The Molecular Role of Immune Cells in Dilated Cardiomyopathy
Abstract
:1. Introduction
2. Etiology of DCM
2.1. Genetic DCM
2.2. Acquired DCM
3. Inflammatory Processes in DCM
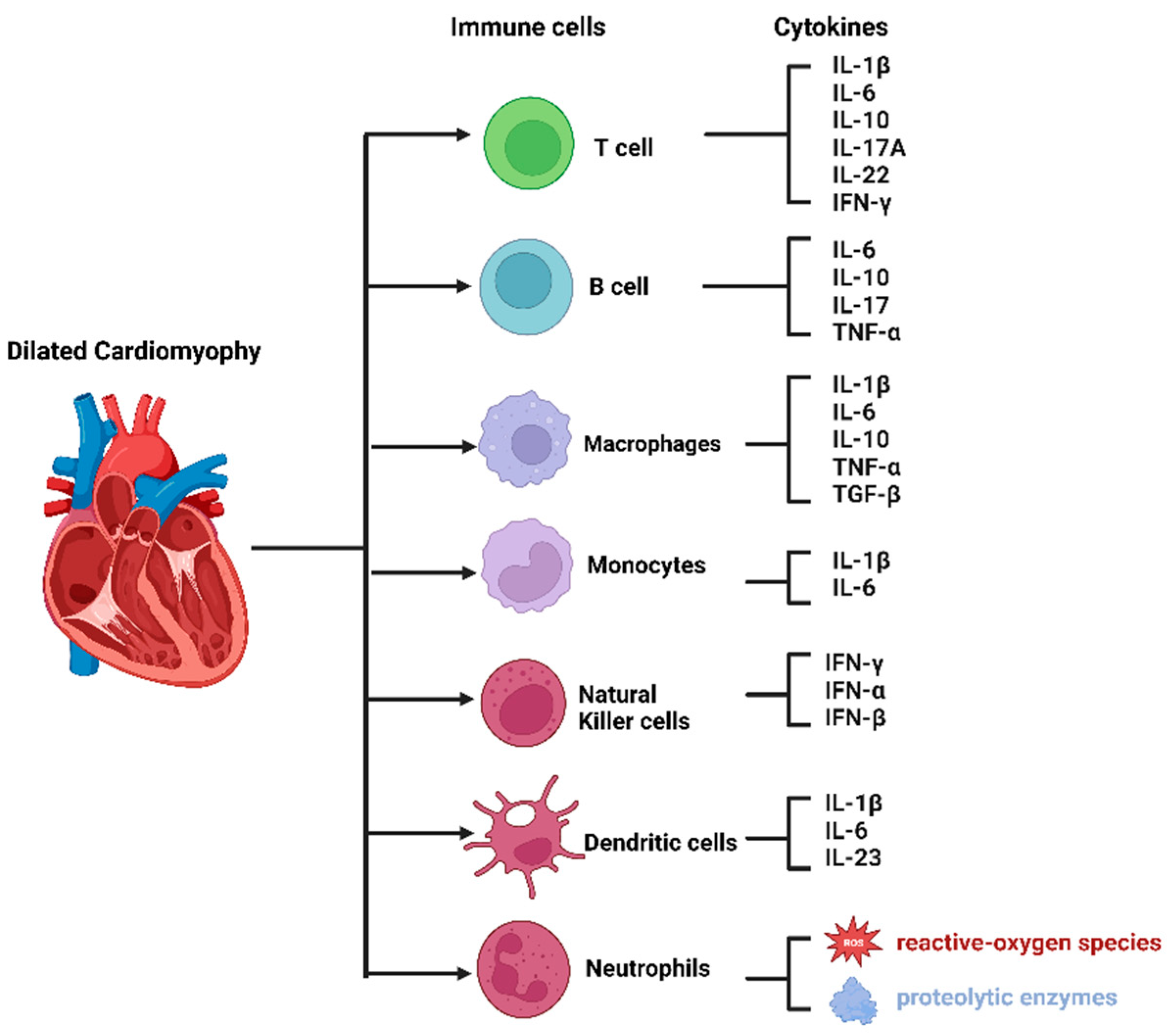
4. The Composition of Immune Cells in the Heart
4.1. Innate Immune Cells
4.2. Adaptive Immune Cells
4.3. Resident Immune Cells in the Heart
4.4. Recruited Immune Cells Mediate Inflammation Responses in the Heart
5. Cellular Communication between Immune Cells and Other Cardiac Cells in DCM
5.1. The Endothelium Participates in the Recruitment of Immune Cells
5.2. The Interplay between Immune Cells and Cardiomyocytes in DCM
5.3. Immune Cells Contribute to Fibrosis in DCM by Activating Myofibroblasts
6. Regulatory Mechanisms of Immune Cells in DCM
6.1. Monocytes and Macrophages in DCM
6.2. T Cells in DCM
6.3. B Cells in DCM
6.4. Natural Killer Cells in DCM
6.5. Neutrophils in DCM
6.6. Dendritic Cells in DCM
7. The Role of Immune Cells in Other Cardiomyopathies
8. Immunotherapy for DCM
8.1. Immunosuppression Therapy
8.2. Intravenous Immunoglobulin
8.3. Immunoadsorption
8.4. Cell Therapy
8.5. Pharmacological Therapy for DCM
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schultheiss, H.-P.; Fairweather, D.; Caforio, A.L.P.; Escher, F.; Hershberger, R.E.; Lipshultz, S.E.; Liu, P.P.; Matsumori, A.; Mazzanti, A.; McMurray, J.; et al. Dilated cardiomyopathy. Nat. Rev. Dis. Primers 2019, 5, 32. [Google Scholar] [CrossRef]
- Taylor, M.R.G.; Carniel, E.; Mestroni, L. Cardiomyopathy, familial dilated. Orphanet J. Rare Dis. 2006, 1, 27. [Google Scholar] [CrossRef] [Green Version]
- Towbin, J.A.; Lowe, A.M.; Colan, S.D.; Sleeper, L.A.; Orav, E.J.; Clunie, S.; Messere, J.; Cox, G.F.; Lurie, P.R.; Hsu, D.; et al. Incidence, causes, and outcomes of dilated cardiomyopathy in children. JAMA 2006, 296, 1867–1876. [Google Scholar] [CrossRef]
- De Noronha, S.V.; Sharma, S.; Papadakis, M.; Desai, S.; Whyte, G.; Sheppard, M.N. Aetiology of sudden cardiac death in athletes in the United Kingdom: A pathological study. Heart Br. Card. Soc. 2009, 95, 1409–1414. [Google Scholar] [CrossRef]
- Rodrigues, P.; Joshi, A.; Williams, H.; Westwood, M.; Petersen, S.E.; Zemrak, F.; Schilling, R.J.; Kirkby, C.; Wragg, A.; Manisty, C.; et al. Diagnosis and Prognosis in Sudden Cardiac Arrest Survivors without Coronary Artery Disease: Utility of a Clinical Approach Using Cardiac Magnetic Resonance Imaging. Circ. Cardiovasc. Imaging 2017, 10, e006709. [Google Scholar] [CrossRef]
- Reichart, D.; Magnussen, C.; Zeller, T.; Blankenberg, S. Dilated cardiomyopathy: From epidemiologic to genetic phenotypes: A translational review of current literature. J. Intern. Med. 2019, 286, 362–372. [Google Scholar] [CrossRef] [Green Version]
- Castelli, G.; Fornaro, A.; Ciaccheri, M.; Dolara, A.; Troiani, V.; Tomberli, B.; Olivotto, I.; Gensini, G.F. Improving survival rates of patients with idiopathic dilated cardiomyopathy in Tuscany over 3 decades: Impact of evidence-based management. Circ. Heart Fail. 2013, 6, 913–921. [Google Scholar] [CrossRef] [Green Version]
- Seferović, P.M.; Polovina, M.; Bauersachs, J.; Arad, M.; Ben Gal, T.; Lund, L.H.; Felix, S.B.; Arbustini, E.; Caforio, A.L.P.; Farmakis, D.; et al. Heart failure in cardiomyopathies: A position paper from the Heart Failure Association of the European Society of Cardiology. Eur. J. Heart Fail. 2019, 21, 553–576. [Google Scholar] [CrossRef] [Green Version]
- Mann, D.L. Innate immunity and the failing heart: The cytokine hypothesis revisited. Circ. Res. 2015, 116, 1254–1268. [Google Scholar] [CrossRef]
- Kühl, U.; Noutsias, M.; Seeberg, B.; Schultheiss, H.P. Immunohistological evidence for a chronic intramyocardial inflammatory process in dilated cardiomyopathy. Heart Br. Card. Soc. 1996, 75, 295–300. [Google Scholar] [CrossRef] [Green Version]
- Peters, S.; Johnson, R.; Birch, S.; Zentner, D.; Hershberger, R.E.; Fatkin, D. Familial Dilated Cardiomyopathy. Heart Lung Circ. 2020, 29, 566–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harding, D.; Chong, M.H.A.; Lahoti, N.; Bigogno, C.M.; Prema, R.; Mohiddin, S.A.; Marelli-Berg, F. Dilated cardiomyopathy and chronic cardiac inflammation: Pathogenesis, diagnosis and therapy. J. Intern. Med. 2023, 293, 23–47. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.N.; Mestroni, L.; Taylor, M.R.G. Genetics of dilated cardiomyopathy. Curr. Opin. Cardiol. 2021, 36, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Piran, S.; Liu, P.; Morales, A.; Hershberger, R.E. Where genome meets phenome: Rationale for integrating genetic and protein biomarkers in the diagnosis and management of dilated cardiomyopathy and heart failure. J. Am. Coll. Cardiol. 2012, 60, 283–289. [Google Scholar] [CrossRef] [Green Version]
- Ammirati, E.; Frigerio, M.; Adler, E.D.; Basso, C.; Birnie, D.H.; Brambatti, M.; Friedrich, M.G.; Klingel, K.; Lehtonen, J.; Moslehi, J.J.; et al. Management of Acute Myocarditis and Chronic Inflammatory Cardiomyopathy: An Expert Consensus Document. Circ. Heart Fail. 2020, 13, e007405. [Google Scholar] [CrossRef]
- George, A.; Figueredo, V.M. Alcoholic cardiomyopathy: A review. J. Card. Fail. 2011, 17, 844–849. [Google Scholar] [CrossRef] [Green Version]
- Monda, E.; Palmiero, G.; Rubino, M.; Verrillo, F.; Amodio, F.; Di Fraia, F.; Pacileo, R.; Fimiani, F.; Esposito, A.; Cirillo, A.; et al. Molecular Basis of Inflammation in the Pathogenesis of Cardiomyopathies. Int. J. Mol. Sci. 2020, 21, 6462. [Google Scholar] [CrossRef]
- Tschöpe, C.; Ammirati, E.; Bozkurt, B.; Caforio, A.L.P.; Cooper, L.T.; Felix, S.B.; Hare, J.M.; Heidecker, B.; Heymans, S.; Hübner, N.; et al. Myocarditis and inflammatory cardiomyopathy: Current evidence and future directions. Nat. Rev. Cardiol. 2021, 18, 169–193. [Google Scholar] [CrossRef]
- Caforio, A.L.; Keeling, P.J.; Zachara, E.; Mestroni, L.; Camerini, F.; Mann, J.M.; Bottazzo, G.F.; McKenna, W.J. Evidence from family studies for autoimmunity in dilated cardiomyopathy. Lancet Lond. Engl. 1994, 344, 773–777. [Google Scholar] [CrossRef]
- Volkova, M.; Russell, R. Anthracycline cardiotoxicity: Prevalence, pathogenesis and treatment. Curr. Cardiol. Rev. 2011, 7, 214–220. [Google Scholar] [CrossRef] [Green Version]
- Tucker, N.R.; Chaffin, M.; Fleming, S.J.; Hall, A.W.; Parsons, V.A.; Bedi, K.C.; Akkad, A.-D.; Herndon, C.N.; Arduini, A.; Papangeli, I.; et al. Transcriptional and Cellular Diversity of the Human Heart. Circulation 2020, 142, 466–482. [Google Scholar] [CrossRef]
- Litviňuková, M.; Talavera-López, C.; Maatz, H.; Reichart, D.; Worth, C.L.; Lindberg, E.L.; Kanda, M.; Polanski, K.; Heinig, M.; Lee, M.; et al. Cells of the adult human heart. Nature 2020, 588, 466–472. [Google Scholar] [CrossRef]
- Jung, S.-H.; Hwang, B.-H.; Shin, S.; Park, E.-H.; Park, S.-H.; Kim, C.W.; Kim, E.; Choo, E.; Choi, I.J.; Swirski, F.K.; et al. Spatiotemporal dynamics of macrophage heterogeneity and a potential function of Trem2hi macrophages in infarcted hearts. Nat. Commun. 2022, 13, 4580. [Google Scholar] [CrossRef] [PubMed]
- Hua, X.; Hu, G.; Hu, Q.; Chang, Y.; Hu, Y.; Gao, L.; Chen, X.; Yang, P.-C.; Zhang, Y.; Li, M.; et al. Single-Cell RNA Sequencing to Dissect the Immunological Network of Autoimmune Myocarditis. Circulation 2020, 142, 384–400. [Google Scholar] [CrossRef]
- Abplanalp, W.T.; John, D.; Cremer, S.; Assmus, B.; Dorsheimer, L.; Hoffmann, J.; Becker-Pergola, G.; Rieger, M.A.; Zeiher, A.M.; Vasa-Nicotera, M.; et al. Single-cell RNA-sequencing reveals profound changes in circulating immune cells in patients with heart failure. Cardiovasc. Res. 2021, 117, 484–494. [Google Scholar] [CrossRef] [Green Version]
- Pinto, A.R.; Ilinykh, A.; Ivey, M.J.; Kuwabara, J.T.; D’Antoni, M.L.; Debuque, R.; Chandran, A.; Wang, L.; Arora, K.; Rosenthal, N.A.; et al. Revisiting Cardiac Cellular Composition. Circ. Res. 2016, 118, 400–409. [Google Scholar] [CrossRef]
- Barin, J.G.; Rose, N.R.; Ciháková, D. Macrophage diversity in cardiac inflammation: A review. Immunobiology 2012, 217, 468–475. [Google Scholar] [CrossRef] [Green Version]
- Puhl, S.-L.; Steffens, S. Neutrophils in Post-myocardial Infarction Inflammation: Damage vs. Resolution? Front. Cardiovasc. Med. 2019, 6, 25. [Google Scholar] [CrossRef] [Green Version]
- Silvestre-Roig, C.; Braster, Q.; Ortega-Gomez, A.; Soehnlein, O. Neutrophils as regulators of cardiovascular inflammation. Nat. Rev. Cardiol. 2020, 17, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Blanton, R.M.; Carrillo-Salinas, F.J.; Alcaide, P. T-cell recruitment to the heart: Friendly guests or unwelcome visitors? Am. J. Physiol. Heart Circ. Physiol. 2019, 317, H124–H140. [Google Scholar] [CrossRef] [PubMed]
- Rock, K.L.; Lai, J.-J.; Kono, H. Innate and adaptive immune responses to cell death. Immunol. Rev. 2011, 243, 191–205. [Google Scholar] [CrossRef] [Green Version]
- Rurik, J.G.; Aghajanian, H.; Epstein, J.A. Immune Cells and Immunotherapy for Cardiac Injury and Repair. Circ. Res. 2021, 128, 1766–1779. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.; Martinez-Pomares, L. Physiological roles of macrophages. Pflug. Arch. 2017, 469, 365–374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frangogiannis, N.G. Cardiac fibrosis: Cell biological mechanisms, molecular pathways and therapeutic opportunities. Mol. Asp. Med. 2019, 65, 70–99. [Google Scholar] [CrossRef] [PubMed]
- Zaman, R.; Epelman, S. Resident cardiac macrophages: Heterogeneity and function in health and disease. Immunity 2022, 55, 1549–1563. [Google Scholar] [CrossRef]
- Banerjee, D.; Tian, R.; Cai, S. The Role of Innate Immune Cells in Cardiac Injury and Repair: A Metabolic Perspective. Curr. Cardiol. Rep. 2023, 25, 631–640. [Google Scholar] [CrossRef]
- Wagner, M.J.; Khan, M.; Mohsin, S. Healing the Broken Heart; The Immunomodulatory Effects of Stem Cell Therapy. Front. Immunol. 2020, 11, 639. [Google Scholar] [CrossRef] [Green Version]
- Epelman, S.; Liu, P.P.; Mann, D.L. Role of innate and adaptive immune mechanisms in cardiac injury and repair. Nat. Rev. Immunol. 2015, 15, 117–129. [Google Scholar] [CrossRef]
- McKenna, W.J.; Caforio, A.L.P. Myocardial Inflammation and Sudden Death in the Inherited Cardiomyopathies. Can. J. Cardiol. 2022, 38, 427–438. [Google Scholar] [CrossRef]
- Swirski, F.K.; Nahrendorf, M. Cardioimmunology: The immune system in cardiac homeostasis and disease. Nat. Rev. Immunol. 2018, 18, 733–744. [Google Scholar] [CrossRef]
- Cihakova, D.; Barin, J.G.; Afanasyeva, M.; Kimura, M.; Fairweather, D.; Berg, M.; Talor, M.V.; Baldeviano, G.C.; Frisancho, S.; Gabrielson, K.; et al. Interleukin-13 protects against experimental autoimmune myocarditis by regulating macrophage differentiation. Am. J. Pathol. 2008, 172, 1195–1208. [Google Scholar] [CrossRef] [Green Version]
- Bracamonte-Baran, W.; Čiháková, D. Cardiac Autoimmunity: Myocarditis. Adv. Exp. Med. Biol. 2017, 1003, 187–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilhelmi, M.H.; Leyh, R.G.; Wilhelmi, M.; Haverich, A. Upregulation of endothelial adhesion molecules in hearts with congestive and ischemic cardiomyopathy: Immunohistochemical evaluation of inflammatory endothelial cell activation. Eur. J. Cardio-Thorac. Surg. Off. J. Eur. Assoc. Cardio-Thorac. Surg. 2005, 27, 122–127. [Google Scholar] [CrossRef] [Green Version]
- Jaakkola, K.; Jalkanen, S.; Kaunismäki, K.; Vänttinen, E.; Saukko, P.; Alanen, K.; Kallajoki, M.; Voipio-Pulkki, L.M.; Salmi, M. Vascular adhesion protein-1, intercellular adhesion molecule-1 and P-selectin mediate leukocyte binding to ischemic heart in humans. J. Am. Coll. Cardiol. 2000, 36, 122–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liao, J.K. Linking endothelial dysfunction with endothelial cell activation. J. Clin. Investig. 2013, 123, 540–541. [Google Scholar] [CrossRef] [Green Version]
- Baselet, B.; Sonveaux, P.; Baatout, S.; Aerts, A. Pathological effects of ionizing radiation: Endothelial activation and dysfunction. Cell. Mol. Life Sci. CMLS 2019, 76, 699–728. [Google Scholar] [CrossRef] [Green Version]
- Tong, M.; Sadoshima, J. Mitochondrial autophagy in cardiomyopathy. Curr. Opin. Genet. Dev. 2016, 38, 8–15. [Google Scholar] [CrossRef] [Green Version]
- Frangogiannis, N.G. Regulation of the inflammatory response in cardiac repair. Circ. Res. 2012, 110, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Lafuse, W.P.; Wozniak, D.J.; Rajaram, M.V.S. Role of Cardiac Macrophages on Cardiac Inflammation, Fibrosis and Tissue Repair. Cells 2020, 10, 51. [Google Scholar] [CrossRef]
- Murphy, A.M.; Wong, A.L.; Bezuhly, M. Modulation of angiotensin II signaling in the prevention of fibrosis. Fibrogenesis Tissue Repair 2015, 8, 7. [Google Scholar] [CrossRef] [Green Version]
- Frangogiannis, N.G. Cardiac fibrosis. Cardiovasc. Res. 2021, 117, 1450–1488. [Google Scholar] [CrossRef]
- Ma, Z.-G.; Yuan, Y.-P.; Wu, H.-M.; Zhang, X.; Tang, Q.-Z. Cardiac fibrosis: New insights into the pathogenesis. Int. J. Biol. Sci. 2018, 14, 1645–1657. [Google Scholar] [CrossRef] [Green Version]
- Kong, P.; Christia, P.; Frangogiannis, N.G. The pathogenesis of cardiac fibrosis. Cell. Mol. Life Sci. CMLS 2014, 71, 549–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nevers, T.; Salvador, A.M.; Velazquez, F.; Ngwenyama, N.; Carrillo-Salinas, F.J.; Aronovitz, M.; Blanton, R.M.; Alcaide, P. Th1 effector T cells selectively orchestrate cardiac fibrosis in nonischemic heart failure. J. Exp. Med. 2017, 214, 3311–3329. [Google Scholar] [CrossRef] [Green Version]
- Bansal, S.S.; Ismahil, M.A.; Goel, M.; Patel, B.; Hamid, T.; Rokosh, G.; Prabhu, S.D. Activated T Lymphocytes are Essential Drivers of Pathological Remodeling in Ischemic Heart Failure. Circ. Heart Fail. 2017, 10, e003688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blyszczuk, P.; Müller-Edenborn, B.; Valenta, T.; Osto, E.; Stellato, M.; Behnke, S.; Glatz, K.; Basler, K.; Lüscher, T.F.; Distler, O.; et al. Transforming growth factor-β-dependent Wnt secretion controls myofibroblast formation and myocardial fibrosis progression in experimental autoimmune myocarditis. Eur. Heart J. 2017, 38, 1413–1425. [Google Scholar] [CrossRef] [Green Version]
- Sivakumar, P.; Gupta, S.; Sarkar, S.; Sen, S. Upregulation of lysyl oxidase and MMPs during cardiac remodeling in human dilated cardiomyopathy. Mol. Cell. Biochem. 2008, 307, 159–167. [Google Scholar] [CrossRef]
- Zhang, W.; Chancey, A.L.; Tzeng, H.-P.; Zhou, Z.; Lavine, K.J.; Gao, F.; Sivasubramanian, N.; Barger, P.M.; Mann, D.L. The development of myocardial fibrosis in transgenic mice with targeted overexpression of tumor necrosis factor requires mast cell-fibroblast interactions. Circulation 2011, 124, 2106–2116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geissmann, F.; Jung, S.; Littman, D.R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 2003, 19, 71–82. [Google Scholar] [CrossRef] [Green Version]
- Maekawa, Y.; Anzai, T.; Yoshikawa, T.; Asakura, Y.; Takahashi, T.; Ishikawa, S.; Mitamura, H.; Ogawa, S. Prognostic significance of peripheral monocytosis after reperfused acute myocardial infarction: A possible role for left ventricular remodeling. J. Am. Coll. Cardiol. 2002, 39, 241–246. [Google Scholar] [CrossRef] [Green Version]
- Riad, A.; Gross, S.; Witte, J.; Feldtmann, R.; Wagner, K.B.; Reinke, Y.; Weitmann, K.; Empen, K.; Beug, D.; Westermann, D.; et al. MD-2 is a new predictive biomarker in dilated cardiomyopathy and exerts direct effects in isolated cardiomyocytes. Int. J. Cardiol. 2018, 270, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Pugin, J.; Stern-Voeffray, S.; Daubeuf, B.; Matthay, M.A.; Elson, G.; Dunn-Siegrist, I. Soluble MD-2 activity in plasma from patients with severe sepsis and septic shock. Blood 2004, 104, 4071–4079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feldtmann, R.; Kümmel, A.; Chamling, B.; Strohbach, A.; Lehnert, K.; Gross, S.; Loerzer, L.; Riad, A.; Lindner, D.; Westermann, D.; et al. Myeloid differentiation factor-2 activates monocytes in patients with dilated cardiomyopathy. Immunology 2022, 167, 40–53. [Google Scholar] [CrossRef] [PubMed]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte chemoattractant protein-1 (MCP-1): An overview. J. Interferon Cytokine Res. Off. J. Int. Soc. Interferon Cytokine Res. 2009, 29, 313–326. [Google Scholar] [CrossRef]
- Jiang, Y.; Beller, D.I.; Frendl, G.; Graves, D.T. Monocyte chemoattractant protein-1 regulates adhesion molecule expression and cytokine production in human monocytes. J. Immunol. 1992, 148, 2423–2428. [Google Scholar] [CrossRef]
- Carty, C.L.; Heagerty, P.; Heckbert, S.R.; Enquobahrie, D.A.; Jarvik, G.P.; Davis, S.; Tracy, R.P.; Reiner, A.P. Association of genetic variation in serum amyloid-A with cardiovascular disease and interactions with IL6, IL1RN, IL1beta and TNF genes in the Cardiovascular Health Study. J. Atheroscler. Thromb. 2009, 16, 419–430. [Google Scholar] [CrossRef] [Green Version]
- Bironaite, D.; Daunoravicius, D.; Bogomolovas, J.; Cibiras, S.; Vitkus, D.; Zurauskas, E.; Zasytyte, I.; Rucinskas, K.; Labeit, S.; Venalis, A.; et al. Molecular mechanisms behind progressing chronic inflammatory dilated cardiomyopathy. BMC Cardiovasc. Disord. 2015, 15, 26. [Google Scholar] [CrossRef] [Green Version]
- Lehmann, M.H.; Kühnert, H.; Müller, S.; Sigusch, H.H. Monocyte chemoattractant protein 1 (MCP-1) gene expression in dilated cardiomyopathy. Cytokine 1998, 10, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Nakamura, K.; Kusano, K.F.; Nakamura, Y.; Ohta-Ogo, K.; Nagase, S.; Sakuragi, S.; Ohe, T. Expression of monocyte chemoattractant protein-1 in idiopathic dilated cardiomyopathy. Int. J. Cardiol. 2008, 126, 427–429. [Google Scholar] [CrossRef] [Green Version]
- Weinberger, T.; Schulz, C. Myocardial infarction: A critical role of macrophages in cardiac remodeling. Front. Physiol. 2015, 6, 107. [Google Scholar] [CrossRef]
- Heidt, T.; Courties, G.; Dutta, P.; Sager, H.B.; Sebas, M.; Iwamoto, Y.; Sun, Y.; Da Silva, N.; Panizzi, P.; van der Laan, A.M.; et al. Differential contribution of monocytes to heart macrophages in steady-state and after myocardial infarction. Circ. Res. 2014, 115, 284–295. [Google Scholar] [CrossRef]
- Epelman, S.; Lavine, K.J.; Beaudin, A.E.; Sojka, D.K.; Carrero, J.A.; Calderon, B.; Brija, T.; Gautier, E.L.; Ivanov, S.; Satpathy, A.T.; et al. Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation. Immunity 2014, 40, 91–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lavine, K.J.; Epelman, S.; Uchida, K.; Weber, K.J.; Nichols, C.G.; Schilling, J.D.; Ornitz, D.M.; Randolph, G.J.; Mann, D.L. Distinct macrophage lineages contribute to disparate patterns of cardiac recovery and remodeling in the neonatal and adult heart. Proc. Natl. Acad. Sci. USA 2014, 111, 16029–16034. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, G.; Bredemeyer, A.; Li, W.; Zaitsev, K.; Koenig, A.L.; Lokshina, I.; Mohan, J.; Ivey, B.; Hsiao, H.-M.; Weinheimer, C.; et al. Tissue Resident CCR2− and CCR2+ Cardiac Macrophages Differentially Orchestrate Monocyte Recruitment and Fate Specification Following Myocardial Injury. Circ. Res. 2019, 124, 263–278. [Google Scholar] [CrossRef]
- Warner, N.; Núñez, G. MyD88: A critical adaptor protein in innate immunity signal transduction. J. Immunol. 2013, 190, 3–4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, N.R.; Mohan, J.; Kopecky, B.J.; Guo, S.; Du, L.; Leid, J.; Feng, G.; Lokshina, I.; Dmytrenko, O.; Luehmann, H.; et al. Resident cardiac macrophages mediate adaptive myocardial remodeling. Immunity 2021, 54, 2072–2088.e7. [Google Scholar] [CrossRef]
- Ma, Y.; Mouton, A.J.; Lindsey, M.L. Cardiac macrophage biology in the steady-state heart, the aging heart, and following myocardial infarction. Transl. Res. J. Lab. Clin. Med. 2018, 191, 15–28. [Google Scholar] [CrossRef] [Green Version]
- Gombozhapova, A.; Rogovskaya, Y.; Shurupov, V.; Rebenkova, M.; Kzhyshkowska, J.; Popov, S.V.; Karpov, R.S.; Ryabov, V. Macrophage activation and polarization in post-infarction cardiac remodeling. J. Biomed. Sci. 2017, 24, 13. [Google Scholar] [CrossRef] [Green Version]
- Boniakowski, A.E.; Kimball, A.S.; Jacobs, B.N.; Kunkel, S.L.; Gallagher, K.A. Macrophage-Mediated Inflammation in Normal and Diabetic Wound Healing. J. Immunol. 2017, 199, 17–24. [Google Scholar] [CrossRef] [Green Version]
- Nakayama, T.; Sugano, Y.; Yokokawa, T.; Nagai, T.; Matsuyama, T.-A.; Ohta-Ogo, K.; Ikeda, Y.; Ishibashi-Ueda, H.; Nakatani, T.; Ohte, N.; et al. Clinical impact of the presence of macrophages in endomyocardial biopsies of patients with dilated cardiomyopathy. Eur. J. Heart Fail. 2017, 19, 490–498. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Bauersachs, J.; Langer, H.F. Immune mechanisms in heart failure. Eur. J. Heart Fail. 2017, 19, 1379–1389. [Google Scholar] [CrossRef] [Green Version]
- Nian, M.; Lee, P.; Khaper, N.; Liu, P. Inflammatory cytokines and postmyocardial infarction remodeling. Circ. Res. 2004, 94, 1543–1553. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, S.D.; Frangogiannis, N.G. The Biological Basis for Cardiac Repair after Myocardial Infarction: From Inflammation to Fibrosis. Circ. Res. 2016, 119, 91–112. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, G.; Schneider, C.; Wong, N.; Bredemeyer, A.; Hulsmans, M.; Nahrendorf, M.; Epelman, S.; Kreisel, D.; Liu, Y.; Itoh, A.; et al. The human heart contains distinct macrophage subsets with divergent origins and functions. Nat. Med. 2018, 24, 1234–1245. [Google Scholar] [CrossRef] [PubMed]
- Yap, J.; Cabrera-Fuentes, H.A.; Irei, J.; Hausenloy, D.J.; Boisvert, W.A. Role of Macrophages in Cardioprotection. Int. J. Mol. Sci. 2019, 20, 2474. [Google Scholar] [CrossRef] [Green Version]
- Baumeier, C.; Escher, F.; Aleshcheva, G.; Pietsch, H.; Schultheiss, H.-P. Plasminogen activator inhibitor-1 reduces cardiac fibrosis and promotes M2 macrophage polarization in inflammatory cardiomyopathy. Basic Res. Cardiol. 2021, 116, 1. [Google Scholar] [CrossRef]
- Davani, E.Y.; Dorscheid, D.R.; Lee, C.-H.; van Breemen, C.; Walley, K.R. Novel regulatory mechanism of cardiomyocyte contractility involving ICAM-1 and the cytoskeleton. Am. J. Physiol. Heart Circ. Physiol. 2004, 287, H1013–H1022. [Google Scholar] [CrossRef] [Green Version]
- Simms, M.G.; Walley, K.R. Activated macrophages decrease rat cardiac myocyte contractility: Importance of ICAM-1-dependent adhesion. Am. J. Physiol. 1999, 277, H253–H260. [Google Scholar] [CrossRef]
- Gasparini, S.; Fonfara, S.; Kitz, S.; Hetzel, U.; Kipar, A. Canine Dilated Cardiomyopathy: Diffuse Remodeling, Focal Lesions, and the Involvement of Macrophages and New Vessel Formation. Vet. Pathol. 2020, 57, 397–408. [Google Scholar] [CrossRef] [Green Version]
- Dong, C. Cytokine Regulation and Function in T Cells. Annu. Rev. Immunol. 2021, 39, 51–76. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.V.; Connors, T.J.; Farber, D.L. Human T Cell Development, Localization, and Function throughout Life. Immunity 2018, 48, 202–213. [Google Scholar] [CrossRef] [Green Version]
- Meng, X.; Yang, J.; Dong, M.; Zhang, K.; Tu, E.; Gao, Q.; Chen, W.; Zhang, C.; Zhang, Y. Regulatory T cells in cardiovascular diseases. Nat. Rev. Cardiol. 2016, 13, 167–179. [Google Scholar] [CrossRef]
- Stephenson, E.; Savvatis, K.; Mohiddin, S.A.; Marelli-Berg, F.M. T-cell immunity in myocardial inflammation: Pathogenic role and therapeutic manipulation. Br. J. Pharmacol. 2017, 174, 3914–3925. [Google Scholar] [CrossRef] [Green Version]
- Barin, J.G.; Čiháková, D. Control of inflammatory heart disease by CD4+ T cells. Ann. N. Y. Acad. Sci. 2013, 1285, 80–96. [Google Scholar] [CrossRef]
- Laroumanie, F.; Douin-Echinard, V.; Pozzo, J.; Lairez, O.; Tortosa, F.; Vinel, C.; Delage, C.; Calise, D.; Dutaur, M.; Parini, A.; et al. CD4+ T cells promote the transition from hypertrophy to heart failure during chronic pressure overload. Circulation 2014, 129, 2111–2124. [Google Scholar] [CrossRef]
- Nevers, T.; Salvador, A.M.; Grodecki-Pena, A.; Knapp, A.; Velázquez, F.; Aronovitz, M.; Kapur, N.K.; Karas, R.H.; Blanton, R.M.; Alcaide, P. Left Ventricular T-Cell Recruitment Contributes to the Pathogenesis of Heart Failure. Circ. Heart Fail. 2015, 8, 776–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amador, C.A.; Barrientos, V.; Peña, J.; Herrada, A.A.; González, M.; Valdés, S.; Carrasco, L.; Alzamora, R.; Figueroa, F.; Kalergis, A.M.; et al. Spironolactone decreases DOCA-salt-induced organ damage by blocking the activation of T helper 17 and the downregulation of regulatory T lymphocytes. Hypertension 2014, 63, 797–803. [Google Scholar] [CrossRef] [Green Version]
- Wei, Y.; Yu, K.; Wei, H.; Su, X.; Zhu, R.; Shi, H.; Sun, H.; Luo, Q.; Xu, W.; Xiao, J.; et al. CD4+ CD25+ GARP+ regulatory T cells display a compromised suppressive function in patients with dilated cardiomyopathy. Immunology 2017, 151, 291–303. [Google Scholar] [CrossRef] [Green Version]
- Kong, Q.; Li, X.; Wu, W.; Yang, F.; Liu, Y.; Lai, W.; Pan, X.; Gao, M.; Xue, Y. Increased circulating T-helper 22 cells in patients with dilated cardiomyopathy. Mol. Med. Rep. 2014, 10, 359–364. [Google Scholar] [CrossRef] [Green Version]
- Nindl, V.; Maier, R.; Ratering, D.; De Giuli, R.; Züst, R.; Thiel, V.; Scandella, E.; Di Padova, F.; Kopf, M.; Rudin, M.; et al. Cooperation of Th1 and Th17 cells determines transition from autoimmune myocarditis to dilated cardiomyopathy. Eur. J. Immunol. 2012, 42, 2311–2321. [Google Scholar] [CrossRef]
- Boehm, U.; Klamp, T.; Groot, M.; Howard, J.C. Cellular responses to interferon-gamma. Annu. Rev. Immunol. 1997, 15, 749–795. [Google Scholar] [CrossRef]
- Afanasyeva, M.; Wang, Y.; Kaya, Z.; Park, S.; Zilliox, M.J.; Schofield, B.H.; Hill, S.L.; Rose, N.R. Experimental autoimmune myocarditis in A/J mice is an interleukin-4-dependent disease with a Th2 phenotype. Am. J. Pathol. 2001, 159, 193–203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eriksson, U.; Kurrer, M.O.; Sebald, W.; Brombacher, F.; Kopf, M. Dual role of the IL-12/IFN-gamma axis in the development of autoimmune myocarditis: Induction by IL-12 and protection by IFN-gamma. J. Immunol. 2001, 167, 5464–5469. [Google Scholar] [CrossRef]
- Eriksson, U.; Kurrer, M.O.; Bingisser, R.; Eugster, H.P.; Saremaslani, P.; Follath, F.; Marsch, S.; Widmer, U. Lethal autoimmune myocarditis in interferon-gamma receptor-deficient mice: Enhanced disease severity by impaired inducible nitric oxide synthase induction. Circulation 2001, 103, 18–21. [Google Scholar] [CrossRef] [Green Version]
- Afanasyeva, M.; Georgakopoulos, D.; Belardi, D.F.; Bedja, D.; Fairweather, D.; Wang, Y.; Kaya, Z.; Gabrielson, K.L.; Rodriguez, E.R.; Caturegli, P.; et al. Impaired up-regulation of CD25 on CD4+ T cells in IFN-gamma knockout mice is associated with progression of myocarditis to heart failure. Proc. Natl. Acad. Sci. USA 2005, 102, 180–185. [Google Scholar] [CrossRef]
- Guo, Y.; Wu, W.; Cen, Z.; Li, X.; Kong, Q.; Zhou, Q. IL-22-producing Th22 cells play a protective role in CVB3-induced chronic myocarditis and dilated cardiomyopathy by inhibiting myocardial fibrosis. Virol. J. 2014, 11, 230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barin, J.G.; Baldeviano, G.C.; Talor, M.V.; Wu, L.; Ong, S.; Fairweather, D.; Bedja, D.; Stickel, N.R.; Fontes, J.A.; Cardamone, A.B.; et al. Fatal eosinophilic myocarditis develops in the absence of IFN-γ and IL-17A. J. Immunol. 2013, 191, 4038–4047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baldeviano, G.C.; Barin, J.G.; Talor, M.V.; Srinivasan, S.; Bedja, D.; Zheng, D.; Gabrielson, K.; Iwakura, Y.; Rose, N.R.; Cihakova, D. Interleukin-17A is dispensable for myocarditis but essential for the progression to dilated cardiomyopathy. Circ. Res. 2010, 106, 1646–1655. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Ong, S.; Talor, M.V.; Barin, J.G.; Baldeviano, G.C.; Kass, D.A.; Bedja, D.; Zhang, H.; Sheikh, A.; Margolick, J.B.; et al. Cardiac fibroblasts mediate IL-17A-driven inflammatory dilated cardiomyopathy. J. Exp. Med. 2014, 211, 1449–1464. [Google Scholar] [CrossRef]
- Myers, J.M.; Cooper, L.T.; Kem, D.C.; Stavrakis, S.; Kosanke, S.D.; Shevach, E.M.; Fairweather, D.; Stoner, J.A.; Cox, C.J.; Cunningham, M.W. Cardiac myosin-Th17 responses promote heart failure in human myocarditis. JCI Insight 2016, 1, e85851. [Google Scholar] [CrossRef] [Green Version]
- Kolls, J.K.; Lindén, A. Interleukin-17 family members and inflammation. Immunity 2004, 21, 467–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cortez, D.M.; Feldman, M.D.; Mummidi, S.; Valente, A.J.; Steffensen, B.; Vincenti, M.; Barnes, J.L.; Chandrasekar, B. IL-17 stimulates MMP-1 expression in primary human cardiac fibroblasts via p38 MAPK- and ERK1/2-dependent C/EBP-beta, NF-kappaB, and AP-1 activation. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H3356–H3365. [Google Scholar] [CrossRef] [Green Version]
- Baba, A. Autoantigen estimation and simple screening assay against cardiodepressant autoantibodies in patients with dilated cardiomyopathy. Ther. Apher. Dial. Off. Peer-Rev. J. Int. Soc. Apher. Jpn. Soc. Apher. Jpn. Soc. Dial. Ther. 2008, 12, 109–116. [Google Scholar] [CrossRef]
- Sakaguchi, S.; Yamaguchi, T.; Nomura, T.; Ono, M. Regulatory T cells and immune tolerance. Cell 2008, 133, 775–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, L.; Jin, H.; Li, H. GARP: A surface molecule of regulatory T cells that is involved in the regulatory function and TGF-β releasing. Oncotarget 2016, 7, 42826–42836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dawson, N.A.J.; Vent-Schmidt, J.; Levings, M.K. Engineered Tolerance: Tailoring Development, Function, and Antigen-Specificity of Regulatory T Cells. Front. Immunol. 2017, 8, 1460. [Google Scholar] [CrossRef]
- Kraj, P.; Ignatowicz, L. The mechanisms shaping the repertoire of CD4+ Foxp3+ regulatory T cells. Immunology 2018, 153, 290–296. [Google Scholar] [CrossRef] [Green Version]
- Tang, H.; Zhong, Y.; Zhu, Y.; Zhao, F.; Cui, X.; Wang, Z. Low responder T cell susceptibility to the suppressive function of regulatory T cells in patients with dilated cardiomyopathy. Heart Br. Card. Soc. 2010, 96, 765–771. [Google Scholar] [CrossRef]
- Fantini, M.C.; Becker, C.; Monteleone, G.; Pallone, F.; Galle, P.R.; Neurath, M.F. Cutting edge: TGF-beta induces a regulatory phenotype in CD4+CD25− T cells through Foxp3 induction and down-regulation of Smad7. J. Immunol. 2004, 172, 5149–5153. [Google Scholar] [CrossRef] [Green Version]
- Rezende, R.M.; Oliveira, R.P.; Medeiros, S.R.; Gomes-Santos, A.C.; Alves, A.C.; Loli, F.G.; Guimarães, M.A.F.; Amaral, S.S.; da Cunha, A.P.; Weiner, H.L.; et al. Hsp65-producing Lactococcus lactis prevents experimental autoimmune encephalomyelitis in mice by inducing CD4+LAP+ regulatory T cells. J. Autoimmun. 2013, 40, 45–57. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Z.-F.; Tang, T.-T.; Dong, W.-Y.; Li, Y.-Y.; Xia, N.; Zhang, W.-C.; Zhou, S.-F.; Yuan, J.; Liao, M.-Y.; Li, J.-J.; et al. Defective circulating CD4+LAP+ regulatory T cells in patients with dilated cardiomyopathy. J. Leukoc. Biol. 2015, 97, 797–805. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, W.; Han, Z. Decreased circulating follicular regulatory T cells in patients with dilated cardiomyopathy. Braz. J. Med. Biol. Res. Rev. Bras. Pesqui. Medicas Biol. 2021, 54, e11232. [Google Scholar] [CrossRef] [PubMed]
- Daniels, M.D.; Hyland, K.V.; Wang, K.; Engman, D.M. Recombinant cardiac myosin fragment induces experimental autoimmune myocarditis via activation of Th1 and Th17 immunity. Autoimmunity 2008, 41, 490–499. [Google Scholar] [CrossRef] [Green Version]
- Efthimiadis, I.; Skendros, P.; Sarantopoulos, A.; Boura, P. CD4+/CD25+ T-Lymphocytes and Th1/Th2 regulation in dilated cardiomyopathy. Hippokratia 2011, 15, 335–342. [Google Scholar] [PubMed]
- Vdovenko, D.; Eriksson, U. Regulatory Role of CD4+ T Cells in Myocarditis. J. Immunol. Res. 2018, 2018, 4396351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, R.; Wan, Q.; Kozhaya, L.; Fujii, H.; Unutmaz, D. Identification of a regulatory T cell specific cell surface molecule that mediates suppressive signals and induces Foxp3 expression. PLoS ONE 2008, 3, e2705. [Google Scholar] [CrossRef] [Green Version]
- Lindberg, E.; Andersson, B.; Hörnquist, E.H.; Magnusson, Y. Impaired activation of IFN-gamma+CD4+ T cells in peripheral blood of patients with dilated cardiomyopathy. Cell. Immunol. 2010, 263, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Wang, K.; Li, Y.; Xia, N.; Nie, S.; Lv, B.; Zhang, M.; Tu, X.; Li, Q.; Tang, T.; et al. Down-regulation of microRNA-451a facilitates the activation and proliferation of CD4+ T cells by targeting Myc in patients with dilated cardiomyopathy. J. Biol. Chem. 2017, 292, 6004–6013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abston, E.D.; Coronado, M.J.; Bucek, A.; Onyimba, J.A.; Brandt, J.E.; Frisancho, J.A.; Kim, E.; Bedja, D.; Sung, Y.; Radtke, A.J.; et al. TLR3 deficiency induces chronic inflammatory cardiomyopathy in resistant mice following coxsackievirus B3 infection: Role for IL-4. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R267–R277. [Google Scholar] [CrossRef] [Green Version]
- Quách, T.D.; Rodríguez-Zhurbenko, N.; Hopkins, T.J.; Guo, X.; Hernández, A.M.; Li, W.; Rothstein, T.L. Distinctions among Circulating Antibody-Secreting Cell Populations, Including B-1 Cells, in Human Adult Peripheral Blood. J. Immunol. 2016, 196, 1060–1069. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Liu, J.; Burrows, P.D.; Wang, J.-Y. B Cell Development and Maturation. Adv. Exp. Med. Biol. 2020, 1254, 1–22. [Google Scholar] [CrossRef]
- Hofmann, K.; Clauder, A.-K.; Manz, R.A. Targeting B Cells and Plasma Cells in Autoimmune Diseases. Front. Immunol. 2018, 9, 835. [Google Scholar] [CrossRef]
- Li, Y.; Huang, Y.; Wu, W.; Wei, B.; Qin, L. B Cells Increase Myocardial Inflammation by Suppressing M2 Macrophage Polarization in Coxsackie Virus B3-Induced Acute Myocarditis. Inflammation 2019, 42, 953–960. [Google Scholar] [CrossRef]
- Guo, Y.; Cen, Z.; Wei, B.; Wu, W.; Zhou, Q. Increased circulating interleukin 10-secreting B cells in patients with dilated cardiomyopathy. Int. J. Clin. Exp. Pathol. 2015, 8, 8107–8114. [Google Scholar]
- Jiao, J.; Lu, Y.-Z.; Xia, N.; Wang, Y.-Q.; Tang, T.-T.; Nie, S.-F.; Lv, B.-J.; Wang, K.-J.; Wen, S.; Li, J.-Y.; et al. Defective Circulating Regulatory B Cells in Patients with Dilated Cardiomyopathy. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2018, 46, 23–35. [Google Scholar] [CrossRef]
- Meier, L.A.; Binstadt, B.A. The Contribution of Autoantibodies to Inflammatory Cardiovascular Pathology. Front. Immunol. 2018, 9, 911. [Google Scholar] [CrossRef] [Green Version]
- Du, Y.; Yan, L.; Wang, J.; Zhan, W.; Song, K.; Han, X.; Li, X.; Cao, J.; Liu, H. β1-Adrenoceptor autoantibodies from DCM patients enhance the proliferation of T lymphocytes through the β1-AR/cAMP/PKA and p38 MAPK pathways. PLoS ONE 2012, 7, e52911. [Google Scholar] [CrossRef]
- Yuan, J.; Cao, A.-L.; Yu, M.; Lin, Q.-W.; Yu, X.; Zhang, J.-H.; Wang, M.; Guo, H.-P.; Liao, Y.-H. Th17 cells facilitate the humoral immune response in patients with acute viral myocarditis. J. Clin. Immunol. 2010, 30, 226–234. [Google Scholar] [CrossRef]
- Tang, Q.; Cen, Z.; Lu, J.; Dong, J.; Qin, L.; Lu, F.; Wu, W. The abnormal distribution of peripheral B1 cells and transition B cells in patients with idiopathic dilated cardiomyopathy: A pilot study. BMC Cardiovasc. Disord. 2022, 22, 78. [Google Scholar] [CrossRef]
- Störk, S.; Boivin, V.; Horf, R.; Hein, L.; Lohse, M.J.; Angermann, C.E.; Jahns, R. Stimulating autoantibodies directed against the cardiac beta1-adrenergic receptor predict increased mortality in idiopathic cardiomyopathy. Am. Heart J. 2006, 152, 697–704. [Google Scholar] [CrossRef]
- Iwata, M.; Yoshikawa, T.; Baba, A.; Anzai, T.; Mitamura, H.; Ogawa, S. Autoantibodies against the second extracellular loop of beta1-adrenergic receptors predict ventricular tachycardia and sudden death in patients with idiopathic dilated cardiomyopathy. J. Am. Coll. Cardiol. 2001, 37, 418–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jane-wit, D.; Altuntas, C.Z.; Johnson, J.M.; Yong, S.; Wickley, P.J.; Clark, P.; Wang, Q.; Popović, Z.B.; Penn, M.S.; Damron, D.S.; et al. Beta 1-adrenergic receptor autoantibodies mediate dilated cardiomyopathy by agonistically inducing cardiomyocyte apoptosis. Circulation 2007, 116, 399–410. [Google Scholar] [CrossRef] [Green Version]
- Vazquez-Tello, A.; Halwani, R.; Li, R.; Nadigel, J.; Bar-Or, A.; Mazer, B.D.; Eidelman, D.H.; Al-Muhsen, S.; Hamid, Q. IL-17A and IL-17F expression in B lymphocytes. Int. Arch. Allergy Immunol. 2012, 157, 406–416. [Google Scholar] [CrossRef]
- Yu, M.; Wen, S.; Wang, M.; Liang, W.; Li, H.-H.; Long, Q.; Guo, H.-P.; Liao, Y.-H.; Yuan, J. TNF-α-secreting B cells contribute to myocardial fibrosis in dilated cardiomyopathy. J. Clin. Immunol. 2013, 33, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Siwik, D.A.; Chang, D.L.; Colucci, W.S. Interleukin-1beta and tumor necrosis factor-alpha decrease collagen synthesis and increase matrix metalloproteinase activity in cardiac fibroblasts in vitro. Circ. Res. 2000, 86, 1259–1265. [Google Scholar] [CrossRef]
- Theiss, A.L.; Simmons, J.G.; Jobin, C.; Lund, P.K. Tumor necrosis factor (TNF) alpha increases collagen accumulation and proliferation in intestinal myofibroblasts via TNF receptor 2. J. Biol. Chem. 2005, 280, 36099–36109. [Google Scholar] [CrossRef] [Green Version]
- Yang, M.; Rui, K.; Wang, S.; Lu, L. Regulatory B cells in autoimmune diseases. Cell. Mol. Immunol. 2013, 10, 122–132. [Google Scholar] [CrossRef] [Green Version]
- Lighaam, L.C.; Unger, P.-P.A.; Vredevoogd, D.W.; Verhoeven, D.; Vermeulen, E.; Turksma, A.W.; Ten Brinke, A.; Rispens, T.; van Ham, S.M. In vitro-Induced Human IL-10+ B Cells Do Not Show a Subset-Defining Marker Signature and Plastically Co-express IL-10 with Pro-Inflammatory Cytokines. Front. Immunol. 2018, 9, 1913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ong, S.; Rose, N.R.; Čiháková, D. Natural killer cells in inflammatory heart disease. Clin. Immunol. 2017, 175, 26–33. [Google Scholar] [CrossRef] [Green Version]
- Yokoyama, A. Natural killer cells in dilated cardiomyopathy. Tohoku J. Exp. Med. 1988, 154, 335–344. [Google Scholar] [CrossRef] [Green Version]
- Kanda, T.; Yokoyama, T.; Suzuki, T.; Murata, K. Functional abnormalities of circulating natural killer cell subpopulations in patients with dilated cardiomyopathy. Tohoku J. Exp. Med. 1992, 168, 529–537. [Google Scholar] [CrossRef] [Green Version]
- Ong, S.; Ligons, D.L.; Barin, J.G.; Wu, L.; Talor, M.V.; Diny, N.; Fontes, J.A.; Gebremariam, E.; Kass, D.A.; Rose, N.R.; et al. Natural killer cells limit cardiac inflammation and fibrosis by halting eosinophil infiltration. Am. J. Pathol. 2015, 185, 847–861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abel, A.M.; Yang, C.; Thakar, M.S.; Malarkannan, S. Natural Killer Cells: Development, Maturation, and Clinical Utilization. Front. Immunol. 2018, 9, 1869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gianchecchi, E.; Delfino, D.V.; Fierabracci, A. Natural Killer Cells: Potential Biomarkers and Therapeutic Target in Autoimmune Diseases? Front. Immunol. 2021, 12, 616853. [Google Scholar] [CrossRef]
- Molofsky, A.B.; Van Gool, F.; Liang, H.-E.; Van Dyken, S.J.; Nussbaum, J.C.; Lee, J.; Bluestone, J.A.; Locksley, R.M. Interleukin-33 and Interferon-γ Counter-Regulate Group 2 Innate Lymphoid Cell Activation during Immune Perturbation. Immunity 2015, 43, 161–174. [Google Scholar] [CrossRef] [Green Version]
- Yndestad, A.; Landrø, L.; Ueland, T.; Dahl, C.P.; Flo, T.H.; Vinge, L.E.; Espevik, T.; Frøland, S.S.; Husberg, C.; Christensen, G.; et al. Increased systemic and myocardial expression of neutrophil gelatinase-associated lipocalin in clinical and experimental heart failure. Eur. Heart J. 2009, 30, 1229–1236. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Martínez, E.; Buonafine, M.; Boukhalfa, I.; Ibarrola, J.; Fernández-Celis, A.; Kolkhof, P.; Rossignol, P.; Girerd, N.; Mulder, P.; López-Andrés, N.; et al. Aldosterone Target NGAL (Neutrophil Gelatinase-Associated Lipocalin) Is Involved in Cardiac Remodeling after Myocardial Infarction through NFκB Pathway. Hypertension 2017, 70, 1148–1156. [Google Scholar] [CrossRef]
- Tawfeek, M.S.K.; Raafat, D.M.; Saad, K.; Idriss, N.K.; Sayed, S.; Fouad, D.A.; El-Houfey, A.A. Plasma levels of neutrophil gelatinase-associated lipocalin in children with heart failure. Ther. Adv. Cardiovasc. Dis. 2016, 10, 30–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avci, A.; Alizade, E.; Fidan, S.; Yesin, M.; Guler, Y.; Kargin, R.; Esen, A.M. Neutrophil/lymphocyte ratio is related to the severity of idiopathic dilated cardiomyopathy. Scand. Cardiovasc. J. SCJ 2014, 48, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Araújo, F.D.d.R.; Silva, R.M.F.d.L.; Oliveira, C.A.L.; Meira, Z.M.A. Neutrophil-to-lymphocyte ratio used as prognostic factor marker for dilated cardiomyopathy in childhood and adolescence. Ann. Pediatr. Cardiol. 2019, 12, 18–24. [Google Scholar] [CrossRef]
- Choi, J.-H.; Do, Y.; Cheong, C.; Koh, H.; Boscardin, S.B.; Oh, Y.-S.; Bozzacco, L.; Trumpfheller, C.; Park, C.G.; Steinman, R.M. Identification of antigen-presenting dendritic cells in mouse aorta and cardiac valves. J. Exp. Med. 2009, 206, 497–505. [Google Scholar] [CrossRef]
- Pistulli, R.; König, S.; Drobnik, S.; Kretzschmar, D.; Rohm, I.; Lichtenauer, M.; Fritzenwanger, M.; Mall, G.; Mall, G.; Figulla, H.-R.; et al. Decrease in dendritic cells in endomyocardial biopsies of human dilated cardiomyopathy. Eur. J. Heart Fail. 2013, 15, 974–985. [Google Scholar] [CrossRef]
- Kim, S.J.; Diamond, B. Modulation of tolerogenic dendritic cells and autoimmunity. Semin. Cell Dev. Biol. 2015, 41, 49–58. [Google Scholar] [CrossRef]
- Durai, V.; Murphy, K.M. Functions of Murine Dendritic Cells. Immunity 2016, 45, 719–736. [Google Scholar] [CrossRef] [Green Version]
- Vitali, C.; Mingozzi, F.; Broggi, A.; Barresi, S.; Zolezzi, F.; Bayry, J.; Raimondi, G.; Zanoni, I.; Granucci, F. Migratory, and not lymphoid-resident, dendritic cells maintain peripheral self-tolerance and prevent autoimmunity via induction of iTreg cells. Blood 2012, 120, 1237–1245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kania, G.; Blyszczuk, P.; Eriksson, U. Mechanisms of cardiac fibrosis in inflammatory heart disease. Trends Cardiovasc. Med. 2009, 19, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Athanassopoulos, P.; Balk, A.H.M.M.; Vaessen, L.M.B.; Caliskan, K.; Takkenberg, J.J.M.; Weimar, W.; Bogers, A.J.J.C. Blood dendritic cell levels and phenotypic characteristics in relation to etiology of end-stage heart failure: Implications for dilated cardiomyopathy. Int. J. Cardiol. 2009, 131, 246–256. [Google Scholar] [CrossRef]
- Chelko, S.P.; Asimaki, A.; Lowenthal, J.; Bueno-Beti, C.; Bedja, D.; Scalco, A.; Amat-Alarcon, N.; Andersen, P.; Judge, D.P.; Tung, L.; et al. Therapeutic Modulation of the Immune Response in Arrhythmogenic Cardiomyopathy. Circulation 2019, 140, 1491–1505. [Google Scholar] [CrossRef]
- Zhang, X.-Z.; Zhang, S.; Tang, T.-T.; Cheng, X. Bioinformatics and Immune Infiltration Analyses Reveal the Key Pathway and Immune Cells in the Pathogenesis of Hypertrophic Cardiomyopathy. Front. Cardiovasc. Med. 2021, 8, 696321. [Google Scholar] [CrossRef] [PubMed]
- El Hadi, H.; Freund, A.; Desch, S.; Thiele, H.; Majunke, N. Hypertrophic, Dilated, and Arrhythmogenic Cardiomyopathy: Where Are We? Biomedicines 2023, 11, 524. [Google Scholar] [CrossRef] [PubMed]
- Chintanaphol, M.; Orgil, B.-O.; Alberson, N.R.; Towbin, J.A.; Purevjav, E. Restrictive cardiomyopathy: From genetics and clinical overview to animal modeling. Rev. Cardiovasc. Med. 2022, 23, 108. [Google Scholar] [CrossRef]
- Negri, F.; De Luca, A.; Fabris, E.; Korcova, R.; Cernetti, C.; Grigoratos, C.; Aquaro, G.D.; Nucifora, G.; Camici, P.G.; Sinagra, G. Left ventricular noncompaction, morphological, and clinical features for an integrated diagnosis. Heart Fail. Rev. 2019, 24, 315–323. [Google Scholar] [CrossRef]
- Weintraub, R.G.; Semsarian, C.; Macdonald, P. Dilated cardiomyopathy. Lancet Lond. Engl. 2017, 390, 400–414. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Yabluchanskiy, A.; Iyer, R.P.; Cannon, P.L.; Flynn, E.R.; Jung, M.; Henry, J.; Cates, C.A.; Deleon-Pennell, K.Y.; Lindsey, M.L. Temporal neutrophil polarization following myocardial infarction. Cardiovasc. Res. 2016, 110, 51–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daseke, M.J.; Valerio, F.M.; Kalusche, W.J.; Ma, Y.; DeLeon-Pennell, K.Y.; Lindsey, M.L. Neutrophil proteome shifts over the myocardial infarction time continuum. Basic Res. Cardiol. 2019, 114, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaufman, D.B.; Shapiro, R.; Lucey, M.R.; Cherikh, W.S.; Bustami, R.T.; Dyke, D.B. Immunosuppression: Practice and trends. Am. J. Transplant. Off. J. Am. Soc. Transplant. Am. Soc. Transpl. Surg. 2004, 4 (Suppl. 9), 38–53. [Google Scholar] [CrossRef]
- Hufnagel, G.; Pankuweit, S.; Richter, A.; Schönian, U.; Maisch, B. The European Study of Epidemiology and Treatment of Cardiac Inflammatory Diseases (ESETCID). First epidemiological results. Herz 2000, 25, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.W.; O’Connell, J.B.; Herskowitz, A.; Rose, N.R.; McManus, B.M.; Billingham, M.E.; Moon, T.E. A clinical trial of immunosuppressive therapy for myocarditis. The Myocarditis Treatment Trial Investigators. N. Engl. J. Med. 1995, 333, 269–275. [Google Scholar] [CrossRef]
- Brown, C.A.; O’Connell, J.B. Implications of the Myocarditis Treatment Trial for clinical practice. Curr. Opin. Cardiol. 1996, 11, 332–336. [Google Scholar] [CrossRef]
- Frustaci, A.; Chimenti, C.; Calabrese, F.; Pieroni, M.; Thiene, G.; Maseri, A. Immunosuppressive therapy for active lymphocytic myocarditis: Virological and immunologic profile of responders versus nonresponders. Circulation 2003, 107, 857–863. [Google Scholar] [CrossRef] [Green Version]
- Frustaci, A.; Russo, M.A.; Chimenti, C. Randomized study on the efficacy of immunosuppressive therapy in patients with virus-negative inflammatory cardiomyopathy: The TIMIC study. Eur. Heart J. 2009, 30, 1995–2002. [Google Scholar] [CrossRef]
- Camargo, P.R.; Snitcowsky, R.; da Luz, P.L.; Mazzieri, R.; Higuchi, M.L.; Rati, M.; Stolf, N.; Ebaid, M.; Pileggi, F. Favorable effects of immunosuppressive therapy in children with dilated cardiomyopathy and active myocarditis. Pediatr. Cardiol. 1995, 16, 61–68. [Google Scholar] [CrossRef]
- Kleinert, S.; Weintraub, R.G.; Wilkinson, J.L.; Chow, C.W. Myocarditis in children with dilated cardiomyopathy: Incidence and outcome after dual therapy immunosuppression. J. Heart Lung Transplant. Off. Publ. Int. Soc. Heart Transplant. 1997, 16, 1248–1254. [Google Scholar]
- Camargo, P.R.; Okay, T.S.; Yamamoto, L.; Del Negro, G.M.B.; Lopes, A.A. Myocarditis in children and detection of viruses in myocardial tissue: Implications for immunosuppressive therapy. Int. J. Cardiol. 2011, 148, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Hia, C.P.P.; Yip, W.C.L.; Tai, B.C.; Quek, S.C. Immunosuppressive therapy in acute myocarditis: An 18 year systematic review. Arch. Dis. Child. 2004, 89, 580–584. [Google Scholar] [CrossRef] [Green Version]
- Bayry, J.; Misra, N.; Latry, V.; Prost, F.; Delignat, S.; Lacroix-Desmazes, S.; Kazatchkine, M.D.; Kaveri, S.V. Mechanisms of action of intravenous immunoglobulin in autoimmune and inflammatory diseases. Transfus. Clin. Biol. J. Soc. Fr. Transfus. Sang. 2003, 10, 165–169. [Google Scholar] [CrossRef]
- Maisch, B.; Pankuweit, S. Current treatment options in (peri)myocarditis and inflammatory cardiomyopathy. Herz 2012, 37, 644–656. [Google Scholar] [CrossRef] [PubMed]
- Dennert, R.; Velthuis, S.; Schalla, S.; Eurlings, L.; van Suylen, R.-J.; van Paassen, P.; Tervaert, J.W.C.; Wolffs, P.; Goossens, V.J.; Bruggeman, C.; et al. Intravenous immunoglobulin therapy for patients with idiopathic cardiomyopathy and endomyocardial biopsy-proven high PVB19 viral load. Antivir. Ther. 2010, 15, 193–201. [Google Scholar] [CrossRef]
- Heidendael, J.F.; Den Boer, S.L.; Wildenbeest, J.G.; Dalinghaus, M.; Straver, B.; Pajkrt, D. Intravenous immunoglobulins in children with new onset dilated cardiomyopathy. Cardiol. Young 2018, 28, 46–54. [Google Scholar] [CrossRef]
- Prasad, A.N.; Chaudhary, S. Intravenous immunoglobulin in children with acute myocarditis and/or early dilated cardiomyopathy. Indian Pediatr. 2014, 51, 583–584. [Google Scholar] [CrossRef]
- McNamara, D.M.; Holubkov, R.; Starling, R.C.; Dec, G.W.; Loh, E.; Torre-Amione, G.; Gass, A.; Janosko, K.; Tokarczyk, T.; Kessler, P.; et al. Controlled trial of intravenous immune globulin in recent-onset dilated cardiomyopathy. Circulation 2001, 103, 2254–2259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hazebroek, M.R.; Henkens, M.T.H.M.; Raafs, A.G.; Verdonschot, J.A.J.; Merken, J.J.; Dennert, R.M.; Eurlings, C.; Abdul Hamid, M.A.; Wolffs, P.F.G.; Winkens, B.; et al. Intravenous immunoglobulin therapy in adult patients with idiopathic chronic cardiomyopathy and cardiac parvovirus B19 persistence: A prospective, double-blind, randomized, placebo-controlled clinical trial. Eur. J. Heart Fail. 2021, 23, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Fassbender, C.; Klingel, R.; Köhler, W. Immunoadsorption for autoimmune encephalitis. Atheroscler. Suppl. 2017, 30, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Hohenstein, B.; Bornstein, S.R.; Aringer, M. Immunoadsorption for connective tissue disease. Atheroscler. Suppl. 2013, 14, 185–189. [Google Scholar] [CrossRef]
- Yamaji, K. Immunoadsorption for collagen and rheumatic diseases. Transfus. Apher. Sci. Off. J. World Apher. Assoc. Off. J. Eur. Soc. Haemapheresis 2017, 56, 666–670. [Google Scholar] [CrossRef]
- Bian, R.-T.; Wang, Z.-T.; Li, W.-Y. Immunoadsorption treatment for dilated cardiomyopathy: A PRISMA-compliant systematic review and meta-analysis. Medicine 2021, 100, e26475. [Google Scholar] [CrossRef]
- Bulut, D.; Scheeler, M.; Niedballa, L.M.; Miebach, T.; Mügge, A. Effects of immunoadsorption on endothelial function, circulating endothelial progenitor cells and circulating microparticles in patients with inflammatory dilated cardiomyopathy. Clin. Res. Cardiol. Off. J. Ger. Card. Soc. 2011, 100, 603–610. [Google Scholar] [CrossRef]
- Dandel, M.; Englert, A.; Wallukat, G.; Riese, A.; Knosalla, C.; Stein, J.; Hetzer, R. Immunoadsorption can improve cardiac function in transplant candidates with non-ischemic dilated cardiomyopathy associated with diabetes mellitus. Atheroscler. Suppl. 2015, 18, 124–133. [Google Scholar] [CrossRef]
- Dörffel, W.V.; Felix, S.B.; Wallukat, G.; Brehme, S.; Bestvater, K.; Hofmann, T.; Kleber, F.X.; Baumann, G.; Reinke, P. Short-term hemodynamic effects of immunoadsorption in dilated cardiomyopathy. Circulation 1997, 95, 1994–1997. [Google Scholar] [CrossRef]
- Dörffel, W.V.; Wallukat, G.; Dörffel, Y.; Felix, S.B.; Baumann, G. Immunoadsorption in idiopathic dilated cardiomyopathy, a 3-year follow-up. Int. J. Cardiol. 2004, 97, 529–534. [Google Scholar] [CrossRef]
- Müller, J.; Wallukat, G.; Dandel, M.; Bieda, H.; Brandes, K.; Spiegelsberger, S.; Nissen, E.; Kunze, R.; Hetzer, R. Immunoglobulin adsorption in patients with idiopathic dilated cardiomyopathy. Circulation 2000, 101, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Staudt, A.; Hummel, A.; Ruppert, J.; Dörr, M.; Trimpert, C.; Birkenmeier, K.; Krieg, T.; Staudt, Y.; Felix, S.B. Immunoadsorption in dilated cardiomyopathy: 6-month results from a randomized study. Am. Heart J. 2006, 152, 712.e1–712.e6. [Google Scholar] [CrossRef] [PubMed]
- Felix, S.B.; Staudt, A. Immunoadsorption as treatment option in dilated cardiomyopathy. Autoimmunity 2008, 41, 484–489. [Google Scholar] [CrossRef]
- Ohlow, M.-A.; Brunelli, M.; Schreiber, M.; Lauer, B. Therapeutic effect of immunoadsorption and subsequent immunoglobulin substitution in patients with dilated cardiomyopathy: Results from the observational prospective Bad Berka Registry. J. Cardiol. 2017, 69, 409–416. [Google Scholar] [CrossRef] [Green Version]
- Felix, S.B.; Staudt, A.; Dörffel, W.V.; Stangl, V.; Merkel, K.; Pohl, M.; Döcke, W.D.; Morgera, S.; Neumayer, H.H.; Wernecke, K.D.; et al. Hemodynamic effects of immunoadsorption and subsequent immunoglobulin substitution in dilated cardiomyopathy: Three-month results from a randomized study. J. Am. Coll. Cardiol. 2000, 35, 1590–1598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bulut, D.; Scheeler, M.; Wichmann, T.; Börgel, J.; Miebach, T.; Mügge, A. Effect of protein A immunoadsorption on T cell activation in patients with inflammatory dilated cardiomyopathy. Clin. Res. Cardiol. Off. J. Ger. Card. Soc. 2010, 99, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Ameling, S.; Herda, L.R.; Hammer, E.; Steil, L.; Teumer, A.; Trimpert, C.; Dörr, M.; Kroemer, H.K.; Klingel, K.; Kandolf, R.; et al. Myocardial gene expression profiles and cardiodepressant autoantibodies predict response of patients with dilated cardiomyopathy to immunoadsorption therapy. Eur. Heart J. 2013, 34, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, G.; Dörr, M.; Sappa, P.K.; Ameling, S.; Dhople, V.; Steil, L.; Klingel, K.; Empen, K.; Beug, D.; Völker, U.; et al. Endomyocardial proteomic signature corresponding to the response of patients with dilated cardiomyopathy to immunoadsorption therapy. J. Proteom. 2017, 150, 121–129. [Google Scholar] [CrossRef]
- Bulut, D.; Creutzenberg, G.; Mügge, A. The number of regulatory T cells correlates with hemodynamic improvement in patients with inflammatory dilated cardiomyopathy after immunoadsorption therapy. Scand. J. Immunol. 2013, 77, 54–61. [Google Scholar] [CrossRef]
- Kallwellis-Opara, A.; Staudt, A.; Trimpert, C.; Noutsias, M.; Kühl, U.; Pauschinger, M.; Schultheiss, H.-P.; Grube, M.; Böhm, M.; Baumann, G.; et al. Immunoadsorption and subsequent immunoglobulin substitution decreases myocardial gene expression of desmin in dilated cardiomyopathy. J. Mol. Med. 2007, 85, 1429–1435. [Google Scholar] [CrossRef] [Green Version]
- Ameling, S.; Bhardwaj, G.; Hammer, E.; Beug, D.; Steil, L.; Reinke, Y.; Weitmann, K.; Grube, M.; Trimpert, C.; Klingel, K.; et al. Changes of myocardial gene expression and protein composition in patients with dilated cardiomyopathy after immunoadsorption with subsequent immunoglobulin substitution. Basic Res. Cardiol. 2016, 111, 53. [Google Scholar] [CrossRef]
- Rong, S.-L.; Wang, Z.-K.; Zhou, X.-D.; Wang, X.-L.; Yang, Z.-M.; Li, B. Efficacy and safety of stem cell therapy in patients with dilated cardiomyopathy: A systematic appraisal and meta-analysis. J. Transl. Med. 2019, 17, 221. [Google Scholar] [CrossRef] [PubMed]
- Hare, J.M.; DiFede, D.L.; Rieger, A.C.; Florea, V.; Landin, A.M.; El-Khorazaty, J.; Khan, A.; Mushtaq, M.; Lowery, M.H.; Byrnes, J.J.; et al. Randomized Comparison of Allogeneic Versus Autologous Mesenchymal Stem Cells for Nonischemic Dilated Cardiomyopathy: POSEIDON-DCM Trial. J. Am. Coll. Cardiol. 2017, 69, 526–537. [Google Scholar] [CrossRef] [PubMed]
- Kelkar, A.A.; Butler, J.; Schelbert, E.B.; Greene, S.J.; Quyyumi, A.A.; Bonow, R.O.; Cohen, I.; Gheorghiade, M.; Lipinski, M.J.; Sun, W.; et al. Mechanisms Contributing to the Progression of Ischemic and Nonischemic Dilated Cardiomyopathy: Possible Modulating Effects of Paracrine Activities of Stem Cells. J. Am. Coll. Cardiol. 2015, 66, 2038–2047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagaya, N.; Kangawa, K.; Itoh, T.; Iwase, T.; Murakami, S.; Miyahara, Y.; Fujii, T.; Uematsu, M.; Ohgushi, H.; Yamagishi, M.; et al. Transplantation of mesenchymal stem cells improves cardiac function in a rat model of dilated cardiomyopathy. Circulation 2005, 112, 1128–1135. [Google Scholar] [CrossRef] [Green Version]
- Vrtovec, B.; Poglajen, G.; Sever, M.; Lezaic, L.; Domanovic, D.; Cernelc, P.; Haddad, F.; Torre-Amione, G. Effects of intracoronary stem cell transplantation in patients with dilated cardiomyopathy. J. Card. Fail. 2011, 17, 272–281. [Google Scholar] [CrossRef]
- Tompkins, B.A.; Rieger, A.C.; Florea, V.; Banerjee, M.N.; Natsumeda, M.; Nigh, E.D.; Landin, A.M.; Rodriguez, G.M.; Hatzistergos, K.E.; Schulman, I.H.; et al. Comparison of Mesenchymal Stem Cell Efficacy in Ischemic Versus Nonischemic Dilated Cardiomyopathy. J. Am. Heart Assoc. 2018, 7, e008460. [Google Scholar] [CrossRef] [Green Version]
- Mao, C.; Hou, X.; Wang, B.; Chi, J.; Jiang, Y.; Zhang, C.; Li, Z. Intramuscular injection of human umbilical cord-derived mesenchymal stem cells improves cardiac function in dilated cardiomyopathy rats. Stem Cell Res. Ther. 2017, 8, 18. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Zhou, G.; Chen, Y.; Liu, S.; Chen, F.; Xie, L.; Wang, W.; Zhang, Y.; Wang, T.; Lai, X.; et al. Human umbilical cord mesenchymal stem cells alleviate interstitial fibrosis and cardiac dysfunction in a dilated cardiomyopathy rat model by inhibiting TNF-α and TGF-β1/ERK1/2 signaling pathways. Mol. Med. Rep. 2018, 17, 71–78. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Zhou, Y.; Lai, X.; Zhou, G.; Wang, H.; Feng, X.; Chen, Y.; Wu, Y.; Wang, T.; Ma, L. Human Umbilical Cord Mesenchymal Stem Cells Alleviate Myocardial Endothelial-Mesenchymal Transition in a Rat Dilated Cardiomyopathy Model. Transplant. Proc. 2019, 51, 936–941. [Google Scholar] [CrossRef]
- Orphanou, N.; Papatheodorou, E.; Anastasakis, A. Dilated cardiomyopathy in the era of precision medicine: Latest concepts and developments. Heart Fail. Rev. 2022, 27, 1173–1191. [Google Scholar] [CrossRef] [PubMed]
- McMurray, J.J.V.; Packer, M.; Desai, A.S.; Gong, J.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.C.; Solomon, S.D.; Swedberg, K.; et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 2014, 371, 993–1004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, A.L.; Ziesche, S.; Yancy, C.W.; Carson, P.; Ferdinand, K.; Taylor, M.; Adams, K.; Olukotun, A.Y.; Ofili, E.; Tam, S.W.; et al. Early and sustained benefit on event-free survival and heart failure hospitalization from fixed-dose combination of isosorbide dinitrate/hydralazine: Consistency across subgroups in the African-American Heart Failure Trial. Circulation 2007, 115, 1747–1753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Digitalis Investigation Group. The effect of digoxin on mortality and morbidity in patients with heart failure. N. Engl. J. Med. 1997, 336, 525–533. [Google Scholar] [CrossRef]

| Reference | ||
|---|---|---|
| Endothelial cells | Secretes adhesion molecules (P-selectin, E-selectin, ICAM-1, and VCAM-1), which promote the migration of immune cells. | [43,44,45] |
| Cardiomyocytes | Releasing DAMPs, triggering the production of various chemokines, such as CCL-2, CCL-5, CCL-7, CXCL-1, CXCL-2, and CXCL-8. | [47,48] |
| Myofibroblasts | Promotes tissue development, wound repair, and fibrosis. | [51,52] |
| Immune cells | ||
| Monocytes/Macrophages | Secreted cytokines (MCP-1, IL-6, IL-1β, TNF-α, and TGF-β) regulate inflammatory responses and maintain cardiac homeostasis. | [70,71,72,74,82,83] |
| T cells |
| [99,101,102,103,104,125] |
| B cells |
| [142,143,144,150,151,152] |
| Natural killer cells | Against acute viral pathogens related to myocarditis and cardiac fibrosis. | [28,174,175] |
| Neutrophil | Secreting neutrophil gelatinase-related lipocalin and facilitating endothelial cell activation and cytokine release through the formation of neutrophil extracellular traps. | [40,163] |
| Dendritic cells | As antigen-presenting cells, they transport cellular debris and antigens to T cells. | [30,167] |
| Genetic mutations | LMNA, TTN, MYH7, PLN, DSP, and SCN5a. | [12,13,14] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, E.; Zhou, R.; Li, T.; Hua, Y.; Zhou, K.; Li, Y.; Luo, S.; An, Q. The Molecular Role of Immune Cells in Dilated Cardiomyopathy. Medicina 2023, 59, 1246. https://doi.org/10.3390/medicina59071246
Wang E, Zhou R, Li T, Hua Y, Zhou K, Li Y, Luo S, An Q. The Molecular Role of Immune Cells in Dilated Cardiomyopathy. Medicina. 2023; 59(7):1246. https://doi.org/10.3390/medicina59071246
Chicago/Turabian StyleWang, Enping, Ruofan Zhou, Tiange Li, Yimin Hua, Kaiyu Zhou, Yifei Li, Shuhua Luo, and Qi An. 2023. "The Molecular Role of Immune Cells in Dilated Cardiomyopathy" Medicina 59, no. 7: 1246. https://doi.org/10.3390/medicina59071246







