HBV Prevention and Treatment in Countries of Central Asia and the Caucasus
Abstract
:1. Introduction
2. Prevalence
3. Blood Donor Screening
4. HBV Vaccination
5. Treatment for HBV
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Burke, J. Post-Soviet World: What You Need to Know about the 15 States. The Guardian. 2014. Available online: https://www.theguardian.com/world/2014/jun/09/-sp-profiles-post-soviet-states (accessed on 24 July 2020).
- Gvozdetsky, N.A.; Bruk, S.I.; Owen, L. Caucasus. Encyclopædia Britannica. 2019. Available online: https://www.britannica.com/place/Caucasus (accessed on 22 July 2020).
- Hermann, W.; Linn, J. (Eds.) Central Asia and the Caucasus: At the Crossroads of Eurasia in the 21st Century; SAGE Publications: New Delhi, India, 2011. [Google Scholar]
- Worldometer. Countries in the World by Population. 2020. Available online: https://www.worldometers.info/world-population/population-by-country/ (accessed on 20 June 2020).
- YourFreeTemplates.com. Free Central Asia and Caucasus Editable Map. 2017. Available online: https://yourfreetemplates.com/free-central-asia-caucasus-editable-map/ (accessed on 20 June 2020).
- Lazarus, J.V.; Shete, P.B.; Eramova, I.; Merkinaite, S.; Matic, S. HIV/hepatitis coinfection in eastern Europe and new pan-European approaches to hepatitis prevention and management. Int. J. Drug Policy 2007, 18, 426–432. Available online: https://www.sciencedirect.com/science/article/pii/S0955395907000126?casa_token=S0AA2MLiC2QAAAAA:HCRgbtBPWa2np3X_i1NTV8HVBpSf9pPQnUqbbu1ybPi0axUEtfWJfbWieYXJGHOTc0R_vBaP4Mg (accessed on 24 June 2020). [CrossRef] [PubMed]
- Demirchyan, A.; Mirzoyan, L.; Thompson, M.E. Synthesis of the Existing Data on Hepatitis B in Armenia; American University of Armenia: Yerevan, Armenia, 2000. [Google Scholar]
- Ghazinyan, H.; Asoyan, A.; Mkhitaryan, A.; Melik-Andreasyan, G. Updating HBV status in Armenia. In Proceedings of the EASL Special Conference: Optimal Management of HBV infection, Athens, Greece, 25–27 September 2014; Available online: https://livertree.easl.eu/easl/2014/athens/62141/hasmik.levon.ghazinyan.updating.hepatitis.b.virus.(hbv).status.in.armenia.html?f=p6m3e757 (accessed on 1 August 2020).
- Kravchenko, N.; Maistat, L.; Golovin, S.; Nikelsen, T.; Aliyev, A.; Harantyunyan, A.; Biryukov, S.; Gulov, K.; Jamolov, P.; Pashaev, E.; et al. Otchet “Gepatit V i S v regione Vostochnoĭ Evropy i T͡Sentralʹnoĭ Azii: Epidemii͡a i Otvetnye Mery”. 2017. Available online: http://mv.ecuo.org/download/otchet-gepatit-v-i-s-v-regione-vostochnoj-evropy-i-tsentralnoj-azii-epidemiya-i-otvetnye-mery/ (accessed on 1 August 2020).
- Mamedov, M.; Dadasheva, A.; Kadyrova, A.; Tagizade, R.; Mikhailov, M. Serologicheskie Markery Infekt͡siĭ, Vyzvannykh Virusami Gepatitov v i s, u Zhiteleĭ Azerbaĭdzhana iz Grupp s Vysokim Riskom Parenteral’nogo Infit͡sirovanii͡a [Serological Markers of Infections Caused by Hepatitis B and C Viruses in Residents of Azerbaijan from Groups with a High Risk of Parenteral Infection]. Ėpidemiologii͡a vakt͡sinoprofilaktika 2012, 2, 63. Available online: https://cyberleninka.ru/article/n/serologicheskie-markery-infektsiy-vyzvannyh-virusami-gepatitov-v-i-s-u-zhiteley-azerbaydzhana-iz-grupp-s-vysokim-riskom-parenteralnogo (accessed on 24 June 2020).
- Schweitzer, A.; Akmatov, M.K.; Krause, G. Hepatitis B vaccination timing: Results from demographic health surveys in 47 countries. Bull. World Health Organ. 2017, 95, 199. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5328113/ (accessed on 24 June 2020). [CrossRef] [PubMed]
- Butsashvili, M.; Tsertsvadze, T.; McNutt, L.; Kamkamidze, G.; Gvetadze, R.; Badridze, N. Prevalence of hepatitis B, hepatitis C, syphilis and HIV in Georgian blood donors. Eur. J. Epidemiol. 2001, 17, 693–695. Available online: https://link.springer.com/article/10.1023/A:1015566132757 (accessed on 24 June 2020). [CrossRef] [PubMed]
- Stvilia, K.; Meparidze, M.; Tsertsvadze, T.; Sharvadze, L.; Dzigua, L. Prevalence of HBV and HCV infections and high risk behavior for blood born infections among general population of Tbilisi, Georgia. Ann. Biomed. Res. Educ. 2005, 5, 289–298. Available online: http://citeseerx.ist.psu.edu/viewdoc/download? (accessed on 24 June 2020).
- Khochava, M.; Shalamberidze, I.; Jokhtaberidze, T. Problema B i C gepatitov i ikh Registrat͡sii v Gruzii [The Problem of B and C Hepatitis and Their Registration in Georgia]. 2013. Available online: http://elib.grsmu.by/handle/files/15779 (accessed on 24 June 2020).
- Badridze, N.; Chkhartishvili, N.; Abutidze, A.; Gatserelia, L.; Sharvadze, L. Prevalence of hepatitis B and C among HIV positive patients in Georgia and its associated risk factors. Georgian Med. News, 2008; 165, 54–60. Available online: https://pubmed.ncbi.nlm.nih.gov/19124918/(accessed on 24 June 2020).
- Skorikova, S.V.; Burkitbaev, L.; Savchuk, T.; Zhiburt, E. Rasprostranennost’ VICH-, VGS-, VGV-infekt͡siĭ u donorov krovi g. Astany [Prevalence of HIV, HCV, HBV Infections among Blood Donors in Astana]. Voprosy Virusologii 2015, 60. Available online: https://cyberleninka.ru/article/n/rasprostranennost-vich-vgs-vgv-infektsiy-u-donorov-krovi-g-astany (accessed on 24 June 2020).
- Hope, V.; Eramova, I.; Capurro, D.; Donoghoe, M. Prevalence and estimation of hepatitis B and C infections in the WHO European Region: A review of data focusing on the countries outside the European Union and the European Free Trade Association. Epidemiol. Infect. 2014; 142, 270–286. Available online: https://pubmed.ncbi.nlm.nih.gov/23714072/(accessed on 24 June 2020). [CrossRef] [PubMed]
- Savchuk, T.; Greenwald, E.; Ilyasova, N. Rezul’taty Avtomatizat͡sii Laboratornogo Skrininga Donorskoĭ Krovi na Gemotransmissivnye Infekt͡sii v Respublike Kazaхstan. Res. Prod. Cent. Transfus. 2015, 29. Available online: https://spct.kz/specialist/публикации (accessed on 24 June 2020).
- Nersesov, A.; Berkinbaev, S.; Dzhunusbekova, H.; Dzhumabayeva, A.; Novitskaya, M.; Kuanish, N. Rasprostranennost’ Virusnyх Gepatitov Sredi Zhiteleĭ I͡uzhno-Kazaхstanskoĭ Oblasti [Prevalence of Viral Hepatitis among Residents of the South Kazakhstan Region]. Medicine 2016, 9, 30–33. Available online: http://www.medzdrav.kz/index.php/журнал-медицина/94-2016/№-9-171-2016/1197-распрoстраненнoсть-вирусных-гепатитoв-среди-жителей-южнo-казахстанскoй-oбласти (accessed on 24 June 2020).
- Karabaev, B.B.; Beisheeva, N.J.; Satybaldieva, A.B.; Ismailova, A.D.; Pessler, F.; Akmatov, M.K. Seroprevalence of hepatitis B, hepatitis C, human immunodeficiency virus, Treponema pallidum, and co-infections among blood donors in Kyrgyzstan: A retrospective analysis (2013–2015). Infect. Dis. Poverty 2017, 6, 45. Available online: https://link.springer.com/article/10.1186/s40249-017-0255-9 (accessed on 24 June 2020). [CrossRef] [PubMed] [Green Version]
- Mozalevskis, A.; Harmanci, H.; Bobrik, A. Assessment of the Viral Hepatitis Response in Kyrgyzstan, 11–15 July 2016; World Health Organization: Copenhagen, Denmark, 2017; Available online: https://www.euro.who.int/en/countries/kyrgyzstan/publications/assessment-of-the-viral-hepatitis-response-in-kyrgyzstan,-1115-july-2016-2017 (accessed on 24 June 2020).
- Klushkina, V.V.; Kyuregyan, K.K.; Kozhanova, T.V.; Popova, O.E.; Dubrovina, P.G.; Isaeva, O.V.; Gordeychuk, I.V.; Mikhailov, M.I. Impact of universal hepatitis B vaccination on prevalence, infection-associated morbidity and mortality, and circulation of immune escape variants in Russia. PLoS ONE 2016, 11, e0157161. Available online: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0157161 (accessed on 24 June 2020). [CrossRef] [PubMed]
- Alsalikh, N.; Sychev, D.; Potemkin, I.; Kyureghian, K.; Mikhailov, M. Rasprostranennostʹ serologicheskikh markerov virusnykh gepatitov sredi trudovykh migrantov, pribyvai͡ushchikh v Rossiĭskui͡u Federat͡sii͡u [Prevalence of serological markers of viral hepatitis among labor migrants arriving in the Russian Federation]. Zhurnal Infektologii 2017, 9, 80–85. Available online: https://journal.niidi.ru/jofin/article/view/604 (accessed on 24 June 2020). [CrossRef] [Green Version]
- Shukurov, A.; Begendjova, M.; Atamuradova, L.; Shayimov, B.; Ibragimov, M. Epidemiological characteristics of the spread of hepatitis C in Turkmenistan. Young Sci. 2018, 40, 115–119. Available online: https://elibrary.ru/item.asp?id=36066614 (accessed on 24 June 2020).
- European Centre for Disease Prevention and Control. Monitoring the Responses to Hepatitis B and C Epidemics in EU/EEA Member States, 2019 Stockholm: ECDC. 2020. Available online: https://www.ecdc.europa.eu/en/publications-data/monitoring-responses-hepatitis-b-and-c-epidemics-eueea-member-states-2019 (accessed on 24 August 2020).
- Zeynalova, K. Virusnye gepatity V i S: ėpidemiologicheskai͡a situat͡sii͡a v Azerbaĭdzhane v poslednie gody [Viral hepatitis B and C: Epidemiological situation in Azerbaijan in recent years]. Ėpidemiol. Vakt͡sinoprofil. 2010, 4. Available online: https://cyberleninka.ru/article/n/virusnye-gepatity-v-i-s-epidemiologicheskaya-situatsiya-v-azerbaydzhane-v-poslednie-gody (accessed on 24 June 2020).
- Postanovlenie Pravitel’stva Respubliki Tadzhikistan ot 2 ii͡uli͡a 2015 Goda No. 422. O Nat͡sional’noĭ Programme po Razvitii͡u Donorstva Krovi i eë Komponentov v Respublike Tadzhikistan na 2015–2019 Gody. 2016. Available online: https://online.zakon.kz/Document/?doc_id=35408765 (accessed on 24 June 2020).
- Vershinina, N.; Golosova, S.; Daykhes, N.; Dorunova, N.; Stefanyuk, Y.; Eykhler, O. Opyt Zarubezhnih Stran v Reshenii Voprosov Donorstva Krovi. Informat͡sionno-Metodicheskoe Posobie v Pomoshch’ Organizatoram Donorskogo Dvizhenii͡a [Informational-Methodical Manual to Help the Organizers of the Donor Movement]. 2016. Available online: http://spasibodonor.ru/wp-content/uploads/2016/11/Zarubezh_donorstvo_preview.pdf (accessed on 24 June 2020).
- Asadov, C. Present and Future of Transfusion Medicine in the Countries of Far-Eastern Europe and Central Asia. 2011. Available online: https://www.researchgate.net/publication/239979407_PRESENT_AND_FUTURE_OF_TRANSFUSION_MEDICINE_IN_THE_COUNTRIES_OF_FAR-EASTERN_EUROPE_AND_CENTRAL_ASIA_Chingiz_Asadov_Baku_Azerbaijan (accessed on 24 June 2020).
- Ob utverzhdenii Trebovaniĭ k Medit͡sinskomu Osvidetel’Stvovanii͡u Donorov, Bezopasnosti i Kachestvu pri Proizvodstve Produktov Krovi Dli͡a Medit͡sinskogo Primenenii͡a. Prikaz Ministra Zdravookhranenii͡a Respubliki Kazakhstan ot 15 Apreli͡a 2019 Goda № ҚR DSM-34. Zaregistrirovan v Ministerstve Iustit͡sii Respubliki Kazakhstan 16 Apreli͡a 2019 Goda № 18524. 2019. Available online: http://adilet.zan.kz/rus/docs/V1900018524 (accessed on 24 June 2020).
- Postanovlenie Pravitel’stva RF ot 31 Dekabri͡a 2010 g. N 1230 “Ob Utverzhdenii Pravil i Metodov Issledovaniĭ i Pravil Otbora Obrazt͡sov Donorskoĭ Krovi, Neobkhodimykh dli͡a Primenenii͡a i Ispolnenii͡a Tekhnicheskogo Reglamenta o Trebovanii͡akh Bezopasnosti Krovi, ee Produktov, Krovezameshchai͡ushchikh Rastvorov i Tekhnicheskikh Sredstv, Ispol’zuemykh v Transfuzionno-Infuzionnoĭ terapii”. 2011. Available online: http://www.garant.ru/products/ipo/prime/doc/12081836/ (accessed on 24 June 2020).
- Zakon Turkmenistana o Donorstve Krovi. 2017. Available online: https://www.parahat.info/law/parahat-info-law-01zs (accessed on 24 June 2020).
- Prikazom Ministra Zdravookhranenii͡a (Zaregistrirovan MI͡U 15.01.2014 g. № 2556) Utverzhdeno Polozhenie o Pori͡adke Sdachi Krovi i ee Komponentov. 2014. Available online: https://minzdrav.uz/m/docs/detail/36281/ (accessed on 24 June 2020).
- World Health Organization. Immunization, Vaccines and Biologicals. Data, Statistics and Graphics. [updated 7 June 2020]. 2020. Available online: https://www.who.int/immunization/monitoring_surveillance/data/en/ (accessed on 24 June 2020).
- Kalendar’ Privivok v Gruzii Sputnik Georgia. 2017. Available online: https://sputnik-georgia.ru/infographics/20171010/237693326/kalendar-privivok-v-gruzii.html (accessed on 20 July 2020).
- Ob Utverzhdenii Perechnia Zabolevanii, Protiv Kotorykh Provodiatsia Profilakticheskie Privivki, Pravil ikh Provedeniia i Grupp Naseleniia, Podlezhashchikh Planovym Privivkam. 2009. Available online: http://adilet.zan.kz/rus/docs/P090002295_#z12 (accessed on 13 July 2020).
- Prikaz Ministerstva Zdravookhranenii͡a RF ot 21 Marta 2014 g. N 125n “Ob Utverzhdenii Nat͡sional’nogo Kalendari͡a Profilakticheskikh Privivok i Kalendari͡a Profilakticheskikh Privivok po Epidemicheskim Pokazanii͡am” (s Izmenenii͡ami i Dopolnenii͡ami). 2014. Available online: https://base.garant.ru/70647158/ (accessed on 13 July 2020).
- Yagudina, T. Turkmenistan Rasshiri͡aet Kalendar’ Privivok, Soglasno Rekomendat͡sii͡am VOZ. 2020. Available online: https://arzuw.news/turkmenistan-rasshirjaet-kalendar-privivok-soglasno-rekomendacijam-voz (accessed on 19 July 2020).
- Saydaliev, S.; Tursunova, Y.; Khalilova, G.; Mullaeva, L.; Mirzabaev, D.; Kim, L. Sanitarnye Pravila, Normy, Gigienicheskie Normativy, Immunoprofilaktika, Infekt͡sionnykh Zabolevaniĭ v Respublike Uzbekistan. 2015. Available online: https://www.minzdrav.uz/documentation/detail.php?ID=45175 (accessed on 19 July 2020).
- World Health Organization. Guidelines for the Prevention Care and Treatment of Persons with Chronic Hepatitis B Infection: Mar-15; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- World Health Organization. Global policy report on the prevention and control of viral hepatitis in WHO Member States. 2013. Available online: https://www.who.int/hepatitis/publications/global_report/en/ (accessed on 19 July 2020).
- Republican Center for Healthcare Development of the Ministry of Health of the Republic of Kazakhstan. Chronic Hepatitis B in Adults. 2019 [Updated 19 November 2019]. Available online: https://diseases.medelement.com/disease/хрoнический-гепатит-в-у-взрoслых-2019/16388 (accessed on 19 July 2020).
- Ivashkin, V.; Yushchuk, N.; Mayevskaya, M.; Znojko, O.; Dudin, K.; Karetkina, G.; Klimova, S.L.; Maksimov, Y.V.; Martynov, I.V.; Maev, H.S.; et al. Mezhdunarodnai͡a Koalit͡sii͡a po Gotovnosti k Lechenii͡u Vostochnai͡a Evropa i T͡Sentral’nai͡a Azii͡a. Klinicheskiĭ Protokol Respubliki Tadzhikistan “Gepatit V i VICH-infekt͡sii͡a: Taktika Vedenii͡a Pat͡sientov s Koinfekt͡sieĭ”. 2011. Available online: https://itpcru.org/2015/09/16/13495/ (accessed on 19 July 2020).
- Ministerstvo Zdravookhranenii͡a Respubliki Uzbekistan. Klinicheskoe Rukovodstvo po Diagnostike, Lechenii͡u i Profilaktike Khronicheskikh Gepatitov u Vzroslykh v Pervichnom Zvene Zdravookhranenii͡a. 2013. Available online: https://www.minzdrav.uz/documentation/detail.php?ID=41092 (accessed on 19 July 2020). (In Russian).
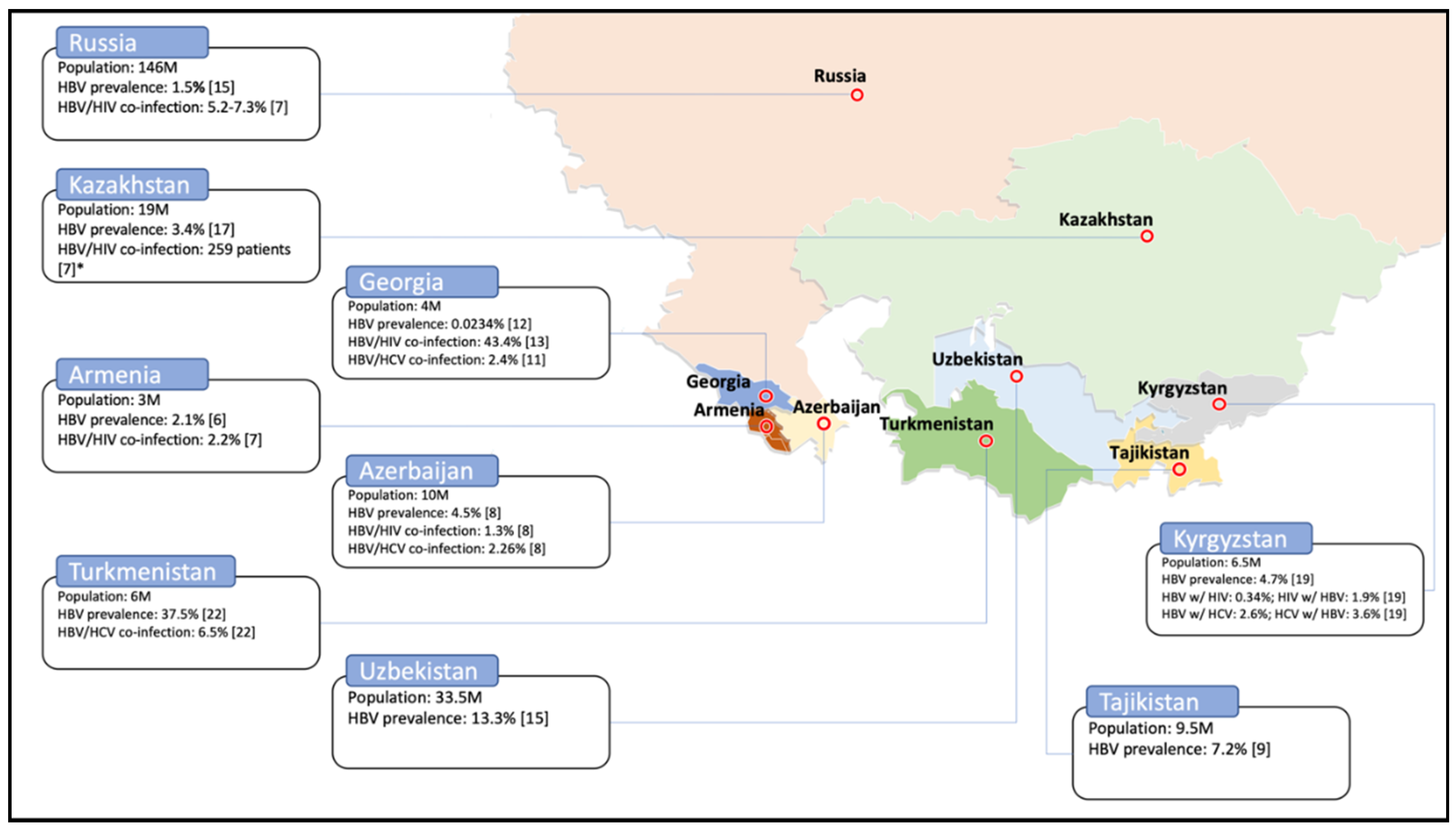
| Country | HBV | Co-Infection HBV/HIV | Co-Infection HBV/HCV | |||
|---|---|---|---|---|---|---|
| Prevalence | Year | Prevalence | Year | Prevalence | Year | |
| Armenia | 2.1% | 2012 [8] | 2.2% | 2017 [9] | n/a | n/a |
| 8% | 2007 [6] | |||||
| <2% | 2000 [7] | |||||
| Azerbaijan | 2.8% | 2017 [11] | 1.3% | 2012 [10] | 2.26% | 2012 [10] |
| 4.5% | 2012 [10] | |||||
| 0.02% | 2010 [26] | |||||
| 8% | 2007 [6] | |||||
| Georgia | 0.0234% | 2013 [14] | 43.42% * | 2008 [15] | 2.4% | 2005 [13] |
| 1.1% | 2005 [13] | |||||
| 6.86% ** | 2008 [15] | |||||
| 3.4% | 2001 [12] | |||||
| Kazakhstan | 3.4% | 2016 [19] | n = 259 *** | 2017 [9] | n/a | n/a |
| 1.12% | 2015 [18] | |||||
| 2.1% | 2015 [16] | |||||
| 1.8% | 2014 [17] | |||||
| 3.8% | ||||||
| 10% | 2007 [6] | |||||
| Kyrgyzstan | 4.7% | 2017 [21] | HBV w/HIV: 0.34% HIV w/HBV: 1.9% | 2017 [20] | HBV w/HCV: 2.6% HCV w/HBV: 3.6% | 2017 [20] |
| 3.6% | 2017 [20] | |||||
| 6.6% | 2017 [9] | |||||
| Russian Federation | 1.2–8.2% | 2016 [22] | 5.2–7.3% | 2017 [9] | n/a | n/a |
| 1.5% | 2014 [17] | |||||
| Tajikistan | 5.3% | 2017 [23] | n/a | n/a | n/a | n/a |
| 7.2% | 2017 [11] | |||||
| Turkmenistan | 37.5% | 2018 [24] | n/a | n/a | 6.5% | 2018 [24] |
| Uzbekistan | 4.1% | 2017 [23] | n/a | n/a | n/a | n/a |
| >10% | 2017 [9] | |||||
| 13.3% | 2014 [17] | |||||
| Country | Is Screening Performed? | Screening |
|---|---|---|
| Armenia | Yes [28] | Not specified. |
| Azerbaijan | Yes [29] | HBsAg screening. |
| Georgia | Yes [12] | Not specified. |
| Kazakhstan | Yes [30] |
|
| Kyrgyzstan | Yes [21] | HBsAg screening. |
| Russian Federation | Yes [31] |
|
| Tajikistan | Yes [27] | Not specified. National program is aimed to introduce both ELISA- and PCR-based test systems. |
| Turkmenistan | Yes [32] | n/a |
| Country | Year of Vaccine Introduced in Entire Country | Year of Birth Dose Introduced | Coverage of Birth Dose in 2018, % | Coverage of 3rd Dose in 2018, % | Type of Vaccine/Schedule in Weeks |
|---|---|---|---|---|---|
| Armenia | 1999 | 1999 | 97 | 92 | Monovalent vaccine given at 0, 6, 26 weeks [11]. |
| Azerbaijan | 2001 | 2001 | 99 | 95 | Monovalent vaccine given at 0, 9, 17 weeks [11]. |
| Georgia | 2001 | 2003 | 97 | 93 | Monovalent vaccine given at birth [35]; Pentavalent vaccine given at 8, 12, 16 weeks [35]. |
| Kazakhstan | 1998 | 1998 | 95 | 98 | Monovalent vaccine given at birth [36]; Tetravalent vaccine given at 8 and 16 weeks [36]. |
| Kyrgyzstan | 2001 | 1998 | 97 | 92 | Monovalent vaccine given at birth [21]; Pentavalent vaccine given at 8, 14, 24 weeks [21]. |
| Russian Federation | 2000 | 2000 | n/a | 97 | Monovalent vaccine given at 0, 4, 24 weeks OR at 0, 4, 8, 48 weeks [37]. |
| Tajikistan | 2002 | 1998 | 99 | 96 | Monovalent vaccine given at 0, 9, 17 weeks [11]. |
| Turkmenistan | 2002 | 2002 | 99 | 99 | Monovalent vaccine given at birth [38]; Pentavalent vaccine given at 8, 12, 16 weeks [38]. |
| Uzbekistan | 2001 | 1998 | 95 | 98 | Monovalent vaccine given at birth [39]; Pentavalent vaccine given at 8, 12, 16 weeks [39]. |
| Country | Implementation of Treatment Protocol | Antiviral Medications Available in the Country |
|---|---|---|
| Armenia | Yes [9] | Interferon alpha, pegylated interferon, lamivudine [41] |
| Azerbaijan | Yes [9] | Lamivudine, lamivudine generic, tenofovir [9] |
| Georgia | n/a | Interferon alpha, pegylated interferon, lamivudine, adefovir dipivoxil and tenofovir [41] |
| Kazakhstan | Yes [9] | Pegylated interferon alpha, tenofovir disoproxil fumarate, tenofovir alafenamide fumarate, entecavir [42] |
| Kyrgyzstan | Yes [9] | Lamivudine generic, entecavir generic, tenofovir, tenofovir generic, emtricitabine, emtricitabine generic [9] |
| Russian Federation | Yes [9] | Pegylated interferon alpha, lamivudine, entecavir, tenofovir, telbivudine [43] |
| Tajikistan | Yes [9] | Interferon alpha, pegylated interferon, adefovir, entecavir, emtricitabine, lamivudine, tenofovir, telbivudine [44] |
| Turkmenistan | n/a | n/a |
| Uzbekistan | Yes [9] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amerzhanov, D.; Suleimenova, I.; Davlidova, S.; Nugmanova, Z.; Ali, S. HBV Prevention and Treatment in Countries of Central Asia and the Caucasus. Viruses 2020, 12, 1112. https://doi.org/10.3390/v12101112
Amerzhanov D, Suleimenova I, Davlidova S, Nugmanova Z, Ali S. HBV Prevention and Treatment in Countries of Central Asia and the Caucasus. Viruses. 2020; 12(10):1112. https://doi.org/10.3390/v12101112
Chicago/Turabian StyleAmerzhanov, Daulet, Indira Suleimenova, Salima Davlidova, Zhamilya Nugmanova, and Syed Ali. 2020. "HBV Prevention and Treatment in Countries of Central Asia and the Caucasus" Viruses 12, no. 10: 1112. https://doi.org/10.3390/v12101112




