Abstract
Ovarian cancer, which ranks eighth among global female cancers and fifth in fatality, poses a significant health challenge owing to its asymptomatic early stages. Understanding the pathogenesis requires extensive research. Recent studies have emphasized the role of the gut and cervicovaginal microbiota in ovarian cancer. This review explores the current understanding of the relationship between the microbiome and ovarian cancer, considering the potential of biomarkers in the serum and various tissues. Insights into the influence of the microbiome on treatments, including surgery and chemotherapy, open doors to innovative approaches, such as fecal microbiome transplantation. This synthesis of recent findings provides crucial insights into the intricate interplay between the microbiome and ovarian cancer, thereby shaping diagnostic and treatment strategies.
1. Introduction
The term “microbiome” encompasses the complete genetic information of the microorganisms inhabiting the human body, surpassing eight million species, which is 360 times more extensive than the human genome [1]. Under normal conditions, indigenous microbiota thrive in diverse regions of the human body, with at least 10,000 species [1]. Notably, the skin hosts approximately 500 species of bacteria and yeast, whereas the gastrointestinal microbiome comprises 500–1000 species, including bacteria, fungi, archaea, and protozoa [1]. Indigenous microbiota reside in areas such as the upper respiratory tract, external genitalia, and vagina [1]. The microbiome plays a pivotal role in various physiological and pathological processes and is intricately linked to the immune system [2]. Recent studies have revealed associations between sleep disorders [3], depression [3], aging [4], and cancer.
Certain microbes, such as Helicobacter pylori in the stomach and Fusobacterium nucleatum in the colon, have been identified as contributors to the risk and progression of stomach cancer and colorectal cancers, respectively [5]. Moreover, studies have revealed that microbiome dysbiosis, even in distant regions, influences cancer progression; for instance, gut microbiota dysbiosis affects hepatocellular carcinoma progression [6] and contributes to breast cancer development [7].
According to the 2020 GLOBOCAN Global Cancer Women’s Cancer Data, ovarian cancer has an incidence rate of 3.4% and mortality rate of 4.7% [8]. Each year, over 300,000 women develop ovarian cancer, which causes approximately 152,000 fatalities. Ovarian cancer is the most lethal gynecological cancer, ranking eighth in global female cancer incidence and fifth in terms of mortality. This high mortality rate is attributed to frequent late-stage diagnoses due to the absence of specific symptoms or definitive diagnostic biomarkers. Although risk factors, such as family history, hyperovulation, endometriosis, and dietary habits, have been known, much remains unclear [9]. Despite its low survival rate, a comprehensive understanding of ovarian cancer remains elusive and warrants further research.
Against this backdrop, studies exploring the nexus between the microbiome and ovarian cancer have recently emerged, aligning with analogous investigations on other cancer types. The cervical vaginal microbiome, which is close to the ovaries, and the gut microbiome, which is already known to be associated with various cancers, are of particular interest. This article reviews the latest discoveries regarding the connection between the microbiome and ovarian cancer, and delves into its potential applications in ovarian cancer diagnosis and treatment.
2. Microbiome and Ovarian Cancer
2.1. Gut Microbiome and Ovarian Cancer
2.1.1. Inflammation
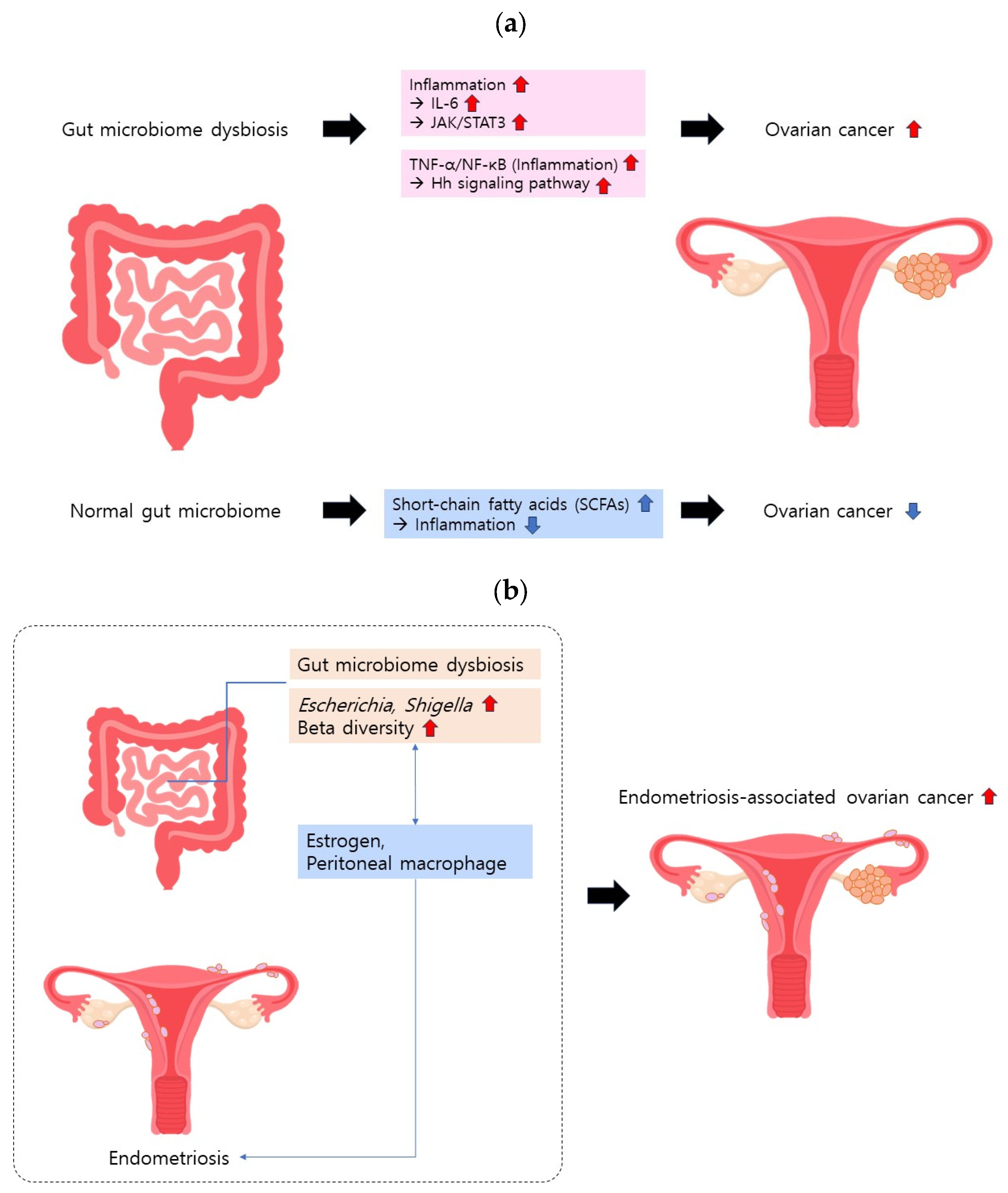
Continued research has underscored the intricate relationship between the gut microbiome and inflammation [10,11,12,13]. One study proposed that the gut microbiome dampens inflammatory responses by influencing immune regulatory cells [11]. Conversely, another study indicated that the gut microbiome may modulate intestinal permeability, thereby intensifying inflammatory responses [12]. Increased intestinal permeability allows microbial products to enter the bloodstream, thereby triggering elevated levels of cytokines and other inflammatory mediators. Other studies have highlighted the anti-inflammatory and immunomodulatory properties of short-chain fatty acids (SCFAs), which are metabolites produced by specific gut microbes [14]. Additionally, studies have suggested that gut microbiota-derived bile acids are involved in inflammation [15]. Remarkably, inflammatory cytokines have been implicated in ovarian cancer [16,17,18] (Figure 1a). A cytokine of particular interest in the gut microbiome is interleukin-6 (IL-6), found to be elevated in the ovarian cancer microenvironment [16]. IL-6 activates the Janus tyrosine kinase/signal transducer and activator of the transcription 3 (JAK/STAT3) pathway, and STAT3 modifies the transcription of several genes promoting cancer [16]. Therefore, IL-6 may promote the development of high-grade ovarian cancer. Notably, Toll-like receptor 5 (TLR5) and the microbiome play crucial roles in IL-6 activation. Rutkowski et al. induced cancer in TLR5-deficient and TLR5-responsive mice via p53 and K-ras gene mutations [19]. Despite similar cancer sizes, TLR5-responsive mice exhibited significantly higher serum IL-6 levels, and ovarian cancer progressed more rapidly than in the TLR5-dificient mice. Differences in microbiome composition, particularly in the Allobaculum, Bacteroides, and Lactobacillus genera, persisted even when the two groups shared the same cage, suggesting that TLR5 influences microbiome composition. Antibiotic treatment eliminating symbiotic bacteria erased the difference in serum IL-6 levels and dissimilarity in tumor growth, affirming the impact of the microbiome on ovarian cancer. Some studies have proposed that inflammatory responses linked to the gut microbiome may influence ovarian cancer carcinogenesis and progression through Hedgehog (Hh) signaling. The Hh signaling pathway has been recognized for its role in epithelial ovarian cancer (EOC) carcinogenesis and progression [20]. Tumor necrosis factor-α (TNF-α), a pro-inflammatory cytokine, has been identified as an activator of Hh signaling through nuclear factor kappa B (NF-κB) activation [21]. Hu et al. demonstrated that the gut microbiome from patients with EOC significantly increased serum TNF-α expression, up-regulated NF-κB, and activated Hh signaling, markedly enhancing EOC development [22]. These findings suggest that the gut microbiome from patients with EOC activates NF-kB, subsequently activating the Hh signaling pathway, potentially contributing to EOC development.
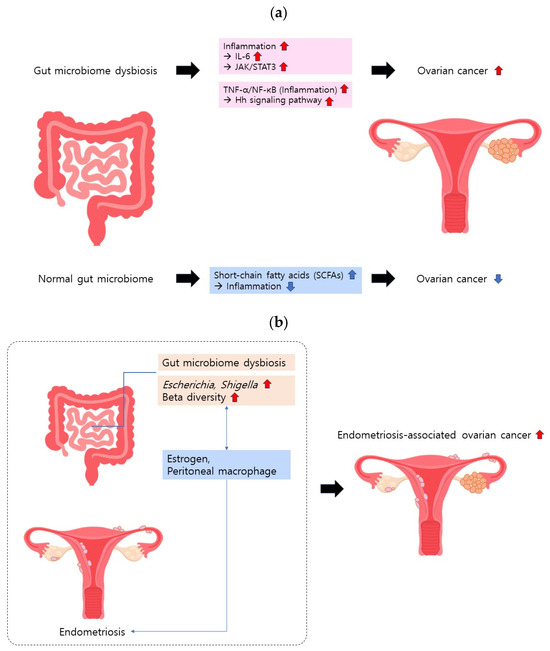
Figure 1.
Gut microbiome and ovarian cancer. (a) Gut microbiome dysbiosis intensifies the inflammatory response, elevating the risk of ovarian cancer. (b) Gut microbiome dysbiosis exacerbates endometriosis, contributing to the development of ovarian cancer. Estrogen and peritoneal macrophages may be involved in endometriosis-associated ovarian cancer development.
2.1.2. Endometriosis
Numerous studies have reported an elevated risk of specific ovarian cancers in women with endometriosis compared to the general population [23,24]. These cancers are classified as endometriosis-associated ovarian carcinomas (EAOCs) and include clear-cell carcinomas, endometrioid ovarian carcinomas, and seromucinous borderline tumors [25]. A clear-cell carcinoma is associated with PIK3CA and ARID1A mutations, whereas endometrioid ovarian cancer is associated with mutations in CTNNB1, PTEN, and ARID1A mutations [24]. Mutations in PTEN [26,27] and ARID1A [21,28] have been identified in endometriosis. Furthermore, hepatocyte nuclear factor-1β (HNF-1β) upregulation has been observed in both ovarian clear-cell carcinoma and endometriosis [24,29,30], implying a connection between endometriosis and specific ovarian cancers. However, the exact association between endometriosis and EAOC remains unclear.
Endometriosis may be linked to dysbiosis of the gut microbiome (Figure 1b). In contrast to women without endometriosis, the gut microbiome of patients with endometriosis was dominated by Escherichia and Shigella [31]. A systematic review revealed that individuals with endometriosis had a higher abundance of Actinobacteria, Firmicutes, Proteobacteria, and Verrucomicrobia in their gut microbiomes than healthy individuals [32]. Conversely, the abundance of Lactobacilli was lower in individuals with endometriosis. A study in rats demonstrated that 42 days after inducing endometriosis, while the alpha diversity of the gut microbiome remained similar, the beta diversity increased [33]. The treatment of these rats with antibiotics reduced endometriotic lesions, and the oral administration of feces reversed this condition.
The precise mechanism linking endometriosis and the gut microbiome remains elusive and presents a complex and multifaceted interplay. Estrogen, which is recognized for its association with the development of endometriosis, is a key player in this interaction [34]. Several studies have proposed a bidirectional relationship between the gut microbiome and estrogen levels in estrogen-related diseases, suggesting the plausible involvement of the gut microbiome in the development or manifestation of endometriosis symptoms [35]. Peritoneal macrophages represent another avenue of exploration for understanding the intricate relationship between endometriosis and the gut microbiome. A previous study revealed that inflammation exacerbated by an abnormal gut microbiome has discernible effects on peritoneal macrophages [36]. Specifically, mice with an altered microbiome exhibit heightened intestinal permeability and leakage of bacterial products, contributing to macrophage dysregulation and an increased likelihood of persistent endometriosis. Conversely, Miller et al. proposed an alternative perspective, suggesting that peritoneal macrophages may exacerbate inflammation [37]. In their study, mice with endometriosis were treated with IL-17, a cytokine known to be elevated during endometriosis, resulting in an increase in the number of M2 peritoneal macrophages. Miller et al. further noted that M2 macrophages can potentially worsen endometriosis.
Collectively, these studies underscore the intricate interplay among inflammation, peritoneal macrophages, and the gut microbiome, which potentially elevates the risk of ovarian cancer by exacerbating endometriosis. However, the precise mechanisms governing this interaction remain unclear. Further research is imperative to unravel the exact mechanisms underlying the interaction between endometriosis and gut microbiome dysbiosis to reveal potential therapeutic targets and interventions.
2.2. Cervicovaginal Microbiome and Ovarian Cancer
2.2.1. Chlamydia
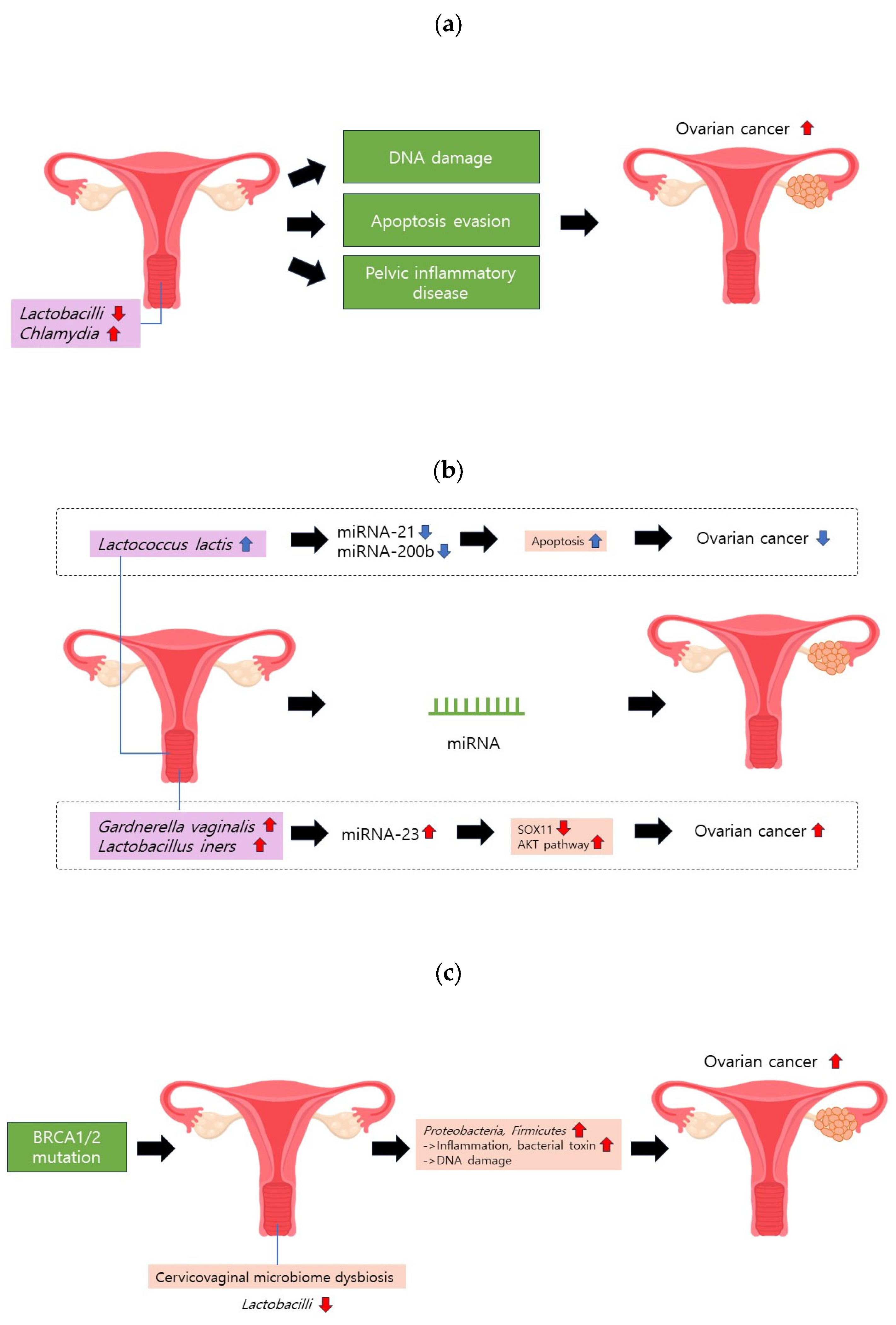
Chlamydia resulting from a Chlamydia trachomatis infection, a prevalent sexually transmitted disease, has been linked to an increased risk of ovarian cancer (Figure 2a). Various studies have identified a high incidence of Chlamydia in ovarian cancer cells [38,39]. Seropositivity for the chlamydial plasmid-encoded Pgp3 antibody is associated with a two-fold higher risk of ovarian cancer [40]. Recent meta-analyses have further substantiated the correlation between chlamydial infections and ovarian cancer risk [41].
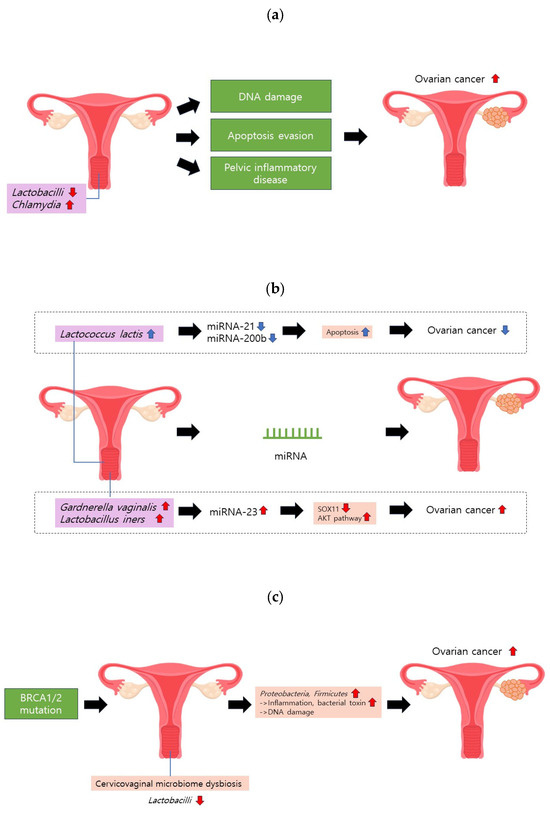
Figure 2.
Cervicovaginal microbiome and ovarian cancer. (a) A reduction in Lactobacilli or an increase in Chlamydia may induce DNA damage, hinder apoptosis, and contribute to pelvic inflammatory disease (PID). These factors collectively elevate the risk of ovarian cancer. (b) Gardnerella vaginalis and Lactobacillus iners can upregulate miRNA-223. This molecular alteration may heighten ovarian cancer risk by diminishing SOX11 expression and activating the AKT pathway. (c) BRCA1/2 mutations can instigate cervicovaginal microbiome dysbiosis, characterized by a decrease in Lactobacilli proportion.
Two primary mechanisms have been proposed to explain how Chlamydia increases the risk of ovarian cancer. First, it induces DNA damage through the production of reactive oxygen species, concurrently impeding the base-excision repair pathway [42]. Second, Chlamydia evades apoptosis by inhibiting the release of mitochondrial caspase 3 and cytochrome C while downregulating p53 [39,42].
Studies have indicated that Chlamydia can ascend to the upper female genital tract, inducing inflammation and damage, potentially leading to pelvic inflammatory disease (PID) and an increased risk of ovarian cancer [43,44,45,46]. Certain Lactobacillus strains, including Lactobacillus crispatus, have demonstrated significant bactericidal effects against Chlamydia trachomatis [47]. In particular, Lactobacillus crispatus exhibits 90% bactericidal activity, mainly attributed to lactic acid production [47,48,49]. This suggests a potential risk reduction for ovarian cancer through the modulation of Chlamydia by Lactobacillus.
2.2.2. Pelvic Inflammatory Disease
Chronic infections arising from PID contribute to the release of tumor-promoting substances such as cytokines, chemokines, and reactive oxygen species, fostering genetic and epigenetic alterations associated with cancer development [46]. However, there is an ongoing debate regarding the correlation between PID and ovarian cancer risk, with some studies reporting no significant association [50,51,52].
Ethnic disparities may contribute to conflicting findings. Zhou et al. noted that PID increased ovarian cancer risk in Asian women, but not in Caucasian women, possibly due to variations in lifestyle and oral contraceptive use [53]. Further complicating matters include inconsistencies within the same ethnic group, as demonstrated by the varying results of Taiwanese studies [52,54]. More comprehensive research controlling for numerous factors is warranted to ascertain the true relationship between PID and ovarian cancer risk.
2.2.3. MicroRNAs
MicroRNAs (miRNAs) are short endogenous noncoding RNAs (18–25 nt) that have recently gained attention for their association with various cancers, including ovarian cancer [55,56,57,58]. The cervicovaginal microbiome may influence miRNA expression, potentially contributing to the OC development of ovarian cancer (Figure 2b).
In a study by Anton et al., Gardnerella vaginalis, which is implicated in bacterial vaginosis, upregulated miRNA-15a, miRNA-143, miRNA-145, miRNA-146, miRNA-223, and miRNA-148 [59]. Furthermore, Lactobacillus iners, which is linked to bacterial vaginosis and adverse pregnancy outcomes, upregulates miRNA-146, miRNA-193b, and miRNA-223 [59,60]. Notably, miRNA-223 is overexpressed in ovarian cancer cells and the serum exosomes of patients with EOC [61,62]. miRNA-223-3p overexpression is associated with reduced levels of the sex-determining region Y-box 11 (SOX11) [61]. They also found that SOX11 overexpression inhibits the growth, migration, and invasion of ovarian cancer cells. Another study has suggested that miRNA-223 overexpression promotes ovarian cancer development by activating the AKT pathway [62]. Conversely, Pan et al. reported the downregulation of miRNA-223 in the exosomes of patients with EOC [63]. Given these conflicting findings, further investigations are needed to clarify the role of miRNA-223 in EOC pathogenesis.
Saadat et al. demonstrated that Lactococcus lactis, a probiotic from the vagina, reduced the expression of miRNA-21 and miRNA-200b and enhanced apoptosis in ovarian cancer cells [64]. The suppression of miRNA-21 has been linked to reduced cancer cell proliferation and tumor growth, whereas the miRNA-200 family plays a role in the initiation and progression of ovarian cancer.
Although promising, the understanding of miRNA–microbiome interactions in ovarian cancer remains limited, necessitating further research to unravel specific miRNA alterations and their implications.
2.2.4. BRCA Mutation
Mutations in BRCA1/2 substantially increase the risk of ovarian cancer, with a risk range of 39–63% in BRCA1 mutation carriers and 16.5–27% in BRCA2 mutation carriers [65,66,67]. Intriguingly, one study proposed the potential interplay between BRCA1/2 mutations and the cervicovaginal microbiome, which influences the risk of ovarian cancer [68] (Figure 2c). In particular, women under 50 years of age with BRCA1 mutations exhibit a reduced proportion of Lactobacilli in their cervicovaginal microbiomes compared to those without mutations. This reduction in Lactobacilli was consistent across disease stages, suggesting a potential causal relationship. Nené et al. introduced mechanistic insights into this decrease in Lactobacilli, linking it to an increase in the progesterone concentration during the luteal phase, which reduced the vaginal glycogen concentration [68]. Given that Lactobacilli thrive in the glycogen metabolism, this unfavorable condition may have contributed to their decrease [68]. Moreover, the evidence suggests that a reduction in Lactobacilli may play a causal role in the development of ovarian cancer. Proteobacteria and Firmicutes, which were predominant in cancer samples, can induce inflammation, release bacterial toxins that damage DNA, and contribute to the development of cancer.
In contrast, progesterone, known for its inhibitory effects on ovarian cancer [69,70,71], raises questions regarding the significance of its role in reducing Lactobacilli and impacting ovarian cancer risk in women with BRCA mutations. Increased progesterone levels in these women may contribute to a decrease in Lactobacilli; however, their impact on ovarian cancer risk remains uncertain. As Nené et al. acknowledged, it is plausible that factors beyond increased progesterone levels may influence the reduction in Lactobacilli and the subsequent increase in ovarian cancer risk in women with BRCA mutations, necessitating further research for a comprehensive understanding of these intricate interactions.
3. Microbiome and the Diagnosis and Treatment of Ovarian Cancer
3.1. Ovarian Cancer Diagnosis and Microbiome Markers
Ovarian cancer has a commendable five-year survival rate, exceeding 90% when diagnosed at stage I [72]. The urgency for an early diagnosis is underscored by the advanced stage at which ovarian cancer is detected. However, the widely employed ovarian cancer biomarker cancer antigen 125 lacks specificity owing to its elevated levels in conditions such as endometriosis and other cancers [73,74]. Hence, it is imperative to establish novel and specific biomarkers for ovarian cancer diagnosis.
Given its potential implications, the microbiome has emerged as a candidate contributor to ovarian cancer development. Although the causative or consequential nature of this association remains unclear, studies have consistently reported altered microbiome compositions in various body sites in patients with ovarian cancer compared to healthy individuals, suggesting a potential role of the microbiome as a diagnostic biomarker (Table 1). One study demonstrated elevated Bacteroides, Prevotella, and Proteobacteria as well as reduced levels of Ruminococcus and Actinobacteria in patients with ovarian cancer [75]. Of note, Prevotella, linked to a proinflammatory state, is associated with cervical and endometrial cancers [76,77]. However, the prospects of using the gut microbiome as a biomarker require further investigation. Miao et al. investigated the peritoneal microbiome and revealed reduced microbial diversity and a distinctive microbial signature in patients with ovarian cancer compared to those with benign adnexal masses [78]. Of these, 18 clusters were highly specific to ovarian cancer pathology.
Another study identified the presence of Brucella, Chlamydia, and Mycoplasma in ovarian cancer tumor cells, with Brucella believed to drive pelvic inflammation, leading to ovarian cancer [38,79]. Chlamydia significantly increases the risk of ovarian cancer, although further research is needed to elucidate Mycoplasma’s role [41]. Additionally, investigations into the dominant taxa in ovarian cancer cells revealed an increased Proteobacteria/Firmicutes ratio and significant changes in specific bacteria such as Acinetobacter and Lactococcus [80]. Notably, the decrease in Lactococcus, a probiotic candidate, and alterations in antibacterial response genes suggest potential microbiota-driven biomarkers for ovarian cancer.
Studies have consistently reported decreased levels of Lactobacilli in the cervicovaginal microbiome of patients with ovarian cancer [68,72,75]. Lactobacilli alone may have limited diagnostic value because they are also reduced in certain cervical cancer types; however, when combined with other biomarkers, they can increase their diagnostic accuracy [81]. Noteworthy is a study by Asangba et al., revealing distinct patterns of bacteria associated with different stages of ovarian cancer, offering potential insights into early diagnosis and prognosis [82].
In serum microbiome studies, Acinetobacter’s increased abundance in patients with ovarian cancer, as seen in the studies by Zhou et al. [80] and Kim et al. [83], aligns with its potential diagnostic significance. Serum samples, which are less invasive and more easily obtainable, represent a promising avenue for microbiome-based biomarker studies.
Exploiting the microbiome as a diagnostic tool holds promise, given its noninvasive nature and compatibility with current liquid-based cytology tests [72]. Ongoing research and clinical applications aim to enhance ovarian cancer prognosis using microbiome-based diagnostics.

Table 1.
Overview of microbiome alterations in gut, peritoneum, cervicovaginal, ovarian cancer tissue, and serum samples from patients with ovarian cancer. Plus sign (+) and minus sign (−) indicate more and less abundant, respectively, in ovarian cancer patients or ovarian cancer cells compared to control.
Table 1.
Overview of microbiome alterations in gut, peritoneum, cervicovaginal, ovarian cancer tissue, and serum samples from patients with ovarian cancer. Plus sign (+) and minus sign (−) indicate more and less abundant, respectively, in ovarian cancer patients or ovarian cancer cells compared to control.
| Location | Microbiome | Relative Abundance |
|---|---|---|
| Gut | Bacteroides [75] | + |
| Prevotella [75] | + | |
| Proteobacteria [75] | + | |
| Ruminococcus [75] | − | |
| Actinobacteria [75] | − | |
| Peritoneum | 18 microbial features [78] | Unique distribution |
| Ovarian Cancer Tissue | Brucella [38] | 76% of patients |
| Chlamydia [38] | 60% of patients | |
| Mycoplasma [38] | 74% of patients | |
| Proteobacteria/firmicutes [80] | + | |
| Acinetobacter [80] | + | |
| Lactococcus [80] | − | |
| Cervicovagina | Lactobacilli [68] | − |
| Mobiluncus curtisii [82] | + → − | |
| Eubacterium rectale [82] | + → − | |
| Fusobacterium nucleatum [82] | + → − | |
| Porphyromonas [82] | + → − | |
| Serum | Acinetobacter [83] | + |
3.2. Ovarian Cancer Treatment and Microbiome Effects
The treatment of ovarian cancer includes surgery and chemotherapy. These treatments were linked to the microbiome (Table 2). A previous study showed that the composition of the gut microbiome was altered after ovarian cancer surgery [84]. The relative proportion of Proteobacteria increased and the relative proportions of Bacteroidetes and Firmicutes decreased after surgery. These microorganisms are also associated with enteritis and colitis [85,86]. After surgery, the number of bacteria producing SCFAs, such as Bacteroidetes, Faecalibacterium, Blautia, Roseburia, and Prevotella, decreased. SCFAs have anti-inflammatory, anti-cancer, and immune effects [14,87]. Therefore, surgery can alter the microbiome composition, which can have various effects on the body.
Platinum-based anti-cancer drugs are commonly used to treat several cancers, including ovarian cancer. However, some studies have shown that these drugs affect the gut microbiome. One study found that certain bacteria, including Bacteroides, Collinsella, and Blautia, increased in number after several cycles of chemotherapy [84]. Bacteroides and Collinsella are associated with rectal cancer [88,89]. Bifidobacterium increased after one–three cycles of chemotherapy [84]. This bacterium plays a crucial role in maintaining the gut microbial balance and has been linked to anti-cancer effects [90].
Cyclophosphamide, in addition to platinum-based chemotherapy, is used to treat severe ovarian cancers. One study reported that it also affected the gut microbiome [91]. In this study, we found that mice treated with cyclophosphamide experienced a breakdown of the small intestinal epithelial barrier and a decrease in the number of Lactobacilli and Enterococci in the small intestine. This indicates that cyclophosphamide may facilitate the movement of bacteria across the intestinal epithelium and surrounding environment. The study found that bacteria present in the small intestine were also present in the mesenteric lymph nodes and spleen. This leads to the activation of the immune response, which increases helper and memory T cells, ultimately promoting anti-cancer effects. These results suggest that cyclophosphamide enhances anti-cancer effects by shifting the gut microbiome to lymphoid organs.
Studies have shown that the vaginal microbiome can affect chemotherapy in ovarian cancer [92]. Gemcitabine, a chemotherapeutic drug used to treat ovarian cancer, was less effective when tumor cells were cocultured with Mycoplasma [92]. This was attributed to the rapid degradation of gemcitabine by pyrimidine nucleoside phosphorylase and cytidine deaminase in Mycoplasma. Another study found that patients with platinum-resistant tumors were more likely to have a vaginal microbiome dominated by Escherichia coli [93].
Furthermore, the combination of surgery and chemotherapy affects the vaginal microbiome. A study on patients with ovarian cancer found that the presence of Lactobacilli decreased when both treatments were applied [75]. The authors attributed this to the dynamics among ovarian cancer, estrogen, and glycogen. Specifically, chemotherapy and an oophorectomy decrease estrogen production which, in turn, reduces glycogen in the vagina, leading to a decrease in Lactobacilli. As discussed in Section 2.2.1 and Section 2.2.3, a reduction in Lactobacilli may promote ovarian cancer progression.
Recent studies have revealed a link between the microbiome and ovarian cancer, leading to attempts to prevent or improve treatment efficiency by transplanting healthy microbes. There are two main types of microbiome transplantation: fecal microbiome transplantation (FMT) and vaginal microbiome transplantation (VMT).
Previous studies have explored the use of FMT for the treatment of other cancers [94,95,96,97]. However, to the best of our knowledge, only one study of FMT as a treatment has been directly relevant to ovarian cancer. Chambers et al. conducted a study which demonstrated that mice treated with antibiotics experienced accelerated ovarian cancer growth and increased cisplatin resistance [98]. However, when these mice were cecally transplanted with a microbiome derived from healthy mice, chemotherapy resistance was mitigated and their lifespan was prolonged. Although there are few studies, there is a glimpse into the potential for FMT in ovarian cancer, and further research could be beneficial for treatment.
No studies have directly addressed the link between VMT and ovarian cancer. Studies have shown that VMT reduces recurrence rates and improves symptoms in patients with bacterial vaginosis [99,100]. Therefore, VMT may be a plausible approach. Based on these findings, we propose investigating the use of VMT to modulate the cervicovaginal microbiome to increase drug responsiveness. This will improve the efficiency of ovarian cancer treatment.

Table 2.
Overview of the relationship between the microbiome and the treatment of ovarian cancer.
Table 2.
Overview of the relationship between the microbiome and the treatment of ovarian cancer.
| Treatment Type | Microbiome | Contents |
|---|---|---|
| Surgical Therapy | Proteobacteria Enterobacteriaceaea | Increase in relative proportion after ovarian cancer surgery [84,101] |
| Bacteroidetes Firmicutes Faecalibacterium Blautia Roseburia Prevotella | Decrease in relative proportion after ovarian cancer surgery [84] | |
| Chemotherapy | Bacteroides Collinsella Blautia | Increase in relative proportion after platinum-based chemotherapy [84] |
| Lactobacilli Enterococci | Relative proportion in small intestine decreased, and relative proportion in mesenteric lymph nodes and spleen increased after cyclophosphamide administration [91] | |
| Bifidobacterium | Increase in relative proportion after one to three cycles of chemotherapy [84] | |
| Mycoplasma | Mycoplasma’s enzymes rapidly break down gemcitabine, reducing its responsiveness to the drug [92] | |
| Chemotherapy with surgery | Lactobacilli | Reduction in relative proportion after oophorectomy and chemotherapy [75] |
4. Discussion and Conclusions
The pivotal role of the microbiome in maintaining human health is evident and dysbiosis has been associated with various diseases, including ovarian cancer. This review highlights the potential repercussions of changes in the gut microbiome, particularly intensifying the inflammatory responses and increasing the risk of endometriosis, thereby increasing the likelihood of ovarian cancer. The IL-6 and Hh signaling pathways are likely involved in the mechanism by which the gut microbiome increases ovarian cancer risk by intensifying the inflammatory response. However, a comprehensive understanding of how endometriosis contributes to ovarian cancer risk requires further research, particularly to elucidate the potential interaction between endometriosis and the gut microbiome, as evidence involving factors such as peritoneal macrophages and estrogen remains limited.
The association between Chlamydia and ovarian cancer risk is intricate, with potential mechanisms including DNA damage or the evasion of apoptosis. However, the possibility of a Chlamydia infection leading to PID and subsequently increasing the risk of ovarian cancer varies across ethnic groups and within the same group. Lifestyle, genetic factors, and research methodologies have been proposed as contributors to these divergent findings, emphasizing the need for further investigation to uncover the factors that influence how PID may lead to ovarian cancer. The relationship between Gardnerella vaginalis, Lactobacillus iners, and ovarian cancer risk, as well as the conflicting results regarding miRNA-223 expression in patients with EOC, necessitates additional exploration. Although studies on miRNAs in ovarian cancer have been conducted, the link between the microbiome and ovarian cancer remains underexplored.
The potential impact of BRCA1/2 mutations on ovarian cancer risk by reducing Lactobacilli requires further research, particularly to reconcile the contradictory findings on the effects of progesterone. Establishing biomarkers for early ovarian cancer diagnosis is imperative for high survival rates, and the microbiome in the gut, peritoneum, ovarian cancer tissue, cervicovaginal area, and serum holds promise as diagnostic markers. However, research on the gut microbiome as a biomarker lags behind that on the cervicovaginal microbiome. Although Mycoplasma has been identified in ovarian cancer tumor cells, its impact on ovarian cancer risk remains inconclusive and requires further investigation. Lactobacilli, which exhibit a consistent decrease in the cervicovaginal microbiome of patients with ovarian, cervical, and precancerous diseases, may be a valuable biomarker, but should be used in combination with other biomarkers to enhance diagnostic accuracy.
In exploring the potential impact of the microbiome on ovarian cancer treatments, this review suggests a plausible influence of both surgical and chemotherapy treatments on the efficacy and adverse effects. Although the microbiome may play a role in the response to chemotherapy, the direct relationship between the microbiome and chemotherapy in ovarian cancer requires further investigation, especially considering the drugs commonly used to treat various cancers. The concept of preventing ovarian cancer through microbiome transplantation from healthy individuals into those at high risk, or improving treatment efficiency through microbiome transplantation in patients with ovarian cancer, underscores the potential connection between ovarian cancer and the microbiome. The limited direct applications of FMT and VMT in ovarian cancer emphasize the need for further exploration, despite encouraging outcomes in related studies. These findings suggest that investigating microbiome transplantation in ovarian cancer is promising and merits further research.
Funding
This work has been supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (NRF-2017R1A5A2014768 and 2022R1A2C1003498).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Talaro, K.P.B. Chess. In Foundations in Microbiology, 10th ed.; McGraw-Hill Education: Columbus, OH, USA, 2017; pp. 402–404. [Google Scholar]
- Takiishi, T.; Fenero, C.I.M.; Câmara, N.O.S. Intestinal barrier and gut microbiota: Shaping our immune responses throughout life. Tissue Barriers 2017, 5, e1373208. [Google Scholar] [CrossRef] [PubMed]
- Wagner-Skacel, J.; Dalkner, N.; Moerkl, S.; Kreuzer, K.; Farzi, A.; Lackner, S.; Painold, A.; Reininghaus, E.Z.; Butler, M.I.; Bengesser, S. Sleep and Microbiome in Psychiatric Diseases. Nutrients 2020, 12, 2198. [Google Scholar] [CrossRef] [PubMed]
- Bana, B.; Cabreiro, F. The Microbiome and Aging. Annu. Rev. Genet. 2019, 53, 239–261. [Google Scholar] [CrossRef] [PubMed]
- Rajagopala, S.V.; Vashee, S.; Oldfield, L.M.; Suzuki, Y.; Venter, J.C.; Telenti, A.; Nelson, K.E. The Human Microbiome and Cancer. Cancer Prev. Res. 2017, 10, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.X.; Schwabe, R.F. The gut microbiome and liver cancer: Mechanisms and clinical translation. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 527–539. [Google Scholar] [CrossRef]
- Chen, J.; Douglass, J.; Prasath, V.; Neace, M.; Atrchian, S.; Manjili, M.H.; Shokouhi, S.; Habibi, M. The microbiome and breast cancer: A review. Breast Cancer Res. Treat. 2019, 178, 493–496. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.; Ralyea, C.; Lockwood, S. Ovarian Cancer: An Integrated Review. Semin. Oncol. Nurs. 2019, 35, 151–156. [Google Scholar] [CrossRef]
- Clemente, J.C.; Manasson, J.; Scher, J.U. The role of the gut microbiome in systemic inflammatory disease. BMJ 2018, 360, j5145. [Google Scholar] [CrossRef]
- Shi, N.; Li, N.; Duan, X.; Niu, H. Interaction between the gut microbiome and mucosal immune system. Mil. Med. Res. 2017, 4, 14. [Google Scholar] [CrossRef]
- Brandsma, E.; Kloosterhuis, N.J.; Koster, M.; Dekker, D.C.; Gijbels, M.J.; Van Der Velden, S.; Ríos-Morales, M.; Van Faassen, M.J.; Loreti, M.G.; De Bruin, A. A proinflammatory gut microbiota increases systemic inflammation and accelerates atherosclerosis. Circ. Res. 2019, 124, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Buford, T.W. (Dis)Trust your gut: The gut microbiome in age-related inflammation, health, and disease. Microbiome 2017, 5, 80. [Google Scholar] [CrossRef]
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The role of short-chain fatty acids in health and disease. Adv. Immunol. 2014, 121, 91–119. [Google Scholar]
- Cai, J.; Sun, L.; Gonzalez, F.J. Gut microbiota-derived bile acids in intestinal immunity, inflammation, and tumorigenesis. Cell Host Microbe 2022, 30, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Browning, L.; Patel, M.R.; Horvath, E.B.; Tawara, K.; Jorcyk, C.L. IL-6 and ovarian cancer: Inflammatory cytokines in promotion of metastasis. Cancer Manag. Res. 2018, 10, 6685–6693. [Google Scholar] [CrossRef]
- Bouras, E.; Karhunen, V.; Gill, D.; Huang, J.; Haycock, P.C.; Gunter, M.J.; Johansson, M.; Brennan, P.; Key, T.; Lewis, S.J. Circulating inflammatory cytokines and risk of five cancers: A Mendelian randomization analysis. BMC Med. 2022, 20, 3. [Google Scholar] [CrossRef]
- Riera-Domingo, C.; Audigé, A.; Granja, S.; Cheng, W.-C.; Ho, P.-C.; Baltazar, F.; Stockmann, C.; Mazzone, M. Immunity, hypoxia, and metabolism–the Ménage à Trois of cancer: Implications for immunotherapy. Physiol. Rev. 2020, 100, 1–102. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, M.R.; Stephen, T.L.; Svoronos, N.; Allegrezza, M.J.; Tesone, A.J.; Perales-Puchalt, A.; Brencicova, E.; Escovar-Fadul, X.; Nguyen, J.M.; Cadungog, M.G. Microbially driven TLR5-dependent signaling governs distal malignant progression through tumor-promoting inflammation. Cancer Cell 2015, 27, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, Y.; Chen, T.; Zhang, Y.; Xu, R.; Wang, W.; Cheng, M.; Chen, Q. Aberrant activation of hedgehog signalling promotes cell migration and invasion via matrix metalloproteinase-7 in ovarian cancer cells. J. Cancer 2019, 10, 990. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Zhang, M.; Zhou, G.; Lin, L.; Han, J.; Wang, Y.; Li, L.; He, Y.; Zeng, Z.; Chen, M. Emerging roles of the Hedgehog signalling pathway in inflammatory bowel disease. Cell Death Discov. 2021, 7, 314. [Google Scholar] [CrossRef]
- Hu, X.; Xu, X.; Zeng, X.; Jin, R.; Wang, S.; Jiang, H.; Tang, Y.; Chen, G.; Wei, J.; Chen, T. Gut microbiota dysbiosis promotes the development of epithelial ovarian cancer via regulating Hedgehog signaling pathway. Gut Microbes 2023, 15, 2221093. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, T.; Chung, H.; Song, Y. Risk and prognosis of ovarian cancer in women with endometriosis: A meta-analysis. Br. J. Cancer 2014, 110, 1878–1890. [Google Scholar] [CrossRef]
- Gadducci, A.; Lanfredini, N.; Tana, R. Novel insights on the malignant transformation of endometriosis into ovarian carcinoma. Gynecol. Endocrinol. 2014, 30, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Králíčková, M.; Laganà, A.S.; Ghezzi, F.; Vetvicka, V. Endometriosis and risk of ovarian cancer: What do we know? Arch. Gynecol. Obstet. 2020, 301, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Krawczyk, N.; Banys-Paluchowski, M.; Schmidt, D.; Ulrich, U.; Fehm, T. Endometriosis-associated malignancy. Geburtshilfe Frauenheilkd. 2016, 76, 176–181. [Google Scholar] [CrossRef] [PubMed]
- McConechy, M.K.; Ding, J.; Senz, J.; Yang, W.; Melnyk, N.; Tone, A.A.; Prentice, L.M.; Wiegand, K.C.; McAlpine, J.N.; Shah, S.P. Ovarian and endometrial endometrioid carcinomas have distinct CTNNB1 and PTEN mutation profiles. Mod. Pathol. 2014, 27, 128–134. [Google Scholar] [CrossRef]
- Chene, G.; Ouellet, V.; Rahimi, K.; Barres, V.; Provencher, D.; Mes-Masson, A.M. The ARID1A pathway in ovarian clear cell and endometrioid carcinoma, contiguous endometriosis, and benign endometriosis. Int. J. Gynecol. Obstet. 2015, 130, 27–30. [Google Scholar] [CrossRef]
- Ito, F.; Yoshimoto, C.; Yamada, Y.; Sudo, T.; Kobayashi, H. The HNF-1β―USP28―Claspin pathway upregulates DNA damage-induced Chk1 activation in ovarian clear cell carcinoma. Oncotarget 2018, 9, 17512. [Google Scholar] [CrossRef]
- Preya, U.H.; Woo, J.-H.; Choi, Y.S.; Choi, J.-H. Hepatocyte nuclear factor-1 beta protects endometriotic cells against apoptotic cell death by up-regulating the expression of antiapoptotic genes. Biol. Reprod. 2019, 101, 686–694. [Google Scholar] [CrossRef]
- Ata, B.; Yildiz, S.; Turkgeldi, E.; Brocal, V.P.; Dinleyici, E.C.; Moya, A.; Urman, B. The endobiota study: Comparison of vaginal, cervical and gut microbiota between women with stage 3/4 endometriosis and healthy controls. Sci. Rep. 2019, 9, 2204. [Google Scholar] [CrossRef]
- Leonardi, M.; Hicks, C.; El-Assaad, F.; El-Omar, E.; Condous, G. Endometriosis and the microbiome: A systematic review. BJOG Int. J. Obstet. Gynaecol. 2020, 127, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Li, D.; Zhang, Z.; Sun, H.; An, M.; Wang, G. Endometriosis induces gut microbiota alterations in mice. Hum. Reprod. 2018, 33, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Clemenza, S.; Vannuccini, S.; Ruotolo, A.; Capezzuoli, T.; Petraglia, F. Advances in targeting estrogen synthesis and receptors in patients with endometriosis. Expert Opin. Investig. Drugs 2022, 31, 1227–1238. [Google Scholar] [CrossRef] [PubMed]
- Flores, R.; Shi, J.; Fuhrman, B.; Xu, X.; Veenstra, T.D.; Gail, M.H.; Gajer, P.; Ravel, J.; Goedert, J.J. Fecal microbial determinants of fecal and systemic estrogens and estrogen metabolites: A cross-sectional study. J. Transl. Med. 2012, 10, 253. [Google Scholar] [CrossRef] [PubMed]
- Thevaranjan, N.; Puchta, A.; Schulz, C.; Naidoo, A.; Szamosi, J.C.; Verschoor, C.P.; Loukov, D.; Schenck, L.P.; Jury, J.; Foley, K.P. Age-associated microbial dysbiosis promotes intestinal permeability, systemic inflammation, and macrophage dysfunction. Cell Host Microbe 2017, 21, 455–466.e454. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.E.; Ahn, S.H.; Marks, R.M.; Monsanto, S.P.; Fazleabas, A.T.; Koti, M.; Tayade, C. IL-17A modulates peritoneal macrophage recruitment and M2 polarization in endometriosis. Front. Immunol. 2020, 11, 108. [Google Scholar] [CrossRef]
- Banerjee, S.; Tian, T.; Wei, Z.; Shih, N.; Feldman, M.D.; Alwine, J.C.; Coukos, G.; Robertson, E.S. The ovarian cancer oncobiome. Oncotarget 2017, 8, 36225. [Google Scholar] [CrossRef]
- Shanmughapriya, S.; Senthilkumar, G.; Vinodhini, K.; Das, B.; Vasanthi, N.; Natarajaseenivasan, K. Viral and bacterial aetiologies of epithelial ovarian cancer. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2311–2317. [Google Scholar] [CrossRef]
- Trabert, B.; Waterboer, T.; Idahl, A.; Brenner, N.; Brinton, L.A.; Butt, J.; Coburn, S.B.; Hartge, P.; Hufnagel, K.; Inturrisi, F. Antibodies against Chlamydia trachomatis and ovarian cancer risk in two independent populations. JNCI J. Natl. Cancer Inst. 2019, 111, 129–136. [Google Scholar] [CrossRef]
- Hosseininasab-Nodoushan, S.-A.; Ghazvini, K.; Jamialahmadi, T.; Keikha, M.; Sahebkar, A. Association of Chlamydia and Mycoplasma infections with susceptibility to ovarian cancer: A systematic review and meta-analysis. Semin. Cancer Biol. 2022, 86, 923–928. [Google Scholar] [CrossRef]
- Gulve, N.; Rudel, T. Chlamydia trachomatis and human herpesvirus 6 infections in ovarian cancer—Casual or causal? PLoS Pathog. 2019, 15, e1008055. [Google Scholar] [CrossRef] [PubMed]
- Brunham, R.C.; Gottlieb, S.L.; Paavonen, J. Pelvic inflammatory disease. N. Engl. J. Med. 2015, 372, 2039–2048. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.; Prabhu, M. Pelvic inflammatory disease: Current concepts in pathogenesis, diagnosis and treatment. Infect. Dis. Clin. 2013, 27, 793–809. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, S.; Jonsson, H.; Lundin, E.; Häggström, C.; Idahl, A. Pelvic inflammatory disease and risk of epithelial ovarian cancer: A national population-based case-control study in Sweden. Am. J. Obstet. Gynecol. 2024, 230, 75.e1–75.e15. [Google Scholar] [CrossRef] [PubMed]
- Piao, J.; Lee, E.J.; Lee, M. Association between pelvic inflammatory disease and risk of ovarian cancer: An updated meta-analysis. Gynecol. Oncol. 2020, 157, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Min, S.; Wang, L.; Zhao, L.; Luo, F.; Lei, W.; Wen, Y.; Luo, L.; Zhou, Q.; Peng, L. Lactobacillus modulates chlamydia infectivity and genital tract pathology in vitro and in vivo. Front. Microbiol. 2022, 13, 877223. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Luna, Y.; Yu, P.; Fan, H. Lactobacilli inactivate Chlamydia trachomatis through lactic acid but not H2O2. PLoS ONE 2014, 9, e107758. [Google Scholar] [CrossRef]
- Mastromarino, P.; Di Pietro, M.; Schiavoni, G.; Nardis, C.; Gentile, M.; Sessa, R. Effects of vaginal lactobacilli in Chlamydia trachomatis infection. Int. J. Med. Microbiol. 2014, 304, 654–661. [Google Scholar] [CrossRef]
- McAlpine, J.N.; Lisonkova, S.; Joseph, K.; McComb, P.F. Pelvic inflammation and the pathogenesis of ovarian cancer: A cohort study. Int. J. Gynecol. Cancer 2014, 24, 1406–1413. [Google Scholar] [CrossRef]
- Rasmussen, C.B.; Jensen, A.; Albieri, V.; Andersen, K.K.; Kjaer, S.K. Is pelvic inflammatory disease a risk factor for ovarian cancer? Cancer Epidemiol. Biomark. Prev. 2017, 26, 104–109. [Google Scholar] [CrossRef]
- Shen, C.-C.; Hu, L.-Y.; Yang, A.C.; Chiang, Y.-Y.; Hung, J.-H.; Tsai, S.-J. Risk of uterine, ovarian and breast cancer following pelvic inflammatory disease: A nationwide population-based retrospective cohort study. BMC Cancer 2016, 16, 839. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zeng, F.; Yuan, J.; Tang, J.; Colditz, G.A.; Tworoger, S.S.; Trabert, B.; Su, X. Pelvic inflammatory disease and the risk of ovarian cancer: A meta-analysis. Cancer Causes Control 2017, 28, 415–428. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-W.; Tu, Y.-Y.; Lin, S.Y.; Su, W.-J.; Lin, W.L.; Lin, W.Z.; Wu, S.-C.; Lai, Y.-L. Risk of ovarian cancer in women with pelvic inflammatory disease: A population-based study. Lancet Oncol. 2011, 12, 900–904. [Google Scholar] [CrossRef] [PubMed]
- Macgregor-Das, A.M.; Das, S. A microRNA’s journey to the center of the mitochondria. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H206–H215. [Google Scholar] [CrossRef] [PubMed]
- Kabzinski, J.; Maczynska, M.; Majsterek, I. MicroRNA as a novel biomarker in the diagnosis of head and neck cancer. Biomolecules 2021, 11, 844. [Google Scholar] [CrossRef] [PubMed]
- Elgeshy, K.M.; Abdel Wahab, A.H.A. The Role, Significance, and Association of MicroRNA-10a/b in Physiology of Cancer. MicroRNA 2022, 11, 118–138. [Google Scholar] [CrossRef]
- Moghadasi, M.; Alivand, M.; Fardi, M.; Moghadam, K.S.; Solali, S. Emerging molecular functions of microRNA-124: Cancer pathology and therapeutic implications. Pathol. Res. Pract. 2020, 216, 152827. [Google Scholar] [CrossRef]
- Anton, L.; Sierra, L.-J.; DeVine, A.; Barila, G.; Heiser, L.; Brown, A.G.; Elovitz, M.A. Common cervicovaginal microbial supernatants alter cervical epithelial function: Mechanisms by which Lactobacillus crispatus contributes to cervical health. Front. Microbiol. 2018, 9, 395751. [Google Scholar] [CrossRef]
- Zheng, N.; Guo, R.; Wang, J.; Zhou, W.; Ling, Z. Contribution of Lactobacillus iners to vaginal health and diseases: A systematic review. Front. Cell. Infect. Microbiol. 2021, 11, 792787. [Google Scholar] [CrossRef]
- Fang, G.; Liu, J.; Wang, Q.; Huang, X.; Yang, R.; Pang, Y.; Yang, M. MicroRNA-223-3p regulates ovarian cancer cell proliferation and invasion by targeting SOX11 expression. Int. J. Mol. Sci. 2017, 18, 1208. [Google Scholar] [CrossRef]
- Zhu, X.; Shen, H.; Yin, X.; Yang, M.; Wei, H.; Chen, Q.; Feng, F.; Liu, Y.; Xu, W.; Li, Y. Macrophages derived exosomes deliver miR-223 to epithelial ovarian cancer cells to elicit a chemoresistant phenotype. J. Exp. Clin. Cancer Res. 2019, 38, 81. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.; Stevic, I.; Müller, V.; Ni, Q.; Oliveira-Ferrer, L.; Pantel, K.; Schwarzenbach, H. Exosomal micro RNA s as tumor markers in epithelial ovarian cancer. Mol. Oncol. 2018, 12, 1935–1948. [Google Scholar] [CrossRef] [PubMed]
- Rahbar Saadat, Y.; Pourseif, M.M.; Zununi Vahed, S.; Barzegari, A.; Omidi, Y.; Barar, J. Modulatory role of vaginal-isolated lactococcus lactis on the expression of miR-21, miR-200b, and TLR-4 in CAOV-4 cells and in silico revalidation. Probiotics Antimicrob. Proteins 2020, 12, 1083–1096. [Google Scholar] [CrossRef]
- Yoshida, R. Hereditary breast and ovarian cancer (HBOC): Review of its molecular characteristics, screening, treatment, and prognosis. Breast Cancer 2021, 28, 1167–1180. [Google Scholar] [CrossRef] [PubMed]
- Tschernichovsky, R.; Goodman, A. Risk-reducing strategies for ovarian cancer in BRCA mutation carriers: A balancing act. Oncologist 2017, 22, 450–459. [Google Scholar] [CrossRef][Green Version]
- Wentzensen, N.; Poole, E.M.; Trabert, B.; White, E.; Arslan, A.A.; Patel, A.V.; Setiawan, V.W.; Visvanathan, K.; Weiderpass, E.; Adami, H.-O. Ovarian cancer risk factors by histologic subtype: An analysis from the ovarian cancer cohort consortium. J. Clin. Oncol. 2016, 34, 2888. [Google Scholar] [CrossRef]
- Nené, N.R.; Reisel, D.; Leimbach, A.; Franchi, D.; Jones, A.; Evans, I.; Knapp, S.; Ryan, A.; Ghazali, S.; Timms, J.F. Association between the cervicovaginal microbiome, BRCA1 mutation status, and risk of ovarian cancer: A case-control study. Lancet Oncol. 2019, 20, 1171–1182. [Google Scholar] [CrossRef]
- McGlorthan, L.; Paucarmayta, A.; Casablanca, Y.; Maxwell, G.L.; Syed, V. Progesterone induces apoptosis by activation of caspase-8 and calcitriol via activation of caspase-9 pathways in ovarian and endometrial cancer cells in vitro. Apoptosis 2021, 26, 184–194. [Google Scholar] [CrossRef]
- Lima, M.A.; Silva, S.V.; Jaeger, R.G.; Freitas, V.M. Progesterone decreases ovarian cancer cells migration and invasion. Steroids 2020, 161, 108680. [Google Scholar] [CrossRef]
- Jeon, S.-Y.; Hwang, K.-A.; Choi, K.-C. Effect of steroid hormones, estrogen and progesterone, on epithelial mesenchymal transition in ovarian cancer development. J. Steroid Biochem. Mol. Biol. 2016, 158, 1–8. [Google Scholar] [CrossRef]
- Morikawa, A.; Kawabata, A.; Shirahige, K.; Akiyama, T.; Okamoto, A.; Sutani, T. Altered cervicovaginal microbiota in premenopausal ovarian cancer patients. Gene 2022, 811, 146083. [Google Scholar] [CrossRef] [PubMed]
- Asante, D.-B.; Calapre, L.; Ziman, M.; Meniawy, T.M.; Gray, E.S. Liquid biopsy in ovarian cancer using circulating tumor DNA and cells: Ready for prime time? Cancer Lett. 2020, 468, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Siu, M.K.; Ngan, H.Y.; Chan, K.K. Molecular biomarkers for the early detection of ovarian cancer. Int. J. Mol. Sci. 2022, 23, 12041. [Google Scholar] [CrossRef]
- Jacobson, D.; Moore, K.; Gunderson, C.; Rowland, M.; Austin, R.; Honap, T.P.; Xu, J.; Warinner, C.; Sankaranarayanan, K.; Lewis, C.M., Jr. Shifts in gut and vaginal microbiomes are associated with cancer recurrence time in women with ovarian cancer. PeerJ 2021, 9, e11574. [Google Scholar] [CrossRef]
- Ley, R.E. Prevotella in the gut: Choose carefully. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 69–70. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Gu, Y.; He, Q.; Huang, J.; Song, Y.; Wan, X.; Li, Y. Integrated analysis of microbiome and transcriptome data reveals the interplay between commensal bacteria and fibrin degradation in endometrial cancer. Front. Cell. Infect. Microbiol. 2021, 11, 748558. [Google Scholar] [CrossRef]
- Miao, R.; Badger, T.C.; Groesch, K.; Diaz-Sylvester, P.L.; Wilson, T.; Ghareeb, A.; Martin, J.A.; Cregger, M.; Welge, M.; Bushell, C. Assessment of peritoneal microbial features and tumor marker levels as potential diagnostic tools for ovarian cancer. PLoS ONE 2020, 15, e0227707. [Google Scholar] [CrossRef]
- Li, C.; Feng, Y.; Yang, C.; Wang, D.; Zhang, D.; Luo, X.; Zhang, H.; Huang, H.; Zhang, H.; Jiang, Y. Association between vaginal microbiota and the progression of ovarian cancer. J. Med. Virol. 2023, 95, e28898. [Google Scholar] [CrossRef]
- Zhou, B.; Sun, C.; Huang, J.; Xia, M.; Guo, E.; Li, N.; Lu, H.; Shan, W.; Wu, Y.; Li, Y. The biodiversity composition of microbiome in ovarian carcinoma patients. Sci. Rep. 2019, 9, 1691. [Google Scholar] [CrossRef]
- Xu, J.; Peng, J.-J.; Yang, W.; Fu, K.; Zhang, Y. Vaginal microbiomes and ovarian cancer: A review. Am. J. Cancer Res. 2020, 10, 743. [Google Scholar]
- Asangba, A.E.; Chen, J.; Goergen, K.M.; Larson, M.C.; Oberg, A.L.; Casarin, J.; Multinu, F.; Kaufmann, S.H.; Mariani, A.; Chia, N. Diagnostic and prognostic potential of the microbiome in ovarian cancer treatment response. Sci. Rep. 2023, 13, 730. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.I.; Kang, N.; Leem, S.; Yang, J.; Jo, H.; Lee, M.; Kim, H.S.; Dhanasekaran, D.N.; Kim, Y.-K.; Park, T. Metagenomic analysis of serum microbe-derived extracellular vesicles and diagnostic models to differentiate ovarian cancer and benign ovarian tumor. Cancers 2020, 12, 1309. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.; Zhang, X.; Fan, Y.; Chen, L.; Ma, X.; Yu, H.; Li, J.; Guan, X.; Zhao, P.; Yang, J. Changes of intestinal microbiota in ovarian cancer patients treated with surgery and chemotherapy. Cancer Manag. Res. 2020, 12, 8125. [Google Scholar] [CrossRef]
- Shin, N.-R.; Whon, T.W.; Bae, J.-W. Proteobacteria: Microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 2015, 33, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Chaput, N.; Lepage, P.; Coutzac, C.; Soularue, E.; Le Roux, K.; Monot, C.; Boselli, L.; Routier, E.; Cassard, L.; Collins, M.; et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann. Oncol. 2017, 28, 1368–1379. [Google Scholar] [CrossRef] [PubMed]
- Routy, B.; Gopalakrishnan, V.; Daillère, R.; Zitvogel, L.; Wargo, J.A.; Kroemer, G. The gut microbiota influences anticancer immunosurveillance and general health. Nat. Rev. Clin. Oncol. 2018, 15, 382–396. [Google Scholar] [CrossRef] [PubMed]
- Ou, J.; Carbonero, F.; Zoetendal, E.G.; DeLany, J.P.; Wang, M.; Newton, K.; Gaskins, H.R.; O’Keefe, S.J. Diet, microbiota, and microbial metabolites in colon cancer risk in rural Africans and African Americans. Am. J. Clin. Nutr. 2013, 98, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Q.; Du, H.; Cheng, X.; Cheng, X.; Tang, Y.; Pan, L.; Wang, Q.; Lin, J. Characteristics of fecal gut microbiota in patients with colorectal cancer at different stages and different sites. Oncol. Lett. 2019, 18, 4834–4844. [Google Scholar] [CrossRef]
- Flórez, A.B.; Sierra, M.; Ruas-Madiedo, P.; Mayo, B. Susceptibility of lactic acid bacteria, bifidobacteria and other bacteria of intestinal origin to chemotherapeutic agents. Int. J. Antimicrob. Agents 2016, 48, 547–550. [Google Scholar] [CrossRef]
- Viaud, S.; Saccheri, F.; Mignot, G.; Yamazaki, T.; Daillère, R.; Hannani, D.; Enot, D.P.; Pfirschke, C.; Engblom, C.; Pittet, M.J. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science 2013, 342, 971–976. [Google Scholar] [CrossRef]
- Voorde, J.V.; Sabuncuoğlu, S.; Noppen, S.; Hofer, A.; Ranjbarian, F.; Fieuws, S.; Balzarini, J.; Liekens, S. Nucleoside-catabolizing enzymes in mycoplasma-infected tumor cell cultures compromise the cytostatic activity of the anticancer drug gemcitabine. J. Biol. Chem. 2014, 289, 13054–13065. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Wang, N.; Han, W.; Ban, M.; Sun, T.; Xu, J. Vaginal and tumor microbiomes in gynecological cancer. Oncol. Lett. 2023, 25, 153. [Google Scholar] [CrossRef] [PubMed]
- Routy, B.; Le Chatelier, E.; Derosa, L.; Duong, C.P.; Alou, M.T.; Daillère, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P. Gut microbiome influences efficacy of PD-1–based immunotherapy against epithelial tumors. Science 2018, 359, 91–97. [Google Scholar] [CrossRef]
- Gopalakrishnan, V.; Spencer, C.N.; Nezi, L.; Reuben, A.; Andrews, M.; Karpinets, T.; Prieto, P.; Vicente, D.; Hoffman, K.; Wei, S. Gut microbiome modulates response to anti–PD-1 immunotherapy in melanoma patients. Science 2018, 359, 97–103. [Google Scholar] [CrossRef]
- Cui, M.; Xiao, H.; Li, Y.; Zhou, L.; Zhao, S.; Luo, D.; Zheng, Q.; Dong, J.; Zhao, Y.; Zhang, X. Faecal microbiota transplantation protects against radiation-induced toxicity. EMBO Mol. Med. 2017, 9, 448–461. [Google Scholar] [CrossRef] [PubMed]
- Hefazi, M.; Patnaik, M.M.; Hogan, W.J.; Litzow, M.R.; Pardi, D.S.; Khanna, S. Safety and efficacy of fecal microbiota transplant for recurrent Clostridium difficile infection in patients with cancer treated with cytotoxic chemotherapy: A single-institution retrospective case series. Mayo Clin. Proc. 2017, 92, 1617–1624. [Google Scholar] [CrossRef] [PubMed]
- Chambers, L.M.; Esakov Rhoades, E.L.; Bharti, R.; Braley, C.; Tewari, S.; Trestan, L.; Alali, Z.; Bayik, D.; Lathia, J.D.; Sangwan, N. Disruption of the gut microbiota confers cisplatin resistance in epithelial ovarian cancer. Cancer Res. 2022, 82, 4654–4669. [Google Scholar] [CrossRef]
- Cohen, C.R.; Wierzbicki, M.R.; French, A.L.; Morris, S.; Newmann, S.; Reno, H.; Green, L.; Miller, S.; Powell, J.; Parks, T. Randomized trial of lactin-V to prevent recurrence of bacterial vaginosis. N. Engl. J. Med. 2020, 382, 1906–1915. [Google Scholar] [CrossRef]
- Lev-Sagie, A.; Goldman-Wohl, D.; Cohen, Y.; Dori-Bachash, M.; Leshem, A.; Mor, U.; Strahilevitz, J.; Moses, A.E.; Shapiro, H.; Yagel, S. Vaginal microbiome transplantation in women with intractable bacterial vaginosis. Nat. Med. 2019, 25, 1500–1504. [Google Scholar] [CrossRef]
- Deng, X.; Li, Z.; Li, G.; Li, B.; Jin, X.; Lyu, G. Comparison of microbiota in patients treated by surgery or chemotherapy by 16S rRNA sequencing reveals potential biomarkers for colorectal cancer therapy. Front. Microbiol. 2018, 9, 1607. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).