The Relationship between Body Composition and Physical Fitness and the Effect of Exercise According to the Level of Childhood Obesity Using the MGPA Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Study 1. Analysis of the Relationship between Body Composition and Physical Fitness in Obese Children (Correlation, Stepwise Multiple Regression Analysis)
2.1.1. Design, Participants
2.1.2. Measures
Flexibility: Sit-and-Reach (Centimeters; cm)
Muscular Strength: Grip Strength (Kilograms; kg)
Muscular Endurance: Sit-Ups (Times)
Agility: Long Jump in Place (Centimeters; cm)
Balance: Standing on One Leg with Eyes Closed (Seconds; s)
2.1.3. Statistical Analyses
2.2. Study 2. Effect on Body Composition and Physical Fitness by the Degree of Obesity in Children after a 16-Week Exercise Intervention
2.2.1. Design, Participants
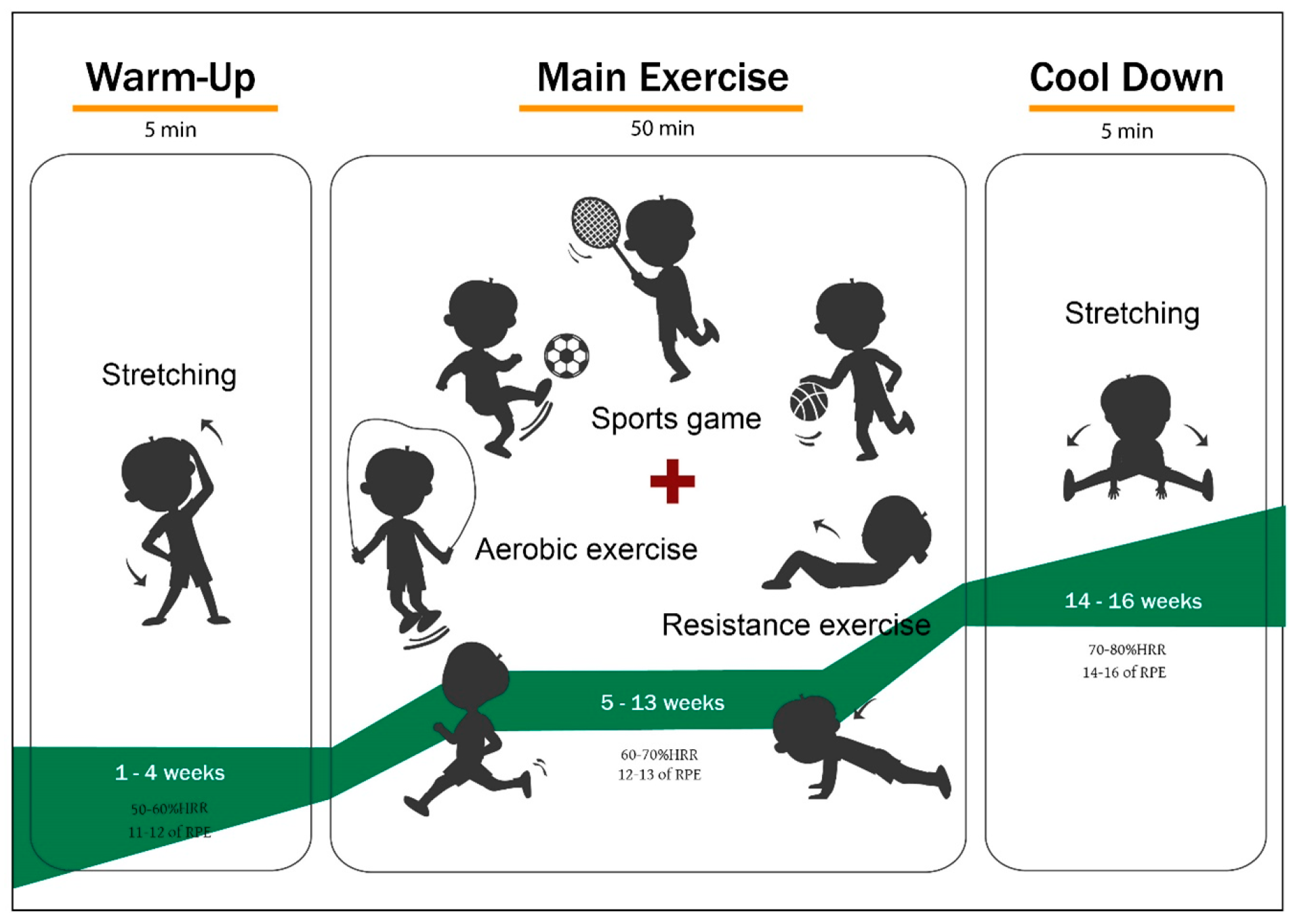
2.2.2. Exercise Program
2.2.3. Statistical Analyses
2.3. Study 3. Parameters on Changes in Body Composition and Physical Fitness after Exercise Program (MGPA)
2.3.1. Design
2.3.2. Statistical Analyses
3. Results
3.1. Study 1
3.1.1. Differences in Body Composition According to the Degree of Obesity
3.1.2. Differences in Physical Fitness According to the Degree of Obesity
3.1.3. Correlation between Body Composition and Physical Fitness by Obesity Degree
3.2. Study 2
3.2.1. Effect of the Exercise Program on Body Composition in Each Group
3.2.2. Effects on Physical Fitness of Each Group after the 16-Week Exercise Program
3.3. Study 3
3.3.1. MGPA: NO Group
3.3.2. MGPA: OV Group
3.3.3. MGPA: OB Group
3.3.4. MGPA: SOB Group
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Simmonds, M.; Llewellyn, A.; Owen, C.G.; Woolacott, N. Predicting adult obesity from childhood obesity: A systematic review and meta-analysis. Obes. Rev. 2016, 17, 95–107. [Google Scholar] [CrossRef]
- Must, A.; Phillips, S.M.; Naumova, E.N. Occurrence and timing of childhood overweight and mortality: Findings from the third harvard growth study. J. Pediatr. 2012, 160, 743–750. [Google Scholar] [CrossRef]
- Serdula, M.K.; Ivery, D.; Coates, R.J.S.; Freedman, D.S.; Williamson, D.F.; Byers, T. Do Obese Children Become Obese Adults? A Review of the Literature. Prev. Med. 1993, 22, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Tyson, N.; Frank, M. Childhood and adolescent obesity definitions as related to BMI, evaluation and management options. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 48, 158–164. [Google Scholar] [CrossRef]
- Kim, J.H.; Yun, S.; Hwang, S.S.; Shim, J.O.; Chae, H.W.; Lee, Y.J.; Lee, J.H.; Kim, S.C.; Lim, D.; Yang, S.W.; et al. The 2017 Korean national growth charts for children and adolescents: Development, improve ment, and prospects. Korean J. Pediatr. 2018, 61, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Solomon, C.G.; Manson, J.E. Obesity and mortality: A review of the epidemiologic data. Am. J. Clin. Nutr. 1997, 66, 1044S–1050S. [Google Scholar] [CrossRef]
- Ortega, F.B.; Lavie, C.J.; Blair, S.N. Obesity and cardiovascular disease. Circ. Res. 2016, 118, 1752–1770. [Google Scholar] [CrossRef]
- Kumar, S.; Kelly, A.S. Review of Childhood Obesity: From Epidemiology, Etiology, and Comorbidities to Clinical Assessment and Treatment. Mayo Clin. Proc. 2017, 92, 251–265. [Google Scholar] [CrossRef]
- Corder, K.; Sharp, S.J.; Atkin, A.J.; Andersen, L.B.; Cardon, G.; Page, A.; Davey, R.; Grøntved, A.; Hallal, P.C.; Janz, K.F.; et al. Age-related patterns of vigorous-intensity physical activity in youth: The International Children’s Accelerometry Database. Prev. Med. Rep. 2016, 4, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Puccinelli, P.J.; da Costa, T.S.; Seffrin, A.; de Lira, C.A.B.; Vancini, R.L.; Nikolaidis, P.T.; Knechtle, B.; Rosemann, T.; Hill, L.; Andrade, M.S. Reduced level of physical activity during COVID-19 pandemic is associated with depression and anxiety levels: An internet- based survey. BMC Public Health 2021, 21, 1–11. [Google Scholar] [CrossRef]
- Pescatello, L.; Riebe, D.; Thompson, P. ACSM’s Guidelines for Exercise Testing and Prescription; Williams, L., Wilkins, Eds.; Wolters Kluwer Health: Philadelphia, PA, USA, 2014. [Google Scholar]
- Landry, B.W.; Driscoll, S.W. Physical activity in children and adolescents. PM&R 2012, 4, 826–832. [Google Scholar] [CrossRef]
- Larsen, M.N.; Nielsen, C.M.; Ørntoft, C.Ø.; Randers, M.B.; Manniche, V.; Hansen, L.; Hansen, P.R.; Bangsbo, J.; Krustrup, P. Physical fitness and Body composition in 8–10 year old danish children are associated with sports club participation. J. Strength Cond. Res. 2017, 31, 3425–3434. [Google Scholar] [CrossRef] [PubMed]
- Alberga, A.S.; Prud’homme, D.; Sigal, R.J.; Goldfield, G.S.; Hadjiyannakis, S.; Phillips, P.; Malcolm, J.; Ma, J.; Doucette, S.; Gougeon, R.; et al. Effects of aerobic training, resistance training, or both on cardiorespiratory and musculoskeletal fitness in adolescents with obesity: The HEARTY trial. Appl. Physiol. Nutr. Metab. 2016, 41, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Delecluse, C.; Colman, V.; Roelants, M.; Verschueren, S.; Derave, W.; Ceux, T.; Eijnde, B.O.; Seghers, J.; Pardaens, K.; Brumagne, S.; et al. Exercise programs for older men: Mode and intensity to induce the highest possible health-related benefits. Prev. Med. 2004, 39, 823–833. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Muñoz, M.; Adsuar, J.C.; Pérez-Gómez, J.; Muñoz-Bermejo, L.; Garcia-Gordillo, M.Á.; Carlos-Vivas, J. Influence of Body Composition on Physical Fitness in Adolescents. Medicina 2020, 56, 328. [Google Scholar] [CrossRef]
- Garson, G. Path Analysis; Statistical Associates Publishing: Asheboro, NC, USA, 2013. [Google Scholar]
- Topa, G.; Leon, M. Theory of planned behavior and smoking: Meta-analysis and SEM model. Subst. Abuse Rehabil. 2010, 3, 23–33. [Google Scholar] [CrossRef]
- Hong, S.H. The Criteria for Selecting Appropriate Fit Indices in Structural Equation Modeling and Their Rationales. Korean J. Clin. Psychol. 2000, 19, 161–177. [Google Scholar]
- Hong, S.; Malik, M.L.; Lee, M.K. Testing configural, metric, scalar, and latent mean invariance across genders in sociotropy and autonomy using a non-western sample. Educ. Psychol. Meas. 2003, 63, 636–654. [Google Scholar] [CrossRef]
- Wronka, I. Association between BMI and height in girls aged 7–18 years—A longitudinal study. Anthropol. Anz. 2013, 70, 319–329. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Karlberg, J. BMI in childhood and its association with height gain, timing of puberty, and final height. Pediatr. Res. 2001, 49, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Katzmarzyk, P.T.; Barreira, T.V.; Broyles, S.T.; Chaput, J.-P.; Fogelholm, M.; Hu, G.; Kuriyan, R.; Kurpad, A.; Lambert, E.V.; Maher, C.; et al. Association between body mass index and body fat in 9–11-year-old children from countries spanning a range of human development. Int. J. Obes. Suppl. 2015, 5, S43–S46. [Google Scholar] [CrossRef][Green Version]
- Kostopoulou, E.; Avgeri, A.; Skiadopoulos, S.; Dimitriou, G.; Giannakopoulos, I. The association between excess weight and body composition measurements in a pediatric population—A cross sectional study. Res. Sq. 2021, 1, 1–19. [Google Scholar] [CrossRef]
- de Moraes, A.M.; Gonçalves, E.M.; de Oliveira Barbeta, V.J.; Guerra-Júnior, G. Cross-sectional study of the association of body composition and physical fitness with bone status in children and adolescents from 11 to 16 years old. BMC Pediatr. 2013, 13, 1–7. [Google Scholar] [CrossRef]
- Chen, W.; Lin, C.C.; Peng, C.T.; Li, C.I.; Wu, H.C.; Chiang, J.; Wu, J.Y.; Huang, P.C. Approaching healthy body mass index norms for children and adolescents from health-related physical fitness. Obes. Rev. 2002, 3, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.C.; Malina, R.M. Body mass index and individual physical fitness tests in Taiwanese youth aged 9–18 years. Int. J. Pediatr. Obes. 2010, 5, 404–411. [Google Scholar] [CrossRef]
- Jürimäe, T.; Hurbo, T.; Jürimäe, J. Relationship of handgrip strength with anthropometric and body composition variables in prepubertal children. HOMO-J. Comp. Hum. Biol. 2009, 60, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Artero, E.G.; España-Romero, V.; Ortega, F.B.; Jiménez-Pavón, D.; Ruiz, J.R.; Vicente-Rodríguez, G.; Bueno, M.; Marcos, A.; Gómez-Martínez, S.; Urzanqui, A.; et al. Health-related fitness in adolescents: Underweight, and not only overweight, as an influencing factor. The AVENA study. Scand. J. Med. Sci. Sports 2010, 20, 418–427. [Google Scholar] [CrossRef]
- Wearing, S.C.; Hennig, E.M.; Byrne, N.M.; Steele, J.R.; Hills, A.P. The impact of childhood obesity on musculoskeletal form. Eur. Heart J. 2006, 7, 209–218. [Google Scholar] [CrossRef]
- Bim, M.A.; de Araújo Pinto, A.; Scarabelot, K.S.; Claumann, G.S.; Pelegrini, A. Handgrip strength and associated factors among Brazilian adolescents: A cross-sectional study. J. Bodyw. Mov. Ther. 2021, 28, 75–81. [Google Scholar] [CrossRef]
- Al-Asadi, J.N. Handgrip strength in medical students: Correlation with body mass index and hand dimensions. Asian J. Med. Sci. 2018, 9, 21–26. [Google Scholar] [CrossRef]
- Liao, Y.; Chang, S.H.; Miyashita, M.; Stensel, D.; Chen, J.F.; Wen, L.T.; Nakamura, Y. Associations between health-related physical fitness and obesity in Taiwanese youth. J. Sports Sci. 2013, 31, 1797–1804. [Google Scholar] [CrossRef]
- Kowal, M.; Sorokowski, P.; Żelaźniewicz, A.; Nowak, J.; Orzechowski, S.; Żurek, A.; Żurek, G. A positive relationship between body height and the testosterone response to physical exercise. Evol. Hum. Behav. 2021, 42, 179–185. [Google Scholar] [CrossRef]
- Martha, P.M.; Rogol, A.D.; Veldhuis, J.D.; Kerrigan, J.R.; Goodman, D.W.; Blizzard, R.M. Alterations in the pulsatile properties of circulating growth hormone concentrations during puberty in boys. J. Clin. Endocrinol. Metab. 1989, 69, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Slentz, C.A.; Duscha, B.D.; Johnson, J.L.; Ketchum, K.; Aiken, L.B.; Samsa, G.P.; Houmard, J.A.; Bales, C.W.; Kraus, W.E. Effects of the amount of exercise on body weight, body composition, and measures of central obesity: STRRIDE—A randomized controlled study. Arch. Intern. Med. 2004, 164, 31–39. [Google Scholar] [CrossRef]
- Halme, T.; Parkkisenniemi, S.; Kujala, U.M.; Nupponen Likes, H. Relationships between standing broad jump, shuttle run and body mass index in children aged three to eight years. J. Sports Med. Phys. Fit. 2009, 49, 395–400. [Google Scholar]
- Chodzko-Zajko, W.J.; Proctor, D.N.; Fiatarone Singh, M.A.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. Exercise and Physical Activity for Older Adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef]
- Hasan, N.A.K.A.K.; Kamal, H.M.; Hussein, Z.A. Relation between body mass index percentile and muscle strength and endurance. Egypt. J. Med. Hum. Genet. 2016, 17, 367–372. [Google Scholar] [CrossRef]
- Truter, L.; Pienaar, A.; Du Toit, D. Relationships between overweight, obesity and physical fitness of nine- to twelve-year-old South African children. S. Afr. Fam. Pract. 2010, 52, 227–233. [Google Scholar] [CrossRef]



| Group | NO (n = 33) | OW (n = 34) | OB (n = 54) | SOB (n = 43) | F | Post-hoc | |
|---|---|---|---|---|---|---|---|
| Variable | |||||||
| BMI (kg/m2) | 18.50 ± 1.98 | 21.94 ± 0.55 | 24.14 ± 0.86 | 27.20 ± 2.31 | 202.664 *** | NO < OW < OB < SOB (Games-Howell) | |
| Height (cm) | 135.52 ± 9.92 | 138.68 ± 10.22 | 140.35 ± 10.09 | 144.77 ± 8.94 | 5.897 *** | NO < SOB (Scheffe) | |
| Weight (kg) | 34.09 ± 5.60 | 42.45 ± 6.52 | 47.86 ± 7.36 | 57.14 ± 7.66 | 73.071 *** | NO < OW < OB < SOB (Scheffe) | |
| %BF (%) | 17.94 ± 4.75 | 21.27 ± 4.29 | 26.53 ± 5.90 | 31.97 ± 7.17 | 43.318 *** | NO, OW < OB < SOB (Games-Howell) | |
| SMM (kg) | 13.26 ± 2.83 | 17.02 ± 3.31 | 16.81 ± 4.22 | 17.87 ± 5.06 | 8.869 *** | NO < OW, OB, SOB (Games-Howell) | |
| Group | NO (n = 33) | OW (n = 34) | OB (n = 54) | SOB (n = 43) | F | Post-hoc | |
|---|---|---|---|---|---|---|---|
| Variable | |||||||
| Muscular strength (kg) | 13.95 ± 3.54 | 16.01 ± 4.10 | 16.09 ± 4.40 | 17.02 ± 4.86 | 3.256 * | NO < SOB (Scheffe) | |
| Flexibility (cm) | 7.26 ± 6.00 | 6.86 ± 4.17 | 6.87 ± 7.71 | 7.04 ± 4.69 | 0.035 | ||
| Muscular endurance (times) | 20.45 ± 10.23 | 18.32 ± 10.17 | 17.39 ± 8.36 | 13.98 ± 8.96 | 3.235 * | SOB < NO (Scheffe) | |
| Power (cm) | 123.65 ± 26.76 | 128.18 ± 16.83 | 119.43 ± 26.16 | 110.36 ± 30.45 | 3.331 * | SOB < OW (Games-Howell) | |
| Balance (s) | 38.96 ± 49.73 | 24.32 ± 18.05 | 20.33 ± 18.27 | 21.05 ± 19.88 | 3.560 * | OB, SOB < NO (Games-Howell) | |
| Variable | Muscular Strength | Flexibility | Muscular Endurance | Power | Balance |
|---|---|---|---|---|---|
| NO | |||||
| BMI | 0.005 | −0.011 | −0.292 | −0.273 | −0.037 |
| Height | 0.522 ** | −0.235 | 0.162 | −0.042 | 0.232 |
| Weight | 0.467 ** | 0.458 ** | −0.045 | −0.225 | 0.212 |
| %BF | 0.073 | 0.038 | −0.224 | −0.577 *** | 0.252 |
| SMM | 0.097 | −0.177 | −0.070 | 0.182 | −0.337 |
| OW | |||||
| BMI | 0.294 | −0.203 | 0.001 | 0.196 | 0.063 |
| Height | 0.748 *** | 0.104 | 0.119 | 0.331 | 0.188 |
| Weight | 0.765 *** | 0.084 | 0.106 | 0.323 | 0.173 |
| %BF | 0.078 | 0.274 | −0.144 | −0.223 | −0.141 |
| SMM | 0.670 *** | −0.005 | 0.101 | 0.336 | 0.209 |
| OB | |||||
| BMI | 0.227 | −0.268 | −0.063 | −0.027 | −0.017 |
| Height | 0.688 *** | −0.067 | 0.251 | 0.285 * | 0.376 ** |
| Weight | 0.689 *** | −0.127 | 0.227 | 0.258 | 0.350 ** |
| %BF | −0.022 | 0.046 | −0.004 | −0.640 *** | 0.113 |
| SMM | 0.273 * | −0.222 | 0.052 | 0.635 *** | −0.052 |
| SOB | |||||
| BMI | −0.134 | 0.019 | −0.104 | −0.255 | −0.064 |
| Height | 0.704 *** | −0.307 * | 0.279 | 0.239 | 0.055 |
| Weight | 0.561 *** | −0.250 | 0.202 | 0.064 | 0.014 |
| %BF | 0.103 | −0.057 | −0.197 | −0.604 *** | 0.193 |
| SMM | −0.056 | 0.003 | 0.243 | 0.635 *** | −0.288 |
| Group | Variables | Muscular Strength | Flexibility | Muscular Endurance | Power | Balance | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| b | β | t | b | β | t | b | β | t | b | β | t | b | β | t | ||
| NO | Pre_F | 0.956 | 0.935 | 8.289 *** | 0.921 | 0.861 | 11.328 *** | 1.106 | 0.971 | 11.117 *** | 0.795 | 0.942 | 11.941 *** | 0.834 | 0.887 | 8.012 *** |
| Diff_ Height | −0.120 | −0.047 | −0.687 | 0.339 | 0.086 | 1.139 | 0.789 | 0.130 | 1.322 | −0.113 | −0.013 | −0.160 | −1.232 | −0.061 | −0.935 | |
| Diff_ Weight | 1.143 | 0.406 | 4.025 *** | −0.447 | −0.102 | −1.062 | 1.065 | 0.158 | 1.309 | 0.265 | 0.027 | 0.267 | 5.074 | 0.227 | 2.719 ** | |
| Diff_ %BF | −0.341 | −0.081 | −0.973 | 0.359 | 0.055 | 0.769 | 2.862 | 0.284 | 3.130 ** | 1.241 | 0.085 | 0.959 | 11.134 | 0.333 | 3.981 *** | |
| Diff_ SMM | −1.611 | −0.186 | −1.219 | 2.863 | 0.213 | 2.084 * | 2.928 | 0.141 | 1.190 | 4.756 | 0.159 | 1.627 | 13.681 | 0.198 | 2.819 ** | |
| Sex | −0.531 | −0.059 | −0.896 | −0.382 | −0.027 | −0.443 | 1.815 | 0.085 | 1.099 | 1.710 | 0.055 | 0.856 | −6.052 | −0.085 | −1.738 | |
| OW | Pre_F | 0.772 | 0.614 | 10.082 *** | 1.151 | 1.271 | 2.979 ** | 0.675 | 0.665 | 3.309 *** | 0.829 | 0.921 | 16.121 *** | 0.794 | 0.836 | 5.429 *** |
| Diff_ Height | 0.772 | 0.291 | 4.150 *** | 1.286 | 0.497 | 1.972 * | 1.334 | 0.220 | 0.683 | 2.326 | 0.215 | 2.885 ** | 1.100 | 0.111 | 0.449 | |
| Diff_ Weight | −0.290 | −0.058 | −1.199 | 1.143 | 0.233 | 0.798 | 0.684 | 0.060 | 0.228 | 3.567 | 0.175 | 3.040 ** | 0.945 | 0.050 | 0.247 | |
| Diff_ %BF | −0.446 | −0.170 | −3.316 *** | 1.385 | 0.541 | 1.689 | −1.547 | −0.258 | −1.039 | 1.373 | 0.128 | 2.217 * | −2.678 | −0.273 | −1.458 | |
| Diff_ SMM | 1.629 | 0.119 | 2.475* | −2.386 | −0.178 | −0.857 | −4.852 | −0.155 | −0.555 | 9.589 | 0.171 | 2.729 ** | −2.928 | −0.057 | −0.293 | |
| Sex | 0.769 | 0.084 | 2.294 * | 4.627 | 0.517 | 1.428 | 2.042 | 0.098 | 0.531 | −0.377 | −0.010 | −0.214 | −5.257 | −0.153 | −1.087 | |
| OB | Pre_F | 1.051 | 0.839 | 12.173 *** | 0.886 | 0.893 | 14.562 *** | 0.924 | 0.756 | 9.213 *** | 0.669 | 0.852 | 6.767 *** | 0.902 | 0.713 | 5.362 *** |
| Diff_ Height | 0.391 | 0.096 | 1.200 | 0.963 | 0.150 | 2.141 * | 1.445 | 0.216 | 2.093 * | 1.687 | 0.113 | 0.819 | 2.507 | 0.127 | 0.872 | |
| Diff_ Weight | −0.230 | −0.084 | −0.809 | −0.049 | −0.011 | −0.125 | 1.515 | 0.338 | 2.510 * | 0.005 | 0.000 | 0.003 | −3.563 | −0.270 | −1.334 | |
| Diff_ %BF | 0.521 | 0.376 | 1.361 | −0.449 | −0.205 | −0.845 | −2.075 | −0.909 | −2.514 * | −0.063 | −0.012 | −0.026 | 8.527 | 1.269 | 2.186 * | |
| Diff_ SMM | 0.909 | 0.487 | 1.683 | −0.382 | −0.129 | −0.506 | −2.692 | −0.875 | −2.299 * | 3.058 | 0.447 | 0.892 | 12.923 | 1.426 | 2.396 * | |
| Sex | 0.783 | 0.076 | 1.234 | −0.361 | −0.022 | −0.373 | −1.711 | −0.101 | −1.266 | 0.058 | 0.002 | 0.014 | −6.916 | −0.139 | −1.228 | |
| SOB | Pre_F | 0.826 | 0.691 | 8.352 *** | 0.824 | 0.794 | 7.035 *** | 0.622 | 0.548 | 3.821 *** | 0.460 | 0.637 | 5.900 *** | 0.405 | 0.395 | 3.151 ** |
| Diff_ Height | 0.787 | 0.278 | 3.471 *** | 0.123 | 0.059 | 0.334 | 2.759 | 0.594 | 3.414 *** | −1.168 | −0.114 | −0.807 | 2.735 | 0.273 | 1.610 | |
| Diff_ Weight | −0.067 | −0.035 | −0.430 | −0.023 | −0.016 | −0.093 | −0.684 | −0.218 | −1.264 | −1.292 | −0.187 | −1.312 | −0.180 | −0.027 | −0.157 | |
| Diff_ %BF | −0.051 | −0.052 | −0.299 | 0.006 | 0.009 | 0.023 | 0.622 | 0.388 | 0.947 | −0.326 | −0.092 | −0.297 | 0.872 | 0.252 | 0.701 | |
| Diff_ SMM | 0.092 | 0.077 | 0.493 | 0.124 | 0.141 | 0.425 | 0.436 | 0.223 | 0.628 | 3.050 | 0.708 | 2.544 * | 2.360 | 0.560 | 1.798 | |
| Sex | 1.505 | 0.115 | 1.710 | −1.430 | −0.149 | −1.406 | 5.884 | 0.275 | 2.139 * | 16.446 | 0.349 | 3.830 *** | 3.484 | 0.076 | 0.764 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, C.K.; Sim, Y.K.; Lee, J.-H.; Yook, J.S.; Ha, S.-M.; Seo, E.C.; So, W.-Y.; Kim, H.R.; Jeong, W.-M.; Goo, B.O.; et al. The Relationship between Body Composition and Physical Fitness and the Effect of Exercise According to the Level of Childhood Obesity Using the MGPA Model. Int. J. Environ. Res. Public Health 2022, 19, 487. https://doi.org/10.3390/ijerph19010487
Lee CK, Sim YK, Lee J-H, Yook JS, Ha S-M, Seo EC, So W-Y, Kim HR, Jeong W-M, Goo BO, et al. The Relationship between Body Composition and Physical Fitness and the Effect of Exercise According to the Level of Childhood Obesity Using the MGPA Model. International Journal of Environmental Research and Public Health. 2022; 19(1):487. https://doi.org/10.3390/ijerph19010487
Chicago/Turabian StyleLee, Chae Kwan, Young Kyun Sim, Jae-Hoon Lee, Jang Soo Yook, Soo-Min Ha, Eun Chul Seo, Wi-Young So, Hyun Ryun Kim, Woo-Min Jeong, Bong Oh Goo, and et al. 2022. "The Relationship between Body Composition and Physical Fitness and the Effect of Exercise According to the Level of Childhood Obesity Using the MGPA Model" International Journal of Environmental Research and Public Health 19, no. 1: 487. https://doi.org/10.3390/ijerph19010487
APA StyleLee, C. K., Sim, Y. K., Lee, J.-H., Yook, J. S., Ha, S.-M., Seo, E. C., So, W.-Y., Kim, H. R., Jeong, W.-M., Goo, B. O., Chung, J.-W., & Ha, M.-S. (2022). The Relationship between Body Composition and Physical Fitness and the Effect of Exercise According to the Level of Childhood Obesity Using the MGPA Model. International Journal of Environmental Research and Public Health, 19(1), 487. https://doi.org/10.3390/ijerph19010487






