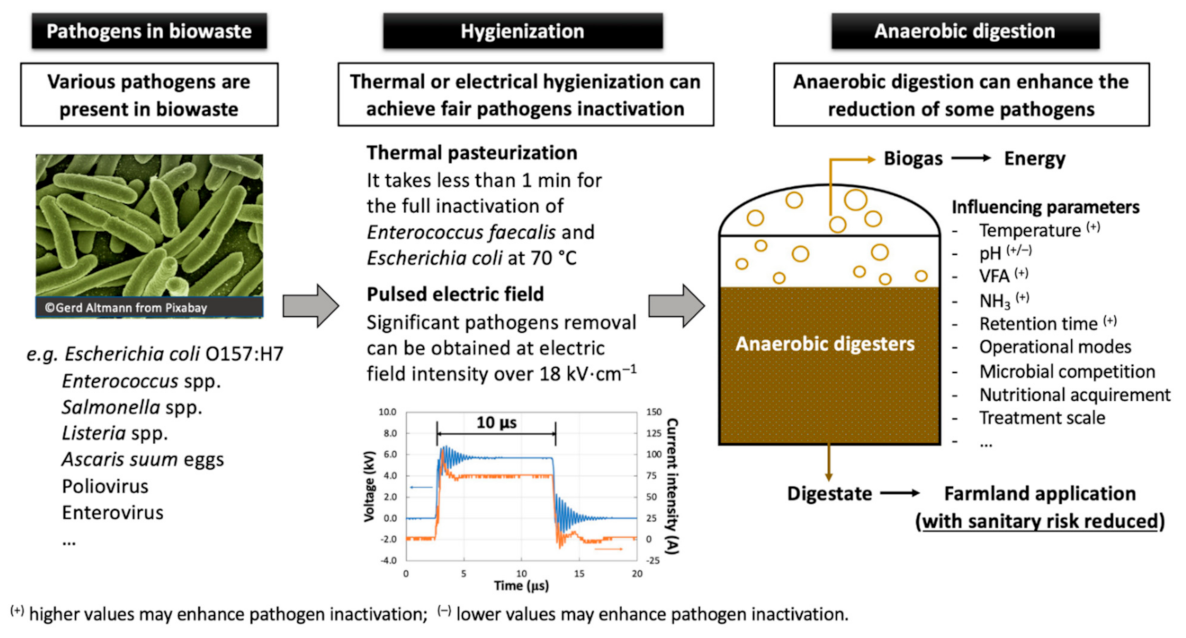
Abstract
Animal by-products (ABP) can be valorized via anaerobic digestion (AD) for biogas energy generation. The digestate issued from AD process is usually used to fertilize farming land for agricultural activities, which may cause potential sanitary risk to the environment. The European Union (EU) requires that certain ABP be thermally pasteurized in order to minimize this sanitary risk. This process is called hygienization, which can be replaced by alternative nonthermal technologies like pulsed electric field (PEF). In the present study, Enterococcus faecalis ATCC 19433 and Escherichia coli ATCC 25922 were used as indicator bacteria. Their resistance to thermal pasteurization and PEF treatment were characterized. Results show that Ent. faecalis and E. coli are reduced by 5 log10 in less than 1 min during thermal pasteurization at 70 °C. The critical electric field strength was estimated at 18 kV∙cm−1 for Ent. faecalis and 1 kV∙cm−1 for E. coli. “G+” bacteria Ent. faecalis are generally more resistant than “G−” bacteria E. coli. AD process also plays an important role in pathogens inactivation, whose performance depends on the microorganisms considered, digestion temperature, residence time, and type of feedstock. Thermophilic digestion is usually more efficient in pathogens removal than mesophilic digestion.
1. Introduction
Reduction of CO2 emission, production of renewable energy as well as promotion of innovative technological advances have nowadays become major objectives for waste valorization leading to circular economy. Anaerobic digestion is a manually controlled microbiological treatment approach converting organic matter of waste (i.e., feedstock) into biogas, which can be used for renewable energy generation [1]. Its coproduct, digestate, is the liquid and solid residue in anaerobic digesters after anaerobic digestion (AD) process. Digestate is rich in mineral elements like N, P, and K and, therefore, can be reused as fertilizers and soil amendment by spreading on farm lands. This “return to soil” policy of digestate is drawing increasing concerns about the sanitary risk to the environment and human being. Contamination of feedstock by various pathogens, such as Salmonella spp., Listeria spp., Giardia spp., Cryptosporidium parvum, and Campylobacter spp. [2,3] and other emerging contaminants like antibiotic resistance genes (ARG) [4], could be transmitted to the environment through waste and digestate land application if they are not properly handled [5]. In 2002, the United States Environmental Protection Agency (US EPA) disclosed that the spreading of biosolids (sewage sludge) on farm lands might cause health problem to nearby local habitants [6]. Afterwards many studies confirm this sanitary risk, especially for anaerobic digestate [7,8].
Animal by-products (ABP) are co-products originated from food industry (e.g., various waste from livestock raising, slaughterhouse and food processing), which are not suitable for human usage. The use, transport, transformation and disposal of ABP are strictly regulated by the European council and parliament [9]. ABP are divided into three categories according to their risk level. Only ABP from Categories 2 and 3 could be valorized through anaerobic digestion in biogas plants (BGP). However, the European Union (EU) requires that these ABP should be pasteurized prior to AD. The process is called hygienization, carried out at 70 °C for at least 60 min. The maximum particle size of ABP entering hygienizers should be kept below 12 mm. Its objective is to reduce the concentration of Enterococcus faecalis or Salmonella senftenberg by a factor of 105 (i.e., 5 log10) and thermal-resistant virus by 3 log10 in ABP [10]. Hygienization consumes huge amount of energy and is considered too prudent in terms of treatment efficiency versus treatment temperature and time [11]. According to Liu et al. (2019) [5], almost 6–25% of the primary energy produced from biogas is consumed by thermally-related hygienization processes in European BGP [5]. This consumption reduces the ecological and financial advantages of biogas energy production.
Pulsed electric field (PEF) is an emerging biomass pretreatment technology. PEF can be applied to liquid or solid materials having biological structure. The intensive electric field (up to 50 kV·cm−1) is delivered to products in forms of short pulses (in the order of milliseconds), giving rise to the electroporation of product cell membrane [12]. Once the electric field intensity exceeds a certain threshold, the induced electroporation on cell membrane disorders microbial activities and therefore, inactivates microorganisms present in the treated materials. This technology has been widely applied for the non-thermal pasteurization of food products [13] and various mechanisms have been discussed for a better knowledge of the process [12,14,15]. PEF was firstly proposed by the same authors of the present paper to substitute for thermal hygienization of ABP by using a batch [16] and a continuous treatment system [17]. The inactivation kinetics of indicator bacteria and the effect of energy input were systematically studied in these studies. The integration of ohmic heating into PEF treatment was proposed to achieve an enhanced synergistic effect of microbial reduction in ABP [17]. Apart from effects on microbial reduction, PEF is able to enhance the biochemical methane potential (BMP) of various biowaste [5].
In addition to thermal and alternative hygienization processes, anaerobic digestion itself was reported to play an important role in certain pathogens inactivation. Many papers have studied the fate of various sporulating and non-sporulating microorganisms during AD treatment and different conclusions were drawn, depending on the AD operational modes, substrates digested and microorganisms considered. Grim et al. (2015) [18] developed integrated thermophilic sanitation operated in anaerobic digesters for ABP hygienization at a Swedish full-scale BGP. Zhao and Liu (2019) [19] resumes in a recent study the key factors determining the reliability of AD process as a possible barrier for deactivation of pathogenic agents in sewage sludge.
Enterococcus faecalis (Gram-positive bacteria, “G+”) and Escherichia coli (Gram-negative bacteria, “G−”) are two microorganisms often serving as indicators characterizing the performance of hygienization process [20,21,22]. As mentioned above, Ent. faecalis is also chosen by EU regulation No. 142/2011 [10] as indicator bacteria. However, the resistance of these two species of bacteria to different hygienization technologies has not been clearly studied. This is the basic information for those who select Ent. faecalis and E. coli as indicator bacteria with regard to various hygienization treatment technologies (i.e., thermal, electrical, and AD stress). It gives practical information to the development of alternative technologies for waste hygienization, helpful for a larger application of innovative technologies such as PEF and the possible integration of hygienization step into the anaerobic digestion process.
The present study characterizes the resistance of indicator microorganisms to conventional thermal pasteurization, alternative PEF treatment and anaerobic digestion. This paper involves (1) the study of inactivation kinetics of Ent. faecalis and E. coli during conventional pasteurization by offering modeling parameters, (2) an investigation of the key characteristics of two indicator bacteria under PEF stress and (3) a literature overview about the effect of AD on the hygienization of biowaste.
2. Materials and Methods
2.1. Suspension Preparation
2.1.1. Indicator Bacteria Incubation
Two strains, Ent. faecalis ATCC 19433 and E. coli ATCC 25922 (Collection of Institut Pasteur, Paris, France), were used as indicator bacteria. Porous beads containing the related strains were incubated at 37 ± 1 °C in nutrient broth (CM0001, Oxoid™, Basingstoke, UK) until achieving respective stationary physiological phase (24 h for Ent. faecalis and 20 h for E. coli [16,17]) since the microorganisms are found the most resistant to heat and PEF treatment at stationary phase [23]. The incubated suspension with target indicator bacteria was then inoculated into different media (nutrient broth or mixed ABP) for thermal and PEF treatment.
2.1.2. Mixed Animal By-Products
The mixed ABP (mixture of fishery waste and pig slurry) came from the feedstock of a local BGP (SEM LIGER, Locminé, France). This ABP had been subjected to thermal pasteurization at 70 °C for an hour conforming to EU regulation No. 142/2011 [10]. No Enterococcus spp. or E. coli were found in this mixed ABP after pasteurization. The collected ABP was analyzed for its physico-chemical properties. The pH value of the ABP was 6.75. Its total solids content was of 14.9%, 94.5% of which was volatile solids. More detailed information is available in Liu et al. (2019) [16].
2.2. Thermal Pasteurization
2.2.1. Thermal Treatment Protocol
The thermal resistance of two indicator bacteria was investigated using the method developed by Le Jean et al. (1994) [24]. 24.3 ± 0.8 μL of the diluted bacterial suspension was put in 75-μL micro-haematocrit capillary tubes (Brand™, Wertheim, Germany) by capillary force. The two ends of capillary tubes were sealed by micro-flame. The initial cell count in capillary tubes varied between 106 and 107 MPN∙mL−1 or CFU∙mL−1.
The capillary tubes containing the bacterial suspension were subjected to different temperatures (55, 60, 65, and 70 °C) for different treatment times. They were simultaneously immersed in a water bath maintained at the desired temperature by a hot plate (Stuart Co., Stone, UK) (see Figure S1 in Supplementary Materials). Time required for the medium in the tube to reach the desired temperature was estimated to be 2.8 s according to previous studies [24,25]. This short rise time can therefore be considered negligible compared to the holding times for the experiments.
Numerous sampling intervals were used to give the bacterial inactivation kinetics as accurate as possible. At each time step, three capillary tubes were removed and cooled by immersion in ice water. The tubes were rinsed with alcohol and then with sterile distilled water. The rinsed tubes were then kept in ice water (0 °C) for viable counts within two hours of handling.
2.2.2. Culturable Bacteria Count
The viable fraction of indicator bacteria present in each capillary tube was counted using Most Probable Number (MPN) method, recommended by the United State Food and Drug Administration [26] and the United States Department of Agriculture [27].
The capillary tubes, without treatment (i.e., controls) or after treatment, were broken into sterile microplates (PS-96 wells, Corning, New York, NY, USA) pre-filled with 216 μL of autoclaved nutrient broth per well. Serial dilutions were performed by micropipettes using disposable sterile filter tips (VWR, Radnor, PA, USA). Before each dilution, the suspensions were stirred several times by the micropipette. The microplates were then incubated at 37 °C for 24 h. The controls (nutrient broth without indicator bacteria in the capillary tubes and in the microplates) were realized in parallel to check for potential contamination. All of the manipulations were carried out under aseptic conditions.
After 24 h incubation, the results (cloudy or not) were noted for each dilution. The data were then entered into the software developed by Dr Blodgett from the US FDA (Division of Mathematics) in order to exert the viable fraction of indicator bacteria in all tubes [26].
2.2.3. Inactivation Kinetics Modeling
Weibull model, as shown in Equation (1), was used to realize the inactivation kinetics of two indicator bacteria after pasteurization at four studied temperatures.
where N(t) is the culturable bacteria count at time t (MPN∙mL−1), N0 is the initial bacteria count (MPN∙mL−1), t is the treatment time (s), α is the scale parameter of Weibull model (s) and β is the shape parameter of Weibull model (–).
According to van Boekel (2002) [28], the scale parameter α of Weibull model strongly depends on the external circumstances like temperature. As a result, the Arrhenius equation (c.f. Equation (2)) was proposed as secondary model to study the possible relationship between the Weibull model’s scale parameter α and the studied temperatures.
where A is the pre-exponential factor (s−1), Ea is the activation energy for the reaction (kJ∙mol−1), R is the universal gas constant (R = 8.314 J∙K−1∙mol−1) and T is the pasteurization temperature (°C).
Van Boekel (2002) [28] indicates that the shape parameter β is an intrinsic parameter of one particular species of bacteria, independent of or weakly dependent on the external environment.
A parameter estimating the time to achieve 5-log10 reduction of indicator bacteria, i.e., 5-D value, is proposed. It can be calculated using Weibull parameters as shown in Equation (3) [29].
where α is the scale parameter of Weibull model (s) and β is the shape parameter of Weibull model (–).
2.3. PEF Treatmennt as Alternative Hygienization
2.3.1. PEF Treatment Protocol
The resistance of indicator bacteria to PEF exposure in nutrient broth (NB) and mixed ABP was characterized in electroporation cuvettes (1-mm gap 90 μL, VWR, Belgium). The experimental setting of PEF treatment was described by Liu et al. (2019) [16]. It was composed of a high voltage generator (SR2.5-P-600, Technix, Créteil, France) and a pulse signal generator (TGP 110, TTI Thurlby Thandar instrument, Bruz, France) regulated by an oscilloscope (OX8022-20 MHz, Metcix, Paris, France). All of them were connected to an electrical modulator (AHTPM2.5, Effitech, Pau, France) sending high voltage pulses to electroporation cuvettes (see Figures S2 and S3 in Supplementary Materials). The electric generator could give voltage up to 2500 V.
The electric field intensity (E) varied from 0 to 25 kV∙cm−1 with the energy input kept constant at 350 J∙mL−1 for each electric field intensity studied. The energy input from high voltage generator can be calculated by the following Equation (4) [30]:
where WPEF is the energy input (J∙mL−1), tPEF is the effective PEF treatment time (s), U is the voltage applied (V), I is the electric current intensity (A), E is the electric field intensity (kV∙cm−1), σ is the electrical conductivity (σ = 0.2 S∙m−1), ntrain is the number of treatment trains (–), f is the repetition frequency (f = 40 Hz), ttrain is the treatment time per train (s) and τ is the pulse width (τ = 2 μs). The suspension temperature in cuvettes was kept under 45 °C in order to minimize the possible microbial damage from ohmic heating, realized by a cooling system consisting of forced ventilation. After one trial, the treated electroporation cuvette was put into iced water pending further microbiological analysis within 2 h.
2.3.2. Culturable Bacteria Count
The culturable fraction of indicator bacteria was counted in triplicates using spread plate method on selective agars. Detailed information could be found in Liu et al. (2019) [16]. Serial dilutions of the PEF-treated suspension were performed under aseptic conditions. 0.1 mL of each dilution was spread, for Ent. faecalis, on Slanetz and Bartley agar (CM0377, Oxoid™, Basingstoke, UK) and for E. coli, on Tryptone Bile X-glucuronide Agar (CM0945, Oxoid™, Basingstoke, UK). Incubation of Ent. faecalis and E. coli in a ventilated oven was carried out at 37 °C for 48 h and 24 h, respectively, before counting.
2.3.3. Estimation of Critical Electric Field Intensity
Critical electric field strength is the intensity of electric field above which a significant inactivation ratio of a given microorganism is observed. This is a key parameter characterizing the resistance of a specific bacteria species to PEF exposure. In order to find this value for two indicator bacteria, the inactivation ratios as a function of applied field strength were modeled by Fermi’s model [31] whose formular is shown in Equation (5).
where N(E) is the inactivation ratio obtained at the studied electric field intensity E (CFU∙mL−1), N0 is the initial bacteria count (CFU∙mL−1), E is the electric field intensity (kV∙cm−1), Ec is the critical electric field intensity (kV∙cm−1) and kc is the Fermi’s model parameter (kV∙cm−1).
2.4. Microbial Inactivation during AD
The inactivation of bacteria during anaerobic digestion was studied by conducting a literature review of published work. The databases used include Web of Science™ (Clarivate Analytics, Philadelphia, PA, USA), Scopus® (Elsevier, Amsterdam, The Netherlands), and Google Scholar™ (Alphabet, Mountain View, CA, USA). In addition to the indicator bacteria mentioned in Section 2.1.1 (Ent. faecalis and E. coli), the information about other important microorganisms like Staphylococcus aureus, coliforms, Salmonella spp., Campylobacter spp., and Clostridium perfringens is also given in this study.
2.5. Data Processing
The achieved experimental and literature data were processed by Microsoft Excel 2016 (Microsoft®, Redmond, WA, USA). The non-linear regression for kinetic modeling was realized by R studio (R Studio Inc., Boston, MA, USA). The modeling goodness was evaluated according to adjusted R2, sum of squared errors (SSE) and root mean squared errors (RMSE) [32].
3. Results
3.1. Resistance to Thermal Treatment
3.1.1. Thermal Inactivation Kinetics
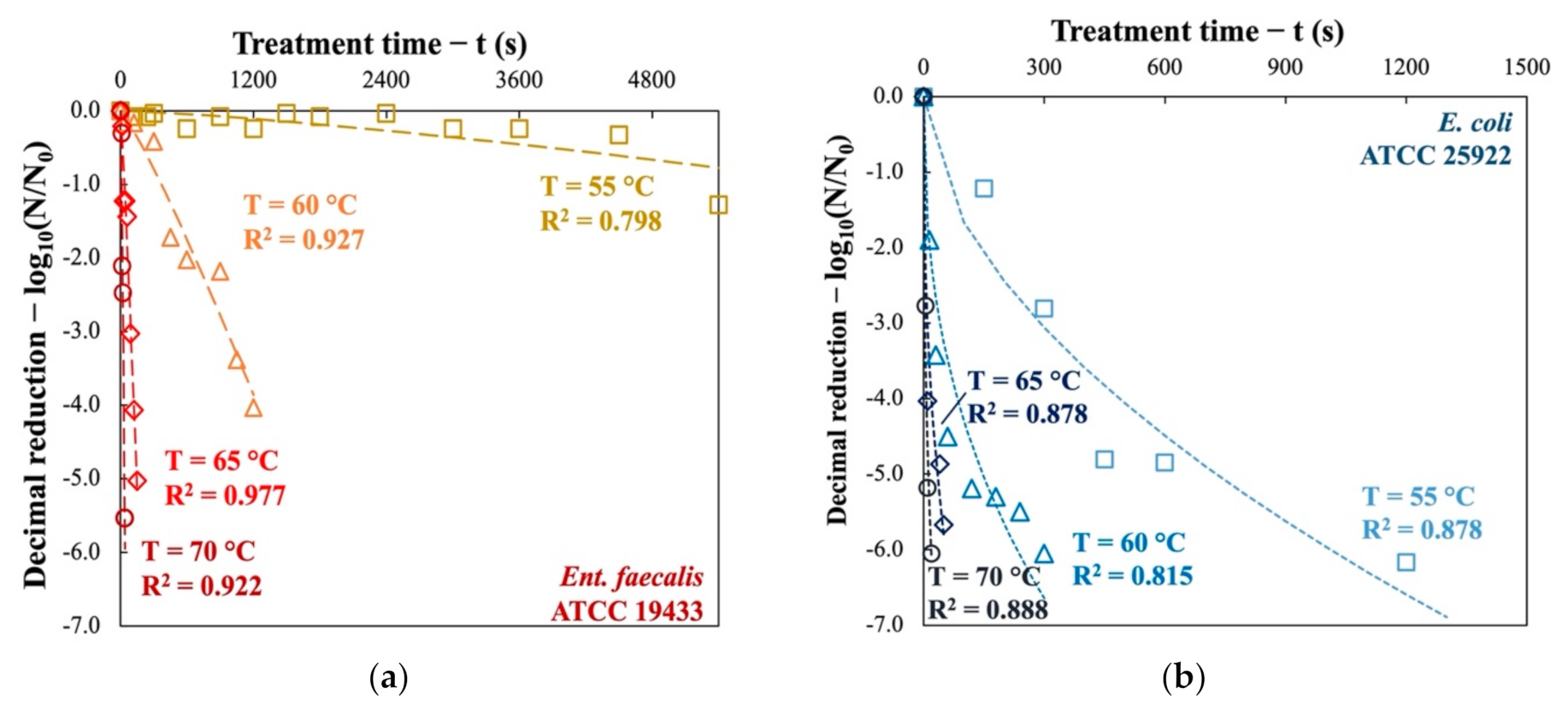
The thermal inactivation kinetics of the two indicator bacteria were performed at 55, 60, 65, and 70 °C. The cultivable fraction was counted using the MPN method and the decimal reductions are presented as a function of processing time in Figure 1. It is evident that the treatment temperature (T) played an important role in the effectiveness of pasteurization. However, the two indicator bacteria behave differently when exposed to the same heat stress.
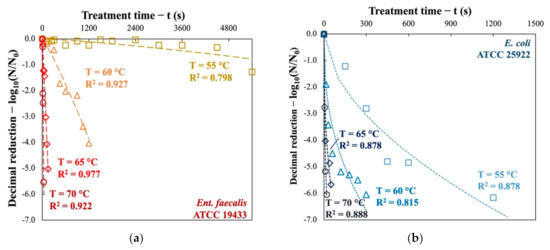
Figure 1.
Inactivation kinetics of (a) Ent. faecalis and (b) E. coli subjected to thermal pasteurization at four temperatures in nutrient broth.
The “G+” bacteria Ent. faecalis are slightly inactivated (0.24 log10 and 1.28 log10) at 55 °C after treatment of 1.0 h and 1.5 h respectively, whereas at 60 °C, a 20 min treatment is sufficient to achieve 4.0-log10 reduction. When the pasteurization temperature is increased from 65 °C to 70 °C, a reduction of more than 5 log10 can be achieved in only 150 s and 35 s, respectively.
As for the “G−” bacteria E. coli, it is more sensitive to heat and its inactivation kinetics are totally different from Ent. faecalis. Pasteurization at 55 °C can easily cause a 5-log10 reduction in 20 min. As the temperature rises, the inactivation kinetics are much faster: at 70 °C, less than 10 s are sufficient for a decimal reduction of more than 6 log10.
It should be noted that the shape of the inactivation curves for Ent. faecalis differs from that of E. coli. The former has a concave shape, whereas for the latter’s shape is convex. The concave shape may be due to the fact that Ent. faecalis, initially resistant to heat stress, accumulate sublethal thermal damage at the beginning of treatment. As treatment time moves on, the bacteria are no longer able to survive pasteurization. On the contrary, the convex shape could be explained by the vulnerability of most of the E. coli population to thermal pasteurization while a certain fraction of the bacteria is more resistant to heat stress.
3.1.2. Curves Modeling and Activation Energy
Inactivation kinetics of two indicator bacteria subjected to thermal pasteurization were modeled using the Weibull model (Equation (1)). This model assumes a non-homogeneous distribution under external stress in a population of samples (population of Ent. faecalis and E. coli in our case). The modelled curves are presented in Figure 1. Table 1 also summarizes the parameter values and the quality of the modeling used. It can be seen that the modeling of microbial destruction kinetics is satisfactory. The modeling of Ent. faecalis is generally better than that of E. coli.

Table 1.
Modeling results of thermal inactivation kinetics of two indicator bacteria using Weibull model.
Since the inactivation curves do not follow first order kinetics (log-linear function), 5-D values estimated by Weibull model instead of 1-D values were calculated. Table 1 gives the time to achieve a reduction of 5 log10 (5-D, see Equation (3)) in indicator bacteria. This performance corresponds to the EU criterion about the hygienization efficiency. It can be seen that the 5-D value of Ent. faecalis is reduced by a factor of 10 when the temperature is increased by every 5 °C between 55 °C and 65 °C, whereas it is reduced by a factor of 5 between 65 °C and 70 °C. It indicates that the Ent. faecalis strain has a higher resistance to heat stress. Pasteurization at 55 °C has almost no effect on the inactivation of this “G+” strain.
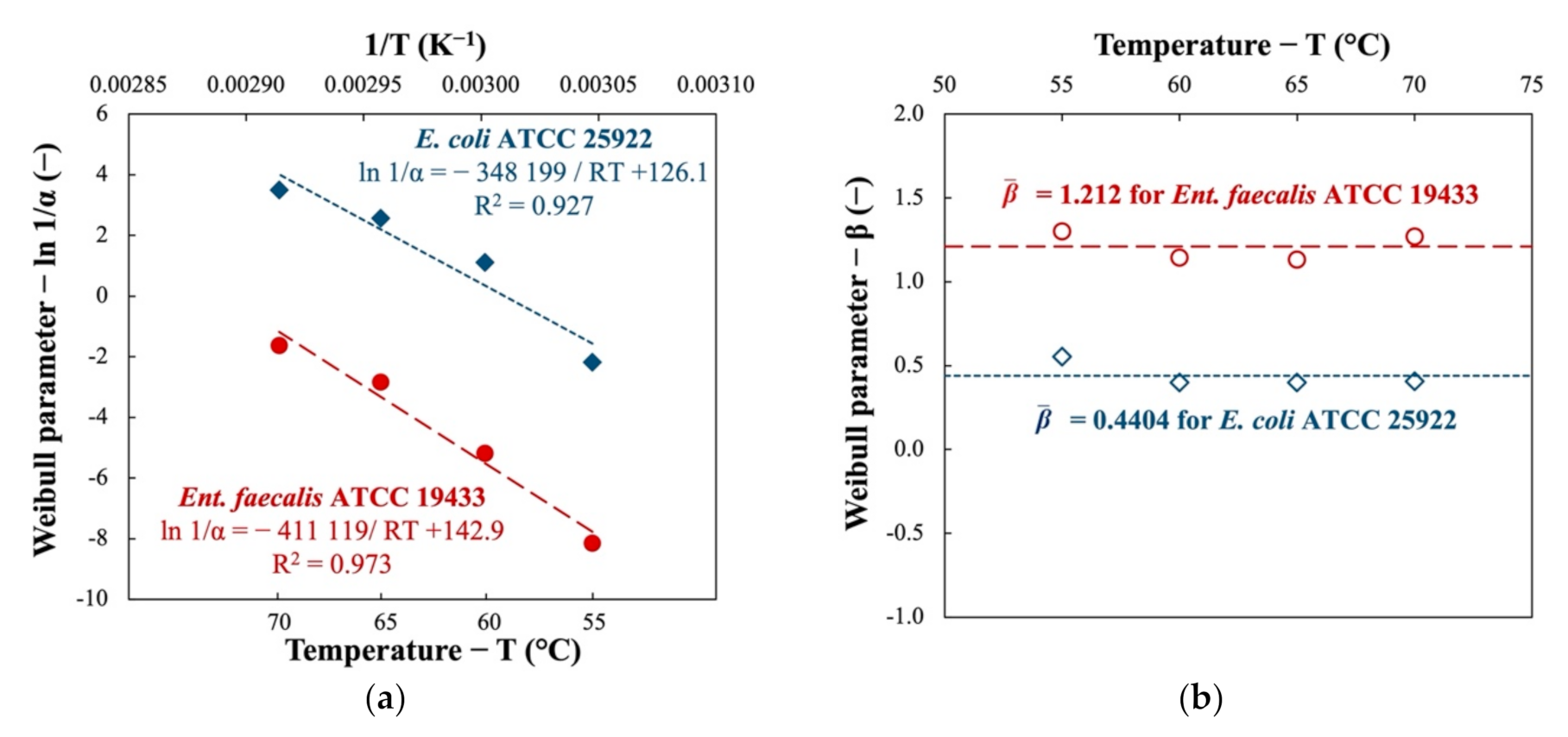
The relationship between the parameters of the Weibull model and pasteurization temperatures was studied (see Figure 2). When plotting the ln(1/α) against the inverse of the temperature (1/T), a linear function described by the Arrhenius equation (Equation (2)) was established with satisfactory modeling performance. The activation energy for pasteurization of the two indicator bacteria is therefore calculated as shown in Figure 2a (Ea = 411.1 kJ∙mol−1 and 348.2 kJ∙mol−1 for Ent. faecalis and E. coli, respectively). The difference in activation energy suggests the difference in resistance of the studied bacteria to thermal pasteurization.
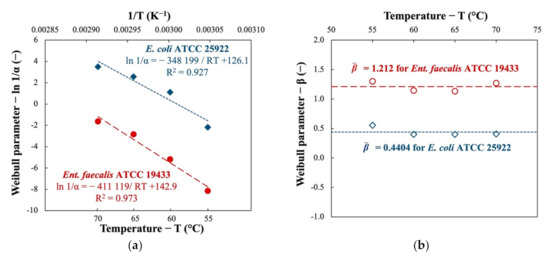
Figure 2.
Dependence of (a) Weibull scale parameter α and (b) Weibull shape parameter β on the treatment temperature during thermal pasteurization of two indicator bacteria in nutrient broth.
As illustrated by Figure 2b, the values of Weibull shape parameter β do not vary much as the treatment temperature changes (coherent to the statement in Section 2.2.3). The mean value of β is found 1.121 for Ent. faecalis and 0.4404 for E. coli.
3.2. Resistance to PEF Treatment—Critical Electric Field Strength
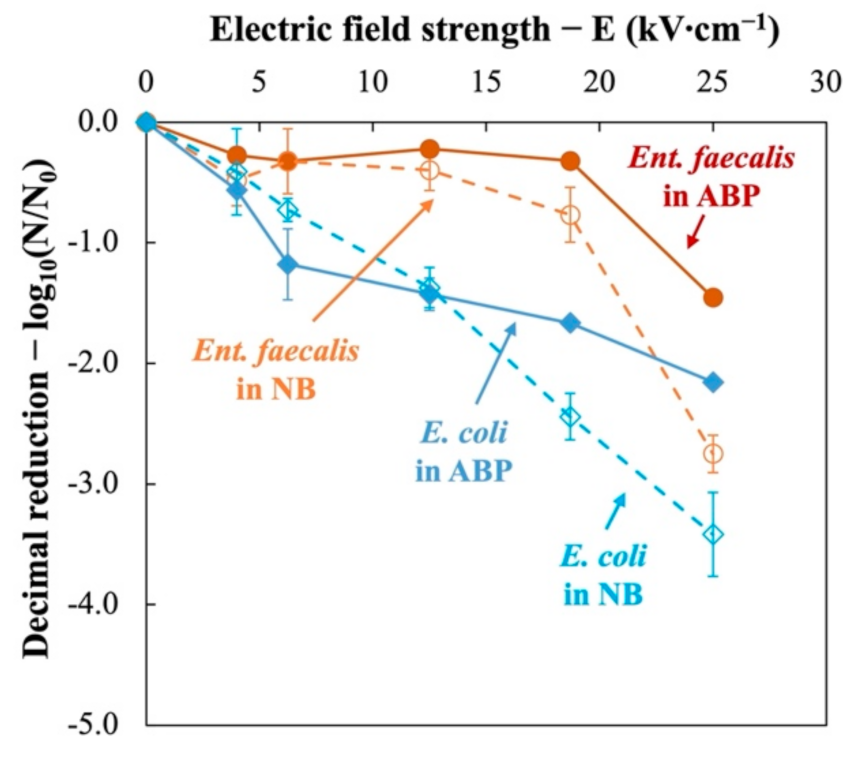
The obtained inactivation ratios of two indicator bacteria in nutrient broth (NB) and mixed animal by-products (ABP) were plotted against the applied electric field strength (0, 4.0, 6.3, 12.5, 18.7, and 25.0 kV∙cm−1).
Figure 3 illustrates the behavior of two indicator bacteria treated at 350 J∙mL−1 at different electric fields, in the nutrient broth or in animal by-products. It can be deduced that PEF treatments are generally less effective in ABP than in NB. This justifies the previous findings that animal by-products provide a protective effect to microorganisms against electrical treatment [16].
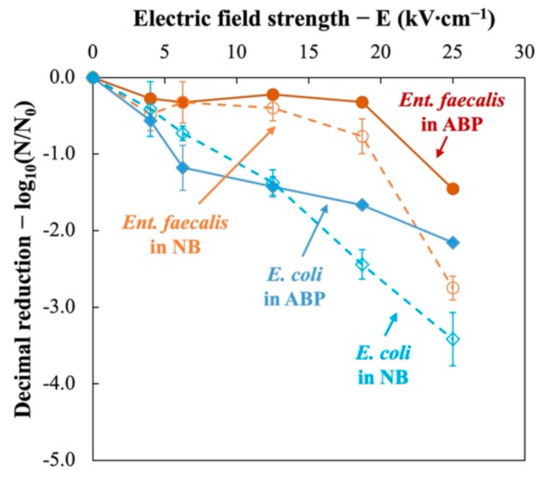
Figure 3.
Inactivation kinetics of Ent. faecalis (red) and E. coli (blue) subjected to pulsed electric field (PEF) treatment in nutrient broth (NB, dotted lines) and animal by-products (ABP, solid lines).
For Ent. faecalis, slight inactivation (<0.4 log10) can be obtained when electric field varied between 0 and 18.5 kV∙cm−1. When electric field intensity increases from 18.5 kV∙cm−1 to 25 kV∙cm−1, significant reduction was observed (1.45 log10 in ABP and 2.75 log10 in NB). Nevertheless, E. coli presents a different inactivation curves. It was inactivated since the very beginning and almost linear inactivation curves are obtained (2.16 log10 in ABP and 3.42 log10 in NB at 25 kV∙cm−1).
The Fermi model (Equation (5)) was used to estimate the critical electric field strength values (Ec) of indicator bacteria. Table 2 summarizes the modeling results. The critical field strengths are estimated at 18.0 kV∙cm−1 and 1.0 kV∙cm−1, respectively, for Ent. faecalis and E. coli. They are close to the theoretical Ec values, 16.5 kV∙cm−1 for Ent. faecalis and 3.34 kV∙cm−1 for E. coli, calculated by Heinz et al. (2001) [33].

Table 2.
Modeling results of PEF inactivation kinetics of two indicator bacteria using Fermi’s model.
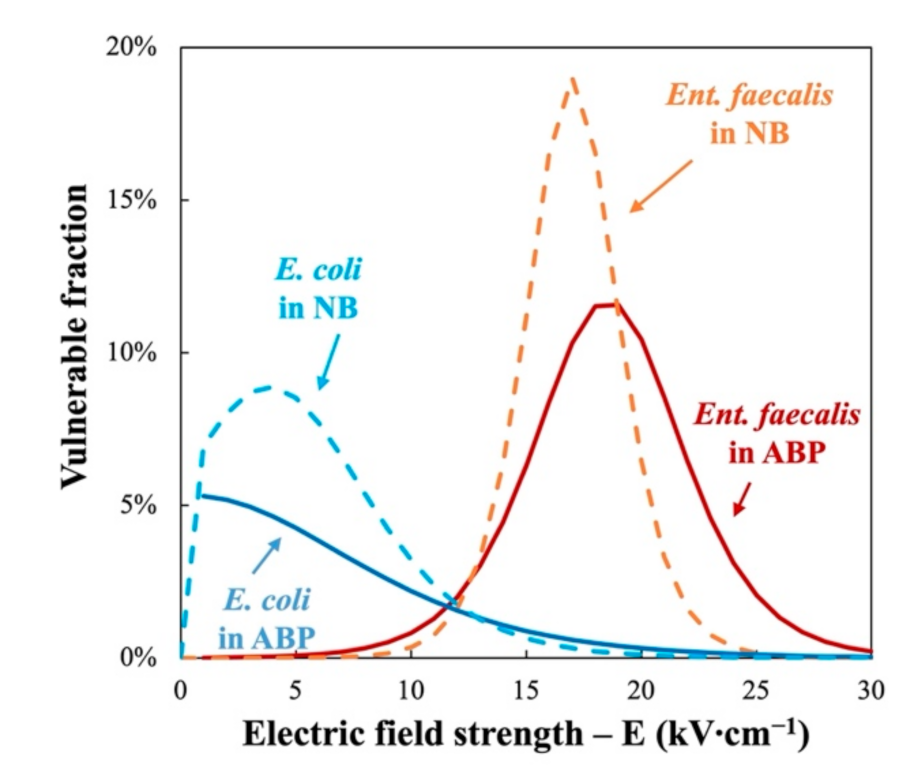
The derivative of the Fermi’s modelled bacterial inactivation curves in Figure 4 shows the distribution of the vulnerable fraction of the two indicator bacteria as a function of electric field. It can be seen that there is a much larger PEF-resistant fraction in Ent. faecalis than in E. coli. 90% of E. coli is vulnerable to electric field strength less than 10 kV∙cm−1, as compared with 20 kV∙cm−1 for Ent. faecalis treated in the same ABP at 350 J∙mL−1. This confirms that “G+” bacteria like Ent. faecalis are generally more resistant to PEF treatment than “G−” bacteria like E. coli [15,16]. In addition, the mixture of ABP has a protective effect compared to NB.
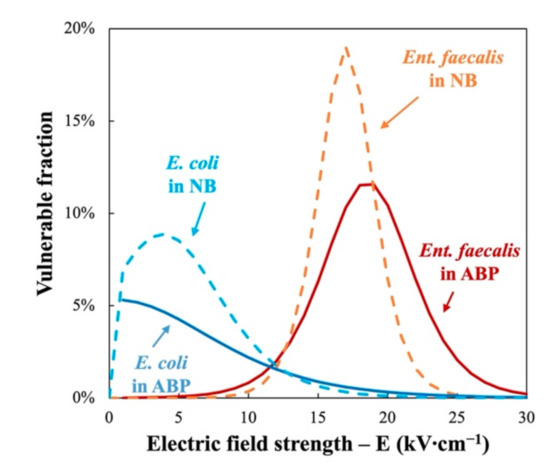
Figure 4.
Distribution of vulnerable fraction of Ent. faecalis (red) and E. coli (blue) with regard to PEF treatment in nutrient broth (NB, dotted curves) and animal by-products (ABP, solid curves).
3.3. Resistance to AD Process
Table 3 reviews the literature data about the effect of anaerobic digestion on the inactivation of pathogen agents present in ABP like animal manure and slaughterhouse waste.

Table 3.
Literature data about the efficiency of anaerobic digestion (AD) process on pathogens inactivation in animal by-products.
Various microorganisms are discussed including bacteria like Bacillus spp. (G+), Clostridium spp. (G+), enterococcus spp. (G+), Listeria spp. (G+), Mycobacterium spp. (considered as G+), Staphylococcus aureus (G+), Campylobacter spp. (G–), E. coli (G–), Salmonella spp. (G–), Yersinia spp. (G–), total and fecal coliforms (G–), eggs of eukaryotes like Ascaris suum, Eimeria tenella, virus like poliovirus and bacteriophages like coliphages. Table 4 and Table 5 list, not exhaustively, several examples of pathogens removal during AD of sewage sludge and other biowaste like municipal solid waste (MSW) and green waste.

Table 4.
Literature data about the efficiency of AD process on pathogens inactivation in sewage sludge.

Table 5.
Literature data about the efficiency of AD process on pathogens inactivation in other biowaste.
The pathogens inactivation efficiency largely depends on the AD operational conditions such as temperature, retention time and treatment scale. Generally, thermophilic AD process (T > 50 °C) tends to be more critical to the survival of microorganisms. Mesophilic AD (35 °C < T < 40 °C) may have effect on certain microorganisms but its performance is limited. A longer retention time in anaerobic digesters may cause a relatively higher microbial removal efficiency. Treatment on lager scales (e.g., full scale BGP) has more difficulty in pathogens reduction than on laboratory scales.
“G−” bacteria are generally more vulnerable to the stress brought by anaerobic conditions than “G+” bacteria. Mesophilic AD has little effect on certain sporulating bacteria like Clostridium spp., bacillus spp. and their respective resistant forms (spores).
When it comes to the efficiency difference among substrates treated by AD (i.e., comparison among Table 3, Table 4, Table 5), pathogens present in sewage sludge and other biowaste are more vulnerable to anaerobic digestion than in animal by-products. This may be due to the variation in chemical composition of substrates that lead to difference in physico-chemical conditions during AD processes, e.g., pH evolution, volatile fatty acids (VFA) production and NH3 accumulation [19].
The mechanisms behind these phenomena are to be discussed in Section 4.
4. Discussion
4.1. Mechanisms
Resistance of Ent. faecalis ATCC 19433 and E. coli ATCC 25922 to the thermal pasteurization was characterized. The 5-D values found by Weibull model were coherent with the reported data [65,66]. The activation energy (Ea) for the inactivation of two indicator bacteria was estimated at 411.1 kJ∙mol−1 and 348.2 kJ∙mol−1. These values correspond to the activation energy range for proteins denaturation [67,68]. It indicates that certain proteins and enzymes critical to microbial activities are denatured giving rise to the bacterial inactivation during thermal treatment. As for PEF treatment, the critical electric field strengths are estimated at 18.0 kV∙cm−1 and 1.0 kV∙cm−1 for Ent. faecalis and E. coli, respectively, close to the theoretical Ec values given by Heinz et al. (2001) [33]. Similar to thermal inactivation, “G+” bacteria Ent. faecalis has a higher resistance to PEF exposure than E. coli.
The difference in chemical compositions constituting cell wall may explain the resistance variation between “G+” and “G−” bacteria to thermal and PEF treatment. “G+” bacteria have a thicker cell wall with multi-layered peptidoglycan and teichoic acids preventing penetration of toxic substances into cells [68]. This rigid cell wall resists the heat transfer and PEF stress that disturb microbial activities [16,17,69,70,71]. As for “G−” bacteria like E. coli, their cell wall consists of a monolayer of peptidoglycan covered by a lipid-rich membrane, which is therefore vulnerable to any physical and chemical stress [68,69].
The difference in electrical treatment efficiency between ABP and nutrient broth reveals that the chemical composition of the products is a very important parameter when evaluating the PEF hygienization efficiency. The changes in electrical conductivity, osmolarity and water activity in the suspension has an impact on bacterial survival [32]. Similar protective effect was seen during the PEF pasteurization of products rich in proteins (E. coli in skim milk) [72], lipids (E. coli, Lactobacillus brevis, Pseudomonas fluorescens, and Saccharomyces cerevisiae in full cream milk) [73], and polysaccharides (Pseudomonas spp. in xanthan gum) [74]. The interaction of indicator bacteria with other microorganisms could also be a reason explaining the efficiency difference in ABP and nutrient broth. In non-sterilized conditions like the case of ABP mixture, microorganisms could form biofilm or aggregates. Then microorganisms situated in the core of aggregates could be protected by the external layers of biofilm or aggregates. Those microbes, as a result, receive less electrical stress and therefore, survive during the treatment [75].
The literature review about the AD effect on pathogen inactivation indicates that its performance depends on the microorganisms considered, digestion temperature, residence time and type of feedstock. Thermophilic digestion is usually more efficient in pathogens removal than mesophilic digestion. “G+” bacteria can persist better than “G−” bacteria. Spores can hardly be influenced by AD process. Figure 5 summarizes different AD parameters that have an impact on pathogens removal during AD treatment of biowaste.
Figure 5.
Descriptive diagram of various AD parameters serving for sanitary control.
Aitken et al. (2005) found the energy activation of 580 kJ∙mol−1 and 510 kJ∙mol−1 for deactivation of Ascaris suum and poliovirus respectively during sludge thermophilic anaerobic digestion operated at 49–53 °C [51]. Differently, Popat et al. (2010) revealed an activation energy of 39 kJ∙mol−1 for poliovirus during sludge AD operated at 51–55 °C. This value refers to the activation energy of RNA inactivation [76]. From the perspectives of molecular microbiology, two different mechanisms could be attributed to explain the microbial inactivation during AD: capsid protein denaturation at higher temperature (T > 55 °C) and DNA or RNA inactivation at lower temperature (T < 55 °C) [77]. The types of AD feedstock (substrates) can also influence the inactivation efficiency of pathogens during AD. Large molecules like lipids, proteins and polysaccharides are degraded into small molecules like VFA and NH3. VFA can cause rapid local pH changes stressing microorganisms. Free VFA and NH3 can penetrate into cells with ease, which disturbs microbial activities. In addition, the competition of various microorganisms present in anaerobic digesters may also be a factor influencing pathogens inactivation performance during AD [19,68].
4.2. Perspectives
The present paper studies the microbial resistance of indicator bacteria to thermal pasteurization, alternative PEF treatment and AD processes. It helps a better grasp of the knowledge related to the hygienization process of biowaste for biogas production.
Conventional thermal pasteurization, conforming to EU No. 142/2011 [10], is found energy consuming. 6–25% of primary energy from biogas could be used to feed this process [5]. The objectives and operational conditions of hygienization have to be reviewed to support the sustainable development of the industry of biogas production. Innovative technologies like alternative hygienization solutions are thus to be studied. In fact, many attempts have been made. In Sweden, Grim et al. (2015) [18] proposed the integrated thermophilic sanitation (ITS) by keeping the substrates at 52 °C for 10 h in thermophilic digesters and for a minimum hydraulic retention time (HRT) of 7 d to dispense from EU pasteurization obligation. They succeeded in achieving similar microbial reduction and got the approval from Swedish Board of Agriculture. Liu et al. (2019 and 2020) [16,17] have studied the possible application of PEF on the alternative hygienization of ABP.
In addition, research focus have been put into the removal of emerging microbial contaminants like ARG. As mentioned in Section 1, studies [4,78] found that ARG could be hardly reduced through traditional waste treatment technologies. Derongs et al. (2020) [79] revealed the role of anaerobic digestate as possible carrier of antibiotic-resistant Clostridium perfringens into the environment. The removal of ARG has to be taken into consideration by future hygienization process. Other infectious pathogens like bacterial spores and African Swine Fever Virus should be included in hygienization objectives as well. More studies are needed to clarify the health risk and their fate during conventional hygienization and AD treatment.
In addition to pathogen removal, hygienization, no matter thermal or alternative (e.g., PEF, microwave and ultrasound), may serve as pretreatment of substrates influencing their biomethane potential [80]. Many studies on ABP [81], biowaste [82] and green waste [83] were realized and around 0–50% BMP increase could be achieved [5]. However, a systematic study combining effect of hygienization on both BMP enhancement and pathogens removal is rare.
Therefore, future strategy of selecting pretreatment methods for biogas feedstock should rely on their sanitary aspect, feedstock types, methane potential intensification, energy consumption, potential efficiency dealing with emerging biological contaminants (e.g., ARG, African Swine Fever Virus (ASFV), and spores) and environmental impact evaluation carried out by life cycle assessment (LCA) approaches.
5. Conclusions
The present study focuses on the characterization of microbial resistance to traditional and innovative hygienization, including thermal pasteurization, PEF treatment and AD process. The thermal inactivation kinetics of Ent. faecalis and E. coli were presented. The values of critical electric field strength of two indicator bacteria are estimated. After a literature review, the behavior of various microorganisms like virus, bacteria, eukaryotes, and phages during anaerobic digestion is summarized. Possible mechanisms explaining the difference in microbial resistance are given. The paper also discusses the perspectives of the research focus concerning hygienization of biowaste. They are the essential information for academic and industrial partners to practice hygienization of biowaste in a more sustainable way.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/en14071938/s1, Figure S1: Photography of capillary tubes in water bath heated at 60 °C by hot plate for pasteurization treatment of indicator bacteria, Figure S2: Schematic description of PEF treatment system, Figure S3: Photography and schematic description of electroporation cuvettes used for PEF treatment of indicator bacteria.
Author Contributions
Experiments, X.L.; Conceptualization, X.L., T.L., and J.-L.L.; methodology, X.L., T.L., and J.-L.L.; software, X.L.; validation, T.L. and J.-L.L.; formal analysis, X.L.; investigation, X.L.; resources, T.L. and J.-L.L.; data curation, X.L.; writing—original draft preparation, X.L.; writing—review and editing, T.L. and J.-L.L.; visualization, X.L.; supervision, T.L. and J.-L.L.; project administration, T.L and J.-L.L.; funding acquisition, T.L. and J.-L.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research consisted in a PhD program jointly funded by Regional Council of Brittany, France (grant number ARED-HYDATE), Departmental Council of Morbihan, France (grant number CD56-HYDATE), European Regional Development Fund—CPER SMART AGRI APIVALE and French mixed economy company LIGER (grant number 2017_00212).
Acknowledgments
The authors thank Yves Lemée and Magali Le Fellic for their valuable support.
Conflicts of Interest
The authors declare no conflict of interest.
Nomenclature
| ABP | Animal by-products |
| AD | Anaerobic digestion |
| Adjusted R2 | Adjusted coefficient of determination |
| ARG | Antibiotic resistance genes |
| ASFV | African Swine Fever Virus |
| BGP | Biogas plants |
| BMP | Biochemical methane potential |
| CFU | Colony-forming unit |
| EU | European Union |
| G+ | Gram-positive bacteria |
| G– | Gram-negative bacteria |
| HRT | Hydraulic retention time |
| LCA | Life cycle assessment |
| MAD | Mesophilic anaerobic digestion |
| MPN | Most probable number |
| MSW | Municipal solid waste |
| NB | Nutrient broth |
| PEF | Pulsed electric field |
| RMSE | Root mean squared errors |
| SSE | Sum of squared errors |
| TAD | Thermophilic anaerobic digestion |
| US EPA | the United States Environmental Protection Agency |
| US FDA | the United States Food and Drug Administration |
| VFA | Volatile fatty acids |
| VS | Volatile solids |
| WAS | Waste activated sludge |
| WWTP | Wastewater treatment plant |
References
- Rekleitis, G.; Haralambous, K.-J.; Loizidou, M.; Aravossis, K. Utilization of Agricultural and Livestock Waste in Anaerobic Digestion (A.D): Applying the Biorefinery Concept in a Circular Economy. Energies 2020, 13, 4428. [Google Scholar] [CrossRef]
- Hutchison, M.L.; Walters, L.D.; Avery, S.M.; Synge, B.A.; Moore, A. Levels of Zoonotic Agents in British Livestock Manures. Lett. Appl. Microbiol. 2004, 39, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Le Maréchal, C.; Druilhe, C.; Repérant, E.; Boscher, E.; Rouxel, S.; Roux, S.L.; Poëzévara, T.; Ziebal, C.; Houdayer, C.; Nagard, B.; et al. Evaluation of the Occurrence of Sporulating and Nonsporulating Pathogenic Bacteria in Manure and in Digestate of Five Agricultural Biogas Plants. Microbiol. Open 2019, 8, e872. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Yu, Z.; Shao, L.; Zhou, Y.; Lü, F. Fate of Antibiotics and Antibiotic-Resistance Genes in a Full-Scale Restaurant Food Waste Treatment Plant: Implications of the Roles beyond Heavy Metals and Mobile Genetic Elements. J. Environ. Sci. 2019. [Google Scholar] [CrossRef]
- Liu, X.; Lendormi, T.; Lanoisellé, J.-L. Overview of Hygienization Pretreatment for Pasteurization and Methane Potential Enhancement of Biowaste: Challenges, State of the Art and Alternative Technologies. J. Clean. Prod. 2019, 236, 117525. [Google Scholar] [CrossRef]
- Lewis, D.; Gattie, D. Pathogen Risks from Applying Sewage Sludge to Land. Environ. Sci. Technol. 2002, 286A–293A. [Google Scholar] [CrossRef]
- Maynaud, G.; Pourcher, A.-M.; Ziebal, C.; Cuny, A.; Druilhe, C.; Steyer, J.-P.; Wéry, N. Persistence and Potential Viable but Non-Culturable State of Pathogenic Bacteria during Storage of Digestates from Agricultural Biogas Plants. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef]
- Coelho, J.J.; Prieto, M.L.; Dowling, S.; Hennessy, A.; Casey, I.; Woodcock, T.; Kennedy, N. Physical-Chemical Traits, Phytotoxicity and Pathogen Detection in Liquid Anaerobic Digestates. Waste Manag. 2018, 78, 8–15. [Google Scholar] [CrossRef]
- European Union. Regulation (EC) No 1069/2009 of the European Parliament and of the Council of 21 October 2009 Laying down Health Rules as Regards Animal By-Products and Derived Products Not Intended for Human Consumption and Repealing Regulation (EC) No 1774/2002 (Animal by-Products Regulation). Off. J. Eur. Union 2009, 52. [Google Scholar] [CrossRef]
- European Union. Commission Regulation (EU) No 142/2011 of 25 February 2011 Implementing Regulation (EC) No 1069/2009 of the European Parliament and of the Council Laying down Health Rules as Regards Animal By-Products and Derived Products Not Intended for Human Consumption and Implementing Council Directive 97/78/EC as Regards Certain Samples and Items Exempt from Veterinary Checks at the Border under That Directive. Off. J. Eur. Union 2011, 54. [Google Scholar] [CrossRef]
- Liu, X.; Lendormi, T.; Lanoisellé, J.-L. A Review of Hygienization Technology of Biowastes for Anaerobic Digestion: Effect on Pathogen Inactivation and Methane Production. Chem. Eng. Trans. 2018, 70, 529–534. [Google Scholar] [CrossRef]
- Vorobiev, E.; Lebovka, N. Processing of Foods and Biomass Feedstocks by Pulsed Electric Energy; Springer International Publishing: Cham, Switzerland, 2020. [Google Scholar]
- Garner, A.L. Pulsed Electric Field Inactivation of Microorganisms: From Fundamental Biophysics to Synergistic Treatments. Appl. Microbiol. Biotechnol. 2019, 1–13. [Google Scholar] [CrossRef]
- Kotnik, T.; Rems, L.; Tarek, M.; Miklavčič, D. Membrane Electroporation and Electropermeabilization: Mechanisms and Models. Annu. Rev. Biophys. 2019, 48. [Google Scholar] [CrossRef]
- Wang, M.-S.; Wang, L.-H.; Bekhit, A.E.-D.A.; Yang, J.; Hou, Z.-P.; Wang, Y.-Z.; Dai, Q.-Z.; Zeng, X.-A. A Review of Sublethal Effects of Pulsed Electric Field on Cells in Food Processing. J. Food Eng. 2018, 223, 32–41. [Google Scholar] [CrossRef]
- Liu, X.; Lendormi, T.; Le Fellic, M.; Lemée, Y.; Lanoisellé, J.-L. Hygienization of Mixed Animal By-Product Using Pulsed Electric Field: Inactivation Kinetics Modeling and Recovery of Indicator Bacteria. Chem. Eng. J. 2019, 368, 1–9. [Google Scholar] [CrossRef]
- Liu, X.; Lendormi, T.; Le Fellic, M.; Lemée, Y.; Lanoisellé, J.-L. Hygienization of Mixed Animal By-Product Using Pulsed Electric Field in a Continuous Treatment System: Synergistic Effect with Ohmic Heating on the Inactivation of Indicator Bacteria. Waste Manag. 2020, 118, 18–26. [Google Scholar] [CrossRef]
- Grim, J.; Malmros, P.; Schnürer, A.; Nordberg, Å. Comparison of Pasteurization and Integrated Thermophilic Sanitation at a Full-Scale Biogas Plant—Heat Demand and Biogas Production. Energy 2015, 79, 419–427. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, Y. Is Anaerobic Digestion a Reliable Barrier for Deactivation of Pathogens in Biosludge? Sci. Total Environ. 2019, 668, 893–902. [Google Scholar] [CrossRef]
- Sobsey, M.D.; Khatib, L.A.; Hill, V.R.; Alocilja, E.; Pillai, S. Pathogens in Animal Wastes and the Impacts of Waste Management Practices on Their Survival, Transport and Fate. In Animal Agriculture and the Environment: National Center for Manure and Animal Waste Management White Papers; Rice, J.M., Caldwell, D.F., Humenik, F.J., Eds.; ASABE: St. Joseph, MI, USA, 2006; pp. 609–666. [Google Scholar] [CrossRef]
- Sahlström, L. A Review of Survival of Pathogenic Bacteria in Organic Waste Used in Biogas Plants. Bioresour. Technol. 2003, 87, 161–166. [Google Scholar] [CrossRef]
- Tallon, P.; Magajna, B.; Lofranco, C.; Leung, K.T. Microbial Indicators of Faecal Contamination in Water: A Current Perspective. Water Air Soil Pollut. 2005, 166, 139–166. [Google Scholar] [CrossRef]
- Somolinos, M.; García, D.; Mañas, P.; Condón, S.; Pagán, R. Effect of Environmental Factors and Cell Physiological State on Pulsed Electric Fields Resistance and Repair Capacity of Various Strains of Escherichia coli. Int. J. Food Microbiol. 2008, 124, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Le Jean, G.; Abraham, G.; Debray, E.; Candau, Y.; Piar, G. Kinetics of Thermal Destruction of Bacillus Stearothermophilus Spores Using a Two Reaction Model. Food Microbiol. 1994, 11, 229–241. [Google Scholar] [CrossRef]
- Abraham, G.; Debray, E.; Candau, Y.; Piar, G. Mathematical Model of Thermal Destruction of Bacillus Stearothermophilus Spores. Appl. Environ. Microbiol. 1990, 56, 3073–3080. [Google Scholar] [CrossRef] [PubMed]
- US FDA. Laboratory Methods—Bacteriological Analytical Manual (BAM). Available online: https://www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm2006949.htm (accessed on 28 April 2017).
- US DA. Laboratory Guidebook—Most Probable Number Procedure and Tables. 2014. Available online: https://www.fsis.usda.gov/news-events/publications/microbiology-laboratory-guidebook (accessed on 29 March 2021).
- Van Boekel, M.A.J.S. On the Use of the Weibull Model to Describe Thermal Inactivation of Microbial Vegetative Cells. Int. J. Food Microbiol. 2002, 74, 139–159. [Google Scholar] [CrossRef]
- Huang, K.; Yu, L.; Liu, D.; Gai, L.; Wang, J. Modeling of Yeast Inactivation of PEF-Treated Chinese Rice Wine: Effects of Electric Field Intensity, Treatment Time and Initial Temperature. Food Res. Int. 2013, 54, 456–467. [Google Scholar] [CrossRef]
- Zhang, Q.; Monsalve-González, A.; Qin, B.-L.; Barbosa-Cánovas, G.V.; Swanson, B.G. Inactivation of Saccharomyces cerevisiae in Apple Juice by Square-Wave and Exponential-Decay Pulsed Electric Fields. J. Food Process Eng. 1994, 17, 469–478. [Google Scholar] [CrossRef]
- Peleg, M. A Model of Microbial Survival after Exposure to Pulsed Electric Fields. J. Sci. Food Agric. 1995, 67, 93–99. [Google Scholar] [CrossRef]
- Liu, X. Hygiénisation par Technologie Électrique des déchets alimentaires en vue de leur méthanisation. Ph.D. Thesis, Université Bretagne Sud (Université Bretagne Loire), Lorient, France, 2019. [Google Scholar]
- Heinz, V.; Alvarez, I.; Angersbach, A.; Knorr, D. Preservation of Liquid Foods by High Intensity Pulsed Electric Fields—Basic Concepts for Process Design. Trends Food Sci. Technol. 2001, 12, 103–111. [Google Scholar] [CrossRef]
- Olsen, J.E.; Jørgensen, J.B.; Nansen, P. On the Reduction of Mycobacterium paratuberculosis in Bovine Slurry Subjected to Batch Mesophilic or Thermophilic Anaerobic Digestion. Agric. Wastes 1985, 13, 273–280. [Google Scholar] [CrossRef]
- Elmerdahl Olsen, J.; Errebo Larsen, H. Bacterial Decimation Times in Anaerobic Digestions of Animal Slurries. Biol. Wastes 1987, 21, 153–168. [Google Scholar] [CrossRef]
- Shih, J.C.H. Ecological Benefits of Anaerobic Digestion. Poult. Sci. 1987, 66, 946–950. [Google Scholar] [CrossRef]
- Forshell, L.P. Survival of Salmonellas and Ascaris suum eggs in a Thermophilic Biogas Plant. In Environment and Animal Health. Proceedings of the 6th International Congress on Animal Hygiene, Skara, Sweden, 14–17 June 1988; Sveriges Lantbruksuniversitet: Skara, Sweden, 1988; Volume II, pp. 612–618. [Google Scholar]
- Kearney, T.E.; Larkin, M.J.; Frost, J.P.; Levett, P.N. Survival of Pathogenic Bacteria during Mesophilic Anaerobic Digestion of Animal Waste. J. Appl. Bacteriol. 1993, 75, 215–219. [Google Scholar] [CrossRef]
- Lund, B.; Jensen, V.F.; Have, P.; Ahring, B. Inactivation of Virus during Anaerobic Digestion of Manure in Laboratory Scale Biogas Reactors. Antonie Leeuwenhoek 1996, 69, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Paavola, T.; Syväsalo, E.; Rintala, J. Co-Digestion of Manure and Biowaste According to the EC Animal By-Products Regulation and Finnish National Regulations. Water Sci. Technol. 2006, 53, 223–231. [Google Scholar] [CrossRef]
- Iranpour, R.; Cox, H.H.J. Evaluation of Thermophilic Anaerobic Digestion Processes for Full-Scale Class A Biosolids Disinfection at Hyperion Treatment Plant. Biotechnol. Bioeng. 2007, 97, 19–39. [Google Scholar] [CrossRef]
- Bagge, E.; Persson, M.; Johansson, K.-E. Diversity of Spore-forming Bacteria in Cattle Manure, Slaughterhouse Waste and Samples from Biogas Plants. J. Appl. Microbiol. 2010, 109, 1549–1565. [Google Scholar] [CrossRef]
- Massé, D.; Gilbert, Y.; Topp, E. Pathogen Removal in Farm-Scale Psychrophilic Anaerobic Digesters Processing Swine Manure. Bioresour. Technol. 2011, 102, 641–646. [Google Scholar] [CrossRef]
- Pandey, P.K.; Soupir, M.L. Escherichia coli Inactivation Kinetics in Anaerobic Digestion of Dairy Manure under Moderate, Mesophilic and Thermophilic Temperatures. AMB Expr. 2011, 1, 18. [Google Scholar] [CrossRef]
- Manser, N.D.; Wald, I.; Ergas, S.J.; Izurieta, R.; Mihelcic, J.R. Assessing the Fate of Ascaris suum Ova during Mesophilic Anaerobic Digestion. Environ. Sci. Technol. 2015, 49, 3128–3135. [Google Scholar] [CrossRef]
- Mazzone, P.; Corneli, S.; Di Paolo, A.; Maresca, C.; Felici, A.; Biagetti, M.; Ciullo, M.; Sebastiani, C.; Pezzotti, G.; Leo, S.; et al. Survival of Mycobacterium avium Subsp. Paratuberculosis in the Intermediate and Final Digestion Products of Biogas Plants. J. Appl. Microbiol. 2018, 125, 36–44. [Google Scholar] [CrossRef]
- Tápparo, D.C.; Viancelli, A.; do Amaral, A.C.; Fongaro, G.; Steinmetz, R.L.R.; Magri, M.E.; Barardi, C.R.M.; Kunz, A. Sanitary Effectiveness and Biogas Yield by Anaerobic Co-Digestion of Swine Carcasses and Manure. Environ. Technol. 2018, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nolan, S.; Waters, N.R.; Brennan, F.; Auer, A.; Fenton, O.; Richards, K.; Bolton, D.J.; Pritchard, L.; O’Flaherty, V.; Abram, F. Toward Assessing Farm-Based Anaerobic Digestate Public Health Risks: Comparative Investigation with Slurry, Effect of Pasteurization Treatments, and Use of Miniature Bioreactors as Proxies for Pathogen Spiking Trials. Front. Sustain. Food Syst. 2018, 2. [Google Scholar] [CrossRef]
- Jepsen, S.-E.; Krause, M.; Grüttner, H. Reduction of Fecal Streptococcus and Salmonella by Selected Treatment Methods for Sludge and Organic Waste. Water Sci. Technol. 1997, 36, 203–210. [Google Scholar] [CrossRef]
- De Luca, G.; Zanetti, F.; Fateh-Moghadm, P.; Stampi, S. Occurrence of Listeria monocytogenes in Sewage Sludge. Zent. Hyg. Umweltmed. 1998, 201, 269–277. [Google Scholar]
- Aitken, M.D.; Sobsey, M.D.; Blauth, K.E.; Shehee, M.; Crunk, P.L.; Walters, G.W. Inactivation of Ascaris suum and Poliovirus in Biosolids under Thermophilic Anaerobic Digestion Conditions. Environ. Sci. Technol. 2005, 39, 5804–5809. [Google Scholar] [CrossRef]
- Aitken, M.D.; Sobsey, M.D.; Shehee, M.; Blauth, K.E.; Hill, V.R.; Farrell, J.B.; Nappier, S.P.; Walters, G.W.; Crunk, P.L.; Van Abel, N. Laboratory Evaluation of Thermophilic-Anaerobic Digestion to Produce Class A Biosolids. 2. Inactivation of Pathogens and Indicator Organisms in a Continuous-Flow Reactor Followed by Batch Treatment. Water Environ. Res. 2005, 77, 3028–3036. [Google Scholar] [CrossRef]
- Iranpour, R.; Cox, H.H.J.; Oh, S.; Fan, S.; Kearney, R.J.; Abkian, V.; Haug, R.T. Thermophilic-Anaerobic Digestion to Produce Class A Biosolids: Initial Full-Scale Studies at Hyperion Treatment Plant. Water Environ. Res. 2006, 78, 170–180. [Google Scholar] [CrossRef]
- Iranpour, R.; Cox, H.H.J.; Fan, S.; Abkian, V.; Minamide, T.; Kearney, R.J.; Haug, R.T. Full-Scale Class A Biosolids Production by Two-Stage Continuous-Batch Thermophilic Anaerobic Digestion at the Hyperion Treatment Plant, Los Angeles, California. Water Environ. Res. 2006, 78, 2244–2252. [Google Scholar] [CrossRef]
- Lloret, E.; Salar, M.J.; Blaya, J.; Pascual, J.A. Two-Stage Mesophilic Anaerobic–Thermophilic Digestion for Sludge Sanitation to Obtain Advanced Treated Sludge. Chem. Eng. J. 2013, 230, 59–63. [Google Scholar] [CrossRef]
- Scaglia, B.; D’Imporzano, G.; Garuti, G.; Negri, M.; Adani, F. Sanitation Ability of Anaerobic Digestion Performed at Different Temperature on Sewage Sludge. Sci. Total Environ. 2014, 466–467, 888–897. [Google Scholar] [CrossRef]
- Engeli, H.; Edelmann, W.; Fuchs, J.; Rottermann, K. Survival of Plant Pathogens and Weed Seeds during Anaerobic Digestion. Water Sci. Technol. 1993, 27, 69–76. [Google Scholar] [CrossRef]
- Ryckeboer, J.; Cops, S.; Coosemans, J. The Fate of Plant Pathogens and Seeds During Anaerobic Digestion and Aerobic Composting of Source Separated Household Wastes. Compost Sci. Util. 2002, 10, 204–216. [Google Scholar] [CrossRef]
- Termorshuizen, A.J.; Volker, D.; Blok, W.J.; ten Brummeler, E.; Hartog, B.J.; Janse, J.D.; Knol, W.; Wenneker, M. Survival of Human and Plant Pathogens during Anaerobic Mesophilic Digestion of Vegetable, Fruit, and Garden Waste. Eur. J. Soil Biol. 2003, 39, 165–171. [Google Scholar] [CrossRef]
- Schnürer, A.; Schnürer, J. Fungal Survival during Anaerobic Digestion of Organic Household Waste. Waste Manag. 2006, 26, 1205–1211. [Google Scholar] [CrossRef]
- Wagner, A.O.; Gstraunthaler, G.; Illmer, P. Survival of Bacterial Pathogens during the Thermophilic Anaerobic Digestion of Biowaste: Laboratory Experiments and in Situ Validation. Anaerobe 2008, 14, 181–183. [Google Scholar] [CrossRef]
- Rounsefell, B.D.; O’Sullivan, C.A.; Chinivasagam, N.; Batstone, D.; Clarke, W.P. Fate of Pathogen Indicators in a Domestic Blend of Food Waste and Wastewater through a Two-Stage Anaerobic Digestion System. Water Sci. Technol. 2013, 67, 366–373. [Google Scholar] [CrossRef]
- Seruga, P.; Krzywonos, M.; Paluszak, Z.; Urbanowska, A.; Pawlak-Kruczek, H.; Niedźwiecki, Ł.; Pińkowska, H. Pathogen Reduction Potential in Anaerobic Digestion of Organic Fraction of Municipal Solid Waste and Food Waste. Molecules 2020, 25, 275. [Google Scholar] [CrossRef]
- Qi, G.; Pan, Z.; Yamamoto, Y.; Andriamanohiarisoamanana, F.J.; Yamashiro, T.; Iwasaki, M.; Ihara, I.; Tangtaweewipat, S.; Umetsu, K. The Survival of Pathogenic Bacteria and Plant Growth Promoting Bacteria during Mesophilic Anaerobic Digestion in Full-Scale Biogas Plants. Anim. Sci. J. 2019, 90, 297–303. [Google Scholar] [CrossRef]
- Magnus, C.A.; Ingledew, W.M.; McCurdy, A.R. Thermal Resistance of Streptococci Isolated from Pasteurized Ham. Can. Inst. Food Sci. Technol. J. 1986, 19, 62–67. [Google Scholar] [CrossRef]
- Cunault, C.; Pourcher, A.M.; Burton, C.H. Using Temperature and Time Criteria to Control the Effectiveness of Continuous Thermal Sanitation of Piggery Effluent in Terms of Set Microbial Indicators. J. Appl. Microbiol. 2011, 111, 1492–1504. [Google Scholar] [CrossRef]
- Bischof, J.C. Thermal Stability of Proteins. Ann. N.Y. Acad. Sci. 2005, 1066, 12–33. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Xie, S.H.; Dennehy, C.; Lawlor, P.G.; Hu, Z.H.; Wu, G.X.; Zhan, X.M.; Gardiner, G.E. Inactivation of Pathogens in Anaerobic Digestion Systems for Converting Biowastes to Bioenergy: A Review. Renew. Sustain. Energy Rev. 2020, 120, 109654. [Google Scholar] [CrossRef]
- Cebrián, G.; Condón, S.; Mañas, P. Physiology of the Inactivation of Vegetative Bacteria by Thermal Treatments: Mode of Action, Influence of Environmental Factors and Inactivation Kinetics. Foods 2017, 6, 107. [Google Scholar] [CrossRef] [PubMed]
- García, D.; Gómez, N.; Mañas, P.; Raso, J.; Pagán, R. Pulsed Electric Fields Cause Bacterial Envelopes Permeabilization Depending on the Treatment Intensity, the Treatment Medium PH and the Microorganism Investigated. Int. J. Food Microbiol. 2007, 113, 219–227. [Google Scholar] [CrossRef]
- Sharma, P.; Bremer, P.; Oey, I.; Everett, D.W. Bacterial Inactivation in Whole Milk Using Pulsed Electric Field Processing. Int. Dairy J. 2014, 35, 49–56. [Google Scholar] [CrossRef]
- Martín, O.; Qin, B.L.; Chang, F.J.; Barbosa-Cánovas, G.V.; Swanson, B.G. Inactivation of Escherichia coli in Skim Milk by High Intensity Pulsed Electric Fields. J. Food Process Eng. 1997, 20, 317–336. [Google Scholar] [CrossRef]
- Grahl, T.; Märkl, H. Killing of Microorganisms by Pulsed Electric Fields. Appl. Microbiol. Biotechnol. 1996, 45, 148–157. [Google Scholar] [CrossRef]
- Ho, S.Y.; Mittal, G.S.; Cross, J.D.; Griffiths, M.W. Inactivation of Pseudomonas fluorescens by High Voltage Electric Pulses. J. Food Sci. 1995, 60, 1337–1340. [Google Scholar] [CrossRef]
- Donsì, G.; Ferrari, G.; Pataro, G. Inactivation Kinetics of Saccharomyces cerevisiae by Pulsed Electric Fields in a Batch Treatment Chamber: The Effect of Electric Field Unevenness and Initial Cell Concentration. J. Food Eng. 2007, 78, 784–792. [Google Scholar] [CrossRef]
- Popat, S.C.; Yates, M.V.; Deshusses, M.A. Kinetics of Inactivation of Indicator Pathogens during Thermophilic Anaerobic Digestion. Water Res. 2010, 44, 5965–5972. [Google Scholar] [CrossRef]
- Cunault, C. Développement d’une Méthode D’hygiénisation Thermique des Effluents Au Moyen D’échangeurs de Chaleur (Application Au Lisier Porcin). Ph.D. Thesis, Université Rennes, Rennes, France, 2012. [Google Scholar]
- He, P.; Zhou, Y.; Shao, L.; Huang, J.; Yang, Z.; Lü, F. The Discrepant Mobility of Antibiotic Resistant Genes: Evidence from Their Spatial Distribution in Sewage Sludge Flocs. Sci. Total Environ. 2019, 697, 134176. [Google Scholar] [CrossRef]
- Derongs, L.; Druilhe, C.; Ziebal, C.; Le Maréchal, C.; Pourcher, A.-M. Characterization of Clostridium perfringens Isolates Collected from Three Agricultural Biogas Plants over a One-Year Period. IJERPH 2020, 17, 5450. [Google Scholar] [CrossRef]
- Orlando, M.-Q.; Borja, V.-M. Pretreatment of Animal Manure Biomass to Improve Biogas Production: A Review. Energies 2020, 13, 3573. [Google Scholar] [CrossRef]
- Luste, S.; Luostarinen, S. Anaerobic Co-Digestion of Meat-Processing by-Products and Sewage Sludge—Effect of Hygienization and Organic Loading Rate. Bioresour. Technol. 2010, 101, 2657–2664. [Google Scholar] [CrossRef]
- Liu, X.; Souli, I.; Chamaa, M.-A.; Lendormi, T.; Sabourin, C.; Lemée, Y.; Boy, V.; Chaira, N.; Ferchichi, A.; Morançais, P.; et al. Effect of Thermal Pretreatment at 70 °C for One Hour (EU Hygienization Conditions) of Various Organic Wastes on Methane Production under Mesophilic Anaerobic Digestion. AIMS Environ. Sci. 2018, 5, 117–129. [Google Scholar] [CrossRef]
- Chamaa, M.A. Couplage de la Méthanisation et des Électrotechnologies: Intentisification de la Production de Biogaz et du Séchage du Digestat. Ph.D. Thesis, Université Bretagne Sud, Lorient, France, 2017. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).