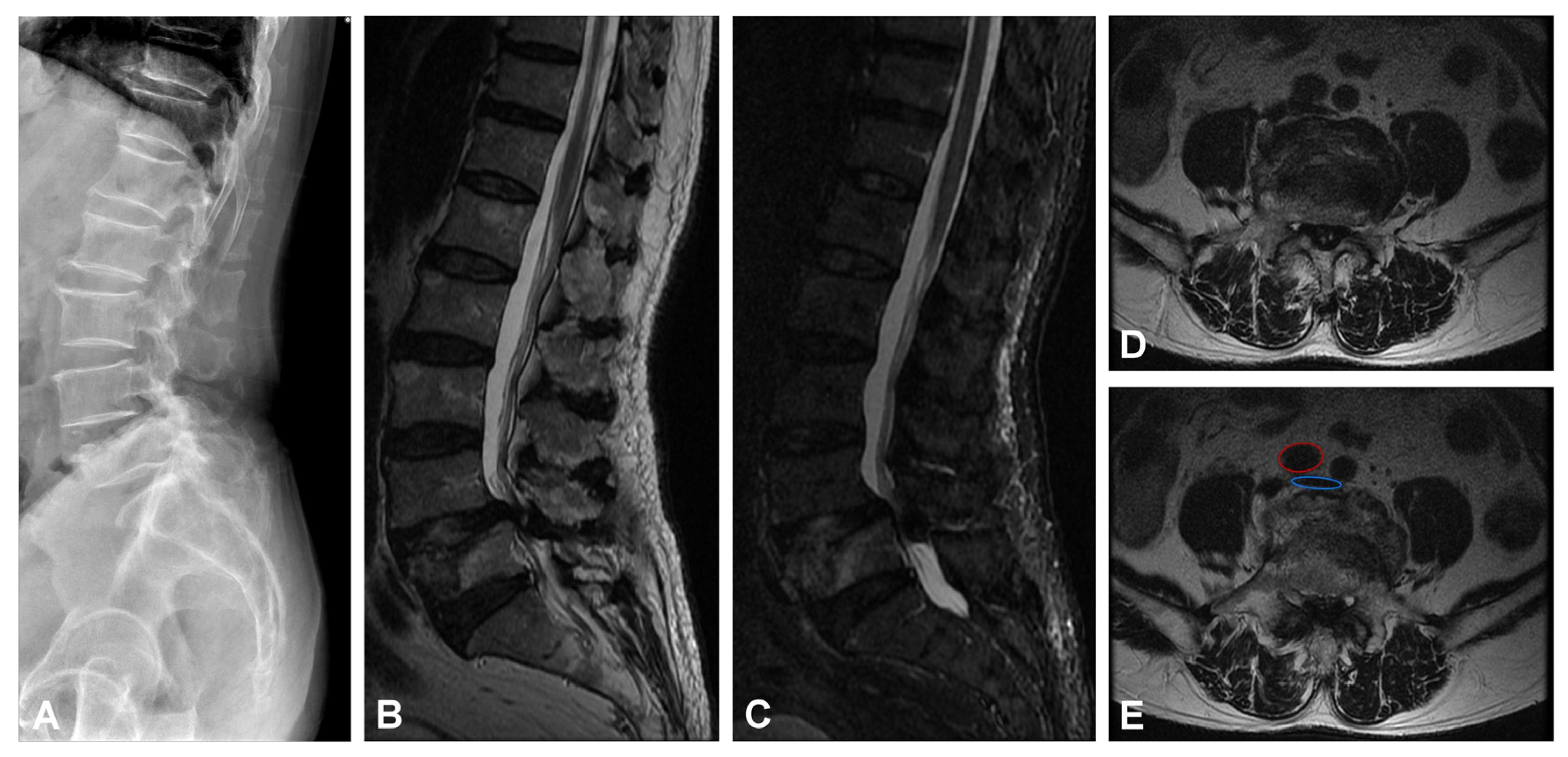
Figure 1.
Images of preoperative. (A) Lateral X-ray view of the lumbar spine. (B) Sagittal T2EI FSE MRI and (C) sagittal STIR MRI L5 old compression fracture, L4/5 spondylolisthesis. (D,E) Axial T2WI FSE MRI showed L4/5 lateral recess stenosis and severe central stenosis. Images (E) showed the pre-operative vascular position of the L5 vertebral body, which is indicated by a red circle symbol (right common iliac artery) and a blue circle symbol (inferior vena cava around the bifurcation).
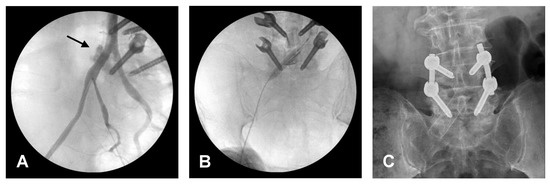
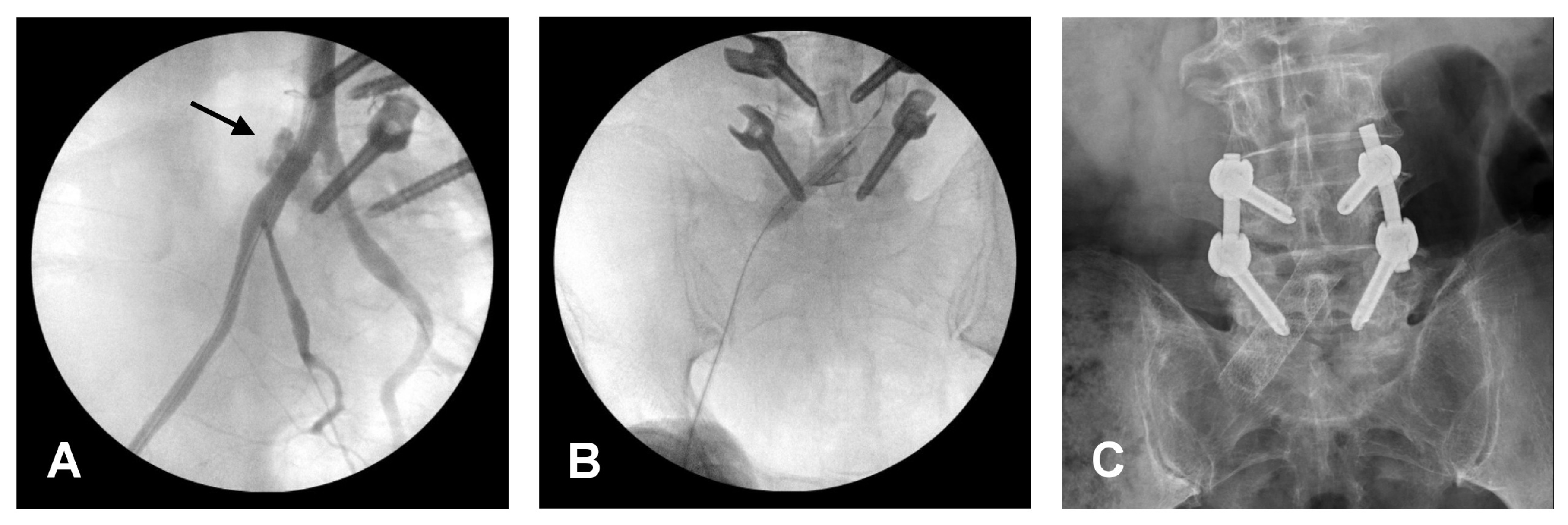
Figure 2.
Intra-operative angiography and post-operative X-ray. (A) Contrast extravasation vias the right common iliac artery, which is indicated by a black arrow. (B) A stent was placed to repair the laceration. (C) Demonstrate stent insertion.
The endovascular procedure began with resuscitative endovascular balloon occlusion of the aorta (REBOA) at the abdomen aorta level to stop the bleeding. A stent graft was placed in the injured right common iliac artery (Figure 2B,C). After the endovascular procedure, the hemodynamic status became stable. The patient was then shifted to a right decubitus position, and the rods and bone substitute were inserted to complete the spinal surgery.
In the intensive care unit, the patient developed acute abdomen compartment syndrome with intra-abdominal pressure (IAP) (20 mmHg), measured indirectly using the bladder technique. Nasogastric tubes were inserted for intestinal pressure decompression. IAP, lactate, liver enzyme, and creatinine levels were closely monitored as a reference to decide when it was appropriate to perform an exploratory laparotomy.
Post-operative oliguria and an increased creatinine level confirmed acute kidney injury (AKI), which may have been caused by hemorrhagic shock or abdominal compartment syndrome. The AKI did not resolve as expected; however, the shock status of the patient improved without inotropic agent usage—the IAP level did not increase, and lactate and liver enzyme levels decreased (Table 1). Therefore, we performed bedside abdomen echocardiography which revealed that a hematoma compressed the bilateral renal arteries. Based on this finding, continuous venovenous hemofiltration (CVVH) was initiated on postoperative day (POD) 8 to provide temporary renal support for the AKI. After a week of CVVH therapy, renal function gradually recovered even though the retroperitoneal hematoma surrounding the renal arteries and kidney was detected on a CT scan (Figure 3A,B). The patient was discharged 31 days postoperatively without renal, bowel, or neurologic sequelae. A month after discharge, the patient followed up at an outpatient clinic. Back pain and neurologic symptoms had improved. His renal function returned to its preoperative condition. His abdomen was soft and free of tenderness. A follow-up abdominal CT showed total resorption of the retroperitoneal hematoma and no evidence of pseudoaneurysm and arteriovenous (AV) fistula. (Figure 3C).

Table 1.
Pateint’s Laboratory Data during Hospitalization.
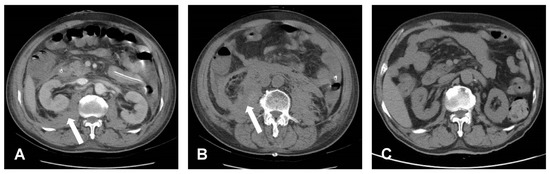
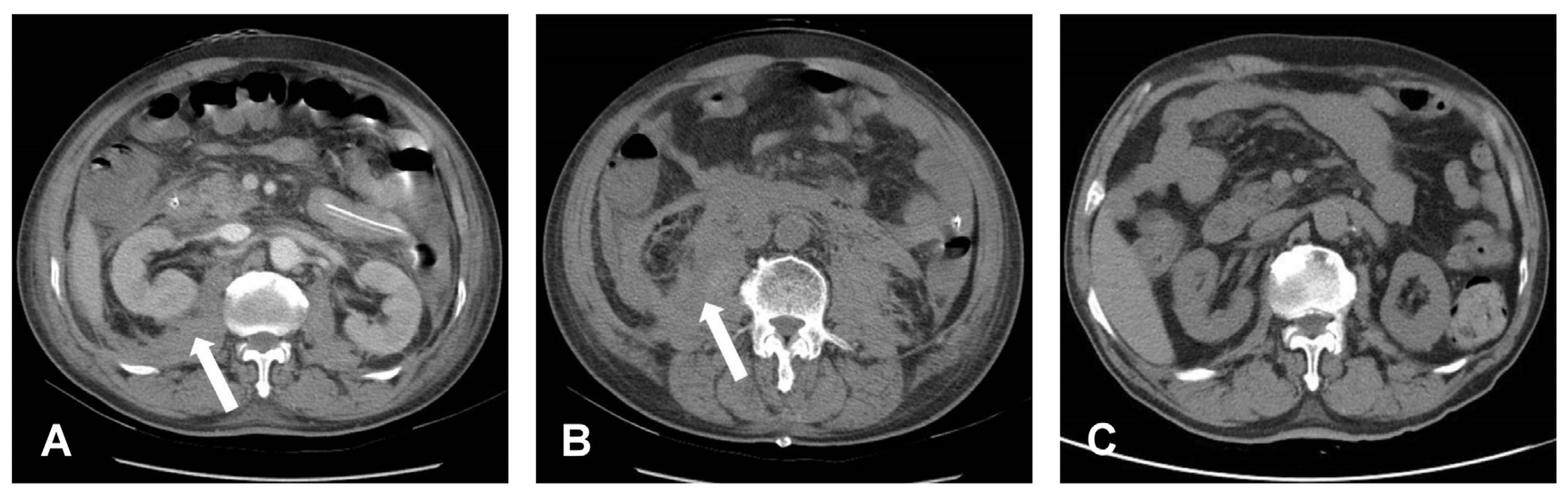
Figure 3.
Post-operative CT. (A,B) Bilateral retroperitoneal hematoma, especially at the right side, which is indicated by a white arrow symbol. (C) Post-operative three months showed totally resolution of retroperitoneal hematoma.
3. Discussion
Iatrogenic vascular injury during lumbar spine surgery is a significant complication because it is difficult to manage and potentially fatal [1,2]. Although the incidence of major vascular injury is rare (0.01–0.05%) [2,3], the mortality rate could reach high at 10–65% [2,4]. According to Papadoulas et al. [2], the incidence of AV fistula, laceration, and pseudoaneurysm due to spine surgery-related vascular injury is 66%, 33%, and 3%, respectively. The three most common clinical presentations of vascular laceration are hypotension (77%), active bleeding (53%), and distended abdomen 20% [2,5]. The symptoms of these complications may present intraoperatively or during the perioperative period. The clinical presentations of AV fistula and pseudoaneurysm can be chronic. AV fistulas usually mimic congestive heart failure with accompanying leg edema. Pseudoaneurysms usually present as new-onset lumbar back pain or neurological symptoms [2,4,6]. They could be diagnosed up to months or years after operation and treated in an elective setting [7,8].
In contrast, most symptoms of vascular laceration may present intraoperatively and should be tended to immediately; on some occasions, the prone position in which the patients are operated on can cause a degree of vascular compression that can temporally tamponade the vascular tears, causing such tears to go unnoticed, thus highly clinical suspicion is required during the perioperative period [9]. Our patient’s tachycardia and hypotension progressed rapidly, and pulseless electrical activity was detected. Later, the right common iliac artery laceration was detected by angiography, the diagnostic tool for detecting vascular injuries. This process is compatible with that described in the previous literature [2,4,10].
Treatment for such vascular injuries can be performed through open or endovascular surgery. Table 2 lists some advantages and disadvantages of open versus endovascular treatment for vascular injuries related to spine surgery and also gives a literature review of the reported cases regarding the injured vessel, injured type, diagnosis timing, and mortality. Open surgery enables the control of acute bleeding and has been traditionally described and recommended to repair these lacerations [10,11].

Table 2.
Advantages and disadvantages of open versus endovascular treatment for vascular injuries related to spine surgery and literature review of previous cases.
Shih et al. described a case that used laparotomy with the abdomen aorta clamped at the level of the celiac axis. However, massive retroperitoneal hematoma complicated the vascular repair. Although the injury to the vessel was repaired, the patient died in the operating room [12]. For our patient, we did not choose open surgery for the following reasons. First, when performing an open laparotomy, compartment pressure is released. This can cause the wound to active bleed, further complicating the vessel repair. Second, the lesion site of the great vessel that adhered anteriorly to the vertebra might have made the repair difficult. Third, since a vein was close to the aorta, the concomitant venous injury was possible. Lastly, an open laparotomy may have caused intestinal exposure and organ swelling, making the abdominal wall difficult to close, contributing to perioperative morbidity and mortality rates [13].
Zajko et al. first reported endovascular repair in 1995. Using a stent graft, they successfully treated a common iliac artery-to-inferior vena cava fistula [14]. The endovascular approach would avoid the above-mentioned disadvantages of the open method. Since then, endovascular stenting repair techniques have been increasingly applied to vascular injuries following lumbar spine surgery. However, almost all of them were applied in postoperative cases (e.g., AV fistula and pseudoaneurysm) [8,15,16,17,18] (Table 2). This technique is rarely applied in vascular laceration due to a lack of availability of the related personnel and facilities in an emergent setting [10]. For this reason, the current literature suggests that primary suture should be considered a first-line treatment [6,14,15,19].
Before the placement of the stent, REBOA is first performed during the same endovascular procedure. In REBOA, a balloon occludes the artery over or above the injury level to help achieve hemostasis, control initial bleeding, and stabilize blood pressure. This technique has made endovascular repair easier, safer, and faster [20]. Although the endovascular approach might salvage the devastating complication of large vessel laceration, there are limitations to this technique. First, the personnel and facilities should be available in a short time. Second, the risk of stenosis, thrombogenesis, and long-term use of anti-thrombotic medication is inevitable.
The abdominal compartment pressure due to the massive hematoma was not relieved after endovascular repair. However, the necessity of surgical decompression was douted [21,22]. In this case, close monitoring of abdominal pressure, optimization of systemic perfusion, indwelling nasogastric tube for gastrointestinal and colonic decompression, and temporary continuous renal replacement are successful strategies we strongly recommend [23].
4. Conclusions
Iatrogenic vessel injury due to TLIF is a major complication that increases perioperative morbidity and mortality. A sudden occurrence of refractory hypotension indicated a great vessel injury. Endovascular surgery is a promising approach to control bleeding and treating vascular injury.
Author Contributions
Investigation: C.-M.C., K.-L.L. and I.-H.C.; writing—original draft: C.-M.C.; writing—review and editing: K.-L.L. and I.-H.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This report was approved by the Research Ethics Committee Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation (No. CR111-03).
Informed Consent Statement
Written informed consent was obtained from the patient to publish the case report and associated images.
Data Availability Statement
Data sharing not applicable.
Acknowledgments
The authors are grateful to the cardiovascular surgeon Jin-You Jhan, who immediately performed endovascular intervention to save the patient’s life, and Yu Ru Kou for the valuable suggestions for the revision of this manuscript.
Conflicts of Interest
The authors have no conflict of interest to disclose.
Abbreviations
AKI—acute kidney injury; AV—arteriovenous; CVVH—continuous venovenous hemofiltration; IAP—intra-abdominal pressure; L—lumbar; REBOA—resuscitative endovascular balloon occlusion of the aorta; TLIF—transforaminal lumbar interbody fusion
References
- Vaccaro, A.R.; Kepler, C.K.; Rihn, J.A.; Suzuki, H.; Ratliff, J.K.; Harrop, J.S.; Morrison, W.B.; Limthongkul, W.; Albert, T.J. Anatomical Relationships of the Anterior Blood Vessels to the Lower Lumbar Intervertebral Discs: Analysis based on magnetic resonance imaging of patients in the prone position. J. Bone Jt. Surg. 2012, 94, 1088–1094. [Google Scholar] [CrossRef]
- Papadoulas, S.; Konstantinou, D.; Kourea, H.; Kritikos, N.; Haftouras, N.; Tsolakis, J. Vascular Injury Complicating Lumbar Disc Surgery. A Systematic Review. Eur. J. Vasc. Endovasc. Surg. 2002, 24, 189–195. [Google Scholar] [CrossRef] [Green Version]
- Ewah, M.B.; Calder, M.I. Intraoperative death during lumbar discectomy. Br. J. Anaesth. 1991, 66, 721–723. [Google Scholar] [CrossRef] [PubMed]
- Goodkin, R.; Lewis, L.L. Vascular and Visceral Injuries Associated with Lumbar Disc Surgery: Medicolegal Implications. Surg. Neurol. 1998, 49, 358–372. [Google Scholar] [CrossRef]
- Van Zitteren, M.; Fan, B.; Lohle, P.N.; de Nie, J.C.; Malefijt, J.D.W.; Vriens, P.W.; Heyligers, J.M. A Shift Toward Endovascular Repair for Vascular Complications in Lumbar Disc Surgery During the Last Decade. Ann. Vasc. Surg. 2013, 27, 810–819. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.S.; Kim, D.J.; Kim, H.S.; Lee, H.K.; Na Choi, S.J.; Chung, S.Y. Vascular Complications Related to Posterior Lumbar Disc Surgery. Vasc. Spéc. Int. 2017, 33, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.P.; Farha, G.J. Arteriovenous Fistula and Isolated Vascular Injuries Secondary to Intervertebral Disk Surgery. Ann. Surg. 1965, 161, 524–531. [Google Scholar] [CrossRef]
- Momoh, A.O.; Barshes, N.R.; Kougias, P.; Huynh, T.T.; Lin, P.H. Endovascular treatment of postlaminectomy aortic pseudoaneurysm. J. Vasc. Surg. 2008, 47, 1083–1085. [Google Scholar] [CrossRef] [Green Version]
- Canaud, L.; Hireche, K.; Joyeux, F.; D’Annoville, T.; Berthet, J.-P.; Marty-Ané, C.; Alric, P. Endovascular Repair of Aorto-iliac Artery Injuries after Lumbar-spine Surgery. Eur. J. Vasc. Endovasc. Surg. 2011, 42, 167–171. [Google Scholar] [CrossRef] [Green Version]
- Szolar, D.H.; Preidler, K.W.; Steiner, H.; Riepl, T.; Flaschka, G.; Stiskal, M.; Moelleken, S.; Norman, D. Vascular complications in lumbar disk surgery: Report of four cases. Neuroradiology 1996, 38, 521–525. [Google Scholar] [CrossRef]
- Düz, B.; Kaplan, M.; Günay, C.; Ustünsöz, B.; Uğurel, M.S. Iliocaval arteriovenous fistula following lumbar disc surgery: Endovascular treatment with a Stent-graft. Turk Neurosurg. 2008, 18, 245–248. [Google Scholar]
- Lee, C.H.; Shih, T.Y.; Wu, Y.C.; Chen, K.H.; Pan, C.C.; Shih, C.M.; Hung, L.K. Rescue of Life-Threatening Great Vessel Injury during Posterior Spine Surgery. Biomed. J. Sci. Tech. Res. 2020, 24, 18594–18598. [Google Scholar] [CrossRef]
- Nam, T.-K.; Park, S.-W.; Shim, H.-J.; Hwang, S.-N. Endovascular Treatment for Common Iliac Artery Injury Complicating Lumbar Disc Surgery: Limited Usefulness of Temporary Balloon Occlusion. J. Korean Neurosurg. Soc. 2009, 46, 261–264. [Google Scholar] [CrossRef]
- Inamasu, J.; Guiot, B.H. Vascular injury and complication in neurosurgical spine surgery. Acta Neurochir. 2006, 148, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.J.; Oh, J.H.; Yoon, Y. Percutaneous endovascular stent-graft for iliac pseudoaneurysm following lumbar discectomy. Cardiovasc. Interv. Rad. 2000, 23, 475–477. [Google Scholar] [CrossRef] [PubMed]
- Giugliano, G.; Laurenzano, E.; Rengo, C.; De Rosa, G.; Brevetti, L.; Sannino, A.; Perrino, C.; Chiariotti, L.; Schiattarella, G.G.; Serino, F.; et al. Abdominal aortic aneurysm in patients affected by intermittent claudication: Prevalence and clinical predictors. BMC Surg. 2012, 12 (Suppl. 1). [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.-K.; Choe, W.J.; Koh, Y.-C.; Park, S.W. Endovascular Management of Great Vessel Injury Following Lumbar Microdiscectomy. Korean J. Spine 2013, 10, 264–267. [Google Scholar] [CrossRef]
- Yan, G.-W.; Li, H.-W.; Yang, G.-Q.; Bhetuwal, A.; Liu, J.-P.; Li, Y.; Fu, Q.-S.; Zhao, L.-W.; Chen, H.; Hu, N.; et al. Iatrogenic arteriovenous fistula of the iliac artery after lumbar discectomy surgery: A systematic review of the last 18 years. Quant. Imaging Med. Surg. 2019, 9, 1163–1175. [Google Scholar] [CrossRef]
- Liu, B.; Ye, K.; Gao, S.; Liu, K.; Feng, H.; Zhou, F.; Tian, Y. The summary of experience of abdominal vascular injury related to posterior lumbar surgery. Int. Orthop. 2019, 43, 2191–2198. [Google Scholar] [CrossRef]
- Malina, M.; Veith, F.; Ivancev, K.; Sonesson, B. Balloon Occlusion of the Aorta During Endovascular Repair of Ruptured Abdominal Aortic Aneurysm. J. Endovasc. Ther. 2005, 12, 556–559. [Google Scholar] [CrossRef]
- Sosa, G.; Gandham, N.; Landeras, V.; Calimag, A.P.; Lerma, E. Abdominal compartment syndrome. Dis. Mon. 2019, 65, 5–19. [Google Scholar] [CrossRef] [PubMed]
- De Laet, I.E.; Malbrain, M.; De Waele, J.J. A Clinician’s Guide to Management of Intra-abdominal Hypertension and Abdominal Compartment Syndrome in Critically Ill Patients. Crit. Care 2020, 24, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheatham, M.L.; Malbrain, M.L.N.G.; Kirkpatrick, A.; Sugrue, M.; Parr, M.; De Waele, J.; Balogh, Z.; Leppäniemi, A.; Olvera, C.; Ivatury, R.; et al. Results from the International Conference of Experts on Intra-abdominal Hypertension and Abdominal Compartment Syndrome. II. Recommendations. Intensiv. Care Med. 2007, 33, 951–962. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).