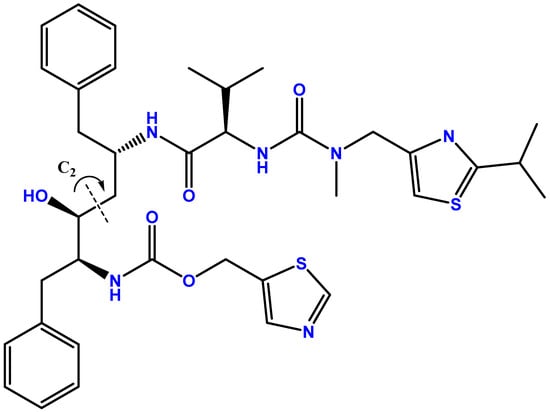
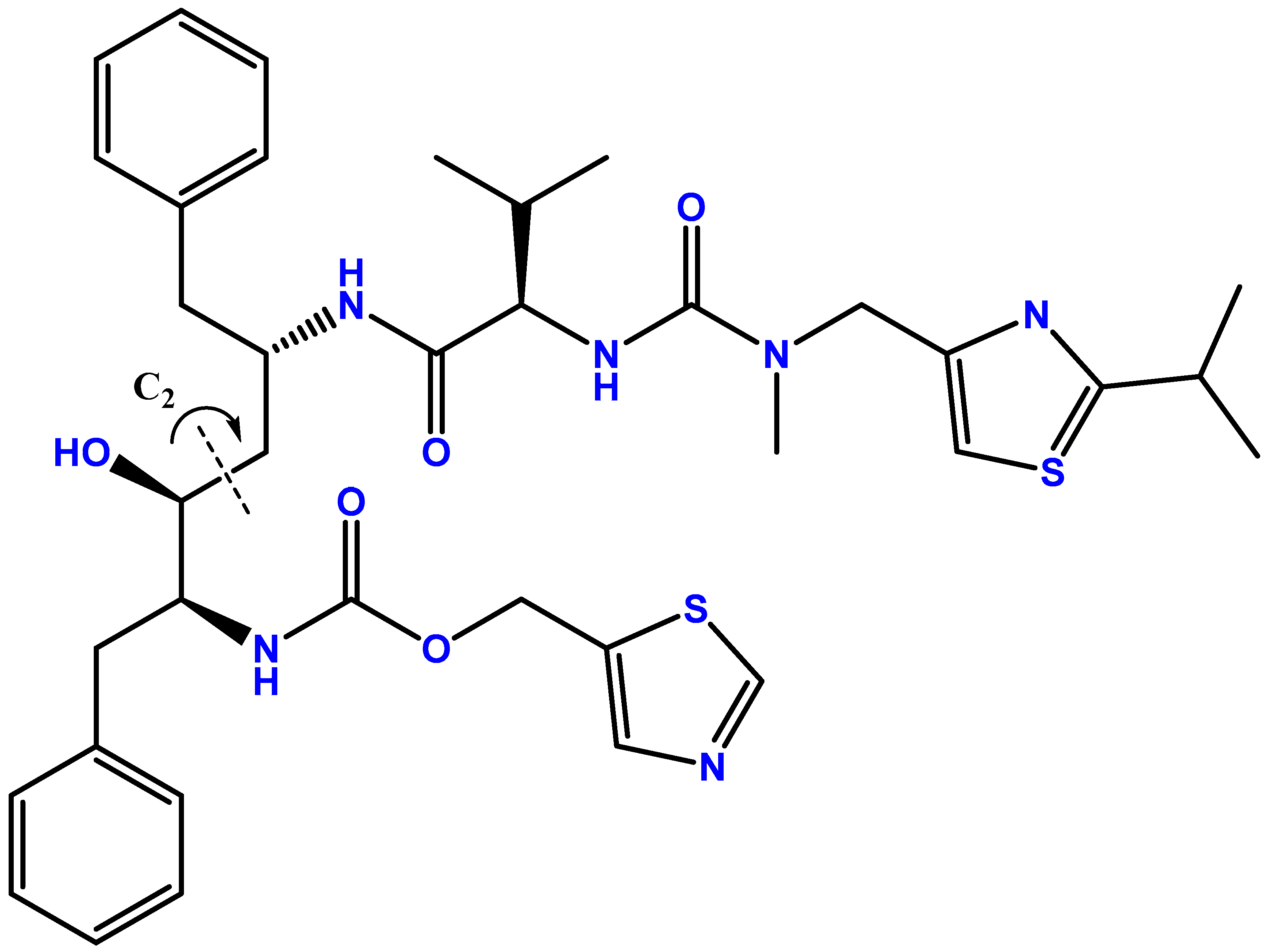
Abstract
Ritonavir is a protease inhibitor initially developed for HIV treatment that is now used as a pharmacokinetic booster for other antiretrovirals due to it being a cytochrome P450 3A4 enzyme and P-glycoprotein inhibitor. Consequently, ritonavir is of special interest for repurposing in other diseases. It had an important role in battling the COVID-19 pandemic as a part of the developed drug Paxlovid® in association with nirmatrelvir and has shown effects in hepatitis and other pathogenic diseases. Ritonavir has also shown promising results in overcoming drug resistance and enhancing the efficacy of existing chemotherapeutic agents in oncology. Evidence of cancer repurposing potential was demonstrated in cancers such as ovarian, prostate, lung, myeloma, breast, and bladder cancer, with several mechanisms of action presented. In vitro studies indicate that ritonavir alone can inhibit key pathways involved in cancer cell survival and proliferation, causing apoptosis, cell cycle arrest, endoplasmic reticulum stress, and metabolic stress due to the inhibition of molecules like heat shock protein 90 and cyclin-dependent kinases. Ritonavir also causes resistant cells to become sensitized to anticancer drugs like gemcitabine or docetaxel. These findings indicate that repurposing ritonavir, either on its own or in combination with other medications, could be a promising approach for treating various diseases. This is particularly relevant in cancer therapy, where ritonavir repurposing is the central focus of this review.
1. Introduction
Since its first licensing in 1996, the powerful HIV protease inhibitor ritonavir has been crucial in the treatment of HIV/AIDS [1]. Its function has changed over time from that of a standalone antiviral to that of an essential part of combination treatments, especially because of its capacity to inhibit the cytochrome P450 3A4 enzyme, which raises the effectiveness of other antiretrovirals [2]. Ritonavir’s dual function as a pharmacokinetic enhancer and therapeutic agent has broadened its use beyond the treatment of HIV, with the potential for this drug to be repurposed in the treatment of various illnesses and even some types of cancer. Despite its effectiveness, ritonavir’s intricate pharmacological interactions with different cytochrome enzymes provide substantial difficulties in clinical practice that must be carefully managed to maximize treatment results. To demonstrate the continued development of this adaptable medication, this review will examine the pharmacological profile of ritonavir, its current therapeutic uses, and the possibility of repurposing it to treat cancer and other viral diseases.
3. Current Clinical Uses
Ritonavir, initially approved for use nearly 20 years ago, has seen significant clinical application primarily as part of combination therapy for HIV treatment. Initially, high-dose ritonavir was shown to be potent in reducing plasma HIV RNA levels, using doses of 600 mg twice daily [8]. However, it was quickly learned that long-term monotherapy resulted in the emergence of mutations and HIV-1 strains resistant to ritonavir and other protease inhibitors like indinavir and saquinavir. Consequently, its use in combination with other antiretrovirals is preferred to prevent resistance development [2].
Ritonavir is extensively used as a pharmacokinetic enhancer or “booster” in HIV therapy. Lower doses significantly boost the pharmacokinetic profiles of other protease inhibitors (PIs) by inhibiting the cytochrome P450 (CYP) 3A4 enzyme, leading to higher drug concentrations, reduced pill burden, and simplified dosing regimens. This enhancement is crucial for drugs like saquinavir, atazanavir, darunavir, and others, where ritonavir increases their exposure, efficacy, and clinical effectiveness [9]. When used in combination as an enhancer for other HIV drugs, lower ritonavir doses are used, typically between 100 and 200 mg twice daily [10].
Ritonavir has significant interactions with cytochrome P450 (CYP) enzymes, both as an inhibitor and an inducer, which can impact the metabolism of various drugs. Ritonavir is a potent inhibitor of the CYP3A subfamily, particularly CYP3A4, and, although some theories have been suggested, the specific mechanisms of inhibition are not yet completely understood [11]. This irreversible inhibition is a primary mechanism for its use as a pharmacokinetic enhancer, as it increases the plasma levels of co-administered drugs that are metabolized by CYP3A4. By inhibiting CYP3A4, ritonavir reduces the metabolism of these drugs, thereby enhancing their therapeutic effects and duration [12]. In addition to its inhibitory effects, ritonavir can also induce the expression of several CYP enzymes, though to a lesser extent. These include CYP1A2, CYP2B6, CYP2C9, and CYP2C19. Ritonavir induces these enzymes through activation of the pregnane X receptor (PXR), which increases the transcription of these CYP genes. This induction can lead to increased metabolism of drugs that are substrates of these enzymes, potentially reducing their efficacy [13].
The consequences of CYP inhibition and induction by ritonavir are significant. The inhibition of CYP3A4 results in higher plasma concentrations of drugs metabolized by this enzyme, enhancing their therapeutic effects and potentially increasing the risk of adverse effects. Conversely, the induction of other CYP enzymes can lead to increased metabolism of drugs, potentially reducing their plasma levels and therapeutic efficacy. The dual role of ritonavir as an inhibitor and inducer of CYP enzymes can complicate drug–drug interactions, requiring careful management and dose adjustments of co-administered drugs. Ritonavir’s ability to both inhibit and induce various CYP enzymes necessitates careful consideration of potential drug interactions in clinical practice to ensure optimal therapeutic outcomes and minimize adverse effects [14].
Apart from the effect on CYP enzymes, ritonavir has also been shown to inhibit another important class of compounds: the drug transporters. Ritonavir is known to inhibit P-gp expression and activity, which can alter the pharmacokinetics of co-administered drugs [15]. For example, ritonavir affects the distribution of indinavir and significantly increases the systemic exposure of fexofenadine, a P-gp substrate [16]. Furthermore, ritonavir acts as an inhibitor of breast cancer resistance protein (BCRP), alongside saquinavir and nelfinavir, enhancing intracellular drug concentrations by inhibiting BCRP-mediated efflux. This inhibition is clinically significant, as it can affect the bioavailability of drugs that are substrates for BCRP [17]. OATP1B1, OATP1B3, and OATP2B1 are organic anion-transporting polypeptides that play crucial roles in the hepatic uptake of various endogenous compounds and xenobiotics, including multiple drugs. Ritonavir exhibits strong inhibitory effects on both OATP1B1 and OATP1B3. Additionally, ritonavir effectively inhibits OATP2B1, although the inhibitory effects are substrate-dependent. This illustrates the complexity of ritonavir’s interaction with different OATP isoforms, emphasizing the importance of context in understanding these interactions [18]. Figure 2 shows a visual representation of ritonavir’s inhibition or induction of drug transporters and metabolizers and the consequent effect on co-administered drugs’ concentration and effect.
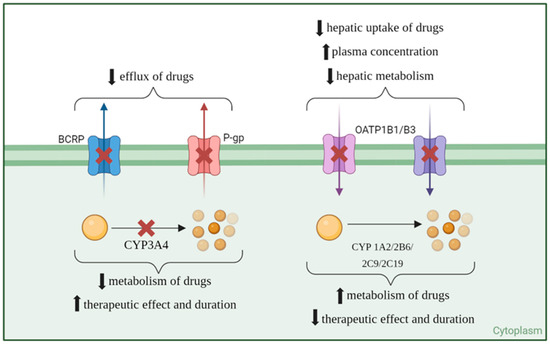
Figure 2.
Inhibition (red cross) and induction (green upvote arrow) of transporters and metabolization enzymes of co-administered drugs (yellow ball) by ritonavir.
4. Potential for Repurposing for Infectious Diseases
4.1. COVID-19
In the context of the COVID-19 pandemic, ritonavir has been combined with nirmatrelvir to form the drug Paxlovid (Pfizer, NY, USA). Nirmatrelvir is a protease inhibitor of the SARS-CoV-2 virus’ 3CL protease, a key enzyme for viral replication [19], while ritonavir boosts its concentration to effective levels. This combination was specifically developed to target SARS-CoV-2, the virus responsible for COVID-19 [20].
Early clinical trials demonstrated that nirmatrelvir/ritonavir (NMV-r) could reduce the risk of hospitalization or death in high-risk, unvaccinated COVID-19 outpatients when administered within five days of symptom onset [21]. As a result of these positive outcomes, regulatory agencies granted emergency use authorizations (EUAs) for Paxlovid for the treatment of COVID-19 in high-risk patients with dosages of 300 mg of nirmatrelvir and 100 mg of ritonavir twice daily [22].
The World Health Organization (WHO) strongly recommended using NMV-r in patients at the highest risk of severe illness or hospitalization due to COVID-19. This includes specific guidance for its use in pregnant and lactating women. However, the WHO did not recommend NMV-r for patients with severe COVID-19, suggesting using other treatments like corticosteroids and interleukin-6 receptor blockers instead. Additionally, it was conditionally recommended for those with non-severe illnesses at the lowest risk of hospitalization [23].
Two studies on the use of NMV-r for COVID-19 treatment in vaccinated patients highlight its effectiveness. The first study found that for adults aged 50 and older or those with underlying health conditions, NMV-r administered within five days of a mild-to-moderate COVID-19 diagnosis provides similar protection against hospitalization as three or more doses of the monovalent mRNA vaccine alone. However, the greatest protection was achieved with both NMV-r treatment and vaccination [24]. The second study, a meta-analysis of seven observational studies with 224,238 vaccinated patients, supports these findings. NMV-r reduced the risk of hospitalization or death in vaccinated patients with mild-to-moderate COVID-19 compared to those who did not receive NMV-r, with greater effectiveness observed in patients aged 50–65 than in those over 65. Despite the protective effect of vaccines, NMV-r still offered significant clinical benefits. Concerns about a viral rebound phenomenon were noted, but evidence suggests this occurs infrequently and with mild symptoms [25].
4.2. Hepatitis
Ritonavir, when used as a booster in combination with other direct-acting antivirals, has shown high effectiveness and safety in treating hepatitis C. A real-world study involving 58 patients with chronic hepatitis or compensated hepatic cirrhosis and genotype 1 HCV infection treated with ritonavir-boosted paritaprevir and ombitasvir for 12 weeks reported a sustained virological response at 24 weeks of 96.6%, with no severe adverse events and only mild-to-moderate adverse events [26]. The combination of ombitasvir/paritaprevir/ritonavir plus dasabuvir, with or without ribavirin, has proven effective in treating HCV genotype 1 infection [27]. Clinical trials, such as the AVIATOR study, provided critical data for optimizing dosages and combinations, resulting in high SVR rates and low relapse rates [28]. Overall, ritonavir-boosted therapies offer a robust option for managing hepatitis C, particularly in patients with genotype 1 and 4 infections, and even in those with complex conditions like renal impairment [29] and post-transplant scenarios [30]. The combination regimens demonstrate high efficacy and manageable safety profiles, both in clinical trials and real-world settings.
Chronic hepatitis E virus (HEV) infection predominantly occurs in immunocompromised patients, such as organ transplant recipients, HIV-infected patients, those receiving anticancer therapy, and those on immunosuppressants [31]. Ritonavir was shown on screening tests to have the potential to inhibit HEV growth [32]. A recent study has found that ritonavir inhibits HEV internalization, a crucial point in the life cycle of this virus. Ritonavir with ribavirin, which inhibits HEV RNA replication, resulted in potent inhibition of HEV growth in both genotypes HEV-3 and HEV-4 in cultured cells, outperforming ribavirin monotherapy. This combination showed additive effects without significant cytotoxicity and decreased HEV RNA levels to undetectable amounts in both culture supernatants and intracellularly [33]. The study concluded that combining ritonavir and ribavirin offers a novel, effective strategy for treating chronic HEV infection, warranting further in vivo studies.
4.3. Toxoplasmosis
Toxoplasmic encephalitis is a severe issue for immunocompromised individuals, especially those with HIV/AIDS, which has a high mortality risk [34]. Lopinavir boosted with ritonavir (L/r) alone and loaded into poly(lactic-co-glycolic acid) (PLGA) nanoparticles was studied in mice infected with the virulent RH strain of T. gondii. Treatment with these formulations significantly reduced mortality rates and parasite numbers in the peritoneal fluid and liver. This effectiveness is likely due to the inhibition of crucial aspartyl protease enzymes in T. gondii, preventing parasite egress from host cells and thereby reducing infection of new cells. The study also found that L/r treatments caused significant morphological changes, suggesting apoptosis and autophagy, which further reduced the parasite’s ability to invade and reproduce [35]. Other pathogenic diseases where ritonavir can be seen boosting the effect of drugs are lymphatic filariasis and onchocerciasis [36], leishmaniasis [37], and Histoplasma capsulatum [38].
Table 1 summarizes the infectious diseases where ritonavir has been shown to be a potential drug for repurposing, along with the effects and modes of action.

Table 1.
Repurposing opportunities of ritonavir in infectious diseases.
6. Conclusions
The goal of this review was to explore the potential repurposing of ritonavir, a well-known HIV protease inhibitor, for the treatment of cancer and other infectious diseases. Ritonavir was first created to treat HIV/AIDS, but it has shown a lot of promise outside of its initial use. This is mainly because of its capacity to inhibit P-glycoprotein and cytochrome P450 3A4, which increases the efficacy of other treatment drugs.
The information gathered here highlights the potential of ritonavir in cancer therapy, as it has demonstrated success in treating a variety of cancer type models, including ovarian, lung, breast, and bladder tumors, as well as in overcoming drug resistance. Research on ritonavir in oncology is justified because of the ways by which it exhibits anticancer effects, including apoptosis induction, cell cycle arrest, and treatment sensitization of cancer cells.
In addition to its oncologic potential, this review also looked at the use of ritonavir in the treatment of various infectious diseases, such as hepatitis virus infection and the COVID-19 pandemic. Ritonavir’s effective inclusion in the COVID-19 Paxlovid treatment regimen is an excellent example of how repurposed drugs may be used to meet critical public health requirements.
Overall, this review highlights the significance of repurposing existing drugs, such as ritonavir, to address novel treatment difficulties. According to the results, ritonavir may prove to be an effective treatment for illnesses and resistant tumors. However, most of these works used in vitro models, and the results need to be validated with preclinical, and afterward—and if promising results are obtained—clinical studies to thoroughly explore this promising treatment option.
Author Contributions
Conceptualization, N.V.; methodology, M.P.; formal analysis, M.P. and N.V.; investigation, M.P.; writing—original draft preparation, M.P.; writing—review and editing, N.V.; supervision, N.V.; project administration, N.V.; funding acquisition, N.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research was financed by Fundo Europeu de Desenvolvimento Regional (FEDER) funds through the COMPETE 2020 Operational Programme for Competitiveness and Internationalisation (POCI), Portugal 2020, and by Portuguese funds through Fundação para a Ciência e a Tecnologia (FCT) in the framework of projects IF/00092/2014/CP1255/CT0004 and CHAIR in Onco-Innovation. FCT for funding M.P. Ph.D. grant (2021.07450.BD).
Data Availability Statement
Not applicable.
Acknowledgments
M.P. acknowledges FCT for funding her Ph.D. grant (2021.07450.BD).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- DrugBank. Ritonavir. Available online: https://go.drugbank.com/drugs/DB00503 (accessed on 9 July 2024).
- Schmit, J.C.; Ruiz, L.; Clotet, B.; Raventos, A.; Tor, J.; Leonard, J.; Desmyter, J.; De Clercq, E.; Vandamme, A.M. Resistance-related mutations in the HIV-1 protease gene of patients treated for 1 year with the protease inhibitor ritonavir (ABT-538). Aids 1996, 10, 995–999. [Google Scholar] [CrossRef]
- Kempf, D.J.; Norbeck, D.W.; Codacovi, L.; Wang, X.C.; Kohlbrenner, W.E.; Wideburg, N.E.; Paul, D.A.; Knigge, M.F.; Vasavanonda, S.; Craig-Kennard, A.; et al. Structure-based, C2 symmetric inhibitors of HIV protease. J. Med. Chem. 1990, 33, 2687–2689. [Google Scholar] [CrossRef] [PubMed]
- Whitesell, J.K. C2 symmetry and asymmetric induction. Chem. Rev. 1989, 89, 1581–1590. [Google Scholar] [CrossRef]
- Hull, M.W.; Montaner, J.S. Ritonavir-boosted protease inhibitors in HIV therapy. Ann. Med. 2011, 43, 375–388. [Google Scholar] [CrossRef]
- FDA. NORVIR (Ritonavir). Available online: www.accessdata.fda.gov/drugsatfda_docs/label/2017/209512lbl.pdf (accessed on 9 July 2024).
- Lledó-García, R.; Nácher, A.; Prats-García, L.; Casabó, V.G.; Merino-Sanjuán, M. Bioavailability and pharmacokinetic model for ritonavir in the rat. J. Pharm. Sci. 2007, 96, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Danner, S.A.; Carr, A.; Leonard, J.M.; Lehman, L.M.; Gudiol, F.; Gonzales, J.; Raventos, A.; Rubio, R.; Bouza, E.; Pintado, V.; et al. A short-term study of the safety, pharmacokinetics, and efficacy of ritonavir, an inhibitor of HIV-1 protease. European-Australian Collaborative Ritonavir Study Group. N. Engl. J. Med. 1995, 333, 1528–1533. [Google Scholar] [CrossRef]
- Larson, K.B.; Wang, K.; Delille, C.; Otofokun, I.; Acosta, E.P. Pharmacokinetic enhancers in HIV therapeutics. Clin. Pharmacokinet. 2014, 53, 865–872. [Google Scholar] [CrossRef]
- EMA. Norvir. Available online: www.ema.europa.eu/en/documents/product-information/norvir-epar-product-information_en.pdf (accessed on 11 July 2024).
- Greenblatt, D.J.; Harmatz, J.S. Ritonavir is the best alternative to ketoconazole as an index inhibitor of cytochrome P450-3A in drug–drug interaction studies. Br. J. Clin. Pharmacol. 2015, 80, 342–350. [Google Scholar] [CrossRef]
- Loos, N.H.C.; Beijnen, J.H.; Schinkel, A.H. The Mechanism-Based Inactivation of CYP3A4 by Ritonavir: What Mechanism? Int. J. Mol. Sci. 2022, 23, 9866. [Google Scholar] [CrossRef]
- Marzolini, C.; Gibbons, S.; Khoo, S.; Back, D. Cobicistat versus ritonavir boosting and differences in the drug–drug interaction profiles with co-medications. J. Antimicrob. Chemother. 2016, 71, 1755–1758. [Google Scholar] [CrossRef]
- Loos, N.H.C.; Beijnen, J.H.; Schinkel, A.H. The inhibitory and inducing effects of ritonavir on hepatic and intestinal CYP3A and other drug-handling proteins. Biomed. Pharmacother. 2023, 162, 114636. [Google Scholar] [CrossRef] [PubMed]
- Drewe, J.; Gutmann, H.; Fricker, G.; Török, M.; Beglinger, C.; Huwyler, J. HIV protease inhibitor ritonavir: A more potent inhibitor of P-glycoprotein than the cyclosporine analog SDZ PSC 833. Biochem. Pharmacol. 1999, 57, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Storch, C.H.; Theile, D.; Lindenmaier, H.; Haefeli, W.E.; Weiss, J. Comparison of the inhibitory activity of anti-HIV drugs on P-glycoprotein. Biochem. Pharmacol. 2007, 73, 1573–1581. [Google Scholar] [CrossRef]
- Gupta, A.; Zhang, Y.; Unadkat, J.D.; Mao, Q. HIV protease inhibitors are inhibitors but not substrates of the human breast cancer resistance protein (BCRP/ABCG2). J. Pharmacol. Exp. Ther. 2004, 310, 334–341. [Google Scholar] [CrossRef]
- Annaert, P.; Ye, Z.W.; Stieger, B.; Augustijns, P. Interaction of HIV protease inhibitors with OATP1B1, 1B3, and 2B1. Xenobiotica 2010, 40, 163–176. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, L.V. Overview of Targets and Potential Drugs of SARS-CoV-2 According to the Viral Replication. J. Proteome Res. 2021, 20, 49–59. [Google Scholar] [CrossRef]
- Reis, S.; Metzendorf, M.I.; Kuehn, R.; Popp, M.; Gagyor, I.; Kranke, P.; Meybohm, P.; Skoetz, N.; Weibel, S. Nirmatrelvir combined with ritonavir for preventing and treating COVID-19. Cochrane Database Syst. Rev. 2023, 11, Cd015395. [Google Scholar] [CrossRef] [PubMed]
- Hammond, J.; Leister-Tebbe, H.; Gardner, A.; Abreu, P.; Bao, W.; Wisemandle, W.; Baniecki, M.; Hendrick, V.M.; Damle, B.; Simón-Campos, A.; et al. Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19. N. Engl. J. Med. 2022, 386, 1397–1408. [Google Scholar] [CrossRef]
- FDA. Fact Sheet for Healthcare Providers: Emergency Use Authorization for Paxlovid. Available online: www.fda.gov/media/155050/download (accessed on 16 July 2024).
- WHO. Therapeutics and COVID-19: Living Guideline, 10 November 2023. Available online: https://app.magicapp.org/#/guideline/nBkO1E (accessed on 16 July 2024).
- Shah, M.M.; Joyce, B.; Plumb, I.D.; Sahakian, S.; Feldstein, L.R.; Barkley, E.; Paccione, M.; Deckert, J.; Sandmann, D.; Hagen, M.B.; et al. Combined Protection of Vaccination and Nirmatrelvir-Ritonavir against Hospitalization in Adults with Coronavirus Disease 2019. Clin. Infect. Dis. 2024, 79, 108–110. [Google Scholar] [CrossRef]
- Li, H.; Xiang, H.; He, B.; Zhang, Q.; Peng, W. Nirmatrelvir plus ritonavir remains effective in vaccinated patients at risk of progression with COVID-19: A systematic review and meta-analysis. J. Glob. Health 2023, 13, 06032. [Google Scholar] [CrossRef]
- Miyasaka, A.; Yoshida, Y.; Yoshida, T.; Murakami, A.; Abe, K.; Ohuchi, K.; Kawakami, T.; Watanabe, D.; Hoshino, T.; Sawara, K.; et al. The Real-world Efficacy and Safety of Ombitasvir/Paritaprevir/Ritonavir for Hepatitis C Genotype 1. Intern. Med. 2018, 57, 2807–2812. [Google Scholar] [CrossRef] [PubMed]
- Flisiak, R.; Flisiak-Jackiewicz, M. Ombitasvir and paritaprevir boosted with ritonavir and combined with dasabuvir for chronic hepatitis C. Expert. Rev. Gastroenterol. Hepatol. 2017, 11, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Poordad, F.; Agarwal, K.; Younes, Z.; Cohen, D.; Xie, W.; Podsadecki, T. Low relapse rate leads to high concordance of sustained virologic response (SVR) at 12 weeks with SVR at 24 weeks after treatment with ABT-450/ritonavir, ombitasvir, and dasabuvir plus ribavirin in subjects with chronic hepatitis C virus genotype 1 infection in the AVIATOR study. Clin. Infect. Dis. 2015, 60, 608–610. [Google Scholar] [CrossRef]
- Abd-Elsalam, S.; Abo-Amer, Y.E.; El-Abgeegy, M.; Elshweikh, S.A.; Elsergany, H.F.; Ahmed, R.; Elkadeem, M.; Hawash, N.; Soliman, S.; Badawi, R.; et al. Efficacy and safety of ombitasvir/paritaprevir/ritonavir/ribavirin in management of Egyptian chronic hepatitis C virus patients with chronic kidney disease: A real-life experience. Medicine 2020, 99, e21972. [Google Scholar] [CrossRef]
- Tronina, O.; Durlik, M.; Wawrzynowicz-Syczewska, M.; Buivydiene, A.; Katzarov, K.; Kupcinskas, L.; Tolmane, I.; Karpińska, E.; Pisula, A.; Karwowska, K.M.; et al. Real-World Safety and Efficacy of Ombitasvir/Paritaprevir/Ritonavir/+Dasabuvir±Ribavirin (OBV/PTV/r/+DSV±RBV) Therapy in Recurrent Hepatitis C Virus (HCV) Genotype 1 Infection Post-Liver Transplant: AMBER-CEE Study. Ann. Transpl. 2017, 22, 199–207. [Google Scholar] [CrossRef]
- Ma, Z.; de Man, R.A.; Kamar, N.; Pan, Q. Chronic hepatitis E: Advancing research and patient care. J. Hepatol. 2022, 77, 1109–1123. [Google Scholar] [CrossRef]
- Primadharsini, P.P.; Nagashima, S.; Nishiyama, T.; Takahashi, M.; Murata, K.; Okamoto, H. Development of Recombinant Infectious Hepatitis E Virus Harboring the nanoKAZ Gene and Its Application in Drug Screening. J. Virol. 2022, 96, e0190621. [Google Scholar] [CrossRef] [PubMed]
- Primadharsini, P.P.; Nagashima, S.; Takahashi, M.; Murata, K.; Okamoto, H. Ritonavir Blocks Hepatitis E Virus Internalization and Clears Hepatitis E Virus In Vitro with Ribavirin. Viruses 2022, 14, 2440. [Google Scholar] [CrossRef]
- Pereira-Chioccola, V.L.; Vidal, J.E.; Su, C. Toxoplasma gondii infection and cerebral toxoplasmosis in HIV-infected patients. Future Microbiol. 2009, 4, 1363–1379. [Google Scholar] [CrossRef]
- Abou-El-Naga, I.F.; El Kerdany, E.D.; Mady, R.F.; Shalaby, T.I.; Zaytoun, E.M. The effect of lopinavir/ritonavir and lopinavir/ritonavir loaded PLGA nanoparticles on experimental toxoplasmosis. Parasitol. Int. 2017, 66, 735–747. [Google Scholar] [CrossRef]
- Beld, L.; Jung, H.; Bulman, C.A.; Rosa, B.A.; Fischer, P.U.; Janetka, J.W.; Lustigman, S.; Sakanari, J.A.; Mitreva, M. Aspartyl Protease Inhibitors as Anti-Filarial Drugs. Pathogens 2022, 11, 707. [Google Scholar] [CrossRef] [PubMed]
- Alves, É.A.R.; de Miranda, M.G.; Borges, T.K.; Magalhães, K.G.; Muniz-Junqueira, M.I. Anti-HIV drugs, lopinavir/ritonavir and atazanavir, modulate innate immune response triggered by Leishmania in macrophages: The role of NF-κB and PPAR-γ. Int. Immunopharmacol. 2015, 24, 314–324. [Google Scholar] [CrossRef] [PubMed]
- Brilhante, R.S.; Caetano, É.P.; Riello, G.B.; Guedes, G.M.; Castelo-Branco Dde, S.; Fechine, M.A.; Oliveira, J.S.; Camargo, Z.P.; Mesquita, J.R.; Monteiro, A.J.; et al. Antiretroviral drugs saquinavir and ritonavir reduce inhibitory concentration values of itraconazole against Histoplasma capsulatum strains in vitro. Braz. J. Infect. Dis. 2016, 20, 155–159. [Google Scholar] [CrossRef] [PubMed]
- van der Putten, E.; Wosikowski, K.; Beijnen, J.H.; Imre, G.; Freund, C.R. Ritonavir reverses resistance to docetaxel and cabazitaxel in prostate cancer cells with acquired resistance to docetaxel. Cancer Drug Resist. 2024, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Ikezoe, T.; Hisatake, Y.; Takeuchi, T.; Ohtsuki, Y.; Yang, Y.; Said, J.W.; Taguchi, H.; Koeffler, H.P. HIV-1 protease inhibitor, ritonavir: A potent inhibitor of CYP3A4, enhanced the anticancer effects of docetaxel in androgen-independent prostate cancer cells in vitro and in vivo. Cancer Res. 2004, 64, 7426–7431. [Google Scholar] [CrossRef]
- Staal, J.; Beyaert, R. Inflammation and NF-κB Signaling in Prostate Cancer: Mechanisms and Clinical Implications. Cells 2018, 7, 122. [Google Scholar] [CrossRef]
- Skinner, K.T.; Palkar, A.M.; Hong, A.L. Genetics of ABCB1 in Cancer. Cancers 2023, 15, 4236. [Google Scholar] [CrossRef]
- Lima, T.S.; Souza, L.O.; Iglesias-Gato, D.; Elversang, J.; Jørgensen, F.S.; Kallunki, T.; Røder, M.A.; Brasso, K.; Moreira, J.M.A. Itraconazole Reverts ABCB1-Mediated Docetaxel Resistance in Prostate Cancer. Front. Pharmacol. 2022, 13, 869461. [Google Scholar] [CrossRef]
- Mita, A.C.; Figlin, R.; Mita, M.M. Cabazitaxel: More than a new taxane for metastatic castrate-resistant prostate cancer? Clin. Cancer Res. 2012, 18, 6574–6579. [Google Scholar] [CrossRef]
- de Weger, V.A.; Stuurman, F.E.; Hendrikx, J.J.M.A.; Moes, J.J.; Sawicki, E.; Huitema, A.D.R.; Nuijen, B.; Thijssen, B.; Rosing, H.; Keessen, M.; et al. A dose-escalation study of bi-daily once weekly oral docetaxel either as ModraDoc001 or ModraDoc006 combined with ritonavir. Eur. J. Cancer 2017, 86, 217–225. [Google Scholar] [CrossRef]
- Vaishampayan, U.N.; Keessen, M.; Heath, E.I.; Dreicer, R.; Buchler, T.; Árkosy, P.F.; Csoszi, T.; Wiechno, P.J.; Kholtobin, D.; Kopyltsov, E.; et al. A phase 2 randomized study of oral docetaxel plus ritonavir (ModraDoc006/r) in patients with metastatic castration-resistant prostate cancer (mCRPC). J. Clin. Oncol. 2022, 40, 5016. [Google Scholar] [CrossRef]
- Li, X.; Ng, A.S.N.; Mak, V.C.Y.; Chan, K.K.L.; Cheung, A.N.Y.; Cheung, L.W.T. Strategic Combination Therapies for Ovarian Cancer. Curr. Cancer Drug Targets 2020, 20, 573–585. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bryant, C.S.; Chamala, S.; Qazi, A.; Seward, S.; Pal, J.; Steffes, C.P.; Weaver, D.W.; Morris, R.; Malone, J.M.; et al. Ritonavir blocks AKT signaling, activates apoptosis and inhibits migration and invasion in ovarian cancer cells. Mol. Cancer 2009, 8, 26. [Google Scholar] [CrossRef] [PubMed]
- Vélez-Cruz, R.; Johnson, D.G. The Retinoblastoma (RB) Tumor Suppressor: Pushing Back against Genome Instability on Multiple Fronts. Int. J. Mol. Sci. 2017, 18, 1776. [Google Scholar] [CrossRef]
- Narasimha, A.M.; Kaulich, M.; Shapiro, G.S.; Choi, Y.J.; Sicinski, P.; Dowdy, S.F. Cyclin D activates the Rb tumor suppressor by mono-phosphorylation. eLife 2014, 3, e02872. [Google Scholar] [CrossRef]
- Winterhoff, B.; Teoman, A.; Freyer, L.; Von Bismarck, A.; Dowdy, S.; Schmalfeldt, B.; Kumar, S.; Shridhar, V. The HIV protease inhibitor ritonavir induces cell cycle arrest and apoptosis in the A2780 ovarian cancer cell line in vitro and in vivo. Gynecol. Oncol. 2013, 130, e138. [Google Scholar] [CrossRef]
- Rascio, F.; Spadaccino, F.; Rocchetti, M.T.; Castellano, G.; Stallone, G.; Netti, G.S.; Ranieri, E. The Pathogenic Role of PI3K/AKT Pathway in Cancer Onset and Drug Resistance: An Updated Review. Cancers 2021, 13, 3949. [Google Scholar] [CrossRef]
- Momenimovahed, Z.; Tiznobaik, A.; Taheri, S.; Salehiniya, H. Ovarian cancer in the world: Epidemiology and risk factors. Int. J. Womens Health 2019, 11, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Srirangam, A.; Wang, M.; Blum, J.; Einhorn, L.; Potter, D.A. Ritonavir causes G1 arrest in non-small cell lung cancer (NSCLC), in part, by binding hsp90 and down-regulating Cdk4 and other Hsp90 client proteins. Cancer Res. 2005, 65, 534. [Google Scholar]
- Rong, B.; Yang, S. Molecular mechanism and targeted therapy of Hsp90 involved in lung cancer: New discoveries and developments (Review). Int. J. Oncol. 2018, 52, 321–336. [Google Scholar] [CrossRef]
- Srirangam, A.; Milani, M.; Mitra, R.; Guo, Z.; Rodriguez, M.; Kathuria, H.; Fukuda, S.; Rizzardi, A.; Schmechel, S.; Skalnik, D.G.; et al. The Human Immunodeficiency Virus Protease Inhibitor Ritonavir Inhibits Lung Cancer Cells, in Part, by Inhibition of Survivin. J. Thorac. Oncol. 2011, 6, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Hu, C.; Li, H. Survivin as a novel target protein for reducing the proliferation of cancer cells. Biomed. Rep. 2018, 8, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Arora, L.; Kumar, A.P.; Arfuso, F.; Chng, W.J.; Sethi, G. The Role of Signal Transducer and Activator of Transcription 3 (STAT3) and Its Targeted Inhibition in Hematological Malignancies. Cancers 2018, 10, 327. [Google Scholar] [CrossRef]
- Courtney, D.; Davey, M.G.; Moloney, B.M.; Barry, M.K.; Sweeney, K.; McLaughlin, R.P.; Malone, C.M.; Lowery, A.J.; Kerin, M.J. Breast cancer recurrence: Factors impacting occurrence and survival. Ir. J. Med. Sci. 2022, 191, 2501–2510. [Google Scholar] [CrossRef]
- Srirangam, A.; Mitra, R.; Wang, M.; Gorski, J.C.; Badve, S.; Baldridge, L.; Hamilton, J.; Kishimoto, H.; Hawes, J.; Li, L.; et al. Effects of HIV protease inhibitor ritonavir on Akt-regulated cell proliferation in breast cancer. Clin. Cancer Res. 2006, 12, 1883–1896. [Google Scholar] [CrossRef]
- Li, H.; Prever, L.; Hirsch, E.; Gulluni, F. Targeting PI3K/AKT/mTOR Signaling Pathway in Breast Cancer. Cancers 2021, 13, 3517. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ma, H.; Yao, J. ERα, A Key Target for Cancer Therapy: A Review. Onco Targets Ther. 2020, 13, 2183–2191. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Hendrikx, J.J.; Rottenberg, S.; Schellens, J.H.; Beijnen, J.H.; Huitema, A.D. Development of a Tumour Growth Inhibition Model to Elucidate the Effects of Ritonavir on Intratumoural Metabolism and Anti-tumour Effect of Docetaxel in a Mouse Model for Hereditary Breast Cancer. AAPS J. 2016, 18, 362–371. [Google Scholar] [CrossRef]
- Schellens, J.H.; Malingré, M.M.; Kruijtzer, C.M.; Bardelmeijer, H.A.; van Tellingen, O.; Schinkel, A.H.; Beijnen, J.H. Modulation of oral bioavailability of anticancer drugs: From mouse to man. Eur. J. Pharm. Sci. 2000, 12, 103–110. [Google Scholar] [CrossRef]
- Hendrikx, J.J.; Lagas, J.S.; Song, J.Y.; Rosing, H.; Schellens, J.H.; Beijnen, J.H.; Rottenberg, S.; Schinkel, A.H. Ritonavir inhibits intratumoral docetaxel metabolism and enhances docetaxel antitumor activity in an immunocompetent mouse breast cancer model. Int. J. Cancer 2016, 138, 758–769. [Google Scholar] [CrossRef]
- Gote, V.; Sharma, A.D.; Pal, D. Hyaluronic Acid-Targeted Stimuli-Sensitive Nanomicelles Co-Encapsulating Paclitaxel and Ritonavir to Overcome Multi-Drug Resistance in Metastatic Breast Cancer and Triple-Negative Breast Cancer Cells. Int. J. Mol. Sci. 2021, 22, 1257. [Google Scholar] [CrossRef] [PubMed]
- Asano, M.; Tanaka, S.; Sakaguchi, M. Effects of normothermic microwave irradiation on CD44(+)/CD24(−) in breast cancer MDA-MB-231 and MCF-7 cell lines. Biosci. Biotechnol. Biochem. 2020, 84, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Al-Othman, N.; Alhendi, A.; Ihbaisha, M.; Barahmeh, M.; Alqaraleh, M.; Al-Momany, B.Z. Role of CD44 in breast cancer. Breast Dis. 2020, 39, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Snyder, S.; Murundi, S.; Crawford, L.; Putnam, D. Enabling P-glycoprotein inhibition in multidrug resistant cancer through the reverse targeting of a quinidine-PEG conjugate. J. Control. Release 2020, 317, 291–299. [Google Scholar] [CrossRef]
- Tabas, I.; Ron, D. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat. Cell Biol. 2011, 13, 184–190. [Google Scholar] [CrossRef]
- Sato, A. The human immunodeficiency virus protease inhibitor ritonavir is potentially active against urological malignancies. Onco Targets Ther. 2015, 8, 761–768. [Google Scholar] [CrossRef][Green Version]
- Gaedicke, S.; Firat-Geier, E.; Constantiniu, O.; Lucchiari-Hartz, M.; Freudenberg, M.; Galanos, C.; Niedermann, G. Antitumor effect of the human immunodeficiency virus protease inhibitor ritonavir: Induction of tumor-cell apoptosis associated with perturbation of proteasomal proteolysis. Cancer Res. 2002, 62, 6901–6908. [Google Scholar]
- Sato, A.; Asano, T.; Okubo, K.; Isono, M.; Asano, T. Ritonavir and ixazomib kill bladder cancer cells by causing ubiquitinated protein accumulation. Cancer Sci. 2017, 108, 1194–1202. [Google Scholar] [CrossRef]
- Xie, J.; Wan, N.; Liang, Z.; Zhang, T.; Jiang, J. Ixazomib—The first oral proteasome inhibitor. Leuk. Lymphoma 2019, 60, 610–618. [Google Scholar] [CrossRef]
- Chen, X.; Shi, C.; He, M.; Xiong, S.; Xia, X. Endoplasmic reticulum stress: Molecular mechanism and therapeutic targets. Signal Transduct. Target. Ther. 2023, 8, 352. [Google Scholar] [CrossRef]
- Sato, A.; Asano, T.; Okubo, K.; Isono, M.; Asano, T. Nelfinavir and Ritonavir Kill Bladder Cancer Cells Synergistically by Inducing Endoplasmic Reticulum Stress. Oncol. Res. 2018, 26, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Okubo, K.; Isono, M.; Asano, T.; Sato, A. Lopinavir-Ritonavir Combination Induces Endoplasmic Reticulum Stress and Kills Urological Cancer Cells. Anticancer. Res. 2019, 39, 5891–5901. [Google Scholar] [CrossRef] [PubMed]
- Okubo, K.; ReßIng, N.; Schulz, W.A.; Hansen, F.K.; Asano, T.; Sato, A. The Dual Histone Deacetylase-Proteasome Inhibitor RTS-V5 Acts Synergistically With Ritonavir to Induce Endoplasmic Reticulum Stress in Bladder Cancer Cells. Anticancer. Res. 2021, 41, 5987–5996. [Google Scholar] [CrossRef] [PubMed]
- Batchu, R.B.; Gruzdyn, O.V.; Bryant, C.S.; Qazi, A.M.; Kumar, S.; Chamala, S.; Kung, S.T.; Sanka, R.S.; Puttagunta, U.S.; Weaver, D.W.; et al. Ritonavir-Mediated Induction of Apoptosis in Pancreatic Cancer Occurs via the RB/E2F-1 and AKT Pathways. Pharmaceuticals 2014, 7, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Mandigo, A.C.; Yuan, W.; Xu, K.; Gallagher, P.; Pang, A.; Guan, Y.F.; Shafi, A.A.; Thangavel, C.; Sheehan, B.; Bogdan, D.; et al. RB/E2F1 as a Master Regulator of Cancer Cell Metabolism in Advanced Disease. Cancer Discov. 2021, 11, 2334–2353. [Google Scholar] [CrossRef]
- Zhao, L.; Zhao, Y.; Schwarz, B.; Mysliwietz, J.; Hartig, R.; Camaj, P.; Bao, Q.; Jauch, K.W.; Guba, M.; Ellwart, J.W.; et al. Verapamil inhibits tumor progression of chemotherapy-resistant pancreatic cancer side population cells. Int. J. Oncol. 2016, 49, 99–110. [Google Scholar] [CrossRef]
- Pontious, C.; Kaul, S.; Hong, M.; Hart, P.A.; Krishna, S.G.; Lara, L.F.; Conwell, D.L.; Cruz-Monserrate, Z. Cathepsin E expression and activity: Role in the detection and treatment of pancreatic cancer. Pancreatology 2019, 19, 951–956. [Google Scholar] [CrossRef]
- Keliher, E.J.; Reiner, T.; Earley, S.; Klubnick, J.; Tassa, C.; Lee, A.J.; Ramaswamy, S.; Bardeesy, N.; Hanahan, D.; Depinho, R.A.; et al. Targeting cathepsin E in pancreatic cancer by a small molecule allows in vivo detection. Neoplasia 2013, 15, 684–693. [Google Scholar] [CrossRef]
- Spencer, N.Y.; Stanton, R.C. The Warburg Effect, Lactate, and Nearly a Century of Trying to Cure Cancer. Semin. Nephrol. 2019, 39, 380–393. [Google Scholar] [CrossRef]
- McBrayer, S.K.; Cheng, J.C.; Singhal, S.; Krett, N.L.; Rosen, S.T.; Shanmugam, M. Multiple myeloma exhibits novel dependence on GLUT4, GLUT8, and GLUT11: Implications for glucose transporter-directed therapy. Blood 2012, 119, 4686–4697. [Google Scholar] [CrossRef]
- Weir, P.; Donaldson, D.; McMullin, M.F.; Crawford, L. Metabolic Alterations in Multiple Myeloma: From Oncogenesis to Proteasome Inhibitor Resistance. Cancers 2023, 15, 1682. [Google Scholar] [CrossRef]
- Dalva-Aydemir, S.; Bajpai, R.; Martinez, M.; Adekola, K.U.; Kandela, I.; Wei, C.; Singhal, S.; Koblinski, J.E.; Raje, N.S.; Rosen, S.T.; et al. Targeting the metabolic plasticity of multiple myeloma with FDA-approved ritonavir and metformin. Clin. Cancer Res. 2015, 21, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Schaff, L.R.; Mellinghoff, I.K. Glioblastoma and Other Primary Brain Malignancies in Adults: A Review. JAMA 2023, 329, 574–587. [Google Scholar] [CrossRef]
- Kast, R.E.; Ramiro, S.; Lladó, S.; Toro, S.; Coveñas, R.; Muñoz, M. Antitumor action of temozolomide, ritonavir and aprepitant against human glioma cells. J. Neurooncol 2016, 126, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Rauschenbach, L.; Wieland, A.; Reinartz, R.; Kebir, S.; Till, A.; Darkwah Oppong, M.; Dobersalske, C.; Ullrich, V.; Ahmad, A.; Jabbarli, R.; et al. Drug repositioning of antiretroviral ritonavir for combinatorial therapy in glioblastoma. Eur. J. Cancer 2020, 140, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Gratton, R.; Tricarico, P.M.; Guimaraes, R.L.; Celsi, F.; Crovella, S. Lopinavir/Ritonavir Treatment Induces Oxidative Stress and Caspaseindependent Apoptosis in Human Glioblastoma U-87 MG Cell Line. Curr. HIV Res. 2018, 16, 106–112. [Google Scholar] [CrossRef]
- Hsu, A.; Granneman, G.R.; Bertz, R.J. Ritonavir: Clinical pharmacokinetics and interactions with other anti-HIV agents. Clin. Pharmacokinet. 1998, 35, 275–291. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).