Effect of Chemical Surface Texturing on the Superhydrophobic Behavior of Micro–Nano-Roughened AA6082 Surfaces
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Superhydrophobic Surfaces
- HNO3/HCl etching: The cleaned sample was dipped in a mixture of HNO3 and HCl in an ultra-pure water (1:3:3 ratio in vol.) solution for 60 min.
- HF/HCl etching: The aluminum specimens were dipped for 15 s in a HCl/HF acidic solution (73 vol.% HCl, 5 vol.% HF and 22 vol.% bi-distilled water).
- Boiling water treatment: Aluminum substrates were pretreated in boiling water for 5 min and dried at 70 °C for 60 min.
2.3. Sample Characterization
3. Results and Discussion
3.1. Surface Morphology
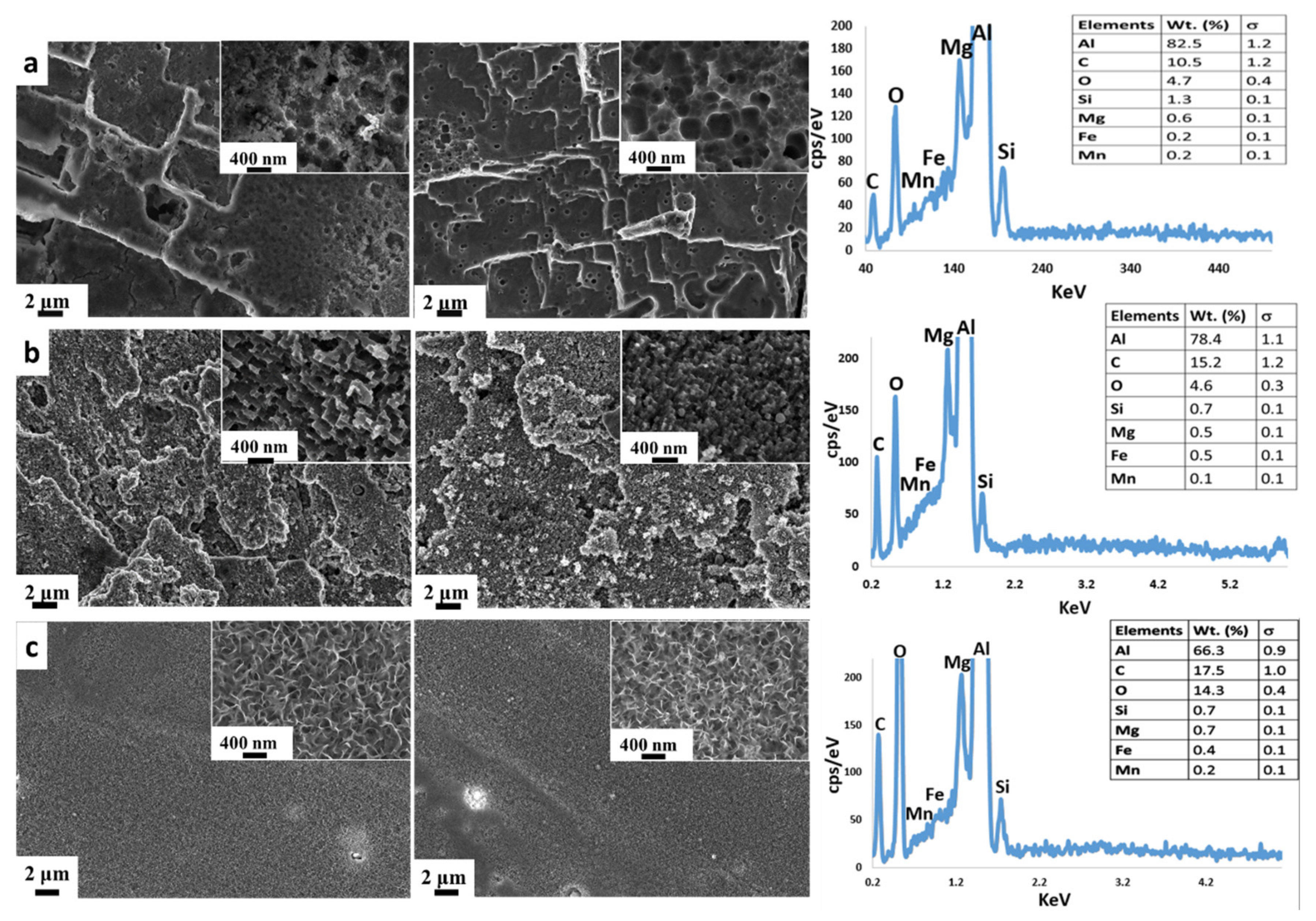
- HNO3/HCl etching: The surface was characterized by a bimodal structure. The coupled action of hydrochloric and nitric acids generated a micro-scale plate-like profile with numerous nano-scale pits (Figure 1a). This morphology is a result of the substrate interaction with this specific acidic solution. It is well known that aluminum alloys react strenuously with hydrochloric acid with a preferential attack of higher energy areas such as dislocations and grain boundaries, usually inducing large, rectangle-shaped pits [74]. However, due to the severe aggressiveness of the solution, the etched areas should be characterized by a non-homogeneous and uniform distribution and size (wider than 1 μm). In such a context, Oh et al. [75] suggested to not use only hydrochloric acid for aluminum etching but a mixture of acidic solution in order to reduce the dissolution rate on the local area and to obtain smaller pits. According to Singh et al. [76], oxidizing acids, such as nitric acid, have less effective corrosive actions on aluminum alloy. The combination of the hydrochloric and nitric acid action can be considered a successful approach to create a hierarchical structure as well as the micro-scale platelet structure that can be seen in the SEM image (Figure 1a).
- HF/HCl etching: The surface morphology of aluminum after the HF/HCl etching and the silane coating steps are shown in Figure 1b. A bimodal morphology can be clearly identified on the surface, i.e., a coral-like structure characterized by the presence of a pixelated substructure at the nanoscale level. In addition, in this case, the microstructure is a consequence of the intrinsic substrate heterogeneity. Indeed, the aluminum alloy has a large number of dislocations and line defects. These local defects are more reactive than other substrate areas in this acidic etchant [75]. Meanwhile, the impurities in the neighborhood of these defects could also magnify the chemical etching reaction. The dissolution phenomena in the aluminum induced by hydrofluoric acid starts after a relatively high immersion time (30–50 min) [77]. According to Straumanis et al. [77], the addition of a small quantity of hydrofluoric acid to other acids allows the electrochemical dissolution of the alloy to be promoted. In addition, HF acid reacts with Si-rich precipitates, favoring a selective dissolution in its neighboring area, thus influencing the large corrosion phenomena induced by a HCl acid solution. The selective dissolution favored by HF, coupled with the wide and general action of HCl, could be considered responsible for the coral-like structure obtained (Figure 1b).
- Boiling water treatment: An environmentally friendly and simple technique to obtain a nanostructured surface was applied. The absence of caustic reagents and special equipment in the whole preparation procedure make it industrially scalable and a cost-effective method. The SEM micrographs of the Al alloy surfaces treated with boiling water are shown in Figure 1c, before and after silanization. At the nano-scale level, the surfaces appeared to be covered by an oxi-hydroxide layer with 3D flower-like structures. This flower-like cluster consists of numerous petals with a thickness of ~30 nm. These contiguous petals overlap and connect to each other, resulting in a complex micro–nano structure. This microstructure is due to the formation of a protective film composed mainly of boehmite, according to the following reaction:
3.2. Profile Characterization
3.2.1. As-Received
3.2.2. HNO3/HCl Etching
3.2.3. HF/HCl Etching
3.2.4. Boiling Water Treatment
3.3. Roughness Statistical Analysis
3.4. Wettability
3.5. Self Cleaning Behavior
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Appendix A
References
- Wang, D.; Sun, Q.; Hokkanen, M.J.; Zhang, C.; Lin, F.Y.; Liu, Q.; Zhu, S.P.; Zhou, T.; Chang, Q.; He, B.; et al. Design of robust superhydrophobic surfaces. Nature 2020, 582, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Aamir, M.; Giasin, K.; Tolouei-Rad, M.; Vafadar, A. A review: Drilling performance and hole quality of aluminium alloys for aerospace applications. J. Mater. Res. Technol. 2020, 9, 12484–12500. [Google Scholar] [CrossRef]
- Varshney, D.; Kumar, K. Application and use of different aluminium alloys with respect to workability, strength and welding parameter optimization. Ain Shams Eng. J. 2021, 12, 1143–1152. [Google Scholar] [CrossRef]
- Wahid, M.A.; Siddiquee, A.N.; Khan, Z.A. Aluminum alloys in marine construction: Characteristics, application, and problems from a fabrication viewpoint. Mar. Syst. Ocean Technol. 2020, 15, 70–80. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, M.; Zhai, J.; Wang, J.; Jiang, L. Bioinspired construction of Mg-Li alloys surfaces with stable superhydrophobicity and improved corrosion resistance. Appl. Phys. Lett. 2008, 92, 183103. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, C.; Jia, D.; Wellmann, D.; Liu, W. Corrosion Behaviors of Selective Laser Melted Aluminum Alloys: A Review. Metals 2020, 10, 102. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Viademonte, M.P.; Abrahami, S.T.; Hack, T.; Burchardt, M.; Terryn, H. A Review on Anodizing of Aerospace Aluminum Alloys for Corrosion Protection. Coatings 2020, 10, 10. [Google Scholar] [CrossRef]
- Calabrese, L.; Proverbio, E.; Galtieri, G.; Borsellino, C. Effect of corrosion degradation on failure mechanisms of aluminium/steel clinched joints. Mater. Des. 2015, 87, 473–481. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, A.G.; Sun, B.R.; Chen, K.S.; Yu, H.Z. Functional and versatile superhydrophobic coatings via stoichiometric silanization. Nat. Commun. 2021, 12, 982. [Google Scholar] [CrossRef] [PubMed]
- Idumah, C.I.; Obele, C.M.; Emmanuel, E.O.; Hassan, A. Recently Emerging Nanotechnological Advancements in Polymer Nanocomposite Coatings for Anti-corrosion, Anti-fouling and Self-healing. Surf. Interfaces 2020, 21, 100734. [Google Scholar] [CrossRef]
- Chidambaram, D.; Clayton, C.R.; Halada, G.P. The role of hexafluorozirconate in the formation of chromate conversion coatings on aluminum alloys. Electrochim. Acta 2006, 51, 2862–2871. [Google Scholar] [CrossRef]
- Johansen, H.D.; Brett, C.M.A.; Motheo, A.J. Corrosion protection of aluminium alloy by cerium conversion and conducting polymer duplex coatings. Corros. Sci. 2012, 63, 342–350. [Google Scholar] [CrossRef]
- Cai, R.; Sun, M.; Chen, Z.; Munoz, R.; O’Neill, C.; Beving, D.E.; Yan, Y. Ionothermal synthesis of oriented zeolite AEL films and their application as corrosion-resistant coatings. Angew. Chem. Int. Ed. 2008, 47, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Chambers, B.D.; Taylor, S.R. The high throughput assessment of aluminium alloy corrosion using fluorometric methods. Part II—A combinatorial study of corrosion inhibitors and synergistic combinations. Corros. Sci. 2007, 49, 1597–1609. [Google Scholar] [CrossRef]
- Xie, Y.; Chen, H.; Shen, Y.; Tao, J.; Jin, M.; Wu, Y.; Hou, W. Rational Fabrication of Superhydrophobic Nanocone Surface for Dynamic Water Repellency and Anti-icing Potential. J. Bionic Eng. 2019, 16, 27–37. [Google Scholar] [CrossRef]
- Melchers, R.E. Predicting long-term corrosion of metal alloys in physical infrastructure. NPJ Mater. Degrad. 2019, 3, 4. [Google Scholar] [CrossRef]
- Darmanin, T.; Guittard, F. Superoleophobic surfaces with short fluorinated chains? Soft Matter 2013, 9, 5982–5990. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, L.; Qian, H.; Li, X. Superhydrophobic surfaces for corrosion protection: A review of recent progresses and future directions. J. Coat. Technol. Res. 2016, 13, 11–29. [Google Scholar] [CrossRef] [Green Version]
- Khaskhoussi, A.; Calabrese, L.; Proverbio, E. An Easy Approach for Obtaining Superhydrophobic Surfaces and their Applications. Key Eng. Mater. 2019, 813, 37–42. [Google Scholar] [CrossRef]
- Li, X.; Yu, Q.; Chen, X.; Zhang, Q. Microstructures and electrochemical behaviors of casting magnesium alloys with enhanced compression strengths and decomposition rates. J. Magnes. Alloy. 2021. [Google Scholar] [CrossRef]
- Xiu, Y.; Xiao, F.; Hess, D.W.; Wong, C.P. Superhydrophobic optically transparent silica films formed with a eutectic liquid. Thin Solid Films 2009, 517, 1610–1615. [Google Scholar] [CrossRef]
- Chen, Y.-K.; Chang, K.-C.; Wu, K.-Y.; Tsai, Y.-L.; Lu, J.-S.; Chen, H. Fabrication of superhydrophobic silica-based surfaces with high transmittance by using tetraethoxysilane precursor and different polymeric species. Appl. Surf. Sci. 2009, 255, 8634–8642. [Google Scholar] [CrossRef]
- Bhushan, B.; Her, E.K. Fabrication of superhydrophobic surfaces with high and low adhesion inspired from rose petal. Langmuir 2010, 26, 8207–8217. [Google Scholar] [CrossRef]
- Sun, T.; Feng, L.; Gao, A.X.; Jiang, L. Bioinspired Surfaces with Special Wettability. Acc. Chem. Res. 2005, 38, 644–652. [Google Scholar] [CrossRef]
- Teisala, H.; Tuominen, M.; Kuusipalo, J. Adhesion mechanism of water droplets on hierarchically rough superhydrophobic rose petal surface. J. Nanomater. 2011, 2011, 818707. [Google Scholar] [CrossRef]
- Radulović, Ž.; Porter, L.M.; Kim, T.K.; Mulenga, A. Comparative bioinformatics, temporal and spatial expression analyses of Ixodes scapularis organic anion transporting polypeptides. Ticks Tick-Borne Dis. 2014, 5, 287–298. [Google Scholar] [CrossRef] [Green Version]
- Watson, G.S.; Cribb, B.W.; Watson, J.A. Experimental determination of the efficiency of nanostructuring on non-wetting legs of the water strider. Acta Biomater. 2010, 6, 4060–4064. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, L.; Khaskhoussi, A.; Patane, S.; Proverbio, E. Assessment of Super-Hydrophobic Textured Coatings on AA6082 Aluminum Alloy. Coatings 2019, 9, 352. [Google Scholar] [CrossRef] [Green Version]
- Guo, Z.; Liu, W.; Su, B.L. Why so strong for the lotus leaf? Appl. Phys. Lett. 2008, 93, 201909. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Y.; Shi, L.; Li, J.; Guo, Z. Recent progress of double-structural and functional materials with special wettability. J. Mater. Chem. 2012, 22, 799–815. [Google Scholar] [CrossRef]
- She, Z.; Li, Q.; Wang, Z.; Tan, C.; Zhou, J.; Li, L. Highly anticorrosion, self-cleaning superhydrophobic Ni-Co surface fabricated on AZ91D magnesium alloy. Surf. Coat. Technol. 2014, 251, 7–14. [Google Scholar] [CrossRef]
- Isimjan, T.T.; Wang, T.; Rohani, S. A novel method to prepare superhydrophobic, UV resistance and anti-corrosion steel surface. Chem. Eng. J. 2012, 210, 182–187. [Google Scholar] [CrossRef]
- Liu, C.; Su, F.; Liang, J.; Huang, P. Facile fabrication of superhydrophobic cerium coating with micro-nano flower-like structure and excellent corrosion resistance. Surf. Coat. Technol. 2014, 258, 580–586. [Google Scholar] [CrossRef]
- Li, X.; Liang, J.; Shi, T.; Yang, D.; Chen, X.; Zhang, C.; Liu, Z.; Liu, D.; Zhang, Q. Tribological behaviors of vacuum hot-pressed ceramic composites with enhanced cyclic oxidation and corrosion resistance. Ceram. Int. 2020, 46, 12911–12920. [Google Scholar] [CrossRef]
- Li, W.; Kang, Z. Fabrication of corrosion resistant superhydrophobic surface with self-cleaning property on magnesium alloy and its mechanical stability. Surf. Coat. Technol. 2014, 253, 205–213. [Google Scholar] [CrossRef]
- Lai, Y.; Tang, Y.; Gong, J.; Gong, D.; Chi, L.; Lin, C.; Chen, Z. Transparent superhydrophobic/superhydrophilic TiO2-based coatings for self-cleaning and anti-fogging. J. Mater. Chem. 2012, 22, 7420–7426. [Google Scholar] [CrossRef]
- Xie, C.; Li, C.; Xie, Y.; Cao, Z.; Li, S.; Zhao, J.; Wang, M. ZnO/Acrylic Polyurethane Nanocomposite Superhydrophobic Coating on Aluminum Substrate Obtained via Spraying and Co-Curing for the Control of Marine Biofouling. Surf. Interfaces 2021, 22, 100833. [Google Scholar] [CrossRef]
- Li, J.; Shi, L.; Chen, Y.; Zhang, Y.; Guo, Z.; Su, B.L.; Liu, W. Stable superhydrophobic coatings from thiol-ligand nanocrystals and their application in oil/water separation. J. Mater. Chem. 2012, 22, 9774–9781. [Google Scholar] [CrossRef]
- Wang, C.; Yao, T.; Wu, J.; Ma, C.; Fan, Z.; Wang, Z.; Cheng, Y.; Lin, Q.; Yang, B. Facile approach in fabricating superhydrophobic and superoleophilic surface for water and oil mixture separation. ACS Appl. Mater. Interfaces 2009, 1, 2613–2617. [Google Scholar] [CrossRef]
- Shang, H.M.; Wang, Y.; Limmer, S.J.; Chou, T.P.; Takahashi, K.; Cao, G.Z. Optically transparent superhydrophobic silica-based films. Thin Solid Films 2005, 472, 37–43. [Google Scholar] [CrossRef]
- Ma, M.; Hill, R.M. Superhydrophobic surfaces. Curr. Opin. Colloid Interface Sci. 2006, 11, 193–202. [Google Scholar] [CrossRef]
- Ran, M.; Zheng, W.; Wang, H. Fabrication of superhydrophobic surfaces for corrosion protection: A review. Mater. Sci. Technol. 2019, 35, 313–326. [Google Scholar] [CrossRef]
- Manoharan, K.; Bhattacharya, S. Superhydrophobic surfaces review: Functional application, fabrication techniques and limitations. J. Micromanuf. 2019, 2, 59–78. [Google Scholar] [CrossRef]
- Wang, H.; Gao, D.; Meng, Y.; Wang, H.; Wang, E.; Zhu, Y. Corrosion-resistance, robust and wear-durable highly amphiphobic polymer based composite coating via a simple spraying approach. Prog. Org. Coat. 2015, 82, 74–80. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, X.; Gong, Y.; Zhang, B.; Li, H. Superhydrophobic nanocoatings prepared by a novel vacuum cold spray process. Surf. Coat. Technol. 2017, 325, 52–57. [Google Scholar] [CrossRef]
- Zhang, B.; Zhu, Q.; Li, Y.; Hou, B. Facile fluorine-free one step fabrication of superhydrophobic aluminum surface towards self-cleaning and marine anticorrosion. Chem. Eng. J. 2018, 352, 625–633. [Google Scholar] [CrossRef]
- Salehikahrizsangi, P.; Raeissi, K.; Karimzadeh, F.; Calabrese, L.; Proverbio, E. Highly hydrophobic Ni-W electrodeposited film with hierarchical structure. Surf. Coat. Technol. 2018, 344, 626–635. [Google Scholar] [CrossRef]
- Kim, H.-M.; Choi, J.-W.; Kwon, J.-S.; Lee, C.-H.; Kim, B. Super-Hydrophobic Properties of Aluminum Surfaces Synthesized by a Two-Step Chemical Etching Process. J. Nanosci. Nanotechnol. 2019, 19, 6452–6457. [Google Scholar] [CrossRef]
- Lian, Z.; Xu, J.; Wang, Z.; Yu, H. Biomimetic Superlyophobic Metallic Surfaces: Focusing on Their Fabrication and Applications. J. Bionic Eng. 2020, 17, 1–33. [Google Scholar] [CrossRef] [Green Version]
- Sun, R.; Zhao, J.; Li, Z.; Qin, N.; Mo, J.; Pan, Y.; Luo, D. Robust superhydrophobic aluminum alloy surfaces with anti-icing ability, thermostability, and mechanical durability. Prog. Org. Coat. 2020, 147, 105745. [Google Scholar] [CrossRef]
- Guo, F.; Duan, S.; Wu, D.; Matsuda, K.; Wang, T.; Zou, Y. Facile etching fabrication of superhydrophobic 7055 aluminum alloy surface towards chloride environment anticorrosion. Corros. Sci. 2021, 182, 109262. [Google Scholar] [CrossRef]
- Çakir, O. Chemical etching of aluminium. J. Mater. Process. Technol. 2008, 199, 337–340. [Google Scholar] [CrossRef]
- Jiang, B.; Li, M.; Liang, Y.; Bai, Y.; Song, D.; Li, Y.; Luo, J. Etching anisotropy mechanisms lead to morphology-controlled silicon nanoporous structures by metal assisted chemical etching. Nanoscale 2016, 8, 3085–3092. [Google Scholar] [CrossRef]
- Chen, Z.; Guo, Y.; Fang, S. A facial approach to fabricate superhydrophobic aluminum surface. Surf. Interface Anal. 2010, 42, 1–6. [Google Scholar] [CrossRef]
- Ruan, M.; Li, W.; Wang, B.; Luo, Q.; Ma, F.; Yu, Z. Optimal conditions for the preparation of superhydrophobic surfaces on al substrates using a simple etching approach. Appl. Surf. Sci. 2012, 258, 7031–7035. [Google Scholar] [CrossRef]
- Rezayi, T.; Entezari, M.H.; Moosavi, F. The variation of surface free energy of Al during superhydrophobicity processing. Chem. Eng. J. 2017, 322, 181–187. [Google Scholar] [CrossRef]
- Varshney, P.; Mohapatra, S.S.; Kumar, A. Superhydrophobic coatings for aluminium surfaces synthesized by chemical etching process. Int. J. Smart Nano Mater. 2016, 7, 248–264. [Google Scholar] [CrossRef] [Green Version]
- Xiao, X.; Xie, W.; Ye, Z. Preparation of corrosion-resisting superhydrophobic surface on aluminium substrate. Surf. Eng. 2019, 35, 411–417. [Google Scholar] [CrossRef]
- Salehikahrizsangi, P.; Raeissi, K.; Karimzadeh, F.; Calabrese, L.; Patane, S.; Proverbio, E. Erosion-corrosion behavior of highly hydrophobic hierarchical nickel coatings. Colloids Surfaces A Physicochem. Eng. Asp. 2018, 558, 446–454. [Google Scholar] [CrossRef]
- Chu, F.; Wu, X. Fabrication and condensation characteristics of metallic superhydrophobic surface with hierarchical micro-nano structures. Appl. Surf. Sci. 2016, 371, 322–328. [Google Scholar] [CrossRef]
- Kumar, A.; Gogoi, B. Development of durable self-cleaning superhydrophobic coatings for aluminium surfaces via chemical etching method. Tribol. Int. 2018, 122, 114–118. [Google Scholar] [CrossRef]
- Attar, M.R.; Khajavian, E.; Hosseinpour, S.; Davoodi, A. Fabrication of micro–nano-roughened surface with superhydrophobic character on an aluminium alloy surface by a facile chemical etching process. Bull. Mater. Sci. 2020, 43, 31. [Google Scholar] [CrossRef]
- Hooda, A.; Goyat, M.S.; Pandey, J.K.; Kumar, A.; Gupta, R. A review on fundamentals, constraints and fabrication techniques of superhydrophobic coatings. Prog. Org. Coat. 2020, 142, 105557. [Google Scholar] [CrossRef]
- Sun, R.; Zhao, J.; Li, Z.; Mo, J.; Pan, Y.; Luo, D. Preparation of mechanically durable superhydrophobic aluminum surface by sandblasting and chemical modification. Prog. Org. Coat. 2019, 133, 77–84. [Google Scholar] [CrossRef]
- Sun, D.; Böhringer, K.F. An active self-cleaning surface system for photovoltaic modules using anisotropic ratchet conveyors and mechanical vibration. Microsyst. Nanoeng. 2020, 6, 87. [Google Scholar] [CrossRef]
- Lian, Z.; Xu, J.; Yu, Z.; Yu, P.; Yu, H. A simple two-step approach for the fabrication of bio-inspired superhydrophobic and anisotropic wetting surfaces having corrosion resistance. J. Alloys Compd. 2019, 793, 326–335. [Google Scholar] [CrossRef]
- Samanta, A.; Wang, Q.; Shaw, S.K.; Ding, H. Roles of chemistry modification for laser textured metal alloys to achieve extreme surface wetting behaviors. Mater. Des. 2020, 192, 108744. [Google Scholar] [CrossRef]
- Cao, L.; Hu, H.A.; Gao, D. Design and fabrication of micro-textures for inducing a superhydrophobic behavior on hydrophilic materials. Langmuir 2007, 23, 4310–4314. [Google Scholar] [CrossRef]
- Liang, M.; Chen, X.; Xu, Y.; Zhu, L.; Jin, X.; Huang, C. Double-grooved nanofibre surfaces with enhanced anisotropic hydrophobicity. Nanoscale 2017, 9, 16214–16222. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Wang, Y.; Ding, J.; Wang, Q.; Chen, Q. Water condensation on superhydrophobic aluminum surfaces with different low-surface-energy coatings. Appl. Surf. Sci. 2012, 258, 4063–4068. [Google Scholar] [CrossRef]
- Lee, Y.; Ju, K.Y.; Lee, J.K. Stable biomimetic superhydrophobic surfaces fabricated by polymer replication method from hierarchically structured surfaces of al templates. Langmuir 2010, 26, 14103–14110. [Google Scholar] [CrossRef]
- Feng, L.; Che, Y.; Liu, Y.; Qiang, X.; Wang, Y. Fabrication of superhydrophobic aluminium alloy surface with excellent corrosion resistance by a facile and environment-friendly method. Appl. Surf. Sci. 2013, 283, 367–374. [Google Scholar] [CrossRef]
- Varshney, P.; Mohapatra, S.S.; Kumar, A. Fabrication of Mechanically Stable Superhydrophobic Aluminium Surface with Excellent Self-Cleaning and Anti-Fogging Properties. Biomimetics 2017, 2, 2. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Wu, J.; Yu, X.; Wu, H. Low-cost one-step fabrication of superhydrophobic surface on Al alloy. Appl. Surf. Sci. 2011, 257, 7928–7931. [Google Scholar] [CrossRef]
- Oh, H.J.; Lee, J.H.; Ahn, H.J.; Jeong, Y.; Park, N.J.; Kim, S.S.; Chi, C.S. Etching characteristics of high-purity aluminum in hydrochloric acid solutions. Mater. Sci. Eng. A 2007, 448–451, 348–351. [Google Scholar] [CrossRef]
- Singh, D.D.N.; Chaudhary, R.S.; Agarwal, C.V. Corrosion Characteristics of Some Aluminum Alloys in Nitric Acid. J. Electrochem. Soc. 1982, 129, 1869–1874. [Google Scholar] [CrossRef]
- Straumanis, M.E.; Wang, Y.N. The Rate and Mechanism of Dissolution of Purest Aluminum in Hydrofluoric Acid. J. Electrochem. Soc. 1955, 102, 370–381. [Google Scholar] [CrossRef]
- Feng, L.; Yan, Z.; Qiang, X.; Liu, Y.; Wang, Y. Facile formation of superhydrophobic aluminum alloy surface and corrosion-resistant behavior. Appl. Phys. A Mater. Sci. Process. 2016, 122, 165. [Google Scholar] [CrossRef]
- Jafari, R.; Farzaneh, M. Fabrication of superhydrophobic nanostructured surface on aluminum alloy. Appl. Phys. A Mater. Sci. Process. 2011, 102, 195–199. [Google Scholar] [CrossRef] [Green Version]
- Zumelzu, E.; Cabezas, C. Observations on the influence of microstructure on electrolytic tinplate corrosion. Mater. Charact. 1995, 34, 143–148. [Google Scholar] [CrossRef]
- Calabrese, L.; Khaskhoussi, A.; Proverbio, E. Superhydrophobic behavior of modified AA6082 alloy surfaces. Metall. Ital. 2019, 11/12, 6–10. [Google Scholar]
- Khaskhoussi, A.; Calabrese, L.; Proverbio, E. Superhydrophobic Self-Assembled Silane Monolayers on Hierarchical 6082 Aluminum Alloy for Anti-Corrosion Applications. Appl. Sci. 2020, 10, 2656. [Google Scholar] [CrossRef] [Green Version]
- Rahimi, M.; Fojan, P.; Gurevich, L.; Afshari, A. Effects of aluminium surface morphology and chemical modification on wettability. Appl. Surf. Sci. 2014, 296, 124–132. [Google Scholar] [CrossRef]
- Kamusewitz, H.; Possart, W. Wetting and scanning force microscopy on rough polymer surfaces: Wenzel’s roughness factor and the thermodynamic contact angle. Appl. Phys. A Mater. Sci. Process. 2003, 76, 899–902. [Google Scholar]
- Calabrese, L.; Bonaccorsi, L.; Caprì, A.; Proverbio, E. Effect of silane matrix composition on performances of zeolite composite coatings. Prog. Org. Coat. 2016, 101, 100–110. [Google Scholar] [CrossRef]
- Calabrese, L.; Bonaccorsi, L.; Capri, A.; Proverbio, E. Effect of silane matrix on corrosion protection of zeolite based composite coatings. Metall. Ital. 2014, 106, 35–39. [Google Scholar]
- de Gennes, P.-G.; Brochard-Wyart, F.; Quéré, D.; de Gennes, P.-G.; Brochard-Wyart, F.; Quéré, D. Dewetting. In Capillarity and Wetting Phenomena; Springer: New York, NY, USA, 2004; pp. 153–190. [Google Scholar]
- Kulinich, S.A.; Farzaneh, M. Alkylsilane self-assembled monolayers: Modeling their wetting characteristics. Appl. Surf. Sci. 2004, 230, 232–240. [Google Scholar] [CrossRef]
- Lafume, A.; Quéré, D. Superhydrophobic states. Nat. Mater. 2003, 2, 457–460. [Google Scholar] [CrossRef]
- Quére, D.; Lafuma, A.; Bico, J. Slippy and sticky microtextured solids. Nanotechnology 2003, 14, 1109–1112. [Google Scholar]
- Liu, M.; Zheng, Y.; Zhai, J.; Jiang, L. Bioinspired Super-antiwetting Interfaces with Special Liquid-Solid Adhesion. Acc. Chem. Res. 2010, 43, 368–377. [Google Scholar] [CrossRef]
- Horcas, I.; Fernández, R.; Gómez-Rodríguez, J.M.; Colchero, J.; Gómez-Herrero, J.; Baro, A.M. WSXM: A software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 2007, 78, 013705. [Google Scholar] [CrossRef] [PubMed]
- Soliman, H.H.; Gadelmawla, E.S.; Koura, M.M.; Maksoud, T.M.A.; Elewa, I.M. Roughness parameters. J. Mater. Process. Technol. 2002, 123, 133–145. [Google Scholar]







| Samples | Step 1 | Step 2 |
|---|---|---|
| Al-R | – | – |
| Al-RS | – | S18 |
| Al-N | HNO3/HCl etching | – |
| Al-NS | HNO3/HCl etching | S18 |
| Al-F | HF/HCl etching | – |
| Al-FS | HF/HCl etching | S18 |
| Al-W | Boiling water | – |
| Al-WS | Boiling water | S18 |
| Sample | Arith. Roughness Ra (Sa) (nm) | Rms Roughness (Sq) (nm) | Skewness, S | Kurtosis, K |
|---|---|---|---|---|
| Al-R | 10.7 | 16.3 | 0.13 | 3.27 |
| Al-N | 33.1 | 43.1 | 0.08 | 0.80 |
| Al-NS | 14.9 | 21.7 | 0.03 | 2.19 |
| Al-W | 27.5 | 36.4 | 0.17 | 1.41 |
| Al-WS | 30.8 | 37.4 | −0.13 | 0.04 |
| Al-F | 181.0 | 229. | 1.34 | 2.01 |
| Al-FS | 138.0 | 169.8 | −0.51 | 0.18 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khaskhoussi, A.; Calabrese, L.; Patané, S.; Proverbio, E. Effect of Chemical Surface Texturing on the Superhydrophobic Behavior of Micro–Nano-Roughened AA6082 Surfaces. Materials 2021, 14, 7161. https://doi.org/10.3390/ma14237161
Khaskhoussi A, Calabrese L, Patané S, Proverbio E. Effect of Chemical Surface Texturing on the Superhydrophobic Behavior of Micro–Nano-Roughened AA6082 Surfaces. Materials. 2021; 14(23):7161. https://doi.org/10.3390/ma14237161
Chicago/Turabian StyleKhaskhoussi, Amani, Luigi Calabrese, Salvatore Patané, and Edoardo Proverbio. 2021. "Effect of Chemical Surface Texturing on the Superhydrophobic Behavior of Micro–Nano-Roughened AA6082 Surfaces" Materials 14, no. 23: 7161. https://doi.org/10.3390/ma14237161









