Abstract
Background and Objectives: The diagnosis of anaphylaxis comprehensively depends on both situational information and laboratory investigations. For this purpose, serum tryptase concentration is examined as an indicator of systemic mast cell mediator release, linked to an underlying anaphylactic process. Increased levels of tryptase may occur in some events different from anaphylaxis, but usually information from crime scene investigations is lacking and autoptic findings are not specific. For legal reasons, it is required to achieve a definite diagnosis of mast cell degranulation that can lead to a certain diagnosis of death from anaphylaxis. Immunohistochemistry seems to be a relatively simple, reliable, and easily repeatable method that can assist the forensic pathologist in the differential diagnosis of death from anaphylaxis. Materials and Methods: This work provides an overview of the current literature on immunohistochemical methods useful in the determination process of anaphylactic-related deaths. A systematic search, according to the PRISMA statement, was performed in databases to identify studies investigating immunohistochemical targets related to anaphylaxis death. Results: This work underscores the importance of anaphylaxis mediators such as tryptase, CD117, and chymase in the immunohistochemical analysis of anaphylactic deaths. Conclusions: According to the reviewed literature, the diagnosis of death due to anaphylaxis should depend not just on the suspicion of an anaphylactic reaction but also on confirming mast cell degranulation through the identification of IHC positivity for inflammatory mediators, particularly in the respiratory tract.
1. Introduction
Anaphylaxis is an acute, life-threatening, systemic reaction caused by the immediate release of basophils and mast cells’ mediators in response to the exposure to an allergen [1]. It is characterized by various clinical presentations, but the most worrisome symptoms are the obstruction of the upper airway, bronchospasm, and hypotension [2]. The post-mortem diagnosis of fatal anaphylaxis may be challenging. Since death is usually the consequence of asphyxia or rapid shock, it could be difficult to identify useful post-mortem findings; in fact, there may not be sufficient time for specific features to appear. In the case of food, venom, or drug allergy, the specific features are generally present in only 59% of cases [3]. The suspicion of this kind of death arises when there is a history of poorly controlled asthma, insect bite, or medication/food intake immediately prior to collapse. In accordance with the Royal College of Pathologists’ guidelines, the pathologist has to carefully observe the body, looking for sites of stinging or biting by invertebrates, food debris, external signs of shock, and other features that may suggest a rapid mechanism of death. The autopsy could then provide more data, such as airway and lung edema. However, these features are typical but not specific; accordingly, they are not enough to sustain a diagnosis of anaphylactic reaction, especially if there is the hypothesis of a crime (i.e., suspected anaphylaxis due to food intake in a restaurant in a person who has well-known allergies).
An important role in the diagnosis of anaphylaxis is given to laboratory investigations. The serum tryptase concentrations, for example, have been studied as a marker of the release of great and systemic mast cells’ mediators. However, increased levels of tryptase may occur in some events other than anaphylaxis, such as multiple traumas, asphyxia, myocardial infarction, heroine intoxication, systemic mastocytosis, and hypereosinophilic parasitosis [4]. In addition, despite the lack of exhaustive studies on the real post-mortem stability of IgE immunoglobulins, some authors consider the assay on total and specific IgE serum levels (RAST, ELISA procedures) to be a reliable procedure applicable after death. The problem of the laboratory investigations is the intensity and speed of the cytolytic processes and the chemical degradation that could modify the concentration and stability of analytes.
Since autoptic and laboratory data may be absent or inconclusive, it is important to search for new methods to reach the post-mortem diagnosis of anaphylaxis death. Herein, we provide a systematic literature review to collect the current knowledge about the use of immunohistochemistry (IHC), evaluating the possibility of adding a new methodology that could help in the post-mortem diagnosis of anaphylaxis-related death.
2. Materials and Methods
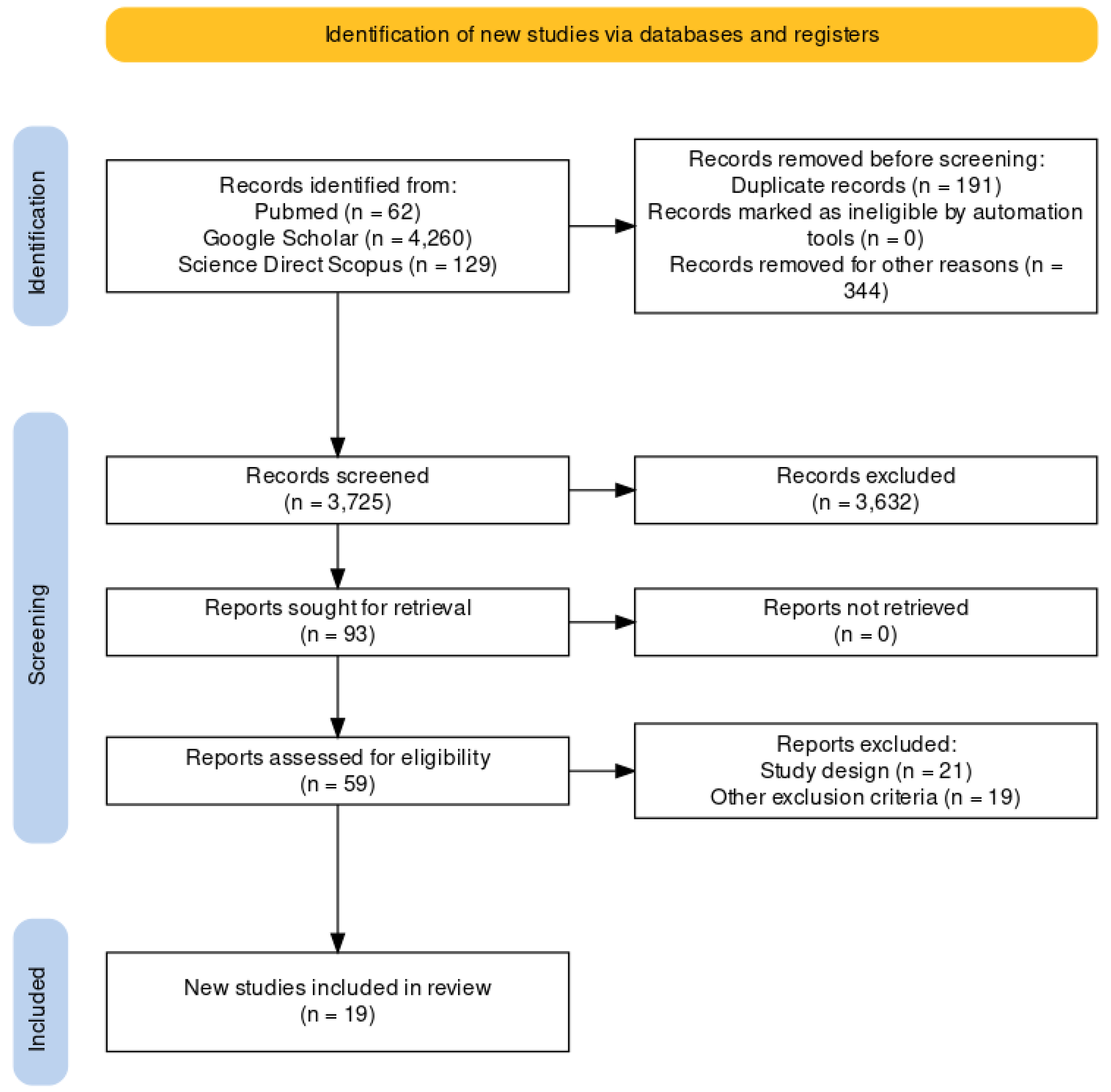
The present systematic review was carried out according to the Preferred Reporting Items for Systematic Review (PRISMA) [5] standards. A methodological appraisal of each study was conducted according to the PRISMA standards, including an evaluation of bias. PRISMA 2020 Statement was applied. It consists of a checklist and a flow diagram (Figure 1).
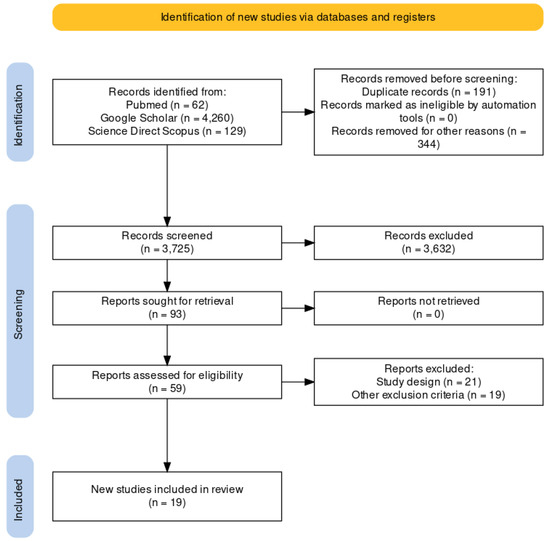
Figure 1.
The selection of papers following PRISMA protocol.
A systematic literature search and critical review of the collected studies were conducted. An electronic search of PubMed, Science Direct Scopus, and Google Scholar from database inception to March 2023 was performed.
Databases were investigated using the following research terms (“anaphylaxis” OR “anaphylactic shock” OR “anaphylactic death” OR “tryptase” OR “anti-tryptase anti-body”) AND (“immunohistochemistry” OR “IHC” OR “immunofluorescence”) AND (“post-mortem” OR “autopsy” OR “forensic”) in all fields [e.g., title, abstract, and keywords].
From this research, a list of abstracts was organized in the form of a dataset and all of the dataset was downloaded in a .nbib file and uploaded to Software Zotero 6-0.30, used as a citation manager.
The research group, following a meeting, established the inclusion and exclusion criteria for the paper, in accordance with the PRISMA standard.
First of all, two investigators (AG and FDD) read all the abstracts found from databases. The bibliographies of all identified papers were examined and cross-referenced to further identify relevant literature.
After selecting abstracts and investigating the bibliographies of related papers, data collection began. One investigator (AG) independently examined papers with titles or abstracts that appeared to be relevant, selecting those that analyzed the use of IHC in anaphylactic death.
The data collection process included study selection and data extraction. Disagreements concerning eligibility among the researchers were resolved by consensus. Preprint articles were excluded, and only papers in English were included.
Data extraction was performed by two investigators (AG and FDD) and verified by additional investigators (AM, ACM, and FDD).
This study was exempt from institutional review board approval, as it did not involve human subjects.
3. Results
A review of the titles and abstracts, as well as a manual search of the reference lists, was carried out. The reference lists of all identified articles were reviewed to find missed literature. This search identified 93 articles, which were then screened based on their abstract. The resulting 93 papers were screened to exclude duplicates, which left 59 articles for further consideration. Non-English papers were excluded [n° 344], and the following inclusion criteria were used: (1) original research articles, (2) reviews and mini-reviews, and (3) case reports/series. These publications were carefully evaluated, considering the main aims of the review. This evaluation left 19 scientific papers comprising original research articles, case reports, and case series (see Figure 1).
We found articles about serum analytes (such as tryptase) and IHC with anti-tryptase antibody (AB), anti-CD117 and anti-chymase. Because of the specific aim of this work, only papers concerning IHC have been included in the systematic review. The selection of appropriate scientific papers was performed; nineteen articles met the inclusion criteria and were included.
3.1. The Study Sample
The study sample, considering all the scientific papers analyzed, comprised 162 cases of anaphylactic death (medium age 45.34—DS 17.24). In Table 1, we provide a summary of the main characteristics of the included studies, while Table 2 and Table 3 show the distribution of our studies per type of allergen.

Table 1.
Main characteristics of the included studies.

Table 2.
Distribution of studies per type of allergen.

Table 3.
Apten distribution shows a predominant role of drugs.
In accordance with the different patterns of positivity of the ABs used to perform the IHC, the included cases have been divided into different groups. The anti-tryptase ABs provided positive results in all the cases; the anti-chymase ABs were positive in 24 up to 162 cases (14.8%) and anti-cd117 Abs were positive in 4/162 cases (2.5%).
Below, we provide a brief description of the immunohistochemical findings which we collected from the included studies, divided per type of AB.
3.2. Anti-Tryptase Antibody
The immunohistochemical pattern with anti-tryptase ABs shows a positivity in the lung tissue (see Table 4). Tryptase indicates the presence of mast cells. We found mast cells in the bronchial walls and in the vicinity of alveoli and capillaries; in some cases the connective interstitium and the pulmonary epithelial cells also resulted positive to these ABs. In the glottis and in the laryngeal wall, the presence of mast cells is also evident. In the minority of cases there were positive findings in myocardial tissue, spleen and stomach, jejunum intestinal tissue, and skin.

Table 4.
Main findings per anti-tryptase.
3.3. Anti-Chimase Antibody
These kinds of ABs present the same patterns as those of the anti-tryptase ABs in the pulmonary tissue with positivity at the site of the bronchial walls and in the vicinity of alveoli and capillaries (Table 5). Despite being less represented in all the cases, in some cases, they are present in the myocardial tissue and in the spleen.

Table 5.
Main findings per anti-chimase.
3.4. Anti-CD117 Antibody
Anti-CD117 ABs are less represented than the anti-tryptase ones, but their pattern of positivity is similar (Table 6). Indeed, in the lung tissue, the typical shapes are starry-like and yard-like. In the laryngeal wall, beyond the typical pattern, we can find positivity in cells with dendritic morphology and in small lymphocytes. Moreover, there is positivity in the myocardium and in the intestinal mucosa.

Table 6.
Main findings per anti-CD117.
4. Discussion
In this study, it was evident that antibiotics, cephalosporins above all, are the main cause of anaphylaxis; contrast agents and other drugs also have an important role. Considering all the 19 articles, in 9 of them the cause of the anaphylaxis was due to antibiotics. Blumentahl et al. underlined that adverse drug reactions account for more than 3% of hospital admissions and complicate the hospitalization of 10–20% of patients [25]. Antibiotics are the commonest cause of life-threatening immune-mediated drug reactions that are considered off target, including anaphylaxis, organ-specific adverse reactions, and severe cutaneous adverse reactions [26].
Our results show that ABs against tryptase, cd117, and chymase (mediators of anaphylaxis) are typically positive in cases of anaphylactic death, indicating the presence of mast cells. The tissue most frequently involved is the lung. Sometimes there is also a halo of positiveness around the cells after degranulation. On microscopical observation, we can find the typical starry-like and yard-like patterns.
Anaphylaxis is considered a systemic, life-threatening disorder triggered by mediators released by mast cells and basophils, activated via allergic (IgE-mediated) or nonallergic (non-IgE-mediated) mechanisms. Hypovolemia and distributive shock usually occur in severe anaphylaxis, and this is known by the historically termed “empty heart syndrome”. The incidence of anaphylaxis is probably underestimated. Publications from the last few years reveal an incidence of from 50 to 103 episodes per 100,000 persons/year. In the United States and United Kingdom, the mortality rate has been estimated to be less than 1 per million. According to the ICD-10, anaphylactic shock occurs as a response to allergen exposure (adverse food reaction, due to serum, adverse effect of drug or medication, possible Hymenoptera venom anaphylaxis events), which leads to the activation of mast cells and basophils, although in some cases, the cause remains unknown [27].
An analytical study of data in the US population shows that there were 2229 anaphylaxis-related deaths between 1999 and 2009 (0.69 per million population). The annual number of deaths ranged from 186 to 225, corresponding to mortality rates of between 0.63 and 0.76 per million. Of all anaphylaxis-related deaths, 75% were due to anaphylactic shock. Overall, 87% of the deaths occurred in a medical facility (inpatient, outpatient/ED, or dead on arrival), and approximately 7% occurred at home [28].
The typical triggers in IgE-mediated anaphylaxis are food; airborne allergens such as pollen, animal dander, aerosolized foods; latex; medications (oral or parenteral), food-dependent exercise-induced anaphylaxis; allergy to mammalian sugars such as galactose-1,3-alpha-galactose (alpha-gal); seminal fluid and hormones, radiocontrast media reactions [29,30], venom, and anesthetic drugs [31]. In the case of non-IgE-mediated anaphylaxis the typical triggers are IV immunoglobulins, NSAIDs (nonsteroidal anti-inflammatory drugs) [32], dialysis membranes, dextrans, iron, biological agents, and heparin.
According to LoVerde et al., clinically, the respiratory tract and the lungs are mostly implicated in cases of anaphylaxis (the typical finding is laryngeal edema). In fact, considering our data, we have noticed that all the antibodies are primarily positive in the lungs and in the laryngeal wall [33]. Death from anaphylaxis usually results from a combination of factors including upper airway obstruction from mucosal edema, asphyxia from bronchospasm, and shock due to massive fluid shifts [34].
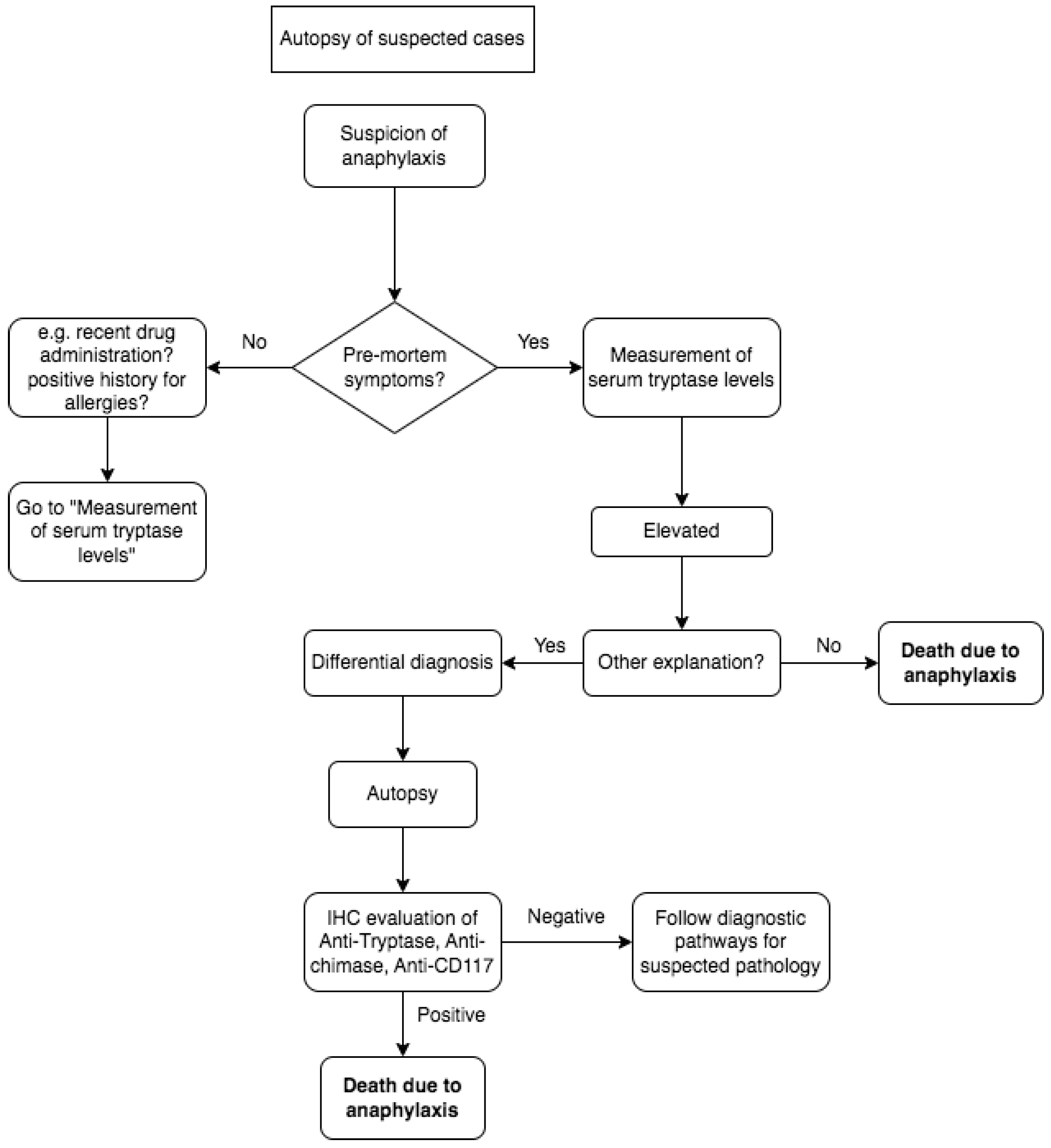
In accordance with the main interpretation of anaphylaxis pathophysiology, mast cells and basophils are the primary effector cells in allergies, directly responding to allergen challenges through immunoglobulin-dependent or independent mechanisms. Upon activation, mast cells and basophils release three major groups of proinflammatory mediators, causing pathological damage that can lead to death. The lethal effects of allergies occur after the activation of mast cells or basophils. Theoretically, the identification of degranulation markers in mast cells and basophils is a definitive event in allergies, and finding them seems to be helpful in the post-mortem diagnosis of anaphylactic death [35,36]. Molecular methods of investigation could be helpful, as is the use of IHC. Our investigation reveals upper airway findings indicating positivity in the immunohistochemical evaluation of anti-tryptase, anti-chimase, and anti-CD117 Abs Tryptase which is consistently observed in all lung tissue samples, particularly in atopic asthmatic subjects, aiding in the identification of mast cells. Conversely, the presence of vacuolated mast cells appears to be indicative of degranulation in cases of anaphylaxis. Notably, certain samples exhibit anti-chymase-positive mast cells in perivascular spaces, displaying a similar pattern [8,14]. The assessment of the laryngeal wall and glottis includes an examination of the Tryptase and CD117 expression patterns, revealing the involvement of perivascular zones with vacuolized mast cells [12,23]. The diagnosis of anaphylaxis-related death is demanded in crime scene investigation, using information from anmnesis and circumstantial data, followed by tryptase serum dosage, and is eventually confirmed by immunohistochemical analysis, as shown in Figure 2.
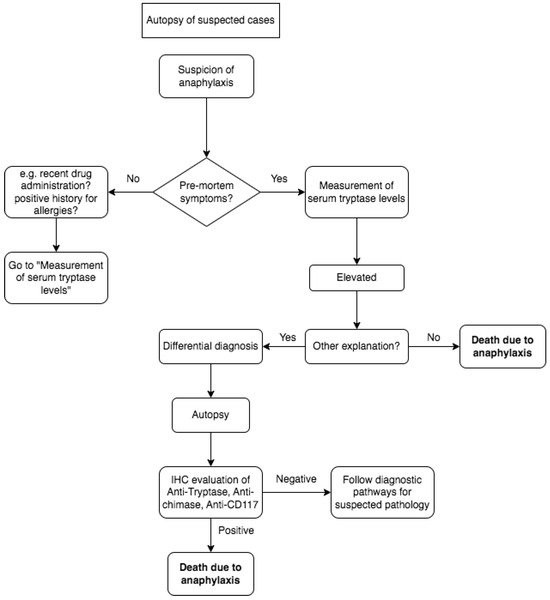
Figure 2.
The diagnosis of anaphylaxis-related death in a suspected case is conducted by performing serum confirmation and/or immunohistochemical analysis, as shown in the flowchart.
In fact, as shown in the results, while anti-tryptase in lung tissues appears to be the most reliable marker, on the other hand, it is too general. So, as found in the literature review, an association with other markers such as anti-chymase and anti-CD117 is required to confirm the diagnosis. These two IHC markers of anaphylaxis seem to be not specific but have a good rate of sensitivity. Further studies are needed to confirm this hypothesis.
There are, however, numerous problems that arise in establishing the diagnosis of anaphylaxis at autopsy, as no typical macroscopic features are present in 41% of cases at autopsy. To corroborate the diagnosis, it could be useful to conduct a histological study of the laryngeal mucosa [3]. Moreover, to reach the diagnosis, an important role is given to the value of serum tryptase, which is considered the most widely used biomarker to confirm a diagnosis of anaphylaxis retrospectively [37]. On the other hand, there is the problem that serum histamine and tryptase levels are not always elevated [38], even in patients with severe manifestations of anaphylaxis including cutaneous, gastrointestinal, respiratory, or cardiovascular compromise [39].
A limit of the present study is the small sample size, due to the lack of articles about this topic. IHC has been relatively recently introduced in forensic investigations, and most of the included papers have been published in the last ten years. Moreover, this is a very targeted topic. Further studies are needed to evaluate how valid IHC could be in the demonstration of fatal anaphylactic shock.
5. Conclusions
Considering all the studies analyzed in this review and the fact that reaching a diagnosis of anaphylactic death could be an issue, IHC represents a fundamental tool for the pathologist. In fact, there are many cases where there is no possibility to perform laboratory tests in search for serum tryptase, or they are useless. Indeed, tryptase is not always elevated in anaphylaxis. Our work underlines the importance of some anaphylaxis mediators such as tryptase, cd117, and chymase in the immunohistochemical analysis in such kind of death. In accordance with the analyzed papers, the diagnosis of death due to anaphylaxis should be based not only on the incidence of anaphylactic reaction, but on the proof of the presence of these inflammatory mediators with IHC, especially in the respiratory tract. Even if further evidence needs to be collected, all the professionals involved in such investigations should be aware of the importance of IHC as a new reliable tool to reach the post-mortem diagnosis of anaphylaxis.
Author Contributions
Conceptualization, A.M., B.G. and F.D.D.; methodology, R.L.R.; software, A.C.M.; validation, F.P., B.G., A.M. and A.G.; formal analysis, A.C.M.; investigation, N.P.; resources, F.D.D.; data curation, F.D.D.; writing—original draft preparation, A.G. and G.N.; writing—review and editing, F.D.D.; visualization, R.L.R. and G.N.; supervision, A.M.; project administration, A.M.; funding acquisition, F.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lieberman, P.; Nicklas, R.A.; Oppenheimer, J.; Kemp, S.F.; Lang, D.M.; Bernstein, D.I.; Bernstein, J.A.; Burks, A.W.; Feldweg, A.M.; Fink, J.N.; et al. The Diagnosis and Management of Anaphylaxis Practice Parameter: 2010 Update. J. Allergy Clin. Immunol. 2010, 126, 477–480.e42. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.G.A.; Mullins, R.J.; Gold, M.S. 2. Anaphylaxis: Diagnosis and Management. Med. J. Aust. 2006, 185, 400. [Google Scholar] [CrossRef] [PubMed]
- Pumphrey, R.S.H. Postmortem Findings after Fatal Anaphylactic Reactions. J. Clin. Pathol. 2000, 53, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Da Broi, U.; Moreschi, C. Post-Mortem Diagnosis of Anaphylaxis: A Difficult Task in Forensic Medicine. Forensic Sci. Int. 2011, 204, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. Int. J. Surg. 2021, 88, 105906. [Google Scholar] [CrossRef] [PubMed]
- Fineschi, V.; Cecchi, R.; Centini, F.; Reattelli, L.P.; Turillazzi, E. Immunohistochemical Quantication of Pulmonary Mast-Cells and Post-Mortem Blood Dosages of Tryptase and Eosinophil Cationic Protein in 48 Heroin-Related Deaths. Forensic Sci. Int. 2001, 120, 189–194. [Google Scholar] [CrossRef]
- Edston, E.; Gidlund, E.; Wickman, M.; Ribbing, H.; Hage-Hamsten, M.V. Increased Mast Cell Tryptase in Sudden Infant Death Ð Anaphylaxis, Hypoxia or Artefact? Clin. Exp. Allergy 1999, 29, 1648–1654. [Google Scholar] [CrossRef]
- Perskvist, N.; Edston, E. Differential Accumulation of Pulmonary and Cardiac Mast Cell-Subsets and Eosinophils between Fatal Anaphylaxis and Asthma Death. Forensic Sci. Int. 2007, 169, 43–49. [Google Scholar] [CrossRef]
- Osawa, M.; Satoh, F.; Horiuchi, H.; Tian, W.; Kugota, N.; Hasegawa, I. Postmortem Diagnosis of Fatal Anaphylaxis during Intravenous Administration of Therapeutic and Diagnostic Agents: Evaluation of Clinical Laboratory Parameters and Immunohistochemistry in Three Cases. Leg. Med. 2008, 10, 143–147. [Google Scholar] [CrossRef]
- Turillazzi, E.; Greco, P.; Neri, M.; Pomara, C.; Riezzo, I.; Fineschi, V. Anaphylactic Latex Reaction during Anaesthesia: The Silent Culprit in a Fatal Case. Forensic Sci. Int. 2008, 179, e5–e8. [Google Scholar] [CrossRef]
- Trani, N.; Bonetti, L.R.; Gualandri, G.; Barbolini, G. Immediate Anaphylactic Death Following Antibiotics Injection: Splenic Eosinophilia Easily Revealed by Pagoda Red Stain. Forensic Sci. Int. 2008, 181, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Unkrig, S.; Hagemeier, L.; Madea, B. Postmortem Diagnostics of Assumed Food Anaphylaxis in an Unexpected Death. Forensic Sci. Int. 2010, 198, e1–e4. [Google Scholar] [CrossRef] [PubMed]
- Luongo, S.; Frontalini, C.; Pesaresi, M.; Valsecchi, M.; Tagliabracci, A. Histopathological Markers for the Diagnosis of Anaphylactic Death. Med. Sci. Law 2011, 51, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Edston, E. Accumulation of Eosinophils, Mast Cells, and Basophils in the Spleen in Anaphylactic Deaths. Forensic Sci. Med. Pathol. 2013, 9, 496–500. [Google Scholar] [CrossRef] [PubMed]
- Comment, L.; Reggiani Bonetti, L.; Mangin, P.; Palmiere, C. Measurement of β-Tryptase in Postmortem Serum, Pericardial Fluid, Urine and Vitreous Humor in the Forensic Setting. Forensic Sci. Int. 2014, 240, 29–34. [Google Scholar] [CrossRef]
- Reggiani Bonetti, L.; Maccio, L.; Trani, N.; Radheshi, E.; Palmiere, C. Splenic Hypereosinophilia in Anaphylaxis-Related Death: Different Assessments Depending on Different Types of Allergens? Int. J. Leg. Med. 2015, 129, 97–103. [Google Scholar] [CrossRef]
- Guo, X.-J. Mast Cell Tryptase and Carboxypeptidase A Expression in Body Fluid and Gastrointestinal Tract Associated with Drug-Related Fatal Anaphylaxis. WJG 2015, 21, 13288. [Google Scholar] [CrossRef]
- Radheshi, E.; Reggiani Bonetti, L.; Confortini, A.; Silingardi, E.; Palmiere, C. Postmortem Diagnosis of Anaphylaxis in Presence of Decompositional Changes. J. Forensic Leg. Med. 2016, 38, 97–100. [Google Scholar] [CrossRef]
- Takahashi, M.; Kondo, T.; Morichika, M.; Kuse, A.; Nakagawa, K.; Ueno, Y. Postmortem Detection of Antibiotic-Specific Immunoglobulin E in the Case of Anaphylactic Death. Forensic Sci. Int. 2016, 266, 14–17. [Google Scholar] [CrossRef]
- Wang, X.; Yin, C.; Su, X.; Su, M. Reliable Postmortem Molecular Diagnosis of Anaphylaxis: Co-Localization of Mast Cell Degranulation and Immunoglobulin E in Allergic Throat Tissues. Am. J. Forensic Med. Pathol. 2020, 41, 249–258. [Google Scholar] [CrossRef]
- D’Errico, S.; Frati, P.; Zanon, M.; Valentinuz, E.; Manetti, F.; Scopetti, M.; Santurro, A.; Fineschi, V. Cephalosporins’ Cross-Reactivity and the High Degree of Required Knowledge. Case Report and Review of the Literature. Antibiotics 2020, 9, 209. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Montana, A.; Liberto, A.; Filetti, V.; Nunno, N.D.; Amico, F.; Salerno, M.; Loreto, C.; Sessa, F. Anaphylactic Death: A New Forensic Workflow for Diagnosis. Healthcare 2021, 9, 117. [Google Scholar] [CrossRef] [PubMed]
- Tambuzzi, S.; Gentile, G.; Boracchi, M.; Di Candia, D.; Bianchi, R.; Zoja, R. Postmortem Diagnostics of Assumed Suicidal Food Anaphylaxis in Prison: A Unique Case of Anaphylactic Death Due to Peach Ingestion. Forensic Sci. Med. Pathol. 2021, 17, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Zhang, C.; Wang, P.; Wang, X.; You, J.; Zhang, G.; Yu, H. Expression of FcεRIα and Tryptase in Human Lung Tissue during Drug-Induced Anaphylactic Death. Forensic Sci. Med. Pathol 2021, 17, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, K.G.; Kuhlen, J.L., Jr.; Weil, A.A.; Varughese, C.A.; Kubiak, D.W.; Banerji, A.; Shenoy, E.S. Adverse Drug Reactions Associated with Ceftaroline Use: A 2-Center Retrospective Cohort. J. Allergy Clin. Immunol. Pract. 2016, 4, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, K.G.; Peter, J.G.; Trubiano, J.A.; Phillips, E.J. Antibiotic Allergy. Lancet 2019, 393, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Bilò, M.B.; Corsi, A.; Martini, M.; Penza, E.; Grippo, F.; Bignardi, D. Fatal Anaphylaxis in Italy: Analysis of Cause-of-death National Data, 2004-2016. Allergy 2020, 75, 2644–2652. [Google Scholar] [CrossRef]
- Ma, L.; Danoff, T.M.; Borish, L. Case Fatality and Population Mortality Associated with Anaphylaxis in the United States. J. Allergy Clin. Immunol. 2014, 133, 1075–1083. [Google Scholar] [CrossRef]
- Järvinen, K.M. Food-Induced Anaphylaxis. Curr. Opin. Allergy Clin. Immunol. 2011, 11, 255–261. [Google Scholar] [CrossRef]
- Iweala, O.I.; Burks, A.W. Food Allergy: Our Evolving Understanding of Its Pathogenesis, Prevention, and Treatment. Curr. Allergy Asthma Rep. 2016, 16, 37. [Google Scholar] [CrossRef]
- Garvey, L. Old, New and Hidden Causes of Perioperative Hypersensitivity. CPD 2017, 22, 6814–6824. [Google Scholar] [CrossRef] [PubMed]
- Dona, I.; Salas, M.; Perkins, J.R.; Barrionuevo, E.; Gaeta, F.; Cornejo-Garcia, J.A.; Campo, P.; Torres, M.J. Hypersensitivity Reactions to Non-Steroidal Anti-Inflammatory Drugs. Curr. Pharm. Des. 2016, 22, 6784–6802. [Google Scholar] [CrossRef] [PubMed]
- LoVerde, D.; Iweala, O.I.; Eginli, A.; Krishnaswamy, G. Anaphylaxis. Chest 2018, 153, 528–543. [Google Scholar] [CrossRef] [PubMed]
- Byard, R.W. Anaphylaxis at Autopsy. Forensic Sci. Med. Pathol. 2017, 13, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Marone, G.; Triggiani, M.; Genovese, A.; Paulis, A.D. Role of Human Mast Cells and Basophils in Bronchial Asthma. In Advances in Immunology; Elsevier: Amsterdam, The Netherlands, 2005; Volume 88, pp. 97–160. ISBN 978-0-12-022488-3. [Google Scholar]
- Pettipher, R.; Hansel, T.T.; Armer, R. Antagonism of the Prostaglandin D2 Receptors DP1 and CRTH2 as an Approach to Treat Allergic Diseases. Nat. Rev. Drug Discov. 2007, 6, 313–325. [Google Scholar] [CrossRef]
- Waterfield, T.; Dyer, E.; Wilson, K.; Boyle, R.J. How to Interpret Mast Cell Tests. Arch. Dis. Child. Educ. Pract. 2016, 101, 246–251. [Google Scholar] [CrossRef]
- Vadas, P.; Liss, G.M.; Simons, F.E.R.; Yeung, J. Platelet-Activating Factor, PAF Acetylhydrolase, and Severe Anaphylaxis. N. Engl. J. Med. 2008, 358, 28–35. [Google Scholar] [CrossRef]
- Burks, A.W.; Jones, S.M.; Boyce, J.A.; Sicherer, S.H.; Wood, R.A.; Assa’ad, A.; Sampson, H.A. NIAID-Sponsored 2010 Guidelines for Managing Food Allergy: Applications in the Pediatric Population. Pediatrics 2011, 128, 955–965. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).