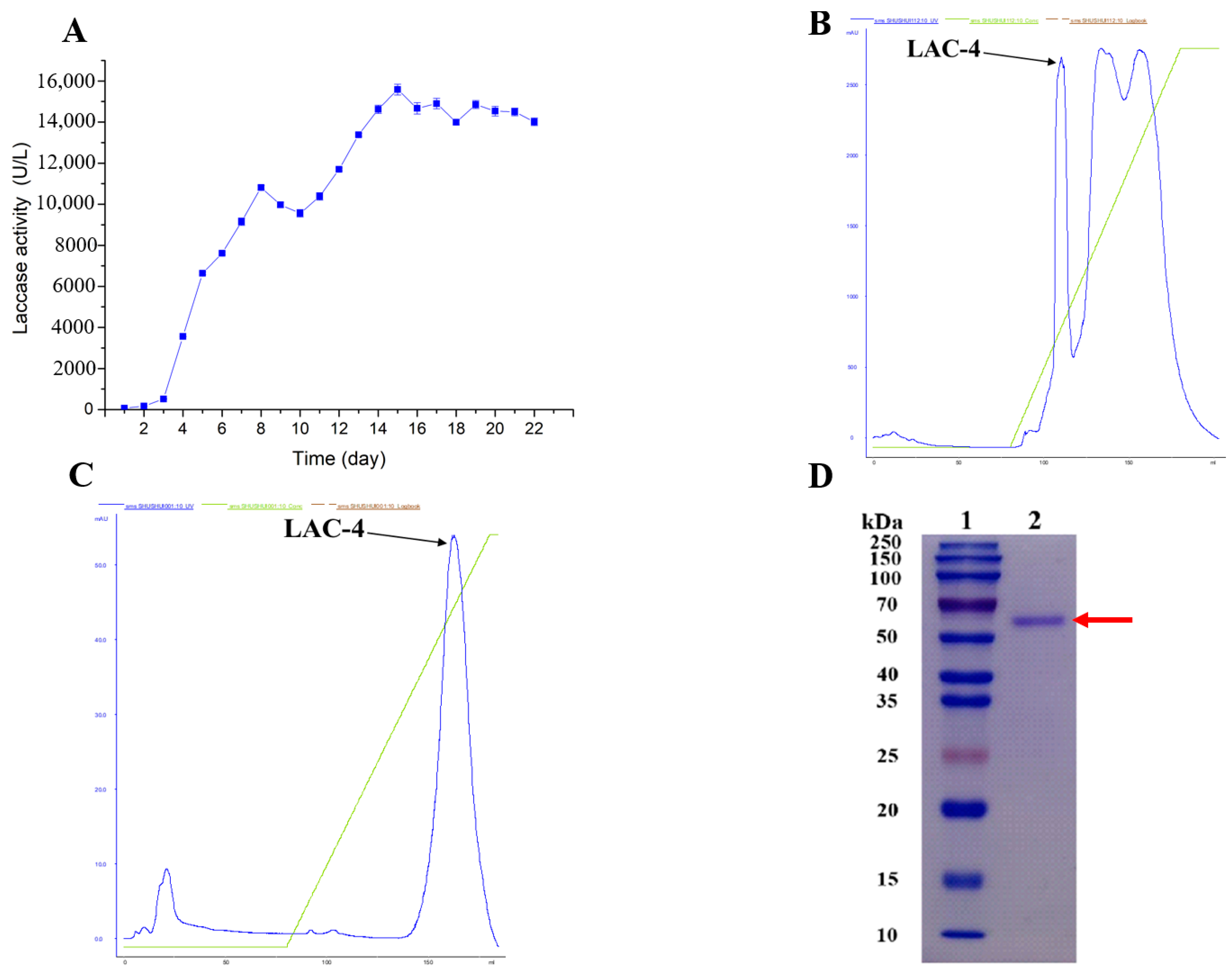
Figure 1.
Purification of LAC-4 laccase from white-rot fungus Ganoderma lucidum. (A) Change curve of laccase production by Ganoderma lucidum in GYP medium. CuSO4 was added on the third day of incubation to induce laccase expression. (B) The chromatographic profile of LAC-4 (anion chromatography). (C) The chromatographic profile of LAC-4 (hydrophobic interaction chromatography). (D) Detection of purified LAC-4 laccase by SDS-PAGE electrophoresis. Lane 1: protein molecular mass marker; Lane 2: purified LAC-4 laccase.
Figure 1.
Purification of LAC-4 laccase from white-rot fungus Ganoderma lucidum. (A) Change curve of laccase production by Ganoderma lucidum in GYP medium. CuSO4 was added on the third day of incubation to induce laccase expression. (B) The chromatographic profile of LAC-4 (anion chromatography). (C) The chromatographic profile of LAC-4 (hydrophobic interaction chromatography). (D) Detection of purified LAC-4 laccase by SDS-PAGE electrophoresis. Lane 1: protein molecular mass marker; Lane 2: purified LAC-4 laccase.
Figure 2.
Enzymatic properties of purified LAC-4 laccase. (A) Effects of different temperatures on LAC-4 activity. (B) Effects of different temperatures on LAC-4 stability. (C) Effects of different pH on LAC-4 activity. (D) LAC-4 stability at different pH values. (E) Effects of different inhibitors (SDS, EDTA-2Na, DTT, NaN3, and mercaptoethanol) on LAC-4 laccase activity.
Figure 2.
Enzymatic properties of purified LAC-4 laccase. (A) Effects of different temperatures on LAC-4 activity. (B) Effects of different temperatures on LAC-4 stability. (C) Effects of different pH on LAC-4 activity. (D) LAC-4 stability at different pH values. (E) Effects of different inhibitors (SDS, EDTA-2Na, DTT, NaN3, and mercaptoethanol) on LAC-4 laccase activity.
Figure 3.
Effects of different metal ions and organic solvents on the LAC-4 laccase activity. (A) CuSO4, (B) MnSO4, (C) ZnSO4, (D) MgSO4, (E) Na2SO4, (F) Comparison of the effects of the metal salt with chloride as the anion and the sulfate ions as the anion on LAC-4 laccase activity (the concentration of metal salt was 100 mM), (G) Comparison of the effects of the metal salt with chloride as the anion and the sulfate ions as the anion on LAC-4 laccase activity (the concentration of metal salt was 200 mM), (H) Concentration of organic solvent was 10% (v/v), (I) Concentration of organic solvent was 20% (v/v), (J) Concentration of organic solvent was 50% (v/v). (H–J): 1: Control, no addition of any organic solvent; 2: Ethylene glycol; 3: Butanediol; 4: Glycerol; 5: Propylene glycol; 6: Ethanol; 7: Methanol; 8: Isopropyl alcohol; 9: Acetonitrile; 10: Acetone; 11: DMSO; 12: DMF.
Figure 3.
Effects of different metal ions and organic solvents on the LAC-4 laccase activity. (A) CuSO4, (B) MnSO4, (C) ZnSO4, (D) MgSO4, (E) Na2SO4, (F) Comparison of the effects of the metal salt with chloride as the anion and the sulfate ions as the anion on LAC-4 laccase activity (the concentration of metal salt was 100 mM), (G) Comparison of the effects of the metal salt with chloride as the anion and the sulfate ions as the anion on LAC-4 laccase activity (the concentration of metal salt was 200 mM), (H) Concentration of organic solvent was 10% (v/v), (I) Concentration of organic solvent was 20% (v/v), (J) Concentration of organic solvent was 50% (v/v). (H–J): 1: Control, no addition of any organic solvent; 2: Ethylene glycol; 3: Butanediol; 4: Glycerol; 5: Propylene glycol; 6: Ethanol; 7: Methanol; 8: Isopropyl alcohol; 9: Acetonitrile; 10: Acetone; 11: DMSO; 12: DMF.
Figure 4.
Effects of different metal ions on the stability of LAC-4 laccase. (A) CaCl2, (B) LiCl, (C) MgCl2, (D) NaCl, (E) KCl, (F) AlCl3, (G) MnCl2, (H) ZnCl2. Control, no addition of any metal ion.
Figure 4.
Effects of different metal ions on the stability of LAC-4 laccase. (A) CaCl2, (B) LiCl, (C) MgCl2, (D) NaCl, (E) KCl, (F) AlCl3, (G) MnCl2, (H) ZnCl2. Control, no addition of any metal ion.
Figure 5.
Effects of different organic solvents on the stability of LAC-4 laccase. (A) Propylene glycol (B) Ethylene glycol (C) Glycerol (D) DMSO (E) Ethanol (F) Acetonitrile (G) Isopropyl alcohol (H) Methanol (I) Acetone. Control, no addition of any organic solvent.
Figure 5.
Effects of different organic solvents on the stability of LAC-4 laccase. (A) Propylene glycol (B) Ethylene glycol (C) Glycerol (D) DMSO (E) Ethanol (F) Acetonitrile (G) Isopropyl alcohol (H) Methanol (I) Acetone. Control, no addition of any organic solvent.
Figure 6.
Degradation of chlorophenols with different structures by LAC-4. (A) Degradation of different concentrations of 2,6-DCP by LAC-4. (B) Degradation of different concentrations of 2,3,6-TCP by LAC-4. (C) Degradation of different concentrations of 3-CP by LAC-4. (D) The time-course degradation curve for the different concentrations of 2,6-DCP. (E) The time-course degradation curve for the different concentrations of 2,3,6-TCP. (F) The ln c-t curve for LAC-4 degradation of 2,6-DCP. (G) The ln c-t curve for LAC-4 degradation of 2,3,6-TCP. (H) Degradation of the 2,6-DCP and 2,3,6-TCP mixture by LAC-4. The concentration of 2,6-DCP and 2,3,6-TCP in the mixture was 100 mg/L, 200 mg/L, respectively.
Figure 6.
Degradation of chlorophenols with different structures by LAC-4. (A) Degradation of different concentrations of 2,6-DCP by LAC-4. (B) Degradation of different concentrations of 2,3,6-TCP by LAC-4. (C) Degradation of different concentrations of 3-CP by LAC-4. (D) The time-course degradation curve for the different concentrations of 2,6-DCP. (E) The time-course degradation curve for the different concentrations of 2,3,6-TCP. (F) The ln c-t curve for LAC-4 degradation of 2,6-DCP. (G) The ln c-t curve for LAC-4 degradation of 2,3,6-TCP. (H) Degradation of the 2,6-DCP and 2,3,6-TCP mixture by LAC-4. The concentration of 2,6-DCP and 2,3,6-TCP in the mixture was 100 mg/L, 200 mg/L, respectively.
Figure 7.
Effects of different concentrations of metal salts and organic solvents on the degradation of 2,6-DCP by LAC-4. (A) CuSO4 (B) MgSO4 (C) MnSO4 (D) ZnSO4 (E) K2SO4 (F) Na2SO4 (G) Comparison of the tolerance of LAC-4 for metal salts with sulfate as the anion and with chloride ions as the anion during the degradation of 2,6-DCP. The concentration of metal salt was 400 mM. Control, no addition of any metal salt. (H) Comparison of the tolerance of LAC-4 for metal salts with sulfate as the anion and with chloride ions as the anion during the degradation of 2,6-DCP. The concentration of metal salt was 600 mM. Control, no addition of any metal salt. (I) Concentration of the organic solvent was 5% (v/v). (J) Concentration of the organic solvent was 10% (v/v). (K) Concentration of the organic solvent was 20% (v/v). (L) Concentration of the organic solvent was 50% (v/v). (M) Concentration of the organic solvent was 70% (v/v). (I–M): 1: Control, no addition of any organic solvent. 2: Ethylene glycol; 3: Glycerol; 4: Butanediol; 5: Propylene glycol; 6: Methanol; 7: Ethanol; 8: DMF; 9: DMSO; 10: Acetone; 11: Isopropyl alcohol; 12: Acetonitrile.
Figure 7.
Effects of different concentrations of metal salts and organic solvents on the degradation of 2,6-DCP by LAC-4. (A) CuSO4 (B) MgSO4 (C) MnSO4 (D) ZnSO4 (E) K2SO4 (F) Na2SO4 (G) Comparison of the tolerance of LAC-4 for metal salts with sulfate as the anion and with chloride ions as the anion during the degradation of 2,6-DCP. The concentration of metal salt was 400 mM. Control, no addition of any metal salt. (H) Comparison of the tolerance of LAC-4 for metal salts with sulfate as the anion and with chloride ions as the anion during the degradation of 2,6-DCP. The concentration of metal salt was 600 mM. Control, no addition of any metal salt. (I) Concentration of the organic solvent was 5% (v/v). (J) Concentration of the organic solvent was 10% (v/v). (K) Concentration of the organic solvent was 20% (v/v). (L) Concentration of the organic solvent was 50% (v/v). (M) Concentration of the organic solvent was 70% (v/v). (I–M): 1: Control, no addition of any organic solvent. 2: Ethylene glycol; 3: Glycerol; 4: Butanediol; 5: Propylene glycol; 6: Methanol; 7: Ethanol; 8: DMF; 9: DMSO; 10: Acetone; 11: Isopropyl alcohol; 12: Acetonitrile.
![Ijerph 19 08150 g007]()
Figure 8.
Phytotoxicities of individual chlorophenols (2,6-DCP, 2,3,6-TCP) and chlorophenol mixtures (2,6-DCP + 2,3,6-TCP) at different concentrations before and after degradation by LAC-4. The plant seed shoots in chlorophenols treated with LAC-4 laccase and the plant seed shoots exposed to untreated chlorophenols were measured, respectively. *, significant difference (p < 0.05); **, very significant difference (p < 0.01). (A) The toxicity measurements for the 2,6-DCP degradation products by LAC-4 on mung bean seed germination. Control: no addition of 2,6-DCP; 100-0: 100 mg/L of 2,6-DCP not treated with LAC-4; 100-1: 100 mg/L of 2,6-DCP treated with LAC-4; 200-0: 200 mg/L of 2,6-DCP not treated with LAC-4; 200-1: 200 mg/L of 2,6-DCP treated with LAC-4; 400-0: 400 mg/L of 2,6-DCP not treated with LAC-4; 400-1: 400 mg/L of 2,6-DCP treated with LAC-4; 600-0: 600 mg/L of 2,6-DCP not treated with LAC-4; 600-1: 600 mg/L of 2,6-DCP treated with LAC-4. (B) The toxicity measurements for the 2,6-DCP degradation products by LAC-4 on wheat seed germination. Control: no addition of 2,6-DCP; 100-0: 100 mg/L of 2,6-DCP not treated with LAC-4; 100-1: 100 mg/L of 2,6-DCP treated with LAC-4; 200-0: 200 mg/L of 2,6-DCP not treated with LAC-4; 200-1: 200 mg/L of 2,6-DCP treated with LAC-4; 400-0: 400 mg/L of 2,6-DCP not treated with LAC-4; 400-1: 400 mg/L of 2,6-DCP treated with LAC-4; 600-0: 600 mg/L of 2,6-DCP not treated with LAC-4; 600-1: 600 mg/L of 2,6-DCP treated with LAC-4. (C) The toxicity measurements for the 2,6-DCP degradation products by LAC-4 on rice seed germination. Control: no addition of 2,6-DCP; 400-0: 400 mg/L of 2,6-DCP not treated with LAC-4; 400-1: 400 mg/L of 2,6-DCP treated with LAC-4; 600-0: 600 mg/L of 2,6-DCP not treated with LAC-4; 600-1: 600 mg/L of 2,6-DCP treated with LAC-4. (D) The toxicity measurements for the 2,3,6-TCP degradation products by LAC-4 on mung bean seed germination. Control: no addition of 2,3,6-TCP; 100-0: 100 mg/L of 2,3,6-TCP not treated with LAC-4; 100-1: 100 mg/L of 2,3,6-TCP treated with LAC-4; 200-0: 200 mg/L of 2,3,6-TCP not treated with LAC-4; 200-1: 200 mg/L of 2,3,6-TCP treated with LAC-4; 400-0: 400 mg/L of 2,3,6-TCP not treated with LAC-4; 400-1: 400 mg/L of 2,3,6-TCP treated with LAC-4; 600-0: 600 mg/L of 2,3,6-TCP not treated with LAC-4; 600-1: 600 mg/L of 2,3,6-TCP treated with LAC-4. (E) The toxicity measurements for the 2,3,6-TCP degradation products by LAC-4 on wheat seed germination. Control: no addition of 2,3,6-TCP; 100-0: 100 mg/L of 2,3,6-TCP not treated with LAC-4; 100-1: 100 mg/L of 2,3,6-TCP treated with LAC-4; 400-0: 400 mg/L of 2,3,6-TCP not treated with LAC-4; 400-1: 400 mg/L of 2,3,6-TCP treated with LAC-4. (F) The toxicity measurements for the 2,3,6-TCP degradation products by LAC-4 on rice seed germination. Control: no addition of 2,3,6-TCP; 100-0: 100 mg/L of 2,3,6-TCP not treated with LAC-4; 100-1: 100 mg/L of 2,3,6-TCP treated with LAC-4; 200-0: 200 mg/L of 2,3,6-TCP not treated with LAC-4; 200-1: 200 mg/L of 2,3,6-TCP treated with LAC-4; 400-0: 400 mg/L of 2,3,6-TCP not treated with LAC-4; 400-1: 400 mg/L of 2,3,6-TCP treated with LAC-4. (G) The toxicity measurements for the mixed chlorophenols (2,6-DCP + 2,3,6-TCP) degradation products by LAC-4 on mung bean seed germination. Control: no addition of mixed chlorophenols; 100-0: mixed chlorophenols (100 mg/L 2,6-DCP + 100 mg/L 2,3,6-TCP) not treated with LAC-4; 100-1: mixed chlorophenols (100 mg/L 2,6-DCP + 100 mg/L 2,3,6-TCP) treated with LAC-4; 200-0: mixed chlorophenols (200 mg/L 2,6-DCP + 200 mg/L 2,3,6-TCP) not treated with LAC-4; 200-1: mixed chlorophenols (200 mg/L 2,6-DCP + 200 mg/L 2,3,6-TCP) treated with LAC-4. (H) The toxicity measurements for the mixed chlorophenols (2,6-DCP + 2,3,6-TCP) degradation products by LAC-4 on wheat seed germination. (I) The toxicity measurements for the mixed chlorophenols (2,6-DCP + 2,3,6-TCP) degradation products by LAC-4 on rice seed germination.
Figure 8.
Phytotoxicities of individual chlorophenols (2,6-DCP, 2,3,6-TCP) and chlorophenol mixtures (2,6-DCP + 2,3,6-TCP) at different concentrations before and after degradation by LAC-4. The plant seed shoots in chlorophenols treated with LAC-4 laccase and the plant seed shoots exposed to untreated chlorophenols were measured, respectively. *, significant difference (p < 0.05); **, very significant difference (p < 0.01). (A) The toxicity measurements for the 2,6-DCP degradation products by LAC-4 on mung bean seed germination. Control: no addition of 2,6-DCP; 100-0: 100 mg/L of 2,6-DCP not treated with LAC-4; 100-1: 100 mg/L of 2,6-DCP treated with LAC-4; 200-0: 200 mg/L of 2,6-DCP not treated with LAC-4; 200-1: 200 mg/L of 2,6-DCP treated with LAC-4; 400-0: 400 mg/L of 2,6-DCP not treated with LAC-4; 400-1: 400 mg/L of 2,6-DCP treated with LAC-4; 600-0: 600 mg/L of 2,6-DCP not treated with LAC-4; 600-1: 600 mg/L of 2,6-DCP treated with LAC-4. (B) The toxicity measurements for the 2,6-DCP degradation products by LAC-4 on wheat seed germination. Control: no addition of 2,6-DCP; 100-0: 100 mg/L of 2,6-DCP not treated with LAC-4; 100-1: 100 mg/L of 2,6-DCP treated with LAC-4; 200-0: 200 mg/L of 2,6-DCP not treated with LAC-4; 200-1: 200 mg/L of 2,6-DCP treated with LAC-4; 400-0: 400 mg/L of 2,6-DCP not treated with LAC-4; 400-1: 400 mg/L of 2,6-DCP treated with LAC-4; 600-0: 600 mg/L of 2,6-DCP not treated with LAC-4; 600-1: 600 mg/L of 2,6-DCP treated with LAC-4. (C) The toxicity measurements for the 2,6-DCP degradation products by LAC-4 on rice seed germination. Control: no addition of 2,6-DCP; 400-0: 400 mg/L of 2,6-DCP not treated with LAC-4; 400-1: 400 mg/L of 2,6-DCP treated with LAC-4; 600-0: 600 mg/L of 2,6-DCP not treated with LAC-4; 600-1: 600 mg/L of 2,6-DCP treated with LAC-4. (D) The toxicity measurements for the 2,3,6-TCP degradation products by LAC-4 on mung bean seed germination. Control: no addition of 2,3,6-TCP; 100-0: 100 mg/L of 2,3,6-TCP not treated with LAC-4; 100-1: 100 mg/L of 2,3,6-TCP treated with LAC-4; 200-0: 200 mg/L of 2,3,6-TCP not treated with LAC-4; 200-1: 200 mg/L of 2,3,6-TCP treated with LAC-4; 400-0: 400 mg/L of 2,3,6-TCP not treated with LAC-4; 400-1: 400 mg/L of 2,3,6-TCP treated with LAC-4; 600-0: 600 mg/L of 2,3,6-TCP not treated with LAC-4; 600-1: 600 mg/L of 2,3,6-TCP treated with LAC-4. (E) The toxicity measurements for the 2,3,6-TCP degradation products by LAC-4 on wheat seed germination. Control: no addition of 2,3,6-TCP; 100-0: 100 mg/L of 2,3,6-TCP not treated with LAC-4; 100-1: 100 mg/L of 2,3,6-TCP treated with LAC-4; 400-0: 400 mg/L of 2,3,6-TCP not treated with LAC-4; 400-1: 400 mg/L of 2,3,6-TCP treated with LAC-4. (F) The toxicity measurements for the 2,3,6-TCP degradation products by LAC-4 on rice seed germination. Control: no addition of 2,3,6-TCP; 100-0: 100 mg/L of 2,3,6-TCP not treated with LAC-4; 100-1: 100 mg/L of 2,3,6-TCP treated with LAC-4; 200-0: 200 mg/L of 2,3,6-TCP not treated with LAC-4; 200-1: 200 mg/L of 2,3,6-TCP treated with LAC-4; 400-0: 400 mg/L of 2,3,6-TCP not treated with LAC-4; 400-1: 400 mg/L of 2,3,6-TCP treated with LAC-4. (G) The toxicity measurements for the mixed chlorophenols (2,6-DCP + 2,3,6-TCP) degradation products by LAC-4 on mung bean seed germination. Control: no addition of mixed chlorophenols; 100-0: mixed chlorophenols (100 mg/L 2,6-DCP + 100 mg/L 2,3,6-TCP) not treated with LAC-4; 100-1: mixed chlorophenols (100 mg/L 2,6-DCP + 100 mg/L 2,3,6-TCP) treated with LAC-4; 200-0: mixed chlorophenols (200 mg/L 2,6-DCP + 200 mg/L 2,3,6-TCP) not treated with LAC-4; 200-1: mixed chlorophenols (200 mg/L 2,6-DCP + 200 mg/L 2,3,6-TCP) treated with LAC-4. (H) The toxicity measurements for the mixed chlorophenols (2,6-DCP + 2,3,6-TCP) degradation products by LAC-4 on wheat seed germination. (I) The toxicity measurements for the mixed chlorophenols (2,6-DCP + 2,3,6-TCP) degradation products by LAC-4 on rice seed germination.
![Ijerph 19 08150 g008]()
Figure 9.
Gas chromatography (GC) peak spectra of degradation products of 2,6-DCP degraded by LAC-4 at different time points. (A) 0 h, (B) 0.5 h, (C) 3 h, (D) 12 h.
Figure 9.
Gas chromatography (GC) peak spectra of degradation products of 2,6-DCP degraded by LAC-4 at different time points. (A) 0 h, (B) 0.5 h, (C) 3 h, (D) 12 h.
Figure 10.
Mass spectrum (MS) of degradation products of 2,6-DCP degraded by LAC-4. Three products were detected by GC-MS analysis: the m/z values of substances I, II and III were 324, 323.8 and 321.9, respectively.
Figure 10.
Mass spectrum (MS) of degradation products of 2,6-DCP degraded by LAC-4. Three products were detected by GC-MS analysis: the m/z values of substances I, II and III were 324, 323.8 and 321.9, respectively.
Figure 11.
Free radicals generated by 2,6-DCP catalyzed by LAC-4 laccase (A) and the coupling reaction between the free radicals (B). 2,6-DCP was first oxidized by LAC-4 laccase, generating three chemically stable free radicals (A); then, six possible coupling reactions randomly occurred between the free radical monomers (B).
Figure 11.
Free radicals generated by 2,6-DCP catalyzed by LAC-4 laccase (A) and the coupling reaction between the free radicals (B). 2,6-DCP was first oxidized by LAC-4 laccase, generating three chemically stable free radicals (A); then, six possible coupling reactions randomly occurred between the free radical monomers (B).
Figure 12.
Speculation of the degradation pathway of 2,6-DCP catalyzed by LAC-4 laccase. The putative degradation pathway of 2,6-DCP catalyzed by LAC-4 laccase. (a) 2,6-DCP; (b) free radicals; (c) dimer products.
Figure 12.
Speculation of the degradation pathway of 2,6-DCP catalyzed by LAC-4 laccase. The putative degradation pathway of 2,6-DCP catalyzed by LAC-4 laccase. (a) 2,6-DCP; (b) free radicals; (c) dimer products.
Figure 13.
Gas chromatography (GC) peak spectra of degradation products of 2,3,6-TCP degraded by LAC-4 at different time points. (a) 0 h, (b) 0.5 h, (c) 3 h, (d) 12 h.
Figure 13.
Gas chromatography (GC) peak spectra of degradation products of 2,3,6-TCP degraded by LAC-4 at different time points. (a) 0 h, (b) 0.5 h, (c) 3 h, (d) 12 h.
Figure 14.
Mass spectrum (MS) of degradation products of 2,3,6-TCP degraded by LAC-4. (A) Mass spectrum of 2,3,6-TCP. (a1) is the MS detection result of 2,3,6-trichlorophenol (not degraded). (a2) is the database matching result. (B) Two intermediate products were detected by GC-MS: product I (b1), which had a retention time of 16.517 min with a m/z of 391.8, and product II (b2), which had a retention time of 17.801 min with a m/z of 391.8.
Figure 14.
Mass spectrum (MS) of degradation products of 2,3,6-TCP degraded by LAC-4. (A) Mass spectrum of 2,3,6-TCP. (a1) is the MS detection result of 2,3,6-trichlorophenol (not degraded). (a2) is the database matching result. (B) Two intermediate products were detected by GC-MS: product I (b1), which had a retention time of 16.517 min with a m/z of 391.8, and product II (b2), which had a retention time of 17.801 min with a m/z of 391.8.
Figure 15.
Free radicals generated by 2,3,6-TCP catalyzed by LAC-4 laccase (A) and the coupling reaction between the free radicals (B). 2,3,6-TCP was first oxidized by LAC-4 laccase, generating four chemically stable free radicals (A, ①–④); then, ten possible coupling reactions randomly occurred between the free radical monomers (B, (1)–(10)).
Figure 15.
Free radicals generated by 2,3,6-TCP catalyzed by LAC-4 laccase (A) and the coupling reaction between the free radicals (B). 2,3,6-TCP was first oxidized by LAC-4 laccase, generating four chemically stable free radicals (A, ①–④); then, ten possible coupling reactions randomly occurred between the free radical monomers (B, (1)–(10)).
Figure 16.
Speculation of the degradation pathway of 2,3,6-TCP catalyzed by LAC-4 laccase. The putative degradation pathway of 2,3,6-TCP catalyzed by LAC-4 laccase.
Figure 16.
Speculation of the degradation pathway of 2,3,6-TCP catalyzed by LAC-4 laccase. The putative degradation pathway of 2,3,6-TCP catalyzed by LAC-4 laccase.
Figure 17.
The degradation products of an equivalent mixture of 2,6-DCP and 2,3,6-TCP catalyzed by LAC-4 laccase contain four homodimers and three heterodimers.
Figure 17.
The degradation products of an equivalent mixture of 2,6-DCP and 2,3,6-TCP catalyzed by LAC-4 laccase contain four homodimers and three heterodimers.
Table 1.
The kinetic parameters of purified LAC-4 laccase.
Table 1.
The kinetic parameters of purified LAC-4 laccase.
| | Km (mM) | Vmax (mM/s) | Kcat (s−1) | Kcat/Km (s−1mM−1) |
|---|
| ABTS | 0.075 | 3.988 × 10−7 | 9.42 | 126.27 |
| 2,6-DMP | 0.803 | 1.085 × 10−7 | 2.56 | 3.19 |
| Guaiacol | 0.097 | 1.906 × 10−7 | 4.5 | 46.34 |
Table 2.
The kinetic parameters of degradation of 2,6-DCP and 2,3,6-TCP by LAC-4.
Table 2.
The kinetic parameters of degradation of 2,6-DCP and 2,3,6-TCP by LAC-4.
| | Km (mM) | Vmax (mM/s) | Kcat (s−1) | Kcat/Km (s−1mM−1) |
|---|
| 2,6-DCP | 3.636 | 2.74 × 10−3 | 1.23 | 0.338 |
| 2,3,6-TCP | 4.875 | 1.50 × 10−3 | 0.67 | 0.137 |
Table 3.
Effects of different concentrations of 2,6-DCP on the germination rates of wheat seeds before and after degradation by LAC-4 laccase.
Table 3.
Effects of different concentrations of 2,6-DCP on the germination rates of wheat seeds before and after degradation by LAC-4 laccase.
| | 2,6-DCP |
|---|
| Control | 100-0 | 100-1 | 200-0 | 200-1 | 400-0 | 400-1 | 600-0 | 600-1 |
|---|
| wheat seed germination rate (%) | 95 | 95 | 95 | 80 | 95 | 60 | 100 | 0 | 80 |
Table 4.
Effects of different concentrations of 2,6-DCP on the germination rates of rice seeds before and after degradation by LAC-4 laccase.
Table 4.
Effects of different concentrations of 2,6-DCP on the germination rates of rice seeds before and after degradation by LAC-4 laccase.
| | 2,6-DCP |
|---|
| | Control | 400-0 | 400-1 | 600-0 | 600-1 |
|---|
| rice seed germination rate (%) | 95 | 70 | 85 | 0 | 50 |
Table 5.
Effects of different concentrations of 2,3,6-TCP on the germination rates of wheat and rice seeds before and after degradation by LAC-4 laccase.
Table 5.
Effects of different concentrations of 2,3,6-TCP on the germination rates of wheat and rice seeds before and after degradation by LAC-4 laccase.
| | 2,3,6-TCP |
|---|
| Control | 100-0 | 100-1 | 200-0 | 200-1 | 400-0 | 400-1 |
|---|
| wheat seed germination rate (%) | 90 | 35 | 95 | 15 | 95 | 0 | 25 |
| rice seed germination rate (%) | 95 | 75 | 80 | 0 | 80 | 0 | 50 |
Table 6.
Effects of different concentrations of mixed chlorophenol (2,6-DCP + 2,3,6-TCP) on the germination rates of mung bean and wheat seeds before and after degradation by LAC-4 laccase.
Table 6.
Effects of different concentrations of mixed chlorophenol (2,6-DCP + 2,3,6-TCP) on the germination rates of mung bean and wheat seeds before and after degradation by LAC-4 laccase.
| | 2,6-DCP + 2,3,6-TCP |
|---|
| Control | 100-0 | 100-1 | 200-0 | 200-1 |
|---|
| mung bean seed germination rate (%) | 100 | 100 | 100 | 70 | 100 |
| wheat seed germination rate (%) | 95 | 40 | 90 | 0 | 15 |