Abstract
Antioxidants are crucial in counteracting the impact of free radicals, thereby preventing cell damage. Extracts from orange, apricot, and olive tree pruning, classified as by-products according to environmental laws, underwent examination of their antioxidant properties. In the present investigation, data on the antioxidant activity of extracts of these tree by-products were used to calculate the relative antioxidant capacity index (RACI). The latter made it possible to identify Apricot bark maceration extract as having the highest antioxidant index. Further analysis through UHPLC-MS enabled the identification of compounds known for their antioxidant activity, including catechin, epicatechin, and others. Cytotoxicity assays conducted on human hepatocellular carcinoma cell lines (HepG2) confirmed the safety of the chosen extract for cosmetic applications. The extract was then used to formulate a cosmetic cream, which subsequently underwent quality control and proved to be stable, of good consistency, and free of pathogens. The results show that developing a sustainable cosmetic cream prototype is a promising step toward realizing an environmentally friendly product in the circular economy context.
1. Introduction
With growing global attention focused on environmental concerns and diminishing resources, the food industry is placing greater emphasis on implementing environmentally friendly practices and reducing waste. This has prompted a significant shift in focus, whereby agro-food by-products are now seen as precious resources with unexploited potential, challenging their previous classification as mere processing waste. The European Union (EU) has implemented initiatives designed to reduce foodstuff waste by adopting a circular economy model. This strategy significantly emphasizes minimizing, recovering, reusing, and recycling materials to enhance their value, thereby extending their economic usability. In this context, several investigations have evidenced the potential reuse of agricultural by-products in food, pharmaceutical, and cosmetic fields [1]. Specifically, a considerable source of by-products comes from cultivating fruit trees. Within the EU, a collective expanse of 11,301,345 hectares is designated for cultivating fruit trees, predominantly pear and apple trees, grapevines, olive groves, stone fruit, walnut, and citrus trees. The pruning of these trees results in a substantial volume of wood waste. An estimated 25 million tons of wood waste are, indeed, generated in the EU each year by tree pruning activities. Typically, this woody waste is disposed of by burning or crushing it in the field, with the consequent loss of potential immediate economic value.
With the Consolidated Environmental Law, No. 156/2016, prunings and mowings are reclassified in Europe as by-products and not as waste, eliminating their categorization as unusable materials. In this way, extractives derived from pruning have been classified under the botanicals category and are regulated by Annex 1 of the Italian Ministerial Decree of 10 August 2018. Nevertheless, wood derived from tree pruning presents a broad range of possibilities for valorization since it is rich in high-added-value molecules like phenols, thus highlighting the need to breathe new life into this by-product [2,3]. Phenolic compounds have garnered interest from the cosmetic and food industries as they exhibit interesting biological activities, including anti-inflammatory and antioxidant effects, prompting the production of value-added products. Hence, to mitigate issues linked to improper by-product disposal, phenolic-rich extracts from various agro-industrial residues have been utilized to develop value-added products within the food and cosmetic sectors [4]. For instance, recent investigations on sweet orange (Citrus sinensis L.) pruning/wood biomass and bitter orange (Citrus aurantium L.) tree wood biomass showed the presence of different alkaloids, essential oils, and polyphenols with valuable biological activity like antioxidant or antibacterial properties [2]. Similarly, mandarin (Citrus reticulata Blanco) wood waste extracts were shown to be rich in polyphenols, terpenes, and alkaloids, while pomelo (Citrus maxima (Burm.) Merr.) and grapefruit (Citrus paradisi Macfad.) were demonstrated to be rich in flavanols, flavanones, and coumarins [2]. These specialized metabolites make agro-food waste products from the cultivation of fruit trees secondary raw materials that need to be valorized, in a circular economy perspective, for medical purposes, as food additives, or for the formulation of cosmetic products.
The cosmetic industry is a rapidly growing and profitable sector, driven by the annual introduction of more advanced and specialized products. The leading cosmetics categories include make-up, skincare, hair care, toiletries, perfumes, and deodorants. Specifically, skincare products lead the market, accounting for about 36% of the global cosmetic industry. To date, natural products, rich in human health-beneficial specialized metabolites, have been employed to treat various skin conditions, such as inflammation or infection, and to protect against pollution and ultraviolet (UV) radiation [5]. Thus, recognizing the importance of natural products in cosmetics has led to the emergence of so-called biocosmetics as replacements for synthetic cosmetics. Several marked cosmetic preparations are, indeed, prepared with synthetic ingredients such as parabens (which may have carcinogenic properties) and compounds obtained from petroleum and silicon processing (petrolatum and silicones, respectively) due to their ease of preparation, application, storage, aesthetic appeal, and cost efficiency. However, they come with limitations, including potential aftereffects such as skin allergies [6] and damage to the natural skin hydrolipidic film [7]. For this reason, in the actual era of biocosmetics, botanical extracts supporting skin and hair health, texture, and integrity find widespread application in commercial cosmetic formulations. Nowadays, the growing demand for cosmetic formulations with natural and traceable constituents lacking dangerous chemicals meets the needs of consumers in an increasingly health-conscious world [8]. Specifically, global demand for plant-based products is growing by 10% to 15% annually [9]. The United States and Europe represent the two largest distributors of botanical products on the world market, with 41% and 20% market shares, respectively [10]. In 2011, the Personal Care Product Council reported that about one-third of the ingredients in the International Nomenclature Cosmetic Ingredient (INCI) were classified as botanicals obtained through several manufacturing procedures applied to plant materials. The latter, indeed, undergo different processes to obtain extracts, tinctures, waxes, vegetable and essential oils, juices, purified antioxidants, vitamins, lipids, carbohydrates, and other bioactive substances. The Latin binomial name is used in the INCI designation to indicate the specific part of the plant used (such as root or leaf) and the product obtained from its processing (such as oil, juice, or extract). This system provides a standardized way to label and communicate the ingredients used in these products, ensuring transparency and clarity for consumers and regulatory purposes [11]. Nevertheless, the most important natural ingredients in the cosmetics industry are oils, fats, surfactants, emulsifiers, natural colorants, clays, and essential oils. In particular, wood represents an interesting source of ingredients for cosmetic purposes. For instance, biomass residues from Cryptomeria japonica (Thunb. ex L.f.) D.Don (roots, cones, bark, sapwood, heartwood) represent a rich source of specialized metabolites like flavanols, terpenes, and lignans with potential application in cosmeceutical fields [12]. Apart from the healthy properties, like anti-inflammatory and antioxidant properties, noteworthy is also their antimicrobial and antifungal properties, which make it possible to use plant extracts as preservatives in cosmetics. From this perspective, cosmetic science has rapidly improved with the substitution of synthetic preservatives with natural ones [13].
Therefore, building on previous research on the biological and chemical characterization of extracts from orange, olive, and apricot bark and wood [14,15,16] this study aimed to outline the process of developing a natural cosmetic cream prototype for industrial applications. Specifically, the purpose of this study lies in (I) identifying the extract with the highest antioxidant index (RACI), (II) analyzing it using HPLC-MS, (III) performing cell cytotoxicity tests, and (IV) incorporating the characterized extract into a cosmetic cream specifically designed for the production of prototypes.
2. Materials and Methods
2.1. Orchard Biomass Extracts and Antioxidant Evaluation
In the present investigation, extracts and antioxidant results from pruning branches of olive trees (Olea europaea L. cv Coratina), apricot trees (Prunus armeniaca L. cv Pisana), and orange trees (Citrus sinensis L. cv Navellina), obtained in a previous study were used [14,15,16]. Specifically, extracts were obtained using four distinct extraction techniques: maceration (ME), ultrasound-assisted extraction (UAE), accelerated solvent extraction (ASE), and autoclaving (AT). For all extraction methods, 10 g of orchard tree bark and wood biomass, pulverized into powder using a milling machine (Retsch GmbH, Haan, Germany) equipped with a 40-mesh sieve, was subjected to extraction using an ethanol:water mixture (70:30 v/v) as the solvent for ME, UAE, and AT and only water for AT. Further information about the extraction methods applied can be found in our previous studies [14,15,16].
2.2. Relative Antioxidant Capacity Index (RACI) Determination
In the field of antioxidant research, the relative antioxidant capacity index (RACI) has emerged as a valuable tool for standardizing the assessment of antioxidant activity in different samples and studies. This approach addresses the inherent variability of individual antioxidant assays by combining their results into a single normalized metric. The application of RACI thus plays a significant role in the quest for accurate and comparable antioxidant capacity measurements. One of the main advantages of RACI lies in its ability to minimize bias. Antioxidants act through various mechanisms, including electron transfer and hydrogen atom donation. Different assays are adapted to measure these mechanisms, leading to results that are not always directly comparable. By integrating the results of multiple tests, RACI accounts for these differences and offers a representation of an extract’s antioxidant capacity. This process is achieved through normalization, in which raw test values are converted into z-scores (or standard scores), allowing results from different tests to be analyzed and compared collectively [16].
In the case of the present investigation, the relative antioxidant capacity index (RACI) was calculated by comparing antioxidant data from earlier studies on the antioxidant activity of bark (B) and wood (W) extracts of apricot tree (AP), olive tree (OL), and orange tree (OR) [14,15,16]. The z-scores were calculated considering the following formula [16]:
where x is the individual data point (e.g., an antioxidant assay result), µ is the mean (average) of all data points in the dataset, and σ is the standard deviation of the dataset, which measures how spread out the values are around the mean.
2.3. U-HPLC–MS Analysis
Extract phytochemical characterization was performed using ultra-high-performance liquid chromatography (U-HPLC) (Shimadzu, Kyoto, Japan) on a Shimadzu LC–MS-8030 system featuring a Diode Array Detector SPDM20A. Separation was performed at a steady 40 °C on a reversed-phase Phenomenex® Luna 3 μm C18 column (i.d. 150 mm × 3 mm, Phenomenex, Torrance, CA, USA). Elution employed a binary solvent system consisting of water with 0.1% formic acid and acetonitrile with 0.1% formic acid at a 0.4 mL/min flow rate. The sample volume injected was 1.0 μL. Detection employed UV monitoring at 280 nm, along with ion detection in both positive and negative modes through electrospray ionization mass spectrometry (ESI-MS) (Shimadzu, Kyoto, Japan). The operational settings for mass spectrometry included a nitrogen nebulizing gas flow of 3.0 L/min, an interface voltage of 4.5 kV, drying gas at 15 L/min, and a gas pressure of 230 kPa, with the desolvation line and block heater temperatures set at 250 °C and 400 °C, respectively. Data acquisition and analysis were performed using Shimadzu’s LabSolution software (v. 5.42SP4). Peak identification was based on mass spectral data, which were compared to literature references, databases (MassBank Europe, MassBank North America, and PubChem), and reference standards for (+) catechin hydrate and (−) epicatechin, both purchased from Sigma–Aldrich, Louis, MI, USA. The relative quantification of the identified compounds involved measuring the area of each peak in the total ion current (TIC) chromatogram in negative mode. This was achieved by integrating the area of each peak and expressing it as a percentage of the total area of all identified peaks in the chromatogram.
2.4. Cell Culture and Drug Treatment
The HepG2 (human hepatocellular carcinoma cell line) was cultured in DMEM (embedded with 2 mM glutamine, 10% fetal bovine serum, 100 μg/mL streptomycin, and 100 U/mL penicillin) and incubated at 37 °C in a humidified environment containing 5% CO2. The extract, chosen after RACI calculation, and the formulated cream prototype were solubilized in DMSO and tested at different concentrations. The DMSO concentration in the culture medium was kept at 1% (v/v), a level that did not interfere with cell growth when compared to controls without DMSO. For all experiments, DMSO-treated cells were used as the control [17].
2.5. Cytotoxicity Assay
An evaluation of the extract’s possible cytotoxicity was carried out to identify the optimal concentration of the extract to be included in the cream formulation. The extract was tested at 0.250, 0.100, 0.050, 0.025, 0.013, 0.006, 0.003, and 0.002 mg/mL, and cell viability was evaluated after 24, 48, and 78 h. The viability of HepG2 cells was measured using an MTT assay, as reported in previous investigations [18]. The cytotoxicity assay was repeated on the cream with the selected extract and tested at 5.000, 2.000, 1.000, 0.500, and 0.250 mg/mL.
2.6. Cream Formulation
Cream formulations (Table 1) were produced using chemicals acquired from Ecocosmesicreativa di A.M. and distilled water.

Table 1.
Ingredients employed for the formulation of the cream.
2.7. Preparation of Cream
Water and glycerine were poured and stirred in a stainless steel vessel using a mixer (IP10F, Fimar, Rimini, Italy). This blend was then warmed on a Falc hot plate (F60 FALC instrument, Como, Italy) until it reached a temperature of up to 70 °C. While stirring, lactil, xanthan gum, sodium citrate, phytic acid, and sodium dehydroacetate were added to this mixture (Phase A). Separately, in a different container, olive oil, rice oil, sunflower oil, shea butter, AperoxidTLA, Olivem 1000, and vitamin E acetate were combined (Phase B). Phase B was then heated on a hot plate until it reached a temperature of up to 70 °C. Subsequently, Phase A was carefully blended into Phase B to create an emulsion. After allowing the combined mixture to cool down to 30 °C, the chosen extract and Dermosoft Eco 1388 were added while stirring.
2.8. Cream Quality Check
The examination of samples involved measuring the pH with a pH meter (Hanna Instruments, Padova, Italy), determining viscosity using a viscometer (PCE Deutschland GmbH, Meschede, Germany), and evaluating stability and microbial characteristics using Contact Slides 2 and 4 (Liofilchem™, Teramo, Italy). To test the emulsion’s resilience to mechanical stress and potential phase separation, a centrifuge (CN-45, Orma, Taranto, Italy) was used. Contact Slide 2, a dual-medium, plastic-supported, ready-for-use device, is designed for the microbial assessment of both liquids and surfaces, accommodating the presence of disinfectant residues. Its selective medium aids in the isolation and counting of yeasts and molds. Contact Slide 4 offers a two-medium, ready-for-use format for bacterial count, particularly targeting Pseudomonas, yeasts, and molds, ensuring bacteriological control of surfaces with disinfectant inactivation. Quantitative and qualitative limits were determined according to the European Standard EN ISO 17516:2014 Cosmetics–Microbiology–Microbiological limits [19]. This standard, ratified by the CEN on 9 August 2014, serves as a critical reference in the cosmetics sector internationally.
2.9. Statistical Analysis
The tests were conducted thrice, and outcomes were presented as the average ± standard deviation. Statistical evaluations were conducted with GraphPad Prism 5 Software, Inc. (San Diego, CA, USA). A p-value ≤ 0.05 was deemed to signify statistical significance.
3. Results
3.1. Relative Antioxidant Capacity Index (RACI)
Results from the antioxidant activity of apricot tree (AP), olive tree (OL), and orange tree (OR) bark (B) and wood (W) extracts obtained in previous investigations through maceration (ME), accelerated solvent extraction (ASE), autoclaving (AT), and ultrasound-assisted extraction (UAE) [14,15,16] were analyzed and compared by calculating the RACI.
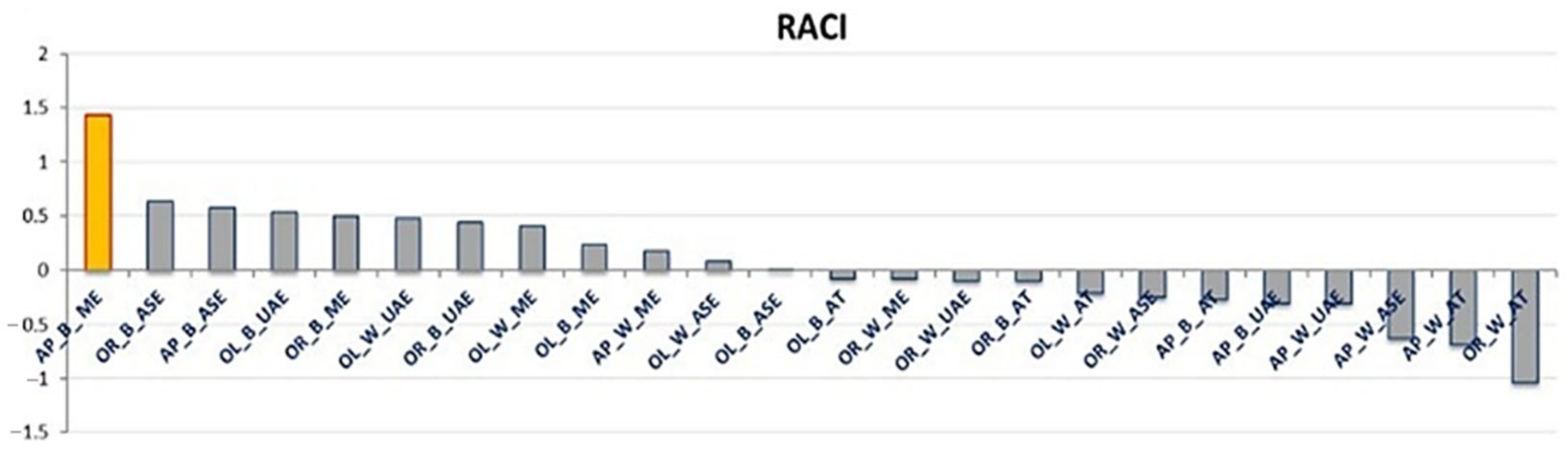
The RACI provides a reliable antioxidant capacity ranking of the samples studied (21), allowing the extract obtained by AP_B_ME to be identified as the most active (RACI value of 1.43) followed by OR_B_ASE and AP_B_ASE (resulting in a RACI value of 0.62 and 0.57, respectively). Contrarily, the OR_W_AT extract showed the lowest RACI value (−1.04), and, in the complex, as shown in Figure 1, the AT techniques resulted in extracts with a lower antioxidant capacity index than the other extractive methods. These results can be explained considering that the AT extraction technique employs water as the extraction solvent, while for the other extractions made, a hydroalcoholic solution was used (70% EtOH/H2O) [14,15,16]. The results obtained confirm previous studies demonstrating that apolar solvents such as ethanol or methanol used alone or in a mixture with water allow for better extraction of phenolic compounds from plant matrices than water alone [20]. Regarding the differences in the wood and bark extracts, it was observed that eight bark and four wood samples had a positive RACI. The different phytochemical compositions of bark and wood could explain these results. It was indeed demonstrated that the sapwood and internal bark contain high levels of nutrients such as sucrose and glycosides. In contrast, although deficient in these nutrients, the external bark is abundant in active molecules involved in protecting plant tissues from environmental threats [21]. This explains the highest antioxidant capacity index of the bark, corroborating similar findings in apple tree studies [22].
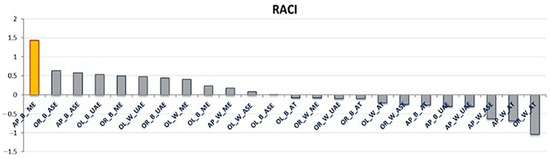
Figure 1.
Relative antioxidant capacity index (RACI) evaluated for the apricot (AP), orange (OR), and olive (OL) tree bark (B) and wood (W) extracts obtained with maceration (ME), ultrasound-assisted extraction (UAE), autoclaving (AT), and accelerated solvent extraction (ASE). The yellow bar indicates the extract with the highest RACI value.
Furthermore, olive extracts are found to be more distributed in the positive part of the RACI graph (Figure 1) than apricot and orange. This can be related to the selected olive trees’ age, which have thus adapted over the years to cope with various abiotic and biotic stresses, resulting in increased production of specialized metabolites [23].
3.2. LC-MS Analysis
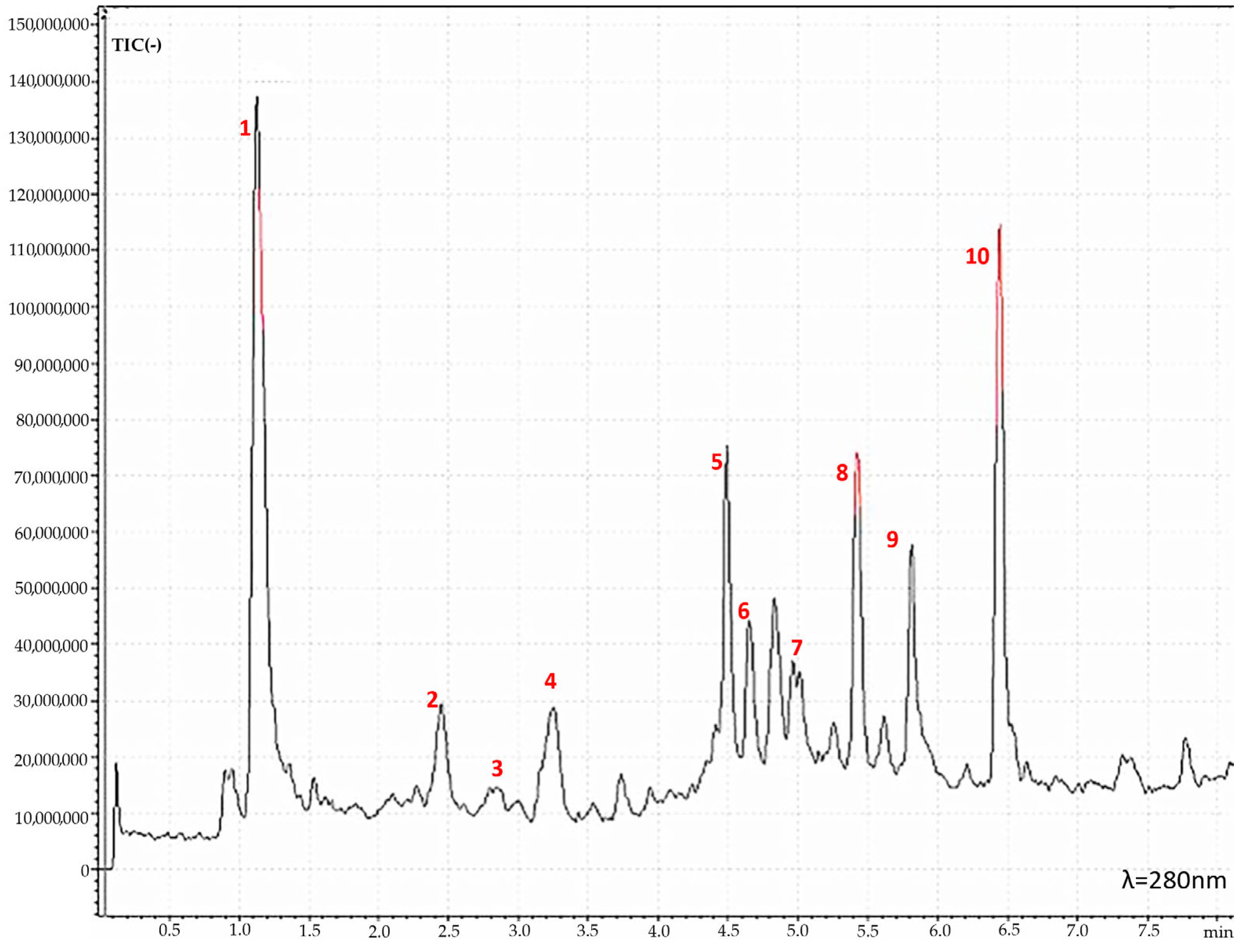
Based on the results from the RACI, the extract from apricot tree bark obtained with maceration (AP_B_ME) was recognized as the most active; hence, its phytochemical characterization was performed using HPLC-MS. Figure 2 represents the AP_B_ME chromatogram from the HPLC-MS analysis, while Table 2 reports the compounds identified.
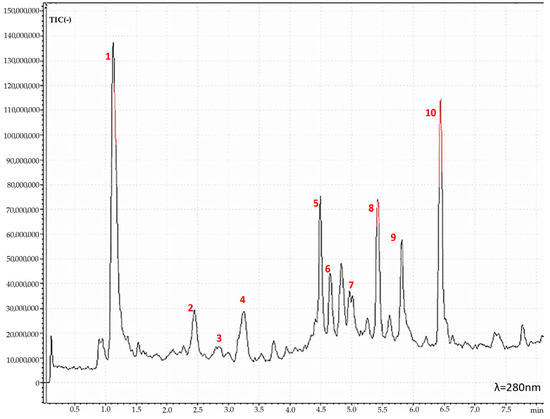
Figure 2.
HPLC-MS chromatogram of apricot tree bark extract obtained with maceration (AP_B_ME) showing the peaks of the active molecules tentatively identified: (1) Unknown, (2) Catechin, (3) Epicatechin, (4) Flavonoid, (5) Hypoprotocetraric Acid, (6) Procyanidin Dimer (Type A), (7) Phenolic Glycosides, (8) Unknown, (9) Scopoletin, and (10) Naringenin.

Table 2.
Molecules identified in apricot tree bark maceration (AP_B_ME) extract using HPLC-MS.
Except for catechin and epicatechin, a tentative identification was made for the other compounds. Epicatechin and catechin are recognized as two of the most important flavonols with significant nutritional relevance for human health [24], showing notable antioxidant and antimutagenic activities [25]. Furthermore, several investigations have demonstrated catechins’ ability to reduce and prevent skin damage by slowing the degradation of the extracellular matrix caused by environmental pollution and ultraviolet radiation. Catechins also have direct beneficial effects on the skin by promoting the synthesis of collagen, inhibiting the formation of matrix metalloproteinase enzymes, and improving the absorption of bio-cosmetic ingredients. These characteristics make catechins of considerable importance for cosmetics formulations [26].
Apart from catechin and epicatechin, the other identified peaks were m/z 191 [Scopoletin + H]−, m/z 271 [Naringenin + H]−, m/z 343 [Hypoprotocetraricacid + H]−, m/z 505 [Flavonoid + H]−, m/z 559 [Phenolic glycosides + H]−, m/z 559 [Phenolic glycosides + H]−, and m/z 575 [procydin dimer A + H]−. Peaks with m/z 505, m/z 559, and m/z 575 were phenolic molecules widely recognized for their anti-inflammatory, antioxidant, vasodilatory, and antithrombotic activities [27,28,29,30]. In the contest of skin protection, phenolic-rich extracts have been suggested as a highly effective starting material for cosmetics with anti-aging properties. Phenolic molecules have indeed been demonstrated to exert anti-hyaluronidase, anti-collagenase, and anti-elastase properties, thereby inhibiting enzymes responsible for visible skin aging signs such as wrinkles. Moreover, phenolic compounds have chromophores in their structures that are responsible for absorbing ultraviolet radiation. This prevents solar radiation from penetrating the skin and provides sun protection (SPF). This characteristic enhances the sun protection offered by cosmetic products formulated with phenolic-rich extracts and counteracts the detrimental effects of oxidative stress following sun exposure. Results from in vitro investigations on epidermal cell lines indeed have demonstrated that pre-treatment with phenolic extracts significantly decreases the formation of free radicals, thereby preventing DNA damage [31]. Specifically, naringenin, found in AP_B_ME at the highest concentration (60.9 mg/g), was found to avoid aging, oxidative damage, and inflammation induced by UVB in a cell-based investigation on human HaCaT keratinocytes [32] and an in vivo investigation on hairless mice skin [33]. On the other hand, scopoletin, the second most abundant compound of AP_B_ME (35.2 mg/g), was observed to enhance melanin production in B16F10 cell lines by upregulating tyrosinase and microphthalmia-associated transcription factor (MITF) through the phosphorylation of cAMP response element-binding protein (CREB) [34]. Therefore, based on the phytochemical profile, AP_B_ME represents a suitable extract to be incorporated in a cosmetic formulation for skin protection [35].
3.3. Apricot Bark Maceration Extract’s Cytotoxic Effect on HepG2 Cells
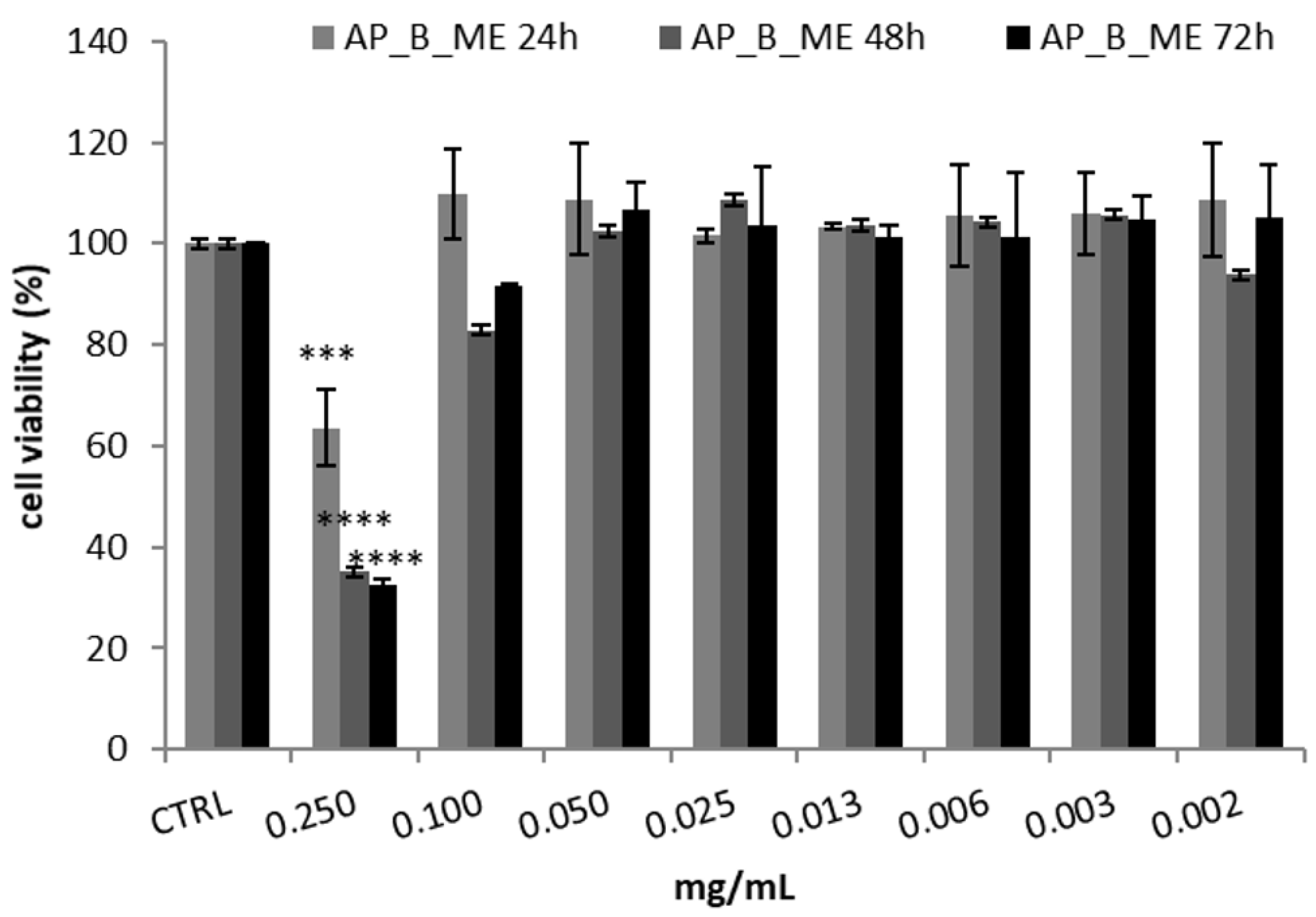
HepG2, a human hepatocellular carcinoma cell line with metabolic competence, is widely employed for in vitro simulations of liver functions in the human body. It exhibits either phase I or phase II enzymatic activities, comprising cytochrome P450 enzymes. Using this cell line for analyzing cosmetic ingredients might seem pointless given the expectation that active cosmetic substances do not typically penetrate the skin. However, its use becomes essential when conducting in vitro tests to assess the possible skin permeation of cosmetic products [36]. Hence, in the present investigation, the HepG2 cell line was chosen to select the concentration of AP_B_ME extract to be included in the cream based on its potential toxicity evaluated with the MTT assay. This decision was taken based on previous studies indicating that cosmetic ingredients tend to target the liver more frequently than other organs [37]. Specifically, the HepG2 cell line was subjected to treatment with various AP_B_ME extract concentrations; cell viability was detected after 24, 48, and 72 h. As reported in Figure 3, only the extract’s highest concentration (0.250 mg/mL) significantly determines a reduction in cell viability at all times. In contrast, the other tested concentrations (0.100–0.002 mg/mL) did not show a cytotoxic effect, exhibiting an IC50 value (i.e., the extract concentration which results in 50% inhibition of cell viability) of 0.138 ± 0.02 and 0.137 ± 0.01 mg/mL after 48 and 72 h treatment, respectively.
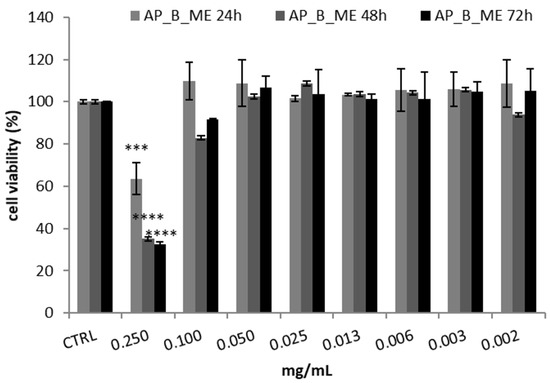
Figure 3.
Cell viability, assessed with the MTT assay, of HepG2 cells treated with apricot tree bark maceration extract (AP_B_ME) at different concentrations. Cell viability was measured after 24, 48, and 72 h. The results are reported as the mean ± SD of three independent experiments (n = 3). **** p < 0.0001 and *** p < 0.001 vs. CTRL.
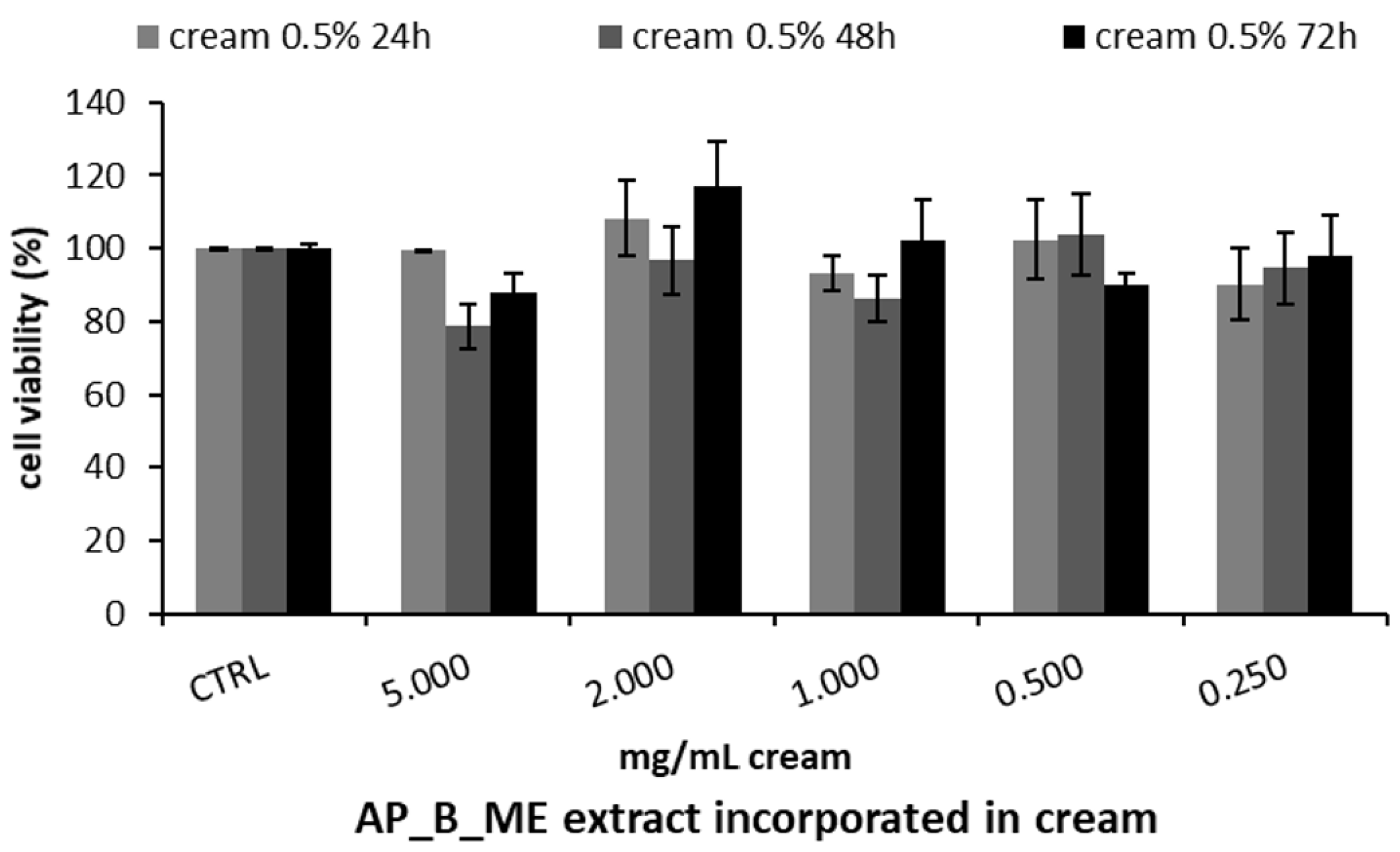
Based on these results, the apricot bark extract obtained with maceration was used to formulate a skin cream at a ratio of 0.5% v/v. Therefore, the MTT test on HepG2 was repeated using different concentrations of the cream enriched with the extract, demonstrating no cytotoxic effect at any of the concentrations tested after 24, 48, and 72 h (Figure 4).
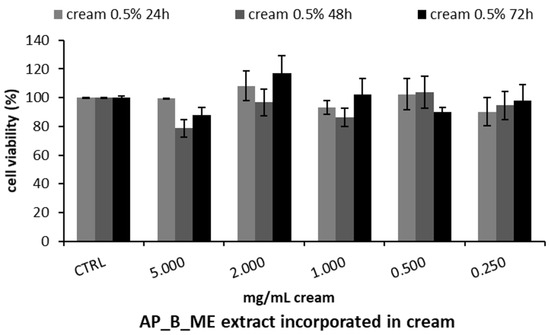
Figure 4.
Cell viability, assessed with the MTT assay, of HepG2 cells treated with different concentrations of apricot tree bark maceration (AP_B_ME) extract included in the cream prototype at 0.5% v/v. Cell viability was measured after 24, 48, and 72 h. The results are expressed as the mean ± SD of three independent experiments (n = 3).
Based on these results, it is possible to speculate that the cosmetic active ingredients can be considered safe in case they reach the bloodstream after skin permeation. These are promising results given using AP_B_ME extract as preservatives, thus avoiding the use of chemicals like parabens, which are parahydroxybenzoic acid esters highly employed for cream preparation by the pharmaceutical and cosmetic industries. An example of paraben used in cosmetics is butylparaben and its derivatives. However, they are categorized as allergens and have reported in vitro toxic effects, causing a reduction in HepG2 cell viability dose dependently above 400 µM [38]. The parabens’ possible toxicity is also supported by regulations issued by the European Commission on 16 April 2015, which mandate that manufacturers must decrease the presence of butylparaben and propylparaben in their cosmetic products. Furthermore, manufacturers are prohibited from incorporating these preservatives into products designed for use with diapers for children under three years of age. For this reason, in the field of biocosmetics, the search for new safe ingredients is demanded, making the results from the present investigation very encouraging for introducing safe, new-generation ingredients in cosmetic products and reaching zero-waste targets.
3.4. Quality Check
Developing and commercializing a cosmetic product requires a precise and detailed process guided by specific legislation [39]. In the cosmetics industry, adherence to Good Manufacturing Practice (GMP) is outlined in UNI EN ISO 22716: 2008 [40]. These guidelines provide detailed instructions for ensuring the quality and safety of cosmetic products throughout their production, quality control, packaging, and transportation processes. Furthermore, European Union Regulation No. 1223/2009 has a pivotal role in overseeing and regulating the entire process of production, packaging, distribution, and trading of cosmetics. The primary objective of this regulation is to safeguard consumer interests and ensure their protection. In particular, following this regulation, a cosmetic launched on the market must ensure human health safety under typical or reasonably anticipated usage conditions.
In the present investigation, quality control analyses of the cream produced were performed following EU directive 1223/2009 (Table 3). The microbial analysis described above aligns with procedures commonly adopted to ensure the microbiological safety of cosmetic formulations, as outlined in Isnaini et al. (2025) [41], and adheres to ISO 21149:2017 [42] for detecting aerobic microorganisms and ISO 22717:2015 [43] for Pseudomonas spp. detection. These measures ensure a thorough evaluation of the product’s safety in compliance with regulatory standards. The confirmed absence of harmful pathogens in our formulation underscores its suitability for consumer use.

Table 3.
Quality check of the cream.
The assessments included visually evaluating the cream’s appearance, consistency, and phase separation. Color (Figure 5) was assessed by visual inspection, while odors were analyzed by smell.

Figure 5.
Aspect of the face cream produced with apricot tree bark maceration (AP_B_ME) extract (Source: Authors’ collection).
The pH, which must range between 4.8 and 5.8, was evaluated using the litmus test. Density was determined using a viscosimeter. A small amount of cream was placed on contact slides 2 and 4 for microbial analysis. After 24 h, the slides were examined for the presence of yeast, molds, bacteria, and Pseudomonas spp. Figure 6 demonstrates that no harmful pathogens were detected.

Figure 6.
Contact Slide 2—Microbial analysis: (a) bacterial counts and (b) yeast and moulds. Contact slide 4—Microbial analyses: (c) Pseudomonas spp. and (d) yeast and molds (Source: Authors’ collection).
4. Conclusions
This study demonstrates the feasibility of developing a prototype of an environmentally friendly cosmetic cream using apricot pruning by-products, highlighting its potential in the context of the circular economy. Specifically, by exploiting the antioxidant properties of different orchard tree biomass extracts throughout the RACI calculation, the apricot bark maceration extract (AP_B_ME) demonstrated the highest antioxidant capacity index. At the same time, UHPLC-MS analysis identified a range of compounds in AP_B_ME, including polyphenols like catechin and epicatechin. Hence, AP_B_ME is used in cosmetic applications. The formulated cream was found to be stable, non-toxic, and pathogen-free, confirming its safety and quality for consumer use. These results provide a solid basis for further industrial development and potential commercialization of sustainable cosmetic products derived from agri-food by-products. This approach not only adds value to pruning residues but also addresses environmental challenges, opening the way for innovative solutions in sustainable cosmetic formulations.
Author Contributions
M.R.B., M.P. and I.F.: conceptualization, investigation, methodology, software, validation, and writing–original draft; M.R.B., C.S., M.P., I.F., L.M. and L.T.: title—related advice and writing—review and editing; M.R.B., L.M. and L.T.: preparation and supervision. All authors have read and agreed to the published version of the manuscript.
Funding
This research received a specific grant of funding from “Region Basilicata, Industrial PhD 4.0” and the European Union Next-Generation EU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR)–MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4–D.D. 1032 17/06/2022, CN00000022). This manuscript reflects only the authors’ views and opinions, neither the European Union nor the European Commission can be considered responsible for them.
Data Availability Statement
Data are available upon request.
Acknowledgments
The PhD program in agricultural, forest, and environmental sciences was coordinated by F. Napolitano at the University of Basilicata, with the great support of my tutor, Luigi Todaro, and my co-tutor, Luigi Milella. A special thanks to Ecocosmesicreativa by A.M. for providing the materials and laboratory facilities for cream prototype development. This study was carried out within the Agritech National Research Center.
Conflicts of Interest
Author Immacolata Faraone was employed by the company Innovative Startup Farmis s.r.l. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Rațu, R.N.; Veleșcu, I.D.; Stoica, F.; Usturoi, A.; Arsenoaia, V.N.; Crivei, I.C.; Postolache, A.N.; Lipșa, F.D.; Filipov, F.; Florea, A.M. Application of Agri-Food By-Products in the Food Industry. Agriculture 2023, 13, 1559. [Google Scholar] [CrossRef]
- Aliaño-González, M.J.; Gabaston, J.; Ortiz-Somovilla, V.; Cantos-Villar, E. Wood waste from fruit trees: Biomolecules and their applications in agri-food industry. Biomolecules 2022, 12, 238. [Google Scholar] [CrossRef] [PubMed]
- Valdebenito, F.; Ramírez-Álvarez, R.; Muñoz, M.A.; Pecchi, G.; Canales, R.; Ormazabal, S.; Muñoz, R.; Alejandro-Martín, S.; Quero, F.; Adam, R. Biomass characterization and solvent extraction as tools to promote phenol production from urban pruning. Fuel 2024, 362, 130830. [Google Scholar] [CrossRef]
- Ferreira, S.M.; Gomes, S.M.; Santos, L. The Chemistry Behind Biological Properties of Agro-industrial Portuguese By-Products. Waste Biomass Valorization 2023, 15, 2721–2733. [Google Scholar] [CrossRef]
- Mahesh, S.K.; Fathima, J.; Veena, V.G. Cosmetic potential of natural products: Industrial applications. In Natural Bio-Active Compounds: Volume 2: Chemistry, Pharmacology and Health Care Practices; Springer: Berlin/Heidelberg, Germany, 2019; pp. 215–250. [Google Scholar] [CrossRef]
- Kuno, N.; Matsumoto, M. Skin-Beautifying Agent, Anti-Aging Agent for the Skin, Whitening Agent and External Agent for the Skin. U.S. Patent 6,682,763, 27 January 2004. [Google Scholar]
- Tam, C.C.; Elston, D.M. Allergic contact dermatitis caused by white petrolatum on damaged skin. DERM 2006, 17, 201–203. [Google Scholar] [CrossRef]
- Joshi, L.S.; Pawar, H.A. Herbal cosmetics and cosmeceuticals: An overview. Nat. Prod. Chem. Res. 2015, 3, 170. [Google Scholar] [CrossRef]
- Ashawat, M.; Banchhor, M.; Saraf, S.; Saraf, S. Herbal Cosmetics: “Trends in Skin Care Formulation”. Pharmacogn. Rev. 2009, 3, 82. [Google Scholar]
- Trüeb, R.M. The value of hair cosmetics and pharmaceuticals. Dermatology 2001, 202, 275–282. [Google Scholar] [CrossRef]
- Ferreira, M.S.; Magalhães, M.C.; Oliveira, R.; Sousa-Lobo, J.M.; Almeida, I.F. Trends in the use of botanicals in anti-aging cosmetics. Molecules 2021, 26, 3584. [Google Scholar] [CrossRef]
- Lima, A.; Arruda, F.; Janeiro, A.; Medeiros, J.; Baptista, J.; Madruga, J.; Lima, E. Biological activities of organic extracts and specialized metabolites from different parts of Cryptomeria japonica (Cupressaceae)—A critical review. Phytochemistry 2023, 206, 113520. [Google Scholar] [CrossRef]
- Rybczyńska-Tkaczyk, K.; Grenda, A.; Jakubczyk, A.; Kiersnowska, K.; Bik-Małodzińska, M. Natural Compounds with Antimicrobial Properties in Cosmetics. Pathogens 2023, 12, 320. [Google Scholar] [CrossRef] [PubMed]
- Bruno, M.R.; Russo, D.; Cetera, P.; Faraone, I.; Lo Giudice, V.; Milella, L.; Todaro, L.; Sinisgalli, C.; Fritsch, C.; Dumarçay, S. Chemical analysis and antioxidant properties of orange-tree (Citrus sinensis L.) biomass extracts obtained via different extraction techniques. Biofuels Bioprod. Biorefin. 2020, 14, 509–520. [Google Scholar] [CrossRef]
- Bruno, M.R.; Russo, D.; Faraone, I.; D’Auria, M.; Milella, L.; Todaro, L. Orchard biomass residues: Chemical composition, biological activity and wood characterization of apricot tree (Prunus armeniaca L.). Biofuels Bioprod. Biorefin. 2021, 15, 377–391. [Google Scholar] [CrossRef]
- Faraone, I.; Russo, D.; Bruno, M.R.; Todaro, L.; D’Auria, M.; Milella, L. Focus on Olea europaea L. pruning by-products: Extraction techniques, biological activity, and phytochemical profile. Biofuels Bioprod. Biorefin. 2021, 15, 1835–1849. [Google Scholar] [CrossRef]
- Armentano, M.F.; Bisaccia, F.; Miglionico, R.; Russo, D.; Nolfi, N.; Carmosino, M.; Andrade, P.B.; Valentão, P.; Diop, M.S.; Milella, L. Antioxidant and proapoptotic activities of Sclerocarya birrea [(A. Rich.) Hochst.] methanolic root extract on the hepatocellular carcinoma cell line HepG2. BioMed Res. Int. 2015, 2015, 561589. [Google Scholar] [CrossRef]
- Russo, D.; Miglionico, R.; Carmosino, M.; Bisaccia, F.; Andrade, P.B.; Valentão, P.; Milella, L.; Armentano, M.F. A comparative study on phytochemical profiles and biological activities of Sclerocarya birrea (A. Rich.) Hochst leaf and bark extracts. Int. J. Mol. Sci. 2018, 19, 186. [Google Scholar] [CrossRef]
- EN ISO 17516:2014; Cosmetics—Microbiology—Microbiological Limits. International Organization for Standardization: Geneva, Switzerland, 2014.
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Gao, H.; Shupe, T.F.; Eberhardt, T.L.; Hse, C.Y. Antioxidant activity of extracts from the wood and bark of Port Orford cedar. J. Wood Sci. 2007, 53, 147–152. [Google Scholar] [CrossRef]
- Withouck, H.; Boeykens, A.; Vanden Broucke, M.; Moreira, M.M.; Delerue-Matos, C.; De Cooman, L. Evaluation of the impact of pre-treatment and extraction conditions on the polyphenolic profile and antioxidant activity of Belgium apple wood. Eur. Food Res. Technol. 2019, 245, 2565–2578. [Google Scholar] [CrossRef]
- Cui, J.; Li, X.; Lu, Z.; Jin, B. Plant secondary metabolites involved in the stress tolerance of long-lived trees. Tree Physiol. 2024, 44, tpae002. [Google Scholar] [CrossRef]
- Yilmaz, Y. Novel uses of catechins in foods. Trends Food Sci. Technol. 2006, 17, 64–71. [Google Scholar] [CrossRef]
- Mendoza-Wilson, A.M.; Glossman-Mitnik, D. Theoretical study of the molecular properties and chemical reactivity of (+)-catechin and (−)-epicatechin related to their antioxidant ability. J. Mol. Struct. Theochem 2006, 761, 97–106. [Google Scholar] [CrossRef]
- Bae, J.; Kim, N.; Shin, Y.; Kim, S.-Y.; Kim, Y.-J. Activity of catechins and their applications. Biomed. Dermatol. 2020, 4, 8. [Google Scholar] [CrossRef]
- Olas, B. The antioxidant, anti-platelet and anti-coagulant properties of phenolic compounds, associated with modulation of hemostasis and cardiovascular disease, and their possible effect on COVID-19. Nutrients 2022, 14, 1390. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Quispe, C.; Zam, W.; Kumar, M.; Cardoso, S.M.; Pereira, O.R.; Ademiluyi, A.O.; Adeleke, O.; Moreira, A.C.; Živković, J. Phenolic bioactives as antiplatelet aggregation factors: The pivotal ingredients in maintaining cardiovascular health. Oxidative Med. Cell. Longev. 2021, 2021, 2195902. [Google Scholar] [CrossRef]
- Wilcox, L.J.; Borradaile, N.M.; Huff, M.W. Antiatherogenic properties of naringenin, a citrus flavonoid. Cardiovasc. Drug Rev. 1999, 17, 160–178. [Google Scholar] [CrossRef]
- Yi, L.; Ma, S.; Ren, D. Phytochemistry and bioactivity of Citrus flavonoids: A focus on antioxidant, anti-inflammatory, anticancer and cardiovascular protection activities. Phytochem. Rev. 2017, 16, 479–511. [Google Scholar] [CrossRef]
- De Lima Cherubim, D.J.; Buzanello Martins, C.V.; Oliveira Fariña, L.; da Silva de Lucca, R.A. Polyphenols as natural antioxidants in cosmetics applications. J. Cosmet. Dermatol. 2020, 19, 33–37. [Google Scholar] [CrossRef]
- El-Mahdy, M.A.; Zhu, Q.; Wang, Q.E.; Wani, G.; Patnaik, S.; Zhao, Q.; Arafa, E.S.; Barakat, B.; Mir, S.N.; Wani, A.A. Naringenin protects HaCaT human keratinocytes against UVB-induced apoptosis and enhances the removal of cyclobutane pyrimidine dimers from the genome. Photochem. Photobiol. 2008, 84, 307–316. [Google Scholar] [CrossRef]
- Martinez, R.M.; Pinho-Ribeiro, F.A.; Steffen, V.S.; Caviglione, C.V.; Vignoli, J.A.; Barbosa, D.S.; Baracat, M.M.; Georgetti, S.R.; Verri, W.A., Jr.; Casagrande, R. Naringenin inhibits UVB irradiation-induced inflammation and oxidative stress in the skin of hairless mice. J. Nat. Prod. 2015, 78, 1647–1655. [Google Scholar] [CrossRef]
- Ahn, M.-J.; Hur, S.-J.; Kim, E.-H.; Lee, S.H.; Shin, J.S.; Kim, M.-K.; Uchizono, J.A.; Whang, W.-K.; Kim, D.-S. Scopoletin from Cirsium setidens increases melanin synthesis via CREB phosphorylation in B16F10 cells. Korean J. Physiol. Pharmacol. Off. J. Korean Physiol. Soc. Korean Soc. Pharmacol. 2014, 18, 307. [Google Scholar] [CrossRef] [PubMed]
- Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The potential of plant phenolics in prevention and therapy of skin disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Prado Corrêa, G.; Marcato, D.C.; Ramos, W.S.; Corrêa, M.A.; Cicarelli, R.M.B.; Isaac, V.L.B. In vitro evaluation of the cytotoxicity and eye irritation potential of preservatives widely used in cosmetics. Braz. J. Pharm. Sci. 2022, 58, e20039. [Google Scholar] [CrossRef]
- Chiari, B.G.; Martini, P.C.; Moraes, J.D.D.; Andréo, R.; Correa, M.A.; Cicarelli, R.M.B.; Isaac, V.L.B. Use of HepG2 cells to assay the safety of cosmetic active substances. Int. J. Res. Cosmet. Sci. 2012, 2, 8–14. [Google Scholar]
- Kizhedath, A.; Wilkinson, S.; Glassey, J. Assessment of hepatotoxicity and dermal toxicity of butyl paraben and methyl paraben using HepG2 and HDFn in vitro models. Toxicol. Vitr. 2019, 55, 108–115. [Google Scholar] [CrossRef]
- McLaughlin, J.L.; Rogers, L.L.; Anderson, J.E. The use of biological assays to evaluate botanicals. Drug Inf. J. 1998, 32, 513–524. [Google Scholar] [CrossRef]
- UNI EN ISO 22716:2008; Cosmetics—Good Manufacturing Practices (GMP)—Guidelines on Good Manufacturing Practices. Ente Nazionale Italiano di Unificazione (UNI): Milan, Italy, 2008.
- Isnaini, N.; Harnelly, E.; Zulkarnain, Z.; Prajaputra, V.; Muhammad, S.; Syahraini, A.; Syaharani, C.P.S.; Nurfaizah, N.; Sarah, Y. Potential of patchouli (Pogostemon cablin) and champaca (Magnolia champaca) oils incorporated in facial wash formulation for effective anti-aging on human skin. J. Pharm. Pharmacogn. Res. 2025, 13, 459–474. [Google Scholar] [CrossRef]
- ISO 21149:2017; Cosmetics—Microbiology—Enumeration and Detection of Aerobic Mesophilic Bacteria. International Organization for Standardization: Geneva, Switzerland, 2017.
- ISO 22717:2015; Cosmetics—Microbiology—Detection of Pseudomonas aeruginosa. International Organization for Standardization: Geneva, Switzerland, 2015.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).