Abstract
Radical hysterectomy (RH) is the standard treatment for early stage cervical cancer, but the surgical approach for locally bulky-size cervical cancer (LBS-CC) is still unclear. We retrospectively compared the outcomes of women with LBS-CC treated with neoadjuvant chemotherapy (NACT) and subsequent RH between the robotic (R-RH) and abdominal approaches (A-RH). Between 2012 and 2014, 39 women with LBS-CC FIGO (International Federation of Gynecology and Obstetrics) stage IB2–IIB were treated with NACT-R-RH (n = 18) or NACT-A-RH (n = 21). Surgical parameters and prognosis were compared. Patient characteristics were not significantly different between the groups, but the NACT-R-RH group had significantly more patients with FIGO stage IIB disease, received multi-agent-based NACT, and had a lower percentage of deep stromal invasion than the NACT-A-RH group. After NACT-R-RH, surgical parameters were better, but survival outcomes, such as disease-free survival (DFS) and overall survival (OS), were significantly worse. On multivariate analysis, FIGO stage IIB contributed to worse DFS (p = 0.003) and worse OS (p = 0.012) in the NACT-A-RH group. Women with LBS-CC treated with NACT-R-RH have better perioperative outcomes but poorer survival outcomes compared with those treated with NACT-A-RH. Thus, patients with FIGO stage IIB LBS-CC disease might not be suitable for surgery after multi-agent-based NACT.
1. Introduction
Surgical treatment for cervical cancer (CC) has been discussed intensively owing to recent advances in minimally invasive surgery (MIS). The technical feasibility of minimally invasive radical hysterectomy (MIS-RH) has been previously described in numerous reports [1,2,3,4,5], but there are increasing concerns regarding the oncological outcomes, especially on the basis of the results of the recent prospective randomized trial, i.e., the international laparoscopic approach to cervical cancer (LACC) trial [6,7,8,9,10]. Conventional abdominal RH (A-RH) showed better disease-free survival (DFS) and overall survival (OS) than MIS-RH [8,9], but the current evidence is still debatable [11,12,13,14,15]. The reason is probably because MIS-RH includes both conventionally laparoscopic RH (L-RH) and robotic RH (R-RH), and the techniques needed for conventional laparoscopic surgery are much more advanced and complicated than those for robotic surgery. L-RH requires more time for surgeons to develop the essential level of skill to perform the procedure effectively and has a longer learning curve. Many studies have compared the feasibility of these two different MIS techniques for treating different types of gynecological diseases, regardless of the malignancy status. Unsurprisingly, the results favor the robotic approach [16,17,18,19,20,21,22]. Therefore, when most MIS-RH procedures in the LACC trial were performed via conventional L-RH (84%) and contributed to poorer survival outcomes [9], one could argue that L-RH is not regularly performed in most cancer centers and the current practice and trend favor R-RH in many countries [12], which is also apparent in Taiwan [23,24].
It is also debatable whether RH can be performed instead of concurrent chemoradiotherapy (CCRT) for the management of locally advanced CC. In addition to the well-known criteria such as FIGO (International Federation of Gynecology and Obstetrics) staging, tumor size is among the most concerning factors with an increased possibility of therapeutic failure, and subsequently decreased DFS and OS, regardless of whether surgery was performed [25,26,27,28,29]. Many strategies have been suggested to overcome this limitation, and neoadjuvant chemotherapy (NACT) has been introduced for treating various types of malignancies [29,30,31,32,33,34] and locally advanced CC [28,29,30]. The main purpose of NACT is to reduce tumor size, thereby facilitating subsequent local treatment. Among patients with CC, those who received NACT for FIGO stage IB2/IIA2 CC were highly likely to undergo successful laparoscopic surgery [30]. Other advantages of NACT include the potential of controlling micro-metastatic disease and reducing the need for postoperative adjuvant radiotherapy with/without chemotherapy (CT) [35,36]. To date, only a few studies have evaluated the feasibility and safety of MIS-RH for patients with CC after NACT [37,38,39,40,41,42,43]. In our previous study, we evaluated the therapeutic effect of RH after NACT in patients with locally bulky-size cervical cancer (LBS-CC ≥ 6 cm). Weekly NACT with only cisplatin significantly decreased tumor size (from 6.4 ± 0.5 cm to 4.5 ± 1.4 cm), reduced blood loss (from 930 ± 356 mL to 558 ± 1328 mL, p < 0.001), and lowered the immediate complication rate (from 32% to 5.7%) during subsequent A-RH [28]. A meta-analysis of 18 studies with 1785 patients with CC (mainly FIGO stage IB2 to IIB) who were treated with cisplatin-based NACT before RH confirmed that the response to NACT was an indicator of progression-free survival (PFS) and OS [35], suggesting that these patients would have better oncological outcomes if the tumors showed a good response to NACT treatment. Therefore, in this retrospective study, we evaluated and compared the surgical and oncological outcomes of conventional A-RH and R-RH in patients with LBS-CC after NACT.
2. Materials and Methods
This retrospective study included patients from two oncological institutions: Taipei Veterans General Hospital (Taipei VGH) and Taipei Medical University Hospital (TMUH) in Taiwan between 2012 and 2014. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the TMU-Joint Institutional Review Board (project identification code: N201511005). A database search identified 39 patients with histologically confirmed LBS-CC (IB3, IIA2, and IIB, based on the 2018 FIGO staging system for cancer of the cervix uteri [25]) treated with platinum-based NACT followed by radical hysterectomy. Eighteen patients underwent NACT followed by robotic radical hysterectomy (i.e., the NACT-R-RH group) and 21 underwent NACT followed by conventional abdominal radical hysterectomy (i.e., the NACT-A-RH group). The medical charts including age, body mass index (BMI), clinical FIGO stage, pre-treatment tumor size on imaging, intra-operative operative parameters, and length of hospital stay (LOS) were assessed and these data collected. Information about the pathological status was also collected, including pathological tumor size, histology grade, number of resected lymph nodes (LNs), LN involvement, resection margin involvement, parametrial involvement, lymphovascular space invasion (LVSI), and deep stromal invasion (DSI). The pathological specimens were reviewed at each institution by board-certified gynecologic pathologists. The data from both the institutions were pooled into a combined analysis. All the patients who underwent R-RH were informed that the robotic procedure could be converted to A-RH if technological issues occurred.
2.1. Neoadjuvant Chemotherapy
All patients with histology-proven CC underwent a complete evaluation before NACT, including a vaginal-pelvic examination, chest radiography, and pelvic computed tomography or magnetic resonance imaging, to evaluate the clinical stage based on the FIGO Clinical Staging of Carcinoma of the Cervix Uteri (2008) and the revised 2018 FIGO staging [25]. Cystoscopy was performed for cases with clinically suspected involvement of the bladder, and colonoscopy was performed for cases with suspected involvement of the rectum. All patients underwent careful evaluation prior to NACT and the following criteria were used: World Health Organization (WHO) performance status of 0–2, adequate bone marrow reserve (absolute granulocyte count ≥ 2000/mL, platelet count ≥ 100,000/mL, and hemoglobin ≥ 10 g/dL); adequate renal, hepatic, pulmonary, and cardiac function; and absence of prior malignant diseases or major surgical illness. Two types of NACT protocols were utilized in this study: platinum-based (40 mg/m2) per week for 3 weeks, or taxane/platinum-based (60 mg/m2, 40 mg/m2) per week for 3 weeks. Surgery was performed at the end of the fourth week. Owing to the retrospective design of this study, the choice of the NACT regimen could not be controlled and it was administered at the discretion of each gynecological oncological surgeon.
2.2. Surgical Procedures
All the patients underwent detailed pre-operative evaluations. RH via either approach was type C1 hysterectomy as defined by the updated classification by Querleu and Morrow [44]. Bilateral pelvic lymph node dissection and para-aortic lymph node dissection were performed for all patients in the current study. A single surgeon (W.-M.L.) performed all the robotic surgeries on Da Vinci Si Surgical System® (Intuitive Surgical Inc.®, 1020 Kifer Road, Sunnyvale, CA, USA). Central docking between the patient’s legs was used, and all 4 robotic arms were used. One assistant trocar was placed in the left lower abdominal quadrant, and initial access was obtained at the level above the umbilicus by using the closed method.
2.3. Pre-Operative Preparation and Post-Operative Care
In both groups, patients received a preventive antibiotic dose of 1 g cefazolin within 30 min before the operation. Elastic compression bandages were applied to the lower extremities to prevent venous thromboembolism. With the patient under general anesthesia, the perineum and vagina were disinfected with povidone-iodine, followed by Foley catheter insertion. The abdomen was disinfected with 0.5% chlorhexidine and tincture of iodine. The operative time was defined from the time of making the skin incision to the completion of skin closure. Estimated blood loss was calculated by the difference in the total amounts of suctioned and irrigation fluids. The postoperative visual analog scale (VAS) pain scores were recorded by nursing staff up to 24 h postoperatively [45,46,47]. On arriving at the recovery room, all patients were immediately administered postoperative opioid (morphine as a 5-mg intravenous injection). Then, postoperative analgesics were administered (morphine as an intravenous infusion with an initial dose of 0.5 mg/h for 1 day and subsequent oral non-steroid anti-inflammatory agent on the next day). Adjuvant therapy was administered according to the presence of risk factors for recurrence considering the final pathology findings such as positive LNs, positive parametrial invasion, positive surgical margins, LVSI, DSI, and tumor size.
2.4. Statistical Analysis
Data were represented as the mean and standard deviation (SD) for continuous variables, and number and percentage for categorical variables. The Student t-test and Fisher exact test were used to compare the differences between the two groups for continuous variables and categorical variables, respectively. DFS was defined as the duration from the date of surgery to the date of diagnosis of local recurrence or distant metastasis. OS was defined as the duration from the date of surgery to the date of death. The DFS probability and OS probability were estimated using the Kaplan–Meier method. The log-rank test was used to compare differences in the survival probabilities for categorical variables. A univariate Cox proportional hazard model was used to quantify the risk effect on survival for each variable. Possible risk factors associated with survival probability at a significance level of 0.10 or less were included in a multivariable Cox proportional hazard model. A backward variable selection method was used to choose the final model. The level of significance was set at 0.05 for each test before the analysis. All data management and analysis were performed using R 3.5.3 [available at http://www.R-project.org/, R Development Core Team (2018), R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria].
3. Results
3.1. Clinical Characteristics and Pathological Status
The baseline clinical characteristics were not significantly different between the two groups, including patient age, BMI, comorbidities, a history of previous abdominal surgery, histology type, and tumor size on imaging (Table 1). However, significantly more patients had FIGO stage IIB CC (n = 17, 81.0%; and n = 4, 22.2%, respectively, all p < 0.001) and more patients were treated with multi-agent NACT (taxane/platinum-based) in the NACT-R-RH group than in the NACT-A-RH group (n = 19, 90.5%; and n = 4, 22.2%, respectively, all p < 0.001). The pathological data, including number of resected LNs, LN involvement, resection margin involvement, parametrial involvement, LVSI, and pathological tumor size, were not significantly different between both groups; however, DSI was found in all 18 patients in the NACT-A-RH group (100%) and in 9 patients in the NACT-R-RH group (42.9%; p < 0.001).

Table 1.
Clinical characteristics and pathological status.
3.2. Perioperative Outcomes and Follow-up Status
Intraoperative findings, such as operative time, estimated blood loss, blood transfusion rate, postoperative VAS score, and LOS were better in the NACT-R-RH group than in the NACT-A-RH group. The incidence of immediate intraoperative complications (bladder, bowel, nerve, and vascular injuries) did not differ between the groups. In the NACT-R-RH group, 2 patients had urinary bladder injury, which was repaired during surgery; in contrast, there were no cases of intraoperative complications in the NACT-A-RH group (Table 2). No conversion to laparotomy was needed in the NACT-R-RH group. The incidence of postoperative complications, such as infection, ileus, and obturator nerve palsy, did not differ between the groups. There were no readmissions due to postoperative complications or re-intervention in either group.

Table 2.
Perioperative outcomes and follow-up status.
The type of adjuvant therapy administered based on patient risk factors was not significantly different between the groups. The median follow-up time was not significantly different between the groups: 49.6 months (95% confidence interval [CI], 45.3–58.4 months) and 43.7 months (95% CI, 55.8–62.4 months) in the NACT-R-RH and NACT-A-RH group, respectively (p = 0.08). At the time of censoring, the sites of recurrences were not significantly different between the two groups. The disease recurrence rate was 47.6% in the NACT-R-RH group and 16.7% in the NACT-A-RH group, without significant difference (p = 0.092). Seven patients (33.3%) in the NACT-R-RH group and 1 (5.6%) in the NACT-A-RH group died of disease (p = 0.049). The perioperative outcomes and follow-up status are summarized in Table 2.
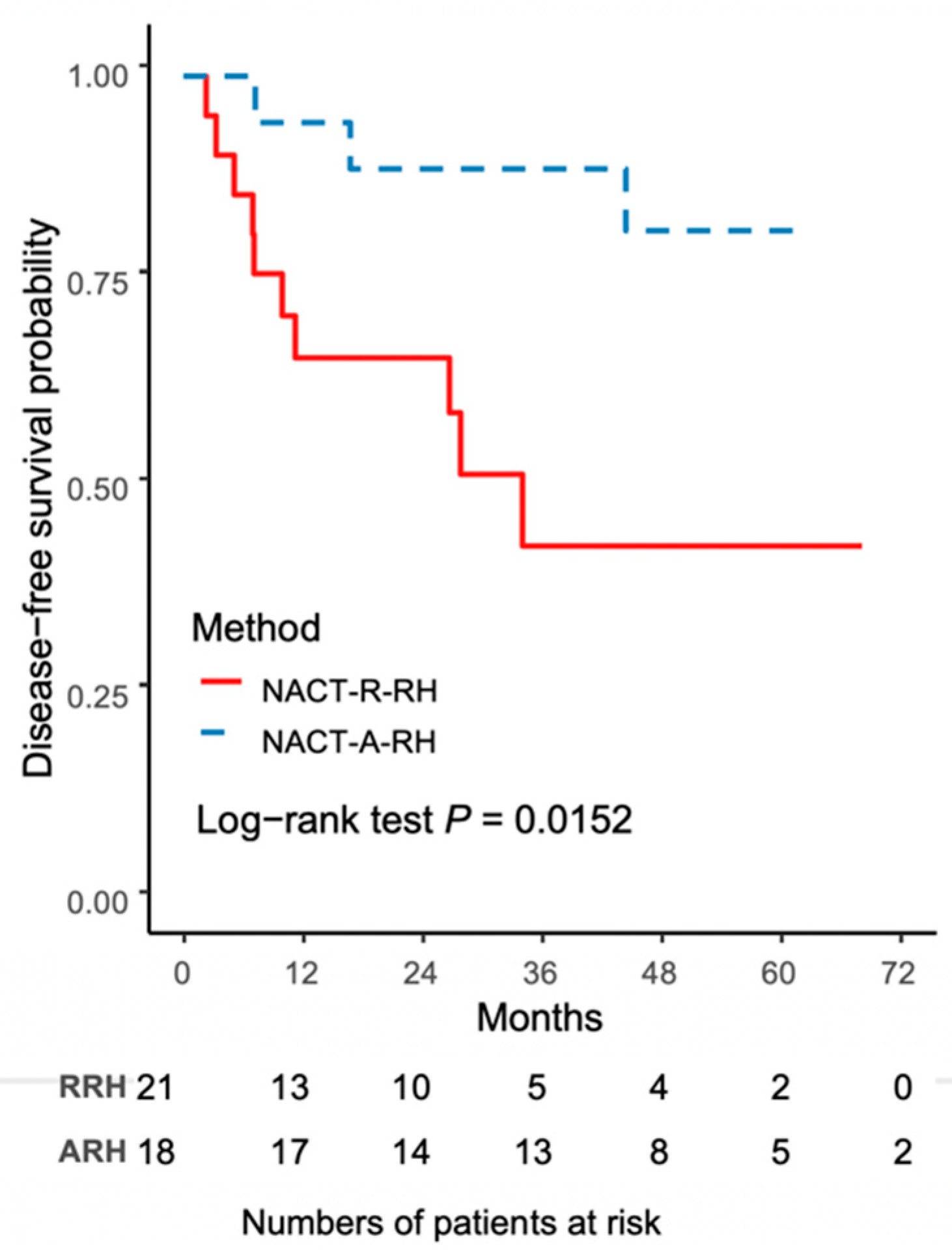
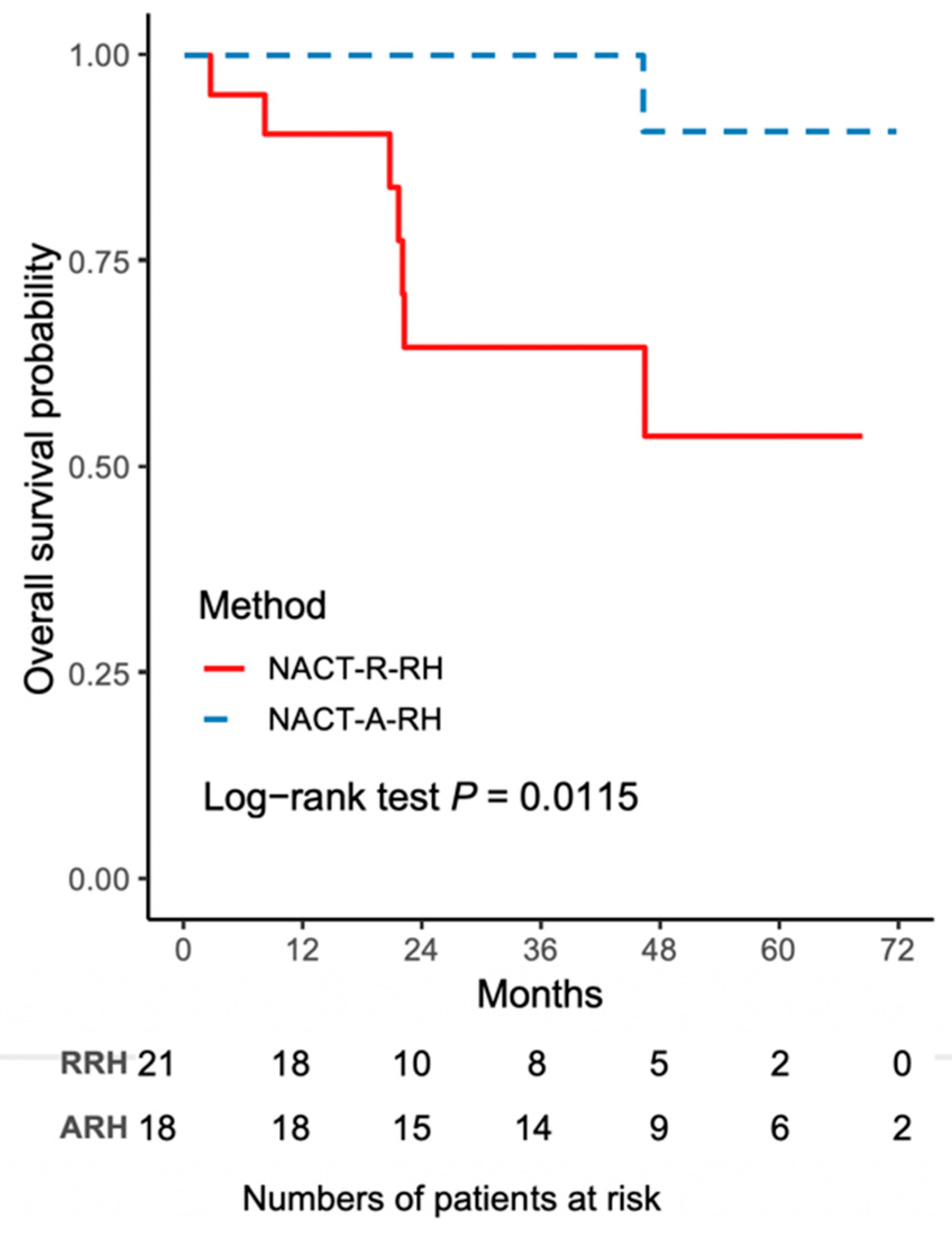
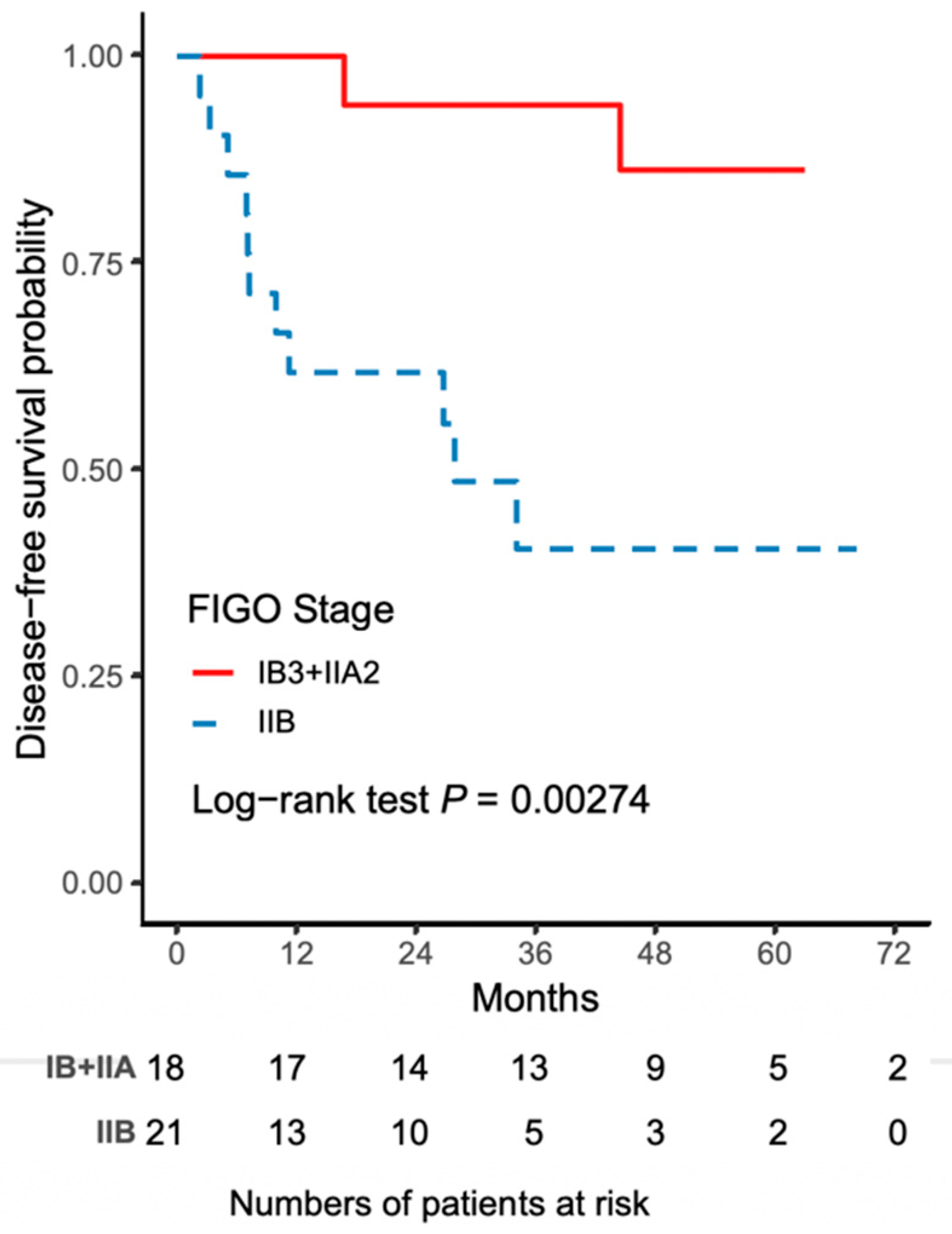
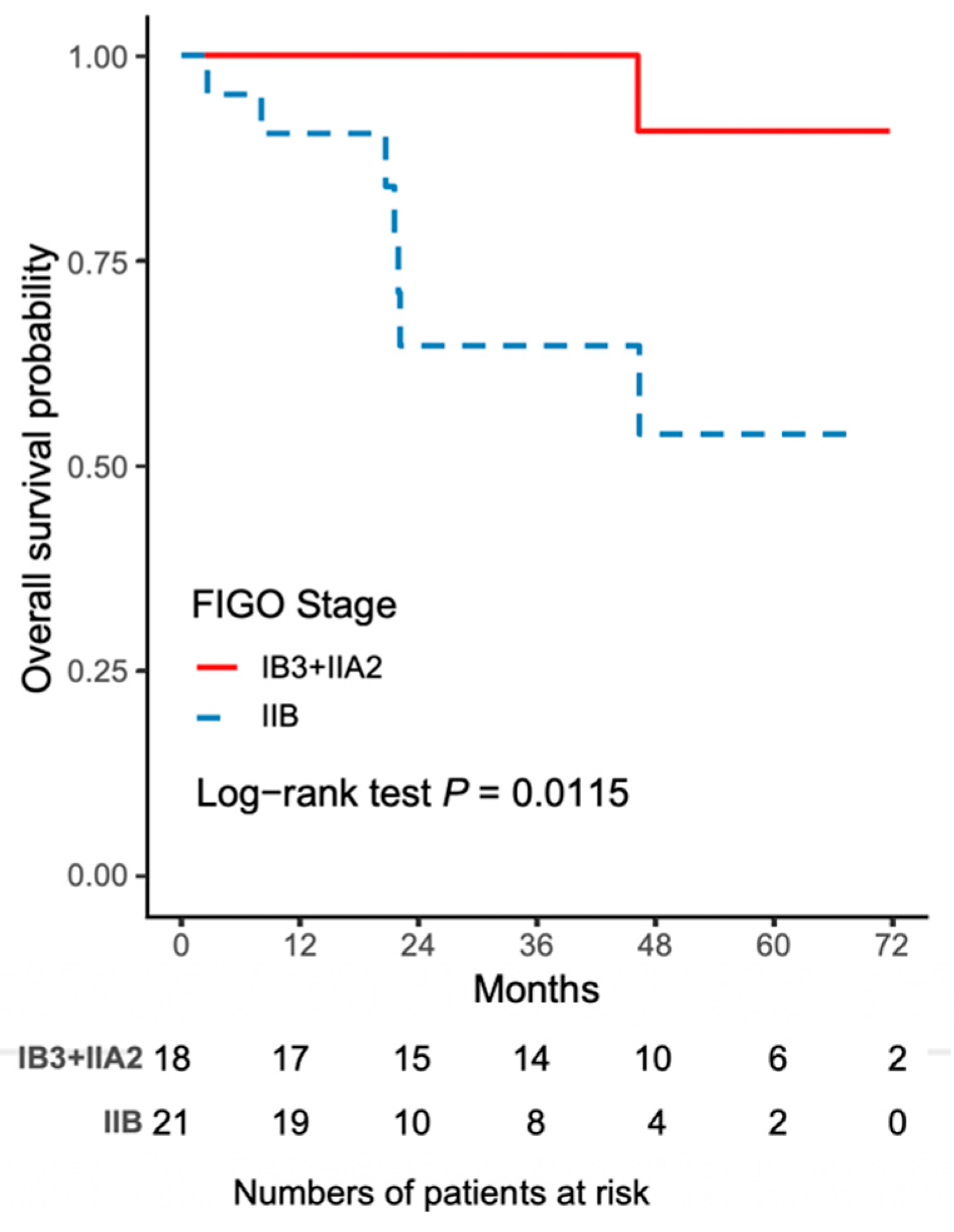
The worse outcome of patients in the NACT-R-RH group was further reflected by the results on Kaplan-Meir survival analysis. The 5-year DFS rate was 43.3% (95% CI 18.8–65.8) for the NACT-R-RH group and 81.5% (95% CI 52.3–93.7) for the NACT-A-RH group (p = 0.0152), as shown in Figure 1. A significantly worse prognosis was noted in patients in the NACT-R-RH group, along with decreased DFS in the 5-year follow-up period. At the end of the first postoperative year, 38% (n = 8) of patients in the NACT-R-RH group experienced recurrence, which was significantly higher than the rate of 5.6% (n = 1) in the NACT-A-RH group, contributing to the worse prognosis in the 5-year follow-up. The worse DFS also contributed to the worse OS. The 5-year OS rate was 53.9% (95% CI 24.6–76.1) for the NACT-R-RH group and 90.9% (95% CI 50.8–98.7) for the NACT-A-RH group (p = 0.0115), as shown in Figure 2. This worse prognosis was significant at the 2-year follow-up; the mortality rate was 52.4% and 16.7% in the NACT-R-RH group and NACT-A-RH group, respectively. To identify the factors that contributed to the worse prognosis, univariate and multivariate analyses were performed. Risk factors associated with worse DFS are shown in Table 3 and Table 4. Univariate analysis for DFS revealed that the surgical approach (R-RH vs. A-RH), FIGO stage (IB3-IIA2 vs. IIB), NACT regimen (taxane/platinum-based vs. platinum-based), LOS, and intra-operative complications might be associated with worse DFS (Table 3). A further multivariate analysis was performed to identify the key factors, and the results showed that FIGO stage IIB and adenosquamous cell carcinoma were independent prognostic factors that shortened the DFS (Table 4). FIGO stage IIB significantly influenced DFS, with a hazard ratio (HR) of 8.71 (95% CI 2.32–48.72, p = 0.001). The tumor type of adenosquamous cell carcinoma also contributed to worse DFS (HR 8.54; 95% CI 1.20–63.21; Table 4). On evaluating the risk factors contributing to worse OS, a history of abdominal surgery, FIGO stage IIB, type of the NACT regimen, operative time, LOS, and intraoperative complications were risk factors that decreased OS (Table 5). On multivariate analysis, FIGO stage IIB was identified as the most important prognostic factor contributing to patient death (Table 6). The Kaplan–Meier curve for 5-year DFS comparing FIGO stage IIB and FIGO stage IB3 + IIA2 disease is shown in Figure 3. The Kaplan–Meier curve for 5-year OS comparing FIGO stage IIB and FIGO stage IB3 + IIA2 disease is shown in Figure 4.
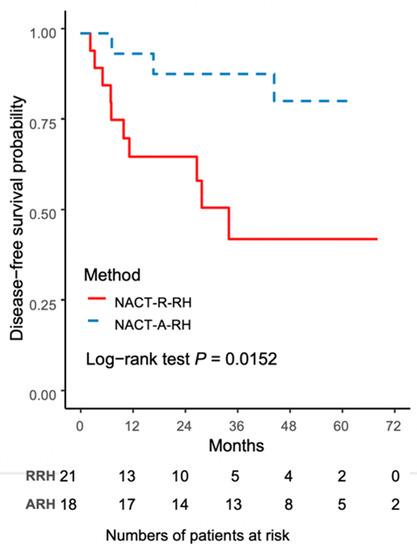
Figure 1.
The Kaplan–Meier curve for 5-year disease-free survival (DFS) comparing NACT-R-RH and NACT-A-RH.
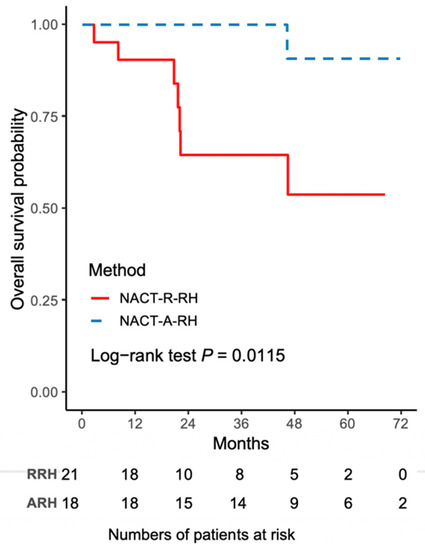
Figure 2.
The Kaplan–Meier curve for 5-year overall survival (OS) comparing NACT-R-RH and NACT-A-RH.

Table 3.
Univariate analysis of disease-free survival.

Table 4.
Multivariate analysis of disease-free survival.

Table 5.
Univariate analysis of overall survival.

Table 6.
Multivariate analysis of overall survival.
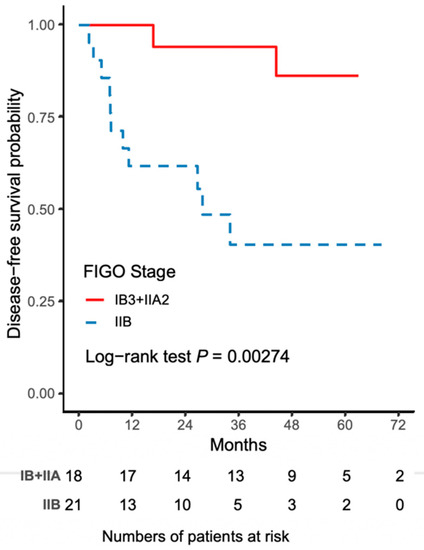
Figure 3.
The Kaplan–Meier curve for 5-year disease-free survival (DFS) comparing FIGO stage IIB and FIGO stage IB3 + IIA2 disease.
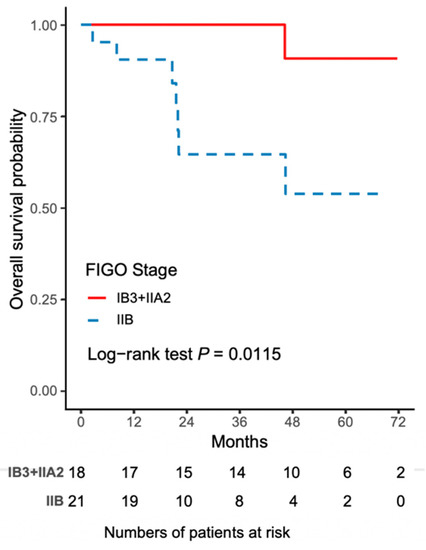
Figure 4.
The Kaplan–Meier curve for 5-year overall survival (OS) comparing FIGO stage IIB and FIGO stage IB3 + IIA2 disease.
4. Discussion
The results of the current study demonstrated that NACT-R-RH resulted in significantly better peri-operative outcomes than NACT-A-RH did, with a shorter operative time, lower estimated blood loss, fewer incidences of intraoperative blood transfusion, lower immediate postoperative and 24-h VAS scores, and shorter LOS, without increasing intraoperative complications; these results strongly indicate the feasibility of R-RH in patients with CC. This observation is not new, as a recent meta-analysis suggested that R-RH had surgical benefits for patients with CC than A-RH did [22]. Although the perioperative benefits of R-RH have been well documented, most studies were performed for early stage CC, regardless of whether patients received NACT treatment [12,16,17,18,19,20,21,22,37,38,39,40,41,42,43]. Some studies did not support the benefits of NACT in patients who were scheduled to undergo surgery, especially patients needing MIS [48,49,50]. One of the frequent observations was the desmoplastic effect of the tissue after NACT, making tissue dissection much more difficult during surgery [48,49,50]. In the current study, although 2 patients in the NACT-R-RH group had bladder injury, compared to none in the NACT-A-RH group, we do not believe that this complication is due to the desmoplastic effect of NACT, because all patients in the NACT-R-RH group underwent complete R-RH and no conversion to laparotomy was necessary. Furthermore, intra- and postoperative parameters, such as operative time, estimated blood loss, the need for blood transfusion, and LOS, were significantly better in the NACT-R-RH group than in the NACT-A-RH group; this finding was similar to the findings of previous studies that showed the benefits of NACT, without increasing the difficulty in performing MIS techniques [40,42,51]. In another study, the median estimated blood loss, operative time, and LOS were significantly better after NACT-R-RH than after NACT-A-RH [51]. In a previous study, although the mean operative time was longer in the NACT-R-RH group than in the NACT-A-RH group, other intra-operative parameters, such as estimated blood loss and LOS, were still significantly better in the NACT-R-RH group [50]. In both studies, no significant difference was observed in the recurrence pattern, DFS, and OS during the 3-year follow-up between NACT-R-RH and NACT-A-RH [50,51]. A recent review of many previous case-control and retrospective case series also demonstrated the safety of NACT-R-RH considering the oncological outcomes [52].
The goal of NACT includes downstaging the tumor to improve the radical curability and safety of surgery and inhibit micro-metastasis and distant metastasis [29,30,31,32,33,34,35,36], as well as allow surgeons to perform less radical surgery without compromising oncological safety [52,53]. An early Cochrane review showed a significant decrease in adverse pathological findings after NACT (odds ratio [OR] 0.54, 95% CI 0.40–0.73, p < 0.0001 for LN status; OR 0.58, 95% CI 0.41–0.82, p = 0.002 for parametrial infiltration), contributing to an improvement in both OS and PFS. These results appear to indicate that NACT may offer benefits over surgery alone for women with LBS-CC, although the effects were less clear for all other pre-specified outcomes [53]. A recent meta-analysis also supported the benefits of decreasing the severity of disease status in patients with LBS-CC treated with NACT, based on the significantly decreased rate of LN metastasis, parametrial infiltration, and recurrence [54]. Thus, NACT is an acceptable and effective procedure for selected patients with LBS-CC, although the analysis did not support advantages in survival (DFS, PFS, and OS) of patients with LBS-CC treated with NACT [54]. In fact, the impact on survival after NACT is still highly debatable, with no consensus about whether it can result in better outcomes [28,29,30,35,36,54,55,56,57,58,59]. Accordingly, the term “acquired treatment response” seems important. A meta-analysis showed varying response rates after NACT for patients with LBS-CC, ranging from 58.5% to 86.5% for the clinical response rate and from 7.5% to 78.8% for the pathological response rate [57]. The clinical response rate predicted favorable DFS (HR 2.36, 95% CI 1.82–3.06) and OS (HR 3.36, 95% CI 2.41–4.69) [57]. Moreover, the pathological response also predicted favorable outcomes such as DFS (HR 3.61, 95% CI 2.0–6.52) and OS (HR 5.45, 95% CI 3.42–8.7) [57]. Another meta-analysis further suggested the importance of an early response, because an early response to NACT is associated with favorable outcomes [58]. However, it is difficult to define “early response”. In the current study, some adverse surgical and pathological parameters seemed to be improved after NACT, although the clinical relevance is unclear. We did not use the same evaluation tools to compare the changes before and after treatment. For example, in the current study, the tumor size decreased by approximately 25% (from 4.6 cm to 3.4 cm in the NACT-R-RH group and from 5.1 cm to 3.6 cm in the NACT-A-RH group, respectively). The size before treatment was calculated using preoperative imaging but the size after treatment was based on postoperative pathological measurement. It is well known that formalin-fixed tissue is significantly smaller than tissue before the process of formalin fixation, with the percentage of shrinkage being approximately 25–30% [28]; therefore, in the current study, tumor size showed almost no response, which might partly explain the worse outcomes.
Another issue in the current study is the use of different NACT regimens for the patients with LBS-CC. In the NACT-R-RH group, more patients received NACT with taxane/platinum-based regimens. It is unclear whether the different NACT regimens contributed to varied outcome in our study. In another study, among women with LBS-CC treated with NACT-RH, taxane/platinum-based regimens had a similar effect on DFS and cause-specific survival compared to non-taxane/platinum-based regimens, irrespectively of the tumor type (squamous cell carcinoma and non-squamous cell carcinoma) [55]. In the current study, the NACT regimen administered was significantly different between the NACT-R-RH and NACT-A-RH groups, and the taxane/platinum-based regimens were administered much more frequently in the NACT-R-RH group. The results from univariate analysis showed that the taxane/platinum-based regimen was associated with worse prognosis, but it may have been influenced by other confounding factors. Further multivariate analysis was performed, and the results did not establish any relationship between different NACT regimens and prognosis. Hence, as the evidence about the beneficial roles of NACT is still not conclusive, further study is warranted.
The worse prognosis of patients with LBS-CC treated with NACT-R-RH demonstrated in the current study led us to re-visit recent publications in the New England Journal of Medicine [8,9]. The LACC trial showed a decreased OS and a 3-fold increase in recurrence in the MIS-RH group than in the A-RH group, both of which were unexpected results [9], suggesting that MIS-RH should be used cautiously. In the current study, FIGO stage IIB was the most important indicator for worse outcomes of patients with LBS-CC treated with NACT-RH. Per the new FIGO staging of cancer of the cervix uteri (2018), invasive carcinoma limited to the cervix with the greatest dimension of ≥2 cm and <4 cm is classified as stage IB2, carcinoma >4 cm as stage IB3, and carcinoma that is limited to the upper two-thirds of the vagina without parametrial invasion and >4 cm as stage IIA2 [25]. This new FIGO staging system of CC emphasizes the importance of tumor size. In a previous study, Wright et al. used the National Cancer Database (n = 62,212, data used were of patients treated between 2004 and 2015) to examine the prognostic performance of the 2018 CC staging system; the 5-year survival rate was 91.6% (95% CI 90.4–92.6%) for stage IB1 CC, 83.3% (95% CI 81.8–84.8%) for stage IB2 CC, and 76.1% (95% CI 74.3–77.8%) for IB3 CC [60]. Another group used the data of The Surveillance, Epidemiology, and End Results Program collected between 1988 and 2014 (n = 8909) to validate this new FIGO CC staging system and also confirmed that this system was valuable for distinguishing survival outcomes of patients with FIGO stage IB CC [61]. In that study, the HR of cause-specific survival of 2018 FIGO stage IB3 CC and 2018 FIGO stage IB2 CC was 4.07 (95% CI 3.33–4.97) and 1.98 (95% CI 1.62–2.41), respectively, compared to that of 2018 FIGO stage IB1 CC [61]. On multivariable analysis for cause-specific survival in the FIGO stage IB cohort based on 2018 FIGO stage IB2, the HR for 2018 FIGO stage IB3 was 2.06 (95% CI 1.76–2.41) and HR for 2018 FIGO stage IB1 was 0.51 (95% CI 0.42–0.95) [61]. Thus, the 2018 FIGO CC staging system reflected the effect of screening and prevention programs for CC [62,63,64,65,66,67], as the proportion of 2018 FIGO stage IB1 (n = 3604, 40.5%) and stage IB2 disease (n = 3620, 40.6%) was similar while the percentage of 2018 FIGO stage IB3 disease was only 18.9% (n = 1685) [61]. It was anticipated that researchers would find trends in the significantly increasing proportion of 2018 FIGO stage IB1 CC and in the decrease in the 2018 FIGO stage IB3 disease during the study period [61]. The data presented in the current study were consistent with the new 2018 FIGO staging system for CC, and we re-considered the treatment strategy for patients with LBS-CC. Although the cut-off value of 2 cm (2018 FIGO stage IB1 CC) might be applicable and RH could be performed as the MIS procedure [68,69], this cut-off is still debatable [8,9]. Therefore, we strongly agree with the notice of the Society of Gynecologic Oncology (SGO) announced in November 2018, that gynecological oncologists should be aware of the emerging data on MIS for CC so that “thorough discussions can be undertaken with patients and shared decision-making can be used when choosing the surgical approach for RH”. Moreover, the results of the LACC trial, together with institutional data, should be discussed with patients before choosing MIS-RH.
To address the potential risks of MIS for gynecological cancer, many researchers have suggested and hypothesized possible mechanisms that contribute to worse prognosis, thereby assisting in minimizing tumor dissemination during surgery [70,71,72,73,74,75,76,77,78]. Most hypotheses are mostly inconclusive and require larger randomized controlled trials. In addition, MIS-RH is indeed a challenging surgical procedure compared with conventional total hysterectomy [78]. As the surgical technique for MIS-RH varies greatly and is extremely difficult to control for with randomization or multivariate adjustment [69], good training to perform the technique delicately is of paramount importance [79,80,81,82,83].
Several limitations of the current study are apparent due to the unfair comparison between two groups. The first limitation is the small sample size of both groups. The wide use of effective universal screening for cervical cancer (pap smear) in developed countries [62,84,85,86,87] has led to a dramatic decrease in LBS-CC prevalence rate. Moreover, the therapeutic choice for patients with LBS-CC is debated [88]; therefore the patients enrolled in the current study were mainly based on shared decision making, which directly influenced the sample size, resulting in an insufficient power to properly compare PFS and OS between the two types of surgery after NACT. Second, based on the same reason shown above, an additional cost is required for receiving robotic surgery, which is not covered by the National Insurance Health Care System in Taiwan [89,90,91,92,93,94], and the risk of selection bias could not be totally avoided, contributing to a significantly greater number of patients with FIGO stage IIB in the robotic group. This is a well-known limitation of the retrospective design. The effects of both limitations were combined and augmented by the confounding effects. Finally, imaging evaluation was not used at the end of NACT treatment. Therefore, the response of the patients to NACT was uncertain.
Despite these limitations, the current study provided valuable data showing that women with LBS-CC treated with NACT followed by surgery required further evaluation, especially for those patients with FIGO stage IIB. Our study confirmed that the seemingly promising perioperative outcomes in the NACT-R-RH group did not translate into better oncological outcomes. In contrast, they were associated with higher recurrence and mortality rates. Therefore, it is clear that any advanced technology (robotic surgery) in cancer treatment should be testified by survival outcomes, regardless of perioperative outcomes. In addition, the current study provided additional evidence that FIGO stage IIB contributed greatly to a poorer prognosis if NACT-R-RH was performed. Therefore, it is reasonable to suppose that NACT-A-RH might be a more appropriate approach in patients with FIGO stage IB3-IIA2 LBS-CC if the patients plan to undergo NACT along with a subsequent surgical intervention.
5. Conclusions
R-RH after NACT in patients with LBS-CC results in better perioperative outcomes but does not contribute to better survival outcomes. In addition, NACT-R-RH is associated with higher rates of recurrence and mortality, and the poor oncological outcomes occur relatively earlier compared to that observed after NACT-A-RH. Patients with FIGO stage IIB LBS-CC should be well informed about the possible inferior survival outcomes before undergoing NACT-R-RH.
Author Contributions
Conceptualization, W.-M.L. and P.-H.W.; formal analysis, J.C.-F.L.; investigation, C.-H.L.; resources, Y.-C.L. and N.-R.L.; data curation, I.-S.C. and W.-H.C.; methodology, P.-H.W.; writing—original draft, C.-H.L.; writing—review and editing, P.-H.W.; supervision, W.-M.L. and P.-H.W.; project administration, W.-M.L. and P.-H.W.
Funding
This research was funded by Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan (DP2-108-21121-01-A-07), and also supported by grants from the Taipei Veterans General Hospital (V108C-085) and from the Ministry of Science and Technology, Executive Yuan (MOST: 106-2314-B-075-061-MY3), Taipei, Taiwan. The authors appreciate the financial support from the Female Cancer Foundation, Taipei, Taiwan.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Willows, K.; Kupets, R.; Diong, C.; Vicus, D.; Covens, A.; Gien, L.T. Rates over time and regional variation of radical minimally invasive surgery for cervical cancer: A population based study. Gynecol. Oncol. 2019, 154, 338–344. [Google Scholar] [CrossRef]
- Behbehani, S.; Suarez-Salvador, E.; Buras, M.; Magtibay, P.; Magrina, J. Mortality rates in laparoscopic and robotic gynecologic oncology surgery—A systemic review and meta-analysis. J. Minim. Invasive Gynecol. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Uppal, S.; Rebecca Liu, J.; Kevin Reynolds, R.; Rice, L.W.; Spencer, R.J. Trends and comparative effectiveness of inpatient radical hysterectomy for cervical cancer in the United States (2012–2015). Gynecol. Oncol. 2019, 152, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Gil-Moreno, A.; Carbonell-Socias, M.; Salicrú, S.; Centeno-Mediavilla, C.; Franco-Camps, S.; Colas, E.; Oaknin, A.; Pérez-Benavente, A.; Díaz-Feijoo, B. Radical hysterectomy: Efficacy and safety in the dawn of minimally invasive techniques. J. Minim. Invasive Gynecol. 2019, 26, 492–500. [Google Scholar] [CrossRef] [PubMed]
- Corrado, G.; Vizza, E.; Legge, F.; Pedone Anchora, L.; Sperduti, I.; Fagotti, A.; Mancini, E.; Gallotta, V.; Zampa, A.; Chiofalo, B.; et al. Comparison of different surgical Approaches for stage IB1 cervical cancer patients: A multi-institution study and a review of the literature. Int. J. Gynecol. Cancer 2018, 28, 1020–1028. [Google Scholar] [CrossRef]
- Cusimano, M.C.; Baxter, N.N.; Gien, L.T.; Moineddin, R.; Liu, N.; Dossa, F.; Willows, K.; Ferguson, S.E. Impact of surgical approach on oncologic outcomes in women undergoing radical hysterectomy for cervical cancer. Am. J. Obstet. Gynecol. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Doo, D.W.; Kirkland, C.T.; Griswold, L.H.; McGwin, G.; Huh, W.K.; Leath, C.A.; Kim, K.H. Comparative outcomes between robotic and abdominal radical hysterectomy for IB1 cervical cancer: Results from a single high volume institution. Gynecol. Oncol. 2019, 153, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Melamed, A.; Margul, D.J.; Chen, L.; Keating, N.L.; Del Carmen, M.G.; Yang, J.; Seagle, B.L.; Alexander, A.; Barber, E.L.; Rice, L.W.; et al. Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N. Engl. J. Med. 2018, 379, 1905–1914. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, P.T.; Frumovitz, M.; Pareja, R.; Lopez, A.; Vieira, M.; Ribeiro, R.; Buda, A.; Yan, X.; Shuzhong, Y.; Chetty, N.; et al. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N. Engl. J. Med. 2018, 379, 1895–1904. [Google Scholar] [CrossRef]
- Fader, A.N. Surgery in cervical cancer. N. Engl. J. Med. 2018, 379, 1955–1957. [Google Scholar] [CrossRef]
- Lee, C.L.; Huang, K.G. Board of Directors of TAMIG. Clinical trial should be more rigorous. Taiwan J. Obstet. Gynecol. 2019, 58, 306–307. [Google Scholar] [CrossRef] [PubMed]
- Falconer, H.; Palsdottir, K.; Stalberg, K.; Dahm-Kähler, P.; Ottander, U.; Lundin, E.S.; Wijk, L.; Kimmig, R.; Jensen, P.T.; Zahl Eriksson, A.G.; et al. Robot-assisted approach to cervical cancer (RACC): An international multi-center, open-label randomized controlled trial. Int. J. Gynecol. Cancer 2019, 29, 1072–1076. [Google Scholar] [CrossRef] [PubMed]
- Martin-Hirsch, P.; Wood, N.; Whitham, N.L.; Macdonald, R.; Kirwan, J.; Anagnostopoulos, A.; Hutson, R.; Theophilou, G.; Otify, M.; Smith, M.; et al. Survival of women with early-stage cervical cancer in the UK treated with minimal access and open surgery. BJOG 2019, 126, 956–959. [Google Scholar] [CrossRef] [PubMed]
- Naumann, R.W. Minimally invasive radical hysterectomy has many benefits compared with open radical hysterectomy: Will the LACC trial cause the premature demise of this procedure? J. Minim. Invasive Gynecol. 2019, 26, 379–380. [Google Scholar] [CrossRef] [PubMed]
- Abdollah, F.; Keeley, J.; Menon, M. Minimally invasive or abdominal radical hysterectomy for cervical cancer. N. Engl. J. Med. 2019, 380, 793. [Google Scholar] [PubMed]
- Alfonzo, E.; Wallin, E.; Ekdahl, L.; Staf, C.; Rådestad, A.F.; Reynisson, P.; Stålberg, K.; Falconer, H.; Persson, J.; Dahm-Kähler, P. No survival difference between robotic and open radical hysterectomy for women with early-stage cervical cancer: Results from a nationwide population-based cohort study. Eur. J. Cancer 2019, 116, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Siesto, G.; Finco, A.; Portuesi, R.A.V.; Romano, F.; Ieda’, N.P.; Vitobello, D. Survival outcomes of robotic radical hysterectomy for early stage cervical cancer: A 9-year study. Int. J. Med. Robot. 2019, 15, e2003. [Google Scholar] [CrossRef] [PubMed]
- Piedimonte, S.; Czuzoj-Shulman, N.; Gotlieb, W.; Abenhaim, H.A. Robotic radical hysterectomy for cervical cancer: A population-based study of adoption and immediate postoperative outcomes in the United States. J. Minim. Invasive Gynecol. 2019, 26, 551–557. [Google Scholar] [CrossRef]
- Zhang, S.S.; Ding, T.; Cui, Z.H.; Lv, Y.; Jiang, R.A. Efficacy of Robotic Radical Hysterectomy for Cervical Cancer Compared with that of Open and Laparoscopic Surgery: A Separate Meta-Analysis of High-Quality Studies. Medicine 2019, 98, e14171. [Google Scholar] [CrossRef]
- Jin, Y.M.; Liu, S.S.; Chen, J.; Chen, Y.N.; Ren, C.C. Robotic radical hysterectomy is superior to laparoscopic radical hysterectomy and open radical hysterectomy in the treatment of cervical cancer. PLoS ONE 2018, 13, e0193033. [Google Scholar] [CrossRef]
- Shah, C.A.; Beck, T.; Liao, J.B.; Giannakopoulos, N.V.; Veljovich, D.; Paley, P. Surgical and oncologic outcomes after robotic radical hysterectomy as compared to open radical hysterectomy in the treatment of early cervical cancer. J. Gynecol. Oncol. 2017, 28, e82. [Google Scholar] [CrossRef] [PubMed]
- Shazly, S.A.; Murad, M.H.; Dowdy, S.C.; Gostout, B.S.; Famuyide, A.O. Robotic radical hysterectomy in early stage cervical cancer: A systematic review and meta-analysis. Gynecol. Oncol. 2015, 138, 457–471. [Google Scholar] [CrossRef]
- Chen, C.H.; Chen, H.H.; Liu, W.M. Feasibility and perioperative outcomes of robotic-assisted radical hysterectomy after neoadjuvant chemotherapy for locally advanced cervical cancer. J. Minim. Invasive Gynecol. 2015, 22, S21. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.L.; Wu, K.Y.; Huang, K.G.; Lee, P.S.; Yen, C.F. Long-term survival outcomes of laparoscopically assisted radical hysterectomy in treating early-stage cervical cancer. Am. J. Obstet. Gynecol. 2010, 203, e1–e7. [Google Scholar] [CrossRef] [PubMed]
- Bhatla, N.; Berek, J.S.; Cuello Fredes, M.; Denny, L.A.; Grenman, S.; Karunaratne, K.; Kehoe, S.T.; Konishi, I.; Olawaiye, A.B.; Prat, J.; et al. Revised FIGO staging for carcinoma of the cervix uteri. Int. J. Gynaecol. Obstet. 2019, 145, 129–135. [Google Scholar] [CrossRef]
- Arimoto, T.; Kawana, K.; Adachi, K.; Ikeda, Y.; Nagasaka, K.; Tsuruga, T.; Yamashita, A.; Oda, K.; Ishikawa, M.; Kasamatsu, T.; et al. Minimization of curative surgery for treatment of early cervical cancer: A review. Jpn. J. Clin. Oncol. 2015, 45, 611–616. [Google Scholar] [CrossRef][Green Version]
- Horn, L.C.; Bilek, K.; Fischer, U.; Einenkel, J.; Hentschel, B. A cut-off value of 2 cm in tumor size is of prognostic value in surgically treated FIGO stage IB cervical cancer. Gynecol. Oncol. 2014, 134, 42–46. [Google Scholar] [CrossRef]
- Wang, P.H.; Chang, Y.H.; Yang, Y.H.; Chang, W.H.; Huang, S.Y.; Lai, C.R.; Juang, C.M.; Chen, Y.J.; Horng, H.C.; Wen, K.C.; et al. Outcome of patients with bulky IB (≥6 cm) cervical squamous cell carcinoma with and without cisplatin-based neoadjuvant chemotherapy. Taiwan J. Obstet. Gynecol. 2014, 53, 330–336. [Google Scholar] [CrossRef]
- Kumar, L.; Pramanik, R.; Kumar, S.; Bhatla, N.; Malik, S. Neoadjuvant chemotherapy in gynaecological cancers—Implications for staging. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 790–801. [Google Scholar] [CrossRef]
- Zhao, H.; He, Y.; Zhu, L.R.; Wang, J.L.; Guo, H.Y.; Xu, T.; Wang, Y.Q.; Yao, Y.; Wu, Y.M. Effect of Neoadjuvant Chemotherapy Followed by Radical Surgery for FIGO Stage IB2/IIA2 Cervical Cancer: A Multi-Center Retrospective Clinical Study. Medicine 2019, 98, e15604. [Google Scholar] [CrossRef]
- Lin, J.W.; Hsu, C.P.; Yeh, H.L.; Chuang, C.Y.; Lin, C.H. The impact of pathological complete response after neoadjuvant chemoradiotherapy in locally advanced squamous cell carcinoma of esophagus. J. Chin. Med. Assoc. 2018, 81, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Kusunoki, S.; Terao, Y.; Hirayama, T.; Fujino, K.; Ujihira, T.; Ota, T.; Takeda, S. Safety and efficacy of neoadjuvant chemotherapy with bevacizumab in advanced-stage peritoneal/ovarian cancer patients. Taiwan J. Obstet. Gynecol. 2018, 57, 650–653. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.H. Neoadjuvant chemotherapy before definite operative approach for women with advanced-stage epithelial ovarian cancer. Taiwan J. Obstet. Gynecol. 2018, 57, 623–624. [Google Scholar] [CrossRef]
- Chen, M.Q.; Lin, Q.L.; Chen, Y.G.; Guo, J.H.; Xu, B.H.; Tian, Y. Neoadjuvant chemotherapy may not benefit esophageal squamous cell carcinoma patients treated with definitive chemoradiotherapy. J. Chin. Med. Assoc. 2017, 80, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Sardi, J.E.; Katsumata, N.; Ryu, H.S.; Nam, J.H.; Chung, H.H.; Park, N.H.; Song, Y.S.; Behtash, N.; Kamura, T.; et al. Efficacy of neoadjuvant chemotherapy in patients with FIGO stage IB1 to IIA cervical cancer: An international collaborative meta-analysis. Eur. J. Surg. Oncol. 2013, 39, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Katsumata, N.; Yoshikawa, H.; Kobayashi, H.; Saito, T.; Kuzuya, K.; Nakanishi, T.; Yasugi, T.; Yaegashi, N.; Yokota, H.; Kodama, S.; et al. Phase III randomised controlled trial of neoadjuvant chemotherapy plus radical surgery vs radical surgery alone for stages IB2, IIA2, and IIB cervical cancer: A Japan Clinical Oncology Group trial (JCOG 0102). Br. J. Cancer 2013, 108, 1957–1963. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, A.; Damiani, G.R.; Loverro, M.; Pirovano, C.; Fachechi, G.; Corso, S.; Trojano, G. Comparison of Robotic and laparoscopic Radical type-B and C hysterectomy for cervical cancer: Long term-outcomes. Acta Biomed. 2017, 88, 289–296. [Google Scholar] [PubMed]
- Raspagliesi, F.; Bogani, G.; Spinillo, A.; Ditto, A.; Bogliolo, S.; Casarin, J.; Leone Roberti Maggiore, U.; Martinelli, F.; Signorelli, M.; Gardella, B.; et al. Introducing nerve-sparing approach during minimally invasive radical hysterectomy for locally-advanced cervical cancer: A multi-institutional experience. Eur. J. Surg. Oncol. 2017, 43, 2150–2156. [Google Scholar] [CrossRef]
- Pellegrino, A.; Damiani, G.R.; Fachechi, G.; Pirovano, C.; Gaetani, M.; Youssef, A. Cost analysis of minimally invasive radical hysterectomy for cervical cancer performed by a single surgeon in an Italian center: An update in gynecologic oncological field. Updates Surg. 2017, 69, 517–522. [Google Scholar] [CrossRef]
- Minig, L.; Zanagnolo, V.; Cárdenas-Rebollo, J.M.; Colombo, N.; Maggioni, A. Feasibility of robotic radical hysterectomy after neoadjuvant chemotherapy in women with locally advanced cervical cancer. Eur. J. Surg. Oncol. 2016, 42, 1372–1377. [Google Scholar] [CrossRef]
- Vizza, E.; Corrado, G.; Mancini, E.; Vici, P.; Sergi, D.; Baiocco, E.; Patrizi, L.; Saltari, M.; Pomati, G.; Cutillo, G. Laparoscopic versus robotic radical hysterectomy after neoadjuvant chemotherapy in locally advanced cervical cancer: A case control study. Eur. J. Surg. Oncol. 2015, 41, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Vizza, E.; Corrado, G.; Zanagnolo, V.; Tomaselli, T.; Cutillo, G.; Mancini, E.; Maggioni, A. Neoadjuvant chemotherapy followed by robotic radical hysterectomy in locally advanced cervical cancer: A multi-institution study. Gynecol. Oncol. 2014, 133, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Vitobello, D.; Siesto, G.; Pirovano, C.; Ieda, N. Surgical outcomes of robotic radical hysterectomy after neoadjuvant chemotherapy for locally advanced cervical cancer: Comparison with early stage disease. Eur. J. Surg. Oncol. 2013, 39, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Querleu, D.; Cibula, D.; Abu-Rustum, N.R. 2017 Update on the Querleu-Morrow Classification of Radical Hysterectomy. Annal. Surg. Oncol. 2017, 24, 3406–3412. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.J.; Hsu, T.F.; Chang, Y.H.; Huang, B.S.; Jiang, L.Y.; Wang, P.H.; Chen, Y.J. Postoperative maintenance levonorgestrel-releasing intrauterine system for symptomatic uterine adenomyoma. Taiwan J. Obstet. Gynecol. 2018, 57, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.D.; Horng, H.C.; Liu, C.H.; Hsiao, S.M.; Chen, Y.J.; Chang, W.H.; Wang, P.H. Comparison of single-port and three-port laparoscopic salpingectomy in the management for tubal pregnancy. J. Chin. Med. Assoc. 2018, 81, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Hsu, T.F.; Huang, B.S.; Tsai, H.W.; Chang, Y.H.; Wang, P.H. Postoperative maintenance levonorgestrel-releasing intrauterine system and endometrioma recurrence: A randomized controlled study. Am. J. Obstet. Gynecol. 2017, 216, e1–e582. [Google Scholar] [CrossRef] [PubMed]
- Daneshmand, S.; Djaladat, H.; Nichols, C. Management of residual mass in nonseminomatous germ cell tumors following chemotherapy. Ther. Adv. Urol. 2011, 3, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Winter, C.; Raman, J.D.; Sheinfeld, J.; Albers, P. Retroperitoneal lymph node dissection after chemotherapy. BJUI 2009, 104, 404–1412. [Google Scholar] [CrossRef]
- Zanagnolo, V.; Minig, L.; Cárdenas-Rebollo, J.M.; Achilarre, M.T.; Garbi, A.; Patrono, M.G.; Colombo, N.; Maggioni, A. Robotic versus open radical hysterectomy in women with locally advanced cervical cancer after neoadjuvant chemotherapy: A Single-institution Experience of Surgical and Oncologic Outcomes. J. Minim. Invasive Gynecol. 2016, 23, 909–916. [Google Scholar] [CrossRef]
- Corrado, G.; Cutillo, G.; Saltari, M.; Mancini, E.; Sindico, S.; Vici, P.; Sergi, D.; Sperduti, I.; Patrizi, L.; Pomati, G.; et al. Surgical and oncological outcome of robotic surgery compared with laparoscopic and abdominal surgery in the management of locally advanced cervical cancer after neoadjuvant chemotherapy. Int. J. Gynecol. Cancer 2016, 26, 539–546. [Google Scholar] [CrossRef] [PubMed]
- van der Velden, J.; Mom, G.H. Tailoring radicality in early cervical cancer: How far can we go? J. Gynecol. Oncol. 2019, 30, e30. [Google Scholar] [CrossRef] [PubMed]
- Rydzewska, L.; Tierney, J.; Vale, C.L.; Symonds, P.R. Neoadjuvant chemotherapy plus surgery versus surgery for cervical cancer. Cochrane Database Syst. Rev. 2012, 12, CD007406. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; He, Y.; Yang, S.L.; Zhao, Q.; Wu, Y.M. Neoadjuvant chemotherapy with radical surgery vs. radical surgery alone for cervical cancer: A systematic review and meta-analysis. Onco. Targets Ther. 2019, 12, 1881–1891. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, K.; Shimada, M.; Yamaguchi, S.; Kigawa, J.; Tokunaga, H.; Tabata, T.; Kodama, J.; Kawana, K.; Mikami, M.; Sugiyama, T. Neoadjuvant chemotherapy with taxane and platinum followed by radical hysterectomy for stage IB2-IIB cervical cancer: Impact of histology type on survival. J. Clin. Med. 2019, 8, 156. [Google Scholar] [CrossRef]
- De la Torre, M. Neoadjuvant chemotherapy in woman with early or locally advanced cervical cancer. Rep. Pract. Oncol. Radiother. 2018, 23, 528–532. [Google Scholar] [CrossRef]
- Zhu, Y.; Yang, J.; Zhang, X.; Chen, D.; Zhang, S. Acquired Treatment Response from Neoadjuvant Chemotherapy Predicts A Favorable Prognosis for Local Advanced Cervical Cancer: A Meta-Analysis. Medicine 2018, 97, e0530. [Google Scholar] [CrossRef]
- Chen, Z.; Shi, Y.; Wang, S.; Lu, Q. Meta-analysis showing that early response to neoadjuvant chemotherapy predicts better survival among cervical cancer patients. Oncotarget 2017, 8, 59609–59617. [Google Scholar] [CrossRef]
- Gong, L.; Lou, J.Y.; Wang, P.; Zhang, J.W.; Liu, H.; Peng, Z.L. Clinical evaluation of neoadjuvant chemotherapy followed by radical surgery in the management of stage IB2-IIB cervical cancer. Int. J. Gynecol. Obstet. 2012, 117, 23–26. [Google Scholar] [CrossRef]
- Wright, J.D.; Matsuo, K.; Huang, Y.; Tergas, A.I.; Hou, J.Y.; Khoury-Collado, F.; St Clair, C.M.; Ananth, C.V.; Neugut, A.I.; Hershman, D.L. Prognostic performance of the 2018 International Federation of Gynecology and Obstetrics Cervical Cancer Staging Guidelines. Obstet. Gynecol. 2019, 134, 49–57. [Google Scholar] [CrossRef]
- Matsuo, K.; Machida, H.; Mandelbaum, R.S.; Konishi, I.; Mikami, M. Validation of the 2018 FIGO cervical cancer staging system. Gynecol. Oncol. 2019, 152, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Vahedpoor, Z.; Behrashi, M.; Khamehchian, T.; Abedzadeh-Kalahroudi, M.; Moravveji, A.; Mohmadi-Kartalayi, M. Comparison of the diagnostic value of the visual inspection with acetic acid (VIA) and Pap smear in cervical cancer screening. Taiwan J. Obstet. Gynecol. 2019, 58, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Sawaya, G.F.; Smith-McCune, K.; Kuppermann, M. Cervical cancer screening: More choices in 2019. JAMA 2019, 321, 2018–2019. [Google Scholar] [CrossRef] [PubMed]
- Chiang, Y.C.; Chen, Y.Y.; Hsieh., S.F.; Chiang, C.J.; You, S.L.; Cheng, W.F.; Lai, M.S.; Chen, C.A. Taiwan Cervical Cancer Prevention Surveillance Center. Screening frequency and histologic type influence the efficacy of cervical cancer screening: A nationwide cohort study. Taiwan J. Obstet. Gynecol. 2017, 56, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Kim, J. The Relationship of Health Beliefs with Information Sources and HPV Vaccine Acceptance among Young Adults in Korea. Int. J. Environ. Res. Public Health 2018, 15, 673. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.K.; Seo, S.S.; Myong, J.P.; Koo, J.W.; Jeong, J. Factors Associated with Cervical Cancer Screening among Married Female Immigrants with Korean Husbands in South Korea. Int. J. Environ. Res. Public Health 2018, 15, 2528. [Google Scholar] [CrossRef]
- Pelullo, C.P.; Esposito, M.R.; Di Giuseppe, G. Human Papillomavirus Infection and Vaccination: Knowledge and Attitudes among Nursing Students in Italy. Int. J. Environ. Res. Public Health 2019, 16, 1770. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.I.; Cho, J.H.; Seol, A.; Kim, Y.I.; Lee, M.; Kim, H.S.; Chung, H.H.; Kim, J.W.; Park, N.H.; Song, Y.S. Comparison of survival outcomes between minimally invasive surgery and conventional open surgery for radical hysterectomy as primary treatment in patients with stage IB1-IIA2 cervical cancer. Gynecol. Oncol. 2019, 153, 3–12. [Google Scholar] [CrossRef]
- Leitao, M.M., Jr. The change in landscape after a new landmark is constructed: Radical hysterectomy for early cervical cancer and Minimally Invasive Surgery. Gynecol. Oncol. 2019, 153, 1–2. [Google Scholar] [CrossRef]
- Wang, P.H.; Yen, M.S.; Yuan, C.C.; Chao, K.C.; Ng, H.T.; Lee, W.L.; Chao, H.T. Port site metastasis after laparoscopic-assisted vaginal hysterectomy for endometrial cancer: Possible mechanisms and prevention. Gynecol. Oncol. 1997, 66, 151–155. [Google Scholar] [CrossRef]
- Wang, P.H.; Yuan, C.C.; Chao, K.C.; Yen, M.S.; Ng, H.T.; Chao, H.T. Squamous cell carcinoma of the cervix after laparoscopic surgery. A case report. J. Reprod. Med. 1997, 42, 801–804. [Google Scholar] [PubMed]
- Wang, P.H.; Yuan, C.C.; Lin, G.; Ng, H.T.; Chao, H.T. Risk factors contributing to early occurrence of port site metastases of laparoscopic surgery for malignancy. Gynecol. Oncol. 1999, 72, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.H.; Yeh, J.Y.; Sheu, B.C. Vaginal hysterectomy for endometrial cancer. Am. J. Obstet. Gynecol. 2009, 201, e12. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.H.; Horng, H.C.; Chen, C.P. Is it safe to use minimally invasive surgery in the management of endometrial cancer? Taiwan J. Obstet. Gynecol. 2016, 55, 155–156. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, M.; Kong, T.W.; Kim, S.; Kim, S.C.; Kim, Y.B.; Kim, J.W.; Park, J.Y.; Suh, D.H.; Shim, S.H.; Lee, K.H.; et al. Minimally invasive surgery for radical hysterectomy in women with cervical cancer: Korean Society of Gynecologic Oncology, Korean Society of Obstetrics and Gynecology, and Korean Society of Gynecologic Endoscopy and Minimally Invasive Surgery position statement. J. Gynecol. Oncol. 2019, 30, e104. [Google Scholar] [PubMed]
- Nezhat, F.R.; Ananth, C.V.; Vintzileos, A.M. The two Achilles heels of surgical randomized controlled trials: Differences in surgical skills and reporting of average performance. Am. J. Obstet. Gynecol. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Ratiu, D.; Luncescu, C.; Morgenstern, B.; Eichler, C.; Grüttner, B.; Ludwig, S.; Mallmann, P.; Thangarajah, F. Comparison of minimally invasive surgery and abdominal surgery among patients with cervical cancer. Anticancer Res. 2019, 39, 2661–2664. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, L.; Wu, M.; Ma, S.; Tan, X.; Zhong, S.; Lang, J. The impact of the surgical routes and learning curve of radical hysterectomy on the survival outcomes in stage IB cervical cancer: A retrospective cohort study. Int. J. Surg. 2019, 68, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Yen, M.S.; Ng, H.T.; Wang, P.H. A delicate surgical method for cancer treatment is welcome. Taiwan J. Obstet. Gynecol. 2012, 51, 329–330. [Google Scholar] [CrossRef]
- Wen, K.C.; Horng, H.C.; Wang, P.H. Hemorrhage: A strong indicator for myomectomy-related complication. J. Chin. Med. Assoc. 2016, 79, 413–414. [Google Scholar] [CrossRef] [PubMed]
- Cheng-Yen Lai, J.; Lai, K.J.; Yi-Yung Yu, E.; Hung, S.T.; Chu, C.Y.; Wang, K.L. Sentinel lymphatic mapping among women with early-stage cervical cancer: A systematic review. Taiwan J. Obstet. Gynecol. 2018, 57, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Shimada, C.; Todo, Y.; Yamazaki, H.; Takeshita, S.; Okamoto, K.; Minobe, S.; Yamashiro, K.; Kato, H. A feasibility study of sentinel lymph node mapping by cervical injection of a tracer in Japanese women with early stage endometrial cancer. Taiwan J. Obstet. Gynecol. 2018, 57, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.T.; Horng, H.C.; Wang, P.H. The role of complete staging surgery for pure endometrioid-type endometrial cancer. J. Chin. Med. Assoc. 2018, 81, 663–664. [Google Scholar] [CrossRef]
- Restivo, V.; Costantino, C.; Marras, A.; Napoli, G.; Scelfo, S.; Scuderi, T.; Casuccio, A.; Cernigliaro, A.; Giusti, A.; Spila Alegiani, S. Pap Testing in a High-Income Country with Suboptimal Compliance Levels: A Survey on Acceptance Factors among Sicilian Women. Int. J. Environ. Res. Public Health 2018, 15, 1804. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Jungsuwadee, P.; Abraham, O.; Ko, D. Shared Decision-Making and Women’s Adherence to Breast and Cervical Cancer Screenings. Int. J. Environ. Res. Public Health 2018, 15, 1509. [Google Scholar] [CrossRef] [PubMed]
- Pikala, M.; Burzyńska, M.; Maniecka-Bryła, I. Years of Life Lost due to Cervical Cancer in Poland in 2000 to 2015. Int. J. Environ. Res. Public Health 2019, 16, 1545. [Google Scholar] [CrossRef] [PubMed]
- Nutthachote, P.; Oranratanaphan, S.; Termrungruanglert, W.; Triratanachat, S.; Chaiwongkot, A.; Baedyananda, F.; Bhattarakosol, P. Comparison of detection rate of high risk HPV infection between self-collected HPV testing and clinician-collected HPV testing in cervical cancer screening. Taiwan J. Obstet. Gynecol. 2019, 58, 477–481. [Google Scholar] [CrossRef] [PubMed]
- da Costa, S.C.S.; Bonadio, R.C.; Gabrielli, F.C.G.; Aranha, A.S.; Dias Genta, M.L.N.; Miranda, V.C.; de Freitas, D.; Abdo Filho, E.; Ferreira, P.A.O.; Machado, K.K.; et al. Neoadjuvant chemotherapy with cisplatin and gemcitabine followed by chemoradiation versus chemoradiation for locally advanced cervical cancer: A randomized phase II trial. J. Clin. Oncol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Chuang, C.S.; Yang, K.W.; Yen, C.M.; Lin, C.L.; Kao, C.H. Risk of Seizures in patients with organophosphate poisoning: A nationwide population-based study. Int. J. Environ. Res. Public Health 2019, 16, 3147. [Google Scholar] [CrossRef]
- Chen, C.K.; Chang, H.T.; Chen, Y.C.; Chiang, S.C.; Chou, H.P.; Chen, T.J. Utilization and Safety of Percutaneous Lung Biopsy: A 10-Year Nationwide Population-Based Study. Int. J. Environ. Res. Public Health 2019, 16, 1316. [Google Scholar] [CrossRef]
- Tong, C.; Wang, Y.H.; Chang, Y.C. Increased Risk of Carotid Atherosclerosis in Male Patients with Chronic Periodontitis: A Nationwide Population-Based Retrospective Cohort Study. Int. J. Environ. Res. Public Health 2019, 16, 2635. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.Y.; Chung, H.J.; Huang, E.Y.; Lin, T.P.; Lin, A.T.L. The perioperative outcomes between renal hilar and non-hilar tumors following robotic-assisted partial nephrectomy (RAPN). J. Chin. Med. Assoc. 2018, 81, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Lin, H.H.; Hsiao, S.M. Comparison of robotic assisted laparoscopic myomectomy with barbed sutures and traditional laparoscopic myomectomy with barbed sutures. Taiwan J. Obstet. Gynecol. 2018, 57, 709–712. [Google Scholar] [CrossRef]
- Lee, C.Y.; Chen, I.H.; Torng, P.L. Robotic myomectomy for large uterine myomas. Taiwan J. Obstet. Gynecol. 2018, 57, 796–800. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).