Aggressive Male Breast Cancer—Clinical and Therapeutic Aspects Correlated with the Histopathological Examination: A Case Report and Literature Review
Abstract
:1. Introduction
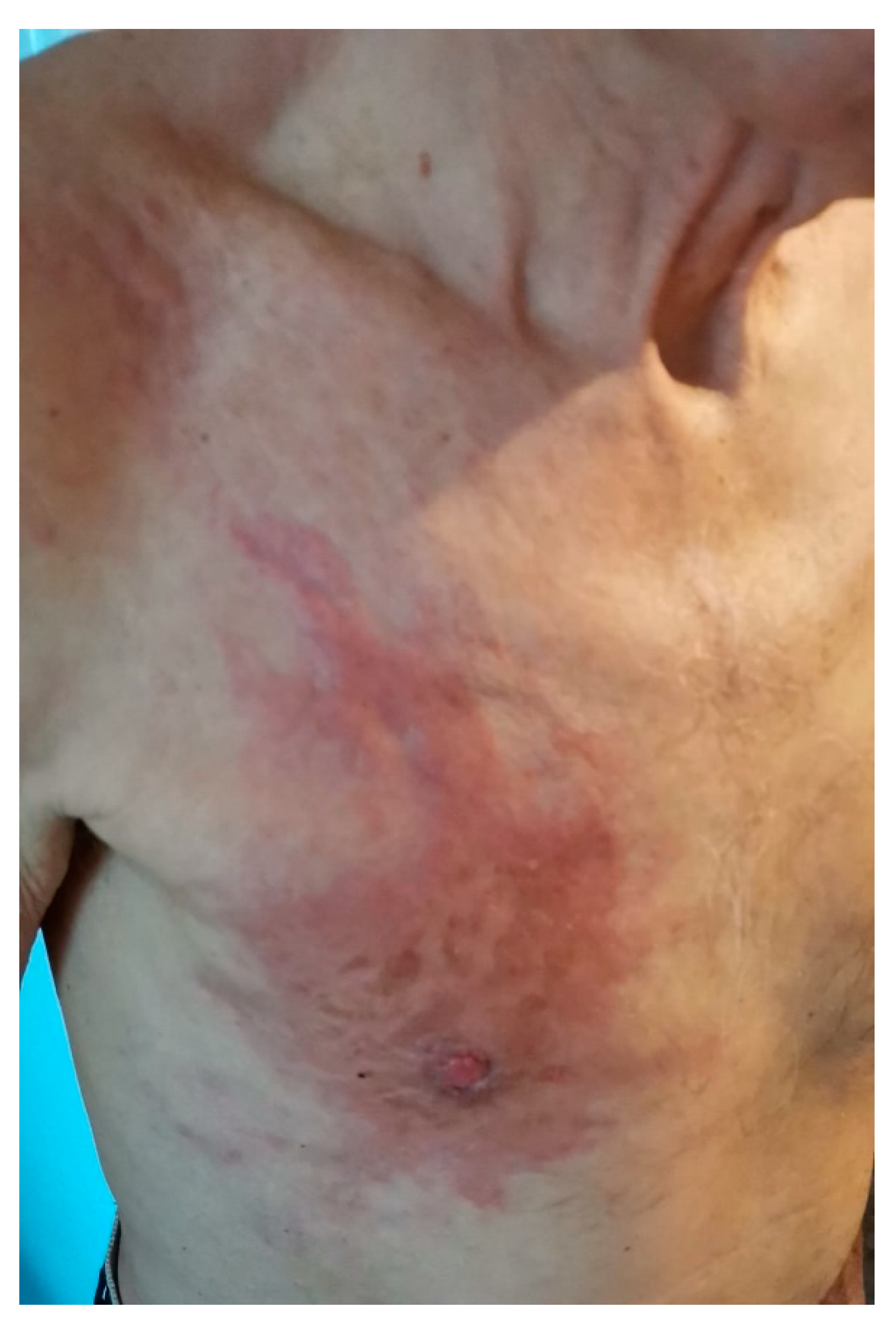
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Korde, L.A.; Zujewski, J.A.; Kamin, L.; Giordano, S.; Domchek, S.; Anderson, W.F.; Bartlett, J.M.; Gelmon, K.; Nahleh, Z.; Bergh, J.; et al. Multidisciplinary meeting on male breast can—cer: Summary and research recommendations. J. Clin. Oncol. 2010, 28, 2114–2122. [Google Scholar] [CrossRef]
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Thun, M.J. Cancer statistics, 2009. CA Cancer J. Clin. 2009, 59, 225–249. [Google Scholar] [CrossRef]
- Darkeh, M.; Azavedo, E. Male Breast Cancer Clinical Features, Risk Factors, and Current Diagnostic and Therapeutic Approaches. Int. J. Clin. Med. 2014, 5, 1068–1086. [Google Scholar] [CrossRef]
- Sosa, A.; Espinoza, S.; Aguilar, R.; Palencia, R. Male Breast Cancer: Case Report. Rev. Colomb. Radiol. 2017, 28, 4810–4815. [Google Scholar]
- Pritzlaff, M.; Summerour, P.; McFarland, R.; Li, S.; Reineke, P.; Dolinsky, J.S.; Goldgar, D.E.; Shimelis, H.; Couch, F.J.; Chao, E.C.; et al. Male breast cancer in a multi-gene panel testing cohort: Insights and unexpected results. Breast Cancer Res. Treat. 2017, 161, 575–586. [Google Scholar] [CrossRef]
- Scatena, C.; Scarpitta, R.; Innocenti, L.; Miccoli, M.; Biancotti, R.; Diodati, L.; Ghilli, M.; Naccarato, A.G. Androgen receptor expression inversely correlates with histological grade and N stage in ER. Breast Cancer Res. Treat. 2020, 182, 55–65. [Google Scholar] [CrossRef]
- Prasad, S.; Kumar-Srivastava, A. Carcinoma breast male: A case report. Am. J. Med. Case Rep. 2014, 2, 48–49. [Google Scholar]
- Khan, L.; Dixon, M. Case report: Male breast cancer. Am. J. Hematol./Oncol. 2016, 12, 11–14. [Google Scholar]
- Giordano, S.H. A review of the diagnosis and management of male breast cancer. Oncologist 2005, 10, 471–479. [Google Scholar] [CrossRef]
- Thomas, D.B. Breast cancer in men. Epidemiol. Rev. 1993, 15, 220–231. [Google Scholar] [CrossRef]
- Zeeshan, S.; Siddiqiui, T.; Shaukat, F.; Tariq, M.U.; Khan, N.; Vohra, L. Male Breast Cancer: The Three Decades’ Experience of a Tertiary Care Hospital in a Lower-Middle Income Country. Cureus 2022, 14, e22670. [Google Scholar] [CrossRef] [PubMed]
- Patten, D.K.; Sharifi, L.K.; Fazel, M. New approaches in the management of male breast cancer. Clin. Breast Cancer 2013, 13, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Li, J.; Zhu, S.; Wu, J.; Li, X.; Liu, Q.; Wei, W.; Sun, S. Poorer breast cancer survival outcomes in males than females might be attributable to tumor subtype. Oncotarget 2016, 7, 87532–87542. [Google Scholar] [CrossRef] [PubMed]
- Lin, N.; Xu, Q.; Wang, B.; Dang, Y.; Lin, C. Male HER-2 positive metastatic breast cancer: A case report. Asian J. Surg. 2020, 43, 1175–1176. [Google Scholar] [CrossRef] [PubMed]
- Alsayed, B.; Abdulla, H.A.; Alaskar, H.; Dhaif, A. Male occult triple-negative breast cancer. BMJ Case Rep. 2019, 12, e229482. [Google Scholar] [CrossRef] [PubMed]
- Takuwa, H.; Tsuji, W.; Shintaku, M.; Yotsumoto, F. Hormone signaling via androgen receptor affects breast cancer and prostate cancer in a male patient: A case report. BMC Cancer 2018, 18, 1282. [Google Scholar] [CrossRef]
- Wang, B.; Wang, L.; Zhao, Z.; Xu, X. Cystic encapsulated papillary carcinoma with ductal carcinoma in situ in the male breast: A clinicopathologic feature with a diagnostic challenge: A case report and review of literature. Medicine 2023, 102, e34388. [Google Scholar] [CrossRef]
- Mukendi, A.M.; Van Den Berg, E.; Pather, S.; Padayachee, R.S. Metachronous or synchronous male breast and prostate cancers a duality to lookout for. F1000Research 2018, 7, 1825. [Google Scholar] [CrossRef]
- Ramakrishna, K.N.; Durland, J.; Ramos, C.; Dhamoon, A.S. Unilateral nipple discharge in a man without a palpable mass diagnosed as breast cancer. BMJ Case Rep. 2020, 13, e236223. [Google Scholar] [CrossRef]
- Cortina, C.S.; Madrigrano, A. Nipple changes in an 85-year-old man. CMAJ 2019, 191, E135. [Google Scholar] [CrossRef]
- Luo, H.; Meng, K.; He, J.; Hu, Z.; Yang, O.; Lan, T.; Su, K.; Yang, H.; Zhan, C.; Xu, H. Intracystic papillary carcinoma of the breast in males: Three case reports. Medicine 2020, 99, e20278. [Google Scholar] [CrossRef] [PubMed]
- Başara Akın, I.; Özgül, H.A.; Gürel, D.; Aksoy, S.; Balcı, P. Elastography findings of encapsulated solid papillary carcinoma of breast in a man. Med. Ultrason. 2019, 21, 491–493. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, S.; Jayant, K.; Agarwal, R.K.; Dayama, K.G.; Arora, S. An unusual case of metastatic male breast cancer to the nasopharynx-review of literature. Ann. Palliat. Med. 2015, 4, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Gautam, S.; Joshi, B.R.; Adhikary, S.; Regmi, S.; Pradhan, A. Male Breast Cancer: A Rare Entity. JNMA J. Nepal. Med. Assoc. 2018, 56, 804–807. [Google Scholar] [CrossRef] [PubMed]
- Sekal, M.; Znati, K.; Harmouch, T.; Riffi, A.A. Apocrine carcinoma of the male breast: A case report of an exceptional tumor. Pan Afr. Med. J. 2014, 19, 294. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.P.; Mannelli, V.K.; Kandola, S.; Marinas, E.B. Pleomorphic sarcoma of the breast. BMJ Case Rep. 2017, 2017, bcr-2017. [Google Scholar] [CrossRef] [PubMed]
- Sucharita, S.; Sahu, N.; Giri, R.; Sahu, S.K. Mammary not otherwise specified-type sarcoma with CD10 expression. J. Cancer Res. Ther. 2023, 19 (Suppl. 1), S454–S457. [Google Scholar] [CrossRef]
- Gucalp, A.; Traina, T.A.; Eisner, J.R.; Parker, J.S.; Selitsky, S.R.; Park, B.H.; Elias, A.D.; Baskin-Bey, E.S.; Cardoso, F. Male breast cancer: A disease distinct from female breast cancer. Breast Cancer Res. Treat. 2019, 173, 37–48. [Google Scholar] [CrossRef]
- Giotta, F.; Acito, L.; Candeloro, G.; Del Medico, P.; Gadaleta-Caldarola, G.; Giordano, G.; Gueli, R.; Lugini, A.; Magri, V.; Mandarŕ, M.; et al. Eribulin in Male Patients with Breast Cancer: The First Report of Clinical Outcomes. Oncologist 2016, 21, 1298–1305. [Google Scholar] [CrossRef]
- Joshi, M.G.; Lee, A.K.; Loda, M.; Camus, M.G.; Pedersen, C.; Heatley, G.J.; Hughes, K.S. Male breast carcinoma: An evaluation of prognostic factors contributing to a poorer outcome. Cancer 1996, 77, 490–498. [Google Scholar] [CrossRef]
- Ottini, L.; Capalbo, C.; Rizzolo, P.; Silvestri, V.; Bronte, G.; Rizzo, S.; Russo, A. HER2-positive male breast cancer: An update. Breast Cancer 2010, 2, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Cutuli, B. Strategies in treating male breast cancer. Expert. Opin. Pharmacother. 2007, 8, 193–202. [Google Scholar] [CrossRef]
- Zaha, D.C. Significance of immunohistochemistry in breast cancer. World J. Clin. Oncol. 2014, 5, 382–392. [Google Scholar] [CrossRef] [PubMed]
- Luna-Moré, S.; Rius, F.; Weil, B.; Jimenez, A.; Bautista, M.D.; Pérez-Mellado, A. EMA: A differentiation antigen related to node metastatic capacity of breast carcinomas. Pathol. Res. Pract. 2001, 197, 419–425. [Google Scholar] [CrossRef]
- Akosa, A.; Van Norden, S.; Tettey, Y. Hormone receptor expression in male breast cancers. Ghana Med. J. 2005, 39, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Li, L.T.; Jiang, G.; Chen, Q.; Zheng, J.N. Ki67 is a promising molecular target in the diagnosis of cancer (review). Mol. Med. Rep. 2015, 11, 1566–1572. [Google Scholar] [CrossRef]
- Nishimura, R.; Osako, T.; Okumura, Y.; Hayashi, M.; Toyozumi, Y.; Arima, N. Ki-67 as a prognostic marker according to breast cancer subtype and a predictor of recurrence time in primary breast cancer. Exp. Ther. Med. 2010, 1, 747–754. [Google Scholar] [CrossRef]
- Nishimura, R.; Osako, T.; Okumura, Y.; Hayashi, M.; Arima, N. Clinical significance of Ki-67 in neoadjuvant chemotherapy for primary breast cancer as a predictor for chemosensitivity and for prognosis. Breast Cancer 2010, 17, 269–275. [Google Scholar] [CrossRef]
- Singhai, R.; Patil, V.W.; Jaiswal, S.R.; Patil, S.D.; Tayade, M.B.; Patil, A.V. E-Cadherin as a diagnostic biomarker in breast cancer. N. Am. J. Med. Sci. 2011, 3, 227–233. [Google Scholar] [CrossRef]
- Ottini, L.; Palli, D.; Rizzo, S.; Federico, M.; Bazan, V.; Russo, A. Male breast cancer. Crit. Rev. Oncol. Hematol. 2010, 73, 141–155. [Google Scholar] [CrossRef]
- Johansen Taber, K.A.; Morisy, L.R.; Osbahr, A.J.; Dickinson, B.D. Male breast cancer: Risk factors, diagnosis, and management (Review). Oncol. Rep. 2010, 24, 1115–1120. [Google Scholar] [CrossRef] [PubMed]
- Sousa, B.; Moser, E.; Cardoso, F. An update on male breast cancer and future directions for research and treatment. Eur. J. Pharmacol. 2013, 717, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Sauder, C.A.M.; Bateni, S.B.; Davidson, A.J.; Nishijima, D.K. Breast Conserving Surgery Compared with Mastectomy in Male Breast Cancer: A Brief Systematic Review. Clin. Breast Cancer 2020, 20, e309–e314. [Google Scholar] [CrossRef] [PubMed]
- Somsekhar, S.P.; Geeta, K.; Jain, R.; Nayyer, R.; Halder, S.; Malik, V.K.; Parikh, P.; Aggarwal, S.; Koul, R. Practical consensus recommendations regarding role of mastectomy in metastatic breast cancer. South Asian J. Cancer 2018, 7, 79–82. [Google Scholar] [CrossRef]
- Altiner, S.; Altiner, Ö.; Büyükkasap, Ç.; Uğraş Dikmen, A.; Pekcici, M.R.; Erel, S. Analysis of Knowledge About Male Breast Cancer Among Patients at Tertiary Medical Center. Am. J. Men’s Health 2023, 17, 15579883231165626. [Google Scholar] [CrossRef]





















| Study [Ref.] | Age (Years) | Side | Size [cm] | Clinical Characteristics | Axillary Lymph Node | Biopsy |
|---|---|---|---|---|---|---|
| Na Lin et al. [14] | 78 | Right breast | 2.5 × 1.1 cm | No tenderness, no skin changes, no bloody fluid overflow at the nipple | Right axillary lymph nodes were palpable | Core needle biopsy—invasive ductal carcinoma |
| Basma Alsayed et al. [15] | 82 | Left axilla | 8 × 7 cm | There were no masses felt in either breast | The overlying skin was erythematous, and there was a sinus discharging serous fluid. | Core-needle biopsy—invasive ductal carcinoma |
| Haruko Takuwa et al. [16] | 69 | Left breast | >6 cm | Tumor mass without skin invasion in the upper-lateral region as well as axillary lymph node swelling | Left axillary lymph node swelling | Core-needle biopsy—invasive ductal carcinoma |
| Bo Wang et al. [17] | 73 | Right breast | 3.6 × 2.3 cm | Hard, smooth and movable lesion was palpated below the right papilla. There were no skin lesions | No palpable axillary lymph node | Breast tumor resection—a little papillary neoplasm of the breast with epithelial atypia and hypertrophy in the fibrous cystic wall with a little DCIS |
| Alain Mwamba Mukendi et al. [18] | 68 | Right breast | Not mentioned | Painless right breast lump | No palpable axillary lymph node | Core biopsy of the right breast—infiltrating ductal carcinoma displaying cribriform features. |
| Karan N Ramakrishna et al. [19] | 69 | Right breast | 0.6 cm mass | Right-sided serous nipple discharge; nipple swelling and pain | No palpable axillary lymph node | Ultrasound-guided biopsy—atypical ductal hyperplasia Excisional biopsy—ductal carcinoma in situ |
| Chandler S Cortina et al. [20] | 85 | Left breast | 2 cm | Pedunculated mass over the nipple and associated nipple and areola enlargement | No palpable lymph node | Excisional biopsy—invasive ductal carcinoma |
| Hua Luo et al. [21] | 70 | Left breast | 2 cm | A well-circumscribed, firm and mobile mass in the left periareolar region. | No palpable lymph node | Excisional biopsy—intracystic papillary carcinoma |
| Hua Luo et al. [21] | 67 | Right breast | 1.5 | A well-circumscribed and firm mass in the right subareolar region. The tumor was fixed | No palpable lymph node | Excisional biopsy—intracystic papillary carcinoma |
| Hua Luo et al. [21] | 76 | Right breast | 1 cm | Mobile lump in the right breast | No palpable lymph node | Lumpectomy—intracystic papillary carcinoma |
| Işıl Başara Akın et al. [22] | 72 | Right breast | 4 cm | A painless, mobile lesion at the retroareolar region of the breast | No palpable lymph node | US-guided core needle biopsy—encapsulated solid papillary carcinoma |
| Swati Agrawal et al. [23] | 65 | Right breast | 3 cm | Palpable mass in his right breast | No palpable lymph node | Fine-needle aspiration cytology—suggestive of malignancy |
| Swotantra Gautam et al. [24] | 78 | Left breast | 3 × 2 cm | A non-tender lump just beneath the left nipple; it was mobile and not adhered to underlying structures | No palpable lymph node | Fine-needle cytology—carcinoma of breast |
| Mohammed Sekal et al. [25] | 70 | Left breast | 4 cm | Nodule presented a rapid augmentation of its volume with adhesion to both superficial and deep plans and inflammatory opposite signs | No palpable lymph node | Fine-needle aspiration cytology—apocrine carcinoma |
| Manoj P Rai et al. [26] | 81 | Left breast | 1.2 × 0.9 cm | Breast mass in the lower inner quadrant | Left axillary lymphadenopathy | Left breast lumpectomy—low-grade pleomorphic sarcoma |
| Soumya Sucharita et al. [27] | 80 | Right breast | 6 cm | A well-circumscribed and firm mass. The corresponding skin surface was normal. | One right axillary lymph node was palpable | Fine-needle aspiration cytology was performed from both the breast mass and axillary lymph node—ductal carcinoma. Lymph node showed the features of reactive hyperplasia |
| Study [Ref.] | ER | PR | HER2 | Ki 67 | Neoadjuvant Chemotherapy | Surgical Treatment | Axillary Lymph Node Hp | Stage | HP of Specimen |
|---|---|---|---|---|---|---|---|---|---|
| [14] | 90% | 80% | Positive | 70% | 4 cycles: paclitaxel, capecitabine and trastuzumab | Modified radical mastectomy | reactive hyperplasia | T2N3M1 | invasive ductal carcinoma |
| [15] | Negative | Negative | Negative | Not determined | No | Left modified radical mastectomy | 4 out of 21 axillary lymph nodes showing metastatic disease | pT3N2M0 | invasive ductal carcinoma |
| [16] | Positive | Negative | Negative | 10% | No | Mastectomy and axillary dissection | 36 out of 39 axillary lymph nodes showing metastatic disease | pT3N3aM0 (stage III) | invasive ductal carcinoma |
| [17] | Positive | Positive (20%) | Negative | 20% | No | Breast tumor resection | no | Not mentioned | ductal carcinoma in situ |
| [18] | Positive (>91%) | Positive (70%) | Negative | 30% | Tamoxifen 4 months | Right mastectomy and right axillary lymph node dissection | not mentioned | T4bN1Mx | invasive ductal carcinoma |
| [19] | Positive | Positive | Not mentioned | Not mentioned | No | Right total mastectomy + right axillary sentinel lymph node | no evidence of tumor spread to the lymph nodes | TisN0cM0 | ductal carcinoma in situ |
| [20] | Positive | Positive | Equivocal | Not mentioned | No | Mastectomy with sentinel node biopsy | no evidence of tumor spread to the lymph nodes | T4bN0M0 | invasive ductal carcinoma |
| [21] | Positive (90%) | Positive (>99%) | Negative | 10% | No | Simple mastectomy with axillary sentinel lymph node biopsy | no positive axillary lymph node was detected. | Not mentioned | intracystic papillary carcinoma |
| [21] | Positive | Positive | Negative | 35% | No | Mastectomy with sentinel lymph node mapping | no positive axillary lymph node was detected. | Not mentioned | intracystic papillary carcinoma with a small focus on invasive carcinoma |
| [21] | Positive | Positive | Negative | 60% | No | Right mastectomy | not made | Not mentioned | intracystic papillary carcinoma |
| [22] | Positive | Positive | Negative | Not mentioned | No | Total mastectomy | not made | Not mentioned | encapsulated solid papillary carcinoma |
| [23] | Positive | Positive | Negative | Not mentioned | No | Right modified radical mastectomy with an axillary lymph node dissection | 5 of 16 axillary nodes involved | T2pN1M0 | invasive adenocarcinoma |
| [24] | Not mentioned | Not mentioned | Not mentioned | Not mentioned | No | Right modified radical mastectomy with an axillary lymph node dissection | no positive axillary lymph node was detected | T2N0M0 | invasive breast carcinoma, NOS |
| [25] | Negative | Negative | Negative | Not mentioned | Palliative chemotherapy | No surgery | no surgery | Stage IV (lung metastases) | no surgery |
| [26] | Positive | Positive | Not mentioned | Not mentioned | No | No other surgery | no surgery | T1acN0M0 | no surgery |
| [27] | Not determined | Not determined | Not determined | Not determined | No | Modified radical mastectomy | lymph node—reactive hyperplasia | Not mentioned | NOS type sarcoma |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrescu, A.-M.; Pirici, N.-D.; Ruxanda, A.-I.; Vasile, L.; Pîrșcoveanu, M.; Paitici, Ș.; Petrescu, G.-S.; Munteanu, A.C.; Matei, R.-A.; Dumitrache, D.; et al. Aggressive Male Breast Cancer—Clinical and Therapeutic Aspects Correlated with the Histopathological Examination: A Case Report and Literature Review. Medicina 2023, 59, 2167. https://doi.org/10.3390/medicina59122167
Petrescu A-M, Pirici N-D, Ruxanda A-I, Vasile L, Pîrșcoveanu M, Paitici Ș, Petrescu G-S, Munteanu AC, Matei R-A, Dumitrache D, et al. Aggressive Male Breast Cancer—Clinical and Therapeutic Aspects Correlated with the Histopathological Examination: A Case Report and Literature Review. Medicina. 2023; 59(12):2167. https://doi.org/10.3390/medicina59122167
Chicago/Turabian StylePetrescu, Ana-Maria, Nicolae-Daniel Pirici, Anca-Ileana Ruxanda, Liviu Vasile, Mircea Pîrșcoveanu, Ștefan Paitici, Gabriel-Sebastian Petrescu, Alexandru Claudiu Munteanu, Ramona-Andreea Matei, Daniel Dumitrache, and et al. 2023. "Aggressive Male Breast Cancer—Clinical and Therapeutic Aspects Correlated with the Histopathological Examination: A Case Report and Literature Review" Medicina 59, no. 12: 2167. https://doi.org/10.3390/medicina59122167





