A Systematic Review and Meta-Analysis Comparing the Effectiveness of Transversus Abdominis Plane Block and Caudal Block for Relief of Postoperative Pain in Children Who Underwent Lower Abdominal Surgeries
Abstract
:1. Introduction
2. Methods
2.1. Electronic Literature Search Strategy
2.2. Data Extraction
2.3. Criteria for Considering Studies for the Review
2.4. Types of Studies
2.5. Data Extraction and Management
2.6. Assessment of the Risk of Bias in the Included Studies
2.7. Assessment of Heterogeneity
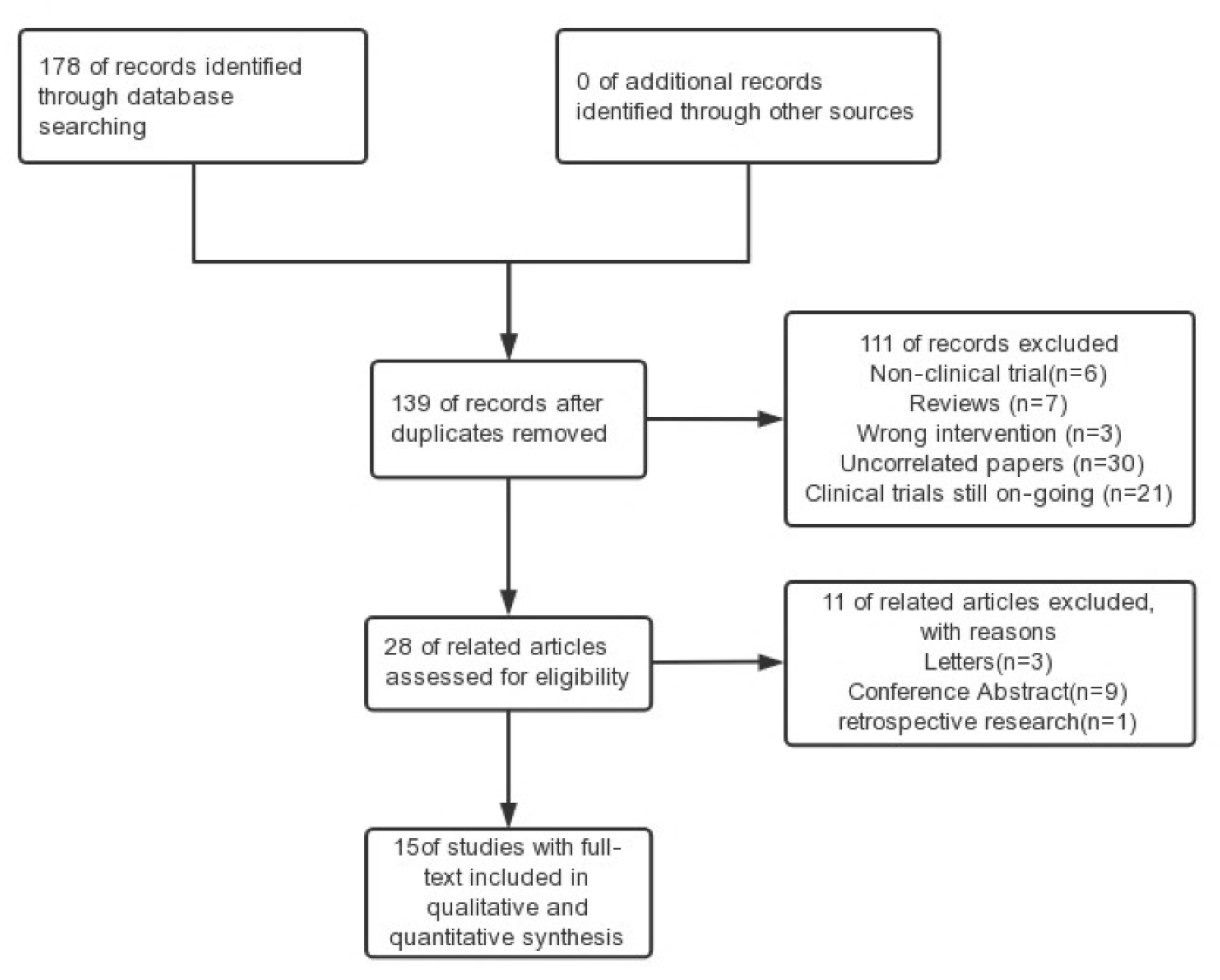
3. Results
3.1. Risk of Bias
3.2. Pain Scores
3.3. Postoperative Pain Scores at 1 h
3.4. Postoperative Pain Scores at 6 h
3.5. Postoperative Pain Scores at 12 h
3.6. Postoperative Pain Scores at 24 h
3.7. Additional Analgesic Requirement
3.8. Total Dose of Rescue Analgesic Given in 24 h
3.9. The Mean Duration of Analgesia
3.10. Intraoperative and Postoperative Hemodynamic Conditions
3.11. Parents’ Satisfaction
3.12. Adverse Effects
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Database | Search Strategy | Result (Numbers) |
|---|---|---|
| PubMed | #1“caudal block”[Title/Abstract] OR “caudal epidural block”[Title/Abstract] OR “CB”[Title/Abstract] OR “CEB”[Title/Abstract] #2“transversus abdominis plane block”[Title/Abstract] OR “TAP”[Title/Abstract] OR “TAPB”[Title/Abstract] #3 #1 AND #2 | 47 |
| Web of Science | transversus abdominis plane block (Theme) AND caudal block (Theme) | 50 |
| The Cochrane Library | #1 (Transversus Abdominis Plane):ti,ab,kw AND (Caudal Block):ti,ab,kw (Word variations have been searched) | 63 |
| Embase | #1 (transversus abdominis plane block or TAPB).ti. (1121) #2 (CB or caudal block).ti. (1988) #3 (transversus abdominis plane block or TAPB).ab. (622) #4 (CB or caudal block).ab. (29227) #5 #1 or #3 (1461) #6 #2 or 4 (29954) #7 #5 and 6 (17) | 17 |
References
- Albrecht, S.; Fechner, J.; Geisslinger, G.; Maass, A.B.; Upadhyaya, B.; Moecke, H.; Haigh, C.; Schüttler, J. Postoperative pain control following remifentanil-based anaesthesia for major abdominal surgery. Anaesthesia 2000, 55, 315–322. [Google Scholar] [CrossRef]
- Lonnqvist, P.A.; Morton, N.S. Postoperative analgesia in infants and children. Br. J. Anaesth. 2005, 95, 59–68. [Google Scholar] [CrossRef]
- Wiegele, M.; Marhofer, P.; Lönnqvist, P.A. Caudal epidural blocks in paediatric patients: A review and practical considerations. Br. J. Anaesth. 2019, 122, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Go, R.; Huang, Y.Y.; Weyker, P.D.; Webb, C.A. Truncal blocks for perioperative pain management: A review of the literature and evolving techniques. Pain Manag. 2016, 6, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Dontukurthy, S.; Mofidi, R. The Role of Interfascial Plane Blocks in Paediatric Regional Anaesthesia: A Narrative Review of Current Perspectives and Updates. Anesthesiol. Res. Pract. 2020, 2020, 8892537. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.D.; Suresh, S. Applications of regional anaesthesia in paediatrics. Br. J. Anaesth. 2013, 111 (Suppl. S1), i114–i124. [Google Scholar] [CrossRef]
- Dobereiner, E.F.; Cox, R.G.; Ewen, A.; Lardner, D.R. Evidence-based clinical update: Which local anesthetic drug for pediatric caudal block provides optimal efficacy with the fewest side effects? Can. J. Anaesth. 2010, 57, 1102–1110. [Google Scholar] [CrossRef]
- Apan, A.; Cuvas, O. Caudal block in adults: New horizons with ultrasound. Minerva Anestesiol. 2013, 79, 1332–1333. [Google Scholar] [CrossRef]
- Rafi, A.N. Abdominal field block: A new approach via the lumbar triangle. Anaesthesia 2001, 56, 1024–1026. [Google Scholar] [CrossRef]
- Tran, D.Q.; Bravo, D.; Leurcharusmee, P.; Neal, J.M. Transversus Abdominis Plane Block: A Narrative Review. Anesthesiology 2019, 131, 1166–1190. [Google Scholar] [CrossRef]
- Bryskin, R.B.; Londergan, B.; Wheatley, R.; Heng, R.; Lewis, M.; Barraza, M.; Mercer, E.; Ye, G. Transversus Abdominis Plane Block Versus Caudal Epidural for Lower Abdominal Surgery in Children: A Double-Blinded Randomized Controlled Trial. Anesth. Analg. 2015, 121, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Hozo, S.P.; Djulbegovic, B.; Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 2005, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dogra, N.; Gupta, A.; Aggarwal, S. Ultrasound-guided transversus abdominis plane block versus caudal block for postoperative analgesia in children undergoing inguinal hernia surgery: A comparative study. J. Anaesthesiol. Clin. Pharmacol. 2020, 36, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Polat, H.; Şentürk, E.; Karadeniz, M.S.; Bingül, E.S.; Emre Demirel, E.; Erginel, B.; Tuğrul, K.M. Effects of ultrasound guided caudal epidural and transversus abdominis plane block on postoperative analgesia in pediatric inguinal hernia repair surgeries. J. Pediatr. Urol. 2023, 19, 213.e211–213.e217. [Google Scholar] [CrossRef] [PubMed]
- Sethi, N.; Pant, D.; Dutta, A.; Koul, A.; Sood, J.; Chugh, P.T. Comparison of caudal epidural block and ultrasonography-guided transversus abdominis plane block for pain relief in children undergoing lower abdominal surgery. J. Clin. Anesth. 2016, 33, 322–329. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.P.; Wang, H.T.; Xu, Y.C.; Lv, H.M.; Yu, Y.; Wang, P.; Pei, X.D.; Zhao, J.W.; Nan, Z.H.; et al. Ultrasound-guided quadratus lumborum block provided more effective analgesia for children undergoing lower abdominal laparoscopic surgery: A randomized clinical trial. Surg. Endosc. 2022, 36, 9046–9053. [Google Scholar] [CrossRef]
- Alkayssi, H. Comparison between Caudal and TAP Blocks Post-Inguinal Surgery Analgesia in Children. NeuroQuantology 2022, 20, 278–283. [Google Scholar] [CrossRef]
- Alsadek, W.M.; Al-Gohari, M.M.; Elsonbaty, M.I.; Nassar, H.M.; Alkonaiesy, R.M. Ultrasound guided TAP block versus ultrasound guided caudal block for pain relief in children undergoing lower abdominal surgeries. Egypt. J. Anaesth. 2015, 31, 155–160. [Google Scholar] [CrossRef]
- Ganesh, B.; Swain, S.; Banerjee, S. Comparison of Ultrasound-Guided Transversus Abdominis Plane Block and Caudal Epidural Block for Pain Relief in Children Undergoing Infraumbilical Surgeries. Anesth. Essays Res. 2021, 15, 161–166. [Google Scholar] [CrossRef]
- Ghodke, S.; Ravindra, M.; Rn, H. A comparative study of caudal block and ultrasound-guided transversus abdominis plane block with levobupivacaine and dexamethasone as additive in extraperitoneal lower abdominal surgeries in pediatrics. Asian J. Pharm. Clin. Res. 2021, 14, 173–176. [Google Scholar] [CrossRef]
- İpek, C.B.; Kara, D.; Yılmaz, S.; Yeşiltaş, S.; Esen, A.; Dooply, S.; Karaaslan, K.; Türköz, A. Comparison of ultrasound-guided transversus abdominis plane block, quadratus lumborum block, and caudal epidural block for perioperative analgesia in pediatric lower abdominal surgery. Turk. J. Med. Sci. 2019, 49, 1395–1402. [Google Scholar] [CrossRef]
- Rautela, M.S.; Sahni, A.; Dalal, N. Is Ultrasound-Guided Transversus Abdominis Plane Block Superior to a Caudal Epidural or Wound Infiltration for Intraoperative and Postoperative Analgesia in Children Undergoing Unilateral Infraumbilical Surgery? A Double-blind Randomized Trial. J. Indian Assoc. Pediatr. Surg. 2022, 27, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.; Bhandary, A.; Shetty, S.R.; Harish, B.G. Comparative study between an ultrasound-guided transversus abdominis plane block and an ultrasound-guided caudal block for postoperative analgesia in children undergoing lower abdominal surgeries—A prospective randomised study. S. Afr. J. Anaesth. Analg. 2021, 27, 223–227. [Google Scholar] [CrossRef]
- Sahin, L.; Soydinc, M.H.; Sen, E.; Cavus, O.; Sahin, M. Comparison of 3 different regional block techniques in pediatric patients. A prospective randomized single-blinded study. Saudi Med. J. 2017, 38, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Vinukonda, M.; Kumar, K.S.; Suggala, K.K. A comparative randomized study of USG guided transversus abdominis plane block versus USG guided caudal block for post-operative analgesia in paediatric unilateral open inguinal hernia repair. Eur. J. Mol. Clin. Med. 2022, 9, 10. [Google Scholar]
- Kodali, V.R.K.; Kandimalla, A.; Vakamudi, M. Comparison of Analgesic Efficacy of Ultrasound-Guided Transversus Abdominus Plane Block and Caudal Block for Inguinal Hernia Repair in Pediatric Population: A Single-Blinded, Randomized Controlled Study. Anesth. Essays Res. 2020, 14, 478–484. [Google Scholar] [CrossRef]
- Hicks, C.L.; von Baeyer, C.L.; Spafford, P.A.; van Korlaar, I.; Goodenough, B. The Faces Pain Scale-Revised: Toward a common metric in pediatric pain measurement. Pain 2001, 93, 173–183. [Google Scholar] [CrossRef]
- Desai, N.; El-Boghdadly, K.; Albrecht, E. Epidural vs. transversus abdominis plane block for abdominal surgery—A systematic review, meta-analysis and trial sequential analysis. Anaesthesia 2021, 76, 101–117. [Google Scholar] [CrossRef]
- El-Boghdadly, K.; Desai, N.; Halpern, S.; Blake, L.; Odor, P.M.; Bampoe, S.; Carvalho, B.; Sultan, P. Quadratus lumborum block vs. transversus abdominis plane block for caesarean delivery: A systematic review and network meta-analysis. Anaesthesia 2021, 76, 393–403. [Google Scholar] [CrossRef]
- Shanthanna, H.; Singh, B.; Guyatt, G. A systematic review and meta-analysis of caudal block as compared to noncaudal regional techniques for inguinal surgeries in children. BioMed Res. Int. 2014, 2014, 890626. [Google Scholar] [CrossRef]
- Hafeman, M.; Greenspan, S.; Rakhamimova, E.; Jin, Z.; Moore, R.P.; Al Bizri, E. Caudal block vs. transversus abdominis plane block for pediatric surgery: A systematic review and meta-analysis. Front. Pediatr. 2023, 11, 1173700. [Google Scholar] [CrossRef] [PubMed]
- Kelley-Quon, L.I.; Cho, J.; Strong, D.R.; Miech, R.A.; Barrington-Trimis, J.L.; Kechter, A.; Leventhal, A.M. Association of Nonmedical Prescription Opioid Use with Subsequent Heroin Use Initiation in Adolescents. JAMA Pediatr. 2019, 173, e191750. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.; Carey, M. A case of liver trauma with a blunt regional anesthesia needle while performing transversus abdominis plane block. Reg. Anesth. Pain Med. 2008, 33, 274–275. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, P.; Chadwick, M. Liver trauma secondary to ultrasound-guided transversus abdominis plane block. Br. J. Anaesth. 2010, 104, 509–510. [Google Scholar] [CrossRef]
- Aleman, R.; Blanco, D.G.; Funes, D.R.; Montorfano, L.; Semien, G.; Szomstein, S.; Lo Menzo, E.; Rosenthal, R.J. Does Transverse Abdominis Plane Block Increase the Risk of Postoperative Urinary Retention after Inguinal Hernia Repair? J. Soc. Laparosc. Robot. Surg. 2021, 25, e2021.00015. [Google Scholar] [CrossRef]
- Salaria, O.N.; Kannan, M.; Kerner, B.; Goldman, H. A Rare Complication of a TAP Block Performed after Caesarean Delivery. Case Rep. Anesthesiol. 2017, 2017, 1072576. [Google Scholar] [CrossRef]
- Kao, S.C.; Lin, C.S. Caudal Epidural Block: An Updated Review of Anatomy and Techniques. BioMed Res. Int. 2017, 2017, 9217145. [Google Scholar] [CrossRef]
- Lundblad, M.; Lönnqvist, P.A.; Eksborg, S.; Marhofer, P. Segmental distribution of high-volume caudal anesthesia in neonates, infants, and toddlers as assessed by ultrasonography. Paediatr. Anaesth. 2011, 21, 121–127. [Google Scholar] [CrossRef]
- Lundblad, M.; Eksborg, S.; Lönnqvist, P.A. Secondary spread of caudal block as assessed by ultrasonography. Br. J. Anaesth. 2012, 108, 675–681. [Google Scholar] [CrossRef]
- Ecoffey, C.; Lacroix, F.; Giaufré, E.; Orliaguet, G.; Courrèges, P. Epidemiology and morbidity of regional anesthesia in children: A follow-up one-year prospective survey of the French-Language Society of Paediatric Anaesthesiologists (ADARPEF). Paediatr. Anaesth. 2010, 20, 1061–1069. [Google Scholar] [CrossRef]
- Tsuchiya, N.; Ichizawa, M.; Yoshikawa, Y.; Shinomura, T. Comparison of ropivacaine with bupivacaine and lidocaine for ilioinguinal block after ambulatory inguinal hernia repair in children. Paediatr. Anaesth. 2004, 14, 468–470. [Google Scholar] [CrossRef] [PubMed]









| Study | Location | Age (Years) | Sample Size (TAPB/CB) | Type of Surgery | Anesthetic Technology | Background Analgesia Rescue Analgesia | Follow Up | Jadad Score | |
|---|---|---|---|---|---|---|---|---|---|
| TAPB | CB | ||||||||
| Kumar, A. et al., 2020 [13] | India | 2–8 | 56/56 | Inguinal hernia surgery | 0.5 mL/kg of 0.2% ropivacaine | 1 mL/kg of 0.2% ropivacaine | Syrup paracetamol 10 mg/kg orally | 24 h | 6 |
| Polat, H. et al., 2022 [14] | Turkey | 1–7 | 30/30 | Pediatric inguinal hernia repair surgeries | 0.5 mL/kg of 0.25% bupivacaine | 1 mL/kg bupivacaine 0.25% | Paracetamol IV or per oral was administered | 24 h | 7 |
| Sethi, N. et al., 2016 [15] | India | 2–6 | 34/36 | Lower abdominal surgery | 0.5 mL/kg of 0.25% bupivacaine | Bupivacaine (0.75 mL/kg of 0.25%) | Oral paracetamol 20 mg/kg | 4 | |
| Zhang, Y. et al., 2022 [16] | China | 1–12 | 60/60 | Lower abdominal laparoscopic surgery | Ropivacaine | Ropivacaine | Tramadol | 24 h | 7 |
| Alkayssi, H. et al., 2022 [17] | Iraq | 3–9 | 20/20 | Post-inguinal surgery (elective inguinal hernia repair surgery) | Bupivacaine 0.25% (0.5 mL/kg) | Bupivacaine 0.25% (1 mL/kg) | Paracetamol 10 mg/kg | 12 h | 5 |
| Alsadek, W.M. et al., 2015 [18] | Egypt | 2–7 | 20/20 | Lower abdominal surgeries | A bolus of 0.5 mL/kg bupivacaine 0.25% | 1.0 mL/kg bupivacaine 0.25% | Paracetamol (acetaminophen) suppository 15 mg/kg | 12 h | 6 |
| Bryskin, R.B. et al., 2015 [11] | America | 1–9 | 24/21 | Lower abdominal surgery | 0.5 mL/kg of 0.25% bupivacaine + epinephrine 1:200,000 | 1 mL/kg of 0.25% bupivacaine + epinephrine 1:200,000 | Morphine 0.05 mg/kg IV every 2 h as needed for moderate to severe pain | 24 h | 7 |
| Ganesh, B. et al., 2021 [19] | India | 2–7 | 25/25 | Infraumbilical surgeries | 0.5 mL·kg−1 of 0.2% Ropivacaine | 1 mL·kg−1 of 0.2% ropivacaine | Injection paracetamol 20 mg/kg, intravenous fentanyl 1 μg/kg | 24 h | 7 |
| Ghodke, S.M. et al., 2021 [20] | India | 1–8 | 25/25 | Extraperitoneal lower abdominal surgeries | 0.5 mL/kg of 0.2% levobupivacaine and 0.1 mg/kg dexamethasone. | 1 mL/kg of 0.2% levobupivacaine and 0.1 mg/kg dexamethasone | Paracetamol 15 mg/kg IV | 24 h | 4 |
| İpek, C.B. et al., 2019 [21] | Turkey | 0.5–14 | 29/30 | Lower abdominal surgery | 0.5 mL kg−1 of 0.25% bupivacaine solution | 0.5 mL kg−1 of 0.25% bupivacaine solution | 10 mg kg−1 of ibuprofen syrup | 24 h | 5 |
| Rautela, M.S. et al., 2022 [22] | India | 3–10 | 40/40 | Unilateral infraumbilical surgery | 0.5 mL/kg of 0.25% bupivacaine | 0.75 mL/kg of 0.25% bupivacaine | Syrup paracetamol 15 mg/kg body weight or injection paracetamol 10 mg/kg body weight | 24 h | 6 |
| Reddy, A. et al., 2021 [23] | India | 2–10 | 31/31 | Lower abdominal surgeries | 0.25% bupivacaine 0.5 mL/kg with 1 µg/kg dexmedetomidine. | 0.25% bupivacaine 0.5 mL/kg with 1 µg/kg dexmedetomidine | NM | 24 h | 6 |
| Sahin, L. et al., 2017 [24] | America | 1–7 | 30/30 | Unilateral lower abdominal surgery | 0.25% levobupivacaine added to 1/200,000 adrenaline at a dose of 0.5 mL/kg | 0.25% levobupivacaine added to 1/200,000 adrenaline at a dose of 0.7 mL/kg | NM | 24 h | 4 |
| Vinukonda, M.K. et al., 2022 [25] | India | 2–8 | 30/30 | Unilateral open inguinal hernia repair | 0.5 mL/kg 0.25% bupivacaine | 0.75/kg 0.2% bupivacaine | Paracetamol IV 15 mg/kg | 24 h | 4 |
| Kodali, V.R.K. et al., 2020 [26] | India | 0.5–8 | 31/31 | Inguinal hernia repair | 0.5 mL·kg−1 of 0.25% bupivacaine | 1 mL·kg−1 of 0.25% bupivacaine | Intravenous paracetamol was given at a dose of 7.5 mg·kg−1 for children weighing < 10 kg and 15 mg·kg−1 for children weighing > 10 kg | 24 h | 6 |
| Outcome | Studies | Participants | Statistical Method | Effect Estimate (95% CI) | p-Value |
|---|---|---|---|---|---|
| Pain score at 1 h (CHEOPS score) | 3 | 212 | SMD (IV,RE) | 1.83 (−1.46 to 5.12) | 0.28 |
| Pain score at 1 h (FLACC score) | 7 | 459 | SMD (IV,RE) | 0.35 (−0.54 to 1.24) | 0.44 |
| Pain score at 6 h (FLACC score) | 8 | 519 | SMD (IV,RE) | −0.10 (−0.44 to −0.23) | 0.55 |
| Pain score at 12 h (FLACC score) | 7 | 459 | SMD (IV,RE) | −0.02 (−0.45 to −0.40) | 0.93 |
| Pain score at 24 h (FLACC score) | 7 | 469 | SMD (IV,RE) | −0.66 (−1.57 to −0.25) | 0.15 |
| Additional analgesic requirement | 7 | 439 | OR (MH,RE) | 0.25 (0.09 to 0.63) | 0.004 |
| Total dose of rescue analgesic given in 24 h | 9 | 673 | SMD (IV,RE) | −0.37 (−1.33 to −0.58) | 0.44 |
| The mean duration of analgesia | 9 | 671 | SMD (IV,RE) | 1.29 (0.01 to 2.57) | 0.05 |
| Parents’ satisfaction | 4 | 282 | SMD (IV,RE) | 0.44 (−0.12 to 1.0) | 0.12 |
| Adverse effects | 5 | 352 | OR (MH,RE) | 0.78 (0.22 to 2.82) | 0.70 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, D.; Sun, Y.; Gong, F.; Yin, Y.; Wang, Y. A Systematic Review and Meta-Analysis Comparing the Effectiveness of Transversus Abdominis Plane Block and Caudal Block for Relief of Postoperative Pain in Children Who Underwent Lower Abdominal Surgeries. Medicina 2023, 59, 1527. https://doi.org/10.3390/medicina59091527
Xiao D, Sun Y, Gong F, Yin Y, Wang Y. A Systematic Review and Meta-Analysis Comparing the Effectiveness of Transversus Abdominis Plane Block and Caudal Block for Relief of Postoperative Pain in Children Who Underwent Lower Abdominal Surgeries. Medicina. 2023; 59(9):1527. https://doi.org/10.3390/medicina59091527
Chicago/Turabian StyleXiao, Dan, Yiyuan Sun, Fang Gong, Yu Yin, and Yue Wang. 2023. "A Systematic Review and Meta-Analysis Comparing the Effectiveness of Transversus Abdominis Plane Block and Caudal Block for Relief of Postoperative Pain in Children Who Underwent Lower Abdominal Surgeries" Medicina 59, no. 9: 1527. https://doi.org/10.3390/medicina59091527





