Abstract
Soybean plants are highly susceptible to Fusarium species, which significantly reduce soybean production and quality. Several Fusarium species have been reported to synthesize mycotoxins, such as trichothecene, which have been related to major human diseases. In November 2021, soybean pods in Nantong municipality, China, showed black necrotic lesions during the harvest stage. The disease incidence reached 69%. The pathogen was identified as Fusarium sulawense via morphological analysis and sequencing of ITS, EF1-α and RPB2 genes. A PCR assay with primers targeting the trichothecene biosynthesis genes suggested that the three isolates could synthesize trichothecenes. The effectiveness of fungicide carbendazim and natural metabolites dipicolinic acid and kojic acid was screened for the management of F. sulawense on postharvest soybean pods. The highest efficacy was obtained when combining 3.8 mg/mL carbendazim and 0.84 mg/mL dipicolinic acid (curative efficacy: 49.1% lesion length inhibition; preventive efficacy: 82.7% lesion length inhibition), or 1.9 mg/mL carbendazim and 0.71 mg/mL kojic acid (preventive efficacy: 84.9% lesion length inhibition). Collectively, this report will lead to a better understanding of the safety hazards found in soybean products in China and reveals the application of dipicolinic and kojic acids to reduce the use of carbendazim.
1. Introduction
In 2020, soybean production reached 353 million tons, with major producing areas in the USA, Brazil, Argentina and China [1]. Soybean-derived food products provide essential proteins, antioxidants and minerals, and are rich in isoflavones [2]. Several studies have shown that soy products can reduce serum concentrations of total cholesterol, while fermented soy products are effective in attenuating the effects of diabetes, blood pressure, cardiac disorders and cancer [3]. Despite their high nutritional and economic relevance, soybean plants are highly susceptible to a wide range of pathogens that reduce crop production and quality [4,5,6]. It has been estimated that, from 1996 to 2016, the total economic loss caused by soybean diseases in the USA was 95.48 billion dollars [7,8].
Fusarium species have been reported to attack different soybean tissues and cause a variety of diseases on soybean plants, such as wilting, root rot, seed rot and pod blight [9,10,11,12]. Some Fusarium species can infect different tissues. For example, F. oxysporum has been reported to cause soybean root rot, cotyledon spot and wilting [13,14,15]. Several screenings of soybean diseases have indicated that Fusarium is the most common fungal pathogen infecting soybean crops [16,17]. Sudden death syndrome, which is caused by F. virguliforme, is among the most important diseases affecting soybean production in the USA [18]. F. acuminatum, F. avenaceum, F. commune, F. culmorum, F. equiseti, F. oxysporum, F. proliferatum, F. redolens, F. torulosum and F. tricinctum were detected in diseased soybean roots in Alberta, Canada [19]. F. avenaceum was the most common species associated with soybean grains in Poland, followed by F. equiseti and F. sporotrichioides [20]. Recently, several Fusarium species, including F. fujikuroi, F. luffae, F. proliferatum and F. sulawense, were found associated with soybean pods in Hubei Province, China [21].
Apart from the enormous economic losses, some Fusarium strains can synthesize toxins, such as 4-acetyl nivalenol, 3-acetyl deoxynivalenol, diacetoxyscirpenol, neosolaniol, nivalenol and zearalenone [22]. Some of these toxins have been reported to induce anorexia, modify DNA by altering methylation and histone acetylation patterns and cause diabetes [23]. Fumonisins, which are synthesized by some Fusarium strains, cause fatal livestock diseases and are considered potentially carcinogenic mycotoxins for humans [24]. F. sulawense, among other species, has been reported to synthesize mycotoxin trichothecene, which can cause breathing problems and lung inflammation [25].
One of the main limitations in the control of soybean diseases caused by Fusarium species is the lack of information regarding the identity of the pathogens that are currently affecting soybean crops and the development of suitable methods for their management. In this study, F. sulawense, also named F. sulawesiense, was identified as the causal agent of soybean pod blight in Nantong municipality, China. Carbendazim, dipicolinic acid and kojic acid were evaluated as antifungal agents for F. sulawense management on soybean.
2. Materials and Methods
2.1. Sample Collection and Fungal Isolation
In November 2021, soybean pod blight symptoms were observed in Nantong municipality, China (120.54° E, 31.58° N). The symptoms were observed in nearly 69% of the pods from a commercial soybean field (3.2 ha). The field contained 3-month-old soybean plants in the harvest stage. Five small pieces of symptomatic soybean pods, approximately 0.3 cm2 in size, were extracted from different plants, surface sterilized in 1.5% NaOCl for 1 min and washed twice with sterile ddH2O. The pathogen was cultured on potato-dextrose-agar (PDA) medium containing chloramphenicol (50 µg/mL) in darkness at 26 °C for 5 days. PDA medium was prepared by boiling 200 g of potatoes in 1 L of water for 30 min. Then, the solution was filtered, and 20 g dextrose and 15 g agar were added (pH = 5.6). Three isolates, NJC31, NJC32 and NJC33 (NJC = Nantong, Jiangsu, China), were obtained via single spore isolation [26].
2.2. Morphological Characterization
A 5-mm-diameter mycelial PDA plug containing NJC31 was incubated in 50 mL yeast extract-peptone-dextrose (YEPD) medium (0.15 g yeast extract, 0.5 g peptone and 1 g glucose in 50 mL ddH2O) at 26 °C and 200 rpm in darkness for 48 h. Fungal morphology was observed using a Leica DM2500 microscope (Germany).
2.3. Sequence Analysis
The three isolates were grown in 100 mL potato-dextrose broth (PDB; prepared as PDA medium but in the absence of agar) at 26 °C and 200 rpm in darkness for 48 h. The genomic DNA was extracted using the Ezup Column Fungi Genomic DNA Purification Kit (Sangon Biotech, China) according to the manufacturer’s instructions. Internal transcribed spacer (ITS) region of rDNA, elongation factor 1-α (EF1-α) and RNA polymerase II second largest subunit (RPB2) partial sequences were amplified using the ITS1/ITS4 (5′-TCCGTAGCTGAACCTGCGG-3′ and 5′-TCCTCCGCTTATTGATATGC-3′, respectively), EF1-728F/EF1-986R (5′-CATCGAGAAGTTCGAGAAGG-3′ and 5′-TACTTGAAGGAACCCTTACC-3′, respectively) and fRPB2-7CF/fRPB2-11aR (5′-ATGGGYAARCAAGCYATGGG-3′ and 5′-GCRTGGATCTTRTCRTCSACC-3′, respectively) primers [27,28,29].
Polymerase Chain Reaction (PCR) amplification was carried out in a 50 μL reaction mixture, which contained 25 μL of 2 × primestar, 1 μL of each primer (10 μM), 1 μL of DNA template (70 ng/μL) and 22 μL of ddH2O using a PCR Thermal Cycler (Techne HEMR 9600, China). The PCR conditions consisted of 2 min of initial denaturation at 98.0 °C, followed by 32 cycles of denaturation at 98 °C for 10 s; primer annealing 30 s at 53.8 °C for ITS, 56.8 °C for EF1-α and 55.9 °C for RPB2; extension at 72 °C for 80 s; and final extension at 72 °C for 2 min. Amplicons were checked by electrophoresis using a 1.0% agarose gel in TAE buffer (1×) and visualized with a UV luminometer (Tanon 1600R, China). DNA sequencing was performed by Sangon Biotech (Shanghai, China).
The obtained sequences were submitted to GenBank under accession numbers ON646105, ON646177 and ON646200 (ITS); ON685317-ON685319 (EF1-α); and ON685320-ON685322 (RPB2) (Table S1 and Figure S1).
2.4. Construction of the Phylogenetic Tree
The phylogenetic tree based on the EF1-α and RPB2 sequences was constructed using MEGA7 with ex-type strains retrieved from GenBank. The ITS gene was not included in the phylogenetic tree due to the ITS gene of some ex-type isolates, including F. lactis CBS 411.97 and F. parvisorum CBS 137236, was not available in GenBank. After removing ambiguous regions and autapomorphic insertions manually, the alignment resulted in three matrices with a length of 594 (EF1-α) and 1646 (RPB2) sites. The evolutionary history was inferred using the Maximum Likelihood method based on the Tamura 3-parameter model. The percentage of trees in which the associated taxa clustered together is shown next to the branches. Initial trees for the heuristic search were obtained automatically by applying the Neighbor-Join and BioNJ algorithms to a matrix of pairwise distances estimated using the Maximum Composite Likelihood (MCL) approach, and then selecting the topology with a superior log likelihood value (−6447.23). A discrete Gamma distribution was used to model evolutionary rate differences among sites (5 categories (+G, parameter = 0.6742)). There were a total of 2241 positions in the final dataset. The number of bootstrap replications was 1000. The tree is drawn to scale, with branch lengths measured in the number of substitutions per site.
Apart from NJC31, NJC32 and NJC33, the EF1-α and RPB2 sequences of 16 ex-type Fusarium isolates, including F. sulawense InaCC F964, F. kotabaruense InaCC F963, F. tanahbumbuense InaCC F965, F. lacertarum CBS 130185, F. lactis CBS 411.97, F. mundagurra NRRL 66235, F. napiforme CBS 748.97, F. nygamai CBS 749.97, F. parvisorum CBS 137236, F. phyllophilum CBS 216.76, F. pseudocircinatum CBS 449.97, F. pseudonugamai CBS 417.97, F. ramigenum CBS 418.98, F. sudanense CBS 454.97, F. terricola CBS 483.94 and F. tjaetaba NRRL 66243 [30], were used for the construction of the phylogenetic tree. Lasiodiplodia theobromae CBS 164.96 was used as an outgroup strain [31].
2.5. Pathogenicity Test
Re-inoculation of NJC31, NJC32 and NJC33 was carried out using detached ‘Lee68′ soybean pods. The pods were surface sterilized for 1 min in 1.5% NaOCl and washed twice with sterile ddH2O. A 0.5 cm2 size wound was cut in each soybean pod using a sterilized knife. The pathogens were inoculated using a 1 × 106 spores/mL (20 μL) conidial suspension, which was prepared by extracting F. sulawense conidia from PDA medium. Each soybean pod contained one inoculation site. Sterilized ddH2O was used in the control experiment. Ten soybean pods were used for each isolate. Experiments were repeated 3 times. The pathogen was recovered from the wounds and the identity of the pathogen was confirmed via sequencing and morphology analyses, following Koch’s postulates.
The disease severity index (DSI) was calculated for each strain using a 3-point scale. The scores were 0 = no symptoms, 1 = mild symptoms (slight discoloration of pods), 2 = obvious lesions (pods were obviously diseased) and 3 = severe lesions (pods showing rot symptoms).
2.6. PCR Assay of Trichothecene Biosynthetic Genes
The trichothecene biosynthetic genes TRI4, TRI5, TRI13NIV and TRI13DON were amplified following the conditions reported by Stepien et al. [32,33]. The primers used in the amplification were 5′-CCCCTGGCTACTCTCGAGA-3′ and 5′-AAGCTTTGAGAACCTTCAC-3′ (TRI4), 5′-AGCGACTACAGGCTTCCCTC-3′ and 5′-AAACCATCCAGTTCTCCATCT-3′ (TRI5), 5′-CCAAATCCGAAAACCGCAG-3′ and 5′-TTGAAAGCTCCAATGTCGTG-3′ (TRI13NIV) and 5′-CATCATGAGACTTGTKRAGTTTGGG-3′ and 5′-GCTAGATCGATTGTTGCATTGAG-3′ (TRI13DON). The PCR amplification was carried out in a 50 μL reaction mixture, which contained 25 μL of 2 × primestar, 1 μL of each primer (10 μM), 1 μL of DNA template and 22 μL of ddH2O using a PCR Thermal Cycler (Techne HEMR 9600). The PCR conditions consisted of 15 min of initial denaturation at 94.0 °C, followed by 35 cycles of denaturation at 94 °C for 60 s, primer annealing at 58.0 °C for 30 s, extension at 72 °C for 60 s and final extension at 72 °C for 10 min.
PCR products were observed by electrophoresis using the procedure indicated in Section 2.3.
2.7. Efficacy of Carbendazim, Dipicolinic Acid and Kojic Acid for the Control of F. sulawense on Soybean Pods
Soybean pods were sterilized and wounded following the conditions indicated in Section 2.5. NJC31 was used in the screening. A 1 × 106 NJC31 spores/mL (20 μL) conidial suspension was prepared as indicated in Section 2.5. For the curative assay, NJC31 conidia was poured into the wound, and the pods were maintained at 28 °C and 90% relative humidity for 24 h. Then, 10 mL of aqueous solutions containing carbendazim (3.8 mg/mL), dipicolinic acid (0.84 mg/mL) or kojic acid (0.71 mg/mL) were sprayed. Technical compounds (and not commercial fungicides) were used in the screening. The concentration of carbendazim was adjusted to the application rate recommended by suppliers, which suggests the use of 0.8 g carbendazim per kg of seeds for the management of Fusarium [34]. Dipicolinic (0.84 mg/mL) and kojic acids (0.71 mg/mL) were applied at the same concentrations previously reported [35,36]. After spraying the antifungal compounds, the soybean pods were kept at 28 °C and 90% relative humidity for 24 h. Sterilized ddH2O (10 mL) was sprayed in the control treatment. The efficacy of the fungicides was calculated according to the lesion length caused by NJC31. Twenty pods were used for each treatment condition. Experiments were repeated 3 times.
For the preventive assay, 10 mL aqueous solutions containing carbendazim (3.8 mg/mL), dipicolinic acid (0.84 mg/mL) or kojic acid (0.71 mg/mL) were sprayed on the wounded soybean pods. After drying at room temperature using a fan, a 1 × 106 NJC31 spores/mL (20 μL) conidial suspension was injected into each wound. Sterilized ddH2O (10 mL) was sprayed in the control treatment. The soybean pods were incubated at 28 °C and 90% relative humidity for 72 h. Twenty pods were used per treatment. Experiments were repeated 3 times.
2.8. Combinations of Carbendazim, Dipicolinic Acid and Kojic Acid for the Control of F. sulawense on Soybean Pods
The following combinations were screened: (1) carbendazim (3.8 mg/mL)/dipicolinic acid (0.84 mg/mL); (2) carbendazim (1.9 mg/mL)/dipicolinic acid (0.84 mg/mL); (3) carbendazim (0.95 mg/mL)/dipicolinic acid (0.84 mg/mL); (4) carbendazim (3.8 mg/mL)/kojic acid (0.71 mg/mL); (5) carbendazim (1.9 mg/mL)/kojic acid (0.71 mg/mL); (6) carbendazim (0.95 mg/mL)/kojic acid (0.71 mg/mL); (7) dipicolinic acid (0.84 mg/mL)/kojic acid (0.71 mg/mL); (8) dipicolinic acid (0.21 mg/mL)/kojic acid (0.71 mg/mL); and (9) dipicolinic acid (0.84 mg/mL)/kojic acid (0.18 mg/mL). Their efficacy was screened in curative and preventive applications as described in Section 2.7. Sterilized ddH2O (10 mL) was sprayed in the control treatment. The soybean pods were incubated at 28 °C and 90% relative humidity for 72 h. Twenty pods were used per treatment condition. Experiments were repeated 3 times.
2.9. Combinations of Carbendazim, Dipicolinic Acid and Kojic Acid for the Control of F. sulawense on Soybean Pods
The protection provided by carbendazim and kojic acid for the control of NJC31 was evaluated at different time points. The preventive assay described in Section 2.7 was followed. The antifungals were applied at two different concentrations: (1) carbendazim (1.9 mg/mL)/kojic acid (0.71 mg/mL) and (2) carbendazim (0.95 mg/mL)/kojic acid (0.71 mg/mL). Then, the pathogen was inoculated after 0, 1, 2, 3, 4, 5, 6 and 7 days. The soybean pods were incubated at 28 °C and 90% relative humidity for 72 h after the inoculation of the pathogen. Twenty pods were used for each treatment condition. Experiments were repeated 3 times.
2.10. Statistical Analysis
Statistical analyses were performed using SPSS software v. 20.0 (IBM Corp., Armonk, NY, USA). The obtained data were processed using one-way ANOVA with post hoc multiple comparisons. Means were considered significantly different when p ≤ 0.05.
3. Results
3.1. F. sulawense Was Identified as the Causal Agent of Soybean Pod Blight in Nantong Municipality
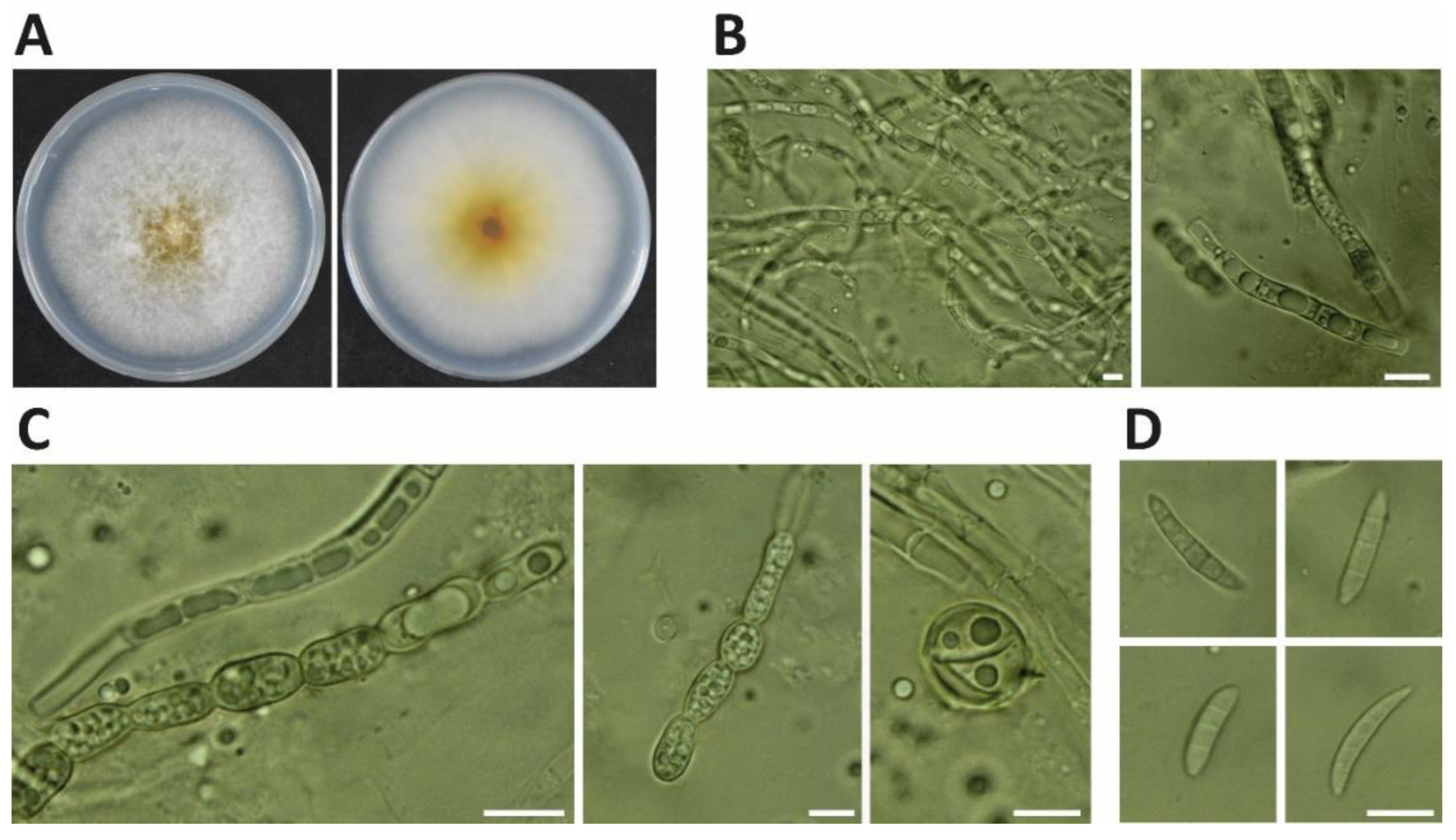
In November 2021, soybean pods (Glycine max cv. ‘Lee68′) at the harvest stage exhibited black necrotic lesions (Figure 1). Three isolates, NJC31, NJC32 and NJC33, were obtained. The colonies of the three isolates showed white mycelium with yellow pigmentation on PDA (Figure 2). Microscope observations revealed the presence of septate mycelium. Linear (60–200 μm long) and round (approximately 10 μm diameter) chlamydospores were observed (number of observations = 89). Fusiform macroconidia, 10 to 15 µm in length, showed a curved shape and contained between 3 and 5 septum separations (number of observations = 56).

Figure 1.
Symptoms caused by Fusarium sulawense and location. (A) Soybean pods (Glycine max cv. ‘Lee68’) in Nantong municipality exhibited black necrotic lesions and were heavily mildewed. (B) A map of China showing the municipalities of Nantong, Jiangsu Province and Jingzhou, Hubei Province. F. sulawense was identified as the causal agent of soybean pod blight in Nantong municipality in 2021 (this work) and in Jingzhou municipality in 2020 [21].
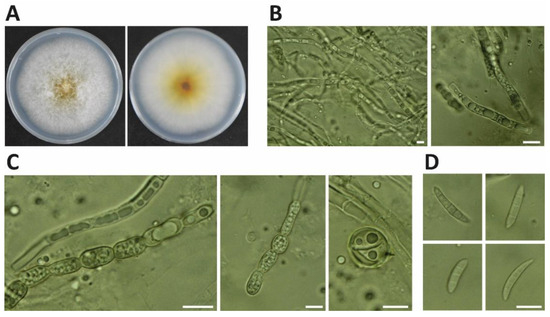
Figure 2.
Morphological characteristics of Fusarium sulawense NJC31 isolated from soybean pods. (A) Colony of F. sulawense on PDA at 26 °C after incubation for 5 days. (B) Septate mycelium. (C) Linear and round chlamydospores (number of observations = 89). (D) Fusiform macroconidia (number of observations = 56) (10–15 μm in length) containing 3–5 septum separations. Bar = 10 μm.
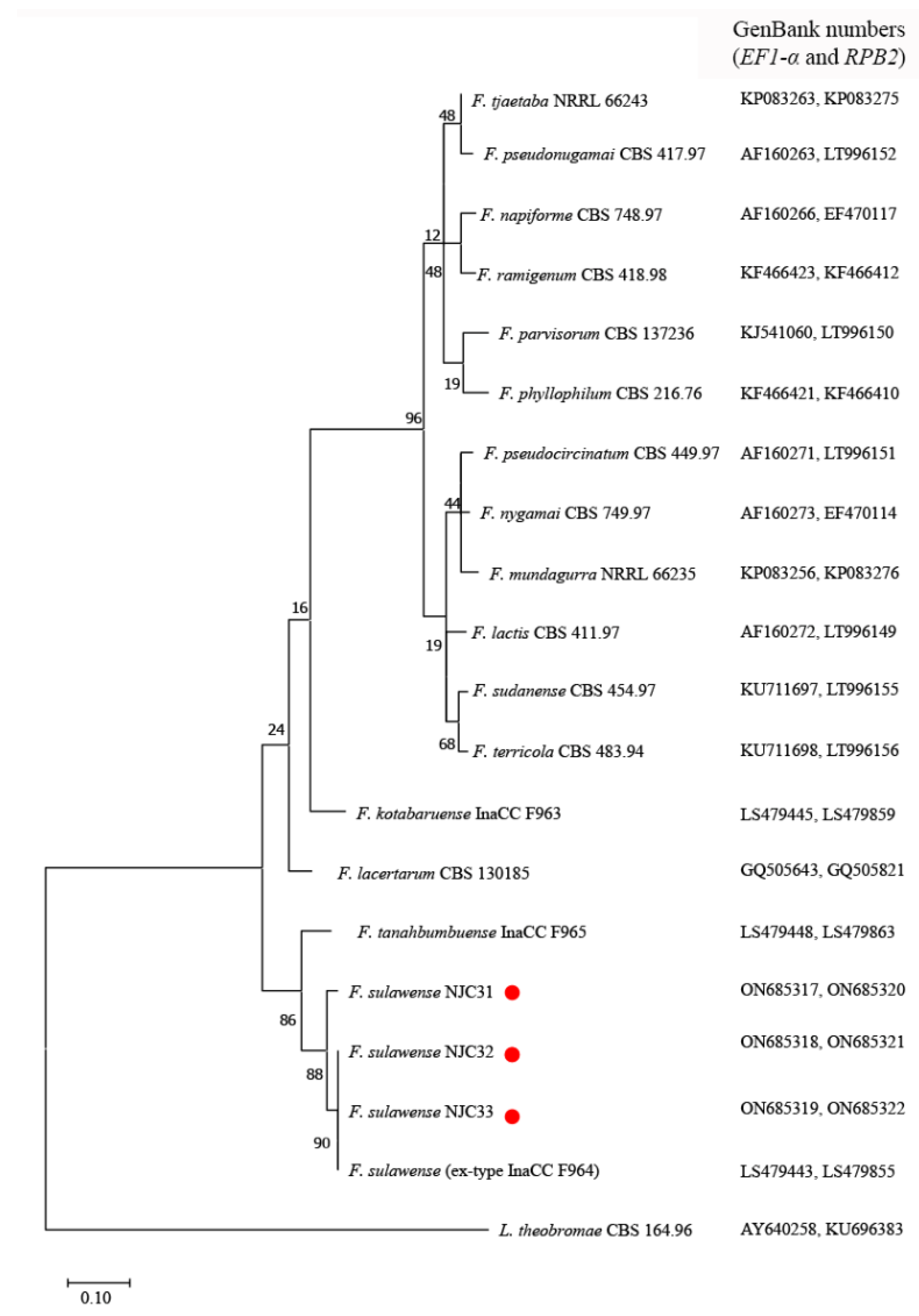
The ITS sequences of NJC31, NJC32 and NJC33 showed 100% identity compared to reference F. sulawense strains LC7919 and LC7920 (GenBank numbers: MK280811 and MK280805), which were also isolated in China [37]. The EF1-α sequence of NJC31 was 100% identical with F. sulawense LC7919, LC7920 and LC12149 (GenBank numbers: MK280811, MK280805 and MK280783), while the EF1-α sequences of NJC32 and NJC33 were 100% identical compared to the sequences from F. sulawense LC12176 and LC12169 (GenBank numbers: MK280839 and MK289756) [37]. The RPB2 sequence of NJC31 showed 98.50% identity with respect to the RPB2 sequences from F. sulawense LC7939, LC7919 and LC12176 (GenBank numbers: MK289795, MK289786 and MK289761); the RPB2 sequence of NJC32 showed 99.89% homology with respect to the RPB2 sequences from LC7939, LC7919 and LC12176; and the RPB2 sequence of NJC33 showed 100% homology concerning the RPB2 sequences from LC7939, LC7919 and LC12176 [37]. The amplified sequences showed 99.59–99.80% (ITS) and 97.63–100% (EF1-α) homology to the corresponding sequences of ex-type strain F. sulawense InaCC F964 (GenBank numbers: LS479410 and LS479443) [30]. Figure 3 shows a phylogenetic tree constructed by combining the EF1-α and RPB2 sequences.
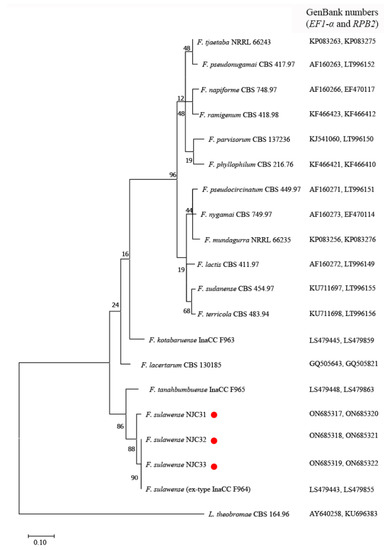
Figure 3.
Phylogenetic analysis of elongation factor 1-α (EF1-α) and RNA polymerase II second largest subunit (RPB2) sequences from Fusarium sulawense strains NJC31, NJC32 and NJC33. The tree was constructed using MEGA7 with reference strains retrieved from GenBank. The evolutionary history was inferred using the Maximum Likelihood method and Tamura 3-parameter model. The tree with the highest log likelihood (−6447.23) is shown. A discrete Gamma distribution was used to model evolutionary rate differences among sites (5 categories (+G, parameter = 0.6742). The rate variation model allowed for some sites to be evolutionarily invariable ([+I], 20.81% sites). Default parameters were used in the analysis. The red points indicate the strains isolated in this work.
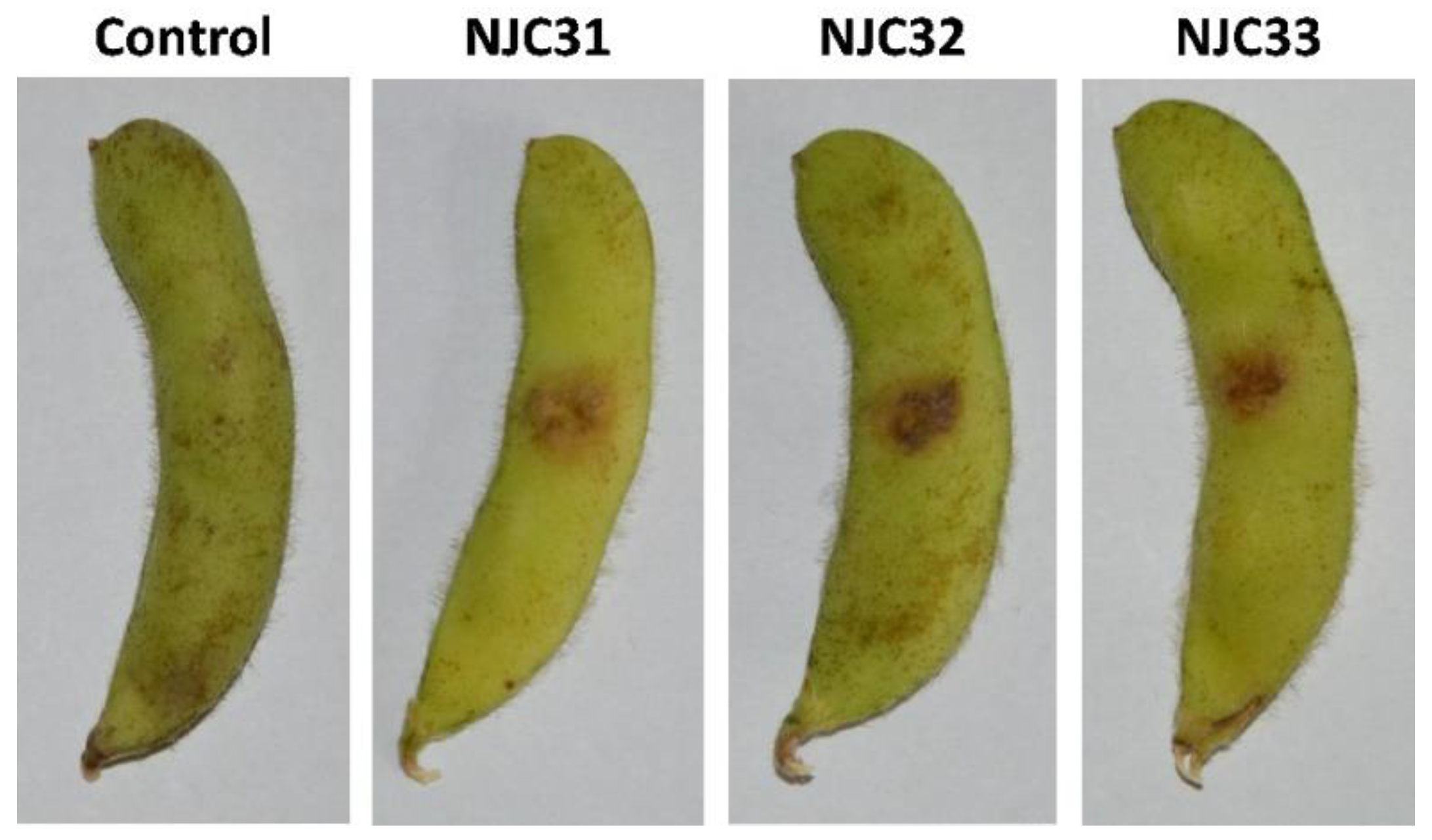
A pathogenicity assay was carried out to confirm the virulence of the isolates (Figure 4). After inoculation of the strains, black-brown necrotic lesions were easily observed with the naked eye. The pathogen was recovered from the lesions and identified, confirming that F. sulawense was the causal agent of rot on soybean pods. The disease severity ranged from 75.1% to 88.4% for NJC31, 76.3% to 85.5% for NJC32 and 77.2% to 84.2% for NJC33, indicating that the three isolates were highly virulent.
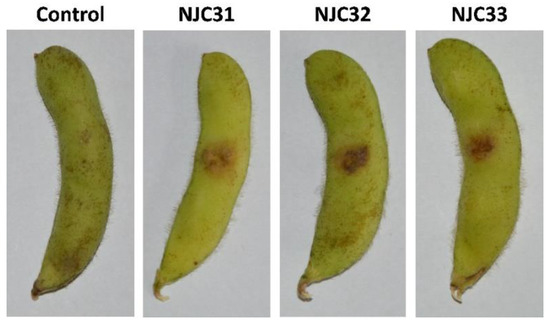
Figure 4.
Pathogenicity assay of Fusarium sulawense strains NJC31, NJC32 and NJC33. A 0.5 cm2 size wound was cut in each soybean pod and the pathogens were inoculated using a 1 × 106 spores/mL (20 μL) conidial suspension. Sterilized ddH2O was used in the control experiment. The pathogen was recovered from the wounds and the identity of the pathogen was confirmed via sequencing and morphology analyses, following Koch’s postulates.
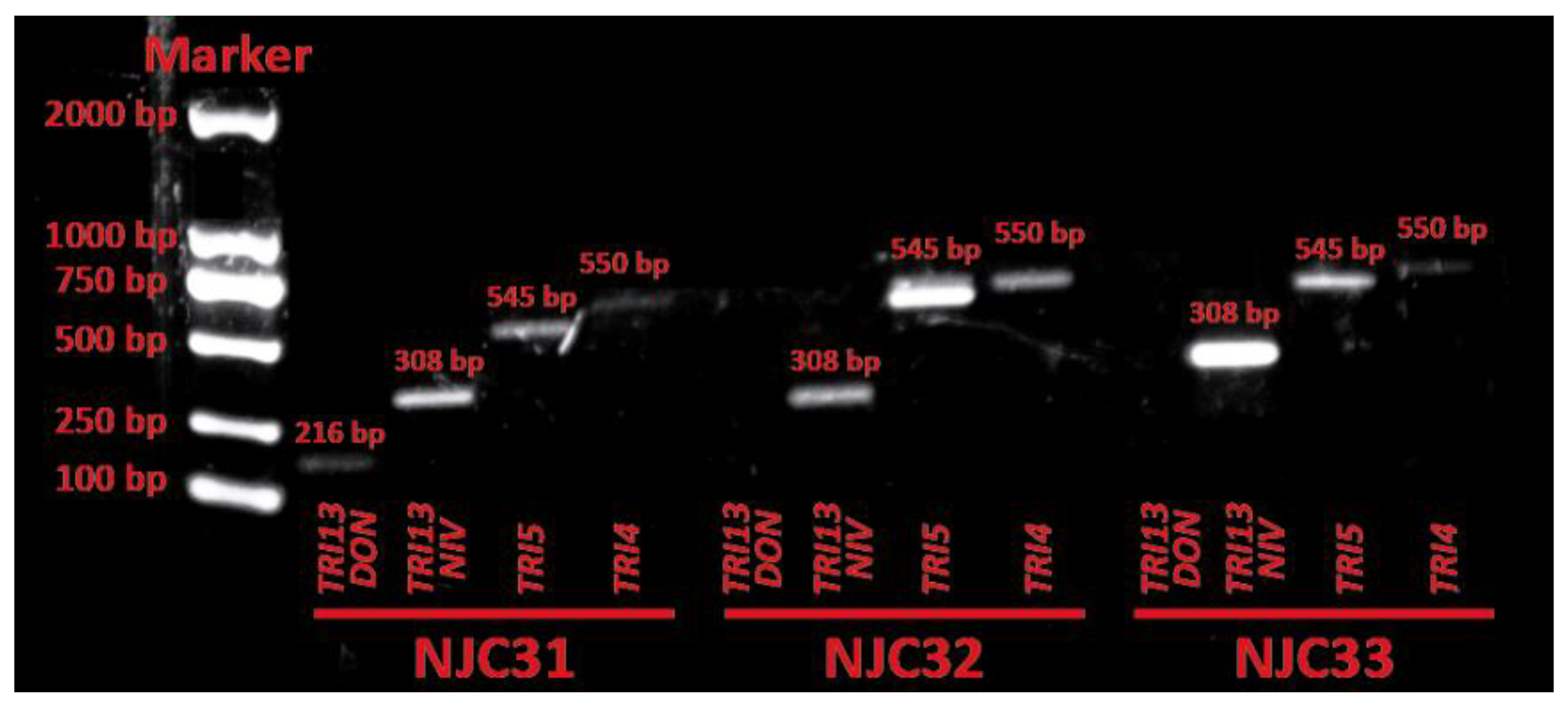
The ability of NJC31, NJC32 and NJC33 to synthesize trichothecenes was evaluated by amplifying the trichothecene biosynthesis genes TRI4, TRI5, TRI13NIV and TRI13DON. The presence of the TRI4 and TRI5 genes is specific for A-trichothecene producers, while the TRI13NIV and TRI13DON genes are involved in the biosynthesis of B-type trichothecenes nivalenol and deoxynivalenol, respectively [32,33]. The product sizes predicted for F. equiseti were used to identify tentatively the PCR products from NJC31, NJC32 and NJC33 [32]. The PCR assay showed the presence of TRI4, TRI5, TRI13NIV and TRI13DON in the DNA of NJC31, while TRI4, TRI5 and TRI13NIV were detected in NJC32 and NJC33 (Figure 5). These results suggest that NJC31 can synthesize A-type trichothecenes, nivalenol and deoxynivalenol, while NJC32 and NJC33 seem to produce only A-type trichothecenes and nivalenol. Unfortunately, no trichothecene biosynthesis genes were obtained in sufficient concentration to allow for sequencing.
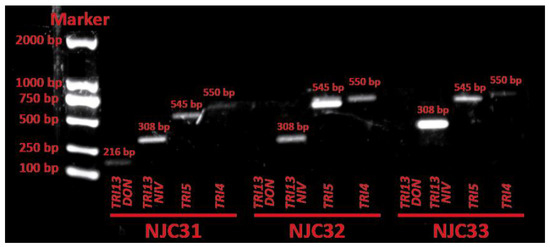
Figure 5.
Electrophoresis analysis of trichothecene biosynthetic genes TRI13DON, TRI13NIV, TRI5 and TRI4 in Fusarium sulawense strains NJC31, NJC32 and NJC33. The product sizes predicted for Fusarium equiseti were used to identify tentatively the PCR products [32].
3.2. Combinations of Carbendazim, Dipicolinic Acid and Kojic Acid Reduced the Symptoms of F. sulawense on Soybean Pods
The preventive and curative efficacies of carbendazim, dipicolinic acid and kojic acid were evaluated by applying the compounds separately and in combination. For the preventive assay, the soybean pods were treated with the fungicides, and then F. sulawense was inoculated. For the curative assay, F. sulawense was inoculated on the soybean pods. After the appearance of the disease symptoms, the antifungal compounds were applied. The curative and preventive assays were performed using NJC31.
Although carbendazim, dipicolinic acid and kojic acid showed preventive efficacy when they were applied separately, none of the fungicides showed curative efficacy. The highest preventive efficacy was observed when using carbendazim (76.2% lesion length inhibition) (Table 1 and Figure 6). In contrast, dipicolinic acid (33.3% lesion length inhibition) and kojic acid (28.6% lesion length inhibition) showed weak preventive efficacies.

Table 1.
Efficacy of carbendazim, dipicolinic acid and kojic acid against Fusarium sulawense on soybean pods.

Figure 6.
Images showing the efficacy of carbendazim, dipicolinic acid and kojic acid for the control of Fusarium sulawense NJC31 symptoms on soybean pods. (A) Symptoms after treatment with carbendazim, dipicolinic acid and kojic acid separately. (B) Symptoms after applying combinations of carbendazim, dipicolinic acid and kojic acid. (C) Symptoms after preventive application with carbendazim and kojic acid. The pathogen was inoculated at different time points after treatment with the fungicides. Control experiments were carried out in the absence of fungicides.
To continue the screening, carbendazim was combined with dipicolinic and kojic acids (Table 2 and Figure 6). Although none of the fungicides showed curative efficacy when they were applied separately, the combinations of carbendazim and dipicolinic acid reduced the lesion length caused by F. sulawense (although the means were not significantly different). In contrast, the combinations of carbendazim and kojic acid were not effective for controlling the pathogen, while the application of 0.84 mg/mL dipicolinic acid and 0.71 mg/mL kojic acid reduced the lesion length by 33.3%.

Table 2.
Efficacy of carbendazim, dipicolinic acid and kojic acid combinations against Fusarium sulawense on soybean pods.
In preventive applications, the combination of 3.8 mg/mL carbendazim and 0.84 mg/mL dipicolinic acid (82.7% lesion length inhibition) slightly increased the efficacy when compared to the single application of carbendazim (76.2% lesion length inhibition). The highest efficacy was detected when using 1.9 mg/mL carbendazim and 0.71 mg/mL kojic acid (84.9% lesion length inhibition). The concentration of carbendazim could be reduced by combining this fungicide with dipicolinic and kojic acids without an obvious decrease in antifungal activity.
Combinations of 1.9 mg/mL carbendazim and 0.71 mg/mL kojic acid could protect soybean pods from F. sulawense infection for 7 days without any obvious decrease in efficacy (Table 3 and Figure 6). The inhibitory activity ranged from 76.2% to 81.7% during the 7 days of study. These results indicate that the combination of 1.9 mg/mL carbendazim and 0.71 mg/mL kojic acid can be a suitable mixture to provide extended protection of soybean pods. In contrast, the combination of 0.95 mg/mL carbendazim and 0.71 mg/mL kojic acid provided limited protection, with only 50.8% and 26.9% inhibitory activities after 4 and 6 days, respectively.

Table 3.
Preventive efficacy of carbendazim and kojic acid combinations for the control of Fusarium sulawense on soybean pods at different time points.
4. Discussion
F. sulawense is a novel Fusarium species that was designated in 2019 [30,38]. F. sulawense was isolated for the first time on bananas in Indonesia in 2014, and it has been indicated that this species is endemic to this crop [30]. This species belongs to the F. incarnatum-equiseti complex, which is a phylogenetically rich complex that contains more than 30 pathogenic species [39]. F. sulawense has been reported to infect numerous crops in tropical and equatorial areas. For example, it was reported to cause rot on melon fruits in Brazil in 2020 [40]. F. sulawense was also found to cause rot on papaya, leaf spot on mango and blight on plums in China [41,42,43]. This species was identified as the causal agent of malformations on Swietenia macrophylla and head blight of wheat in Mexico [25,27]. F. sulawense was discovered to be the cause of sesame stem rot in Pakistan [44], and rice sheath rot in Indonesia [45]. As indicated in the Introduction Section, F. sulawense was reported to cause soybean pod blight in Hubei, China, in July–August 2020 [30]. In agreement with the symptoms observed in Hubei, F. sulawense caused black-brown necrotic lesions on soybean pods in Nantong municipality, and the pods were heavily mildewed. Hubei and Jiangsu are located in subtropical areas on the Yangtze River, confirming the spread of F. sulawense to higher latitudes. This study suggests that F. sulawense may become an important soybean pathogen across China in the coming years. All spreads caused by F. sulawense in China, Brazil and Mexico were reported from 2020 to date, suggesting that this is a new pathogen in these countries and has spread from Indonesia to other parts of the world. According to the reports, this species can only be found in certain plants but does not have a cosmopolitan distribution. F. graminearum, F. pseudograminearum and F. meridionale have also been identified as the causal agents of soybean pod blight in Argentina and Canada [12,46]. Some Diaporthe species, such as D. aspalathi, D. sojae and D. longicolla, have been reported to be among the main pathogens producing soybean pod blight in Canada and Brazil [47,48,49].
Morphology analyses confirmed the presence of microconidia, fusiform macroconidia, septate mycelium and chlamydospores, which are consistent with the common morphology of Fusarium species [29,50,51]. Maryani et al. analyzed the morphological characteristics of the F. sulawense ex-type strain InaCC F964 [30]. This strain also formed white mycelium with yellow pigmentation on PDA medium, and showed very similar structures, such as 10–15 μm conidia and elongated chlamydospores. The number of septum separations in the conidia was similar, 3-6 septum separations, and in both cases the conidia showed a curved shape. Although the morphology analysis indicated that the strains isolated in this study showed very similar morphology to F. sulawense ex-type InaCC F964, this analysis could not confirm the specific species given that most Fusarium species show similar morphological characteristics. For this reason, a sequencing analysis was carried out. This analysis was performed by sequencing the ITS, EF1-α and RPB2 genes, which are common genes used for the identification of Fusarium species [52,53].
Zhao et al. indicated that the identified strains from the F. fujikuroi species complex showed higher virulence on soybean pods than the strains from the F. incarnatum-equiseti species complex [21]. In that study, the disease incidence ranged from 63.3% to 80%, which is similar to the disease incidence observed in this study (69%). Although F. proliferatum 39 YZU 201408 was reported to exhibit high virulence with 72.2–90.0% disease severity, the virulence of the F. sulawense strains isolated in Hubei was under 50% [21]. The F. sulawense strains isolated in this work showed higher virulence (75.1–88.4% disease severity) compared to the F. sulawense strains isolated in Hubei; however, both assays were not carried out using the same soybean variety. Further research is necessary to examine and compare the virulence of different Fusarium species on soybean pods. In this report, the three isolates were F. sulawense strains, suggesting that F. sulawense is the main agent causing soybean pod blight symptoms in Nantong. However, it would be interesting to extend the screening to more fields and plants in the area to identify all the pathogens and evaluate the spread of this disease in Jiangsu Province.
F. incarnatum-equiseti species are known to synthesize mycotoxins, such as zearalenone and trichothecene [22,25,54]. Although isolates NJC31, NJC32 and NJC33 were found to contain several trichothecene biosynthesis genes, further chemical analyses are necessary to confirm the presence of trichothecenes in the secretions of and their structures. The ability of F. sulawense to produce trichothecenes was previously reported by Montoya-Martinez et al. [25]. As far as we know, F. sulawense has never been reported to synthesize zearalenone. However, further studies are necessary to confirm this chemotype. Trichothecenes are also synthetized by F. graminearum and are commonly found in cereal grains [55]. Although A-type trichothecenes are more toxic to mammals than B-type trichothecenes, B-type trichothecenes are more common [56]. Trichothecenes can produce hypoxia, which in turn promotes oxidative stress in human cells [57]. The toxic effects of trichothecenes are stronger in growing children than in adults [58]. The obtained results suggest that the F. sulawense strains isolated in Nantong can produce both A-type and B-type trichothecenes, indicating that the presence of these F. sulawense isolates on soybean pods may pose important health hazards. For this reason, the occurrence of F. sulawense on soybean must be carefully monitored and controlled.
Carbendazim, which shows a mode of action based on the inhibition of microtubule polymerization in fungal cells [59], is a commercial fungicide commonly used for the management of soybean diseases [60]. Despite carbendazim’s broad range of antifungal activity and wide use, the production and use of carbendazim have been restricted in several countries due to its toxic effects [61]. Carbendazim has been related to major human diseases, such as cancer [62], and is known to produce embryotoxicity, germ cell apoptosis, teratogenesis, infertility, hepatocellular dysfunction, hematopoiesis and developmental toxicity in different mammalian species [63]. For these reasons, methods to decrease the use of this fungicide are urgently needed. On the other hand, dipicolinic and kojic acids are experimental compounds and their antifungal activities have been screened against different fungal plant pathogens [35,36,64]. As far as we know, no toxic effects were reported for these compounds, which is an important advantage compared to carbendazim. Dipicolinic and kojic acids are unexpensive (dipicolinic acid ≈ 1.3 EUR/g; koijc acid ≈ 0.3 EUR/g), showing similar price in comparison to carbendazim (≈ 0.9 EUR/g), and are produced in large scale. Kojic acid has been reported to show higher stability than dipicolinic acid and carbendazim in pear fruit [64]. In contrast with carbendazim, which is produced by chemical synthesis, dipicolinic and kojic acids are natural secondary metabolites. Dipicolinic acid is produced by Bacillus species and is a common component of bacterial spores [65], while kojic acid is synthesized by some Aspergillus strains and is widely used in the cosmetic industry to protect skin from UV radiation [66]. Dipiconilic acid was reported to inhibit chitin synthesis in Valsa pyri [35], whereas kojic acid was demonstrated to block the biosynthesis of melanin in Sclerotinia sclerotiorum [36].
All antifungal compounds showed higher preventive than curative efficacies. Separately, none of the compounds could reduce the disease symptoms in the curative assay. This highlights the difficulty in controlling F. sulawense after observing rot symptoms. Although none of the compounds showed any curative efficacy against the pathogen, the combination of dipicolinic acid and carbendazim reduced soybean pod blight symptoms. Interestingly, the combination of dipicolinic acid and carbendazim enhanced the curative and preventive efficacies of the commercial fungicide. These results suggest that dipicolinic acid can be a suitable complement to reduce the concentration and use of carbendazim. These results can be explained considering that dipicolinic acid is capable of damaging F. sulawense cell wall, which may facilitate the entry of carbendazim inside the pathogen cells [67]. As far as we know, this is the first study regarding the combination of carbendazim and dipicolinic acid for the control of fungal diseases. Previously, it was observed that combining carbendazim and kojic acid enhanced the ability of the commercial fungicide to control S. sclerotiorum on soybean pods [36].
Interestingly, combinations of carbendazim and kojic acid showed high protection ability inhibiting the pathogen’s advancement for 7 days. This highlights the potential application of this combination to control F. sulawense on soybean pods. It must be noted that the maturation of soybean pods requires approximately three weeks, and during this time, the pods are highly susceptible to fungal pathogen infections [68,69].
As far as we know, this is the first method for the management of F. sulawense. Only one study regarding the management of Fusarium species on soybean was reported to date and involved the use of captan, fludioxonil, azoxystrobin, trifloxystrobin and pyraclostrobin for the control of F. graminearum on soybean [70]. The highest efficacies were observed when using captan and fludioxonil. In contrast with the previous report, this study also involved the use of natural compounds.
5. Conclusions
In summary, new F. sulawense strains were identified in Nantong municipality as the causal agents of soybean pod rot. Given that, in 2020, F. sulawense was also found causing rot on soybean pods cultivated in Hubei, this new report suggests that F. sulawense may become a common soybean pathogen in China over the next few years. Several biosynthesis genes were amplified in the F. sulawense isolates, suggesting that they may be able to produce A-type and B-type trichothecenes and, for this reason, can pose an important safety hazard. Carbendazim, dipicolinic acid and kojic acid were screened for the control of F. sulawense on soybean, revealing that combinations of carbendazim and dipicolinic acid, and carbendazim and kojic acid, are a suitable method to control this pathogen. Although combinations of carbendazim and kojic acid only showed preventive efficacy, combinations of carbendazim and dipicolinic acid showed both preventive and curative abilities. This is the first study regarding the combination of carbendazim and dipicolinic acid. The obtained results suggest that dipicolinic and kojic acids can be suitable complements to reduce the use of carbendazim. Collectively, this report will lead to a better understanding of the potential safety hazards found in soybean products in China and reveals the potential use of natural metabolites dipicolinic and kojic acids for the control of Fusarium species in soybean.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijerph191710531/s1, Table S1: Sequences of the Fusarium sulawense strains isolated from soybean pods in China, Figure S1: Agarose gel showing the amplicons of ITS, EF1-α, RPB2 genes.
Author Contributions
Conceptualization, X.-C.S. and P.L.; methodology, Q.S., S.-L.Z., Y.-J.X., M.-T.X. and X.C.; formal analysis, Q.S., S.-L.Z., Y.-J.X. and M.-T.X.; investigation, Q.S., S.-L.Z., Y.-J.X. and M.-T.X.; resources, S.-Y.W., X.-C.S. and P.L.; data curation, D.D.H.-B.; writing—original draft preparation, P.L.; writing—review and editing, D.D.H.-B.; visualization, P.L.; supervision, P.L.; project administration, S.-Y.W.; funding acquisition, S.-Y.W., X.-C.S. and P.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China, grant number 32172441, the Nantong Applied Research Program, grant number JC2020103, the Rural Revitalization Science and Technology Project of Shaanxi Province, grant number 2022FP-01, the Social and Livelihood Project of Nantong, grant number MS12020069, and the Large Instruments Open Foundation of Nantong University, grant numbers KFJN2130, KFJN2135 and KFJN2233.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Food and Agriculture Organization of the United Nations (FAO). 2020. Available online: http://www.fao.org/faostat/en/#data/QCL/visualize (accessed on 1 May 2022).
- Wang, S.Y.; Zhang, Y.J.; Zhu, G.Y.; Shi, X.C.; Chen, X.; Herrera-Balandrano, D.D.; Liu, F.Q.; Laborda, P. Occurrence of isoflavones in soybean sprouts and strategies to enhance their content: A review. J. Food Sci. 2022, 87, 1961–1982. [Google Scholar] [CrossRef]
- Jayachandran, M.; Xu, B.J. An insight into the health benefits of fermented soy products. Food Chem. 2019, 271, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Bandara, A.Y.; Weerasooriya, D.K.; Bradley, C.A.; Allen, T.W.; Esker, P.D. Dissecting the economic impact of soybean diseases in the United States over two decades. PLoS ONE 2020, 15, e0231141. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.M.; Kandoth, P.K.; Warren, S.D.; Yeckel, G.; Heinz, R.; Alden, J.; Yang, C.L.; Jamai, A.; El-Mellouki, T.; Juvale, P.S.; et al. A soybean cyst nematode resistance gene points to a new mechanism of plant resistance to pathogens. Nature 2012, 492, 256. [Google Scholar] [CrossRef]
- Ma, Z.C.; Zhu, L.; Song, T.Q.; Wang, Y.; Zhang, Q.; Xia, Y.Q.; Qiu, M.; Lin, Y.C.; Li, H.Y.; Kong, L.; et al. A paralogous decoy protects Phytophthora sojae apoplastic effector PsXEG1 from a host inhibitor. Science 2017, 355, 710–714. [Google Scholar] [CrossRef] [PubMed]
- Peltier, A.J.; Bradley, C.A.; Chilvers, M.I.; Malvick, D.K.; Mueller, D.S.; Wise, K.A.; Esker, P.D. Biology, yield loss and control of Sclerotinia stem rot of soybean. J. Integr. Pest Manag. 2012, 3, B1–B7. [Google Scholar] [CrossRef] [Green Version]
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P. The global burden of pathogens and pests on major food crops. Nat. Ecol. Evol. 2019, 3, 430. [Google Scholar] [CrossRef] [PubMed]
- Hui, Y.; Nelson, B. Effects of soil type, temperature and moisture on development of Fusarium root rot of soybean by Fusarium solani (FSSC 11) and Fusarium tricinctum. Plant Dis. 2022. [Google Scholar] [CrossRef]
- Liu, B.; Wei, H.H.; Shen, W.S.; Smith, H. Long-term effect of non-irrigation and irrigation on soil Pythium, Fusarium, and Rhizoctonia communities and their relation with seed-rot, root-rot, and damping-off of soybean. Eur. J. Plant Pathol. 2020, 158, 297–314. [Google Scholar] [CrossRef]
- Zheng, N.; Zhang, L.P.; Ge, F.Y.; Huang, W.K.; Kong, L.A.; Peng, D.L.; Liu, S.M. Conidia of one Fusarium solani isolate from a soybean-production field enable to be virulent to soybean and make soybean seedlings wilted. J. Integr. Agric. 2018, 17, 2042–2053. [Google Scholar] [CrossRef]
- Chiotta, M.L.; Zanon, M.S.A.; Palazzini, J.M.; Scandiani, M.M.; Formento, A.N.; Barros, G.G.; Chulze, S.N. Pathogenicity of Fusarium graminearum and F. meridionale on soybean pod blight and trichothecene accumulation. Plant Pathol. 2016, 65, 1492–1497. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.G.; Jiang, W.Y.; Li, S.; Jiang, D.; Wang, R.T.; Ji, P.; Pan, H.Y. Occurrence of cotyledon spot on soybean caused by Fusarium oxysporum in China. Plant Dis. 2019, 103, 2967. [Google Scholar] [CrossRef]
- Cruz, D.R.; Leandro, L.F.S.; Munkvold, G.P. Effects of temperature and pH on Fusarium oxysporum and soybean seedling disease. Plant Dis. 2019, 103, 3234–3243. [Google Scholar] [CrossRef]
- Chang, K.F.; Hwang, S.F.; Conner, R.L.; Ahmed, H.U.; Zhou, Q.; Turnbull, G.D.; Strelkov, S.E.; McLaren, D.L.; Gossen, B.D. First report of Fusarium proliferatum causing root rot in soybean (Glycine max L.) in Canada. Crop Prot. 2015, 67, 52–58. [Google Scholar] [CrossRef]
- Chang, X.L.; Li, H.J.; Naeem, M.; Wu, X.L.; Yong, T.W.; Song, C.; Liu, T.G.; Chen, W.Q.; Yang, W.Y. Diversity of the seedborne fungi and pathogenicity of Fusarium species associated with intercropped soybean. Pathogens 2020, 9, 531. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.L.; Dai, H.; Wang, D.P.; Zhou, H.H.; He, W.Q.; Fu, Y.; Ibrahim, F.; Zhou, Y.; Gong, G.S.; Shang, J.; et al. Identification of Fusarium species associated with soybean root rot in Sichuan Province, China. Eur. J. Plant Pathol. 2018, 151, 563–577. [Google Scholar] [CrossRef]
- Pimentel, M.F.; Arnao, E.; Warner, A.J.; Subedi, A.; Rocha, L.F.; Srour, A.; Bond, J.; Fakhoury, A.M. Trichoderma isolates inhibit Fusarium virguliforme growth, reduce root rot, and induce defense-related genes on soybean seedlings. Plant Dis. 2020, 104, 1949–1959. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.X.; Li, N.N.; Chang, K.F.; Hwang, S.F.; Strelkov, S.E.; Conner, R.L.; McLaren, D.L.; Fu, H.T.; Harding, M.W.; Turnbull, G.D. Genetic diversity and aggressiveness of Fusarium species isolated from soybean in Alberta, Canada. Crop Prot. 2018, 105, 49–58. [Google Scholar] [CrossRef]
- Zelechowski, M.; Molcan, T.; Bilska, K.; Myszczynski, K.; Olszewski, J.; Karpiesiuk, K.; Wyrebek, J.; Kulik, T. Patterns of diversity of Fusarium fungi contaminating soybean grains. Toxins 2022, 13, 884. [Google Scholar] [CrossRef]
- Zhao, L.; Wei, X.; Zheng, T.; Gou, Y.N.; Wang, J.; Deng, J.X.; Li, M.J. Evaluation of pathogenic Fusarium spp. associated with soybean seed (Glycine max L.) in Hubei Province, China. Plant Dis. 2022, in press. [Google Scholar] [CrossRef]
- Lu, Y.A.; Qiu, J.B.; Wang, S.F.; Xu, J.H.; Ma, G.Z.; Shi, J.R.; Bao, Z.H. Species diversity and toxigenic potential of Fusarium incarnatum-equiseti species complex isolates from rice and soybean in China. Plant Dis. 2021, 105, 2628–2636. [Google Scholar] [CrossRef] [PubMed]
- Pierzgalski, A.; Bryla, M.; Kanabus, J.; Modrzewska, M.; Podolska, G. Updated review of the toxicity of selected Fusarium toxins and their modified forms. Toxins 2021, 13, 768. [Google Scholar] [CrossRef] [PubMed]
- Kamle, M.; Mahato, D.K.; Devi, S.; Lee, K.E.; Kang, S.G.; Kumar, P. Fumonisins: Impact on agriculture, food, and human health and their management strategies. Toxins 2019, 11, 328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montoya-Martinez, A.C.; O’Donnell, K.; Busman, M.; Vaughan, M.M.; McCormick, S.P.; Santillan-Mendoza, R.; Pineda-Vaca, D.; Clapes-Garduno, L.; Fernandez-Pavia, S.P.; Ploetz, R.C.; et al. Weeds harbor Fusarium species that cause malformation disease of economically important trees in Western Mexico. Plant Dis. 2022, 106, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Su, Y.Y.; Cai, L. An optimized protocol of single spore isolation for fungi. Cryptogam. Mycol. 2013, 34, 349–356. [Google Scholar] [CrossRef]
- Leyva-Mir, S.G.; Garcia-Leon, E.; Camacho-Tapia, M.; Villasenor-Mir, H.E.; Leyva-Madrigal, K.Y.; Mora-Romero, G.A.; Tovar-Pedraza, J.M. Occurrence of the Fusarium incarnatum-equiseti species complex causing Fusarium head blight of wheat in Mexico. Plant Dis. 2022, in press. [Google Scholar] [CrossRef]
- Sunpapao, A.; Suwannarach, N.; Kumla, J.; Dumhai, R.; Riangwong, K.; Sanguansub, S.; Wanchana, S.; Arikit, S. Morphological and molecular identification of plant pathogenic fungi associated with dirty panicle disease in coconuts (Cocos nucifera) in Thailand. J. Fungi 2022, 8, 335. [Google Scholar] [CrossRef]
- Xie, Y.; Sun, Q.; Zhang, S.; Shi, X.; Herrera-Balandrano, D.D.; Wang, S.; Laborda, P. First report of Fusarium acuminatum causing leaf blight on garlic in China. Plant Dis. 2022, in press. [Google Scholar] [CrossRef]
- Maryani, N.; Sandoval-Denis, M.; Lombard, L.; Crous, P.W.; Kema, G.H.J. New endemic Fusarium species hitch-hiking with pathogenic Fusarium strains causing Panama disease in small-holder banana plots in Indonesia. Persoonia 2019, 43, 48–69. [Google Scholar] [CrossRef] [Green Version]
- Xiao, X.E.; Wang, W.; Crous, P.W.; Wang, H.K.; Jiao, C.; Huang, F.; Pu, Z.X.; Zhu, Z.R.; Li, H.Y. Species of Botryosphaeriaceae associated with citrus branch diseases in China. Persoonia 2021, 47, 106–135. [Google Scholar] [CrossRef]
- Stepien, L.; Gromadzka, K.; Chelkowski, J. Polymorphism of mycotoxin biosynthetic genes among Fusarium equiseti isolates from Italy and Poland. J. Appl. Genet. 2012, 53, 227–236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nicholson, P.; Simpson, D.R.; Wilson, A.H.; Chandler, E.; Thomsett, M. Detection and differentiation of trichothecene and enniatin-producing Fusarium species on small-grain cereals. Eur. J. Plant Pathol. 2004, 110, 503–514. [Google Scholar] [CrossRef]
- Tonu Agrotech Ltd. 2022. Available online: http://www.tonuagrotech.com/products_info/Carbendazim-50--SC-363774.html (accessed on 1 May 2022).
- Song, X.G.; Han, M.H.; He, F.; Wang, S.Y.; Li, C.H.; Wu, G.C.; Huang, Z.G.; Liu, D.; Liu, F.Q.; Laborda, P.; et al. Antifungal mechanism of dipicolinic acid and its efficacy for the biocontrol of pear Valsa canker. Front. Microbiol. 2020, 11, 958. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.Y.; Shi, X.C.; Wang, S.Y.; Wang, B.; Laborda, P. Antifungal mechanism and efficacy of kojic acid for the control of Sclerotinia sclerotiorum in soybean. Front. Plant Sci. 2022, 13, 845698. [Google Scholar] [CrossRef]
- Wang, M.M.; Chen, Q.; Diao, Y.Z.; Duan, W.J.; Cai, L. Fusarium incarnatum-equiseti complex from China. Persoonia 2019, 43, 70–89. [Google Scholar] [CrossRef] [Green Version]
- Xia, J.W.; Sandoval-Denis, M.; Crous, P.W.; Zhang, X.G.; Lombard, L. Numbers to names—Restyling the Fusarium incarnatum-equiseti species complex. Persoonia 2019, 43, 186–221. [Google Scholar] [CrossRef]
- Jedidi, I.; Jurado, M.; Cruz, A.; Trabelsi, M.M.; Said, S.; Gonzalez-Jaen, M.T. Phylogenetic analysis and growth profiles of Fusarium incarnatum-equiseti species complex strains isolated from Tunisian cereals. Int. J. Food Microbiol. 2021, 353, 109297. [Google Scholar] [CrossRef]
- Lima, E.N.; Oster, A.H.; Bordallo, P.N.; Araujo, A.A.C.; Silva, D.E.M.; Lima, C.S. A novel lineage in the Fusarium incarnatum-equisetis pecies complex is one of the causal agents of fusarium rot on melon fruits in Northeast Brazil. Plant Pathol. 2021, 70, 133–143. [Google Scholar] [CrossRef]
- Yi, R.H.; Lian, T.; Su, J.J.; Chen, J. First report of internal black rot on Carica papaya fruit caused by Fusarium sulawesiense in China. Plant Dis. 2022, 106, 319. [Google Scholar] [CrossRef]
- Lu, M.; Zhang, Y.; Li, Q.; Huang, S.; Tang, L.; Chen, X.; Guo, T.; Mo, J.; Ma, L. First report of leaf blight caused by Fusarium pernambucanum and Fusarium sulawesiense on plum in Sichuan, China. Plant Dis. 2022, in press. [Google Scholar] [CrossRef]
- Guo, Z.N.; Yu, Z.H.; Li, Q.L.; Tang, L.H.; Guo, T.X.; Huang, S.P.; Mo, J.Y.; Hsiang, T.; Luo, S.M. Fusarium species associated with leaf spots of mango in China. Microb. Pathog. 2021, 150, 104736. [Google Scholar] [CrossRef] [PubMed]
- Kamran, M.; Ullah, I.; Ul Haq, M.E.; Iqba, M.; Abbas, H.; Nazir, S.; Ali, S.; Khan, Q.A.T.; Idrees, M. First report of Fusarium sulawesiensis (FIESC-16) associated with stem rot of sesame in Pakistan. Plant Dis. 2020, 104, 1554. [Google Scholar] [CrossRef]
- Pramunadipta, S.; Widiastuti, A.; Wibowo, A.; Suga, H.; Priyatmojo, A. Identification and pathogenicity of Fusarium spp. associated with the sheath rot disease of rice (Oryza sativa) in Indonesia. J. Plant Pathol. 2022, 104, 251–267. [Google Scholar] [CrossRef]
- Xue, A.G.; Cober, E.; Voldeng, H.D.; Babcock, C.; Clear, R.M. Evaluation of the pathogenicity of Fusarium graminearum and Fusarium pseudograminearum on soybean seedlings under controlled conditions. Can. J. Plant Pathol. 2007, 29, 35–40. [Google Scholar] [CrossRef]
- Abdelmagid, A.; Hafez, M.; Lawley, Y.; Rehal, P.K.; Daayf, F. First report of pod and stem blight and seed decay caused by Diaporthe longicolla on soybean in Western Canada. Plant Dis. 2022, 106, 1061. [Google Scholar] [CrossRef]
- Udayanga, D.; Castlebury, L.A.; Rossman, A.Y.; Chukeatirote, E.; Hyde, K.D. The Diaporthe sojae species complex: Phylogenetic re-assessment of pathogens associated with soybean, cucurbits and other field crops. Fungal Biol. 2015, 119, 383–407. [Google Scholar] [CrossRef]
- Brumer, B.B.; Lopes-Caitar, V.S.; Chicowski, A.S.; Beloti, J.D.; Castanho, F.M.; da Silva, D.C.G.; de Carvalho, S.; Lopes, I.O.N.; Soares, R.M.; Seixas, C.D.S.; et al. Morphological and molecular characterization of Diaporthe (anamorph Phomopsis) complex and pathogenicity of Diaporthe aspalathi isolates causing stem canker in soybean. Eur. J. Plant Pathol. 2018, 151, 1009–1025. [Google Scholar] [CrossRef]
- Sun, Q.; Xie, Y.J.; Chen, T.M.; Zhang, J.P.; Laborda, P.; Wang, S.Y. First report of Fusarium avenaceum causing canker disease on kiwi tree in China. Plant Dis. 2022, 106, 1762. [Google Scholar] [CrossRef]
- Crous, P.W.; Lombard, L.; Sandoval-Denis, M.; Seifert, K.A.; Schroers, H.J.; Chaverri, P.; Gene, J.; Guarro, J.; Hirooka, Y.; Bensch, K.; et al. Fusarium: More than a node or a foot-shaped basal cell. Stud. Mycol. 2021, 98, 100116. [Google Scholar] [CrossRef]
- Piacentini, K.C.; Rocha, L.O.; Savi, G.D.; Carnielli-Queiroz, L.; Fontes, L.D.; Correa, B. Assessment of toxigenic Fusarium species and their mycotoxins in brewing barley grains. Toxins 2019, 11, 31. [Google Scholar] [CrossRef] [Green Version]
- Yin, Q.; Shi, X.; Zhu, Z.; Tian, L.; Wang, Y.; Zhao, X.; Jia, Z.; Ma, L. First report of Fusarium solani (FSSC 6) associated with the root rot of Magnolia denudata in China. Plant Dis. 2022, in press. [Google Scholar] [CrossRef] [PubMed]
- Tralamazza, S.M.; Piacentini, K.C.; Savi, G.D.; Carnielli-Queiroz, L.; Fontes, L.D.; Martins, C.S.; Correa, B.; Rocha, L.O. Wild rice (O. latifolia) from natural ecosystems in the Pantanal region of Brazil: Host to Fusarium incarnatum-equiseti species complex and highly contaminated by zearalenone. Int. J. Food Microbiol. 2021, 345, 109127. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Srivastava, S.; Dewangan, J.; Divakar, A.; Rath, S.K. Global occurrence of deoxynivalenol in food commodities and exposure risk assessment in humans in the last decade: A survey. Crit. Rev. Food Sci. Nutr. 2020, 60, 1346–1374. [Google Scholar] [CrossRef] [PubMed]
- Foroud, N.A.; Baines, D.; Gagkaeva, T.Y.; Thakor, N.; Badea, A.; Steiner, B.; Burstmayr, M.; Burstmayr, H. Trichothecenes in cereal grains—An update. Toxins 2019, 11, 634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- You, L.; Zhao, Y.Y.; Kuca, K.; Wang, X.; Oleksak, P.; Chrienova, Z.; Nepovimova, E.; Jacevic, V.; Wu, Q.H.; Wu, W.D. Hypoxia, oxidative stress, and immune evasion: A trinity of the trichothecenes T-2 toxin and deoxynivalenol (DON). Arch. Toxicol. 2021, 95, 1899–1915. [Google Scholar] [CrossRef]
- Maresca, M. From the gut to the brain: Journey and pathophysiological effects of the food-associated trichothecene mycotoxin deoxynivalenol. Toxins 2013, 5, 784–820. [Google Scholar] [CrossRef]
- Rama, E.M.; Bortolan, S.; Vieira, M.L.; Ceccatto Gerardin, D.C.; Moreira, E.G. Reproductive and possible hormonal effects of carbendazim. Regul. Toxicol. Pharmacol. 2014, 69, 476–486. [Google Scholar] [CrossRef]
- Molin, C.; Ribeiro, N.R.; Matusomoto, M.N.; Lutkemeyer, A.J.; Bordignon, K.B.; Ferreira, M.L.B.; Barbieri, M.; Deuner, C.C.; Huzar-Novakowiski, J. Seed treatment for controlling damping-off caused by Globisporangium irregulare and Globisporangium ultimum var. sporangiiferum in soybean from southern Brazil. Crop Prot. 2021, 149, 105782. [Google Scholar]
- Wang, S.Y.; Shi, X.C.; Liu, F.Q.; Laborda, P. Chromatographic methods for detection and quantification of carbendazim in food. J. Agric. Food Chem. 2020, 68, 11880–11894. [Google Scholar] [CrossRef]
- Singh, S.; Singh, N.; Kumar, V.; Datta, S.; Wani, A.B.; Singh, D.; Singh, K.; Singh, J. Toxicity, monitoring and biodegradation of the fungicide carbendazim. Environ. Chem. Lett. 2016, 14, 317–329. [Google Scholar] [CrossRef]
- Lopez-Pacheco, I.Y.; Silva-Nunez, A.; Salinas-Salazar, C.; Arevalo-Gallegos, A.; Lizarazo-Holguin, L.A.; Barcelo, D.; Iqbal, H.M.N.; Parra-Saldivar, R. Anthropogenic contaminants of high concern: Existence in water resources and their adverse effects. Sci. Total Environ. 2019, 690, 1068–1088. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.Y.; Chen, Y.; Wang, S.Y.; Shi, X.C.; Herrera-Balandrano, D.D.; Polo, V.; Laborda, P. Peel diffusion and antifungal efficacy of different fungicides in pear fruit: Structure-diffusion-activity relationships. J. Fungi 2022, 8, 547. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Wang, X.C.; Han, M.H.; Song, X.G.; Yang, D.J.; Wang, S.Y.; Laborda, P.; Shi, X.C. Enhanced spoVF operon increases host attachment and biocontrol ability of Bacillus subtilis for the management of Ceratocystis fimbriata in sweet potato. Biol. Control 2021, 161, 104651. [Google Scholar] [CrossRef]
- Saeedi, M.; Eslamifar, M.; Khezri, K. Kojic acid applications in cosmetic and pharmaceutical preparations. Biomed. Pharmacother. 2019, 110, 582–593. [Google Scholar] [CrossRef]
- Santoso, P.; Minamihata, K.; Ishimine, Y.; Taniguchi, H.; Komada, T.; Sato, R.; Goto, M.; Takashima, T.; Taira, T.; Kamiya, N. Enhancement of the antifungal activity of chitinase by palmitoylation and the synergy of palmitoylated chitinase with amphotericin B. ACS Infect. Dis. 2022, 8, 1051–1061. [Google Scholar] [CrossRef]
- Boufleur, T.R.; Ciampi-Guillardi, M.; Tikami, I.; Rogerio, F.; Thon, M.R.; Sukno, S.A.; Massola, N.S.; Baroncelli, R. Soybean anthracnose caused by Colletotrichum species: Current status and future prospects. Mol. Plant Pathol. 2021, 22, 393–409. [Google Scholar] [CrossRef]
- Molina, J.P.E.; Navarro, B.L.; Allen, T.W.; Godoy, C.V. Soybean target spot caused by Corynespora cassiicola: A resurgent disease in the Americas. Trop. Plant Pathol. 2022, 47, 315–331. [Google Scholar] [CrossRef]
- Ellis, M.L.; Broders, K.D.; Paul, P.A.; Dorrance, A.E. Infection of soybean seed by Fusarium graminearum and effect of seed treatments on disease under controlled conditions. Plant Dis. 2011, 95, 401–407. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).