Management of Solid Waste Containing Fluoride—A Review
Abstract
:1. Introduction
2. Anthropogenic Sources of Contamination with Fluorine Compounds
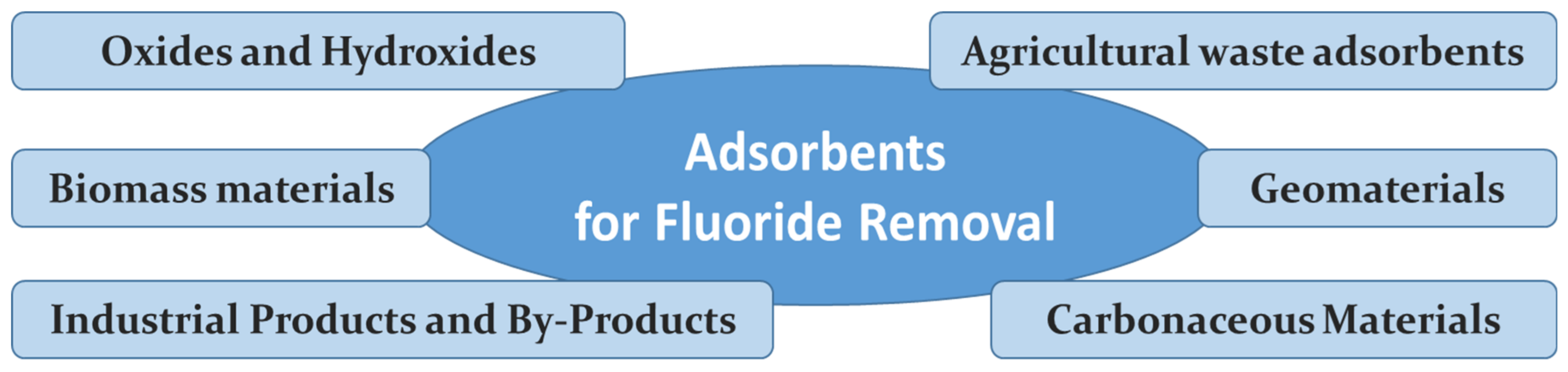
3. Selected Types of Reagents for Fluoride Removal
4. Industrial Waste, By-Product, and Biomass as Fluoride Adsorbents
5. Fluoride Wastes Removal in Industrial Processes
5.1. Industrial Production of Aluminium Fluoride
5.2. Industrial Waste from Semiconductor Factories
6. A New Concept(s) for the Production and Management of Fluoride Adsorbents
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gupta, N.; Yadav, K.K.; Kumar, V. A review on current status of municipal solid waste management in India. J. Environ. Sci. 2015, 37, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Melaré, A.V.D.S.; González, S.M.; Faceli, K.; Casadei, V. Technologies and decision support systems to aid solid-waste management: A systematic review. Waste Manag. 2017, 59, 567–584. [Google Scholar] [CrossRef] [PubMed]
- Bing, X.; Bloemhof, J.M.; Ramos, T.R.P.; Barbosa-Povoa, A.; Wong, C.Y.; van der Vorst, J.G. Research challenges in municipal solid waste logistics management. Waste Manag. 2016, 48, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Hettiarachchi, H.; Meegoda, J.N.; Ryu, S. Organic Waste Buyback as a Viable Method to Enhance Sustainable Municipal Solid Waste Management in Developing Countries. Int. J. Environ. Res. Public Health 2018, 15, 2483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ouda, O.K.M.; Raza, S.A.; Nizami, A.S.; Rehan, M.; Al-Waked, R.; Korres, N.E. Waste to energy potential: A case study of Saudi Arabia. Renew. Sustain. Energy Rev. 2016, 61, 328–340. [Google Scholar] [CrossRef]
- Sadef, Y.; Nizami, A.-S.; Batool, S.A.; Chaudary, M.N.; Ouda, O.K.M.; Asam, Z.-U.; Habib, K.; Rehan, M.; Demirbas, A. Waste-to-energy and recycling value for developing integrated solid waste management plan in Lahore. Energy Sources Part B Econ. Plan. Policy 2016, 11, 569–579. [Google Scholar] [CrossRef]
- Sawadogo, M.; Tanoh, S.T.; Sidibé, S.; Kpai, N.; Tankoano, I. Cleaner production in Burkina Faso: Case study of fuel briquettes made from cashew industry waste. J. Clean. Prod. 2018, 195, 1047–1056. [Google Scholar] [CrossRef]
- Ghisolfi, V.; Chaves, G.D.L.D.; Siman, R.R.; Xavier, L.H. System dynamics applied to closed loop supply chains of desktops and laptops in Brazil: A perspective for social inclusion of waste pickers. Waste Manag. 2017, 60, 14–31. [Google Scholar] [CrossRef]
- Matter, A.; Ahsan, M.; Marbach, M.; Zurbrügg, C. Impacts of policy and market incentives for solid waste recycling in Dhaka, Bangladesh. Waste Manag. 2015, 39, 321–328. [Google Scholar] [CrossRef]
- Ferronato, N.; Torretta, V. Waste Mismanagement in Developing Countries: A Review of Global Issues. Int. J. Environ. Res. Public Health 2019, 16, 1060. [Google Scholar] [CrossRef] [Green Version]
- Rudelis, V.; Dambrauskas, T.; Grineviciene, A.; Baltakys, K. The Prospective Approach for the Reduction of Fluoride Ions Mobility in Industrial Waste by Creating Products of Commercial Value. Sustainability 2019, 11, 634. [Google Scholar] [CrossRef] [Green Version]
- Angin, D. Utilization of activated carbon produced from fruit juice industry solid waste for the adsorption of Yellow 18 from aqueous solutions. Bioresour. Technol. 2014, 168, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Bujak, J.W. Thermal utilization (treatment) of plastic waste. Energy 2015, 90, 1468–1477. [Google Scholar] [CrossRef]
- Moh, Y. Solid waste management transformation and future challenges of source separation and recycling practice in Malaysia. Resour. Conserv. Recycl. 2017, 116, 1–14. [Google Scholar] [CrossRef]
- Shen, C.-W.; Tran, P.P.; Ly, P.T.M. Chemical Waste Management in the U.S. Semiconductor Industry. Sustainability 2018, 10, 1545. [Google Scholar] [CrossRef] [Green Version]
- Stonys, R.; Kuznetsov, D.; Krasnikovs, A.; Škamat, J.; Baltakys, K.; Antonovič, V.; Černašėjus, O. Reuse of ultrafine mineral wool production waste in the manufacture of refractory concrete. J. Environ. Manag. 2016, 176, 149–156. [Google Scholar] [CrossRef]
- Yilmaz, O.; Kara, B.Y.; Yetis, U. Hazardous waste management system design under population and environmental impact considerations. J. Environ. Manag. 2017, 203, 720–731. [Google Scholar] [CrossRef] [Green Version]
- Woźniak, J.; Pactwa, K. Overview of Polish Mining Wastes with Circular Economy Model and Its Comparison with Other Wastes. Sustainability 2018, 10, 3994. [Google Scholar] [CrossRef] [Green Version]
- Siddique, R. Utilization of Industrial By-products in Concrete. Procedia Eng. 2014, 95, 335–347. [Google Scholar] [CrossRef] [Green Version]
- Dyachenko, A.; Petlin, I.; Malyutin, L. The Research of Sulfuric Acidic Recycling of Aluminum Industry Fluorine-containing Waste Products. Procedia Chem. 2014, 11, 10–14. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Zhang, H.; Zhang, Z.; Shao, L.; He, P. Treatment and resource recovery from inorganic fluoride-containing waste produced by the pesticide industry. J. Environ. Sci. 2015, 31, 21–29. [Google Scholar] [CrossRef] [PubMed]
- de Beer, M.; Doucet, F.; Maree, J.; Liebenberg, L. Synthesis of high-purity precipitated calcium carbonate during the process of recovery of elemental sulphur from gypsum waste. Waste Manag. 2015, 46, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Baek, C.; Seo, J.; Choi, M.; Cho, J.; Ahn, J.; Cho, K. Utilization of CFBC Fly Ash as a Binder to Produce In-Furnace Desulfurization Sorbent. Sustainability 2018, 10, 4854. [Google Scholar] [CrossRef] [Green Version]
- Vaičiukynienė, D.; Vaitkevičius, V.; Kantautas, A.; Sasnauskas, V. Utilization of by-product waste silica in concrete-based materials. Mater. Res. 2012, 15, 561–567. [Google Scholar] [CrossRef]
- Iljina, A.; Baltakys, K.; Baltakys, M.; Siauciunas, R. Neutralization and removal of compounds containing fluoride ions from waste silica gel. Rom. J. Mater. 2014, 44, 265–271. [Google Scholar]
- Iljina, A.; Baltakys, K.; Bankauskaite, A.; Eisinas, A.; Kitrys, S. The stability of formed CaF2 and its influence on the thermal behavior of C–S–H in CaO–silica gel waste-H2O system. J. Therm. Anal. 2016, 127, 221–228. [Google Scholar] [CrossRef]
- Vaičiukynienė, D.; Kantautas, A.; Vaitkevičius, V.; Jakevičius, L.; Rudžionis, Ž.; Paškevičius, M. Effects of ultrasonic treatment on zeolite NaA synthesized from by-product silica. Ultrason. Sonochem. 2015, 27, 515–521. [Google Scholar] [CrossRef]
- Baltakys, K.; Iljina, A.; Bankauskaite, A. Thermal properties and application of silica gel waste contaminated with F− ions for C-S-H synthesis. J. Therm. Anal. 2015, 121, 145–154. [Google Scholar] [CrossRef]
- Hydrothermal Synthesis of Calcium Sulfoaluminate–Belite Cement from Industrial Waste Materials—Advances in Engineering. Available online: https://advanceseng.com/hydrothermal-synthesis-calcium-sulfoaluminate-belite-cement-industrial-waste-materials/ (accessed on 9 January 2022).
- Shen, Z.; Jin, F.; O’Connor, D.; Hou, D. Solidification/Stabilization for Soil Remediation: An Old Technology with New Vitality. Environ. Sci. Technol. 2019, 53, 11615–11617. [Google Scholar] [CrossRef] [Green Version]
- Ma, W.; Chen, D.; Pan, M.; Gu, T.; Zhong, L.; Chen, G.; Yan, B.; Cheng, Z. Performance of chemical chelating agent stabilization and cement solidification on heavy metals in MSWI fly ash: A comparative study. J. Environ. Manag. 2019, 247, 169–177. [Google Scholar] [CrossRef]
- Chen, W.; Wang, F.; Li, Z.; Li, Q. A comprehensive evaluation of the treatment of lead in MSWI fly ash by the combined cement solidification and phosphate stabilization process. Waste Manag. 2020, 114, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.-S.; Du, Y.-J.; Zhou, A.; Zhang, M.; Li, J.-S.; Zhou, S.-J.; Xia, W.-Y. Geoenvironmental properties of industrially contaminated site soil solidified/stabilized with a sustainable by-product-based binder. Sci. Total Environ. 2020, 765, 142778. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.-L.; Zhao, L.-Y.; Yuan, Z.-J.; Li, D.-Q.; Morrison, L. Assessment of the long-term leaching characteristics of cement-slag stabilized/solidified contaminated sediment. Chemosphere 2020, 267, 128926. [Google Scholar] [CrossRef]
- Solidification/Stabilization-ITRC. Available online: https://itrcweb.org/itrcwebsite/teams/projects/solidificationstabilization (accessed on 2 March 2022).
- Kizinievic, O.; Kizinievic, V.; Trambitski, Y.; Voisniene, V. Application of paper sludge and clay in manufacture of composite materials: Properties and biological susceptibility. J. Build. Eng. 2022, 48, 104003. [Google Scholar] [CrossRef]
- Jayarathne, T.; Stockwell, C.E.; Yokelson, R.J.; Nakao, S.; Stone, E.A. Emissions of Fine Particle Fluoride from Biomass Burning. Environ. Sci. Technol. 2014, 48, 12636–12644. [Google Scholar] [CrossRef] [PubMed]
- Nayak, B.; Samant, A.; Patel, R.; Misra, P.K. Comprehensive Understanding of the Kinetics and Mechanism of Fluoride Removal over a Potent Nanocrystalline Hydroxyapatite Surface. ACS Omega 2017, 2, 8118–8128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bodzek, M.; Konieczny, K. Open Access (CC BY-NC 4K.0) Fluorki w środowisku wodnym-zagrożenia i metody usuwania Fluorine in the Water Environment—Hazards and Removal Methods. Eng. Prot. Environ. 2018, 21, 113–141. [Google Scholar] [CrossRef]
- Tang, W.; Kovalsky, P.; He, D.; Waite, T.D. Fluoride and nitrate removal from brackish groundwaters by batch-mode capacitive deionization. Water Res. 2015, 84, 342–349. [Google Scholar] [CrossRef]
- Wang, L.; Sun, N.; Wang, Z.; Han, H.; Yang, Y.; Liu, R.; Hu, Y.; Tang, H.; Sun, W. Self-assembly of mixed dodecylamine–dodecanol molecules at the air/water interface based on large-scale molecular dynamics. J. Mol. Liq. 2019, 276, 867–874. [Google Scholar] [CrossRef]
- Claveau-Mallet, D.; Wallace, S.; Comeau, Y. Removal of phosphorus, fluoride and metals from a gypsum mining leachate using steel slag filters. Water Res. 2013, 47, 1512–1520. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, H.; Jiang, X.; Lv, G.; Yan, J. Study on the evolution and transformation of Cl during Co-incineration of a mixture of rectification residue and raw meal of a cement kiln. Waste Manag. 2019, 84, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Guth, S.; Hüser, S.; Roth, A.; Degen, G.; Diel, P.; Edlund, K.; Eisenbrand, G.; Engel, K.-H.; Epe, B.; Grune, T.; et al. Toxicity of fluoride: Critical evaluation of evidence for human developmental neurotoxicity in epidemiological studies, animal experiments and in vitro analyses. Arch. Toxicol. 2020, 94, 1375–1415. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for Drinking-Water Quality 3rd Edition Incorporating the First and Second Addenda; Recommendations Geneva 2008 WHO Library Cataloguing-in-Publication Data; World Health Organization: Geneva, Switzerland, 2008; Volume 1. [Google Scholar]
- Karthikeyan, G.; Shunmugasundarraj, A. Isopleth mapping and in-situ fluoride dependence on water quality in the krishnagiri block of tamil nadu in south india. Res. Rep. 2000, 33, 121–127. [Google Scholar]
- Rao, N.S. Groundwater quality: Focus on fluoride concentration in rural parts of Guntur district, Andhra Pradesh, India. Hydrol. Sci. J. 2003, 48, 835–847. [Google Scholar] [CrossRef]
- Viswanathan, G.; Jaswanth, A.; Gopalakrishnan, S.; Ilango, S.S.; Aditya, G. Determining the optimal fluoride concentration in drinking water for fluoride endemic regions in South India. Sci. Total Environ. 2009, 407, 5298–5307. [Google Scholar] [CrossRef]
- Abdelgawad, A.; Watanabe, K.; Takeuchi, S.; Mizuno, T. The origin of fluoride-rich groundwater in Mizunami area, Japan—Mineralogy and geochemistry implications. Eng. Geol. 2009, 108, 76–85. [Google Scholar] [CrossRef]
- Rafique, T.; Naseem, S.; Bhanger, M.I.; Usmani, T.H. Fluoride ion contamination in the groundwater of Mithi sub-district, the Thar Desert, Pakistan. Environ. Earth Sci. 2008, 56, 317–326. [Google Scholar] [CrossRef]
- Meenakshi; Maheshwari, R. Fluoride in drinking water and its removal. J. Hazard. Mater. 2006, 137, 456–463. [Google Scholar] [CrossRef]
- Habuda-Stanić, M.; Ravančić, M.E.; Flanagan, A. A Review on Adsorption of Fluoride from Aqueous Solution. Materials 2014, 7, 6317–6366. [Google Scholar] [CrossRef]
- Waghmare, S.S.; Arfin, T. Fluoride removal by industrial, agricultural and biomass wastes as adsorbents: Review. J. Adv. Res. Innov. Ideas Educ. 2015, 1, 628–653. [Google Scholar]
- Tomar, V.; Kumar, D. A critical study on efficiency of different materials for fluoride removal from aqueous media. Chem. Cent. J. 2013, 7, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, P.S.; Suganya, S.; Srinivas, S.; Priyadharshini, S.; Karthika, M.; Karishma Sri, R.; Lichtfouse, E. Treatment of fluoride-contaminated water. A review. Environ. Chem. Lett. 2019, 17, 1707–1726. [Google Scholar] [CrossRef] [Green Version]
- Waghmare, S.S.; Arfin, T. Fluoride Removal by Clays, Geomaterials, Minerals, Low Cost Materials and Zeolites by Ad-sorption: A Review. Int. J. Sci. Eng. Technol. Res. 2015, 4, 3663–3676. [Google Scholar]
- Waghmare, S.S.; Arfin, T. Fluoride Removal from Water by various techniques: Review. IJISET—Int. J. Innov. Sci. Eng. Technol. 2015, 2, 560–571. [Google Scholar]
- Rao, N. Fluoride and Environment—A Review. In Proceedings of the 3rd International Conference on Environment and Health, Chennai, India, 15–17 December 2003; pp. 386–399. [Google Scholar]
- Schlesinger, W.H.; Klein, E.M.; Vengosh, A. Global Biogeochemical Cycle of Fluorine. Glob. Biogeochem. Cycles 2020, 34, e2020GB006722. [Google Scholar] [CrossRef]
- He, J.; Yang, Y.; Wu, Z.; Xie, C.; Zhang, K.; Kong, L.; Liu, J. Review of fluoride removal from water environment by adsorption. J. Environ. Chem. Eng. 2020, 8, 104516. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Kumar, E.; Sillanpää, M. Fluoride removal from water by adsorption—A review. Chem. Eng. J. 2011, 171, 811–840. [Google Scholar] [CrossRef]
- Wang, L.; Sun, N.; Tang, H.; Sun, W. A Review on Comprehensive Utilization of Red Mud and Prospect Analysis. Minerals 2019, 9, 362. [Google Scholar] [CrossRef] [Green Version]
- Arora, H.C.; Chattopadhya, S.N. A study on the effluent disposal of superphosphate fertilizer factory. Environ. Health 1994, 16, 140–150. [Google Scholar]
- Mourad, N.; Sharshar, T.; Elnimr, T.; Mousa, M. Radioactivity and fluoride contamination derived from a phosphate fertilizer plant in Egypt. Appl. Radiat. Isot. Incl. Data Instrum. Methods Use Agric. Ind. Med. 2009, 67, 1259–1268. [Google Scholar] [CrossRef]
- Shen, J.; Schaefer, A. Removal of fluoride and uranium by nanofiltration and reverse osmosis: A review. Chemosphere 2014, 117, 679–691. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.-S.; Li, K.-C. Production of insulating glass ceramics from thin film transistor-liquid crystal display (TFT-LCD) waste glass and calcium fluoride sludge. J. Clean. Prod. 2013, 57, 335–341. [Google Scholar] [CrossRef]
- Ponsot, I.; Falcone, R.; Bernardo, E. Stabilization of fluorine-containing industrial waste by production of sintered glass-ceramics. Ceram. Int. 2013, 39, 6907–6915. [Google Scholar] [CrossRef]
- Sujana, M.G.; Thakur, R.S.; Das, S.N.; Rao, S.B. Defluorination of Waste Water. Asian J. Chem. 1997, 9, 561–570. [Google Scholar]
- Shen, F.; Chen, X.; Gao, P.; Chen, G. Electrochemical removal of fluoride ions from industrial wastewater. Chem. Eng. Sci. 2003, 58, 987–993. [Google Scholar] [CrossRef]
- Blagojevic, S.; Jakovljevic, M.; Radulovic, M. Content of fluorine in soils in the vicinity of aluminium plant in Podgorica. J. Agric. Sci. 2002, 47, 1–8. [Google Scholar] [CrossRef]
- Paulson, E.G. Reducing fluoride in industrial wastewater. Chem. Eng. 1997, 84, 89–94. [Google Scholar]
- Khatibikamal, V.; Torabian, A.; Janpoor, F.; Hoshyaripour, G. Fluoride removal from industrial wastewater using electrocoagulation and its adsorption kinetics. J. Hazard. Mater. 2010, 179, 276–280. [Google Scholar] [CrossRef]
- Drouiche, N.; Aoudj, S.; Hecini, M.; Ghaffour, N.; Lounici, H.; Mameri, N. Study on the treatment of photovoltaic wastewater using electrocoagulation: Fluoride removal with aluminium electrodes—Characteristics of products. J. Hazard. Mater. 2009, 169, 65–69. [Google Scholar] [CrossRef]
- Drouiche, N.; Djouadi-Belkada, F.; Ouslimane, T.; Kefaifi, A.; Fathi, J.; Ahmetovic, E. Photovoltaic solar cells industry wastewater treatment. Desalination Water Treat. 2013, 51, 5965–5973. [Google Scholar] [CrossRef]
- Huang, C. Precipitate flotation of fluoride-containing wastewater from a semiconductor manufacturer. Water Res. 1999, 33, 3403–3412. [Google Scholar] [CrossRef]
- Paudyal, H.; Pangeni, B.; Inoue, K.; Kawakita, H.; Ohto, K.; Alam, S. Removal of Fluoride from Aqueous Solution by Using Porous Resins Containing Hydrated Oxide of Cerium(IV) and Zirconium(IV). J. Chem. Eng. Jpn. 2012, 45, 331–336. [Google Scholar] [CrossRef]
- Hu, C.-Y.; Lo, S.; Kuan, W.; Lee, Y. Removal of fluoride from semiconductor wastewater by electrocoagulation–flotation. Water Res. 2005, 39, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Warmadewanthi, B.; Liu, J.C. Selective separation of phosphate and fluoride from semiconductor wastewater. Water Sci. Technol. 2009, 59, 2047–2053. [Google Scholar] [CrossRef]
- Neuwahl, F.; Cusano, G.; Benavides, J.G.; Holbrook, S.; Roudier, S. Best Available Techniques (BAT) Reference Document for Waste Incineration; European Union: Luxembourg, 2006. [Google Scholar]
- Ghosh, A.; Mukherjee, K.; Ghosh, S.K.; Saha, B. Sources and toxicity of fluoride in the environment. Res. Chem. Intermed. 2012, 39, 2881–2915. [Google Scholar] [CrossRef]
- Dreveton, A. Overview of the Fluorochemicals Industrial Sectors. Procedia Eng. 2016, 138, 240–247. [Google Scholar] [CrossRef] [Green Version]
- Chlebna-Sokol, D. Wpływ Ponadoptymalnych Stężeń Fluorków w Wodzie Pitnej na Rozwój Biologiczny i Stan Zdrowia Dzieci w Wieku Szkolnym; Polska Akademia Nauk: Łódź, Poland, 1995. [Google Scholar]
- Ndiaye, P.; Moulin, P.; Dominguez, L.; Millet, J.; Charbit, F. Removal of fluoride from electronic industrial effluentby RO membrane separation. Desalination 2005, 173, 25–32. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Sun, N.; Sun, W.; Hu, Y.; Tang, H. Precipitation Methods Using Calcium-Containing Ores for Fluoride Removal in Wastewater. Minerals 2019, 9, 511. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Fan, Q.; Wang, S.; Liu, Y.; Zhou, A.; Fan, L. Adsorptive removal of fluoride from aqueous solutions using Al-humic acid-La aerogel composites. Chem. Eng. J. 2016, 306, 174–185. [Google Scholar] [CrossRef]
- Zeng, G.; Ling, B.; Li, Z.; Luo, S.; Sui, X.; Guan, Q. Fluorine removal and calcium fluoride recovery from rare-earth smelting wastewater using fluidized bed crystallization process. J. Hazard. Mater. 2019, 373, 313–320. [Google Scholar] [CrossRef]
- Raghav, S.; Nehra, S.; Kumar, D. Adsorptive removal studies of fluoride in aqueous system by bimetallic oxide incorporated in cellulose. Process. Saf. Environ. Prot. Trans. Inst. Chem. Eng. Part B 2019, 127, 211–225. [Google Scholar] [CrossRef]
- Chigondo, M.; Paumo, H.K.; Bhaumik, M.; Pillay, K.; Maity, A. Hydrous CeO2-Fe3O4 decorated polyaniline fibers nanocomposite for effective defluoridation of drinking water. J. Colloid Interface Sci. 2018, 532, 500–516. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, M.; Banerjee, A.; Pramanick, P.P.; Sarkar, A.R. Use of laterite for the removal of fluoride from contaminated drinking water. J. Colloid Interface Sci. 2006, 302, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Oguz, E. Adsorption of fluoride on gas concrete materials. J. Hazard. Mater. 2005, 117, 227–233. [Google Scholar] [CrossRef]
- Alagumuthu, G.; Veeraputhiran, V.; Venkataraman, R. Adsorption Isotherms on Fluoride Removal: Batch Techniques. Arch. Appl. Sci. Res. 2010, 2, 170–185. Available online: https://www.academia.edu/2555273/Adsorption_Isotherms_on_Fluoride_Removal_Batch_Techniques (accessed on 9 January 2022).
- Chaudhary, M.; Maiti, A. Defluoridation by highly efficient calcium hydroxide nanorods from synthetic and industrial wastewater. Colloids Surf. A Physicochem. Eng. Asp. 2019, 561, 79–88. [Google Scholar] [CrossRef]
- Venditti, F.; Cuomo, F.; Giansalvo, G.; Giustini, M.; Cinelli, G.; Lopez, F. Fluorides decontamination by means of Aluminum polychloride based commercial coagulant. J. Water Process. Eng. 2018, 26, 182–186. [Google Scholar] [CrossRef]
- Tolkou, A.K.; Mitrakas, M.; Katsoyiannis, I.A.; Ernst, M.; Zouboulis, A.I. Fluoride removal from water by composite Al/Fe/Si/Mg pre-polymerized coagulants: Characterization and application. Chemosphere 2019, 231, 528–537. [Google Scholar] [CrossRef]
- Turner, B.D.; Binning, P.; Stipp, S.L.S. Fluoride Removal by Calcite: Evidence for Fluorite Precipitation and Surface Adsorption. Environ. Sci. Technol. 2005, 39, 9561–9568. [Google Scholar] [CrossRef]
- El-Gohary, F.; Tawfik, A.; Mahmoud, U. Comparative study between chemical coagulation/precipitation (C/P) versus coagulation/dissolved air flotation (C/DAF) for pre-treatment of personal care products (PCPs) wastewater. Desalination 2010, 252, 106–112. [Google Scholar] [CrossRef]
- Saha, S. Treatment of aqueous effluent for fluoride removal. Water Res. 1993, 27, 1347–1350. [Google Scholar] [CrossRef]
- Reardon, E.J.; Wang, Y. A Limestone Reactor for Fluoride Removal from Wastewaters. Environ. Sci. Technol. 2000, 34, 3247–3253. [Google Scholar] [CrossRef]
- Gong, W.-X.; Qu, J.-H.; Liu, R.-P.; Lan, H.-C. Effect of aluminum fluoride complexation on fluoride removal by coagulation. Colloids Surf. A Physicochem. Eng. Asp. 2012, 395, 88–93. [Google Scholar] [CrossRef]
- Herath, H.M.A.S.; Kawakami, T.; Tafu, M. Repeated Heat Regeneration of Bone Char for Sustainable Use in Fluoride Removal from Drinking Water. Healthcare 2018, 6, 143. [Google Scholar] [CrossRef] [Green Version]
- Asimeng, B.O.; Fianko, J.R.; Kaufmann, E.E.; Tiburu, E.K.; Hayford, C.F.; Anani, P.A.; Dzikunu, O.K. Preparation and characterization of hydroxyapatite from Achatina achatina snail shells: Effect of carbonate substitution and trace elements on defluoridation of water. J. Asian Ceram. Soc. 2018, 6, 205–212. [Google Scholar] [CrossRef] [Green Version]
- Kodama, H.; Kabay, N. Reactivity of inorganic anion exchanger BiPbO2(NO3) with fluoride ions in solution. Solid State Ion. 2001, 141-142, 603–607. [Google Scholar] [CrossRef]
- Chubar, N.; Samanidou, V.; Kouts, V.; Gallios, G.; Kanibolotsky, V.; Strelko, V.; Zhuravlev, I. Adsorption of fluoride, chloride, bromide, and bromate ions on a novel ion exchanger. J. Colloid Interface Sci. 2005, 291, 67–74. [Google Scholar] [CrossRef]
- Hänninen, K.; Kaukonen, A.M.; Murtomäki, L.; Hirvonen, J.T. Mechanistic evaluation of factors affecting compound loading into ion-exchange fibers. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2007, 31, 306–317. [Google Scholar] [CrossRef]
- Ruixia, L.; Jinlong, G.; Hongxiao, T. Adsorption of Fluoride, Phosphate, and Arsenate Ions on a New Type of Ion Exchange Fiber. J. Colloid Interface Sci. 2002, 248, 268–274. [Google Scholar] [CrossRef]
- Meenakshi, S.; Viswanathan, N. Identification of selective ion-exchange resin for fluoride sorption. J. Colloid Interface Sci. 2007, 308, 438–450. [Google Scholar] [CrossRef]
- Paudyal, H.; Pangeni, B.; Inoue, K.; Kawakita, H.; Ohto, K.; Ghimire, K.N.; Alam, S. Preparation of novel alginate based anion exchanger from Ulva japonica and its application for the removal of trace concentrations of fluoride from water. Bioresour. Technol. 2013, 148, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Tian, J. Removal of fluoride and arsenate from aqueous solution by hydrocalumite via precipitation and anion exchange. Chem. Eng. J. 2013, 231, 121–131. [Google Scholar] [CrossRef]
- Hichour, M.; Persin, F.; Molénat, J.; Sandeaux, J.; Gavach, C. Fluoride removal from diluted solutions by Donnan dialysis with anion-exchange membranes. Desalination 1999, 122, 53–62. [Google Scholar] [CrossRef]
- Amor, Z.; Malki, S.; Taky, M.; Bariou, B.; Mameri, N.; Elmidaoui, A. Optimization of fluoride removal from brackish water by electrodialysis. Desalination 1998, 120, 263–271. [Google Scholar] [CrossRef]
- Ahamad, K.U.; Mahanta, A.; Ahmed, S. Removal of Fluoride from Groundwater by Adsorption onto Brick Powder–Alum–Calcium-Infused Adsorbent. Adv. Waste Manag. 2019, 231–242. [Google Scholar] [CrossRef]
- Hashim, K.S.; Shaw, A.; Al Khaddar, R.; Pedrola, M.O.; Phipps, D. Defluoridation of drinking water using a new flow column-electrocoagulation reactor (FCER)—Experimental, statistical, and economic approach. J. Environ. Manag. 2017, 197, 80–88. [Google Scholar] [CrossRef]
- Cui, H.; Qian, Y.; An, H.; Sun, C.; Zhai, J.; Li, Q. Electrochemical removal of fluoride from water by PAOA-modified carbon felt electrodes in a continuous flow reactor. Water Res. 2012, 46, 3943–3950. [Google Scholar] [CrossRef]
- Lin, J.-Y.; Raharjo, A.; Hsu, L.-H.; Shih, Y.-J.; Huang, Y.-H. Electrocoagulation of tetrafluoroborate (BF4−) and the derived boron and fluorine using aluminum electrodes. Water Res. 2019, 155, 362–371. [Google Scholar] [CrossRef]
- Tahaikt, M.; Achary, I.; Sahli, M.M.; Amor, Z.; Taky, M.; Alami, A.; Boughriba, A.; Hafsi, M.; Elmidaoui, A. Defluoridation of Moroccan groundwater by electrodialysis: Continuous operation. Desalination 2006, 189, 215–220. [Google Scholar] [CrossRef]
- Renuka, P.; Pushpanjali, K. Review on Defluoridation Techniques of Water. Int. J. Eng. Sci. 2013, 2, 86–94. [Google Scholar]
- Sahli, M.M.; Annouar, S.; Tahaikt, M.; Mountadar, M.; Soufiane, A.; Elmidaoui, A. Fluoride removal for underground brackish water by adsorption on the natural chitosan and by electrodialysis. Desalination 2007, 212, 37–45. [Google Scholar] [CrossRef]
- Zuo, Q.; Chen, X.; Li, W.; Chen, G. Combined electrocoagulation and electroflotation for removal of fluoride from drinking water. J. Hazard. Mater. 2008, 159, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Ergun, E.; Tor, A.; Cengeloglu, Y.; Kocak, I. Electrodialytic removal of fluoride from water: Effects of process parameters and accompanying anions. Sep. Purif. Technol. 2008, 64, 147–153. [Google Scholar] [CrossRef]
- Kabay, N.; Arar, Ö.; Samatya, S.; Yüksel, Ü.; Yüksel, M. Separation of fluoride from aqueous solution by electrodialysis: Effect of process parameters and other ionic species. J. Hazard. Mater. 2008, 153, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Mameri, N.; Lounici, H.; Belhocine, D.; Grib, H.; Piron, D.; Yahiat, Y. Defluoridation of Sahara water by small plant electrocoagulation using bipolar aluminium electrodes. Sep. Purif. Technol. 2001, 24, 113–119. [Google Scholar] [CrossRef]
- Arar, O.; Yavuz, E.; Yuksel, U.; Kabay, N. Separation of Low Concentration of Fluoride from Water by Electrodialysis (ED) in the Presence of Chloride and Sulfate Ions. Sep. Sci. Technol. 2009, 44, 1562–1573. [Google Scholar] [CrossRef]
- Un, U.T.; Koparal, A.S.; Ogutveren, U.B. Fluoride removal from water and wastewater with a bach cylindrical electrode using electrocoagulation. Chem. Eng. J. 2013, 223, 110–115. [Google Scholar] [CrossRef]
- Karabelas, A.; Yiantsios, S.; Metaxiotou, Z.; Andritsos, N.; Akiskalos, A.; Vlachopoulos, G.; Stavroulias, S. Water and materials recovery from fertilizer industry acidic effluents by membrane processes. Desalination 2001, 138, 93–102. [Google Scholar] [CrossRef]
- Sehn, P. Fluoride removal with extra low energy reverse osmosis membranes: Three years of large scale field experience in Finland. Desalination 2008, 223, 73–84. [Google Scholar] [CrossRef]
- Guo, L.; Hunt, B.J.; Santschi, P.H. Ultrafiltration behavior of major ions (Na, Ca, Mg, F, Cl, and SO4) in natural waters. Water Res. 2001, 35, 1500–1508. [Google Scholar] [CrossRef]
- Lhassani, A.; Rumeau, M.; Benjelloun, D.; Pontie, M. Selective demineralization of water by nanofiltration Application to the defluorination of brackish water. Water Res. 2001, 35, 3260–3264. [Google Scholar] [CrossRef]
- Hu, K.; Dickson, J.M. Nanofiltration membrane performance on fluoride removal from water. J. Membr. Sci. 2006, 279, 529–538. [Google Scholar] [CrossRef]
- Malaisamy, R.; Talla-Nwafo, A.; Jones, K.L. Polyelectrolyte modification of nanofiltration membrane for selective removal of monovalent anions. Sep. Purif. Technol. 2011, 77, 367–374. [Google Scholar] [CrossRef]
- Ghosh, D.; Sinha, M.; Purkait, M. A comparative analysis of low-cost ceramic membrane preparation for effective fluoride removal using hybrid technique. Desalination 2013, 327, 2–13. [Google Scholar] [CrossRef]
- Chakrabortty, S.; Roy, M.; Pal, P. Removal of fluoride from contaminated groundwater by cross flow nanofiltration: Transport modeling and economic evaluation. Desalination 2013, 313, 115–124. [Google Scholar] [CrossRef]
- Yadav, K.K.; Kumar, S.; Pham, Q.B.; Gupta, N.; Rezania, S.; Kamyab, H.; Yadav, S.; Vymazal, J.; Kumar, V.; Tri, D.Q.; et al. Fluoride contamination, health problems and remediation methods in Asian groundwater: A comprehensive review. Ecotoxicol. Environ. Saf. 2019, 182, 109362. [Google Scholar] [CrossRef]
- Jeihanipour, A.; Shen, J.; Abbt-Braun, G.; Huber, S.A.; Mkongo, G.; Schäfer, A.I. Seasonal variation of organic matter characteristics and fluoride concentration in the Maji ya Chai River (Tanzania): Impact on treatability by nanofiltration/reverse osmosis. Sci. Total Environ. 2018, 637-638, 1209–1220. [Google Scholar] [CrossRef]
- Grzegorzek, M.; Majewska-Nowak, K. The use of micellar-enhanced ultrafiltration (MEUF) for fluoride removal from aqueous solutions. Sep. Purif. Technol. 2018, 195, 1–11. [Google Scholar] [CrossRef]
- Sequeira, E.A.T.; Miranda, V.M.; Solache-Ríos, M.; Hernández, I.L. Aluminum and lanthanum effects in natural materials on the adsorption of fluoride ions. J. Fluor. Chem. 2013, 148, 6–13. [Google Scholar] [CrossRef]
- Saxena, A.; Patel, A. Role of Bioremediation as a Low-Cost Adsorbent for Excessive Fluoride Removal in Groundwater. In Handbook of Environmental Materials Management; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1–32. [Google Scholar] [CrossRef]
- Mohan, D.; Singh, K.P.; Singh, V.K. Wastewater treatment using low cost activated carbons derived from agricultural byproducts—A case study. J. Hazard. Mater. 2008, 152, 1045–1053. [Google Scholar] [CrossRef]
- Alagumuthu, G.; Rajan, M. Kinetic and equilibrium studies on fluoride removal by zirconium (IV): Impregnated groundnut shell carbon. Chem. Ind. 2010, 64, 295–304. [Google Scholar] [CrossRef] [Green Version]
- Alagumuthu, G.; Veeraputhiran, V.; Venkataraman, R. Fluoride sorption using Cynodon dactylon based activated carbon. Chem. Ind. 2011, 65, 23–35. [Google Scholar] [CrossRef]
- Alagumuthu, G.; Rajan, M. Equilibrium and kinetics of adsorption of fluoride onto zirconium impregnated cashew nut shell carbon. Chem. Eng. J. 2010, 158, 451–457. [Google Scholar] [CrossRef]
- Daifullah, A.A.M.; Yakout, S.M.; A Elreefy, S. Adsorption of fluoride in aqueous solutions using KMnO4-modified activated carbon derived from steam pyrolysis of rice straw. J. Hazard. Mater. 2007, 147, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Montoya, V.; Ramírez-Montoya, L.A.; Bonilla-Petriciolet, A.; Montes-Moran, M.A. Optimizing the removal of fluoride from water using new carbons obtained by modification of nut shell with a calcium solution from egg shell. Biochem. Eng. J. 2012, 62, 1–7. [Google Scholar] [CrossRef]
- Meenakshi, S. Studies on Defluoridation of Water with a Few Adsorbents and Development of an Indigenous Defluoridation Unit for Do-mestic Use; The Gandhigram Rural Institute: Tamil Nadu, India, 1992. [Google Scholar]
- Malay, D.K.; Salim, A.J. Salim, Comparative Study of Batch Adsorption of Fluoride Using Commercial and Natural Adsorbent. Res. J. Chem. Sci. 2011, 1, 68–75. [Google Scholar]
- Viswanathan, N.; Meenakshi, S. Role of metal ion incorporation in ion exchange resin on the selectivity of fluoride. J. Hazard. Mater. 2009, 162, 920–930. [Google Scholar] [CrossRef]
- Viswanathan, N.; Meenakshi, S. Effect of metal ion loaded in a resin towards fluoride retention. J. Fluor. Chem. 2008, 129, 645–653. [Google Scholar] [CrossRef]
- Vardhan, C.V.; Karthikeyan, J. Removal of Fluoride from Water Using Low-Cost Materials. In Proceedings of the Fifteenth International Water Technology Conference, IWTC-15, Alexandria, Egypt, 28–30 May 2011. [Google Scholar]
- Coetzee, P.; Coetzee, L.; Puka, R.; Mubenga, S. Characterisation of selected South African clays for defluoridation of natural waters. Water SA 2004, 29, 331–338. [Google Scholar] [CrossRef] [Green Version]
- Yadav, A.K.; Kaushik, C.P.; Haritash, A.K.; Kansal, A.; Rani, N. Defluoridation of groundwater using brick powder as an adsorbent. J. Hazard. Mater. 2006, 128, 289–293. [Google Scholar] [CrossRef]
- Malakootian, M.; Moosazadeh, M.; Yousefi, N.; Fatehizadeh, A. Fluoride removal from aqueous solution by pumice: Case study on Kuhbonan water. Afr. J. Environ. Sci. Technol. 2011, 5, 299–306. [Google Scholar] [CrossRef]
- Chidambaram, S.; Ramanathan, A.; Vasudevan, S. Fluoride removal studies in water using natural materials: Technical note. Water SA 2004, 29, 339–344. [Google Scholar] [CrossRef] [Green Version]
- Maliyekkal, S.M.; Shukla, S.; Philip, L.; Nambi, I.M. Enhanced fluoride removal from drinking water by magnesia-amended activated alumina granules. Chem. Eng. J. 2008, 140, 183–192. [Google Scholar] [CrossRef]
- Tripathy, S.S.; Raichur, A. Abatement of fluoride from water using manganese dioxide-coated activated alumina. J. Hazard. Mater. 2008, 153, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Lavecchia, R.; Medici, F.; Piga, L.; Rinaldi, G.; Zuorro, A. Fluoride removal from water by adsorption on a high-alumina content bauxite. Chem. Eng. Trans. 2012, 26, 225–230. [Google Scholar] [CrossRef]
- Teutli-Sequeira, A.; Solache-Rios, M.; Balderas-Hernández, P. Modification Effects of Hematite with Aluminum Hydroxide on the Removal of Fluoride Ions from Water. Water Air Soil Pollut. 2011, 223, 319–327. [Google Scholar] [CrossRef]
- Shan, Y.; Guo, H. Fluoride adsorption on modified natural siderite: Optimization and performance. Chem. Eng. J. 2013, 223, 183–191. [Google Scholar] [CrossRef]
- Sajidu, S.; Kayira, C.; Masamba, W.; Mwatseteza, J. Defluoridation of Groundwater Using Raw Bauxite: Rural Domestic Defluoridation Technology. Environ. Nat. Resour. Res. 2012, 2, 1. [Google Scholar] [CrossRef]
- Goswami, D.; Das, A.K. Removal of fluoride from drinking water using a modified fly ash adsorbent. J. Sci. Ind. Res. 2006, 65, 77–79. [Google Scholar]
- Sundaram, C.S.; Viswanathan, N.; Meenakshi, S. Defluoridation chemistry of synthetic hydroxyapatite at nano scale: Equilibrium and kinetic studies. J. Hazard. Mater. 2008, 155, 206–215. [Google Scholar] [CrossRef]
- Sundaram, C.S.; Viswanathan, N.; Meenakshi, S. Uptake of fluoride by nano-hydroxyapatite/chitosan, a bioinorganic composite. Bioresour. Technol. 2008, 99, 8226–8230. [Google Scholar] [CrossRef] [PubMed]
- Salifu, A.; Petrusevski, B.; Ghebremichael, K.; Modestus, L.; Buamah, R.; Aubry, C.; Amy, G. Aluminum (hydr)oxide coated pumice for fluoride removal from drinking water: Synthesis, equilibrium, kinetics and mechanism. Chem. Eng. J. 2013, 228, 63–74. [Google Scholar] [CrossRef]
- Tembhurkar, A.R.; Dongre, S. Studies on fluoride removal using adsorption process. J. Environ. Sci. Eng. Technol. 2006, 48, 151–156. [Google Scholar]
- Nath, S.K.; Dutta, R.K. Fluoride removal from water using crushed limestone. Indian J. Chem. Technol. 2010, 17, 120–125. [Google Scholar]
- Bhargava, D.; Killedar, D. Fluoride adsorption on fishbone charcoal through a moving media adsorber. Water Res. 1992, 26, 781–788. [Google Scholar] [CrossRef]
- Çengeloglu, Y. Removal of fluoride from aqueous solution by using red mud. Sep. Purif. Technol. 2002, 28, 81–86. [Google Scholar] [CrossRef]
- Asgari, G.; Roshani, B.; Ghanizadeh, G. The investigation of kinetic and isotherm of fluoride adsorption onto functionalize pumice stone. J. Hazard. Mater. 2012, 217–218, 123–132. [Google Scholar] [CrossRef]
- Chen, N.; Zhang, Z.; Feng, C.; Li, M.; Zhu, D.; Sugiura, N. Studies on fluoride adsorption of iron-impregnated granular ceramics from aqueous solution. Mater. Chem. Phys. 2011, 125, 293–298. [Google Scholar] [CrossRef]
- Kamble, S.P.; Jagtap, S.; Labhsetwar, N.K.; Thakare, D.; Godfrey, S.; Devotta, S.; Rayalu, S.S. Defluoridation of drinking water using chitin, chitosan and lanthanum-modified chitosan. Chem. Eng. J. 2007, 129, 173–180. [Google Scholar] [CrossRef]
- Viswanathan, N.; Meenakshi, S. Development of chitosan supported zirconium(IV) tungstophosphate composite for fluoride removal. J. Hazard. Mater. 2010, 176, 459–465. [Google Scholar] [CrossRef]
- Jagtap, S.; Thakre, D.; Wanjari, S.; Kamble, S.; Labhsetwar, N.; Rayalu, S. New modified chitosan-based adsorbent for defluoridation of water. J. Colloid Interface Sci. 2009, 332, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Sujana, M.; Mishra, A.; Acharya, B. Hydrous ferric oxide doped alginate beads for fluoride removal: Adsorption kinetics and equilibrium studies. Appl. Surf. Sci. 2013, 270, 767–776. [Google Scholar] [CrossRef]
- Liang, P.; Zhang, Y.; Wang, D.; Xu, Y.; Luo, L. Preparation of mixed rare earths modified chitosan for fluoride adsorption. J. Rare Earths 2013, 31, 817–822. [Google Scholar] [CrossRef]
- Davila-Rodriguez, J.L.; Escobar-Barrios, V.; Rangel-Mendez, J.R. Removal of fluoride from drinking water by a chitin-based biocomposite in fixed-bed columns. J. Fluor. Chem. 2012, 140, 99–103. [Google Scholar] [CrossRef]
- Swain, S.K.; Patnaik, T.; Patnaik, P.; Jha, U.; Dey, R. Development of new alginate entrapped Fe(III)–Zr(IV) binary mixed oxide for removal of fluoride from water bodies. Chem. Eng. J. 2013, 215-216, 763–771. [Google Scholar] [CrossRef]
- Shams, M.; Nabizadeh Nodehi, R.; Hadi Dehghani, M.; Younesian, M.; Hossein Mahvia, A. Efficiency of granular ferric hydroxide (GFH) for removal of fluoride from water. Fluoride 2010, 43, 61–66. [Google Scholar]
- Chai, L.; Wang, Y.; Zhao, N.; Yang, W.; You, X. Sulfate-doped Fe3O4/Al2O3 nanoparticles as a novel adsorbent for fluoride removal from drinking water. Water Res. 2013, 47, 4040–4049. [Google Scholar] [CrossRef]
- Liu, R.; Gong, W.; Lan, H.; Yang, T.; Liu, H.; Qu, J. Simultaneous removal of arsenate and fluoride by iron and aluminum binary oxide: Competitive adsorption effects. Sep. Purif. Technol. 2012, 92, 100–105. [Google Scholar] [CrossRef]
- García-Sánchez, J.; Solache-Ríos, M.; Miranda, V.M.; Morelos, C.S. Removal of fluoride ions from drinking water and fluoride solutions by aluminum modified iron oxides in a column system. J. Colloid Interface Sci. 2013, 407, 410–415. [Google Scholar] [CrossRef]
- Kang, D.; Yu, X.; Tong, S.; Ge, M.; Zuo, J.; Cao, C.; Song, W. Performance and mechanism of Mg/Fe layered double hydroxides for fluoride and arsenate removal from aqueous solution. Chem. Eng. J. 2013, 228, 731–740. [Google Scholar] [CrossRef]
- Wu, H.-X.; Wang, T.-J.; Chen, L.; Jin, Y.; Zhang, Y.; Dou, X.-M. Granulation of Fe–Al–Ce hydroxide nano-adsorbent by immobilization in porous polyvinyl alcohol for fluoride removal in drinking water. Powder Technol. 2011, 209, 92–97. [Google Scholar] [CrossRef]
- Dou, X.; Zhang, Y.; Wang, H.; Wang, T.; Wang, Y. Performance of granular zirconium–iron oxide in the removal of fluoride from drinking water. Water Res. 2011, 45, 3571–3578. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, N.; Prabhu, S.M.; Meenakshi, S. Development of amine functionalized co-polymeric resins for selective fluoride sorption. J. Fluor. Chem. 2013, 153, 143–150. [Google Scholar] [CrossRef]
- Ganvir, V.; Das, K. Removal of fluoride from drinking water using aluminum hydroxide coated rice husk ash. J. Hazard. Mater. 2011, 185, 1287–1294. [Google Scholar] [CrossRef]
- Mourabet, M.; El Rhilassi, A.; El Boujaady, H.; Bennani-Ziatni, M.; El Hamri, R.; Taitai, A. Removal of fluoride from aqueous solution by adsorption on hydroxyapatite (HAp) using response surface methodology. J. Saudi Chem. Soc. 2015, 19, 603–615. [Google Scholar] [CrossRef] [Green Version]
- Mourabet, M.; El Rhilassi, A.; El Boujaady, H.; Bennani-Ziatni, M.; El Hamri, R.; Taitai, A. Removal of fluoride from aqueous solution by adsorption on Apatitic tricalcium phosphate using Box–Behnken design and desirability function. Appl. Surf. Sci. 2012, 258, 4402–4410. [Google Scholar] [CrossRef]
- Zhang, D.; Luo, H.; Zheng, L.; Wang, K.; Li, H.; Wang, Y.; Feng, H. Utilization of waste phosphogypsum to prepare hydroxyapatite nanoparticles and its application towards removal of fluoride from aqueous solution. J. Hazard. Mater. 2012, 241–242, 418–426. [Google Scholar] [CrossRef]
- Dou, X.; Mohan, D.; Pittman, C.U.; Yang, S. Remediating fluoride from water using hydrous zirconium oxide. Chem. Eng. J. 2012, 198-199, 236–245. [Google Scholar] [CrossRef]
- Swain, S.K.; Patnaik, T.; Singh, V.; Jha, U.; Patel, R.; Dey, R. Kinetics, equilibrium and thermodynamic aspects of removal of fluoride from drinking water using meso-structured zirconium phosphate. Chem. Eng. J. 2011, 171, 1218–1226. [Google Scholar] [CrossRef]
- Poursaberi, T.; Hassanisadi, M.; Torkestani, K.; Zare, M. Development of zirconium (IV)-metalloporphyrin grafted Fe3O4 nanoparticles for efficient fluoride removal. Chem. Eng. J. 2012, 189-190, 117–125. [Google Scholar] [CrossRef]
- Swain, S.K.; Mishra, S.; Patnaik, T.; Patel, R.; Jha, U.; Dey, R. Fluoride removal performance of a new hybrid sorbent of Zr(IV)–ethylenediamine. Chem. Eng. J. 2012, 184, 72–81. [Google Scholar] [CrossRef]
- Koilraj, P.; Kannan, S. Aqueous fluoride removal using ZnCr layered double hydroxides and their polymeric composites: Batch and column studies. Chem. Eng. J. 2013, 234, 406–415. [Google Scholar] [CrossRef]
- Wang, J.; Xu, W.; Chen, L.; Jia, Y.; Wang, L.; Huang, X.-J.; Liu, J. Excellent fluoride removal performance by CeO2–ZrO2 nanocages in water environment. Chem. Eng. J. 2013, 231, 198–205. [Google Scholar] [CrossRef]
- Chen, L.; Wang, T.-J.; Wu, H.-X.; Jin, Y.; Zhang, Y.; Dou, X. Optimization of a Fe–Al–Ce nano-adsorbent granulation process that used spray coating in a fluidized bed for fluoride removal from drinking water. Powder Technol. 2011, 206, 291–296. [Google Scholar] [CrossRef]
- Zhao, B.; Zhang, Y.; Dou, X.; Wu, X.; Yang, M. Granulation of Fe–Al–Ce trimetal hydroxide as a fluoride adsorbent using the extrusion method. Chem. Eng. J. 2012, 185–186, 211–218. [Google Scholar] [CrossRef]
- Sivasankar, V.; Murugesh, S.; Rajkumar, S.; Darchen, A. Cerium dispersed in carbon (CeDC) and its adsorption behavior: A first example of tailored adsorbent for fluoride removal from drinking water. Chem. Eng. J. 2013, 214, 45–54. [Google Scholar] [CrossRef]
- Mandal, S.; Tripathy, S.; Padhi, T.; Sahu, M.K.; Patel, R.K. Removal efficiency of fluoride by novel Mg-Cr-Cl layered double hydroxide by batch process from water. J. Environ. Sci. 2013, 25, 993–1000. [Google Scholar] [CrossRef]
- Zhang, T.; Li, Q.; Xiao, H.; Mei, Z.; Lu, H.; Zhou, Y. Enhanced fluoride removal from water by non-thermal plasma modified CeO2/Mg–Fe layered double hydroxides. Appl. Clay Sci. 2013, 72, 117–123. [Google Scholar] [CrossRef]
- Wajima, T.; Umeta, Y.; Narita, S.; Sugawara, K. Adsorption behavior of fluoride ions using a titanium hydroxide-derived adsorbent. Desalination 2009, 249, 323–330. [Google Scholar] [CrossRef]
- Chen, L.; He, B.-Y.; He, S.; Wang, T.-J.; Su, C.-L.; Jin, Y. Fe―Ti oxide nano-adsorbent synthesized by co-precipitation for fluoride removal from drinking water and its adsorption mechanism. Powder Technol. 2012, 227, 3–8. [Google Scholar] [CrossRef]
- Babaeivelni, K.; Khodadoust, A.P. Adsorption of fluoride onto crystalline titanium dioxide: Effect of pH, ionic strength, and co-existing ions. J. Colloid Interface Sci. 2013, 394, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Eskandarpour, A.; Onyango, M.S.; Ochieng, A.; Asai, S. Removal of fluoride ions from aqueous solution at low pH using schwertmannite. J. Hazard. Mater. 2008, 152, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, X.; Liu, L.; Chen, F. Fluoride removal by Fe(III)-loaded ligand exchange cotton cellulose adsorbent from drinking water. Carbohydr. Polym. 2008, 72, 144–150. [Google Scholar] [CrossRef]
- Yu, X.; Tong, S.; Ge, M.; Zuo, J. Removal of fluoride from drinking water by cellulose@hydroxyapatite nanocomposites. Carbohydr. Polym. 2013, 92, 269–275. [Google Scholar] [CrossRef]
- Gogoi, P.K.; Baruah, R. Fluoride removal from water by adsorption on acid activated kaolinite clay. Indian J. Chem. Technol. 2008, 15, 500–503. [Google Scholar]
- Meenakshi, S.; Sundaram, C.S.; Sukumar, R. Enhanced fluoride sorption by mechanochemically activated kaolinites. J. Hazard. Mater. 2008, 153, 164–172. [Google Scholar] [CrossRef]
- Guo, Q.; Reardon, E.J. Fluoride removal from water by meixnerite and its calcination product. Appl. Clay Sci. 2012, 56, 7–15. [Google Scholar] [CrossRef]
- Suzuki, T.; Nakamura, A.; Niinae, M.; Nakata, H.; Fujii, H.; Tasaka, Y. Immobilization of fluoride in artificially contaminated kaolinite by the addition of commercial-grade magnesium oxide. Chem. Eng. J. 2013, 233, 176–184. [Google Scholar] [CrossRef]
- Lebedynets, M.; Sprynskyy, M.; Sakhnyuk, I.; Zbytniewski, R.; Golembiewski, R.; Buszewski, B. Adsorption of Ammonium Ions onto a Natural Zeolite: Transcarpathian Clinoptilolite. Adsorpt. Sci. Technol. 2016, 22, 731–741. [Google Scholar] [CrossRef] [Green Version]
- Erdem, E.; Karapinar, N.; Donat, R. The removal of heavy metal cations by natural zeolites. J. Colloid Interface Sci. 2004, 280, 309–314. [Google Scholar] [CrossRef]
- Rahmani, A.; Nouri, J.; Ghadiri, S.K.; Mahvi, A.; Zare, M.R. Adsorption of fluoride from water by Al3+ and Fe3+ pretreated natural Iranian zeolites. Int. J. Environ. Res. 2010, 4, 607–614. [Google Scholar] [CrossRef]
- Wang, S.; Peng, Y. Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- Sun, Y.; Fang, Q.; Dong, J.; Cheng, X.; Xu, J. Removal of fluoride from drinking water by natural stilbite zeolite modified with Fe(III). Desalination 2011, 277, 121–127. [Google Scholar] [CrossRef]
- Gómez-Hortigüela, L.; Pérez-Pariente, J.; García, R.; Chebude, Y.; Díaz, I. Natural zeolites from Ethiopia for elimination of fluoride from drinking water. Sep. Purif. Technol. 2013, 120, 224–229. [Google Scholar] [CrossRef]
- Sasaki, K.; Fukumoto, N.; Moriyama, S.; Yu, Q.; Hirajima, T. Chemical regeneration of magnesium oxide used as a sorbent for fluoride. Sep. Purif. Technol. 2012, 98, 24–30. [Google Scholar] [CrossRef]
- Tor, A.; Danaoglu, N.; Arslan, G.; Cengeloglu, Y. Removal of fluoride from water by using granular red mud: Batch and column studies. J. Hazard. Mater. 2009, 164, 271–278. [Google Scholar] [CrossRef]
- Ning, W.; Zhao-Kun, L.; Jun, W.; Li, S.; Ying, Z.; Jie-Wei, W. Preparation of Modified Red Mud with Aluminum and Its Adsorption Characteristics on Fluoride Removal. Chin. J. Inorg. Chem. 2009, 25, 849–854. [Google Scholar]
- Lv, G.; Wu, L.; Liao, L.; Zhang, Y.; Li, Z. Preparation and characterization of red mud sintered porous materials for water defluoridation. Appl. Clay Sci. 2013, 74, 95–101. [Google Scholar] [CrossRef]
- Tor, A.; Danaoglu, N.; Arslan, G.; Cengeloglu, Y. Removal of Fluoride From Drinking Water Using Red Mud Introduction. Int. J. Sci. Technol. Res. 2013, 2, 120–122. [Google Scholar]
- Zhu, S.; Zhu, D.; Wang, X. Removal of fluorine from red mud (bauxite residue) by electrokinetics. Electrochim. Acta 2017, 242, 300–306. [Google Scholar] [CrossRef]
- Kemer, B.; Ozdes, D.; Gundogdu, A.; Bulut, V.N.; Duran, C.; Soylak, M. Removal of fluoride ions from aqueous solution by waste mud. J. Hazard. Mater. 2009, 168, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Sujana, M.; Thakur, R.; Rao, S. Removal of Fluoride from Aqueous Solution by Using Alum Sludge. J. Colloid Interface Sci. 1998, 206, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Nigussie, W.; Zewge, F.; Chandravanshi, B. Removal of excess fluoride from water using waste residue from alum manufacturing process. J. Hazard. Mater. 2007, 147, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Mahramanlioglu, M.; Kizilcikli, I.; Bicer, I.O. Adsorption of fluoride from aqueous solution by acid treated spent bleaching earth. J. Fluor. Chem. 2002, 115, 41–47. [Google Scholar] [CrossRef]
- Malakootian, M.; Fatehizadeh, A.; Yousefi, N.; Ahmadian, M.; Moosazadeh, M. Fluoride removal using Regenerated Spent Bleaching Earth (RSBE) from groundwater: Case study on Kuhbonan water. Desalination 2011, 277, 244–249. [Google Scholar] [CrossRef]
- Nemade, P.D.; Vasudeva Rao, A.; Alappat, B.J. Removal of fluorides from water using low cost adsorbents. Water Supply 2002, 2, 311–317. [Google Scholar] [CrossRef]
- Xue, Y.; Hou, H.; Zhu, S.; Zha, J. Utilization of municipal solid waste incineration ash in stone mastic asphalt mixture: Pavement performance and environmental impact. Constr. Build. Mater. 2009, 23, 989–996. [Google Scholar] [CrossRef]
- Geethamani, C.; Ramesh, S.; Gandhimathi, R.; Nidheesh, P. Alkali-treated fly ash for the removal of fluoride from aqueous solutions. Desalin. Water Treat. 2013, 52, 3466–3476. [Google Scholar] [CrossRef]
- Ramesh, S.; Gandhimathi, R.; Nidheesh, P.; Taywade, M. Batch and Column Operations for the Removal of Fluoride from Aqueous Solution Using Bottom Ash. Environ. Res. Eng. Manag. 2012, 60, 12–20. [Google Scholar] [CrossRef]
- Zhang, G.; Sun, G.; Chen, Z.; Evrendilek, F.; Liu, J. Water-soluble fluorine detoxification mechanisms of spent potlining incineration in response to calcium compounds. Environ. Pollut. 2020, 266, 115420. [Google Scholar] [CrossRef]
- Xu, X.; Li, Q.; Cui, H.; Pang, J.; An, H.; Wang, W.; Zhai, J. Column-mode fluoride removal from aqueous solution by magnesia-loaded fly ash cenospheres. Environ. Technol. 2012, 33, 1409–1415. [Google Scholar] [CrossRef] [PubMed]
- Mondal, N.K.; Bhaumik, R.; Baur, T.; Das, B.; Roy, P.; Datta, P.R.A.J.K. Studies on Defluoridation of Water by Tea Ash: An Unconventional Biosorbent. Chem. Sci. Trans. 2012, 1, 239–256. [Google Scholar] [CrossRef]
- Gupta, N.; Gupta, V.; Singh, A.P.; Singh, R.P. Defluoridation of Groundwater using Low Cost Adsorbent like Bagasse Dust, Aluminium Treated Bagasse Flyash, Bone Powder and Shell Powder. Bonfring Int. J. Ind. Eng. Manag. Sci. 2014, 4, 72–75. [Google Scholar] [CrossRef]
- Jadhav, A.S.; Jadhav, M.V. Use of Maize Husk Fly Ash as an Adsorbent for Removal of Fluoride from Water. Int. J. Recent Dev. Eng. Technol. 2014, 2, 2. Available online: www.ijrdet.com (accessed on 25 July 2021).
- Gupta, V.K.; Ali, I.; Saini, V.K. Defluoridation of wastewaters using waste carbon slurry. Water Res. 2007, 41, 3307–3316. [Google Scholar] [CrossRef]
- Cinarli, A.; Bicer, O.; Mahramanlioglu, M. Removal of fluoride using the adsorbents produced from mining waste. Fresenius Environ. Bull. 2005, 14, 520–525. [Google Scholar]
- Kumari, M.; Adhikari, K.; Dutta, S. Fluoride removal using shale: A mine waste. In India Water Week-Efficient Water Management: Challenges and Opportunities; Government of India, Ministry of Water Resources: New Delhi, India, 2013; p. 157. [Google Scholar]
- Islam, M.; Patel, R. Thermal activation of basic oxygen furnace slag and evaluation of its fluoride removal efficiency. Chem. Eng. J. 2011, 169, 68–77. [Google Scholar] [CrossRef]
- Lai, Y.D.; Liu, J.C. Fluoride Removal from Water with Spent Catalyst. Sep. Sci. Technol. 1996, 31, 2791–2803. [Google Scholar] [CrossRef]
- Tsai, C.Y.; Liu, J.C. Fluoride removal from water with iron-coated spent catalyst. Chinese J. Environ. Eng. 1999, 9, 107–114. [Google Scholar]
- Das, N.; Pattanaik, P.; Das, R. Defluoridation of drinking water using activated titanium rich bauxite. J. Colloid Interface Sci. 2005, 292, 1–10. [Google Scholar] [CrossRef]
- Chaudhari, V.S.; Sasane, V.V. Investigation of Optimum Operating Parameters for Removal of Fluoride Using Naturally Available Geomaterial. Int. J. Eng. Res. Technol. 2014, 3, 2123–2128. [Google Scholar]
- Kang, W.-H.; Kim, E.-I.; Park, J.-Y. Fluoride removal capacity of cement paste. Desalination 2007, 202, 38–44. [Google Scholar] [CrossRef]
- Oguz, E. Equilibrium isotherms and kinetics studies for the sorption of fluoride on light weight concrete materials. Colloids Surf. A Physicochem. Eng. Asp. 2007, 295, 258–263. [Google Scholar] [CrossRef]
- Kagne, S.; Jagtap, S.; Dhawade, P.; Kamble, S.; Devotta, S.; Rayalu, S. Hydrated cement: A promising adsorbent for the removal of fluoride from aqueous solution. J. Hazard. Mater. 2008, 154, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Ayoob, S.; Gupta, A. Insights into isotherm making in the sorptive removal of fluoride from drinking water. J. Hazard. Mater. 2008, 152, 976–985. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Cui, J.; Wei, Z. Study on the fluoride adsorption of various apatite materials in aqueous solution. J. Fluor. Chem. 2009, 130, 1035–1041. [Google Scholar] [CrossRef]
- Gao, S.; Sun, R.; Wei, Z.; Zhao, H.; Li, H.; Hu, F.; Gao, S.; Sun, R. Size-dependent defluoridation ultrasonic and microwave combined technique. J. Hazard. Mater. 2011, 185, 29–37. [Google Scholar]
- Poinern, G.E.J.; Ghosh, M.K.; Ng, Y.-J.; Issa, T.B.; Anand, S.; Singh, P. Defluoridation behavior of nanostructured hydroxyapatite synthesized through an ultrasonic and microwave combined technique. J. Hazard. Mater. 2011, 185, 29–37. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, N.; Wei, W.; Cui, J.; Wei, Z. Enhanced adsorption of fluoride from aqueous solution onto nanosized hydroxyapatite by low-molecular-weight organic acids. Desalination 2011, 276, 161–168. [Google Scholar] [CrossRef]
- Nie, Y.; Hu, C.; Kong, C. Enhanced fluoride adsorption using Al (III) modified calcium hydroxyapatite. J. Hazard. Mater. 2012, 233–234, 194–199. [Google Scholar] [CrossRef]
- Cheng, H. Reuse Research Progress on Waste Clay Brick. Procedia Environ. Sci. 2016, 31, 218–226. [Google Scholar] [CrossRef] [Green Version]
- Kagne, S.; Jagtap, S.; Thakare, D.; Devotta, S.; Rayalu, S.S. Bleaching powder: A versatile adsorbent for the removal of fluoride from aqueous solution. Desalination 2009, 243, 22–31. [Google Scholar] [CrossRef]
- Chen, N.; Zhang, Z.; Feng, C.; Li, M.; Zhu, D.; Chen, R.; Sugiura, N. An excellent fluoride sorption behavior of ceramic adsorbent. J. Hazard. Mater. 2009, 183, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Bibi, S.; Farooqi, A.; Hussain, K.; Haider, N. Evaluation of industrial based adsorbents for simultaneous removal of arsenic and fluoride from drinking water. J. Clean. Prod. 2015, 87, 882–896. [Google Scholar] [CrossRef]
- Krysztafkiewicz, A.; Rager, B.; Maik, M. Silica recovery from waste obtained in hydrofluoric acid and aluminum fluoride production from fluosilicic acid. J. Hazard. Mater. 1996, 48, 31–49. [Google Scholar] [CrossRef]
- Alusilica|Alufluor. Available online: https://www.alufluor.com/alusilica/ (accessed on 1 February 2018).
- Şener, Ş.; Sener, E.; Karagüzel, R. Solid waste disposal site selection with GIS and AHP methodology: A case study in Senirkent–Uluborlu (Isparta) Basin, Turkey. Environ. Monit. Assess. 2010, 173, 533–554. [Google Scholar] [CrossRef]
- Kaminskas, R.; Kubiliūtė, R. Artificial pozzolana from silica gel waste–clay–limestone composite. Adv. Cem. Res. 2014, 26, 155–168. [Google Scholar] [CrossRef]
- Li, X.; He, S.; Feng, C.; Zhu, Y.; Pang, Y.; Hou, J.; Luo, K.; Liao, X. Non-Competitive and Competitive Adsorption of Pb2+, Cd2+ and Zn2+ Ions onto SDS in Process of Micellar-Enhanced Ultrafiltration. Sustainability 2018, 10, 92. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Y.; Du, X.; Gao, C.; Yu, Z. Adsorption Behavior of Inorganic and Organic Phosphate by Iron Manganese Plaques on Reed Roots in Wetlands. Sustainability 2018, 10, 4578. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Chen, Q.; Rao, P.; Yan, L.; Shakib, A.; Shen, G. Formulating and Optimizing a Novel Biochar-Based Fertilizer for Simultaneous Slow-Release of Nitrogen and Immobilization of Cadmium. Sustainability 2018, 10, 2740. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Wang, X.-Q.; Wang, D.-F. Immobilization of Heavy Metals in Sewage Sludge during Land Application Process in China: A Review. Sustainability 2017, 9, 2020. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Tae, S.; Kim, R. Theoretical Study on the Production of Environment-Friendly Recycled Cement Using Inorganic Construction Wastes as Secondary Materials in South Korea. Sustainability 2018, 10, 4449. [Google Scholar] [CrossRef] [Green Version]
- Kurosaki, H. Reduction of Fluorine-containing Industrial Waste Using Aluminum-solubility Method. Oki Tech. Rev. 1998, 63, 53–56. [Google Scholar]
- Adhikari, S.; Kayastha, M.S.; Ghimire, D.C.; Aryal, H.R.; Adhikary, S.; Takeuchi, T.; Murakami, K.; Kawashimo, Y.; Uchida, H.; Wakita, K.; et al. Improved Photovoltaic Properties of Heterojunction Carbon Based Solar Cell. J. Surf. Eng. Mater. Adv. Technol. 2013, 3, 178–183. [Google Scholar] [CrossRef] [Green Version]
- Seshan, H. Handbook of Thin—Film Deposition Processes and Techniques, 2nd ed.; Noyes Publications: Park Ridge, NJ, USA, 2002. [Google Scholar]
- Zueva, S.B.; BIrloaga, I.; Ferella, F.; Baturina, E.V.; Corradini, V.; Veglio, F. Mitigation of Fluorine-Containing Waste Resulting from Chemical Vapour Deposition Used In Manufacturing Of Silicon Solar Cells. Processes 2021, 9, 1745. [Google Scholar] [CrossRef]
- Aldaco, R.; Garea, A.; Irabien, A. Calcium fluoride recovery from fluoride wastewater in a fluidized bed reactor. Water Res. 2007, 41, 810–818. [Google Scholar] [CrossRef]
- Shin, C.-H.; Kim, J.-Y.; Kim, J.-Y.; Kim, H.-S.; Lee, H.-S.; Mohapatra, D.; Ahn, J.-W.; Ahn, J.-G.; Bae, W. A solvent extraction approach to recover acetic acid from mixed waste acids produced during semiconductor wafer process. J. Hazard. Mater. 2009, 162, 1278–1284. [Google Scholar] [CrossRef]
- Lee, T.-C.; Liu, F.-J. Recovery of hazardous semiconductor-industry sludge as a useful resource. J. Hazard. Mater. 2009, 165, 359–365. [Google Scholar] [CrossRef]
- Lee, T.-C.; Lin, K.-L.; Su, X.-W.; Lin, K.-K. Recycling CMP sludge as a resource in concrete. Constr. Build. Mater. 2012, 30, 243–251. [Google Scholar] [CrossRef]
- Da, Y.; He, T.; Shi, C.; Wang, M.; Feng, Y. Potential of preparing cement clinker by adding the fluorine-containing sludge into raw meal. J. Hazard. Mater. 2021, 403, 123692. [Google Scholar] [CrossRef]
- Olejarczyk, M.; Urbaniak, W.; Rykowska, I. Wapno posodowe jako składnik sorbentów jonów fluorkowych. In Proceedings of the II Ogólnopolska Przyrodnicza Konferencja Naukowa “Mater naturae”, Online, 11 December 2020; pp. 41–42, ISBN 978-83-66261-98-3. [Google Scholar]
- Wajima, T.; Rakovan, J.F. Removal of fluoride ions using calcined paper sludge. J. Therm. Anal. 2013, 113, 1027–1035. [Google Scholar] [CrossRef]
- Frías, M.; García, R.; Vigil, R.; Ferreiro, S. Calcination of art paper sludge waste for the use as a supplementary cementing material. Appl. Clay Sci. 2008, 42, 189–193. [Google Scholar] [CrossRef]
- Henry, C.L. Nitrogen Dynamics of Pulp and Paper Sludge Amendment to Forest Soils. Water Sci. Technol. 1991, 24, 417–425. [Google Scholar] [CrossRef]
- Tripepi, R.R.; Zhang, X.; Campbell, A.G. Use of Raw and Composted Paper Sludge as a Soil Additive or Mulch for Cottonwood Plants. Compos. Sci. Util. 1996, 4, 26–36. [Google Scholar] [CrossRef]
- Dell’Abate, M.T.; Benedetti, A.; Sequi, P. Thermal Methods of Organic Matter Maturation Monitoring During a Composting Process. J. Therm. Anal. 2000, 61, 389–396. [Google Scholar] [CrossRef]
- Barriga, S.; Méndez, A.; Cámara, J.; Guerrero, F.; Gascó, G. Agricultural valorisation of de-inking paper sludge as organic amendment in different soils: Thermal study. J. Therm. Anal. Calorim. 2010, 99, 981–986. [Google Scholar] [CrossRef]
- Méndez, A.; Barriga, S.; Guerrero, F.; Gascó, G. Thermal analysis of growing media obtained from mixtures of paper mill waste materials and sewage sludge. J. Therm. Anal. 2010, 104, 213–221. [Google Scholar] [CrossRef]
- Rodríguez, O.; Frías, M.; de Rojas, M.I.S. Influence of the calcined paper sludge on the development of hydration heat in blended cement mortars. J. Therm. Anal. 2008, 92, 865–871. [Google Scholar] [CrossRef]
- Melo, C.R.; Angioletto, E.; Riella, H.G.; Peterson, M.; Rocha, M.R.; Melo, A.R.; Silva, L.; Strugale, S. Production of metakaolin from industrial cellulose waste. J. Therm. Anal. 2012, 109, 1341–1345. [Google Scholar] [CrossRef]
- Król, D.; Poskrobko, S. Waste and fuels from waste Part, I. Analysis of thermal decomposition. J. Therm. Anal. Calorim. 2012, 109, 619–628. [Google Scholar] [CrossRef]
- Taş, S.; Yürüm, Y. Co-firing of biomass with coals: Part 2. Thermogravimetric kinetic analysis of co-combustion of fir (Abies bornmulleriana) wood with Beypazari lignite. J. Therm. Anal. Calorim. 2012, 107, 293–298. [Google Scholar] [CrossRef]
- Méndez, A.; Barriga, S.; Saa, A.; Gascó, G. Removal of malachite green by adsorbents from paper industry waste materials: Thermal analysis. J. Therm. Anal. Calorim. 2010, 99, 993–998. [Google Scholar] [CrossRef]
- Olejarczyk, M.; Urbaniak, W.; Rykowska, I. Reclamation materials based on post-soda lime. In Practical Aspects of Remediation, Recultivation and Revitalization; Kołwzan, B., Bukowski, Z., Eds.; Publishing House UKW: Bydgoszcz, Poland, 2022. [Google Scholar]
- Waciński, W.; Olejarczyk, M.; Urbaniak, W.; Rykowska, I. Sorbent, especially for removing aqueous solutions of ions in the form of sparingly soluble salts and how it is maintained. Pol. Pat. Appl. 2022, 440956. [Google Scholar]
- Waciński, W.; Olejarczyk, M.; Urbaniak, W.; Rykowska, I. Method of removing fluoride ions from contaminated waters, especially sewage. Pol. Pat. Appl. 2022, 440957. [Google Scholar]
| Authors | Title | Aim |
|---|---|---|
| Habuda-Stanić M. et al., 2014 [52] | Review on Adsorption of Fluoride from Aqueous Solution | A list of various adsorbents (oxides and hydroxides, biosorbents, geomaterials, carbonaceous materials, and industrial by-products) and their modifications is discussed. This survey showed that various adsorbents, especially binary and trimetal oxides and hydroxides, have good potential for fluoride removal from aquatic environments. |
| Waghmare S.S. et al., 2015 [53] | Fluoride removal by industrial, agricultural and biomass wastes as adsorbents: a review | Reviews the fluoride uptake capacities of industrial by-products, agricultural wastes, and biomass materials from plants, grass, etc., and their modified forms as adsorbents in batch and column performance. |
| Tomar V. et al., 2013 [54] | A critical study on efficiency of different materials for fluoride removal from aqueous media | An extensive list of adsorbents for fluoride removal is compiled. In particular, nanomaterial-based adsorbents might be promising adsorbents for environmental and purification purposes. |
| Kumar P.S., 2019 [39] | Treatment of fluoride-contaminated water: a review | Reviews the origin of fluoride, the analysis of fluoride derivatives, and the technologies to remove fluoride from water, using different adsorbent types. |
| Nagendra Rao C.R. 2003 [58] | Fluoride and environment—a review | Current information on fluoride presence in the environment and its effects on human health, as well as basic methods of defluoridation. |
| Schlesinger W.H. et al., 2020 [59] | Global Biogeochemical Cycle of Fluorine | Synthesis of what is currently known about the natural and anthropogenic fluxes of fluorine. |
| He J. et al., 2020 [60] | Review of fluoride removal from water environment by adsorption | The recent developments in fluoride removal from the water environment by adsorption methods. Based on the review, four technical strategies of adsorption method, including nano-surface effect, structural memory effect, anti-competitive adsorption, and ionic sieve effect, were proposed. |
| Bhatnagar A. et al., 2011 [61] | Fluoride removal from water by adsorption—a review | An extensive list of various adsorbents from literature has been compiled, and their adsorption capacities under various conditions (pH, initial fluoride concentration, temperature, contact time, adsorbent surface charge, etc.) for fluoride removal are presented. |
| Bodzek M. et al., 2018 [39] | Fluorine in the Water Environment-Hazards and Removal Methods, Engineering and Protection of Environment | Detailed information on recent researchers’ efforts in the field of fluoride removal during potable water production. The contaminant elimination methods have been broadly divided in three sections, i.e., coagulation/precipitation, adsorption, and membrane techniques. Both precipitation with the use of calcium salts or coagulation with aluminium sulphate and ferric salts followed by sedimentation are used for fluorine removal. In electrocoagulation, a coagulant is generated in situ by means of oxidation of anode usually made of aluminium or iron. |
| Wang L. et al. 2019 [62] | A Review on Comprehensive Utilization of Red Mud and Prospect Analysis | Comprehensive utilization methods for reducing red mud (RM) environmental pollution and divides the comprehensive utilization of RM into three aspects: the effective extraction of valuable components, resource transformation, and environmental application. |
| Adsorbent | Concentration Range (mg/L) | pH Range | Contact Time (min) | Model Used to Calculate Adsorption Capacity | Maximum Adsorption Capacity (mg/g) | Ref. |
|---|---|---|---|---|---|---|
| Waste mud | - | 2–8 | 0–480 | Langmuir and Freundlich | 27.2 | [220] |
| Red Mud | 5–150 | 4.7 | 15–540 | Freundlich | 0.851 | [215] |
| 5 | 4.7 | 360 | Redlich–Peterson and Freundlich | 0.644 | [215] | |
| 100–1000 | 5.5 | 120 | Langmuir and Freundlich | 3.12 and 6.29 | [165] | |
| Modified red mud with AlCl3 (MRMA), heat activated red mud (MRMAH) | - | 7–8 | Langmuir | MRMA-68.07 MRMAH-91.28 | [216] | |
| Zirconium hydroxide modified red mud porous material Zr-modified RMPM | - | 3 | 60 | pseudo-second-order rate kinetics and pore diffusion models | 0.6 | [217] |
| Red mud | - | 5.5 | 120 | - | [218] | |
| Alum sludge | - | 5.5–6.5 | - | - | 5.35 | [221] |
| Sludge produced during the manufacturing of aluminium sulphate (alum) from kaolin | 10 | 3–8 | - | - | 332.5 | [222] |
| Spent Bleach Earth (SBE) | - | 3.5 | - | - | 7.75 | [223] |
| Fly ash A and S | - | - | - | Freundlich | 1.22 (A) 1.01 (S) | [226] |
| Calcium hydroxide treated fly ash (CFA) | 10 | 7 | 120 | 10.86 | [227] | |
| Bottom ash | - | 6 | 105 | BDST | 16.26 | [228] |
| Magnesia-loaded fly ash cenospheres (MLC) | 10 | - | - | Thomas | 5.884 | [230] |
| aluminium-treated bagasse fly ash (ABF) | 1–10 | 6 | 300 | - | 10 | [232] |
| Maize husk fly ash | 2.0 g/50 mL | 2 | 120 | Redlich-Peterson | [233] | |
| Activated tea ash (AcTAP) | 6 | 180 | Langmuir | 8.55 | [231] | |
| Waste carbon slurry obtained from fuel oil | 15 | 7.58 | 120 | Langmuir | 4.861 | [234] |
| Coal mining waste | - | 3.5 | - | Langmuir | 15.67 | [235] |
| Shale (coal mine waste) in the form of native shale (NS) and heat activated shale (HAS) at 350 °C, 450 °C and 550 °C | 10-HAS550 | 3 | 24 h | Langmuir | 0.358 | [236] |
| Blast furnace slag generated from steel industry | 10 mg/l | 6–10 | 35 | Langmuir | 8.07 | [237] |
| Spent catalyst (a by-product of petrochemical industry) | - | 4 | - | 28 | [238] | |
| Iron coated spent catalyst | - | 5.5–6.0 | - | Langmuir | 7.2–20.7 | [239] |
| Thermally activated titanium rich bauxite (TRB) | 10 | 5.5–6.5 | - | Langmuir | 3.8 | [240] |
| High alumina (81.5%) content bauxite | - | - | - | Freundlich | 3.125 | [243] |
| Bauxite | 10 | 6 | 90 | Freundlicha, Langmuira Tempkina, | 3 | [241] |
| Hydrated cement (HC), brick powder (BP) marble powder (MP). | 30 | 7 8 7 | 60 | Langmuir | 1.72 0.84 0.18 | [254] |
| Bleaching powder | - | 6–10 | - | - | - | [244] |
| Rice husk ash, which was coated with aluminium hydroxide | 10–60 | 7 | 60 | 15.08 | [183] | |
| Activated rice husk ash (ARHA) | 100 | Langmuir | 0.402 | [231] | ||
| Ceramic adsorbents consisting of Kanuma mud, with zeolite, starch, and FeSO4·7H2O | 20–100 | 4–11 | 0–48 h | pseudo-second-order | 2.16 | [253] |
| Porous granular ceramic adsorbents containing dispersed aluminium and iron oxides | 10 | 4–9 | 48 h | Langmuir and Freundlich | 1.79 | [249] |
| Iron-impregnated granular ceramics | 7, 4 | Langmuir and Freundlich | - | [167] | ||
| Recycled phosphogypsum in a form of HAP nanoparticles | 7 | Langmuir-Freundlich | 19.742–25 °C 26.108–35 °C 36.914–45 °C 40.818–55 °C | [186] | ||
| HAP-calcium phosphate based bioceramic | - | - | - | Langmuir and pseudo-second-order | 32.57 | [250] |
| HAP Apatitic tricalcium phosphate. | up to 20 up to 60 | 4.16 4 | Langmuir Langmuir | 13.88–25 °C 14.70–30 °C 15.15–37 °C | [118,119] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olejarczyk, M.; Rykowska, I.; Urbaniak, W. Management of Solid Waste Containing Fluoride—A Review. Materials 2022, 15, 3461. https://doi.org/10.3390/ma15103461
Olejarczyk M, Rykowska I, Urbaniak W. Management of Solid Waste Containing Fluoride—A Review. Materials. 2022; 15(10):3461. https://doi.org/10.3390/ma15103461
Chicago/Turabian StyleOlejarczyk, Małgorzata, Iwona Rykowska, and Włodzimierz Urbaniak. 2022. "Management of Solid Waste Containing Fluoride—A Review" Materials 15, no. 10: 3461. https://doi.org/10.3390/ma15103461
APA StyleOlejarczyk, M., Rykowska, I., & Urbaniak, W. (2022). Management of Solid Waste Containing Fluoride—A Review. Materials, 15(10), 3461. https://doi.org/10.3390/ma15103461






