Machine Learning Prediction of Quantum Yields and Wavelengths of Aggregation-Induced Emission Molecules
Abstract
:1. Introduction
2. Materials and Methods
3. Result and Discussions
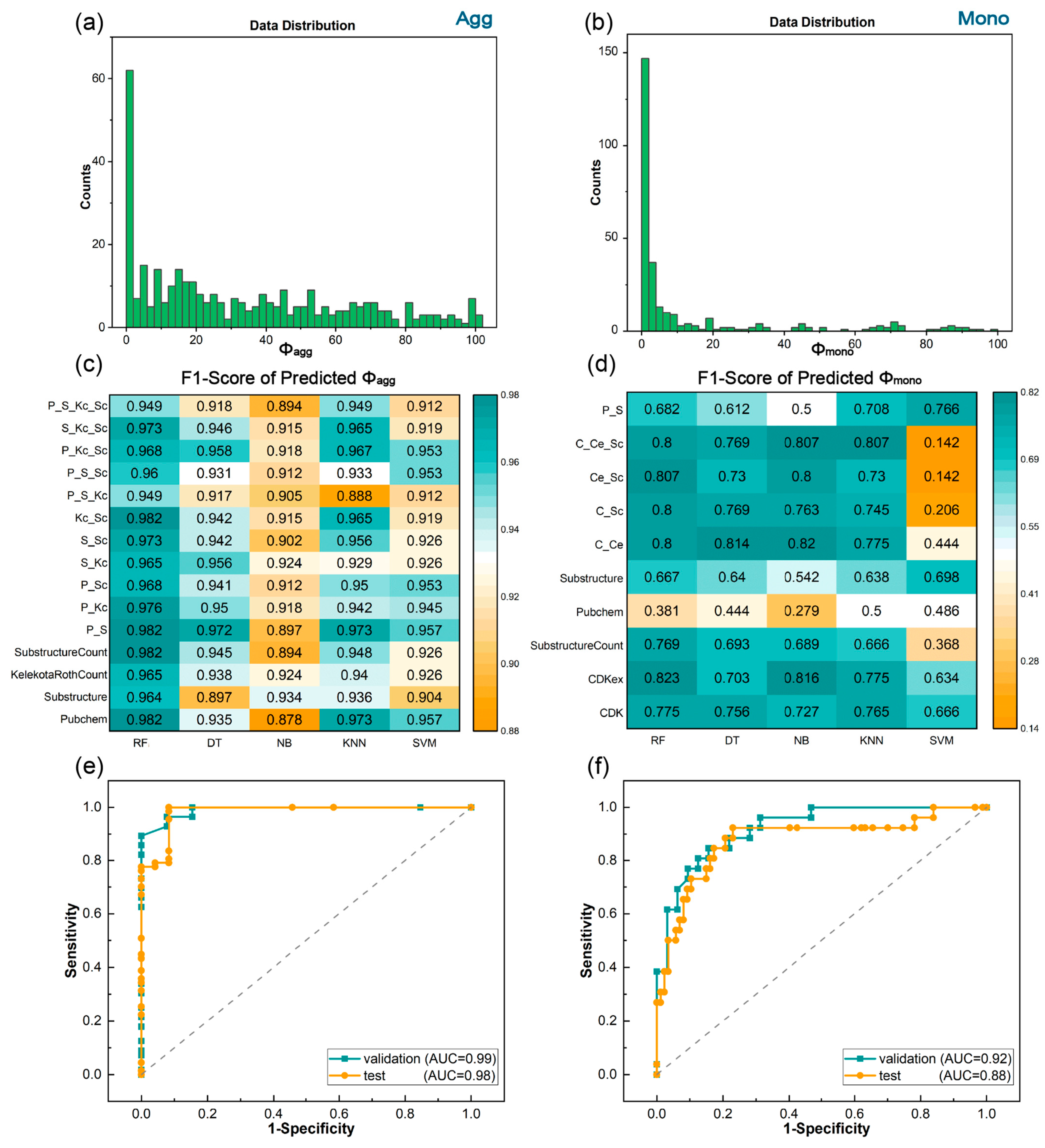
3.1. Prediction of Quantum Yields in the Aggregated and Monomeric States
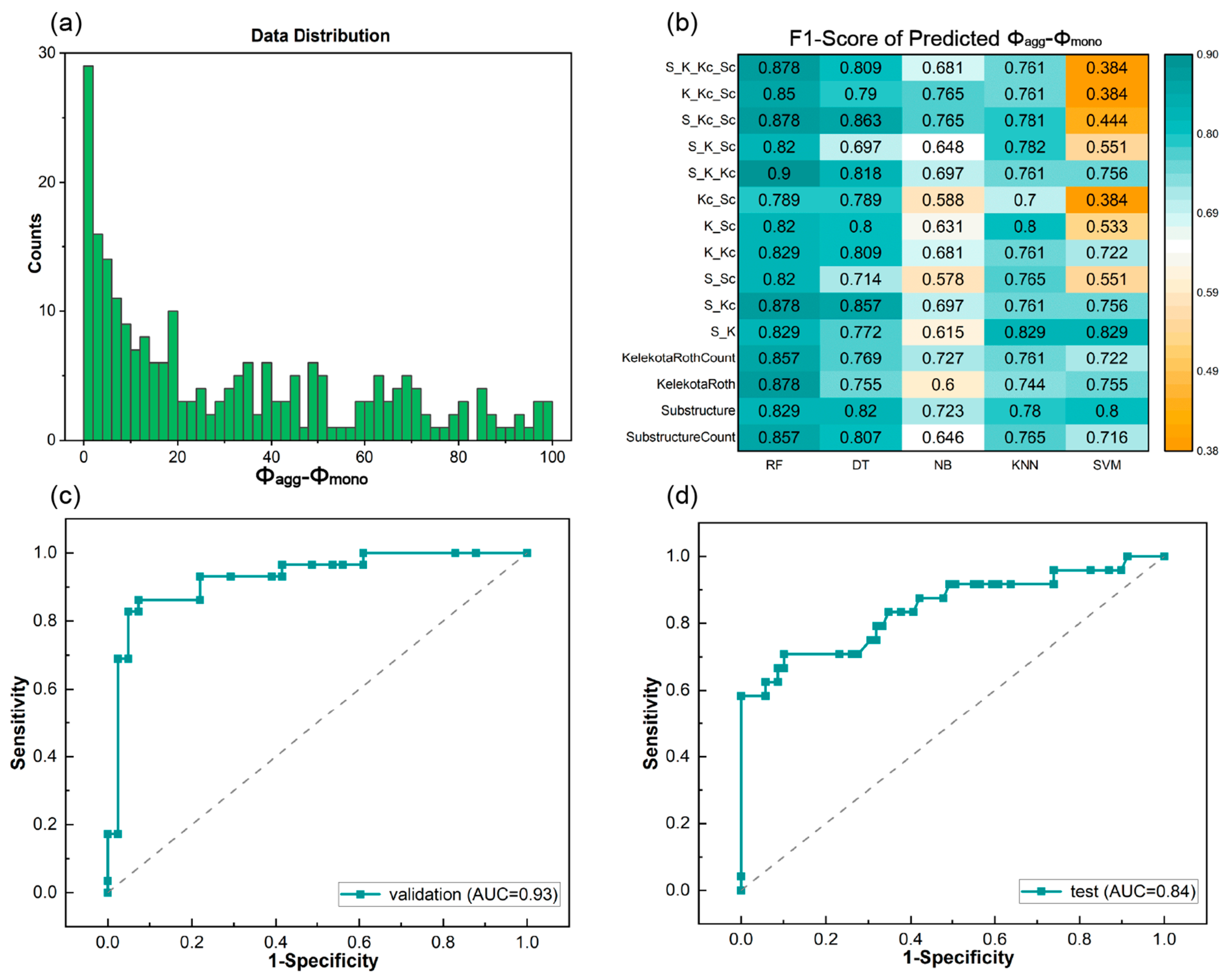
3.2. Prediction of the Quantum Yield Difference between the Aggregated and Monomeric States
3.3. Prediction of Emission Wavelengths and Absorption Wavelengths
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharma, K.; Abbas, B. A study of highly efficient organic light emitting transistors that outperforms organic light emitting diodes. Opt. Quantum Electron. 2023, 55, 338. [Google Scholar] [CrossRef]
- Kim, J.J.; Lee, J.; Yang, S.P.; Kim, H.G.; Kweon, H.S.; Yoo, S.; Jeong, K.H. Biologically inspired organic light-emitting diodes. Nano Lett. 2016, 16, 2994–3000. [Google Scholar] [CrossRef] [PubMed]
- Adachi, C.; Sandanayaka, A.S.D. The leap from organic light-emitting diodes to organic semiconductor laser diodes. CCS Chem. 2020, 2, 1203–1216. [Google Scholar] [CrossRef]
- Mizzoni, S.; Ruggieri, S.; Sickinger, A.; Riobé, F.; Guy, L.; Roux, M.; Micouin, G.; Banyasz, A.; Maury, O.; Baguenard, B.; et al. Circularly polarized activity from two photon excitable europium and samarium chiral bioprobes. J. Mater. Chem. C 2023, 11, 4188–4202. [Google Scholar] [CrossRef]
- Tateo, S.; Shinchi, H.; Matsumoto, H.; Nagata, N.; Hashimoto, M.; Wakao, M.; Suda, Y. Optimized immobilization of single chain variable fragment antibody onto non-toxic fluorescent nanoparticles for efficient preparation of a bioprobe. Colloids Surf. B Biointerfaces 2023, 224, 113192. [Google Scholar] [CrossRef] [PubMed]
- Chua, M.H.; Chin, K.L.O.; Loh, X.J.; Zhu, Q.; Xu, J. Aggregation-induced emission-active nanostructures: Beyond biomedical applications. ACS Nano 2023, 17, 1845–1878. [Google Scholar] [CrossRef]
- Silva, L.R.G.; Carvalho, J.H.S.; Stefano, J.S.; Oliveira, G.G.; Prakash, J.; Janegitz, B.C. Electrochemical sensors and biosensors based on nanodiamonds: A review. Mater. Today Commun. 2023, 35, 106142. [Google Scholar] [CrossRef]
- Nepfumbada, C.; Mthombeni, N.H.; Sigwadi, R.; Ajayi, R.F.; Feleni, U.; Mamba, B.B. Functionalities of electrochemical fluoroquinolone sensors and biosensors. Environ. Sci. Pollut. Res. Int. 2024, 31, 3394–3412. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Fang, M.; Li, Z. Organic luminescent materials: The concentration on aggregates from aggregation-induced emission. Aggregate 2020, 1, 6–18. [Google Scholar] [CrossRef]
- Mei, J.; Leung, N.L.; Kwok, R.T.; Lam, J.W.; Tang, B.Z. Aggregation-induced emission: Together we shine, united we soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef]
- Fang, M.; Yang, J.; Li, Z. Light emission of organic luminogens: Generation, mechanism and application. Prog. Mater. Sci. 2022, 125, 100914. [Google Scholar] [CrossRef]
- Yuan, W.Z.; Lu, P.; Chen, S.; Lam, J.W.; Wang, Z.; Liu, Y.; Kwok, H.S.; Ma, Y.; Tang, B.Z. Changing the behavior of chromophores from aggregation-caused quenching to aggregation-induced emission: Development of highly efficient light emitters in the solid state. Adv. Mater. 2010, 22, 2159–2163. [Google Scholar] [CrossRef] [PubMed]
- Kakumachi, S.; Ba Nguyen, T.; Nakanotani, H.; Adachi, C. Abrupt exciton quenching in blue fluorescent organic light-emitting diodes around turn-on voltage region. Chem. Eng. J. 2023, 471, 144516. [Google Scholar] [CrossRef]
- Ghazy, A.; Lastusaari, M.; Karppinen, M. Excitation wavelength engineering through organic linker choice in luminescent atomic/molecular layer deposited lanthanide-organic thin films. Chem. Mater. 2023, 35, 5988–5995. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Li, Z. The strong light-emission materials in the aggregated state: What happens from a single molecule to the collective group. Adv. Sci. 2017, 4, 1600484. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Lam, J.W.; Tang, B.Z. Aggregation-induced emission: Phenomenon, mechanism and applications. Chem. Commun. 2009, 29, 4332–4353. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, J.; Jiang, C.; Yao, C.; Xi, X. Effective design strategy for aggregation-induced emission and thermally activated delayed fluorescence emitters achieving 18% external quantum efficiency pure-blue oleds with extremely low roll-off. ACS Appl. Mater. Interfaces 2021, 13, 57713–57724. [Google Scholar] [CrossRef]
- Ra, H.S.; Lee, S.H.; Jeong, S.J.; Cho, S.; Lee, J.S. Advances in heterostructures for optoelectronic devices: Materials, properties, conduction mechanisms, device applications. Small Methods 2024, 8, e2300245. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.G.; Kim, G.-Y.; Han, J.-I.; Kim, S.; Kim, J.P. Enhancement of lipid productivity of chlorella sp. Using light-converting red fluorescent films based on aggregation-induced emission. ACS Sustain. Chem. Eng. 2020, 8, 15888–15897. [Google Scholar] [CrossRef]
- Li, X.; Yang, H.; Zheng, P.; Lin, D.; Zhang, Z.; Kang, M.; Wang, D.; Tang, B.Z. Aggregation-induced emission materials: A platform for diverse energy transformation and applications. J. Mater. Chem. A 2023, 11, 4850–4875. [Google Scholar] [CrossRef]
- Kim, E.; Koh, M.; Lim, B.J.; Park, S.B. Emission wavelength prediction of a full-color-tunable fluorescent core skeleton, 9-aryl-1,2-dihydropyrrolo[3,4-b]indolizin-3-one. J. Am. Chem. Soc. 2011, 133, 6642–6649. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Qian, J.; Liu, Y.; Zhu, N.; Xu, B.; Ho, C.-L.; Tian, W.; Wong, W.-Y. Imidazole-containing cyanostilbene-based molecules with aggregation-induced emission characteristics: Photophysical and electroluminescent properties. New J. Chem. 2019, 43, 1844–1850. [Google Scholar] [CrossRef]
- Finencio, B.M.; Santos, F.A.; Parreira, R.L.T.; Orenha, R.P.; Lima, S.M.; Andrade, L.H.C.; Ventura, M.; da Silva de Laurentiz, R. Luminescent properties of beta-(hydroxyaryl)-butenolides and fluorescence quenching in water. J. Fluoresc. 2024. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Zhang, H.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission: New vistas at the aggregate level. Angew. Chem. Int. Ed. 2020, 59, 9888–9907. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.; Drown, B.S.; Hergenrother, P.J. Access to a structurally complex compound collection via ring distortion of the alkaloid sinomenine. Org. Lett. 2016, 18, 4852–4855. [Google Scholar] [CrossRef] [PubMed]
- Sanz-Velasco, A.; Amargos-Reyes, O.; Kahari, A.; Lipinski, S.; Cavinato, L.M.; Costa, R.D.; Kostiainen, M.A.; Anaya-Plaza, E. Controlling aggregation-induced emission by supramolecular interactions and colloidal stability in ionic emitters for light-emitting electrochemical cells. Chem. Sci. 2024, 15, 2755–2762. [Google Scholar] [CrossRef]
- Hennefarth, M.R.; King, D.S.; Gagliardi, L. Linearized pair-density functional theory for vertical excitation energies. J. Chem. Theory Comput. 2023, 19, 7983–7988. [Google Scholar] [CrossRef] [PubMed]
- Baum, Z.J.; Yu, X.; Ayala, P.Y.; Zhao, Y.; Watkins, S.P.; Zhou, Q. Artificial intelligence in chemistry: Current trends and future directions. J. Chem. Inf. Model. 2021, 61, 3197–3212. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Sunoj, R.B. Molecular machine learning for chemical catalysis: Prospects and challenges. Acc. Chem. Res. 2023, 56, 402–412. [Google Scholar] [CrossRef]
- Hagg, A.; Kirschner, K.N. Open-source machine learning in computational chemistry. J. Chem. Inf. Model. 2023, 63, 4505–4532. [Google Scholar] [CrossRef]
- De Angelis, F. The impact of machine learning in energy materials research: The case of halide perovskites. ACS Energy Lett. 2023, 8, 1270–1272. [Google Scholar] [CrossRef]
- Noto, N.; Yada, A.; Yanai, T.; Saito, S. Machine-learning classification for the prediction of catalytic activity of organic photosensitizers in the nickel(ii)-salt-induced synthesis of phenols. Angew. Chem. Int. Ed. 2023, 62, e202219107. [Google Scholar] [CrossRef] [PubMed]
- Janjua, M.R.S.A.; Irfan, A.; Hussien, M.; Ali, M.; Saqib, M.; Sulaman, M. Machine-learning analysis of small-molecule donors for fullerene based organic solar cells. Energy Technol. 2022, 10, 2200019. [Google Scholar] [CrossRef]
- Pfluger, P.M.; Glorius, F. Molecular machine learning: The future of synthetic chemistry? Angew. Chem. Int. Ed. 2020, 59, 18860–18865. [Google Scholar] [CrossRef]
- Wigh, D.S.; Goodman, J.M.; Lapkin, A.A. A review of molecular representation in the age of machine learning. WIREs Comput. Mol. Sci. 2022, 12, e1603. [Google Scholar] [CrossRef]
- Tkatchenko, A. Machine learning for chemical discovery. Nat. Commun. 2020, 11, 4125. [Google Scholar] [CrossRef] [PubMed]
- Ju, C.W.; Bai, H.; Li, B.; Liu, R. Machine learning enables highly accurate predictions of photophysical properties of organic fluorescent materials: Emission wavelengths and quantum yields. J. Chem. Inf. Model. 2021, 61, 1053–1065. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Liu, Y.; Yan, J.; Yan, Z.Y.; Wu, Y.; Ru, Z.; Liao, J.Y.; Miao, X.; Qian, L. Prediction of maximum absorption wavelength using deep neural networks. J. Chem. Inf. Model. 2022, 62, 1368–1375. [Google Scholar] [CrossRef] [PubMed]
- Senanayake, R.D.; Yao, X.; Froehlich, C.E.; Cahill, M.S.; Sheldon, T.R.; McIntire, M.; Haynes, C.L.; Hernandez, R. Machine learning-assisted carbon dot synthesis: Prediction of emission color and wavelength. J. Chem. Inf. Model. 2022, 62, 5918–5928. [Google Scholar] [CrossRef]
- Mahato, K.D.; Kumar, U. Optimized machine learning techniques enable prediction of organic dyes photophysical properties: Absorption wavelengths, emission wavelengths, and quantum yields. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2024, 308, 123768. [Google Scholar] [CrossRef]
- Zhou, J.; Huang, B.; Yan, Z.; Bunzli, J.G. Emerging role of machine learning in light-matter interaction. Light Sci. Appl. 2019, 8, 84. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Wang, K.; Lian, Z.; Yang, X.; Huang, W.; Qin, A.; Wang, Q.; Tian, J.; Tang, B.; Zhang, S. Prediction and understanding of aie effect by quantum mechanics-aided machine-learning algorithm. Chem. Commun. 2018, 54, 7955–7958. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Liu, X.; Cai, P.; Li, J.; Wang, X.; Liu, B. Machine-learning-assisted accurate prediction of molecular optical properties upon aggregation. Adv. Sci. 2022, 9, e2101074. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fan, M.; Xu, Z.; Jiang, Y.; Ding, H.; Li, Z.; Shu, K.; Zhao, M.; Feng, G.; Yong, K.T.; et al. Machine-learning screening of luminogens with aggregation-induced emission characteristics for fluorescence imaging. J. Nanobiotechnol. 2023, 21, 107. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Ramsundar, B.; Feinberg, E.N.; Gomes, J.; Geniesse, C.; Pappu, A.S.; Leswing, K.; Pande, V. Moleculenet: A benchmark for molecular machine learning. Chem. Sci. 2018, 9, 513–530. [Google Scholar] [CrossRef] [PubMed]
- Grisoni, F.; Ballabio, D.; Todeschini, R.; Consonni, V. Molecular descriptors for structure-activity applications: A hands-on approach. Methods Mol. Biol. 2018, 1800, 3–53. [Google Scholar] [PubMed]
- Riniker, S.; Landrum, G.A. Similarity maps—A visualization strategy for molecular fingerprints and machine-learning methods. J. Cheminform. 2013, 5, 43. [Google Scholar] [CrossRef] [PubMed]
- Capecchi, A.; Probst, D.; Reymond, J.L. One molecular fingerprint to rule them all: Drugs, biomolecules, and the metabolome. J. Cheminform. 2020, 12, 43. [Google Scholar] [CrossRef]
- Yang, J.; Cai, Y.; Zhao, K.; Xie, H.; Chen, X. Concepts and applications of chemical fingerprint for hit and lead screening. Drug Discov. Today 2022, 27, 103356. [Google Scholar] [CrossRef]
- Motiei, L.; Margulies, D. Molecules that generate fingerprints: A new class of fluorescent sensors for chemical biology, medical diagnosis, and cryptography. Acc. Chem. Res. 2023, 56, 1803–1814. [Google Scholar] [CrossRef]
- Dong, J.; Cao, D.S.; Miao, H.Y.; Liu, S.; Deng, B.C.; Yun, Y.H.; Wang, N.N.; Lu, A.P.; Zeng, W.B.; Chen, A.F. Chemdes: An integrated web-based platform for molecular descriptor and fingerprint computation. J. Cheminform. 2015, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Yap, C.W. Padel-descriptor: An open source software to calculate molecular descriptors and fingerprints. J. Comput. Chem. 2011, 32, 1466–1474. [Google Scholar] [CrossRef]
- Giudici, P.; Gramegna, A.; Raffinetti, E. Machine learning classification model comparison. Socio-Econ. Plan. Sci. 2023, 87, 101560. [Google Scholar] [CrossRef]
- Pruneski, J.A.; Williams, R.J., 3rd; Nwachukwu, B.U.; Ramkumar, P.N.; Kiapour, A.M.; Martin, R.K.; Karlsson, J.; Pareek, A. The development and deployment of machine learning models. Knee Surg. Sports Traumatol. Arthrosc. 2022, 30, 3917–3923. [Google Scholar] [CrossRef]
- Hatanaka, M.; Kato, H.; Sakai, M.; Kariya, K.; Nakatani, S.; Yoshimura, T.; Inagaki, T. Insights into the luminescence quantum yields of cyclometalated iridium(iii) complexes: A density functional theory and machine learning approach. J. Phys. Chem. A 2023, 127, 7630–7637. [Google Scholar] [CrossRef]
- Rish, A.J.; Henson, S.R.; Velez-Silva, N.L.; Nahid Hasan, M.; Drennen, J.K.; Anderson, C.A. Application of a wavelength angle mapper for variable selection in iterative optimization technology predictions of drug content in pharmaceutical powder mixtures. Int. J. Pharm. 2023, 643, 123261. [Google Scholar] [CrossRef]
- Smajic, A.; Grandits, M.; Ecker, G.F. Using jupyter notebooks for re-training machine learning models. J. Cheminform. 2022, 14, 54. [Google Scholar] [CrossRef]
- Frisch, M.J.T.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian16 Revision c.01; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Janai, M.A.B.; Woon, K.L.; Chan, C.S. Design of efficient blue phosphorescent bottom emitting light emitting diodes by machine learning approach. Org. Electron. 2018, 63, 257–266. [Google Scholar] [CrossRef]
- Mantero, A.; Ishwaran, H. Unsupervised random forests. Stat. Anal. Data Min. 2021, 14, 144–167. [Google Scholar] [CrossRef]
- Walker, A.M.; Cliff, A.; Romero, J.; Shah, M.B.; Jones, P.; Felipe Machado Gazolla, J.G.; Jacobson, D.A.; Kainer, D. Evaluating the performance of random forest and iterative random forest based methods when applied to gene expression data. Comput. Struct. Biotechnol. J. 2022, 20, 3372–3386. [Google Scholar] [CrossRef]
- Biggs, M.; Hariss, R.; Perakis, G. Constrained optimization of objective functions determined from random forests. Prod. Oper. Manag. 2023, 32, 397–415. [Google Scholar] [CrossRef]
- Sandfort, F.; Strieth-Kalthoff, F.; Kühnemund, M.; Beecks, C.; Glorius, F. A structure-based platform for predicting chemical reactivity. Chem 2020, 6, 1379–1390. [Google Scholar] [CrossRef]
- Kang, B.; Seok, C.; Lee, J. Prediction of molecular electronic transitions using random forests. J. Chem. Inf. Model. 2020, 60, 5984–5994. [Google Scholar] [CrossRef] [PubMed]
- Torrisi, S.B.; Carbone, M.R.; Rohr, B.A.; Montoya, J.H.; Ha, Y.; Yano, J.; Suram, S.K.; Hung, L. Random forest machine learning models for interpretable X-ray absorption near-edge structure spectrum-property relationships. npj Comput. Mater. 2020, 6, 109. [Google Scholar] [CrossRef]
- Chen, Y.L.; Li, S.W.; Chi, Y.; Cheng, Y.M.; Pu, S.C.; Yeh, Y.S.; Chou, P.T. Switching luminescent properties in osmium-based beta-diketonate complexes. ChemPhysChem 2005, 6, 2012–2017. [Google Scholar] [CrossRef]
- Obara, S.; Itabashi, M.; Okuda, F.; Tamaki, S.; Tanabe, Y.; Ishii, Y.; Nozaki, K.; Haga, M.-A. Highly phosphorescent iridium complexes containing both tridentate bis(benzimidazolyl)-benzene or -pyridine and bidentate phenylpyridine: Synthesis, photophysical properties, and theoretical study of ir-bis(benzimidazolyl)benzene complex. Inorg. Chem. 2006, 45, 8907–8921. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Yang, L.; Dong, X.; Zhou, L.; Wei, Q.; Ju, H. Cysteine modification of glutathione-stabilized au nanoclusters to red-shift and enhance the electrochemiluminescence for sensitive bioanalysis. Anal. Chem. 2022, 94, 2313–2320. [Google Scholar] [CrossRef]
- Lin, Y.D.; Lu, C.W.; Su, H.C. Long-wavelength light-emitting electrochemical cells: Materials and device engineering. Chemistry 2023, 29, e202202985. [Google Scholar] [CrossRef]



| Descriptors | Φagg | Φmono | Φagg-Φmono | λabs | λem_agg | λem_mono | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AUC | ACC | AUC | ACC | AUC | ACC | r | MRE/% | r | MRE/% | r | MRE/% | |
| MACCS | 0.73 | 0.9 | 0.87 | 0.77 | 0.88 | 0.81 | 0.81 | 7.62 | 0.84 | 5.87 | 0.86 | 7.15 |
| Morgan | 0.82 | 0.82 | 0.86 | 0.84 | 0.83 | 0.81 | 0.85 | 7.02 | 0.76 | 6.82 | 0.83 | 7.56 |
| Atomp | 0.74 | 0.86 | 0.71 | 0.87 | 0.82 | 0.79 | 0.70 | 8.38 | 0.77 | 7.57 | 0.70 | 10.0 |
| Pubchem | 0.92 | 0.97 | 0.60 | 0.56 | 0.89 | 0.80 | 0.75 | 8.59 | 0.80 | 7.21 | 0.83 | 9.80 |
| Substructure | 0.90 | 0.94 | 0.81 | 0.72 | 0.94 | 0.85 | 0.73 | 7.34 | 0.82 | 6.72 | 0.83 | 8.25 |
| Estate | 0.88 | 0.91 | 0.81 | 0.81 | 0.86 | 0.78 | 0.69 | 7.58 | 0.79 | 7.15 | 0.84 | 7.81 |
| CDK | 0.82 | 0.93 | 0.91 | 0.87 | 0.84 | 0.83 | 0.82 | 6.55 | 0.81 | 5.96 | 0.82 | 7.96 |
| CDKex | 0.82 | 0.93 | 0.92 | 0.84 | 0.83 | 0.80 | 0.80 | 6.84 | 0.81 | 6.98 | 0.78 | 8.79 |
| SubstructureCount | 0.92 | 0.93 | 0.87 | 0.84 | 0.90 | 0.89 | 0.75 | 9.00 | 0.83 | 6.91 | 0.82 | 8.03 |
| Atompair2DCount | 0.79 | 0.93 | 0.84 | 0.72 | 0.84 | 0.80 | 0.74 | 8.18 | 0.80 | 8.10 | 0.79 | 8.89 |
| CDKgraphonly | 0.82 | 0.91 | 0.85 | 0.71 | 0.82 | 0.73 | 0.72 | 9.50 | 0.80 | 6.76 | 0.73 | 10.2 |
| KlekotaRoth | 0.88 | 0.95 | 0.86 | 0.79 | 0.94 | 0.90 | 0.72 | 8.66 | 0.83 | 7.20 | 0.84 | 8.37 |
| KlekotaRothCount | 0.90 | 0.94 | 0.82 | 0.78 | 0.93 | 0.87 | 0.76 | 7.91 | 0.82 | 7.00 | 0.84 | 7.95 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bi, H.; Jiang, J.; Chen, J.; Kuang, X.; Zhang, J. Machine Learning Prediction of Quantum Yields and Wavelengths of Aggregation-Induced Emission Molecules. Materials 2024, 17, 1664. https://doi.org/10.3390/ma17071664
Bi H, Jiang J, Chen J, Kuang X, Zhang J. Machine Learning Prediction of Quantum Yields and Wavelengths of Aggregation-Induced Emission Molecules. Materials. 2024; 17(7):1664. https://doi.org/10.3390/ma17071664
Chicago/Turabian StyleBi, Hele, Jiale Jiang, Junzhao Chen, Xiaojun Kuang, and Jinxiao Zhang. 2024. "Machine Learning Prediction of Quantum Yields and Wavelengths of Aggregation-Induced Emission Molecules" Materials 17, no. 7: 1664. https://doi.org/10.3390/ma17071664