Abstract
Numerous studies have been conducted on spent lithium-ion batteries (LIBs) recycled from electric vehicles. Research on pre-processing techniques to safely disassemble spent LIB packs has mainly focused on water-based discharge methods, such as salt-water discharge. However, salt-water discharge corrodes the electrodes and case, causing internal contamination. Therefore, we propose an electrical discharge process that is suitable for the direct recycling and safe disassembly of spent Li-ion batteries. Spent LIBs from electric vehicles (EV) that were scrapped after EV operation were recovered and electrochemically discharged to voltages of 0, 1, 2, and 2.5 V. These discharged spent LIBs were analyzed through X-ray diffraction, scanning electron microscopy, and electrochemical impedance spectroscopy. The spent LIB with a state-of-health (SoH) of 66.8% exhibited significantly increased swelling and bulging when over-discharged. Notably, the discharging of the spent battery to 0 V increased the thickness of the cell by 115%, which could result in a fire and/or explosion. After being discharged to 0 V, the voltage was able to recover to 2.689 V. The appropriate voltage for the discharge process was estimated to be 2.5 V. The proposed electrical discharge process will be suitable for the direct recycling of spent LIBs in the form of pouch cells.
1. Introduction
The disposal of spent LIBs generated from electric vehicles (EVs) has rapidly increased since their introduction 10 years ago [1]. The two main approaches for processing the spent LIBs of EVs are reuse and recycling [2,3]. Many studies have shown that the battery healthcare system can effectively estimate a spent battery’s state of health (SoH) for reuse. However, although EV batteries can be reused in second-life solutions for several years after retirement from EV applications, these batteries will eventually be sent to recycling facilities to collect valuable resources such as lithium, cobalt, nickel, manganese, and even natural graphite from them [2].
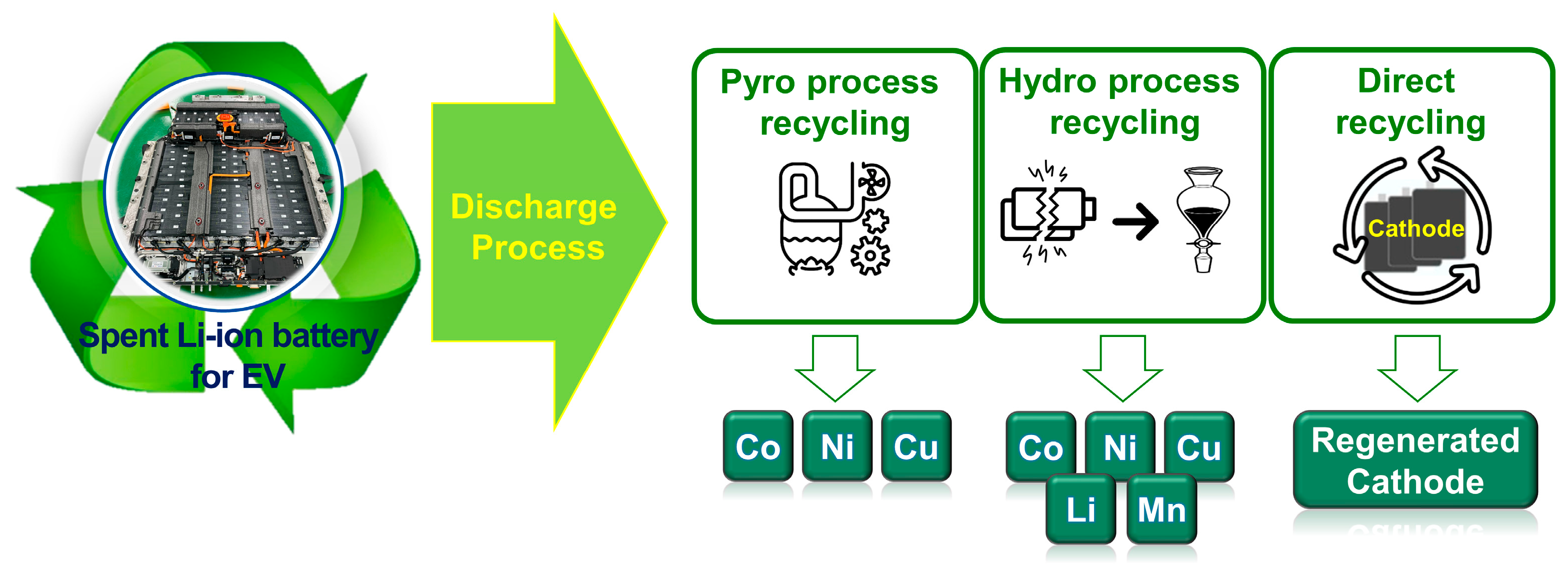
Because there are limited reserves of the rare metals found in spent LIBs [4], researchers are investigating the development of systematic and safe recycling systems. Thus far, three types of recycling methods for spent LIBs have been considered, namely the pyrometallurgy process, hydrometallurgy process, and direct recycling, as shown in Figure 1 [5]. The pyrometallurgy process is widely used for the commercial recovery of cobalt. However, it has the disadvantages of high carbon emissions and difficulty in recovering Li [6,7,8]. In contrast, the hydrometallurgy process is able to recover more Li and Mn than the pyrometallurgy process [9,10,11]; however, it generates large amounts of wastewater and contaminated acid fumes that can severely pollute the environment. In addition, the current important focus is on chemical facilities, research and industry pursuing sustainable and environmentally friendly processes and methods for the recycling of spent batteries [12,13,14,15,16]. Therefore, compared to the pyrometallurgy and hydrometallurgy processes, the direct recycling of batteries [17,18], especially from spent EVs, has gained significant attention as an environmentally friendly approach [19,20]. The discharging of spent LIBs is an essential process in the direct recycling of spent LIBs. This critical step prevents explosions, fires, and toxic gas emissions [21,22,23] that occur as a result of the inadequate sealing and short circuiting of high-energy LIBs. These explosions, fires, and toxic gas emissions can harm human health and the environment and damage the infrastructure necessary for recycling spent LIBs. If the spent LIB materials are damaged, their value for recycling and regeneration may be reduced [24]. The LithoRec project report describes the current discharge technologies used in the LIB recycling process [25]. The report briefly explains the several discharge technologies necessary for both safe and functional LIB recycling [26]. Two short-circuit-based discharge methods are widely employed, namely the resistor-based discharge, which connects both ends of the battery to consume the residual electric energy, and salt-solution-immersion discharge, which immerses the LIB in ionically conducting salt water to form a short circuit and thus effectively induce the electrochemical discharge of the LIB [27]. Among a variety of ionic salts, sodium chloride solutions have been widely used in typical chemical discharge processes [28,29]. Li et al. [30] analyzed the chemical components of wastewater from the salt-water discharge process. They detected “high” levels of aluminum and iron and “moderate” levels of cobalt, lithium, copper, calcium, and manganese. In addition, considerable amounts of zinc, barium, and vanadium were also detected. High concentrations of P were also observed, indicating that the corrosion of the metal collectors and sealing compartments caused electrolyte leakage (i.e., LiPF6). Moreover, metal hydroxides, salts, and metal oxides were also present in the wastewater. Thus, a suitable discharge technology must be developed to prevent the salt-water discharge from damaging the LIB and active materials, which will result in increased costs and additional steps in the recycling process. A potential solution to ensure the safe and effective recycling of spent LIBs from EVs is the electrical discharge method. However, to date, there has been a significant gap in the literature regarding research on the electrical discharge method for spent LIBs. This lack of research hinders the development of appropriate disposal methods for spent LIBs and presents potential environmental and safety hazards. Therefore, research is needed to investigate the electrical discharge method and its effectiveness for the safe disposal of spent LIBs from EVs.
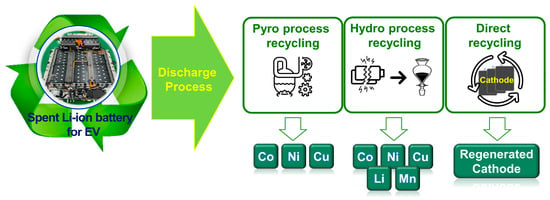
Figure 1.
Schematic illustrations describing the three types of recycling methods for spent LIBs: pyrometallurgy process, hydrometallurgy process, and direct recycling.
In this study, we recovered spent LIBs used in two EVs and observed the state of the modules and cells through electrical discharge. Additionally, we performed a comparative experiment on the salt-water discharge of pouch-type LIBs because they are primarily used in EVs. After discharging the spent LIBs to various voltages, the crystal structures of the cathode and anode were analyzed. We examined the problems caused by over-discharge based on SoH and determined the appropriate discharge voltage. The investigation showed that swelling due to over-discharge became more rapid as SoH decreased. Furthermore, cell volume expansion caused by swelling increased the internal pressure of the cell packaged in the module, leading to a risk of explosion. Therefore, it is essential to discharge pouch cells to an appropriate voltage that can prevent the pouch swelling of LIBs. The results of this study indicate the proper voltage for electrical discharge that is deemed to be applicable for environmentally friendly recycling processes.
2. Experiment
2.1. Disassembly of Spent LIB Packs from EVs
Spent LIB packs were disassembled from two Renault Samsung SM3 ZEs (same model of EV). The spent LIB packs were disassembled to obtain the modules and cells, as shown in Figure 2. Each LIB pack comprised 24 2P4S modules, totaling 192 cells.

Figure 2.
Photographic images of (a) the spent LIB packs from the EV, (b) 24 2P4S modules in the LIB pack, (c) 2P4S modules, (d) single LIB cells disassembled from a 2P4S module, and (e) experimental flowchart.
The two LIB packs with different periods of use were prepared to analyze spent LIBs with different SoHs. Electrical tests could not be performed on the pack units due to safety concerns.
2.2. Discharge Process Using Salt Water
Three LIB cells were prepared to examine the salt-water discharge as a control. For the salt water, NaCl (Daejung, Republic of Korea) was dissolved in deionized (DI) water at 40 °C to a concentration of 10 wt%. The coin (CR 2032, 3 V, Duracell®, Bethel, CT, USA), cylindrical (26650, 3.7 V, Light brothers), and pouch cells (a LIB disassembled from the EV, LG) were subjected to the discharge process in salt water.
2.3. Module Characterization
The SoH of the modules was measured by referring to the manufacturer’s specifications. The specifications of the LIB pack were: 35.9 kWh, a nominal voltage of 360 V, and 74 Ah. The LIB pack comprised 24 modules, and a single module had a 2P4S configuration with eight cells connected in two parallel and four series groups. Accordingly, based on the cut-off voltage of 4.15 to 3 V per cell, the single module was charged to 16.6 V and then discharged to 12 V at a 1/3 C-rate.
2.4. Cell Characterization
The cells were discharged using a charge–discharge testing system (LBT-60V, Arbin, College Station, TX, USA) to 2.5, 2, 1, and 0 V at a 1/3 C-rate, similar to the module characterization above. The constant current/constant voltage (CC/CV) method was used for the discharge. In the CV part, the cell was discharged until the current reached 0.37 A.
2.5. Sample Characterization after Electrochemical Discharge
The swelling thickness of the cells discharged at various voltages was examined by using vernier calipers. The internal resistance was analyzed via electrochemical impedance spectroscopy (EIS, Reference3000, Gamry, Warminster, PA, USA). Electrochemical impedance spectroscopy was conducted in the galvanostatic mode owing to the high capacity of the EV LIB cells in the range of 3 kHz to 0.1 Hz. The discharged cells were disassembled in a glove box filled with pure Ar to recover the cathode and anode for further analysis. X-ray diffraction (XRD, D MAX2200, Rigaku, Tokyo, Japan, Cu Kα, 40 kV/30 mA) was conducted on the recovered cathodes and anodes. The microstructure was analyzed via scanning electron microscopy (SEM, Inspect F50, FEI, Hillsboro, OR, USA).
3. Results and Discussion
Table 1 lists various discharge methods that can be used for discharging spent LIBs. Companies have applied thermal treatments [31], salt bath-based systems [30,32,33,34], or an external circuit. Furthermore, NaCl and other alternative salts have been tested for discharging portable batteries [22]. Among these, NaCl was found to provide the best discharge profile. In addition, Na2S was found to be an adequate discharge medium, whereas solutions containing MgSO4 were unable to fully discharge the battery [22]. In some cases, batteries can be discharged using liquid nitrogen or ultra-low temperature methods in a vacuum atmosphere [21]. However, salt-water discharge is still considered the most industrially appropriate method, and therefore we conducted experiments to compare it to other discharge methods.

Table 1.
Discharge methods for recycling.
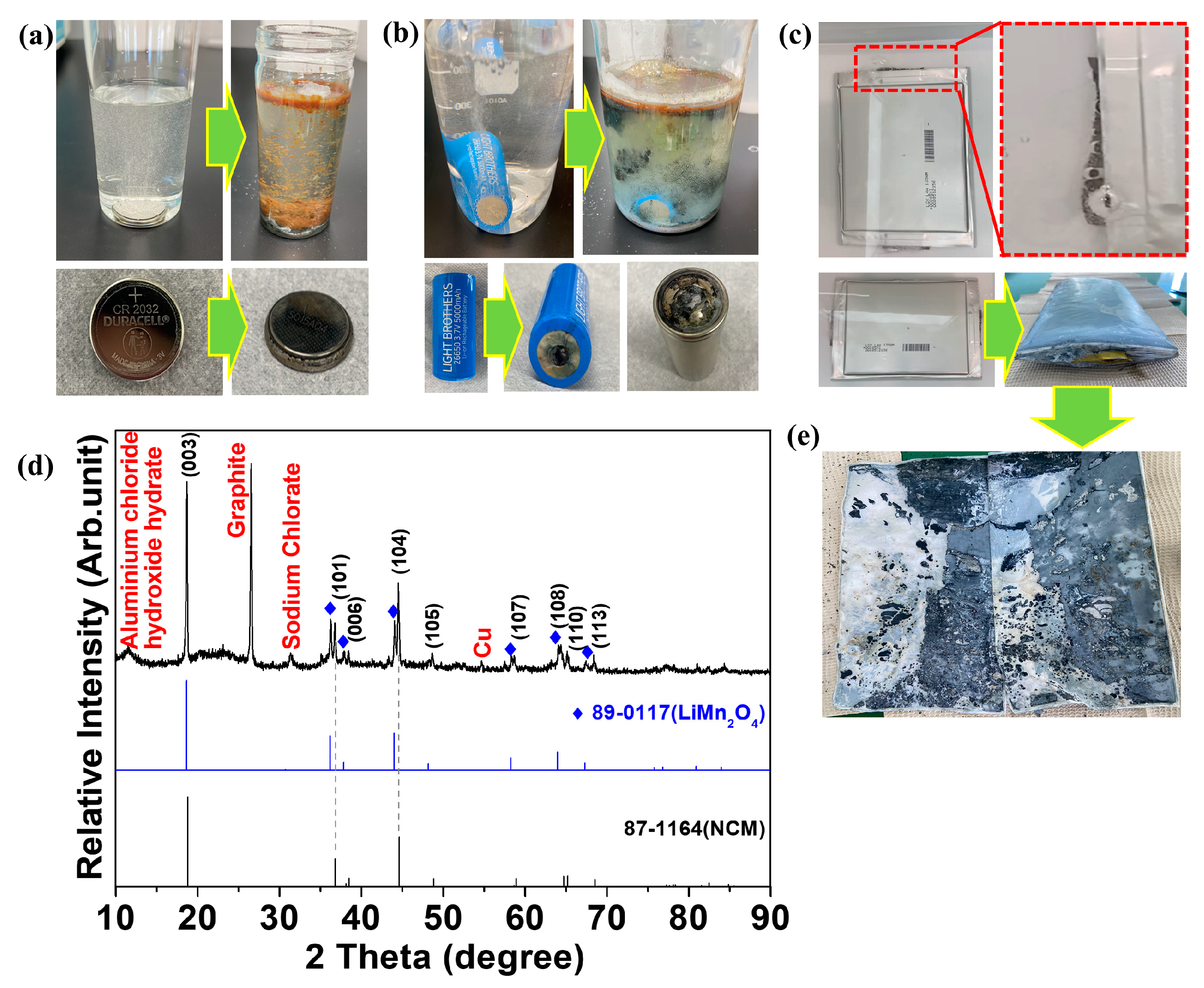
Figure 3 shows the results obtained by discharging various cells using salt water. Figure 3a–c show photos of salt-water discharge applied to coin, cylindrical, and pouch cells, respectively. Chlorine gas was generated as soon as the cells were immersed in salt water. The coin cell shows no significant damage to the cell after being discharged in salt water (Figure 3a). However, as reported by Li et al. [30], various metal oxides were precipitated in the salt water. After being discharged into salt water, the positive electrode in the cylindrical cell was corroded and a hole developed (Figure 3b). This intensive corrosion could be caused by the chlorine gas generated by the positive electrode. Similar to the coin cell, a metal oxide precipitate was observed in the salt water. Moreover, the pungent smell from the cell indicated that the electrolyte solution had leaked. Similar to the cylindrical cell, the positive electrode of the disassembled pouch cell was intensely corroded after being discharged in salt water (Figure 3c). Consequently, the salt water that entered the interior of the pouch cell caused numerous chemical reactions, resulting in volume expansion inside the cell [21,22].
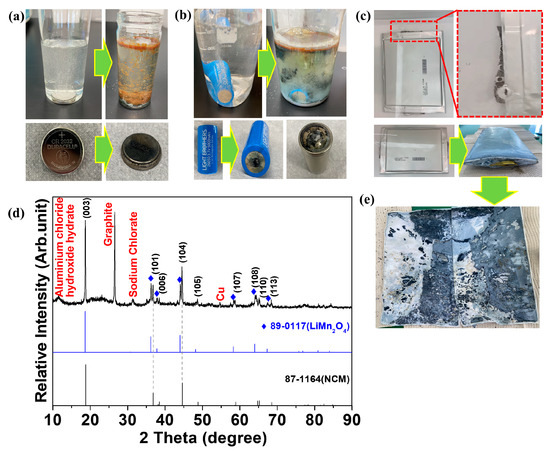
Figure 3.
Discharge in salt water (10% NaCl) of a (a) coin cell, (b) cylindrical cell, and (c) pouch cell. The green arrows indicate the changes after immersing the cells in salt water. (d) X-ray diffraction analysis of the cathode from the swelled pouch cell after the discharge. (e) The photograph of the disassembled pouch cell after the salt water-based discharge.
The cells were then disassembled to examine their internal state. Because the cells were intensely corroded, damage to the current collector occurred simultaneously with the detachment of the active material. Copper and aluminum collectors could not be found in some of the pouch cells, and only the separator and a contaminated lump of powder were observed, as shown in Figure 3e. X-ray diffraction was conducted to identify the components of this contaminated powder. Figure 3d shows the XRD results of the powder recovered from the pouch cell discharged in salt water. Sodium chlorate and aluminum chloride hydroxide hydrate corroded by salt water were identified. The anode electrode was also corroded, and Cu (current collector) and graphite (anode material) peaks were observed. Salt-water discharge requires additional processes, such as addressing environmental pollution caused by wastewater and the handling of corroded pollutants. Thus, we confirmed that salt-water discharge is unsuitable for the recycling of pouch cells.
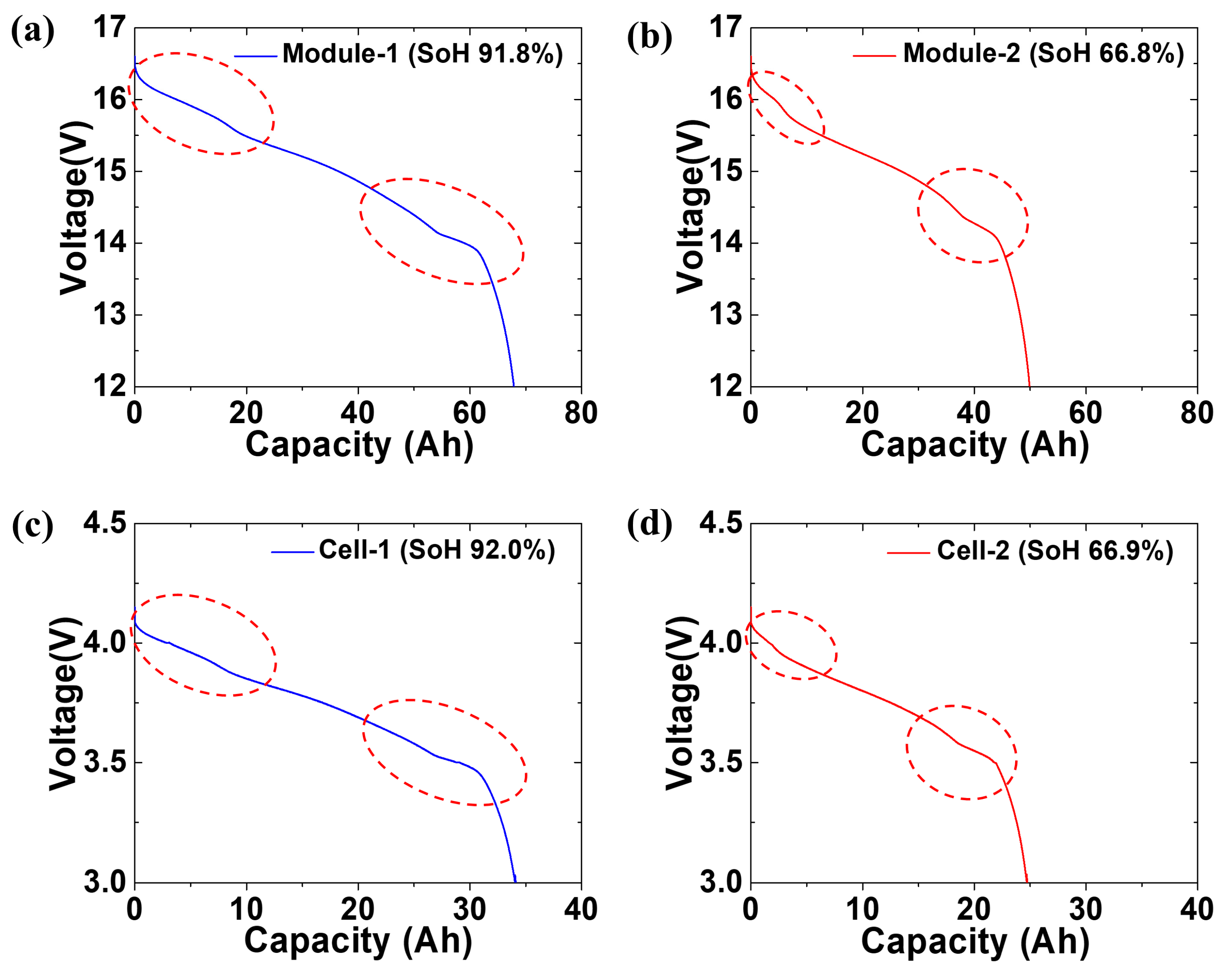
Figure 4 shows the SoH estimation of the spent LIB pack modules and cells recovered from two vehicles. As mentioned in the experimental section, the spent LIB packs were recovered from operating Renault Samsung SM3 ZEs. The modules and cells were prepared before measuring the SoH of the spent LIB packs retrieved from the two vehicles with different periods of use. The battery packs were disassembled into modules and cells, as shown in Figure 2. The LIB pack used in the SM3 EV contained 24 2P4S modules. Based on the specifications, the capacity of the module unit at 100% SoH was 74 Ah. The capacity of the cell unit at 100% SoH was 37 Ah owing to the 2P4S connection of eight cells. The SM3 ZE vehicles with different periods of use were designated 1 and 2, and the recovered modules and cells were numbered 1 and 2. Thus, the module and cell recovered from Vehicle no. 1 were labeled module-1 and cell-1, respectively, and the module and cell recovered from Vehicle no. 2 were labeled module-2 and cell-2, respectively. Figure 4a shows a graph of the discharge profile of module-1. The discharge capacity of module-1 was 67.9 Ah. This corresponded to a SoH of 91.8%. A sudden voltage decrease during the discharge process was not observed. However, two changes in slope during the discharge process were observed. These changes in slope are attributed to the redox coupling of different types of transition metals. Even though the proprietary information of the cathodes was unavailable, the cathode materials used were presumed to be LiNixCoyMn1-x-yO2 (NCM) or a blended composition of NCM and LiMn2O4. Blended cathode materials have previously been developed as a part of the cathode material design method for hybrid, plug-in hybrid, and battery EVs (HEVs, PHEVs, and BEVs, respectively) [35,36,37]. A blended cathode is composed of two or more distinct active material compounds [38]. Numerous studies have reported on the intricate behavior related to Li insertion and extraction kinetics in blended systems [36,39]. When two or more parent cathode materials are physically mixed, they affect the cell state of charge and Li diffusion properties and play a role in coulombic capacity during the cycling process. As a result of these unique characteristics, blended cathode materials are a topic of intense investigation. Figure 4b shows a graph of the discharge profile of module-2. The discharge capacity of module-2 was 49.4 Ah, corresponding to a SoH of 66.8%. In module-2, similar changes in slope were observed during the discharge process. Figure 3c shows the discharge profile graph of cell-1 disassembled from module-1. The discharge capacity of cell-1 was 34 Ah, corresponding to a SoH of 92%, similar to module-1. Figure 4d shows the discharge profile graph of cell-2 disassembled from module-2. The discharge capacity of module-2 was 24.8 Ah, corresponding to a SoH of 66.9%. The difference in SoH between module-1 and module-2 and between cell-1 and cell-2 indicated that the LIB performance varied depending on the operating conditions of EV.
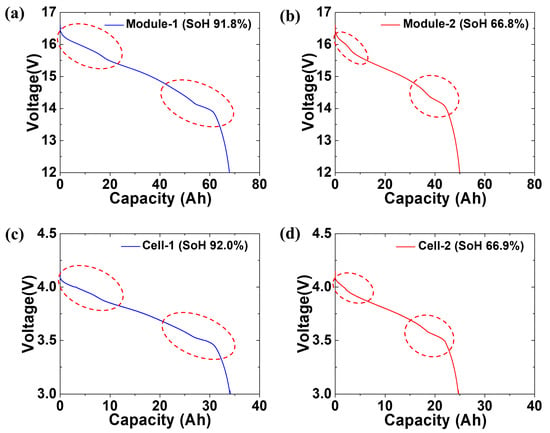
Figure 4.
Polarization curves measured during the electrochemical discharge process of (a) module-1, (b) module-2, (c) cell-1, and (d) cell-2. Dotted circles indicate the redox reactions during the discharge.
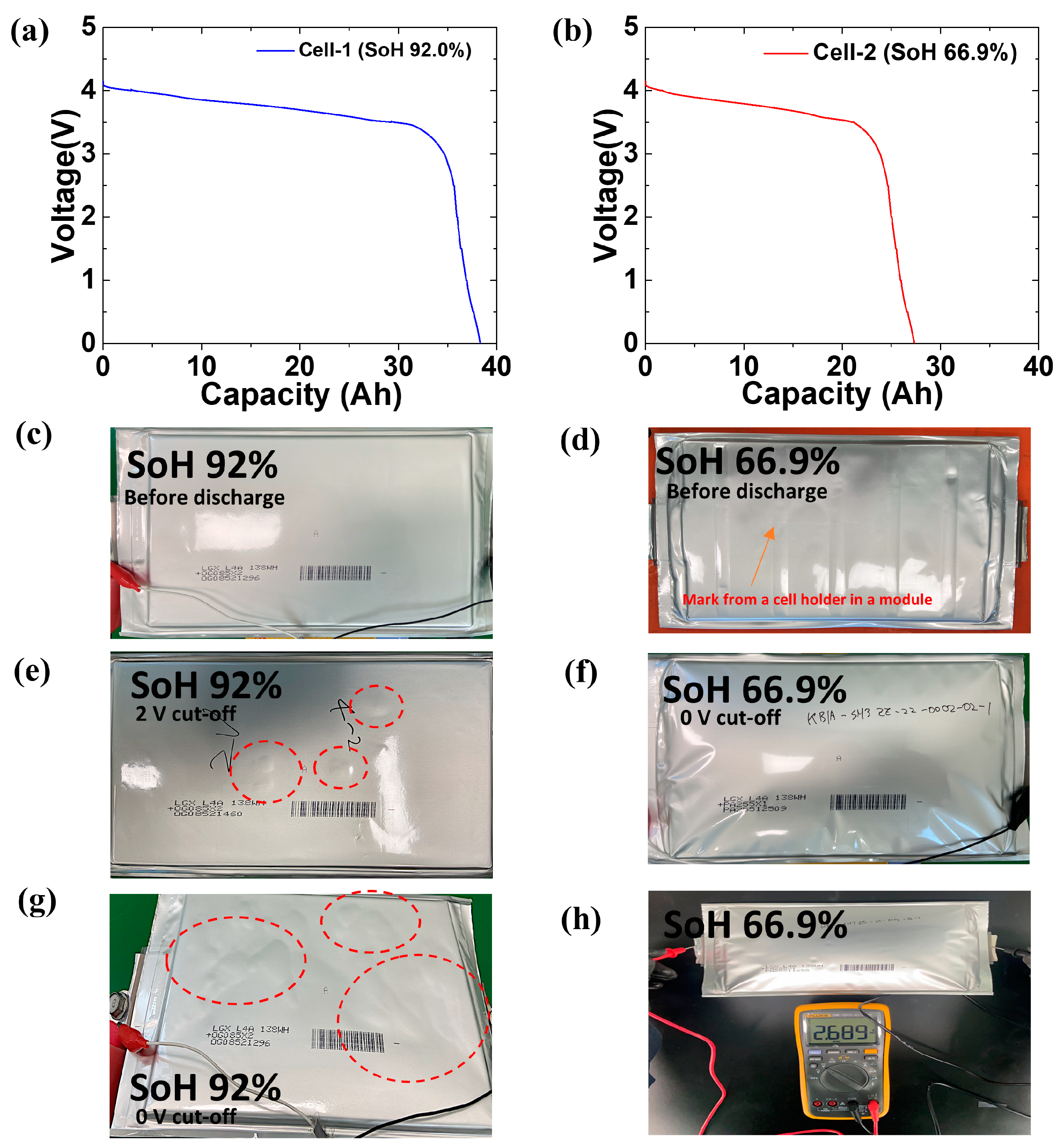
Figure 5 shows the results of discharging the cells to 0 V. Figure 5a shows the discharge profile graph of cell-1 to 0 V. No voltage changes or anomalies owing to side reactions were observed, and the discharge capacity was 38.3 Ah, higher than that shown in Figure 4c. Figure 5b shows the discharge profile graph of cell-2. The discharge capacity of cell-2 was 27.3 Ah. Both cell-1 and cell-2 were discharged from 4.15 V to 0 V, and bulges on cell-1 and intensive swelling on cell-2 were observed. Because the cells were discharged to 0 V, the temperature outside the pouch cell rose to a maximum of 43 °C for both cell-1 and cell-2. Figure 5c,d show photos of the initial state of 4.15 V. Cell-1 (92% SoH) appeared as the initial manufacturing state, and volume expansion was not observed (Figure 5c). Cell-2 (66.9% SoH) had the appearance of swelling, as indicated in Figure 5d, and the exterior of the cell-2 pouch was deformed similar to the shape of the cell holder in the module case. The discharge process was then performed on the cells to 2 V (cut-off). Bulges started forming when the discharge potential reached approximately 2 V (Figure 5e). In cell-2, slight swelling had already progressed, as shown in Figure 5d in the initial state. The bulges enlarged as cell-1 reached the discharge potential of 0 V (Figure 5g). Accordingly, cell-2 (66.9% SoH) exhibited more severe swelling, as shown in Figure 5f. It is worth noting that the intensive swelling of the cell-2 pouch smoothed out the deformation and wrinkles on its surface. The swollen pouch could not be reassembled into the module. We speculate that discharging the cells to 0 V for recycling may cause severe hazards if the cells are installed and fixed in the module or pack. The volumetric expansion of the cell in the module would lead to an explosion or ignition, which would threaten the safety of researchers and could damage the recycling equipment. The cell’s resting potential was measured by using a multimeter after discharging cell-2 to 0 V. As shown in Figure 5h, the resting potential was 2.689 V after resting for 24 h, thus exhibiting the most stable potential difference between the cathode and anode materials. Therefore, we determined that 2.5 V is an appropriate cut-off potential for the discharge.
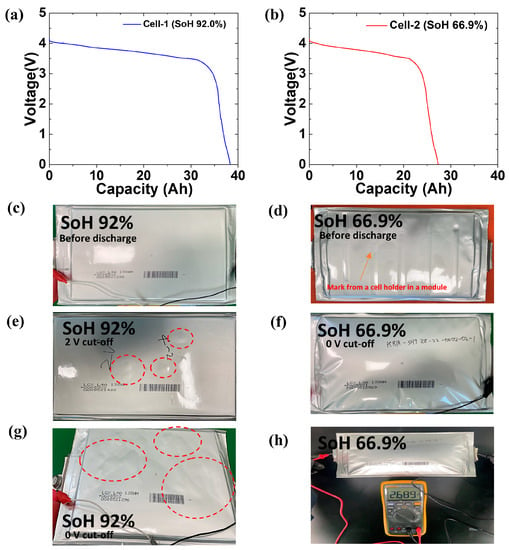
Figure 5.
Polarization curve showing the discharge to 0 V of (a) cell-1 and (b) cell-2. Optical image showing the (c) initial state of cell-1, (d) initial state of cell-2, (e) discharge to 2 V of cell-1, (f) discharge to 0 V on cell-2, (g) discharge to 0 V cell-1 (red circles indicate the bulges on the surface of a cell-1 pouch). (h) Cell potential measurement after 24 h from the discharge to 0 V on a cell-2 pouch.
Because cell-2 exhibited a lower SoH and more intensive swelling than cell-1, cell-2 (66.9% SoH), which is suitable for recycling, was subjected to further analysis. Table 2 presents the changes in the outer thickness of the cell based on the discharge voltage. Cell-1 and cell-2 were discharged to 2.5, 2, 1, and 0 V. Once the cells reached the discharge cut-off potential, the cell thickness was measured. Compared to the as-disassembled cell, no significant change in thickness was observed when discharged to 2.5 V. Both cells exhibited an increase in thickness when the cell potential was below 2 V. Unlike cell-1, which exhibited a decrease in thickness of approximately 19 %, the increase in thickness of cell-2 was approximately 115% at 0 V. This indicates that the LiB cells with low SoH may be prone to swelling under over-discharged potential.

Table 2.
Thicknesses of LIB cell-1 and LIB cell-2 when discharged to various cut-off potentials.
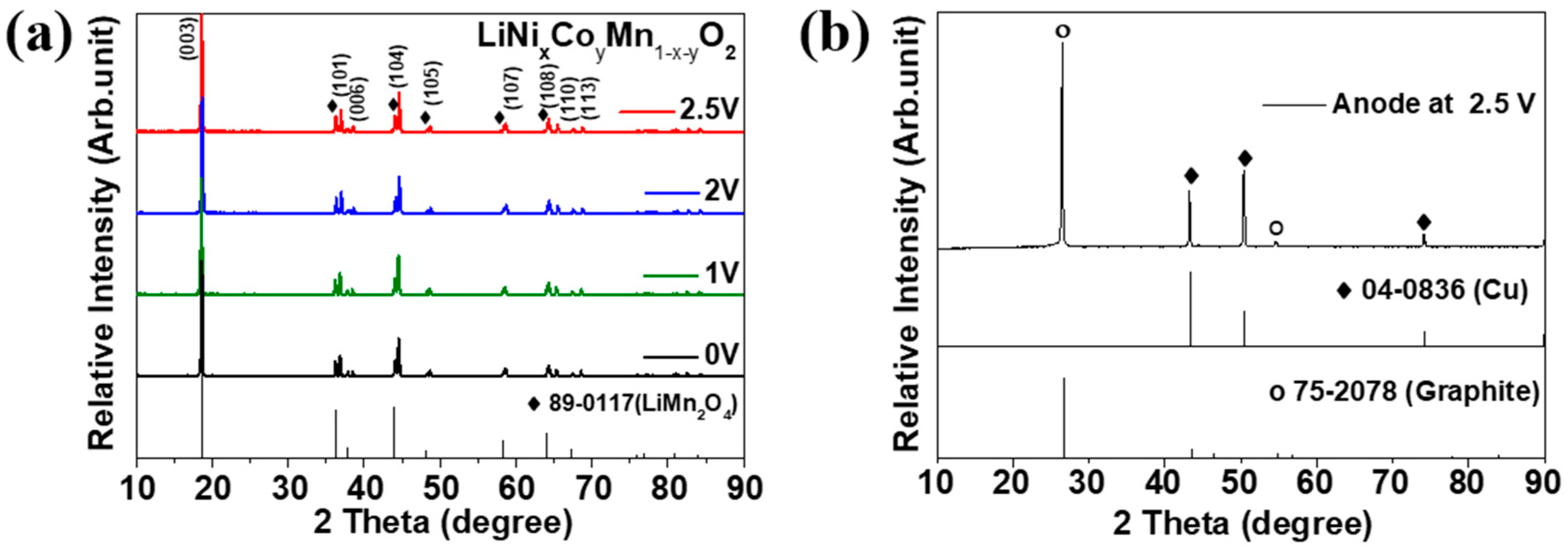
Figure 6 shows SEM images of the recovered cathode discharged to different voltages. According to the SEM images of the microstructure, no anomalies were observed on the surface. Although the electrodes were discharged to each cut-off voltage of 2.5, 2, 1, and even 0 V, no significant cracks or detachments were observed in the electrode layer composed of the active materials and carbon black used as the conductive agent. The discharge process for battery recycling did not significantly change the structure of the electrode layer despite the swelling of the battery. Thus, we cautiously speculate that the swelling of the battery owing to over-discharge to 0 V may result from the decomposition of the electrolyte in spent batteries.

Figure 6.
Scanning electron microscopy images of the recovered cathodes of LIB cell-2 discharged to different voltages: (a) 2.5 V, (b) 2 V, (c) 1 V, and (d) 0 V.
Figure 7 shows the XRD results on the recovered electrodes discharged to each voltage. Figure 7a shows the XRD results of the recovered cathode. No secondary phase was observed at 2.5, 2, 1, and 0 V. Even when cell-2 was discharged from 2.5 V to 0 V, no change in the crystal structure of the cathode material was observed. Thus, the damage to the cathode material is not strongly attributed to swelling. We speculate that blended materials of layered and spinel structures might be used for cathode fabrication. Blended cathode materials have been extensively researched and applied to HEV, PHEV, and BEV. Figure 7b presents the XRD results of the anode discharged to 2.5 V. We examined the Li compound in the anode after cell discharge to 2.5 V was completed. Compounds of Li were not observed, whereas peaks of graphite (anode material) and Cu (current collector) were observed. This demonstrates that there was no Li in the anode even after discharging to 2.5 V. Accordingly, the XRD results in Figure 7 and SEM results in Figure 6 indicate that no critical problems occur in the crystal structure and microstructure changes of the electrode, even when the electrode is discharged to 0 V.
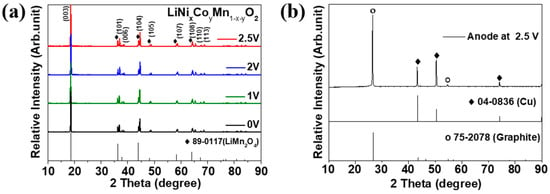
Figure 7.
X-ray diffraction analysis of the (a) cathode of LIB cell-2 discharged to various cut-off potentials and (b) anode of LIB cell-2 discharged to 2.5 V.
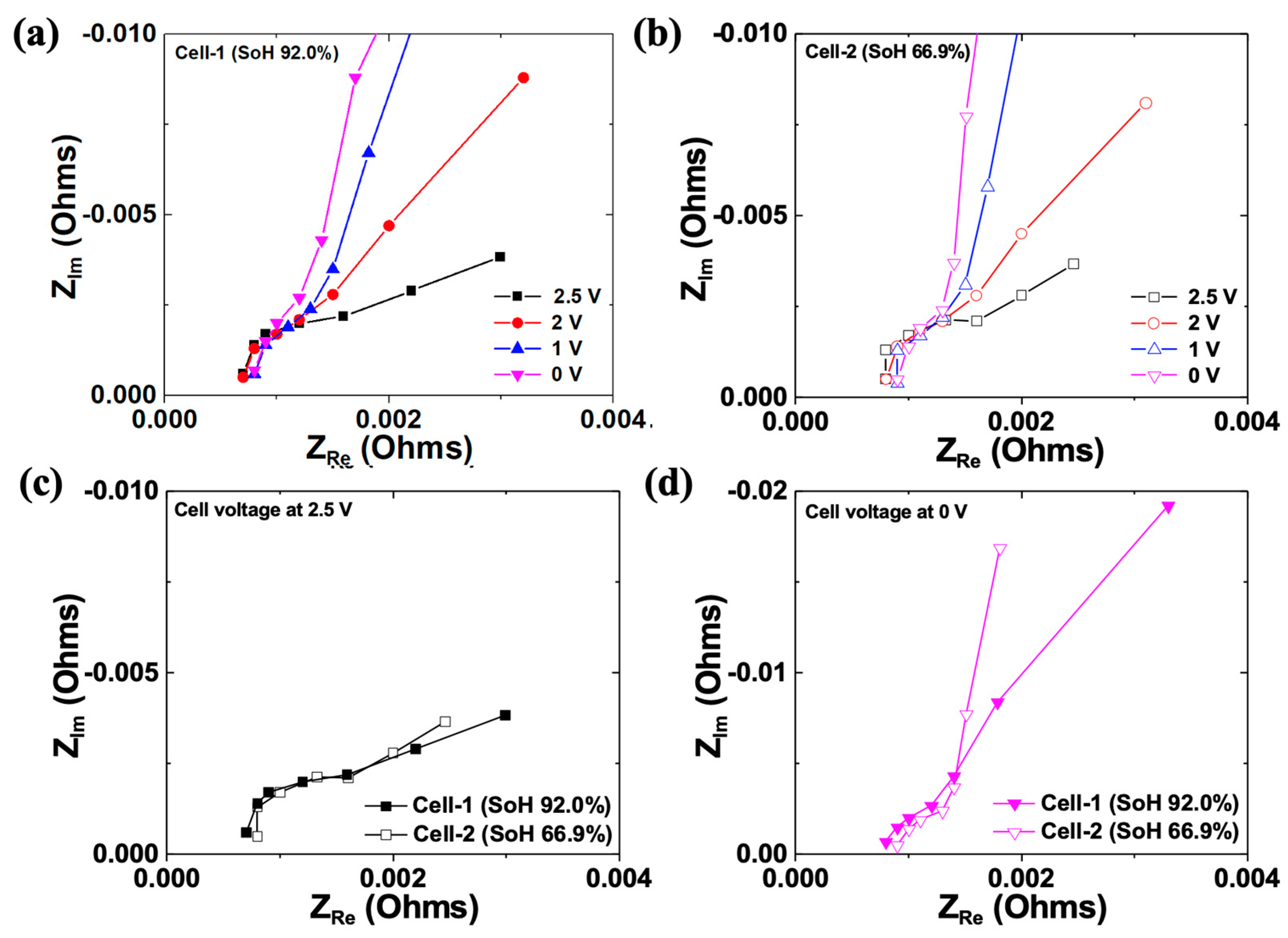
Figure 8 presents the EIS results of cell-1 and cell-2. Electrochemical impedance spectroscopy can execute a non-destructive analysis on the state of the electrode reaction, interfaces, and electrolytes. Through the semi-circle in the middle-frequency region, the resistance based on charge transfer (Rct) at the interface between the electrode and electrolyte can be identified. The tail in the low-frequency region after the semi-circle is called Warburg impedance, relating to diffusion in a medium. Warburg impedance is a type of impedance that arises from the diffusion of ions or molecules in a material. It is characterized by a linear decrease in impedance with the square root of frequency, resulting in a diagonal line on a Nyquist plot with a slope of −1/2. This behavior is related to the diffusion of a charged species in the material, which causes a frequency-dependent phase shift between the voltage and current signals. The Warburg impedance slope can be used to determine the diffusion coefficient and transport properties of the material, making it a valuable tool in electrochemical analyses [40,41,42,43]. Figure 8a shows the EIS results based on the various discharge potentials of cell-1 in a Nyquist plot. Significant changes of the semi-circle were not observed even though the discharge potentials varied. This signifies that almost no change occurred in the interfacial Rct between the electrolyte and electrode. Therefore, we can conclude that no change occurred in the electrode’s composition, crystal structure, or microstructure. However, the finite space Warburg (FSW) behavior appeared profoundly as the cells were discharged below 2 V. For Li-ion batteries, the FSW element appears owing to the highly limited diffusion of lithium ions toward the porous electrode. Thus, FSW behavior at a low discharge potential would be attributed to the limited diffusion behavior resulting from the volumetric expansion, and the discharge current or potential applied to the cell may promote the further decomposition of the electrolyte. Because of the low SoH and low discharge potential applied to cell-2, the FSW behavior observed was profound, especially at 0 V (Figure 8b). Figure 8c shows the EIS results of cell-1 and cell-2 at 2.5 V. Both cells exhibited semi-infinite diffusional behavior in the low-frequency region. The solution resistance (Rs) in cell-1 was smaller than that of cell-2, probably owing to less electrolyte decomposition. Figure 8d shows the EIS results of cell-1 and cell-2 at 0 V. The FSW behavior was predominant in cell-2. We speculate that the high discharge potential (i.e., 0 V) of the low SoH cell might decompose the electrolyte more effectively because of the highly prohibited diffusion of the redox species, resulting in intensive swelling. Thus, when recycling a low SoH battery, the appropriate discharge potential for the safety of the disassembly process must be identified for direct recycling.
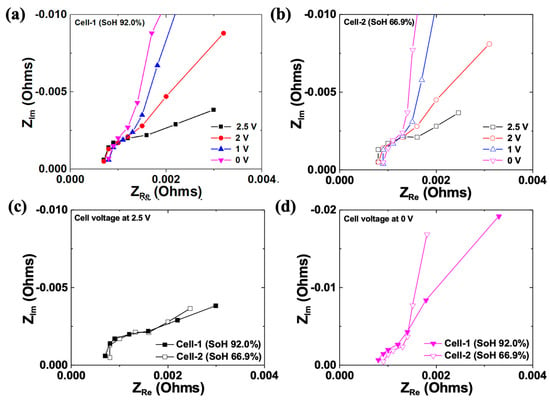
Figure 8.
(a,b) Nyquist plots measured from cell-1 and a cell-2, discharged to various potentials, respectively. (c,d) Comparison of the Nyquist plots of cell-1 and cell-2, discharged to 2.5 and 0V, respectively.
4. Conclusions
This study investigated electrical discharge as a pre-processing method for directly recycling spent LIBs. We confirmed that the pouch cells used in most EV LIB packs are unsuitable for salt-water discharge. We electrically discharged spent LIBs to various voltages under the 1/3C-rate condition and carefully examined the crystal structure through XRD. No changes occurred in the crystal structure of the cathode material, even when over-discharged to 0 V. We also confirmed that no considerable differences in the microstructure were observed via SEM. The EIS results indicated that the resistance of the electrolyte slightly increased, and significant swelling occurred at discharge potentials lower than 2 V, probably owing to the limited diffusion of ions. Even when the spent LIB was over-discharged to 0 V, especially with low SoH, the LIB pouch swelled drastically. After discharge to 0 V, the voltage recovered to approximately 2.5 V in 24 h. We confirmed that the over-discharging of spent LIBs in module and pack units is not recommended because of individual cell swelling. We suggest that an optimal discharge potential of 2.5 V be applied for the safe direct recycling of spent battery packs and modules to ensure the least swelling and minimum degree of recovery of discharged potential. This study provides important insights for the efficient and sustainable recycling of spent LIBs, which are expected to increase in quantity as the demand for EVs and other electronic devices continues to increase. We envision that the findings are adjudged applicable to environmentally friendly recycling processes that require a safe discharge process of spent LIBs. In addition, we contend that they can be applied to an optimized discharge method by applying the deep learning-based capacity estimation method reported by Shen et al. [44].
Author Contributions
Conceptualization, H.L.; methodology, H.L.; software, H.L.; validation, H.L.; formal analysis, H.L.; investigation, H.L.; resources, S.-W.L.; data curation, H.L.; writing—original draft preparation, H.L.; writing—review and editing, H.L., S.-W.L.; visualization, H.L.; supervision, S.-W.L.; project administration, Y.-T.K. and S.-W.L.; funding acquisition, Y.-T.K. and S.-W.L. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (NRF-2017R1C1B2004765 and NRF-2020R1C1C1005743). This work was also funded by the Ministry of Trade, Industry & Energy (MOTIE, Korea). (20019032, Development of manufacturing technology for lithium-based cathode materials with a purity of over 99% using a new eco-friendly method from secondary battery scrap black powder).
Data Availability Statement
Data is contained within the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Gaines, L.; Dai, Q.; Vaughey, J.T.; Gillard, S. Direct Recycling R&D at the ReCell Center. Recycling 2021, 6, 31. [Google Scholar]
- Hua, Y.; Liu, X.; Zhou, S.; Huang, Y.; Ling, H.; Yang, S. Toward Sustainable Reuse of Retired Lithium-ion Batteries from Electric Vehicles. Resour. Conserv. Recycl. 2020, 168, 105249. [Google Scholar] [CrossRef]
- Chen, H.; Shen, J. A degradation-based sorting method for lithium-ion battery reuse. PLoS ONE 2017, 12, e0185922. [Google Scholar] [CrossRef]
- Arshad, F.; Li, L.; Amin, K.; Fan, E.; Manurkar, N.; Ahmad, A.; Yang, J.; Wu, F.; Chen, R. A Comprehensive Review of the Advancement in Recycling the Anode and Electrolyte from Spent Lithium Ion Batteries. ACS Sustain. Chem. Eng. 2020, 8, 13527–13554. [Google Scholar] [CrossRef]
- Chitre, A.; Freake, D.; Lander, L.; Edge, J.; Titirici, M.-M. Towards a More Sustainable Lithium-Ion Battery Future: Recycling LIBs from Electric Vehicles. Batter. Supercaps 2020, 3, 1126–1136. [Google Scholar] [CrossRef]
- Fan, E.; Li, L.; Wang, Z.; Lin, J.; Huang, Y.; Yao, Y.; Chen, R.; Wu, F. Sustainable Recycling Technology for Li-Ion Batteries and Beyond: Challenges and Future Prospects. Chem. Rev. 2020, 120, 7020–7063. [Google Scholar] [CrossRef]
- Ren, G.-X.; Xiao, S.-W.; Xie, M.-Q.; Pan, B.; Chen, J.; Wang, F.-G.; Xia, X. Recovery of valuable metals from spent lithium ion batteries by smelting reduction process based on FeO–SiO2–Al2O3 slag system. Trans. Nonferrous Met. Soc. China 2017, 27, 450–456. [Google Scholar] [CrossRef]
- Xiao, S.; Ren, G.; Xie, M.; Pan, B.; Fan, Y.; Wang, F.; Xia, X. Recovery of Valuable Metals from Spent Lithium-Ion Batteries by Smelting Reduction Process Based on MnO–SiO2–Al2O3 Slag System. J. Sustain. Met. 2017, 3, 703–710. [Google Scholar] [CrossRef]
- Yadav, P.; Jie, C.J.; Tan, S.; Srinivasan, M. Recycling of cathode from spent lithium iron phosphate batteries. J. Hazard. Mater. 2020, 399, 123068. [Google Scholar] [CrossRef]
- Yao, Y.; Zhu, M.; Zhao, Z.; Tong, B.; Fan, Y.; Hua, Z. Hydrometallurgical Processes for Recycling Spent Lithium-Ion Batteries: A Critical Review. ACS Sustain. Chem. Eng. 2018, 6, 13611–13627. [Google Scholar] [CrossRef]
- Fu, Y.; He, Y.; Li, J.; Qu, L.; Yang, Y.; Guo, X.; Xie, W. Improved hydrometallurgical extraction of valuable metals from spent lithium-ion batteries via a closed-loop process. J. Alloys Compd. 2020, 847, 156489. [Google Scholar] [CrossRef]
- Esfahani, M.H.; Zinatloo-Ajabshir, S.; Naji, H.; Marjerrison, C.A.; Greedan, J.E.; Behzad, M. Structural characterization, phase analysis and electrochemical hydrogen storage studies on new pyrochlore SmRETi2O7 (RE = Dy, Ho, and Yb) microstructures. Ceram. Int. 2023, 49, 253–263. [Google Scholar] [CrossRef]
- Zinatloo-Ajabshir, S.; Morassaei, M.S.; Salavati-Niasari, M. Eco-friendly synthesis of Nd2Sn2O7–based nanostructure materials using grape juice as green fuel as photocatalyst for the degradation of erythrosine. Compos. Part B Eng. 2019, 167, 643–653. [Google Scholar] [CrossRef]
- Rezayeenik, M.; Mousavi-Kamazani, M.; Zinatloo-Ajabshir, S. CeVO4/rGO nanocomposite: Facile hydrothermal synthesis, characterization, and electrochemical hydrogen storage. Appl. Phys. A 2022, 129, 47. [Google Scholar] [CrossRef]
- Zinatloo-Ajabshir, S.; Salehi, Z.; Salavati-Niasari, M. Synthesis of dysprosium cerate nanostructures using Phoenix dactylifera extract as novel green fuel and investigation of their electrochemical hydrogen storage and Coulombic efficiency. J. Clean. Prod. 2019, 215, 480–487. [Google Scholar] [CrossRef]
- Wang, Y.; Leung, D.Y.C. A high-performance aluminum-feed microfluidic fuel cell stack. J. Power Sources 2016, 336, 427–436. [Google Scholar] [CrossRef]
- Sethurajan, M.; Gaydardzhiev, S. Bioprocessing of spent lithium ion batteries for critical metals recovery—A review. Resour. Conserv. Recycl. 2020, 165, 105225. [Google Scholar] [CrossRef]
- Piątek, J.; Afyon, S.; Budnyak, T.M.; Budnyk, S.; Sipponen, M.H.; Slabon, A. Sustainable Li-Ion Batteries: Chemistry and Recycling. Adv. Energy Mater. 2020, 11, 2003456. [Google Scholar] [CrossRef]
- Xu, P.; Dai, Q.; Gao, H.; Liu, H.; Zhang, M.; Li, M.; Chen, Y.; An, K.; Meng, Y.S.; Liu, P.; et al. Efficient Direct Recycling of Lithium-Ion Battery Cathodes by Targeted Healing. Joule 2020, 4, 2609–2626. [Google Scholar] [CrossRef]
- Sloop, S.; Crandon, L.; Allen, M.; Koetje, K.; Reed, L.; Gaines, L.; Sirisaksoontorn, W.; Lerner, M. A direct recycling case study from a lithium-ion battery recall. Sustain. Mater. Technol. 2020, 25, e00152. [Google Scholar] [CrossRef]
- Ojanen, S.; Lundström, M.; Santasalo-Aarnio, A.; Serna-Guerrero, R. Challenging the concept of electrochemical discharge using salt solutions for lithium-ion batteries recycling. Waste Manag. 2018, 76, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Torabian, M.M.; Jafari, M.; Bazargan, A. Discharge of lithium-ion batteries in salt solutions for safer storage, transport, and resource recovery. Waste Manag. Res. 2021, 40, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Juarez-Robles, D.; Vyas, A.A.; Fear, C.; Jeevarajan, J.A.; Mukherjee, P.P. Overdischarge and Aging Analytics of Li-Ion Cells. J. Electrochem. Soc. 2020, 167, 090558. [Google Scholar] [CrossRef]
- Gaines, L. The future of automotive lithium-ion battery recycling: Charting a sustainable course. Sustain. Mater. Technol. 2014, 1, 2–7. [Google Scholar] [CrossRef]
- Langner, T.; Sieber, T.; Acker, J. Studies on the deposition of copper in lithium-ion batteries during the deep discharge process. Sci. Rep. 2021, 11, 6316. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhang, F.-S.; He, K.; Zhang, Z.-Y.; Zhang, C.-C. Avoiding thermal runaway during spent lithium-ion battery recycling: A comprehensive assessment and a new approach for battery discharge. J. Clean. Prod. 2022, 380, 135045. [Google Scholar] [CrossRef]
- Arno Kwade, J.D. Recycling of Lithium-Ion Batteries; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Lim, A.E.; Lam, Y.C. Electroosmotic Flow Hysteresis for Fluids with Dissimilar pH and Ionic Species. Micromachines 2021, 12, 1031. [Google Scholar] [CrossRef]
- Lim, A.E.; Lim, C.Y.; Lam, Y.C. Electroosmotic flow hysteresis for dissimilar ionic solutions. Biomicrofluidics 2015, 9, 024113. [Google Scholar] [CrossRef]
- Li, J.; Wang, G.; Xu, Z. Generation and detection of metal ions and volatile organic compounds (VOCs) emissions from the pretreatment processes for recycling spent lithium-ion batteries. Waste Manag. 2016, 52, 221–227. [Google Scholar] [CrossRef]
- Goodship, V.; Stevels, A.; Huisman, J. Waste Electrical and Electronic Equipment (WEEE) Handbook; Woodhead Publishing: Sawston, UK, 2019. [Google Scholar]
- Rouhi, H.; Karola, E.; Serna-Guerrero, R.; Santasalo-Aarnio, A. Voltage behavior in lithium-ion batteries after electrochemical discharge and its implications on the safety of recycling processes. J. Energy Storage 2021, 35, 102323. [Google Scholar] [CrossRef]
- Zhang, T.; He, Y.; Ge, L.; Fu, R.; Zhang, X.; Huang, Y. Characteristics of wet and dry crushing methods in the recycling process of spent lithium-ion batteries. J. Power Sources 2013, 240, 766–771. [Google Scholar] [CrossRef]
- Shaw-Stewart, J.; Alvarez-Reguera, A.; Greszta, A.; Marco, J.; Masood, M.; Sommerville, R.; Kendrick, E. Aqueous solution discharge of cylindrical lithium-ion cells. Sustain. Mater. Technol. 2019, 22, e00110. [Google Scholar] [CrossRef]
- Chikkannanavar, S.B.; Bernardi, D.M.; Liu, L. A review of blended cathode materials for use in Li-ion batteries. J. Power Sources 2014, 248, 91–100. [Google Scholar] [CrossRef]
- Gao, J.; Manthiram, A. Eliminating the irreversible capacity loss of high capacity layered Li[Li0.2Mn0.54Ni0.13Co0.13]O2 cathode by blending with other lithium insertion hosts. J. Power Sources 2009, 191, 644–647. [Google Scholar] [CrossRef]
- Cabana, J.; Kang, S.-H.; Johnson, C.S.; Thackeray, M.M.; Grey, C.P. Structural and electrochemical characterization of composite layered-spinel electrodes containing Ni and Mn for Li-ion batteries. J. Electrochem. Soc. 2009, 156, A730. [Google Scholar] [CrossRef]
- Albertus, P.; Christensen, J.; Newman, J. Experiments on and modeling of positive electrodes with multiple active materials for lithium-ion batteries. J. Electrochem. Soc. 2009, 156, A606. [Google Scholar] [CrossRef]
- Jeong, S.K.; Shin, J.S.; Nahm, K.S.; Prem Kumar, T.; Stephan, A.M. Electrochemical studies on cathode blends of LiMn2O4 and Li[Li1/15Ni1/5Co2/5Mn1/3O2]. Mater. Chem. Phys. 2008, 111, 213–217. [Google Scholar] [CrossRef]
- Sekushin, N.A. Equivalent circuit of Warburg impedance. Russ. J. Electrochem. 2009, 45, 828–832. [Google Scholar] [CrossRef]
- Oldenburger, M.; Bedürftig, B.; Gruhle, A.; Grimsmann, F.; Richter, E.; Findeisen, R.; Hintennach, A. Investigation of the low frequency Warburg impedance of Li-ion cells by frequency domain measurements. J. Energy Storage 2018, 21, 272–280. [Google Scholar] [CrossRef]
- Karden, E.; Buller, S.; De Doncker, R.W. A method for measurement and interpretation of impedance spectra for industrial batteries. J. Power Sources 2000, 85, 72–78. [Google Scholar] [CrossRef]
- Oldenburger, M.; Bedürftig, B.; Gruhle, A.; Richter, E. A new approach to measure the non-linear Butler–Volmer behavior of electrochemical systems in the time domain. J. Energy Storage 2017, 14, 16–21. [Google Scholar] [CrossRef]
- Shen, S.; Sadoughi, M.; Li, M.; Wang, Z.; Hu, C. Deep convolutional neural networks with ensemble learning and transfer learning for capacity estimation of lithium-ion batteries. Appl. Energy 2020, 260, 114296. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).