Pharmacokinetic Variability Drives Palbociclib-Induced Neutropenia in Metastatic Breast Cancer Patients: Drug–Drug Interactions Are the Usual Suspects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Trial Design and Patients
2.2. Endpoint Analysis (Palbociclib Exposure–Toxicity Relationship)
2.3. Pharmacokinetics
2.4. Exposure–Toxicity Analysis
2.5. Exposure-DDI Relationship Analysis
2.6. Statistical Analysis
3. Results
3.1. Patients
3.2. Clinical–Biological Data and Palbociclib-Induced Toxicity
3.3. Palbociclib Pharmacokinetics and Clinicopathological Features
3.4. Palbociclib Exposure and Co-Medication
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sun, W.; O’Dwyer, P.J.; Finn, R.S.; Ruiz-Garcia, A.; Shapiro, G.I.; Schwartz, G.K.; DeMichele, A.; Wang, D. Characterization of Neutropenia in Advanced Cancer Patients Following Palbociclib Treatment Using a Population Pharmacokinetic-Pharmacodynamic Modeling and Simulation Approach. J. Clin. Pharmacol. 2017, 57, 1159–1173. [Google Scholar] [CrossRef] [PubMed]
- Shitara, K.; Matsuo, K.; Takahari, D.; Yokota, T.; Shibata, T.; Ura, T.; Ito, S.; Tajika, M.; Kawai, H.; Muro, K. Neutropenia as a prognostic factor in advanced gastric cancer patients undergoing second-line chemotherapy with weekly paclitaxel. Ann. Oncol. 2010, 21, 2403–2409. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhong, X.; Ma, J.; Sun, W.; Han, H.S.; Soliman, H.H.; Loftus, L.S.; Costa, R.L.B.; Armaghani, A.J.; Soyano-Muller, A.E.; et al. Real-world benefit of combination palbociclib and endocrine therapy for metastatic breast cancer and correlation with neutropenia. Cancer Med. 2021, 10, 7665–7672. [Google Scholar] [CrossRef] [PubMed]
- Thill, M.; Schmidt, M. Management of adverse events during cyclin-dependent kinase 4/6 (CDK4/6) inhibitor-based treatment in breast cancer. Ther. Adv. Med. Oncol. 2018, 10, 175883591879332. [Google Scholar] [CrossRef] [Green Version]
- Sun, W.; Wang, D.D. A Population Pharmacokinetic (Pk) Analysis of Palbociclib (Pd-0332991) in Patients (Pts) with Advanced Solid Tumors. Ann. Oncol. 2014, 25, iv154. [Google Scholar] [CrossRef]
- Yu, Y.; Loi, C.-M.; Hoffman, J.; Wang, D. Physiologically Based Pharmacokinetic Modeling of Palbociclib: PBPK Modeling of Palbociclib. J. Clin. Pharmacol. 2017, 57, 173–184. [Google Scholar] [CrossRef]
- Hu, W.; Sung, T.; Jessen, B.A.; Thibault, S.; Finkelstein, M.B.; Khan, N.K.; Sacaan, A.I. Mechanistic Investigation of Bone Marrow Suppression Associated with Palbociclib and its Differentiation from Cytotoxic Chemotherapies. Clin. Cancer Res. 2016, 22, 2000–2008. [Google Scholar] [CrossRef] [Green Version]
- DeMichele, A.; Clark, A.S.; Tan, K.S.; Heitjan, D.F.; Gramlich, K.; Gallagher, M.; Lal, P.; Feldman, M.; Zhang, P.; Colameco, C.; et al. CDK 4/6 inhibitor palbociclib (PD0332991) in Rb+ advanced breast cancer: Phase II activity, safety, and predictive biomarker assessment. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2015, 21, 995–1001. [Google Scholar] [CrossRef] [Green Version]
- Cazzaniga, M.E.; Danesi, R.; Girmenia, C.; Invernizzi, P.; Elvevi, A.; Uguccioni, M.; on behalf of NetworkER+. Management of toxicities associated with targeted therapies for HR-positive metastatic breast cancer: A multidisciplinary approach is the key to success. Breast Cancer Res. Treat. 2019, 176, 483–494. [Google Scholar] [CrossRef] [Green Version]
- Widmer, N.; Bardin, C.; Chatelut, E.; Paci, A.; Beijnen, J.; Levêque, D.; Veal, G.; Astier, A. Review of therapeutic drug monitoring of anticancer drugs part two—Targeted therapies. Eur. J. Cancer 2014, 50, 2020–2036. [Google Scholar] [CrossRef]
- Bellet, M.; Ahmad, F.; Villanueva, R.; Valdivia, C.; Palomino-Doza, J.; Ruiz, A.; Gonzalez, X.; Adrover, E.; Azaro, A.; Valls-Margarit, M.; et al. Palbociclib and ribociclib in breast cancer: Consensus workshop on the management of concomitant medication. Ther. Adv. Med. Oncol. 2019, 11, 175883591983386. [Google Scholar] [CrossRef]
- Sun, W.; Klamerus, K.J.; Yuhas, L.M.; Pawlak, S.; Plotka, A.; O’Gorman, M.; Kirkovsky, L.; Kosa, M.; Wang, D. Impact of Acid-Reducing Agents on the Pharmacokinetics of Palbociclib, a Weak Base with pH-Dependent Solubility, with Different Food Intake Conditions. Clin. Pharmacol. Drug Dev. 2017, 6, 614–626. [Google Scholar] [CrossRef]
- Momper, J.D.; Yang, J.; Kerr, J.; Saunders, I.; Smith, J.; Shah, M.M. Interaction Between Cyclosporine and Palbociclib in a Renal Transplant Patient: Case Report and Pharmacokinetic Perspective. J. Pharm. Pract. 2020, 33, 912–914. [Google Scholar] [CrossRef]
- Gowarty, J.L.; Herrington, J.D. Verapamil as a culprit of palbociclib toxicity. J. Oncol. Pharm. Pract. Off. Publ. Int. Soc. Oncol. Pharm. Pract. 2019, 25, 743–746. [Google Scholar] [CrossRef]
- Roncato, R.; Angelini, J.; Pani, A.; Cecchin, E.; Sartore-Bianchi, A.; Siena, S.; De Mattia, E.; Scaglione, F.; Toffoli, G. CDK4/6 Inhibitors in Breast Cancer Treatment: Potential Interactions with Drug, Gene, and Pathophysiological Conditions. Int. J. Mol. Sci. 2020, 21, 76350. [Google Scholar] [CrossRef]
- Leenhardt, F.; Gracia, M.; Perrin, C.; Muracciole-Bich, C.; Marion, B.; Roques, C.; Alexandre, M.; Firmin, N.; Pouderoux, S.; Mbatchi, L.; et al. Liquid chromatography–tandem mass spectrometric assay for the quantification of CDK4/6 inhibitors in human plasma in a clinical context of drug-drug interaction. J. Pharm. Biomed. Anal. 2020, 188, 113438. [Google Scholar] [CrossRef]
- Diéras, V.; Rugo, H.S.; Schnell, P.; Gelmon, K.; Cristofanilli, M.; Loi, S.; Colleoni, M.; Lu, D.R.; Mori, A.; Gauthier, E.; et al. Long-term Pooled Safety Analysis of Palbociclib in Combination with Endocrine Therapy for HR+/HER2- Advanced Breast Cancer. JNCI J. Natl. Cancer Inst. 2019, 111, 419–430. [Google Scholar] [CrossRef] [Green Version]
- Diéras, V.; Harbeck, N.; Joy, A.A.; Gelmon, K.; Ettl, J.; Verma, S.; Lu, D.R.; Gauthier, E.; Schnell, P.; Mori, A.; et al. Palbociclib with Letrozole in Postmenopausal Women with ER+/HER2− Advanced Breast Cancer: Hematologic Safety Analysis of the Randomized PALOMA-2 Trial. Oncologist 2019, 24, 1514–1525. [Google Scholar] [CrossRef] [Green Version]
- Zheng, J.; Yu, Y.; Durairaj, C.; Diéras, V.; Finn, R.S.; Wang, D.D. Impact of Dose Reduction on Efficacy: Implications of Exposure-Response Analysis of Palbociclib. Target Oncol. 2020, 16, 69–76. [Google Scholar] [CrossRef]
- Mukai, H.; Shimizu, C.; Masuda, N.; Ohtani, S.; Ohno, S.; Takahashi, M.; Yamamoto, Y.; Nishimura, R.; Sato, N.; Ohsumi, S.; et al. Palbociclib in combination with letrozole in patients with estrogen receptor–positive, human epidermal growth factor receptor 2–negative advanced breast cancer: PALOMA-2 subgroup analysis of Japanese patients. Int. J. Clin. Oncol. 2019, 24, 274–287. [Google Scholar] [CrossRef] [Green Version]
- Finn, R.S.; Rugo, H.S.; Gelmon, K.A.; Cristofanilli, M.; Colleoni, M.; Loi, S.; Colleoni, M.; Lu, D.R.; Mori, A.; Gauthier, E.; et al. Long-Term Pooled Safety Analysis of Palbociclib in Combination with Endocrine Therapy for Hormone Receptor-Positive/Human Epidermal Growth Factor Receptor 2-Negative Advanced Breast Cancer: Updated Analysis with up to 5 Years of Follow-Up. Oncologist 2021, 26, e749–e755. [Google Scholar] [CrossRef] [PubMed]
- Kanbayashi, Y.; Sakaguchi, K.; Ishikawa, T.; Takayama, K.; Taguchi, T. Predictors for development of palbociclib-induced neutropenia in breast cancer patients as determined by ordered logistic regression analysis. Sci. Rep. 2021, 11, 20055. [Google Scholar] [CrossRef] [PubMed]
- Marouille, A.L.; Petit, E.; Kaderbhaï, C.; Desmoulins, I.; Hennequin, A.; Mayeur, D.; Fumet, J.-D.; Ladoire, S.; Tharin, Z.; Ayati, S.; et al. Pharmacokinetic/Pharmacodynamic Model of Neutropenia in Real-Life Palbociclib-Treated Patients. Pharmaceutics 2021, 13, 1708. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Hoffman, J.; Plotka, A.; O’Gorman, M.; Shi, H.; Wang, D. Palbociclib (PD-0332991) pharmacokinetics in subjects with impaired renal function. Cancer Chemother. Pharmacol. 2020, 86, 701–710. [Google Scholar] [CrossRef]
- Molenaar-Kuijsten, L.; Braal, C.L.; Groenland, S.L.; Vries, N.; Rosing, H.; Beijnen, J.H.; Koolen, S.L.W.; Vulink, A.J.E.; van Dongen, M.G.J.; Mathijssen, R.H.J.; et al. Effects of the Moderate CYP3A4 Inhibitor Erythromycin on the Pharmacokinetics of Palbociclib: A Randomized Crossover Trial in Patients with Breast Cancer. Clin. Pharmacol. Ther. 2021, 111, 477–484. [Google Scholar] [CrossRef]
- Del Re, M.; Omarini, C.; Diodati, L.; Palleschi, M.; Meattini, I.; Crucitta, S.; Lorenzini, G.; Isca, C.; Fontana, A.; Livi, L.; et al. Drug–drug interactions between palbociclib and proton pump inhibitors may significantly affect clinical outcome of metastatic breast cancer patients. ESMO Open 2021, 6, 100231. [Google Scholar] [CrossRef]
- Mir, O.; Touati, N.; Lia, M.; Litière, S.; Le Cesne, A.; Sleijfer, S.; Blay, J.-Y.; Leahy, M.; Young, R.; Mathijssen, R.H.J.; et al. Impact of Concomitant Administration of Gastric Acid-Suppressive Agents and Pazopanib on Outcomes in Soft-Tissue Sarcoma Patients Treated within the EORTC 62043/62072 Trials. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2019, 25, 1479–1485. [Google Scholar] [CrossRef] [Green Version]


| Number of Palbociclib Cycles (n) | |
|---|---|
| n | 58 |
| Mean (SD) | 8.9 (3.6) |
| Median (Q1;Q3) | 10.5 (6.0; 12.0) |
| Duration of treatment (months) | |
| n | 58 |
| Mean (SD) | 8.9 (3.8) |
| Median (Q1;Q3) | 11.0 (6.4; 11.3) |
| Dose reduction (n) | |
| At least one dose reduction | |
| No | 39 (67.2%) |
| Yes | 19 (32.8%) |
| If yes: | |
| For hematologic toxicity | 17 (89.5%) |
| For other toxicity | 2 (10.5%) |
| Treatment interruption (n) | |
| At least one treatment interruption | |
| No | 46 (79.3%) |
| Yes | 12 (20.7%) |
| If yes: | |
| For hematologic toxicity | 3 (25.0%) |
| For other toxicity | 9 (75.0%) |
| Neutropenia during the first two cycles (n) | |
| Grade during the first two cycles | |
| Grade 0 | 17 (29.3%) |
| Grade 1 | 1 (1.7%) |
| Grade 2 | 1 (1.7%) |
| Grade 3 | 34 (58.7%) |
| Grade 4 | 5 (8.6%) |
| Incidence of grade 3+ neutropenia during the first two cycles | |
| No | 19 (32.8%) |
| Yes | 39 (67.2%) |
| Variable | Nb Evt/N | Univariable Analysis | Multivariable Analysis | ||||
|---|---|---|---|---|---|---|---|
| OR | 95% IC | p Value ‡ | OR | 95% IC | p Value ‡ | ||
| Clinical variables | |||||||
| Age | p = 0.928 | ||||||
| 5 years increase | 39/58 | 0.99 | (0.80; 1.22) | ||||
| BMI (kg/m²) | p = 0.038 | ||||||
| 1-unit increase | 37/56 | 1.14 | (1.00; 1.31) | ||||
| Missing | 2 | ||||||
| Previous treatment | p = 0.461 | ||||||
| No | 14/19 | 1.00 | Ref | ||||
| Yes | 25/39 | 0.64 | (0.19; 2.14) | ||||
| Laboratory data | |||||||
| Lymphocytes (109/L) | p = 0.101 | ||||||
| 1-unit increase | 39/58 | 0.64 | (0.37; 1.12) | ||||
| Leukocytes (109/L) | p = 0.001 | ||||||
| 1-unit increase | 39/58 | 0.61 | (0.44; 0.84) | ||||
| Neutrophils (109/L) | p = 0.007 | p = 0.002 | |||||
| 1-unit increase | 39/58 | 0.62 | (0.42; 0.92) | 0.56 | (0.36; 0.86) | ||
| Hemoglobin (g/dL) | p = 0.103 | ||||||
| 1-unit increase | 39/57 | 1.43 | (0.92; 2.25) | ||||
| Bilirubin (g/dL) | p = 0.201 | ||||||
| 1-unit increase | 36/55 | 1.11 | (0.94; 1.31) | ||||
| Kidney clearance (mL/min/1.73 m²) | p = 0.538 | ||||||
| 10-unit increase | 39/58 | 0.92 | (0.71; 1.19) | ||||
| Treatment data at D15C1 | |||||||
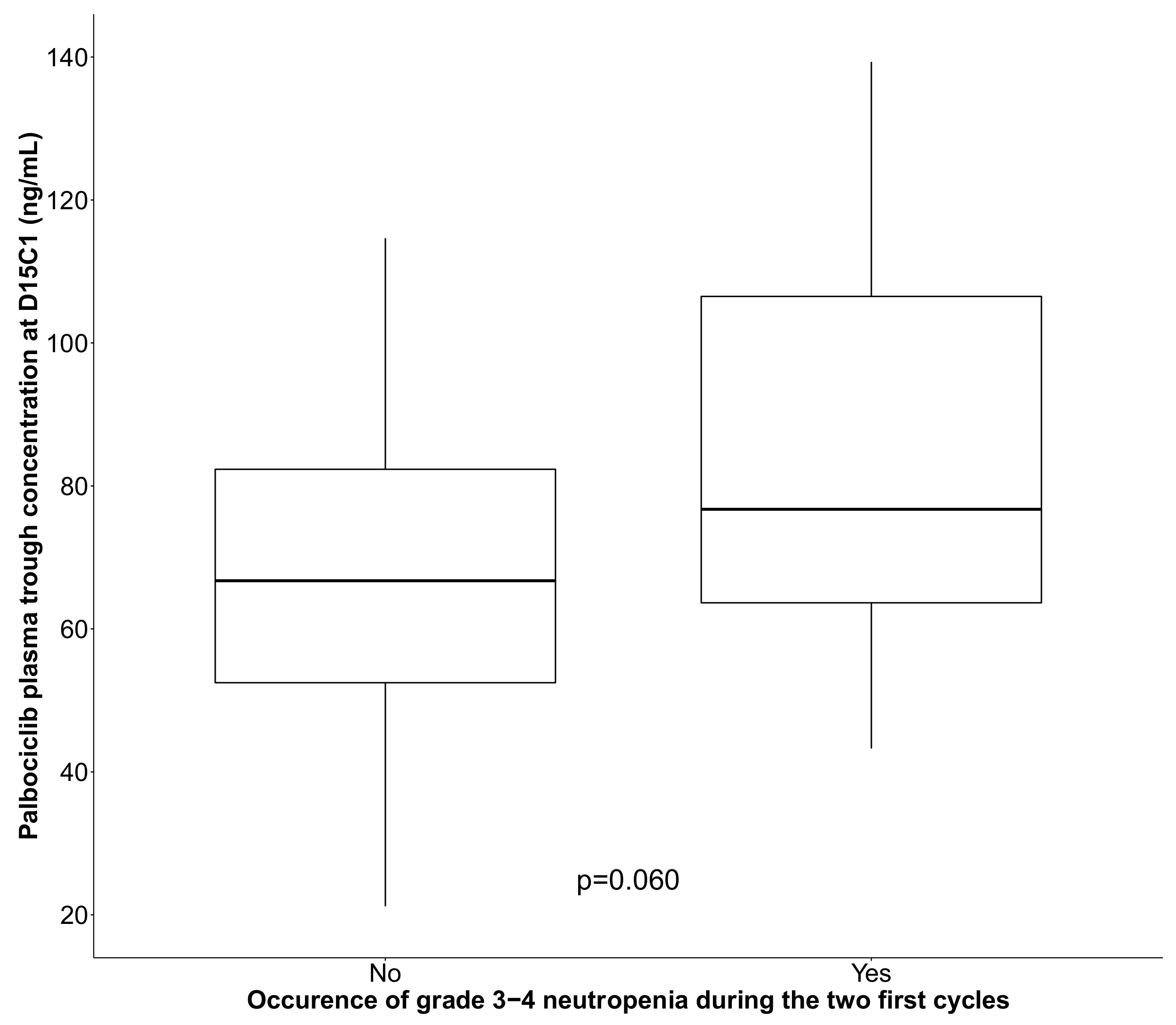
| Palbociclib Ctrough | p = 0.031 | p = 0.008 | |||||
| 10 unit increase | 35/54 | 1.28 | (1.01; 1.64) | 1.42 | (1.06; 1.90) | ||
| CYP3A4 and/or p-gp inhibitor | p = 0.318 | ||||||
| No | 14/19 | 1.00 | Ref | ||||
| Yes | 25/39 | 0.55 | (0.17; 1.77) | ||||
| Antacids | p = 0.183 | ||||||
| No | 28/40 | 1.00 | Ref | ||||
| Yes | 7/14 | 0.43 | (0.12; 1.49) | ||||
| Palbociclib Ctrough | All | Test | ||
|---|---|---|---|---|
| ≤74 ng/mL | >74 ng/mL | |||
| n = 27 | n = 27 | n = 54 | ||
| Sociodemographic and clinical variables at inclusion | ||||
| Age (years) | p = 0.002 | |||
| N | 27 | 27 | 54 | |
| Mean (SD) | 57.1 (12.9) | 67.8 (12.3) | 62.5 (13.6) | |
| Median (Q1;Q3) | 57.0 (48.0; 67.0) | 71.0 (64.0; 76.0) | 65.5 (55.0; 74.0) | |
| Age (Median) | p = 0.003 | |||
| ≤66 years | 20 (74.1%) | 9 (33.3%) | 29 (53.7%) | |
| >66 years | 7 (25.9%) | 18 (66.7%) | 25 (46.3%) | |
| BMI (kg/m²) | p = 0.963 | |||
| N | 25 | 27 | 52 | |
| Mean (SD) | 25.5 (4.9) | 25.5 (4.5) | 25.5 (4.7) | |
| Median (Q1;Q3) | 25.4 (22.1; 29.0) | 24.6 (22.5; 28.1) | 25.0 (22.2; 28.5) | |
| Missing | 2 | 0 | 2 | |
| Alcohol consumption | p = 1.000 | |||
| Non consumer | 23 (85.2%) | 23 (85.2%) | 46 (85.2%) | |
| Former consumer | 0 (0.0%) | 1 (3.7%) | 1 (1.9%) | |
| Consumer | 4 (14.8%) | 3 (11.1%) | 7 (13.0%) | |
| Tobacco consumption | p = 0.322 | |||
| Non-smoker | 17 (63.0%) | 22 (81.5%) | 39 (72.2%) | |
| Former smoker | 5 (18.5%) | 2 (7.4%) | 7 (13.0%) | |
| Smoker | 5 (18.5%) | 3 (11.1%) | 8 (14.8%) | |
| Biological variables at inclusion | ||||
| Creatinine (μmol/L) | p = 0.166 | |||
| N | 27 | 27 | 54 | |
| Mean (SD) | 66.9 (15.9) | 70.1 (13.4) | 68.5 (14.7) | |
| Median (Q1;Q3) | 63.0 (58.0; 70.0) | 66.0 (62.3; 79.0) | 64.5 (59.0; 74.3) | |
| Kidney clearance (ml/min/1.73 m2) | p = 0.017 | |||
| N | 27 | 27 | 54 | |
| Mean (SD) | 93.6 (24.2) | 80.3 (18.2) | 87.0 (22.3) | |
| Median (Q1;Q3) | 96.0 (87.0; 103.0) | 81.0 (67.0; 96.0) | 88.5 (70.0; 100.0) | |
| Albumin (g/L) | p = 0.040 | |||
| N | 21 | 25 | 46 | |
| Mean (SD) | 43.6 (4.5) | 41.3 (4.1) | 42.3 (4.6) | |
| Median (Q1;Q3) | 43.0 (41.7; 47.0) | 42.0 (39.0; 43.5) | 42.0 (40.0; 45.0) | |
| Missing | 6 | 2 | 8 | |
| Variable | n = 52 | |
|---|---|---|
| Coefficient | 95% IC | |
| CYP3A4/P-gp inhibitors | p = 0.035 | |
| No | 1.00 | Ref |
| Yes | 0.22 | (0.01; 0.44) |
| Antacids | p = 0.036 | |
| No | 1.00 | Ref |
| Yes | −0.23 | (−0.46; −0.01) |
| Body surface area at D15C1 | p = 0.787 | |
| 0.5 m² increase | −0.03 | (−0.31; 0.24) |
| Age | p = 0.146 | |
| 5 years increase | 0.03 | (−0.01; 0.06) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leenhardt, F.; Fiteni, F.; Gauthier, L.; Alexandre, M.; Guiu, S.; Firmin, N.; Pouderoux, S.; Viala, M.; Lossaint, G.; Gautier, C.; et al. Pharmacokinetic Variability Drives Palbociclib-Induced Neutropenia in Metastatic Breast Cancer Patients: Drug–Drug Interactions Are the Usual Suspects. Pharmaceutics 2022, 14, 841. https://doi.org/10.3390/pharmaceutics14040841
Leenhardt F, Fiteni F, Gauthier L, Alexandre M, Guiu S, Firmin N, Pouderoux S, Viala M, Lossaint G, Gautier C, et al. Pharmacokinetic Variability Drives Palbociclib-Induced Neutropenia in Metastatic Breast Cancer Patients: Drug–Drug Interactions Are the Usual Suspects. Pharmaceutics. 2022; 14(4):841. https://doi.org/10.3390/pharmaceutics14040841
Chicago/Turabian StyleLeenhardt, Fanny, Frédéric Fiteni, Ludovic Gauthier, Marie Alexandre, Séverine Guiu, Nelly Firmin, Stéphane Pouderoux, Marie Viala, Gerald Lossaint, Chloé Gautier, and et al. 2022. "Pharmacokinetic Variability Drives Palbociclib-Induced Neutropenia in Metastatic Breast Cancer Patients: Drug–Drug Interactions Are the Usual Suspects" Pharmaceutics 14, no. 4: 841. https://doi.org/10.3390/pharmaceutics14040841







