Effect of Herpes Zoster Treatment and Sudden Sensorineural Hearing Loss Using National Health Insurance Claims Data of South Korea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population and Data Collection
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dayan, R.R.; Peleg, R. Herpes zoster—Typical and atypical presentations. Postgrad. Med. 2017, 129, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.H.; Mun, K.H.; Kim, G.W.; Lee, C.S.; Cha, Y.D. Recurrence of herpes zoster in a young woman with IgG3 deficiency. Korean J. Anesthesiol. 2015, 68, 622–623. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.H.; Na, J.I.; Kim, I.S.; Byun, S.Y.; Park, K.C. Population-Based Epidemiologic Study on Herpes Zoster in Korea: Seven-Year Nationwide Analysis from 2010 to 2016. Ann. Dermatol. 2019, 31, 486–488. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.D.; Kang, B.G.; Kim, J.H.; Roh, M.; Jo, D.H. Abdominal distention and constipation followed by herpes zoster infection. Korean J. Anesthesiol. 2013, 65, S143–S144. [Google Scholar] [CrossRef] [PubMed]
- Cohen, B.E.; Durstenfeld, A.; Roehm, P.C. Viral causes of hearing loss: A review for hearing health professionals. Trends Hear. 2014, 29, 18. [Google Scholar] [CrossRef]
- Rossini, B.A.A.; Penido, N.O.; Munhoz, M.S.L.; Bogaz, E.A.; Curi, R.S. Sudden Sensorioneural Hearing Loss and Autoimmune Systemic Diseases. Int. Arch. Otorhinolaryngol. 2017, 21, 213–223. [Google Scholar] [CrossRef]
- Kim, C.H.; Choi, H.; Shin, J.E. Characteristics of hearing loss in patients with herpes zoster oticus. Medicine 2016, 95, e5438. [Google Scholar] [CrossRef]
- Mishra, B.; Panda, N.; Singh, M.P.; Ratho, R.K. Viral infections in sudden hearing loss. Do we have enough evidence? Kathmandu Univ. Med. J. KUMJ 2005, 3, 230–233. [Google Scholar]
- Sheu, J.J.; Keller, J.J.; Chen, Y.H.; Wu, C.S.; Lin, H.C. No increased risk of sudden sensorineural hearing loss following recent herpes zoster: A nationwide population-based study. Acta Oto-Laryngol. 2012, 132, 167–172. [Google Scholar] [CrossRef]
- Arvin, A. Aging, immunity, and the varicella-zoster virus. N. Engl J. Med. 2005, 352, 2266–2267. [Google Scholar] [CrossRef]
- Harpaz, R.; Ortega-Sanchez, I.R.; Seward, J.F. Prevention of herpes zoster: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 2008, 57, 1–30, quiz CE32-34. [Google Scholar]
- Liesegang, T.J. Herpes zoster ophthalmicus natural history, risk factors, clinical presentation, and morbidity. Ophthalmology 2008, 115, S3–S12. [Google Scholar] [CrossRef]
- Yawn, B.P.; Saddier, P.; Wollan, P.C.; St Sauver, J.L.; Kurland, M.J.; Sy, L.S. A population-based study of the incidence and complication rates of herpes zoster before zoster vaccine introduction. Mayo Clin. Proc. 2007, 82, 1341–1349. [Google Scholar] [CrossRef]
- Kawai, K.; Gebremeskel, B.G.; Acosta, C.J. Systematic review of incidence and complications of herpes zoster: Towards a global perspective. BMJ Open 2014, 4, e004833. [Google Scholar] [CrossRef]
- Nguyen, H.Q.; Jumaan, A.O.; Seward, J.F. Decline in mortality due to varicella after implementation of varicella vaccination in the United States. N. Engl. J. Med. 2005, 352, 450–458. [Google Scholar] [CrossRef]
- Davis, M.M.; Patel, M.S.; Gebremariam, A. Decline in varicella-related hospitalizations and expenditures for children and adults after introduction of varicella vaccine in the United States. Pediatrics 2004, 114, 786–792. [Google Scholar] [CrossRef]
- Gershon, A.A.; Breuer, J.; Cohen, J.I.; Cohrs, R.J.; Gershon, M.D.; Gilden, D.; Grose, C.; Hambleton, S.; Kennedy, P.G.; Oxman, M.N.; et al. Varicella zoster virus infection. Nat. Rev. Dis. Primers. 2015, 1, 15016. [Google Scholar] [CrossRef]
- Rauch, S.D. Clinical practice. Idiopathic sudden sensorineural hearing loss. N. Engl. J. Med. 2008, 359, 833–840. [Google Scholar] [CrossRef]
- Choi, J.K.; Park, S.H.; Park, S.; Cho, S.Y.; Lee, H.J.; Kim, S.H.; Choi, S.M.; Lee, D.G.; Choi, J.H.; Yoo, J.H. The changing epidemiology of herpes zoster over a decade in South Korea, 2006–2015. Vaccine 2019, 37, 5153–5160. [Google Scholar] [CrossRef]
- Min, B.M.; Kim, J.H. Epidural catheterization with a subcutaneous injection port for the long-term administration of opioids and local anesthetics to treat zoster-associated pain -a report of two cases. Korean J. Anesthesiol. 2013, 65, 462–467. [Google Scholar] [CrossRef]
- Jung, W.; Lee, B.J.; Kim, S.S.; Lee, Y.J. The effect of Gunn’s intramuscular stimulation for postherpetic neuralgia -A report of 4 cases. Korean J. Anesthesiol. 2010, 58, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, M.; Heman-Ackah, S.E.; Shaikh, J.A.; Roehm, P.C. Sudden sensorineural hearing loss: A review of diagnosis, treatment, and prognosis. Trends Amplif. 2011, 15, 91–105. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Gonzalez, M.A.; Abrante, A.; Lopez-Lorente, C.; Gomez, A.; Dominguez, E.; Esteban, F. Acute-phase inflammatory response in idiopathic sudden deafness: Pathogenic implications. Int. J. Otolaryngol. 2012, 2012, 216592. [Google Scholar] [CrossRef] [PubMed]
- Chau, J.K.; Lin, J.R.; Atashband, S.; Irvine, R.A.; Westerberg, B.D. Systematic review of the evidence for the etiology of adult sudden sensorineural hearing loss. Laryngoscope 2010, 120, 1011–1021. [Google Scholar] [CrossRef]
- Chen, X.; Fu, Y.Y.; Zhang, T.Y. Role of viral infection in sudden hearing loss. J. Int. Med. Res. 2019, 47, 2865–2872. [Google Scholar] [CrossRef]
- Greco, A.; Fusconi, M.; Gallo, A.; Marinelli, C.; Macri, G.F.; De Vincentiis, M. Sudden sensorineural hearing loss: An autoimmune disease? Autoimmun. Rev. 2011, 10, 756–761. [Google Scholar] [CrossRef]
- Veltri, R.W.; Wilson, W.R.; Sprinkle, P.M.; Rodman, S.M.; Kavesh, D.A. The implication of viruses in idiopathic sudden hearing loss: Primary infection or reactivation of latent viruses? Otolaryngol. Head Neck Surg. 1981, 89, 137–141. [Google Scholar] [CrossRef]
- Kuczkowski, J.; Kozlowski, J.; Narozny, W. Acute autoimmune sensorineural hearing loss in pregnant women with Lesniowski-Crohn disease. Otolaryngol. Pol. 2006, 60, 583–585. [Google Scholar]
- Yehudai, D.; Shoenfeld, Y.; Toubi, E. The autoimmune characteristics of progressive or sudden sensorineural hearing loss. Autoimmunity 2006, 39, 153–158. [Google Scholar] [CrossRef]
- Garcia Callejo, F.J.; Marco Algarra, J.; Martinez Beneyto, M.P.; Orts Alborch, M.H.; Morant Ventura, A. Autoimmune identification of sudden hearing loss. Acta Oto-Laryngol. 2003, 123, 168–171. [Google Scholar] [CrossRef]
- Psillas, G.; Arnaoutoglou, M.; Gatsios, T.; Rizos, D.; Koutsouraki, E.; Vital, V. Autoimmune recurrent facial palsy and bilateral sudden sensorineural hearing loss following Ramsay Hunt-like syndrome. Auris Nasus Larynx 2012, 39, 229–232. [Google Scholar] [CrossRef]
- Westerlaken, B.O.; Stokroos, R.J.; Dhooge, I.J.; Wit, H.P.; Albers, F.W. Treatment of idiopathic sudden sensorineural hearing loss with antiviral therapy: A prospective, randomized, double-blind clinical trial. Ann. Otol. Rhinol. Laryngol. 2003, 112, 993–1000. [Google Scholar] [CrossRef]
- Toubi, E.; Ben-David, J.; Kessel, A.; Halas, K.; Sabo, E.; Luntz, M. Immune-mediated disorders associated with idiopathic sudden sensorineural hearing loss. Ann. Otol. Rhinol. Laryngol. 2004, 113, 445–449. [Google Scholar] [CrossRef]
- Wollina, U. Variations in herpes zoster manifestation. Indian J. Med. Res. 2017, 145, 294–298. [Google Scholar] [CrossRef]
- Lee, H.L.; Yeo, M.; Choi, G.H.; Lee, J.Y.; Kim, J.S.; Shin, D.I.; Lee, S.S.; Lee, S.H. Clinical characteristics of headache or facial pain prior to the development of acute herpes zoster of the head. Clin. Neurol. Neurosurg. 2017, 152, 90–94. [Google Scholar] [CrossRef]
- Shin, D.H.; Kim, B.R.; Shin, J.E.; Kim, C.H. Clinical manifestations in patients with herpes zoster oticus. Eur. Arch. Otorhinolaryngol. 2016, 273, 1739–1743. [Google Scholar] [CrossRef]
- Park, S.M.; Han, C.; Lee, J.W.; Kong, T.H.; Seo, Y.J. Does Herpes Virus Reactivation Affect Prognosis in Idiopathic Sudden Sensorineural Hearing Loss? Clin. Exp. Otorhinolaryngol. 2017, 10, 66–70. [Google Scholar] [CrossRef]
- Stokroos, R.J.; Albers, F.W.; Schirm, J. Therapy of idiopathic sudden sensorineural hearing loss: Antiviral treatment of experimental herpes simplex virus infection of the inner ear. Ann. Otol. Rhinol. Laryngol. 1999, 108, 423–428. [Google Scholar] [CrossRef]
- Chandrasekhar, S.S.; Tsai Do, B.S.; Schwartz, S.R.; Bontempo, L.J.; Faucett, E.A.; Finestone, S.A.; Hollingsworth, D.B.; Kelley, D.M.; Kmucha, S.T.; Moonis, G.; et al. Clinical Practice Guideline: Sudden Hearing Loss (Update). Otolaryngol. Head Neck Surg. 2019, 161, S1–S45. [Google Scholar] [CrossRef]
- Merchant, S.N.; Durand, M.L.; Adams, J.C. Sudden deafness: Is it viral? ORL J. Otorhinolaryngol. Relat. Spec. 2008, 70, 52–60. [Google Scholar] [CrossRef]
- Ovet, G.; Alatas, N.; Kocacan, F.N.; Gurcuoglu, S.S.; Gorgulu, H.; Guzelkara, F.; Ovet, H. Sudden sensorineural hearing loss: Is antiviral treatment really necessary? Am. J. Otolaryngol. 2015, 36, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Park, K.H.; Lee, C.K.; Lee, J.D.; Park, M.K.; Lee, B.D. Combination therapy with systemic steroids, an antiviral agent, anticoagulants, and stellate ganglion block for treatment of sudden sensorineural hearing loss. Korean J. Audiol. 2012, 16, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Oxman, M.N.; Levin, M.J.; Johnson, G.R.; Schmader, K.E.; Straus, S.E.; Gelb, L.D.; Arbeit, R.D.; Simberkoff, M.S.; Gershon, A.A.; Davis, L.E.; et al. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N. Engl. J. Med. 2005, 352, 2271–2284. [Google Scholar] [CrossRef] [PubMed]

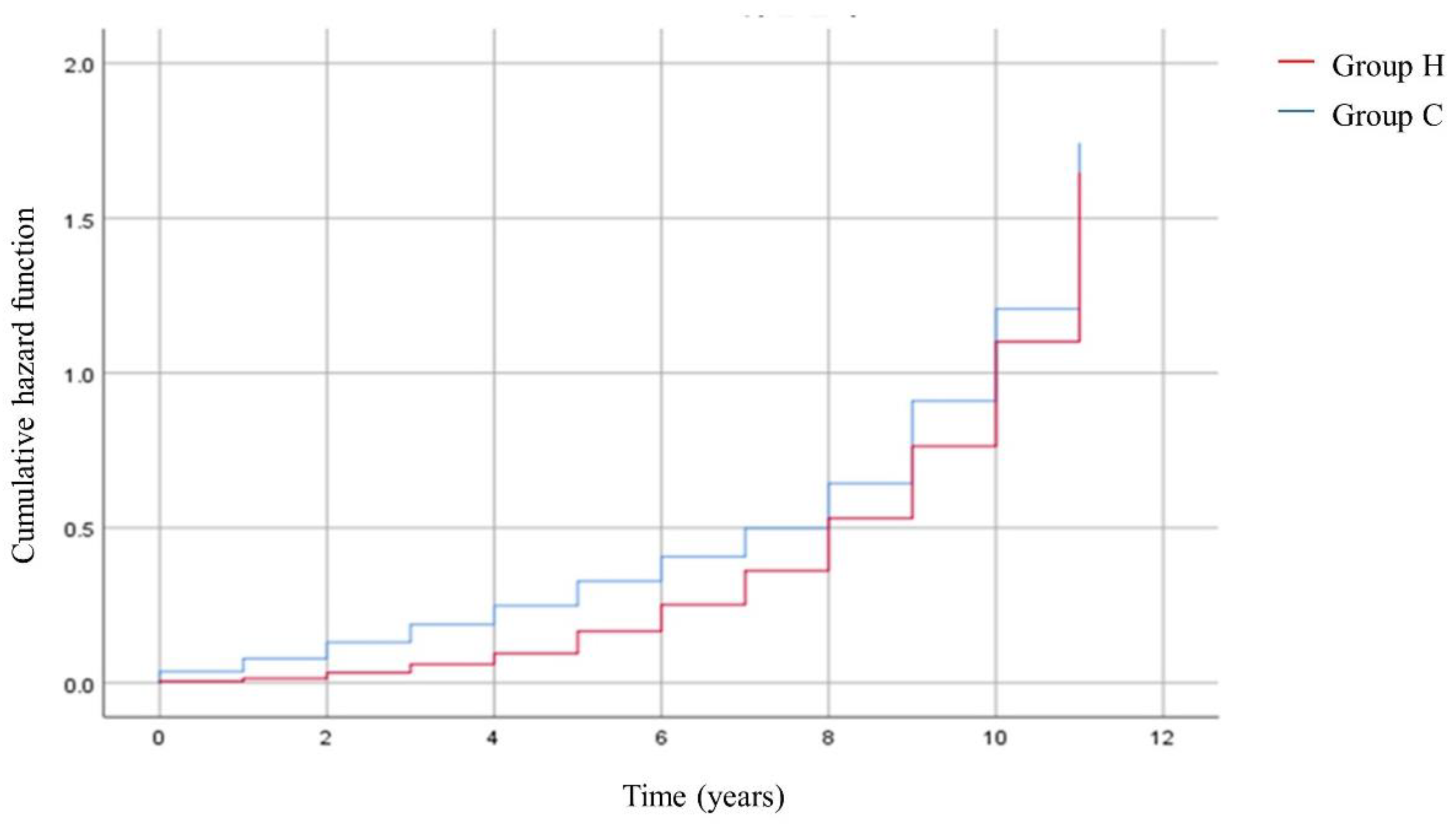
| Group H (n = 35,452) | Group C (n = 35,452) | p Value | |
|---|---|---|---|
| Age (years) | 79.50 ± 5.90 | 79.50 ± 5.90 | 0.999 |
| Sex | |||
| M | 13,500 (38.1) | 13,500 (38.1) | 1.000 |
| F | 21,952 (61.9) | 21,952 (61.9) | |
| Income | |||
| Low | 4285 (12.1) | 4285 (12.1) | 1.000 |
| Middle | 16,316 (46.0) | 16,316 (46.0) | |
| High | 14,851 (41.9) | 14,851 (41.9) | |
| Comorbidities | |||
| Myocardial infarct | 2470 (7.0) | 2224 (6.3) | |
| Cerebrovascular disease | 16,545 (46.7) | 15,005 (42.3) | <0.001 |
| Rheumatic or connective disease (autoimmune disease) | 8163 (23.0) | 6432 (18.1) | <0.001 |
| Diabetes mellitus | 16,693 (47.1) | 14,200 (40.1) | <0.001 |
| Moderate or severe renal failure (chronic renal disease) | 594 (1.7) | 509 (1.4) | 0.010 |
| Otitis media | 8887 (25.1) | 6965 (19.6) | <0.001 |
| Hypertension | 28,688 (80.9) | 26,509 (74.8) | <0.001 |
| SSNHL | 2325 (6.6) | 2143 (6.0) | 0.005 |
| Group | Average (Year) | SE | 95% CI | |
|---|---|---|---|---|
| Lower | Upper | |||
| Group H | 8.703 | 0.059 | 8.588 | 8.818 |
| Group C | 7.773 | 0.078 | 7.620 | 7.927 |
| Overall | 8.257 | 0.049 | 8.161 | 8.353 |
| Group H | Group C | IRR | p Value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | SSNHL | Person-Years | Rate * | n | SSHL | Person-Years | Rate * | |||
| overall | 35,452 | 2325 | 2,715,540 | 8.6 | 35,452 | 2143 | 2,415,324 | 8.9 | 1.085 (1.023–1.151) | 0.006 |
| Year of follow-up | ||||||||||
| <1 | 35,452 | 11 | 35,452 | 3.1 | 35,452 | 75 | 35,452 | 21.2 | 0.147 (0.078–0.276) | 0.002 |
| 1–5 | 35,390 | 345 | 689,031 | 5.0 | 34,326 | 523 | 630,980 | 8.3 | 0.660 (0.576–0.756) | 0.000 |
| 5< | 32,582 | 1969 | 1,991,057 | 9.8 | 28,875 | 1545 | 1,748,892 | 8.8 | 1.274 (1.192–1.362) | 0.000 |
| Variable | Adjusted HR | 95% CI | p Value | |
|---|---|---|---|---|
| Low | high | |||
| Main model | 0.890 | 0.839 | 0.944 | <0.001 |
| Full model | 0.894 | 0.843 | 0.949 | <0.001 |
| Additional covariates | ||||
| Main model + Myocardial infarct | 0.890 | 0.839 | 0.944 | <0.001 |
| Main model + Cerebrovascular disease | 0.889 | 0.838 | 0.943 | <0.001 |
| Main model + Rheumatic or connective disease (autoimmune disease) | 0.891 | 0.840 | 0.945 | <0.001 |
| Main model + diabetes mellitus | 0.891 | 0.841 | 0.945 | <0.001 |
| Main model + Moderate or severe renal failure (chronic renal disease | 0.889 | 0.839 | 0.943 | <0.001 |
| Main model + Otitis media | 0.894 | 0.843 | 0.948 | <0.001 |
| Main model + hypertension | 0.890 | 0.840 | 0.944 | <0.001 |
| Subgroup effects * | ||||
| Sex | ||||
| M | 0.933 | 0.837 | 1.040 | 0.210 |
| F | 0.880 | 0.820 | 0.944 | <.001 |
| Age | ||||
| 60–70 | 0.289 | 0.360 | 2.307 | 0.241 |
| 70–80 | 0.919 | 0.850 | 0.994 | 0.034 |
| 80- | 0.862 | 0.788 | 0.944 | 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Son, H.J.; Choi, E.-J.; Jeong, U.; Choi, Y.J. Effect of Herpes Zoster Treatment and Sudden Sensorineural Hearing Loss Using National Health Insurance Claims Data of South Korea. Medicina 2023, 59, 808. https://doi.org/10.3390/medicina59040808
Son HJ, Choi E-J, Jeong U, Choi YJ. Effect of Herpes Zoster Treatment and Sudden Sensorineural Hearing Loss Using National Health Insurance Claims Data of South Korea. Medicina. 2023; 59(4):808. https://doi.org/10.3390/medicina59040808
Chicago/Turabian StyleSon, Hyo Jung, Eun-Ji Choi, Ukjin Jeong, and Yoon Ji Choi. 2023. "Effect of Herpes Zoster Treatment and Sudden Sensorineural Hearing Loss Using National Health Insurance Claims Data of South Korea" Medicina 59, no. 4: 808. https://doi.org/10.3390/medicina59040808





