Effect of Cathode Microstructure on Electrochemical Properties of Sodium Nickel-Iron Chloride Batteries
Abstract
:1. Introduction
2. Experimental Section
2.1. Preparation of Cathode Granules
2.2. Synthesis Method and Additives of Second Electrolyte
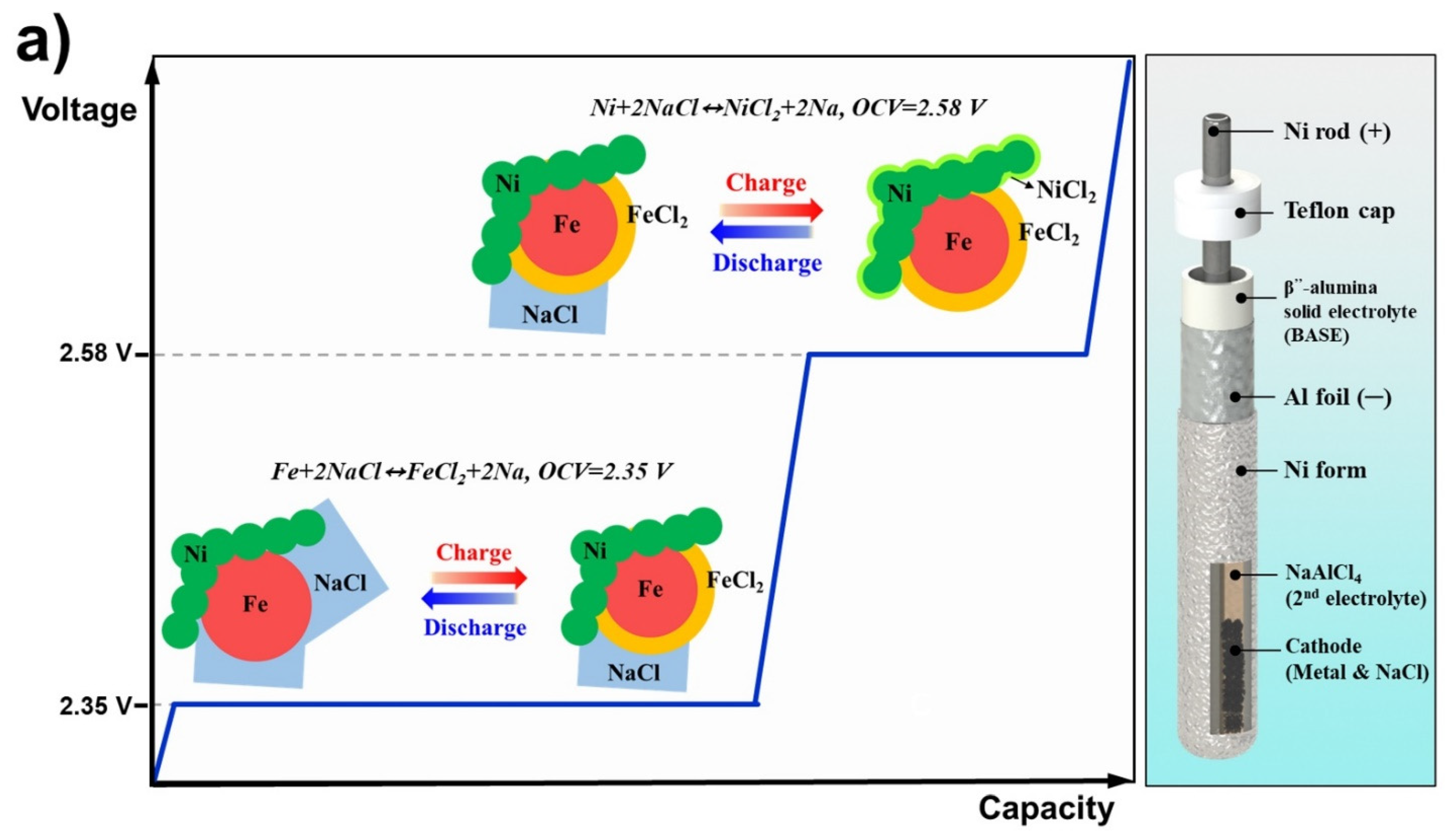
2.3. Configuration of a Simple Test Cell
2.4. Test Condition of Cells
3. Results and Discussion
3.1. Electrochemical Effects of Granule Density and NaCl Size in the Na/NiCl2 Cells
3.2. Application of Small NaCl on the Na/NiCl2 and Na/(Ni,Fe)Cl2 Cell of Metal/NaCl = 1.5/1
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Killer, M.; Farrokhseresht, M.; Paterakis, N.G. Implementation of large-scale Li-ion battery energy storage systems within the EMEA region. Appl. Energy 2020, 260, 114166. [Google Scholar] [CrossRef]
- Zhu, Q.-N.; Wang, Z.-Y.; Wang, J.-W.; Liu, X.-Y.; Yang, D.Y.; Cheng, L.-W.; Tang, M.-Y.; Qin, Y.; Wang, H. Challenges and strategies for ultrafast aqueous zinc-ion batteries. Rare Met. 2021, 40, 309–328. [Google Scholar] [CrossRef]
- Lei, K.-X.; Wang, J.; Chen, C.; Li, S.-Y.; Wang, S.-W.; Zheng, S.-J.; Li, F.-J. Recent progresses on alloy-based anodes for potassium-ion batteries. Rare Met. 2020, 39, 989–1004. [Google Scholar] [CrossRef]
- Skyllas-kazacos, M.; Grossmith, F. Efficient vanadium redox flow cell. J. Electrochem. Soc. 1987, 134, 2950–2953. [Google Scholar] [CrossRef]
- Stefano, S.; Ratnakumar, B.V.; Bankston, C.P. Advanced rechargeable sodium batteries with novel cathodes. J. Power Sources 1990, 29, 301–309. [Google Scholar] [CrossRef] [Green Version]
- Zhan, X.; Bonnett, J.F.; Engelhard, M.H.; Reed, D.M.; Sprenkle, V.L.; Li, G. A High-Performance Na–Al Battery Based on Reversible NaAlCl4 Catholyte. Adv. Energy Mater. 2020, 10, 2001378. [Google Scholar] [CrossRef]
- Zhan, X.; Bowden, M.E.; Lu, X.; Bonnett, J.F.; Lemmon, T.; Reed, D.M.; Sprenkle, V.L.; Li, G. A Low-Cost Durable Na-FeCl2 Battery with Ultrahigh Rate Capability. Adv. Energy Mater. 2020, 10, 1903472. [Google Scholar] [CrossRef]
- Li, G.; Lu, X.; Kim, J.Y.; Viswanathan, V.V.; Meinhardt, K.D.; Engelhard, M.H.; Sprenkle, V.L. An Advanced Na-FeCl2 ZEBRA Battery for Stationary Energy Storage Application. Adv. Energy Mater. 2015, 5, 1500357. [Google Scholar] [CrossRef]
- Keith, T.A.; Thackeray, M.M. The crystal Chemistry of the Na_FeCl2 Battery. J. Electrochem. Soc. 1988, 135, 2121–2123. [Google Scholar]
- Moseley, P.T.; Bones, R.J.; Teagle, D.A.; Bellamy, B.A.; Hawes, W.M. Stability of beta alumina electrolyte in sodium/FeCl2 (Zebra) cells. J. Electrochem. Soc. 1989, 136, 1361–1368. [Google Scholar] [CrossRef]
- Lu, X.; Li, G.; Kim, J.Y.; Lemmon, J.P.; Sprenkle, V.L.; Yang, Z. The effects of temperature on the electrochemical performance of sodium–nickel chloride batteries. J. Power Sources 2012, 215, 288–295. [Google Scholar] [CrossRef]
- Li, G.; Lu, X.; Kim, J.Y.; Lemmon, J.P.; Sprenkle, V.L. Cell degradation of a Na–NiCl2 (ZEBRA) battery. J. Mater. Chem. A 2013, 1, 14935–14942. [Google Scholar] [CrossRef]
- Kim, J.; Jo, S.H.; Bhavaraju, S.; Eccleston, A.; Kang, S.O. Low temperature performance of sodium–nickel chloride batteries with NaSICON solid electrolyte. J. Electroanal. Chem. 2015, 759, 201–206. [Google Scholar] [CrossRef]
- Li, G.; Lu, X.; Kim, J.Y.; Meinhardt, K.D.; Chang, H.J.; Canfield, N.L.; Sprenkle, V.L. Advanced intermediate temperature sodium-nickel chloride batteries with ultra-high energy density. Nat. Commun. 2016, 7, 10683. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, X.; Wang, J.; Gao, X.; Hu, Y.; Wen, Z. Ni-less cathode with 3D free-standing conductive network for planar Na-NiCl2 batteries. Chem. Eng. J. 2020, 387, 124059. [Google Scholar] [CrossRef]
- Gao, X.; Hu, Y.; Li, Y.; Wang, J.; Wu, X.; Yang, J.; Wen, Z. High-Rate and Long-Life Intermediate-Temperature Na-NiCl2 Battery with Dual-Functional Ni-Carbon Composite Nanofiber Network. ACS Appl. Mater. Interfaces 2020, 12, 24767–24776. [Google Scholar] [CrossRef]
- Ahn, C.-W.; Kim, M.; Hahn, B.-D.; Hong, I.; Kim, W.; Moon, G.; Lee, H.; Jung, K.; Park, Y.-C.; Choi, J.-H. Microstructure design of metal composite for active material in sodium nickel-iron chloride battery. J. Power Sources 2016, 329, 50–56. [Google Scholar] [CrossRef]
- Ahn, B.-M.; Ahn, C.-W.; Hahn, B.-D.; Choi, J.-J.; Kim, Y.-D.; Lim, S.-K.; Jung, K.; Park, Y.-C.; Choi, J.-H. Easy approach to realize low cost and high cell capacity in sodium nickel-iron chloride battery. Compos. Part B Eng. 2019, 168, 442–447. [Google Scholar] [CrossRef]
- Kim, J.; Jo, S.H.; Kim, J.-S.; Bhavaraju, S.; Kang, S.O. Investigation of manufacturing parameters for NaCl–Ni granule type cathodes used in low temperature NaSICON sodium-metal chloride batteries. J. Alloy. Compd. 2016, 665, 288–293. [Google Scholar] [CrossRef]
- Kim, S.-M.; Lee, S.-M.; Jung, K.; Park, Y.-C.; Cho, N.-U.; Choi, J.-H.; Kim, H.-S. Influence of Carbon Coating on Beta-Alumina Membrane for Sodium-Nickel Chloride Battery. Bull. Korean Chem. Soc. 2015, 36, 2869–2874. [Google Scholar] [CrossRef]
- Matsumae, Y.; Obata, K.; Ando, A.; Yanagi, M.; Kamei, Y.; Ueno, K.; Dokko, K.; Watanabe, M. Effects of Sulfur Loading, Cathode Porosity, and Electrolyte Amount on Li-S Battery Performance with Solvate Ionic Liquid Electrolyte. Electrochemistry 2019, 87, 254–259. [Google Scholar] [CrossRef] [Green Version]


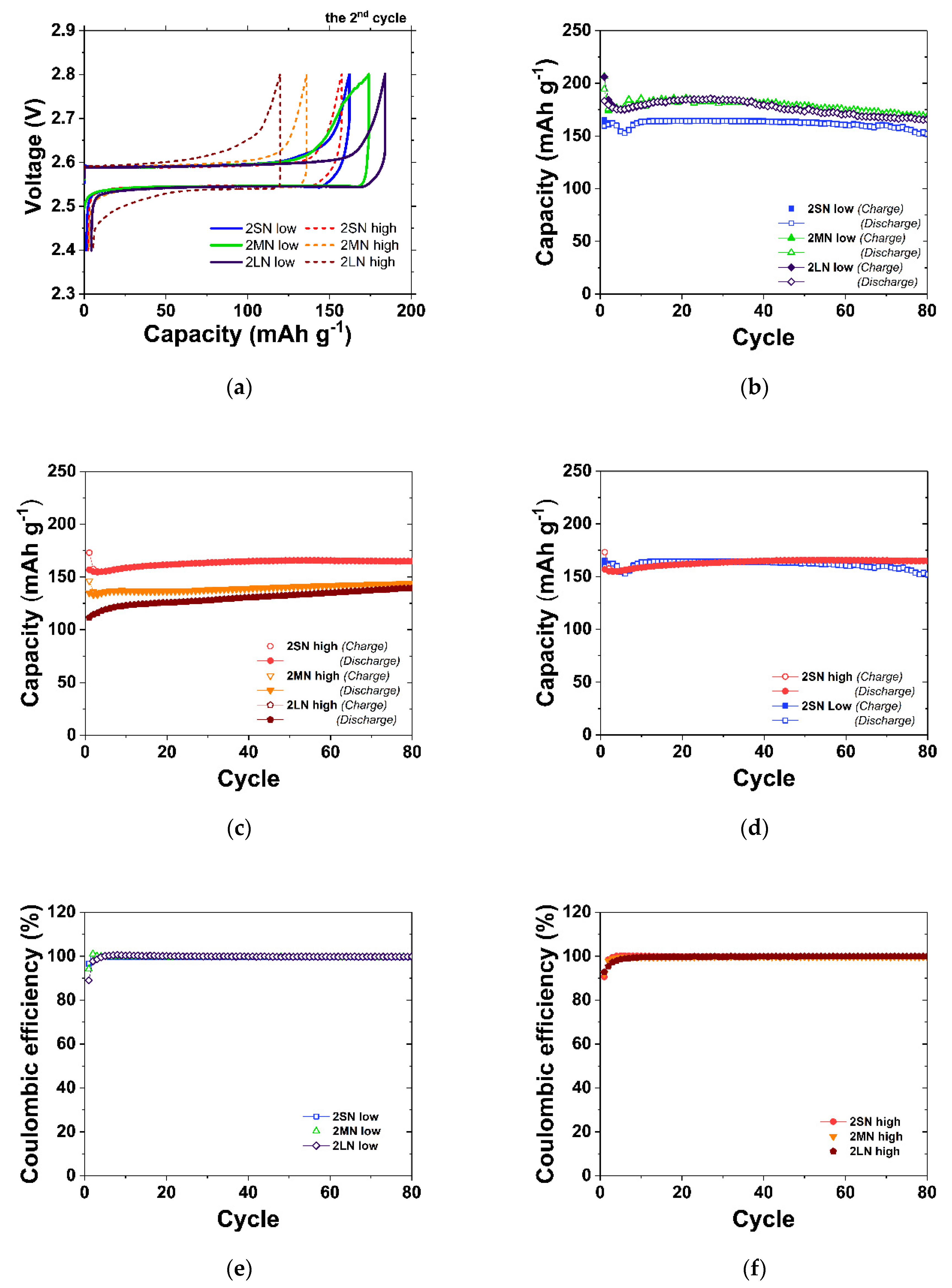


| Cell Type | Cell Name | NaCl Size (μm) | Granule Density (%) | Ratio of Composition | Charge Capacity (mAh g−1) | |||
|---|---|---|---|---|---|---|---|---|
| Ni (wt.%) | Fe (wt.%) | NaCl (wt.%) | Cycle Number | |||||
| 2nd | 80th | |||||||
| Na/NiCl2 | 2SN-low | <38 | 28 | 66.7 | - | 33.3 | 165 | 152 |
| 2SN-high | (Small) | 52 | 157 | 165 | ||||
| 2MN-low | 38–150 | 32 | 66.7 | 33.3 | 174 | 169 | ||
| 2MN-high | (Medium) | 52 | 136 | 143 | ||||
| 2LN-low | 150–300 | 37 | 66.7 | 33.3 | 184 | 166 | ||
| 2LN-high | (Large) | 55 | 120 | 140 | ||||
| Na/(Ni,Fe)Cl2 | 1.5LNF-low | 150–300 | - | 30.1 | 30.1 | 39.8 | 263 | 172 |
| 1.5SNF-high | <38 | 56 | 247 | 224 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahn, B.-M.; Ahn, C.-W.; Hahn, B.-D.; Choi, J.-J.; Kim, Y.-D.; Lim, S.-K.; Choi, J.-H. Effect of Cathode Microstructure on Electrochemical Properties of Sodium Nickel-Iron Chloride Batteries. Materials 2021, 14, 5605. https://doi.org/10.3390/ma14195605
Ahn B-M, Ahn C-W, Hahn B-D, Choi J-J, Kim Y-D, Lim S-K, Choi J-H. Effect of Cathode Microstructure on Electrochemical Properties of Sodium Nickel-Iron Chloride Batteries. Materials. 2021; 14(19):5605. https://doi.org/10.3390/ma14195605
Chicago/Turabian StyleAhn, Byeong-Min, Cheol-Woo Ahn, Byung-Dong Hahn, Jong-Jin Choi, Yang-Do Kim, Sung-Ki Lim, and Joon-Hwan Choi. 2021. "Effect of Cathode Microstructure on Electrochemical Properties of Sodium Nickel-Iron Chloride Batteries" Materials 14, no. 19: 5605. https://doi.org/10.3390/ma14195605






