Abstract
Nanofibers are frequently encountered in daily life as a modern material with a wide range of applications. The important advantages of production techniques, such as being easy, cost effective, and industrially applicable are important factors in the preference for nanofibers. Nanofibers, which have a broad scope of use in the field of health, are preferred both in drug delivery systems and tissue engineering. Due to the biocompatible materials used in their construction, they are also frequently preferred in ocular applications. The fact that they have a long drug release time as a drug delivery system and have been used in corneal tissue studies, which have been successfully developed in tissue engineering, stand out as important advantages of nanofibers. This review examines nanofibers, their production techniques and general information, nanofiber-based ocular drug delivery systems, and tissue engineering concepts in detail.
1. Introduction
Today, biopharmaceutics is increasingly using nanotechnology and nanoscience approaches [1]. Nanotechnology focuses on the creation, characterization, and application of nanomaterials. Nanomaterials are classified into four main categories: zero-dimensional (nanoparticles), one-dimensional (nanofibers), two-dimensional (nanofilms), and three-dimensional (polycrystals) nanomaterials. One-dimensional (1D) nanomaterials have received more attention because of their unique features, which include large surface area, small diameter, and high aspect ratio. The unique characteristics of 1D nanomaterials lead to applications in many areas, such as drug delivery, chemical and biological sensors, supercapacitors, and tissue engineering. As a result, the controlled synthesis of 1D nanomaterials is required for progress in new scientific research. Carbon nanotubes, nanobelts/nanowires, and nanofibers have been some of the 1D nanomaterials attracting much attention in nanotechnology applications [2,3].
Nanofibers have attracted attention among all of these alternatives due to their benefits. Nanofibers have recently been used in drug delivery applications due to their low preparation costs, ability to use a wide range of polymers, and ease of creation using different processes [4,5]. In addition, nanofibers have high porosities, large surface areas, adjustable mechanical properties, and changeable morphologies. These parameters can be customized based on the drug delivery conditions and requirements of various applications. Nanofibers are also a good candidate for tissue engineering due to their extracellular matrix (ECM)-like structure [6].
Nanofiber-based drug delivery systems have opened new opportunities in drug delivery. Nanofibers have been encapsulated with different therapeutic agents, such as small molecular medicines, and biological molecules, such as antibiotics, proteins, DNA, RNA, growth factors, and living cells [7,8]. Additionally, nanofibers can be used for topical, transdermal, and oral drug delivery, while short nanofibers can be injected locally into diseased areas. Furthermore, electrospun nanofibers provide significant benefits in terms of controlling drug release rates by modifying their composition (e.g., hydrophobic and hydrophilic materials) and structure (e.g., homogenous structure, core-sheath structure, and multilayered structure) [9,10,11]. Recently, it has been proven that targeted drug delivery systems can be prepared with stimuli-responsive nanofibers [12]. Thanks to their widespread use, nanofibers have become one of the most frequently studied subjects in the ocular field.
The eye, which has a unique anatomy, is the only vital organ in direct contact with the outside world. It contains many natural barriers to preserve its structural integrity. These anatomical barriers and physiology protect the eye from foreign substances and objects and act as rate-limiting steps for many drugs [13,14]. Therefore, developing ocular drug delivery systems and new-generation ocular tissues has always been a challenging area of interest to pharmaceutical technologists and tissue engineers. Nanofibers are one of the promising approaches to overcome these difficulties encountered in the ocular field, thanks to their features such as adjusting their porosity and fiber diameters, production with biocompatible materials and flexibility, and functionalization with different materials according to the purpose for use [15,16].
2. Nanofibers
Nanofibers are generally defined as filamentous structures less than 1 µm in diameter [17]. With the fiber diameter decreasing to nanometer size, the physical, chemical, and biological properties are highly developed, and nanofibers with a high surface area have emerged [18].
At the beginning of the areas where nanofibers are used the most, biomedical applications such as drug delivery systems, wound dressing, and tissue scaffold studies are coming (Figure 1). Due to the unique functionality and natural morphological properties of nanofibers, they are used as drug delivery systems. The diversity in the processability of nanofibers creates an advantage in terms of drug loading [19,20]. The regulation of the affinity between the polymers that form the structure of nanofibers and the drug also allows the adjustment of drug release profiles. The drug release profile varies according to the polymer properties forming the nanofiber, the production method, the different polymer combinations used, the surface modifications, and the behavior of the drug molecules [16].

Figure 1.
Biomedical applications of nanofiber composites.
Nanofibers have many advantages as a drug delivery system. They can be used for the controlled release of both hydrophilic and lipophilic active ingredients and biological molecules (such as antibiotics, proteins, etc.). Nanofiber morphology, porosity, and composition can also vary the drug release rate. Other advantages of nanofibers are that they have a high surface area/volume ratio and porosity, a structure similar to the extracellular matrix structure of tissue, and their production methods are less costly and simpler than many nanostructured drug delivery systems [21,22,23]. In addition, nanofibers can be designed as a targeted drug delivery system with thermoresponsive, pH-responsive, and electroresponsive properties [24,25,26,27]. Due to these properties, electrospun nanofibers, in particular, are getting more and more attention as a drug delivery system.
In the first trial of nanofibers used as a drug delivery system and produced by electrospinning, tetracycline hydrochloride was used as a model drug. Tetracycline-loaded sustained-release nanofibers were obtained with poly(lactic acid) (PLA) and poly(ethylene vinyl acetate) (PEVA) polymers [28]. In previous studies, nanofibers produced with many different drug molecules and polymers have been used in oral [29], transdermal [30], ocular [31], nasal [32], etc., applications.
2.1. Preparation Methods of Nanofibers
2.1.1. Electrospinning
Electrospinning creates ultrafine fibers using an electric potential with a high voltage and low current. J. F. Cooley, who patented the technology as an “Apparatus for electrically distributing fibers” in 1902, was the first to observe an electrospinning process for such a purpose. The popularity of electrospinning grew at the end of the 20th century, when lots of new publications appeared and continue to appear, covering a wide range of applications for electrospun fibers, including drug delivery, wound healing, tissue engineering, textiles, sensors, cosmetics, and food packaging [33].
Electrospinning is a simple and successful method to produce nanofibers with diameters in the range of 10–1000 nanometers [34]. One of the main advantages of electrospinning is the high degree of control it provides over fiber morphology. This allows researchers to tailor the nanofiber properties to specific applications such as tissue engineering, drug delivery, and filtration. Additionally, electrospinning is a versatile technique that can be used with a wide variety of polymers, including natural and synthetic polymers as well as composites. This versatility allows the production of a wide variety of nanofibers with different properties. In addition, electrospun nanofibers have a high surface area-to-volume ratio, making them useful for a variety of applications [4,5,6,35,36]. Finally, electrospinning can be easily scaled up for mass production, making it suitable for commercial applications [37,38]. However, electrospinning also has some limitations. For example, the production rate is relatively low in conventional electrospinning devices, making it difficult to quickly produce large quantities of nanofibers. Additionally, electrospinning requires the use of high voltages, which can be hazardous, and the use of organic solvents, which can be harmful to the environment and human health. Finally, electrospinning can be costly due to the need for special equipment and the use of high-quality polymers [36,39,40]. In general, electrospinning is a powerful technique with many advantages, but these limitations must be considered when choosing a fabrication method for a particular application.
A polymer melt or solution can be used to manufacture nanofibers. Solution-based electrospinning has accounted for the majority of the research. The standard electrospinning system consists of four main parts: (1) a high electrical field, (2) a conductive capillary tube with a nozzle, (3) a syringe pump, and (4) a collector [41]. A high voltage (usually 10 to 30 kV) is applied to a polymer solution during electrospinning to create a charge on the droplet’s surface. The applied voltage causes a charge movement in the polymer liquid, which can stretch the pendant drop’s shape, which is usually spherical due to surface tension. When the charged polymer liquid’s electrostatic repulsion overcomes the surface tension, a conical shape known as Taylor’s cone is formed, and the jet initiation begins at the cone tip.
Interestingly, flow rate and applied voltage control the two forces that indirectly cause Taylor’s cone to form. As a result, the development of a stable jet is aided by a good balance between the two characteristics. If the polymer liquid has adequate cohesive force, a stable jet is ejected from Taylor’s cone, allowing the polymer chains to stretch and create a uniform filament. The solvent evaporates during this process, and dried polymer fibers are randomly distributed on the collector (Figure 2) [42]. The spinneret is essential in electrospinning since it allows for numerous configurations. The electrospinning process can be classified as needleless, single, coaxial, side-by-side, or triaxial electrospinning, depending on the spinneret [33,43] (Figure 2).
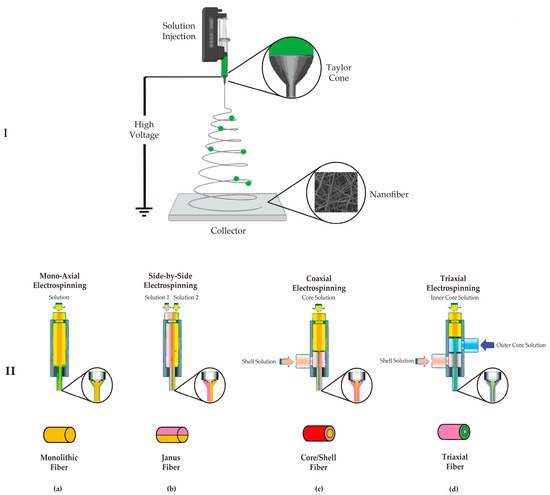
Figure 2.
(I) Schematic diagram of the electrospinning apparatus and Taylor’s cone. (II) Schematic examples of electrospinning spinnerets: (a) monoaxial electrospinning, (b) side-by-side electrospinning, (c) coaxial electrospinning, and (d) triaxial electrospinning.
Numerous factors throughout the electrospinning process influence the nanofiber morphology. These variables are classified as follows: (i) solution parameters, (ii) process parameters, and (iii) environmental parameters. The solution parameters are the type of solvent, the molecular weight of the polymer, the solution concentration, the viscosity of the solution, the conductivity of the solution, the surface tension, the dipole moment, and the dielectric constant. The applied electric field, the distance between the needle tip and the collector, the flow rate, and so on are all process parameters. Ambient parameters that influence the electrospinning process include relative humidity and temperature [44]. The morphology of nanofibers is affected by all factors. All factors impact nanofiber morphology during electrospinning, and none act independently. As a result, to design a nanofiber with the appropriate structure and characteristics, the various parameters must be optimized [44,45]. The effects of the different working parameters on the fiber morphology are summarized in Table 1.

Table 1.
List of parameters affecting properties of electrospun nanofibers.
- −
- Polymers for Electrospinning
Natural polymers (e.g., hyaluronic acid (HA), chitosan (CS), dextran (Dex), gelatin, and collagen), synthetic polymers (e.g., poly(lactic acid) (PLA), poly(lactic-co-glycolic acid) (PLGA), poly(-caprolactone) (PCL)), and their mixtures can all be used to create electrospun nanofibers [46]. Examples of synthetic and natural polymers and the solvents used in the formation of nanofibers for drug delivery are given in Table 2.

Table 2.
Examples of synthetic and natural polymers used for the creation of nanofibers for drug delivery purposes and their solvents.
Nanofibers must fulfill particular requirements regarding mechanical properties, hydrophilicity, shape, and biocompatibility for each application. These characteristics are controlled by the chemical composition of the fiber, namely the polymer structure. The polymer structure has an impact on the loaded drug’s release rate and treatment duration, with three major factors at play: (i) polymer swelling in water, (ii) polymer affinity for the drug, and (iii) polymer degradation rate. In addition, the molecular weight of the polymer impacts some of the physical characteristics of the fiber, such as its thickness and physical stability. Because of the direct relationship between polymer molecular weight and solution viscosity at a constant concentration, molecular weight is also used to control the polymer concentration at which electrospinning may be conducted [63].
- −
- Drug Loading Techniques in the Electrospinning Process
There are many different techniques for loading drugs in the electrospinning process. Some occur during electrospinning, while others occur with surface modification after electrospinning (post-electrospinning). In this review, electrospinning types used for drug loading will first be described and then post-electrospinning surface modification techniques will be discussed.
- −
- Types of electrospinning
Although the most popular method is to blend electrospinning, there are many different types of electrospinning methods. Each type of electrospinning has its own advantages and disadvantages. In Table 3, the advantages and disadvantages of these different electrospinning types are briefly mentioned.

Table 3.
Different types of electrospinning and their advantages and disadvantages.
Blend Electrospinning: The blend electrospinning procedure in which drug/biomolecules are dispersed or dissolved in the polymer solution creates nanofibers with drug/biomolecules scattered throughout the fibers [70]. Despite its simplicity in comparison to other electrospinning techniques, the solvents used to disperse bioactive compounds can cause protein denaturation or loss of biological activity. Furthermore, the inherent charge of biomolecules can frequently cause migration on the jet surface, resulting in dispersion on the surface of nanofibers rather than encapsulation of biomaterials within the fibers. Surface dispersion has been linked to burst biomolecule release [64,70].
Coaxial Electrospinning: Coaxial electrospinning is an enhancement of conventional blend electrospinning such that two nozzles, rather than one, are connected to the high-voltage source. Two distinct solutions are put into each nozzle and pumped out to produce nanofibers with core-shell morphologies. Core-shell nanofibers with superior physicochemical and biological characteristics may be created using synthetic or natural polymers [71].
Electrospinning in a coaxial (or triaxial) direction is thought to be a viable approach for achieving sustained drug release from electrospun core-sheath nanofibers. It is formed by the simultaneous flow of a core and sheath solution from distinct capillaries. The production of core-sheath fibers by triaxial electrospinning, in which the electrospun fiber has a core, intermediate layer, and sheath, has recently been introduced. Multiple drugs may be put into the core-sheath fibers, and their release kinetics can be controlled using coaxial electrospinning. Coaxial spinning has a higher drug loading efficiency than blend spinning. More notably, the first burst release is observed to be lower in coaxially spun core-sheath nanofibers than in blend-spun fibers. After the hydrophilic component of the core dissolves, the core expands or dissolves, generating pores in the shell in core-sheath nanofibers. While drugs are being loaded into the core phase, the shell phase can act as an outer protective layer. In addition, incompatibilities can be solved. For example, hydrophilic drugs in the core phase can be integrated into hydrophobic polymers in the shell phase. Moreover, the shell phase can act as a physical barrier to provide sustained-release kinetics, and by loading the core and shell phases, two separate release patterns can be produced from a single delivery system [42].
Emulsion Electrospinning: In some sources, the emulsion electrospinning method is shown in the blend electrospinning category [72]. The configuration for emulsion electrospinning is similar to the setup for blend electrospinning. Emulsion electrospinning is the simultaneous spinning of two immiscible liquids to produce core-shell nanofibers. Active bioactive compounds and surfactants are allowed to create W/O emulsions before being blended with the polymer matrix solution [73]. The emulsion droplets are stretched into an elliptical shape during the spinning [71].
Moreover, the continuous-phase solvent evaporates quickly, resulting in a viscosity gradient and droplet enrichment in the axial area. Because of the viscosity gradient between the polymer matrix and the elliptical droplet, the core material settles inside the fiber matrix rather than on the polymer’s outer surface. This technology is simpler than coaxial electrospinning and provides sustained release of the loaded cargo components [71,74]. The applied voltage has a substantial effect on the diameters of the nanofibers. Higher applied voltages result in the formation of nanofibers with smaller diameters. Other parameters such as flow rate and spinning distance can also have an impact on fiber morphology. On the other hand, the interface tension between the organic and aqueous phases of the emulsion can harm the bioactive molecules placed within the emulsion electrospun fibers [71].
Melt Electrospinning: Melt electrospinning has risen in popularity in the electrospinning arena where toxicity and solvent accumulation were problems. Instead of using a solution, the polymer melt is used in this method, which is changed from liquid to solid after cooling to achieve the desired product, rather than using solvent evaporation. To produce high-quality fibers with uniform morphology but a wide range of diameters, the polymer-melt flowrate and homogeneous polymer-melt conditions must be controlled. Polymer blends and additives were used to reduce the average diameter of the fibers. The influence of melt temperature on the structure and function of drugs, proteins, and bioactive substances loaded in fibers can be substantial. The flowrate and melt viscosity of melt electrospinning could have an impact on the properties of the produced fibers. The surface wettability of melt electrospun fibers has been enhanced by forming hydroxyl, peroxyl, and N-containing functional groups using oxygen and ammonia plasma, respectively [71].
Gas-Jet Electrospinning: Gas-jet electrospinning is upgrading the conventional melt electrospinning process to produce nanofibers. In this method, polymer solutions are set up into a gas-jet device without the use of an electric field [71]. Additionally, the coaxial jet is surrounded by a tube feeding the heated gas, which can provide sufficient heat near the nozzle. Due to the coflowing gas jet, polymer solutions are exposed to additional stretching and shearing forces [75]. Thus, nanofibers produced in this method become thinner and more homogeneous demonstrating that polylactic acid nanofibers have thinner diameters when the gas flow rate is increased [76].
Side-by-side Electrospinning: Side-by-side electrospinning is a process in which two liquids are applied from separate capillaries. In some sources, it is indicated as a coaxial electrospinning type. However, they are placed adjacent to each other rather than two polymer solutions nested inside each other as in the coaxial process [77]. For example, Yang et al. used a side-by-side electrospinning method to create PVP and ethylcellulose nanofibers with a synergistic release of ciprofloxacin and silver nanoparticles, both of which have antibacterial activity. With this Janus approach, the fibers were able to burst and release ciprofloxacin within 30 min. The antibacterial effect was then maintained for 72 h by the sustained release of silver nanoparticles, significantly inhibiting bacterial growth. Because of this characteristic, this scaffold is a promising method for preventing infections throughout the wound-healing process [78].
- −
- Post-electrospinning surface modification techniques
Several methods have been used to immobilize drugs on the surfaces of electrospun polymeric nanofibers. These can be listed as follows: (i) physical immobilization by simple physical surface adsorption, (ii) layer-by-layer assembly, or (iii) chemical immobilization methods. These methods can provide stronger and more stable drug immobilization on the nanofiber’s surface [79].
Physical Surface Adsorption: Physical surface adsorption is the easiest way to incorporate bioactive substances into membranes and does not rely on chemical treatment. Physical absorption refers to the immersion of electrospun fibers in a liquid solution, where the internal substances tend to adhere to the surface of the scaffolds through electrostatic interaction. Weak nonspecific intermolecular interactions (such as those found in electrostatic interactions, hydrogen bonds, hydrophobic interactions, and Van der Waals forces) are generally established between the surface and peptide sequences [79,80]. Casper et al. created electrospun PEG nanofibers that were functionalized with low molecular weight heparin, a highly sulfated glycosaminoglycan that binds growth factors (GF) for drug delivery and wound healing. The results show that this functionalization technique allows essential fibroblast growth factor (bFGF) to bind to the surface of PEG nanofibers [81]. However, due to the uncontrolled release of drugs, this approach is rarely used for loading proteins or nucleic acids. For example, Wang and colleagues constructed electrospun PLGA/hydroxyapatite (PLGA/HAp) microfibers encapsulated with bone morphogenetic protein-2 (BMP-2). Although the release of adsorbed BMP-2 took more than two weeks, 75% of the proteins were released within the first five days. There was also a distinct burst release phenomenon for gene transfer, and the released gene was rapidly degraded [82].
Layer-By-Layer Assembly (LbL): Generally, layer-by-layer assembly is a sophisticated method of alternating the adsorption of materials onto a surface utilizing opposite interactions, with one layer of material deposited at a time. LbL assembly is a cyclical process that involves the alternating deposition of polymers with opposite charges on their surfaces to build a covering of polyelectrolyte multilayers (PEMs) or a free-standing film [83]. The depositing process can be continued indefinitely until a multilayer film of the required thickness is produced. Electrostatic attraction is the primary driving factor in the assembly process, but hydrogen bonding, hydrophobic, covalent, and biological interactions can also be important. The film’s composition, morphology, and structure may be accurately controlled by this build-up [84]. Xine et al. created cellulose nanofiber mats coated with silk fibroin (SF) (negatively charged) and lysozyme (positively charged). The in vitro and in vivo studies revealed that the mats can enhance wound healing as well as wound infection prevention [85].
Chemical Immobilization: Chemical immobilization refers to the covalent bond that binds agents on the surface of fibers. External enzymes can control the release of agents. The surface characteristics of fibrous membranes can be modified using this technique. Because of the complexity of the immobilizing agents at the membrane surface, covalent immobilization is not a widely used method of loading bioactive compounds [72]. For instance, to immobilize bioactive molecules on the surface of nanofibers for wound healing, the chemical immobilization of primary amine and carboxylate groups has been widely used [86].
2.1.2. Non-Electrospinning Methods
- −
- Interfacial Polymerization
In this process, two different monomers that can be dissolved in two separate phases (such as oil and water) are used. After dissolving the two monomers, they will polymerize at the interface of the emulsion droplet. For example, after dissolving diamine in the water phase, the diacid chloride (oil soluble) is added to the solution and reacts with the first monomer at the interface forming the wall material. Nanofibers can be created by using this method due to homogeneously nucleated growth. Different polymers may also be produced by selecting other monomers; however, most publications refer to polyamide membranes [87].
- −
- Drawing
Another process for producing fibers is drawing. It is similar to spinning. The fact that this process requires just a sharp tip or a micropipette is considered its main advantage. A sharp tip is used in this procedure to draw a droplet of a previously placed polymer solution as liquid fibers. Then, the solvent is evaporated due to the high surface area, allowing the liquid fibers to solidify. Hollow glass micropipettes can be used rather than the sharp tip with a continuous dose of the polymer to avoid the volume shrinking problem that limits the continuous drawing of the fibers and affects their diameter. The micropipette is gently pulled from the liquid and moved at a low speed after being dipped into the droplet using a micromanipulator. Consequently, nanofibers are pulled and deposited on the surface by touching it with the end of the micropipette. To produce nanofiber, this process was repeated several times on each droplet. It can also be used to create continuous nanofibers. Furthermore, exact control of essential drawing parameters such as drawing speed and viscosity can be achieved, allowing for repeatability and control over the dimensions of the produced fibers. Although this method is simple and the dimensions of the fibers can be controlled, it is limited to the laboratory scale because nanofibers are produced one at a time. Moreover, it is a discontinuous process with low performance. To withstand the stress produced by the pulling, only viscoelastic material can be used in this process. In addition, depending on the orifice size, fibers with diameters larger than 100 nm can be produced [87,88].
- −
- Template Synthesis
In this process, using chemical or electrochemical oxidative polymerization, polymeric nanofibers can be produced using a nonporous membrane with several cylindrical pores [87].
One of the features of this technology is the ability to produce nanofibers with varying diameters by using different templates [87]. It implies using a template or mold to achieve a desired material or structure and produce nanofibers. A metal oxide membrane is referred to in the template. In this process, nanofibers are produced by passing polymer solution through pores of nanoscale diameter while applying water pressure on one side, causing extrusion of the polymer and the production of fibers upon contact with the solidifying solution. This technique cannot create nanofibers with long fiber lengths; only a few micrometers are obtained, and the membrane’s pore size controls the diameter of these fibers [88].
- −
- Phase Separation
In phase separation, a polymer is first blended with a solvent before being gelated. The solvent phase is then extracted, leaving the other remaining phase. This system’s main mechanism is separating phases due to physical inconsistency. Some sources describe a thorough process in five steps: (i) polymer dissolving in a solvent at normal temperature or increased temperature; (ii) gelation is the most challenging part of controlling the nanofiber morphology (porosity); (iii) the duration of gelation varied with polymer concentration and gelation temperature; (iv) water extraction of the solvent from the gel; and (v) freezing and freeze-drying under vacuum [89].
It is the minimum equipment needed for the process. It can directly create a nanofiber matrix where mechanical characteristics can be modified by changing the polymer concentration. This process can produce long continuous fibers; however, not all polymers can undergo phase separation and create nanofibers since gelation capacity is required, which limits the use of the phase separation method [87]. Only a few polymers, such as polylactide (PLA) and polyglycolide, have been turned into nanofibers using the phase separation process so far [88].
- −
- Self-Assembly
Self-assembly is a bottom-up nanomaterial production approach in which molecules organize and arrange themselves into patterns or structures via noncovalent forces such as hydrogen bonding, hydrophobic forces, and electrostatic interactions. It is an effective technique for creating very small nanofibers with lengths of several micrometers by forming supramolecular hydrogels via weak interactions such as hydrogen bonding and hydrophobic interactions [90]. The basic process is based on intermolecular interactions that bring tiny units together; the structure of the smaller units of molecules dictates the overall structure of the nanofiber [91]. The major disadvantage of the process is that it is a complex, long, and highly elaborate procedure with low production and a lack of precise control over the fiber dimensions. Furthermore, this process is limited to the production of nanofibers from small active molecules that may self-assemble or be stimulated by an external stimulus [87,90,91].
- −
- Freeze-Drying (FD)
Freeze-drying is a process that involves three main steps: first, the solution is frozen at a low temperature (−70 °C to −80 °C). Then, the frozen sample is placed in a chamber where the pressure is reduced to a few millibars through a partial vacuum. This is known as the primary drying process, which removes ice from the material by direct sublimation. In the next step, most of the unfrozen water in the material is removed by desorption in a secondary drying process. In recent years, freeze-drying has been extensively studied for the production of three-dimensional porous scaffolds [92,93]. In some studies, as a result of the optimization of the freeze-drying method, the diameters of this scaffold are reduced to nanosize, so researchers define this structure as nanofiber [94,95,96,97]. For example, Ma et al. used the freeze-drying method to create chitosan/sodium hyaluronate polyelectrolyte complex fibers. In vitro tests showed that the resulting fibers had good compatibility and no cytotoxicity [98]. In another study, Lee et al. obtained the nanofiber structure of polypyrrole nanoparticles using the freeze-drying method [99].
Moreover, freeze-drying contributes to the production of nanofibers by applying it together with other different methods as well as being used in the phase separation method, as mentioned before [89]. For instance, Lin et al. synthesized ultrafine porous boron nitride nanofiber mats in a two-step production method including freeze-drying and pyrolysis methods. The resulting nanofiber mats have a high surface area and porosity [100]. Many studies also use freeze-drying in combination with the electrospinning method. By combining these two methods, a material with a high surface area and small fiber diameters of electrospun materials can be created, as well as the structural integrity and porosity of freeze-dried samples [101,102]. For example, Tang et al. combined electrospinning and freeze-drying techniques to fabricate membranes, which were found to have increased absorption capacity with increasing porosity [103].
Freeze-drying (FD) has several significant advantages over other methods. It can create porous structures with controllable sizes directly from polymers such as chitin without the requirement for structure-directing additives or pretreatments, which other techniques such as self-assembly and electrospinning cannot.
Furthermore, the freeze-drying technique does not require a high temperature or further leaching step, and the use of water and ice crystals rather than an organic solvent in the scaffold production process makes this process more appropriate for biomedical applications. As a result, it has attracted increased interest in the production of nanofibers. Even though the freeze-drying process has various advantages, it is still challenging to construct scaffolds with hierarchical structures (e.g., vascularized systems) using this method. This method’s nanofiber mats can be used as drug delivery systems, as precursors for highly porous carbon nanofibers, or as templates for manufacturing inorganic fibers [87,104].
- −
- Rotary or Centrifugal Jet Spinning (RJS/CJS)
Rotary or Centrifugal jet spinning (RJS/CJS) (also called forcespinning or rotational jet spinning) is a novel method for the fabrication of nanofibers with controlled size, morphology, and orientation. RJS/CJS differs from electrospinning in that it uses centrifugal force instead of an electric field to produce the spinning process [105,106]. This technique involves the use of a rotating spinneret that ejects a polymer solution or melts through a small orifice (nozzle). At a certain rotational speed, the centrifugal force becomes more powerful than the surface tension of the liquid being rotated, forming liquid jets from the rotating head’s nozzles. These jets are then elongated by the combination of centrifugal force and air friction, ultimately creating nanofibers. The fibers can be collected on a rotating mandrel or a stationary collector, resulting in aligned or randomly oriented fibers, respectively [90,107]. The process of creating nanofibers through RJS/CJS involves controlling the centrifugal force, viscoelasticity, and mass transfer of the solution to manipulate the solution filament into thin nanofibers. The diameter of the resulting nanofibers is greatly influenced by the spinning solution’s elasticity and the solvent’s evaporation rate [108]. In addition, the RJS/CJS process for nanofibers is influenced by multiple factors, including the speed at which the heated structure rotates, the design of the nozzle, the collection system used (such as the distance between the collector and spinning apparatus), and the temperature. These factors affect the shape and appearance of the resulting nanofibers [109,110].
The RJS/CJS technique can be divided into three types, namely melt RJS/CJS, immersion RJS/CJS, and nozzleless RJS/CJS. In melt RJS/CJS, molten polymer is used in the RJS apparatus to create micro and nanofibers without the need for solvents. High temperatures are used to melt the polymer, and additives such as viscosity-reducing agents or plasticizers are used to improve fluidity and reduce fiber diameters [111]. Immersion RJS/CJS involves dropping the extruded polymer into a solution for solidification and/or crosslinking, which minimizes extrusion breakage and bearing in the fibers by reducing surface tension [111,112]. Nozzleless RJS/CJS, on the other hand, does not use a needle for polymer extrusion and instead creates “fingers” by pulling out of a lid-disk gap due to Rayleigh–Taylor instability. This method is an alternative to nozzle RJS systems that can clog when highly viscous polymers are used [111].
Nanofibers produced by the RJS/CJS method have been used with increasing interest in drug delivery systems and tissue engineering studies in recent years. For example, carvedilol was incorporated into nanofibers using an RJS/CRS method, and the nanofibers were then formulated into dispersible tablets. Dissolution studies showed that the drug released quickly and completely from the nanofiber-based tablets, regardless of pH [113]. Another study used PCL and gelatin as model polymers in C-spinning to produce highly aligned ultrafine fibers with smooth surfaces. The resulting mat showed improved hydrophilicity, porosity, and mechanical properties, making it suitable for tissue engineering applications. In vitro and in vivo experiments confirmed the biocompatibility of the scaffolds, which can be used as a wound dressing material [114].
Badrossamay, M. R. et al. engineered nanofiber constructs with robust fiber alignment from collagen, gelatin, and PCL blended using the RJS/CJS method and compared them to electrospun fibers. The RJS/CJS-spun fibers had a higher production rate and retained more protein content on the surface. The authors demonstrated the biofunctionality of RJS/CJS scaffold fibers by testing their ability to support cell growth and maturation with different cell types. The hybrid nanofiber constructs fabricated by RJS/CJS have the potential to be used as a scaffold material for a wide variety of biological tissues and organs as an alternative to electrospinning [115].
By changing the spinneret type, nanofibers of different structures can be obtained in the RJS/CJS method, just as in the electrospinning method. Khang, A. et al. obtained anisotropic two-phase Janus-type nanofiber scaffolding meshes using PCL and gelatin polymers by the centrifugal jet spinning method. The resulting scaffold meshes exhibited a variety of mechanical properties and surface chemistries that are potentially useful in tissue engineering applications. The study provides evidence for the ability of this method to precisely control the orientation of the fiber web and the distribution of different materials within the scaffold [116].
In conclusion, the centrifugal spinning method has become quite popular in drug delivery systems and tissue engineering studies in recent years. The main reasons for this are its high productivity and low solvent usage, as well as being a relatively low-cost nanofiber production method that does not require high voltages [105]. Additionally, successful scale-up studies have been conducted [117]. However, the main disadvantage of centrifugal spinning is that the quality and productivity of fibers are highly affected by material properties and nozzle design [111].
3. Nanofibers as an Ocular System
3.1. Anatomy and Physiological Barriers of the Eye
Anatomically, the eyes consist of two segments, anterior and posterior. The lens, which is a transparent structure responsible for refracting the light coming into the eye, is the border that separates these two parts [118]. The anterior segment includes the cornea, iris, ciliary body, conjunctiva, and anterior surface of the sclera. The choroid, retina, optic nerves, and the posterior surface of the sclera are located in the posterior segment. In both the anterior and posterior segments, the interstitial spaces are filled with aqueous humor and vitreous humor, respectively [13].
The anatomical structure of the eye can be classified independently of its segments, as well as according to the structural features of the layers that make it up. These layers are from outside to inside: Tunica fibrosa oculi (fibrous layer), tunica vasculosa oculi (vascular layer), and tunica interna oculi (neural layer). Tunica fibrosa oculi, the fibrous layer of the eye, consists of the cornea, conjunctiva, and sclera. This layer, which does not contain lymph and blood vessels, surrounds the eyeball from the outside [119]. The cornea is a thin and transparent tissue consisting of five layers (corneal epithelium, Bowman’s membrane, stroma, Descemet’s membrane, and the endothelial layer). The conjunctiva is a mucosal membrane [13]. The sclera is a fibrous tissue consisting of five layers (Tenon’s capsule, episclera, scleral spur, limbus, and posterior sclera) [120], just like the cornea. The tunica vasculosa oculi, located just below the tunica fibrosa oculi, is also known as the uvea. This layer consists of the choroid, ciliary body, and iris. Thanks to the vessels it contains, the blood circulation in the eye tissues and the production of aqueous humor are carried out by the tissues in this layer [13,119]. The tunica interna oculi is the inner layer where the light is transformed into a neural impulse and transmitted to the brain [121].
Since the eye is a vital organ in contact with the external environment, it must maintain its integrity. For this reason, it contains natural anatomical and physiological barriers. All ocular tissues, vessels, and fluids are the natural barriers of the eye. These barriers are the tear film, cornea, conjunctiva, sclera, blood–aqueous humor (iris–ciliary body), lens, and blood–retina barrier [122,123,124]. These barriers, whose main task is protecting the eye from foreign fluids and objects, also act as drug rate-limiting steps. They reduce the penetration of drugs into ocular tissues and their bioavailability. Since each of these barriers has different structures from the others, they also perform rate-restriction functions on drugs with different properties.
The tear film covers the surface of the cornea and conjunctiva. It is the primary protective structure against ocular problems of chemical, mechanical, bacterial, or viral origin. Likewise, it forms the largest barrier among drugs applied topically to the eye [120,125]. Topically applied drugs are usually applied to an area called the cul-de-sac with a volume of 7–10 µL [126]. However, the application dose of any drug is 20–50 µL on average [127]. Due to this sudden increase in the volume of the area, topical drugs such as drops and emulsions applied topically are removed from the eye by nasolacrimal drainage and added to the systemic circulation [123,128]. Around 80% of a topically applied conventional drug is eliminated due to the tear film and nasolacrimal drainage [129].
The cornea acts as an important barrier to conventional topical drugs in the anterior segment of the eye [130]. The epithelium, stroma, and endothelium, which are parts of the cornea, are tissue formations of different polarities. Depending on the arrangement of the cells in the epithelial layer, there is a high shunt resistance and is considered a tight tissue [122]. These tight junctions give the epithelium a lipophilic character. Stroma, conversely, has a highly hydrophilic character due to the collagen fibers in its structure. Therefore, while the epithelium prevents the passage of hydrophilic drugs, the stroma acts as a barrier to the passage of lipophilic drugs. In the endothelium, the innermost layer of the cornea, molecules of up to 70 kDa can pass through passive transport [123,124,130].
The conjunctiva acts as a barrier through the numerous capillaries and lymphatic structures in its structure. It dilutes applied drugs with the blood or lymphatic circulation. There are epithelial cells in the conjunctiva as well as in the cornea. These epithelial cells have a shunt resistance, although not as much as in the corneal epithelium. Points of this resistance are considered tight structures and act as barriers to drugs. The conjunctiva acts as the main barrier for drugs not administered via the corneal route. Compared to the cornea, the conjunctiva is a hydrophilic tissue and more suitable for passing large molecules. For this reason, the direct conjunctival application of ocular drugs is being studied by reducing corneal application to increase the absorption of larger bio-organic molecules such as proteins and peptides. However, considering that most of the drugs used in the clinic are lipophilic and small molecules, it is seen that corneal application will not lose its importance [120,122,130].
The sclera has a hydrophilic character due to the collagen fibers in its structure. For this reason, it acts as a barrier for lipophilic drugs. However, small-sized lipophilic drugs also show scleral permeability similar to hydrophilic drugs. In addition, the size of the molecules and the positive charge are other parameters that reduce the passage through the sclera [124,131].
The iris and ciliary body, the tissues of the tunica vasculosa bulbi form the blood–aqueous barrier (BAB). This barrier is also known as the anterior blood–eye barrier [124]. The ciliary body produces aqueous humor, which stabilizes intraocular pressure. On the other hand, the dense capillaries in this layer contain tight junctions. These tight junctions, the epithelial cells of the iris, and the barrier formed by aqueous humor are highly restrictive for hydrophilic drugs. Small molecules and lipophilic drugs can easily pass the BAB and enter the systemic circulation via the uvea. An inflammation that may occur in this barrier disrupts the integrity of the barrier. This causes the amount of drug passing to the anterior region of the eye to not be controlled [122,124,131,132,133].
The lens is the eye’s tissue, consisting of 65% water and the remainder predominantly proteins. It is distinctly different from other eye tissues with its protein content. The lens is a tissue in which active and passive transport, depending on Na+, are effective. It mainly affects the passage of drug molecules for this reason [122,131].
The blood–retinal barrier (BRB) is formed by the combination of several factors. Primarily, retinal pigment epithelial cells have Na+, K+-ATPase pumps that ensure the balance of Na+ and K+ ions in the eye. On the other hand, the capillaries in their structure have tight connection points. The passage of drug molecules through these tight junctions is the second rate-limiting step. At the same time, molecules that can pass through these capillaries quickly enter the systemic circulation, which reduces the number of drugs in the target area, thus reducing the bioavailability. Finally, the ganglion cells in the retina are nerve cells and form a barrier similar to the cerebrospinal barrier. The BRB acts as a barrier for the passage of proteinaceous and small hydrophilic molecules while allowing the passage of lipophilic molecules. However, these lipophilic molecules are also rapidly distributed and eliminated due to intense systemic circulation [120,122,124,131].
Increasing the ocular bioavailability of a drug to be applied to the eye can only be achieved by overcoming all of these barriers and delivering the drug to the target tissue. Two ways are followed for this purpose. One of them is the application of the drug to the eye in different routes. These routes of administration are classified as topical, periocular (subconjunctival, transscleral, and intravitreal), and systemic [120,124]. Although these different routes, which are actively used today, seem to have solved the problem of low ocular bioavailability, they contain risks as they can cause serious complications such as blindness. For this reason, the second way to increase ocular bioavailability is the development of drug delivery systems [118].
3.2. Drug Delivery System
Conventional dosage forms applied to the eye are solutions, suspensions, ointments, and gels [134,135]. Today, 90% of the drugs on the market fall into this group. These dosage forms usually target the anterior segment of the eye and may have little effect on a possible disease in the posterior segment [136]. On the other hand, they have very low bioavailability due to ocular barriers. Due to their low bioavailability, their dosing frequency during the day is high, which reduces patient compliance. In addition, the fact that gels and ointments cause blurred vision is another factor that reduces patient compliance [137]. To prevent all of these negativities, drug delivery systems targeting certain eye tissues are being developed.
Drug delivery systems are produced by using a suitable polymer/surfactant/lipid, have micro or nano size, and are developed to deliver the loaded drug to the target tissue [138]. Since they target the drug to the tissue where the effect is desired, they allow the use of active pharmaceutical ingredients (API) at lower doses. Thus, a decrease in the rate of side effects may occur. In addition, drug releases can be extended thanks to the polymer/surfactant/lipid used in their preparation. Thus, the dosing frequency can be reduced and patient compliance can be increased [139,140,141]. There are different delivery systems developed for ocular drug targeting. Micro and nanoparticles, micro and nanoemulsions, nanosuspensions, micelles, liposomes, solid lipid nanoparticles, dendrimers, cubosomes, discosomes, niosomes, spanlastics, bilosomes, hydrogels, implants, inserts, lenses, and nanofibers are among the carrier systems developed [118]. While some of these carrier systems have turned into commercial products, some have remained in experimental studies. The most well-known commercial products are Ocusert® (an ocular insert containing pilocarpine) [142], Xelpros® (a pilocarpine-containing micellar system) [143], Modusik-A Ofteno®, Papilock Mini®, and Cequa® (which are micellar systems containing cyclosporine A) [144,145].
Nanofibers have also been used in ocular drug targeting since the first years they were designed as drug delivery systems. Thanks to the materials used in their preparation, they increase the penetration and contact time of drugs with ocular tissues and show high biocompatibility. In addition, their extended-release profile also reduces the dosing frequency [146,147]. However, the features that make nanofibers distinctly superior to other drug delivery systems are that they have a large surface area, high porosity, easily adjustable diameters, and can be combined with other drug delivery systems [15]. Having a large surface area enables the nanofibers to be loaded with more drugs than other drug delivery systems. These features, coupled with extended drug release profiles, allow for further reductions in dosing frequencies. For this reason, nanofibers stand out as a promising approach to the medical treatment of chronic ocular diseases. The fact that porous structures can be produced to be similar to ocular tissues further increases their biocompatibility compared to other carrier systems [148,149,150]. One of the most important features of drug delivery systems is that they are micro or nanosized. The diameter size of nanofibers could be adjusted much more easily than other carrier systems. Thus, the permeation of the drug with ocular tissues can be increased. The studies found that the carrier systems should be smaller than 100 nm to overcome the corneal barriers [151], and the scleral pore openings were between 20–80 nm [13]. Nanofibers can be easily produced in the appropriate diameter according to the target tissue. It also provides an important advantage in that it can be used in combination with other drug delivery systems (nanoparticle, dendrimer, etc.). In recent years, studies in the hydrogel form of nanofibers, which are generally converted into insert or implant form, have also been carried out. Although the drug has been proven to increase penetration and permeation with ocular tissues, it can cause blurred vision, irritation, and watery discharge. Despite these problems, nanofibers may be a viable option for a variety of ocular drug delivery systems [152]. To avoid these side effects and maximize the benefits of using nanofibers for ocular drug delivery, nanofiber properties such as size, surface charge, and polymer type and composition can be optimized, which can significantly affect their interaction with ocular tissues. By optimizing these properties, researchers can minimize the negative effects of nanofiber-based drug delivery systems while maintaining their effectiveness. Thus, the irritation potential of formulations can be reduced by reducing interactions with ocular tissues. In addition, nanofibers can be combined with other delivery systems such as hydrogels or liposomes to increase their performance and reduce their adverse effects. For example, nanofibers prepared with hydrogelizing polymers can provide a protective barrier by minimizing irritation and other side effects. In Table 4, nanofibers developed as an ocular drug delivery system are summarized (in alphabetical order).

Table 4.
Nanofiber-based ocular drug delivery systems.
3.3. Tissue Engineering
Many tissues in the living body cannot regenerate after injury. Even with surgical intervention on these damaged tissues, the tissue cannot regain its former form and return to the former quality of life of the individual [179]. One of the methods developed to solve this problem is tissue engineering. Tissue engineering, also known as regenerative medicine, is the science that deals with the development or production of therapeutic stem cells, tissues, and artificial organs [180]. Tissue engineering is widely used in bone and nerve regeneration, cartilage repair, and cardiovascular and ocular tissue development. Although it may seem simple and feasible, it is a multidisciplinary field of study and contains various difficulties in each step of obtaining tissue from stem cells [109].
The extracellular matrix (ECM) is a heterogeneous, binding network of fibrous glycoproteins with micro- and nanosized pores [181]. It provides the physical scaffold and mechanical stability required for tissue morphogenesis and homeostasis. It is aimed at developing scaffolds similar to ECMs in tissue engineering [148,149,150]. Electrospinning is the widely used method in producing scaffolds produced using biodegradable materials to obtain micro or nanosized fibrous tissue.
The eye has a unique structure and is rich in epithelial cells and neural networks. However, the structural integrity of this organ, which is in contact with the outer surface, may be impaired due to chemical, radiation and burn injuries, and infections caused by contact lenses. Unfortunately, these structural problems can be permanent. For example, the cornea, the outermost transparent layer of the eye, has two main functions. The first is to protect the structures inside the eye, and the second is to refract the light coming from outside and focus it clearly on the retina [182]. Good vision largely depends on corneal epithelial regeneration by limbal epithelial cells [183]. However, structural problems due to environmental factors may lead to limbal stem cell deficiency, and in this case, the act of seeing may not be performed. Therefore, the development of ocular tissues is an important field of study for tissue engineering. It is a great advantage that many biodegradable polymers used in the production of scaffolds are also biocompatible with ocular tissues. Table 5 summarizes the ocular tissue engineering studies with nanofibers.
When the studies are examined, it is generally emphasized that nanofibers produced with different methods and different materials in ocular tissue engineering studies are highly biocompatible with eye tissue and suitable for adhesion and proliferation for different types of cells [184,185]. In addition, successful results have been obtained from anti-inflammatory gene expression studies [186,187]. In other studies, it has been concluded that the prepared nanofibers have better mechanical properties than the amniotic membrane [188]. In some in vivo studies, it has also been observed that cell-cultured nanofibers provide re-epithelialization in the eye [189,190].

Table 5.
Nanofiber-based ocular tissue engineering studies.
Table 5.
Nanofiber-based ocular tissue engineering studies.
| Tissue | Polymer | Comments | References |
|---|---|---|---|
| Limbal stem cell | PHBV |
| [184] |
| Limbal stem cell | PCL |
| [185] |
| Retinal pigment and corneal epithelial cells | PCL |
| [191] |
| Conjunctival epithelial cells | SF/PLCL |
| [192] |
| Retinal ganglion cells | PPy-G/PLGA |
| [193] |
| Ciliary pigment epithelial cells | RADA-16-I peptide |
| [194] |
| Limbal stem cell | dAM/PCL |
| [189] |
| Limbal stem cell | PCL |
| [195] |
| Limbal stem cell | PLA |
| [190] |
| Limbal stem cell | Carbodiimide cross-linked AM |
| [196] |
| Limbal stem cell | PCL/PLA/PLGA/dAM |
| [186] |
| Llimbal stem cell | PCL |
| [197] |
| Limbal stem cell | Silk |
| [198] |
| Retinal progenitor cells | LPG/DPG/RPG |
| [199] |
| Limbal and mesenchymal stem cells | Copolymer PA6/12 |
| [187] |
| Scaffold-based corneal implant | Keratin/PVA |
| [200] |
| Corneal wound dressing | COL/HA/PEO/GA/CS |
| [188] |
| NIH3T3 fibroblast cell | PCLPCL/GEL |
| [201] |
| The lipid phosphate phosphatase-related | E-PA |
| [202] |
COL: collagen, CS: chitosan, dAM: decellularized amniotic membrane, DPG: D-phenylalanine gelators, E-PA: P Lauryl-VVAGE-Am, GA: glutaraldehyde, GEL: gelatin, HA: hyaluronate (HA), LPG: L-phenylalanine gelators, PA: peptide amphiphile, PA6/12: polyamide 6/12 (PA6/12), PCL: polycaprolactone, PEO: polyethylene oxide, PHBV: poly (3-hydroxybutyrate-co-3-hydroxyvalerate), PLA: poly(lactic acid), PLCL: poly(L-lactic acid-co-3-caprolactone), PLGA: poly(lactic-co-glycolic acid), PPy-G: polypyrrole functionalized graphene, PVA: polyvinyl alcohol RPG: a racemic mixture of LPG and DPG, and SF: silk fibroin.
4. Summary
Today, nanofiber technology can be used in many fields, from textiles to sensor production, from drug delivery systems to tissue engineering. Although there are different production methods, electrospinning is the most widely used among these methods. The most important reasons for this are the ease of controlling the fiber morphology, its versatility, and its ability to produce high-quality fiber. In addition, one of its unique advantages is the ability to prepare nanofibers with different properties by using different tools and techniques in the electrospinning method.
As a drug delivery system, nanofibers can be targeted to different tissues in the treatment of many diseases. Ocular applications are one of the most prominent of these areas. Nanofiber-based ocular implants or inserts are frequently studied as drug delivery systems. The high biocompatibility of these systems and the apparent long drug release times are promising in terms of increasing patient compliance and bioavailability in ocular drug administration.
Due to their large surface area, highly porous structure, preparation with different polymers, high drug loading capacity, and special morphologies, nanofibers attract a lot of attention not only as drug delivery systems but also in tissue engineering. Tissue engineering studies are important, especially in ocular tissue destruction, which is frequently encountered due to environmental factors.
In recent years, in particular, positive and promising results from ocular drug delivery systems and tissue engineering studies with nanofibers suggest that nanofiber research will become even more popular in the upcoming years. With the development of the current scale-up studies, nanofibers also offer a tremendous alternative to the conventional drug carrier systems and biomaterials used in ocular targeting.
Author Contributions
Conceptualization, E.U.; writing—original draft preparation, M.E.D.; writing, review, visualization and editing, İ.E.-G.; writing, review and editing, S.G.; review and editing, Y.Ö.; supervision. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Weng, Y.; Liu, J.; Jin, S.; Guo, W.; Liang, X.; Hu, Z. Nanotechnology-based strategies for treatment of ocular disease. Acta Pharm. Sin. B 2017, 7, 281–291. [Google Scholar] [CrossRef]
- Bavatharani, C.; Muthusankar, E.; Wabaidur, S.M.; Alothman, Z.A.; Alsheetan, K.M.; Al-Anazy, M.M.; Ragupathy, D. Electrospinning technique for production of polyaniline nanocomposites/nanofibres for multi-functional applications: A review. Synth. Met. 2021, 271, 116609. [Google Scholar] [CrossRef]
- Kolahalam, L.A.; Viswanath, I.V.K.; Diwakar, B.S.; Govindh, B.; Reddy, V.; Murthy, Y.L.N. Review on nanomaterials: Synthesis and applications. Mater. Today Proc. 2019, 18, 2182–2190. [Google Scholar] [CrossRef]
- Wang, C.; Wang, J.; Zeng, L.; Qiao, Z.; Liu, X.; Liu, H.; Zhang, J.; Ding, J. Fabrication of Electrospun Polymer Nanofibers with Diverse Morphologies. Molecules 2019, 24, 834. [Google Scholar] [CrossRef] [PubMed]
- Kharaghani, D.; Khan, M.Q.; Kim, I.S. Application of Nanofibers in Ophthalmic Tissue Engineering. In Handbook of Nanofibers; Springer International Publishing: Cham, Switzerland, 2019; pp. 649–664. [Google Scholar]
- Ding, J.; Zhang, J.; Li, J.; Li, D.; Xiao, C.; Xiao, H.; Yang, H.; Zhuang, X.; Chen, X. Electrospun polymer biomaterials. Prog. Polym. Sci. 2019, 90, 1–34. [Google Scholar] [CrossRef]
- Hu, X.; Liu, S.; Zhou, G.; Huang, Y.; Xie, Z.; Jing, X. Electrospinning of polymeric nanofibers for drug delivery applications. J. Control. Release 2014, 185, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, S.; Misra, M. Electrospun polymeric nanofibers: New horizons in drug delivery. Eur. J. Pharm. Sci. 2017, 107, 148–167. [Google Scholar] [CrossRef]
- Mendes, A.C.; Gorzelanny, C.; Halter, N.; Schneider, S.W.; Chronakis, I.S. Hybrid electrospun chitosan-phospholipids nanofibers for transdermal drug delivery. Int. J. Pharm. 2016, 510, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Amagat Molas, J.; Chen, M. Injectable PLCL/gelatin core-shell nanofibers support noninvasive 3D delivery of stem cells. Int. J. Pharm. 2019, 568, 118566. [Google Scholar] [CrossRef]
- Akhgari, A.; Shakib, Z.; Sanati, S. A review on electrospun nanofibers for oral drug delivery. Nanomed. J. 2017, 4, 197–207. [Google Scholar] [CrossRef]
- Han, D.; Yu, X.; Chai, Q.; Ayres, N.; Steckl, A.J. Stimuli-Responsive Self-Immolative Polymer Nanofiber Membranes Formed by Coaxial Electrospinning. ACS Appl. Mater. Interfaces 2017, 9, 11858–11865. [Google Scholar] [CrossRef]
- Durgun, M.E.; Güngör, S.; Özsoy, Y. Micelles: Promising Ocular Drug Carriers for Anterior and Posterior Segment Diseases. J. Ocul. Pharmacol. Ther. 2020, 36, jop.2019.0109. [Google Scholar] [CrossRef] [PubMed]
- Özsoy, Y.; Güngör, S.; Kahraman, E.; Durgun, M.E. Polymeric micelles as a novel carrier for ocular drug delivery. In Nanoarchitectonics in Biomedicine; Elsevier: Amsterdam, The Netherlands, 2019; pp. 85–117. [Google Scholar]
- Razavi, M.S.; Ebrahimnejad, P.; Fatahi, Y.; D’Emanuele, A.; Dinarvand, R. Recent Developments of Nanostructures for the Ocular Delivery of Natural Compounds. Front. Chem. 2022, 10, 850757. [Google Scholar] [CrossRef]
- Yoo, H.S.; Kim, T.G.; Park, T.G. Surface-functionalized electrospun nanofibers for tissue engineering and drug delivery. Adv. Drug Deliv. Rev. 2009, 61, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Rabiei, N.; Kish, M.H. Extraction of nanofibers from polymer blends: A brief review. Polym. Adv. Technol. 2019, 30, 813–822. [Google Scholar] [CrossRef]
- Wei, Q.; Tao, D.; Xu, Y. Nanofibers: Principles and manufacture. In Functional Nanofibers and Their Applications; Elsevier: Amsterdam, The Netherlands, 2012; pp. 3–21. [Google Scholar]
- Friuli, V.; Pisani, S.; Conti, B.; Bruni, G.; Maggi, L. Tablet Formulations of Polymeric Electrospun Fibers for the Controlled Release of Drugs with pH-Dependent Solubility. Polymers 2022, 14, 2127. [Google Scholar] [CrossRef] [PubMed]
- Zahmatkeshan, M.; Adel, M.; Bahrami, S.; Esmaeili, F.; Rezayat, S.M.; Saeedi, Y.; Mehravi, B.; Jameie, S.B.; Ashtari, K. Polymer Based Nanofibers: Preparation, Fabrication, and Applications. In Handbook of Nanofibers; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–47. [Google Scholar]
- Esentürk, İ.; Erdal, M.S.; Güngör, S. Electrospinning method to produce drug-loaded nanofibers for topical/transdermal drug delivery applications. J. Fac. Pharm. Istanb. Univ. 2016, 46, 49–64. [Google Scholar]
- Farokhi, M.; Mottaghitalab, F.; Reis, R.L.; Ramakrishna, S.; Kundu, S.C. Functionalized silk fibroin nanofibers as drug carriers: Advantages and challenges. J. Control. Release 2020, 321, 324–347. [Google Scholar] [CrossRef]
- Agrahari, V.; Agrahari, V.; Meng, J.; Mitra, A.K. Electrospun Nanofibers in Drug Delivery. In Emerging Nanotechnologies for Diagnostics, Drug Delivery and Medical Devices; Elsevier: Amsterdam, The Netherlands, 2017; pp. 189–215. [Google Scholar]
- Kajdič, S.; Planinšek, O.; Gašperlin, M.; Kocbek, P. Electrospun nanofibers for customized drug-delivery systems. J. Drug Deliv. Sci. Technol. 2019, 51, 672–681. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, N.; Liu, M.; Cao, Y.; Wang, K.; Wang, J.; Pei, R. Multifunctional Nanofibers for Specific Purification and Release of CTCs. ACS Sens. 2017, 2, 547–552. [Google Scholar] [CrossRef]
- Li, H.; Liu, K.; Williams, G.R.; Wu, J.; Wu, J.; Wang, H.; Niu, S.; Zhu, L.-M. Dual temperature and pH responsive nanofiber formulations prepared by electrospinning. Colloids Surf. B Biointerfaces 2018, 171, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Zheng, Y.; Wang, G.; Lin, Q.; Fan, J. pH- and electro-response characteristics of bacterial cellulose nanofiber/sodium alginate hybrid hydrogels for dual controlled drug delivery. RSC Adv. 2014, 4, 47056–47065. [Google Scholar] [CrossRef]
- Kenawy, E.-R.; Bowlin, G.L.; Mansfield, K.; Layman, J.; Simpson, D.G.; Sanders, E.H.; Wnek, G.E. Release of tetracycline hydrochloride from electrospun poly(ethylene-co-vinylacetate), poly(lactic acid), and a blend. J. Control. Release 2002, 81, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Jia, X.-W.; Liu, Q.; Kong, B.; Wang, H. Fast dissolving oral films for drug delivery prepared from chitosan/pullulan electrospinning nanofibers. Int. J. Biol. Macromol. 2019, 137, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Khoshnevisan, K.; Maleki, H.; Samadian, H.; Shahsavari, S.; Sarrafzadeh, M.H.; Larijani, B.; Dorkoosh, F.A.; Haghpanah, V.; Khorramizadeh, M.R. Cellulose acetate electrospun nanofibers for drug delivery systems: Applications and recent advances. Carbohydr. Polym. 2018, 198, 131–141. [Google Scholar] [CrossRef]
- Grimaudo, M.A.; Concheiro, A.; Alvarez-Lorenzo, C. Crosslinked Hyaluronan Electrospun Nanofibers for Ferulic Acid Ocular Delivery. Pharmaceutics 2020, 12, 274. [Google Scholar] [CrossRef]
- Sofi, H.S.; Abdal-hay, A.; Ivanovski, S.; Zhang, Y.S.; Sheikh, F.A. Electrospun nanofibers for the delivery of active drugs through nasal, oral and vaginal mucosa: Current status and future perspectives. Mater. Sci. Eng. C 2020, 111, 110756. [Google Scholar] [CrossRef]
- Luraghi, A.; Peri, F.; Moroni, L. Electrospinning for drug delivery applications: A review. J. Control. Release 2021, 334, 463–484. [Google Scholar] [CrossRef]
- Megelski, S.; Stephens, J.S.; Chase, D.B.; Rabolt, J.F. Micro- and Nanostructured Surface Morphology on Electrospun Polymer Fibers. Macromolecules 2002, 35, 8456–8466. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef]
- Shi, X.; Zhou, W.; Ma, D.; Ma, Q.; Bridges, D.; Ma, Y.; Hu, A. Electrospinning of Nanofibers and Their Applications for Energy Devices. J. Nanomater. 2015, 2015, 140716. [Google Scholar] [CrossRef]
- Omer, S.; Forgách, L.; Zelkó, R.; Sebe, I. Scale-up of Electrospinning: Market Overview of Products and Devices for Pharmaceutical and Biomedical Purposes. Pharmaceutics 2021, 13, 286. [Google Scholar] [CrossRef] [PubMed]
- Vass, P.; Szabó, E.; Domokos, A.; Hirsch, E.; Galata, D.; Farkas, B.; Démuth, B.; Andersen, S.K.; Vigh, T.; Verreck, G.; et al. Scale-up of electrospinning technology: Applications in the pharmaceutical industry. WIREs Nanomed. Nanobiotechnol. 2020, 12, e1611. [Google Scholar] [CrossRef]
- Nayak, R.; Padhye, R.; Kyratzis, I.L.; Truong, Y.B.; Arnold, L. Recent advances in nanofibre fabrication techniques. Text. Res. J. 2012, 82, 129–147. [Google Scholar] [CrossRef]
- Sill, T.J.; von Recum, H.A. Electrospinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef]
- Xue, J.; Xie, J.; Liu, W.; Xia, Y. Electrospun Nanofibers: New Concepts, Materials, and Applications. Acc. Chem. Res. 2017, 50, 1976–1987. [Google Scholar] [CrossRef]
- Pant, B.; Park, M.; Park, S.-J. Drug Delivery Applications of Core-Sheath Nanofibers Prepared by Coaxial Electrospinning: A Review. Pharmaceutics 2019, 11, 305. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Li, W.; Zhang, F.; Liu, Z.; Zanjanizadeh Ezazi, N.; Liu, D.; Santos, H.A. Electrospun Fibrous Architectures for Drug Delivery, Tissue Engineering and Cancer Therapy. Adv. Funct. Mater. 2019, 29, 1802852. [Google Scholar] [CrossRef]
- Li, Z.; Wang, C. Effects of Working Parameters on Electrospinning. In One-Dimensional Nanostructures: Electrospinning Technique and Unique Nanofibers; Springer: Berlin/Heidelberg, Germany, 2013; pp. 15–28. [Google Scholar]
- Haider, A.; Haider, S.; Kang, I.-K. A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology. Arab. J. Chem. 2018, 11, 1165–1188. [Google Scholar] [CrossRef]
- Kulkarni, A.; Bambole, V.A.; Mahanwar, P.A. Electrospinning of Polymers, Their Modeling and Applications. Polym. Plast. Technol. Eng. 2010, 49, 427–441. [Google Scholar] [CrossRef]
- Lee, H.; Nishino, M.; Sohn, D.; Lee, J.S.; Kim, I.S. Control of the morphology of cellulose acetate nanofibers via electrospinning. Cellulose 2018, 25, 2829–2837. [Google Scholar] [CrossRef]
- Chen, L.; Jiang, H.; Li, Y.; Zimba, B.L.; Yu, X.; Chen, C.; Xiong, G.; Wu, Q. Influences on mechanical properties of chitosan nanofibrous membranes induced by incorporating graphene oxide nanosheets. Mater. Res. Express 2019, 6, 075404. [Google Scholar] [CrossRef]
- Cheah, W.Y.; Show, P.-L.; Ng, I.-S.; Lin, G.-Y.; Chiu, C.-Y.; Chang, Y.-K. Antibacterial activity of quaternized chitosan modified nanofiber membrane. Int. J. Biol. Macromol. 2019, 126, 569–577. [Google Scholar] [CrossRef]
- Wakuda, Y.; Nishimoto, S.; Suye, S.; Fujita, S. Native collagen hydrogel nanofibres with anisotropic structure using core-shell electrospinning. Sci. Rep. 2018, 8, 6248. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Zhu, Y.; Cui, H. Electrospun thyme essential oil/gelatin nanofibers for active packaging against Campylobacter jejuni in chicken. LWT 2018, 97, 711–718. [Google Scholar] [CrossRef]
- Vilchez, A.; Acevedo, F.; Cea, M.; Seeger, M.; Navia, R. Applications of Electrospun Nanofibers with Antioxidant Properties: A Review. Nanomaterials 2020, 10, 175. [Google Scholar] [CrossRef]
- Yao, Q.; Cosme, J.G.L.; Xu, T.; Miszuk, J.M.; Picciani, P.H.S.; Fong, H.; Sun, H. Three dimensional electrospun PCL/PLA blend nanofibrous scaffolds with significantly improved stem cells osteogenic differentiation and cranial bone formation. Biomaterials 2017, 115, 115–127. [Google Scholar] [CrossRef]
- Shababdoust, A.; Zandi, M.; Ehsani, M.; Shokrollahi, P.; Foudazi, R. Controlled curcumin release from nanofibers based on amphiphilic-block segmented polyurethanes. Int. J. Pharm. 2020, 575, 118947. [Google Scholar] [CrossRef]
- Song, Z.; Chiang, S.W.; Chu, X.; Du, H.; Li, J.; Gan, L.; Xu, C.; Yao, Y.; He, Y.; Li, B.; et al. Effects of solvent on structures and properties of electrospun poly(ethylene oxide) nanofibers. J. Appl. Polym. Sci. 2018, 135, 45787. [Google Scholar] [CrossRef]
- Berthet, M.; Gauthier, Y.; Lacroix, C.; Verrier, B.; Monge, C. Nanoparticle-Based Dressing: The Future of Wound Treatment? Trends Biotechnol. 2017, 35, 770–784. [Google Scholar] [CrossRef]
- McLaughlin, A.W.; McDowell, A.; Clarkson, A.N.; Walker, G.F. Characterization of poly(lactic- co -glycolic acid) nanofibers electrospun using a sustainable green chemistry with a low toxicity solvent system. Int. J. Polym. Mater. Polym. Biomater. 2022, 71, 983–992. [Google Scholar] [CrossRef]
- Dias, Y.J.; Sinha-Ray, S.; Pourdeyhimi, B.; Yarin, A.L. Chicago Sky Blue diazo-dye release from poly(methyl methacrylate) (PMMA) electrospun nanofibers. J. Mol. Liq. 2022, 345, 117771. [Google Scholar] [CrossRef]
- Jiang, L.; Jiang, Y.; Stiadle, J.; Wang, X.; Wang, L.; Li, Q.; Shen, C.; Thibeault, S.L.; Turng, L.-S. Electrospun nanofibrous thermoplastic polyurethane/poly(glycerol sebacate) hybrid scaffolds for vocal fold tissue engineering applications. Mater. Sci. Eng. C 2019, 94, 740–749. [Google Scholar] [CrossRef]
- Li, B.; Xia, X.; Chen, J.; Xia, D.; Xu, R.; Zou, X.; Wang, H.; Liang, C. Paclitaxel-loaded lignin particle encapsulated into electrospun PVA/PVP composite nanofiber for effective cervical cancer cell inhibition. Nanotechnology 2021, 32, 015101. [Google Scholar] [CrossRef]
- Pusporini, P.; Edikresnha, D.; Sriyanti, I.; Suciati, T.; Munir, M.M.; Khairurrijal, K. Electrospun polyvinylpyrrolidone (PVP)/green tea extract composite nanofiber mats and their antioxidant activities. Mater. Res. Express 2018, 5, 054001. [Google Scholar] [CrossRef]
- Kishimoto, Y.; Kobashi, T.; Yamanaka, S.; Morikawa, H.; Tamada, Y. Comparisons between silk fibroin nonwoven electrospun fabrics using aqueous and formic acid solutions. Int. J. Polym. Mater. Polym. Biomater. 2018, 67, 462–467. [Google Scholar] [CrossRef]
- Calori, I.R.; Braga, G.; de Jesus, P.d.C.C.; Bi, H.; Tedesco, A.C. Polymer scaffolds as drug delivery systems. Eur. Polym. J. 2020, 129, 109621. [Google Scholar] [CrossRef]
- Buzgo, M.; Mickova, A.; Rampichova, M.; Doupnik, M. Blend electrospinning, coaxial electrospinning, and emulsion electrospinning techniques. In Core-Shell Nanostructures for Drug Delivery and Theranostics; Elsevier: Amsterdam, The Netherlands, 2018; pp. 325–347. [Google Scholar]
- Tijing, L.D.; Woo, Y.C.; Yao, M.; Ren, J.; Shon, H.K. Electrospinning for Membrane Fabrication: Strategies and Applications. In Comprehensive Membrane Science and Engineering; Elsevier: Amsterdam, The Netherlands, 2017; pp. 418–444. [Google Scholar]
- Yoon, J.; Yang, H.-S.; Lee, B.-S.; Yu, W.-R. Recent Progress in Coaxial Electrospinning: New Parameters, Various Structures, and Wide Applications. Adv. Mater. 2018, 30, 1704765. [Google Scholar] [CrossRef]
- Wei, Z. Research Process of Polymer Nanofibers Prepared by Melt Spinning. IOP Conf. Ser. Mater. Sci. Eng. 2018, 452, 022002. [Google Scholar] [CrossRef]
- Teo, W.E.; Ramakrishna, S. A review on electrospinning design and nanofibre assemblies. Nanotechnology 2006, 17, R89–R106. [Google Scholar] [CrossRef]
- Wang, M.-L.; Yu, D.-G.; Bligh, S.W.A. Progress in preparing electrospun Janus fibers and their applications. Appl. Mater. Today 2023, 31, 101766. [Google Scholar] [CrossRef]
- Steyaert, I.; Van der Schueren, L.; Rahier, H.; de Clerck, K. An Alternative Solvent System for Blend Electrospinning of Polycaprolactone/Chitosan Nanofibres. Macromol. Symp. 2012, 321–322, 71–75. [Google Scholar] [CrossRef]
- Shahriar, S.; Mondal, J.; Hasan, M.; Revuri, V.; Lee, D.; Lee, Y.-K. Electrospinning Nanofibers for Therapeutics Delivery. Nanomaterials 2019, 9, 532. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Li, J.; Zhang, X.; Liu, T.; Ding, J.; Chen, X. Electrospun polymer micro/nanofibers as pharmaceutical repositories for healthcare. J. Control. Release 2019, 302, 19–41. [Google Scholar] [CrossRef] [PubMed]
- Nikmaram, N.; Roohinejad, S.; Hashemi, S.; Koubaa, M.; Barba, F.J.; Abbaspourrad, A.; Greiner, R. Emulsion-based systems for fabrication of electrospun nanofibers: Food, pharmaceutical and biomedical applications. RSC Adv. 2017, 7, 28951–28964. [Google Scholar] [CrossRef]
- McClellan, P.; Landis, W.J. Recent Applications of Coaxial and Emulsion Electrospinning Methods in the Field of Tissue Engineering. Biores. Open Access 2016, 5, 212–227. [Google Scholar] [CrossRef]
- Lauricella, M.; Succi, S.; Zussman, E.; Pisignano, D.; Yarin, A.L. Models of polymer solutions in electrified jets and solution blowing. Rev. Mod. Phys. 2020, 92, 035004. [Google Scholar] [CrossRef]
- Zhmayev, E.; Cho, D.; Joo, Y.L. Nanofibers from gas-assisted polymer melt electrospinning. Polymer 2010, 51, 4140–4144. [Google Scholar] [CrossRef]
- Williams, G.R.; Raimi-Abraham, B.T.; Luo, C. (Eds.) Nanofibres in Drug Delivery; UCL Press: London, UK, 2018; ISBN 9781787350182. [Google Scholar]
- Yang, J.; Wang, K.; Yu, D.-G.; Yang, Y.; Bligh, S.W.A.; Williams, G.R. Electrospun Janus nanofibers loaded with a drug and inorganic nanoparticles as an effective antibacterial wound dressing. Mater. Sci. Eng. C 2020, 111, 110805. [Google Scholar] [CrossRef]
- Miguel, S.P.; Figueira, D.R.; Simões, D.; Ribeiro, M.P.; Coutinho, P.; Ferreira, P.; Correia, I.J. Electrospun polymeric nanofibres as wound dressings: A review. Colloids Surf. B Biointerfaces 2018, 169, 60–71. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Nie, W.; Feng, W. Engineering of biomimetic nanofibrous matrices for drug delivery and tissue engineering. J. Mater. Chem. B 2014, 2, 7828–7848. [Google Scholar] [CrossRef]
- Casper, C.L.; Yamaguchi, N.; Kiick, K.L.; Rabolt, J.F. Functionalizing Electrospun Fibers with Biologically Relevant Macromolecules. Biomacromolecules 2005, 6, 1998–2007. [Google Scholar] [CrossRef] [PubMed]
- Nie, H.; Ho, M.-L.; Wang, C.-K.; Wang, C.-H.; Fu, Y.-C. BMP-2 plasmid loaded PLGA/HAp composite scaffolds for treatment of bone defects in nude mice. Biomaterials 2009, 30, 892–901. [Google Scholar] [CrossRef]
- Jeckson, T.A.; Neo, Y.P.; Sisinthy, S.P.; Gorain, B. Delivery of Therapeutics from Layer-by-Layer Electrospun Nanofiber Matrix for Wound Healing: An Update. J. Pharm. Sci. 2021, 110, 635–653. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Qian, Z.; Liu, D.; Liu, H. Surface Modification of Dental Titanium Implant by Layer-by-Layer Electrostatic Self-Assembly. Front. Physiol. 2017, 8, 574. [Google Scholar] [CrossRef] [PubMed]
- Xin, S.; Li, X.; Wang, Q.; Huang, R.; Xu, X.; Lei, Z.; Deng, H. Novel Layer-by-Layer Structured Nanofibrous Mats Coated by Protein Films for Dermal Regeneration. J. Biomed. Nanotechnol. 2014, 10, 803–810. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Duan, X.-P.; Li, Y.-M.; Yang, D.-P.; Long, Y.-Z. Electrospun nanofibers for wound healing. Mater. Sci. Eng. C 2017, 76, 1413–1423. [Google Scholar] [CrossRef]
- Alghoraibi, I.; Alomari, S. Different Methods for Nanofiber Design and Fabrication. In Handbook of Nanofibers; Springer International Publishing: Cham, Switzerland, 2019; pp. 79–124. [Google Scholar]
- Beachley, V.; Wen, X. Polymer nanofibrous structures: Fabrication, biofunctionalization, and cell interactions. Prog. Polym. Sci. 2010, 35, 868–892. [Google Scholar] [CrossRef]
- Almetwally, A.A.; El-Sakhawy, M.; Elshakankery, M.H.; Kasem, M.H. Technology of nano-fibers: Production techniques and properties-Critical review. J. Text. Assoc. 2017, 78, 5–14. [Google Scholar]
- Zhang, X.; Lu, Y. Centrifugal Spinning: An Alternative Approach to Fabricate Nanofibers at High Speed and Low Cost. Polym. Rev. 2014, 54, 677–701. [Google Scholar] [CrossRef]
- Zhang, W.; Yu, X.; Li, Y.; Su, Z.; Jandt, K.D.; Wei, G. Protein-mimetic peptide nanofibers: Motif design, self-assembly synthesis, and sequence-specific biomedical applications. Prog. Polym. Sci. 2018, 80, 94–124. [Google Scholar] [CrossRef]
- Lu, T.; Li, Y.; Chen, T. Techniques for fabrication and construction of three-dimensional scaffolds for tissue engineering. Int. J. Nanomed. 2013, 8, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wakisaka, M. Nanofiber Fabrication Techniques and Its Applicability to Chitosan. Prog. Chem. 2014, 26, 1821–1831. [Google Scholar] [CrossRef]
- Hossain, M.S.; Nakane, K. Development of a high-performance heat dissipation sheet with three-dimensional alumina fibrous structure fabricated by freeze-drying. Results Mater. 2022, 13, 100241. [Google Scholar] [CrossRef]
- Yang, H.; Sugita, N.; Nakane, K. Factors influencing the PVA polymer-assisted freeze-drying synthesis of Al2O3 nanofibers. Ceram. Int. 2019, 45, 16731–16739. [Google Scholar] [CrossRef]
- Vetrik, M.; Pradny, M.; Kobera, L.; Slouf, M.; Rabyk, M.; Pospisilova, A.; Stepanek, P.; Hruby, M. Biopolymer-based degradable nanofibres from renewable resources produced by freeze-drying. RSC Adv. 2013, 3, 15282. [Google Scholar] [CrossRef]
- Ma, G.; Wang, Z.; Chen, J.; Yin, R.; Chen, B.; Nie, J. Freeze-dried chitosan–sodium hyaluronate polyelectrolyte complex fibers as tissue engineering scaffolds. New J. Chem. 2014, 38, 1211. [Google Scholar] [CrossRef]
- Cai, X.; Luan, Y.; Dong, Q.; Shao, W.; Li, Z.; Zhao, Z. Sustained release of 5-fluorouracil by incorporation into sodium carboxymethylcellulose sub-micron fibers. Int. J. Pharm. 2011, 419, 240–246. [Google Scholar] [CrossRef]
- Lee, D.; Zhang, C.; Gao, H. Facile Production of Polypyrrole Nanofibers Using a Freeze-Drying Method. Macromol. Chem. Phys. 2014, 215, 669–674. [Google Scholar] [CrossRef]
- Lin, J.; Xu, L.; Huang, Y.; Li, J.; Wang, W.; Feng, C.; Liu, Z.; Xu, X.; Zou, J.; Tang, C. Ultrafine porous boron nitride nanofibers synthesized via a freeze-drying and pyrolysis process and their adsorption properties. RSC Adv. 2016, 6, 1253–1259. [Google Scholar] [CrossRef]
- Hokmabad, V.R.; Davaran, S.; Aghazadeh, M.; Rahbarghazi, R.; Salehi, R.; Ramazani, A. Fabrication and characterization of novel ethyl cellulose-grafted-poly (ɛ-caprolactone)/alginate nanofibrous/macroporous scaffolds incorporated with nano-hydroxyapatite for bone tissue engineering. J. Biomater. Appl. 2019, 33, 1128–1144. [Google Scholar] [CrossRef]
- Xu, T.; Miszuk, J.M.; Zhao, Y.; Sun, H.; Fong, H. Electrospun Polycaprolactone 3D Nanofibrous Scaffold with Interconnected and Hierarchically Structured Pores for Bone Tissue Engineering. Adv. Healthc. Mater. 2015, 4, 2238–2246. [Google Scholar] [CrossRef]
- Tang, Y.; Liu, Z.; Zhao, K. Fabrication of hollow and porous polystyrene fibrous membranes by electrospinning combined with freeze-drying for oil removal from water. J. Appl. Polym. Sci. 2019, 136, 47262. [Google Scholar] [CrossRef]
- Stojanovska, E.; Canbay, E.; Pampal, E.S.; Calisir, M.D.; Agma, O.; Polat, Y.; Simsek, R.; Gundogdu, N.A.S.; Akgul, Y.; Kilic, A. A review on non-electro nanofibre spinning techniques. RSC Adv. 2016, 6, 83783–83801. [Google Scholar] [CrossRef]
- Xu, H.; Yagi, S.; Ashour, S.; Du, L.; Hoque, M.E.; Tan, L. A Review on Current Nanofiber Technologies: Electrospinning, Centrifugal Spinning, and Electro-Centrifugal Spinning. Macromol. Mater. Eng. 2022, 308, 2200502. [Google Scholar] [CrossRef]
- Hammami, M.A.; Krifa, M.; Harzallah, O. Centrifugal force spinning of PA6 nanofibers—Processability and morphology of solution-spun fibers. J. Text. Inst. 2014, 105, 637–647. [Google Scholar] [CrossRef]
- Edmondson, D.; Cooper, A.; Jana, S.; Wood, D.; Zhang, M. Centrifugal electrospinning of highly aligned polymer nanofibers over a large area. J. Mater. Chem. 2012, 22, 18646. [Google Scholar] [CrossRef]
- Ren, L.; Ozisik, R.; Kotha, S.P.; Underhill, P.T. Highly Efficient Fabrication of Polymer Nanofiber Assembly by Centrifugal Jet Spinning: Process and Characterization. Macromolecules 2015, 48, 2593–2602. [Google Scholar] [CrossRef]
- Duan, Y.; Zhang, Z.; Lu, B.; Chen, B.; Lai, Z. The movement and forces of spinning solution in the nozzle during high-speed centrifugal spinning. J. Eng. Fiber. Fabr. 2019, 14, 155892501982820. [Google Scholar] [CrossRef]
- Gholipour-Kanani, A.; Daneshi, P. A Review on Centrifugal and Electro-Centrifugal Spinning as New Methods of Nanofibers Fabrication. J. Text. Polym. 2022, 10, 41–55. [Google Scholar] [CrossRef]
- Bahú, J.O.; Melo de Andrade, L.R.; Crivellin, S.; Khouri, N.G.; Sousa, S.O.; Fernandes, L.M.I.; Souza, S.D.A.; Concha, L.S.C.; Schiavon, M.I.R.B.; Benites, C.I.; et al. Rotary Jet Spinning (RJS): A Key Process to Produce Biopolymeric Wound Dressings. Pharmaceutics 2022, 14, 2500. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, G.M.; MacQueen, L.A.; Lind, J.U.; Fitzgibbons, S.A.; Chantre, C.O.; Huggler, I.; Golecki, H.M.; Goss, J.A.; Parker, K.K. Production of Synthetic, Para-Aramid and Biopolymer Nanofibers by Immersion Rotary Jet-Spinning. Macromol. Mater. Eng. 2017, 302, 1600365. [Google Scholar] [CrossRef]
- Szabó, P.; Sebe, I.; Stiedl, B.; Kállai-Szabó, B.; Zelkó, R. Tracking of crystalline-amorphous transition of carvedilol in rotary spun microfibers and their formulation to orodispersible tablets for in vitro dissolution enhancement. J. Pharm. Biomed. Anal. 2015, 115, 359–367. [Google Scholar] [CrossRef]
- Loordhuswamy, A.M.; Krishnaswamy, V.R.; Korrapati, P.S.; Thinakaran, S.; Rengaswami, G.D.V. Fabrication of highly aligned fibrous scaffolds for tissue regeneration by centrifugal spinning technology. Mater. Sci. Eng. C 2014, 42, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Badrossamay, M.R.; Balachandran, K.; Capulli, A.K.; Golecki, H.M.; Agarwal, A.; Goss, J.A.; Kim, H.; Shin, K.; Parker, K.K. Engineering hybrid polymer-protein super-aligned nanofibers via rotary jet spinning. Biomaterials 2014, 35, 3188–3197. [Google Scholar] [CrossRef]
- Khang, A.; Ravishankar, P.; Krishnaswamy, A.; Anderson, P.K.; Cone, S.G.; Liu, Z.; Qian, X.; Balachandran, K. Engineering anisotropic biphasic Janus-type polymer nanofiber scaffold networks via centrifugal jet spinning. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 2455–2464. [Google Scholar] [CrossRef]
- Ravishankar, P.; Khang, A.; Laredo, M.; Balachandran, K. Using Dimensionless Numbers to Predict Centrifugal Jet-Spun Nanofiber Morphology. J. Nanomater. 2019, 2019, 4639658. [Google Scholar] [CrossRef]
- Durgun, M.E.; Güngör, S.; Özsoy, Y. Anti-Infective Agents in Ocular Treatment and New Approaches. In Frontiers in Anti-Infective Agents; Bentham Science Publishers Pte. Ltd.: Singapore, 2021; pp. 1–41. [Google Scholar]
- Kansara, V.; Pal, D.; Jain, R.; Mitra, A.K. Identification and Functional Characterization of Riboflavin Transporter in Human-Derived Retinoblastoma Cell Line (Y-79): Mechanisms of Cellular Uptake and Translocation. J. Ocul. Pharmacol. Ther. 2005, 21, 275–287. [Google Scholar] [CrossRef]
- Nettey, H.; Darko, Y.; Bamiro, O.A.; Addo, R.T. Ocular Barriers. In Ocular Drug Delivery: Advances, Challenges and Applications; Springer International Publishing: Cham, Switzerland, 2016; pp. 27–36. [Google Scholar]
- Stenkamp, D.L.; Cameron, D.A. Cellular pattern formation in the retina: Retinal regeneration as a model system. Mol. Vis. 2002, 8, 280–293. [Google Scholar]
- Rupenthal, I.D.; Alany, R.G. Ocular Drug Delivery. In Pharmaceutical Manufacturing Handbook; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 729–767. [Google Scholar]
- Durairaj, C. Ocular Pharmacokinetics. In Pharmacologic Therapy of Ocular Disease; Whitcup, S., Azar, D., Eds.; Springer: Cham, Switzerland, 2016; Volume 242, pp. 31–55. ISBN 978-3-319-58288-7. [Google Scholar]
- Urtti, A. Challenges and obstacles of ocular pharmacokinetics and drug delivery. Adv. Drug Deliv. Rev. 2006, 58, 1131–1135. [Google Scholar] [CrossRef]
- Winter, K.N.; Anderson, D.M.; Braun, R.J. A model for wetting and evaporation of a post-blink precorneal tear film. Math. Med. Biol. 2010, 27, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.W.; Nadeem, A. The Lacrimal Apparatus. In Atlas of Ocular Anatomy; Springer International Publishing: Cham, Switzerland, 2016; pp. 71–76. [Google Scholar]
- Vadlapudi, A.D.; Cholkar, K.; Vadlapatla, R.K.; Mitra, A.K. Aqueous Nanomicellar Formulation for Topical Delivery of Biotinylated Lipid Prodrug of Acyclovir: Formulation Development and Ocular Biocompatibility. J. Ocul. Pharmacol. Ther. 2014, 30, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Dey, S.; Mitra, A.K.; Krishnamoorthy, R. Ocular delivery and therapeutics of proteins and peptides. In Ophthalmic Drug Delivery Systems, 2nd ed.; Revis. Expand.; Routledge: London, UK, 2003; pp. 493–514. [Google Scholar] [CrossRef]
- Durgun, M.E.; Mesut, B.; Hacıoğlu, M.; Güngör, S.; Özsoy, Y. Optimization of the Micellar-Based In Situ Gelling Systems Posaconazole with Quality by Design (QbD) Approach and Characterization by In Vitro Studies. Pharmaceutics 2022, 14, 526. [Google Scholar] [CrossRef]
- Sunkara, G.; Kompella, U.B. Membrane transport processes in the eye. In Ophthalmic Drug Delivery Systems, 2nd ed.; Revis. Expand.; Routledge: London, UK, 2003; pp. 13–58. [Google Scholar] [CrossRef]
- Olsen, T.W.; Aaberg, S.Y.; Geroski, D.H.; Edelhauser, H.F. Human sclera: Thickness and surface area. Am. J. Ophthalmol. 1998, 125, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Pelis, R.M. Drug transport by the blood-aqueous humor barrier of the eye. Drug Metab. Dispos. 2016, 44, 1675–1681. [Google Scholar] [CrossRef]
- Coca-Prados, M. The blood-aqueous barrier in health and disease. J. Glaucoma 2014, 23, S36–S38. [Google Scholar] [CrossRef]
- Wadhwa, S.; Paliwal, R.; Paliwal, S.; Vyas, S. Nanocarriers in Ocular Drug Delivery: An Update Review. Curr. Pharm. Des. 2009, 15, 2724–2750. [Google Scholar] [CrossRef]
- Gaudana, R.; Jwala, J.; Boddu, S.H.S.; Mitra, A.K. Recent perspectives in ocular drug delivery. Pharm. Res. 2009, 26, 1197–1216. [Google Scholar] [CrossRef]
- Kahraman, E.; Durgun, M.E.; Güngör, S.; Özsoy, Y. Polymeric micellar nanocarriers: Topical treatment of inflammatory diseases. In Polymeric Micelles for Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2022; pp. 115–143. [Google Scholar]
- Velpandian, T.; Gupta, S.K. Ocular Pharmacology and Therapeutics: Origin, Principle, Challenges, and Practices. In Pharmacology of Ocular Therapeutics; Springer International Publishing: Cham, Switzerland, 2016; pp. 1–11. [Google Scholar]
- Kahraman, E.; Ÿzhan, G.; Ÿzsoy, Y.; Güngör, S. Polymeric micellar nanocarriers of benzoyl peroxide as potential follicular targeting approach for acne treatment. Colloids Surf. B Biointerfaces 2016, 146, 692–699. [Google Scholar] [CrossRef]
- Torchilin, V.P. Structure and design of polymeric surfactant-based drug delivery systems. J. Control. Release 2001, 73, 137–172. [Google Scholar] [CrossRef]
- Damiani, G.; Eggenhöffner, R.; Pigatto, P.D.M.; Bragazzi, N.L. Nanotechnology meets atopic dermatitis: Current solutions, challenges and future prospects. Insights and implications from a systematic review of the literature. Bioact. Mater. 2019, 4, 380–386. [Google Scholar] [CrossRef]
- Jain, A.K.; Thareja, S. In vitro and in vivo characterization of pharmaceutical nanocarriers used for drug delivery. Artif. Cells Nanomed. Biotechnol. 2019, 47, 524–539. [Google Scholar] [CrossRef] [PubMed]
- Bennett, L. Topical Versus Systemic Ocular Drug Delivery. In Ocular Drug Delivery: Advances, Challenges and Applications; Springer International Publishing: Cham, Switzerland, 2016; pp. 53–74. [Google Scholar]
- Walimbe, T.; Chelerkar, V.; Bhagat, P.; Joshi, A.; Raut, A. Effect of benzalkonium chloride-free latanoprost ophthalmic solution on ocular surface in patients with glaucoma. Clin. Ophthalmol. 2016, 10, 821–827. [Google Scholar] [CrossRef]
- Lallemand, F.; Schmitt, M.; Bourges, J.-L.; Gurny, R.; Benita, S.; Garrigue, J.-S. Cyclosporine A delivery to the eye: A comprehensive review of academic and industrial efforts. Eur. J. Pharm. Biopharm. 2017, 117, 14–28. [Google Scholar] [CrossRef]
- Mandal, A.; Gote, V.; Pal, D.; Ogundele, A.; Mitra, A.K. Ocular Pharmacokinetics of a Topical Ophthalmic Nanomicellar Solution of Cyclosporine (Cequa®) for Dry Eye Disease. Pharm. Res. 2019, 36, 36. [Google Scholar] [CrossRef] [PubMed]
- Gan, L.; Wang, J.; Jiang, M.; Bartlett, H.; Ouyang, D.; Eperjesi, F.; Liu, J.; Gan, Y. Recent advances in topical ophthalmic drug delivery with lipid-based nanocarriers. Drug Discov. Today 2013, 18, 290–297. [Google Scholar] [CrossRef]
- Goyal, R.; Macri, L.K.; Kaplan, H.M.; Kohn, J. Nanoparticles and nanofibers for topical drug delivery. J. Control. Release 2016, 240, 77–92. [Google Scholar] [CrossRef]
- Daley, W.P.; Peters, S.B.; Larsen, M. Extracellular matrix dynamics in development and regenerative medicine. J. Cell Sci. 2008, 121, 255–264. [Google Scholar] [CrossRef]
- Doostmohammadi, M.; Forootanfar, H.; Ramakrishna, S. Regenerative medicine and drug delivery: Progress via electrospun biomaterials. Mater. Sci. Eng. C 2020, 109, 110521. [Google Scholar] [CrossRef] [PubMed]
- Rosso, F.; Giordano, A.; Barbarisi, M.; Barbarisi, A. From Cell-ECM interactions to tissue engineering. J. Cell. Physiol. 2004, 199, 174–180. [Google Scholar] [CrossRef]
- Pepic, I.; Lovric, J.; Filipovic-Grcic, J. Polymeric Micelles in Ocular Drug Delivery: Rationale, Strategies and Challenges. Chem. Biochem. Eng. Q. 2012, 26, 365. [Google Scholar]
- Deepak, A.; Goyal, A.K.; Rath, G. Nanofiber in transmucosal drug delivery. J. Drug Deliv. Sci. Technol. 2018, 43, 379–387. [Google Scholar] [CrossRef]
- Göttel, B.; Lucas, H.; Syrowatka, F.; Knolle, W.; Kuntsche, J.; Heinzelmann, J.; Viestenz, A.; Mäder, K. In Situ Gelling Amphotericin B Nanofibers: A New Option for the Treatment of Keratomycosis. Front. Bioeng. Biotechnol. 2020, 8, 600384. [Google Scholar] [CrossRef] [PubMed]
- Khalil, I.A.; Ali, I.H.; El-Sherbiny, I.M. Noninvasive biodegradable nanoparticles-in-nanofibers single-dose ocular insert: In Vitro, Ex Vivo and In Vivo evaluation. Nanomedicine 2019, 14, 33–55. [Google Scholar] [CrossRef] [PubMed]
- Polat, H.K.; Bozdağ Pehlivan, S.; Özkul, C.; Çalamak, S.; Öztürk, N.; Aytekin, E.; Fırat, A.; Ulubayram, K.; Kocabeyoğlu, S.; İrkeç, M.; et al. Development of besifloxacin HCl loaded nanofibrous ocular inserts for the treatment of bacterial keratitis: In Vitro, Ex Vivo and In Vivo evaluation. Int. J. Pharm. 2020, 585, 119552. [Google Scholar] [CrossRef]
- De Souza, S.O.L.; Guerra, M.C.A.; Heneine, L.G.D.; de Oliveira, C.R.; Junior, A.d.S.C.; Fialho, S.L.; Oréfice, R.L. Biodegradable core-shell electrospun nanofibers containing bevacizumab to treat age-related macular degeneration. J. Mater. Sci. Mater. Med. 2018, 29, 173. [Google Scholar] [CrossRef]
- Lancina, M.G.; Singh, S.; Kompella, U.B.; Husain, S.; Yang, H. Fast Dissolving Dendrimer Nanofiber Mats as Alternative to Eye Drops for More Efficient Antiglaucoma Drug Delivery. ACS Biomater. Sci. Eng. 2017, 3, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Cegielska, O.; Sierakowski, M.; Sajkiewicz, P.; Lorenz, K.; Kogermann, K. Mucoadhesive brinzolamide-loaded nanofibers for alternative glaucoma treatment. Eur. J. Pharm. Biopharm. 2022, 180, 48–62. [Google Scholar] [CrossRef]
- Cejkova, J.; Cejka, C.; Trosan, P.; Zajicova, A.; Sykova, E.; Holan, V. Treatment of alkali-injured cornea by cyclosporine A-loaded electrospun nanofibers—An alternative mode of therapy. Exp. Eye Res. 2016, 147, 128–137. [Google Scholar] [CrossRef]
- Bhattarai, R.S.; Das, A.; Alzhrani, R.M.; Kang, D.; Bhaduri, S.B.; Boddu, S.H.S. Comparison of electrospun and solvent cast polylactic acid (PLA)/poly(vinyl alcohol) (PVA) inserts as potential ocular drug delivery vehicles. Mater. Sci. Eng. C 2017, 77, 895–903. [Google Scholar] [CrossRef]
- Zhang, Z.; Yu, J.; Zhou, Y.; Zhang, R.; Song, Q.; Lei, L.; Li, X. Supramolecular nanofibers of dexamethasone derivatives to form hydrogel for topical ocular drug delivery. Colloids Surf. B Biointerfaces 2018, 164, 436–443. [Google Scholar] [CrossRef]
- Rohde, F.; Walther, M.; Wächter, J.; Knetzger, N.; Lotz, C.; Windbergs, M. In-situ tear fluid dissolving nanofibers enable prolonged viscosity-enhanced dual drug delivery to the eye. Int. J. Pharm. 2022, 616, 121513. [Google Scholar] [CrossRef]
- Park, C.G.; Kim, Y.K.; Kim, S.-N.; Lee, S.H.; Huh, B.K.; Park, M.-A.; Won, H.; Park, K.H.; Choy, Y. Bin Enhanced ocular efficacy of topically-delivered dorzolamide with nanostructured mucoadhesive microparticles. Int. J. Pharm. 2017, 522, 66–73. [Google Scholar] [CrossRef]
- Li, K.; Li, R.; Zou, P.; Li, L.; Wang, H.; Kong, D.; Zheng, G.; Li, L.-L. Glycopeptide-nanotransforrs eyedrops with enhanced permeability and retention for preventing fundus neovascularization. Biomaterials 2022, 281, 121361. [Google Scholar] [CrossRef]
- Singla, J.; Bajaj, T.; Goyal, A.K.; Rath, G. Development of Nanofibrous Ocular Insert for Retinal Delivery of Fluocinolone Acetonide. Curr. Eye Res. 2019, 44, 541–550. [Google Scholar] [CrossRef]
- Yellanki, S.K.; Anna, B.; Kishan, M.R. Preparation and in vivo evaluation of sodium alginate—Poly (vinyl alcohol) electrospun nanofibers of forskolin for glaucoma treatment. Pak. J. Pharm. Sci. 2019, 32, 669–674. [Google Scholar] [PubMed]
- Mehrandish, S.; Mohammadi, G.; Mirzaeei, S. Preparation and functional evaluation of electrospun polymeric nanofibers as a new system for sustained topical ocular delivery of itraconazole. Pharm. Dev. Technol. 2022, 27, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Zhang, S.; Yu, F.; Gong, D.; Lin, J.; Yao, Q.; Fu, Y. Insight into levofloxacin loaded biocompatible electrospun scaffolds for their potential as conjunctival substitutes. Carbohydr. Polym. 2021, 269, 118341. [Google Scholar] [CrossRef] [PubMed]
- Parikh, K.S.; Omiadze, R.; Josyula, A.; Shi, R.; Anders, N.M.; He, P.; Yazdi, Y.; McDonnell, P.J.; Ensign, L.M.; Hanes, J. Ultra-thin, high strength, antibiotic-eluting sutures for prevention of ophthalmic infection. Bioeng. Transl. Med. 2021, 6, e10204. [Google Scholar] [CrossRef]
- Tawfik, E.A.; Alshamsan, A.; Abul Kalam, M.; Raish, M.; Alkholief, M.; Stapleton, P.; Harvey, K.; Craig, D.Q.M.; Barker, S.A. In vitro and in vivo biological assessment of dual drug-loaded coaxial nanofibers for the treatment of corneal abrasion. Int. J. Pharm. 2021, 604, 120732. [Google Scholar] [CrossRef]
- Mirzaeei, S.; Taghe, S.; Asare-Addo, K.; Nokhodchi, A. Polyvinyl Alcohol/Chitosan Single-Layered and Polyvinyl Alcohol/Chitosan/Eudragit RL100 Multi-layered Electrospun Nanofibers as an Ocular Matrix for the Controlled Release of Ofloxacin: An In Vitro and In Vivo Evaluation. AAPS PharmSciTech 2021, 22, 170. [Google Scholar] [CrossRef]
- Yan, D.; Yao, Q.; Yu, F.; Chen, L.; Zhang, S.; Sun, H.; Lin, J.; Fu, Y. Surface modified electrospun poly(lactic acid) fibrous scaffold with cellulose nanofibrils and Ag nanoparticles for ocular cell proliferation and antimicrobial application. Mater. Sci. Eng. C 2020, 111, 110767. [Google Scholar] [CrossRef] [PubMed]
- Mirzaeei, S.; Berenjian, K.; Khazaei, R. Preparation of the Potential Ocular Inserts by Electrospinning Method to Achieve the Prolong Release Profile of Triamcinolone Acetonide. Adv. Pharm. Bull. 2018, 8, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; Al-Kinani, A.A.; Arshad, M.S.; Chang, M.-W.; Alany, R.G.; Ahmad, Z. Development and characterisation of electrospun timolol maleate-loaded polymeric contact lens coatings containing various permeation enhancers. Int. J. Pharm. 2017, 532, 408–420. [Google Scholar] [CrossRef]
- Andreadis, I.I.; Karavasili, C.; Thomas, A.; Komnenou, A.; Tzimtzimis, M.; Tzetzis, D.; Andreadis, D.; Bouropoulos, N.; Fatouros, D.G. In Situ Gelling Electrospun Ocular Films Sustain the Intraocular Pressure-Lowering Effect of Timolol Maleate: In Vitro, Ex Vivo, and Pharmacodynamic Assessment. Mol. Pharm. 2022, 19, 274–286. [Google Scholar] [CrossRef] [PubMed]
- Karavasili, C.; Komnenou, A.; Katsamenis, O.L.; Charalampidou, G.; Kofidou, E.; Andreadis, D.; Koutsopoulos, S.; Fatouros, D.G. Self-Assembling Peptide Nanofiber Hydrogels for Controlled Ocular Delivery of Timolol Maleate. ACS Biomater. Sci. Eng. 2017, 3, 3386–3394. [Google Scholar] [CrossRef] [PubMed]
- Taka, E.; Karavasili, C.; Bouropoulos, N.; Moschakis, T.; Andreadis, D.D.; Zacharis, C.K.; Fatouros, D.G. Ocular Co-Delivery of Timolol and Brimonidine from a Self-Assembling Peptide Hydrogel for the Treatment of Glaucoma: In Vitro and Ex Vivo Evaluation. Pharmaceuticals 2020, 13, 126. [Google Scholar] [CrossRef]
- Moghanizadeh-Ashkezari, M.; Shokrollahi, P.; Zandi, M.; Shokrolahi, F.; Daliri, M.J.; Kanavi, M.R.; Balagholi, S. Vitamin C Loaded Poly(urethane-urea)/ZnAl-LDH Aligned Scaffolds Increase Proliferation of Corneal Keratocytes and Up-Regulate Vimentin Secretion. ACS Appl. Mater. Interfaces 2019, 11, 35525–35539. [Google Scholar] [CrossRef]
- Palispis, W.A.; Gupta, R. Surgical repair in humans after traumatic nerve injury provides limited functional neural regeneration in adults. Exp. Neurol. 2017, 290, 106–114. [Google Scholar] [CrossRef]
- Oerlemans, A.J.; van Hoek, M.E.; van Leeuwen, E.; Dekkers, W.J. Hype and expectations in tissue engineering. Regen. Med. 2014, 9, 113–122. [Google Scholar] [CrossRef]
- Kim, Y.; Ko, H.; Kwon, I.K.; Shin, K. Extracellular Matrix Revisited: Roles in Tissue Engineering. Int. Neurourol. J. 2016, 20, S23–S29. [Google Scholar] [CrossRef] [PubMed]
- Beuerman, R.W.; Pedroza, L. Ultrastructure of the human cornea. Microsc. Res. Tech. 1996, 33, 320–335. [Google Scholar] [CrossRef]
- Eghrari, A.O.; Riazuddin, S.A.; Gottsch, J.D. Overview of the Cornea. Prog. Mol. Biol. Transl. Sci. 2015, 134, 7–23. [Google Scholar]
- Baradaran-Rafii, A.; Biazar, E.; Heidari-keshel, S. Cellular Response of Stem Cells on Nanofibrous Scaffold for Ocular Surface Bioengineering. ASAIO J. 2015, 61, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Mohanty, S.; Gupta, D.; Jassal, M.; Agrawal, A.K.; Tandon, R. Cellular response of limbal epithelial cells on electrospun poly-ε-caprolactone nanofibrous scaffolds for ocular surface bioengineering: A preliminary in vitro study. Mol. Vis. 2011, 17, 2898–2910. [Google Scholar]
- Liu, H.; Zhou, Z.; Lin, H.; Wu, J.; Ginn, B.; Choi, J.S.; Jiang, X.; Chung, L.; Elisseeff, J.H.; Yiu, S.; et al. Synthetic Nanofiber-Reinforced Amniotic Membrane via Interfacial Bonding. ACS Appl. Mater. Interfaces 2018, 10, 14559–14569. [Google Scholar] [CrossRef]
- Zajicova, A.; Pokorna, K.; Lencova, A.; Krulova, M.; Svobodova, E.; Kubinova, S.; Sykova, E.; Pradny, M.; Michalek, J.; Svobodova, J.; et al. Treatment of Ocular Surface Injuries by Limbal and Mesenchymal Stem Cells Growing on Nanofiber Scaffolds. Cell Transplant. 2010, 19, 1281–1290. [Google Scholar] [CrossRef]
- Ye, J.; Shi, X.; Chen, X.; Xie, J.; Wang, C.; Yao, K.; Gao, C.; Gou, Z. Chitosan-modified, collagen-based biomimetic nanofibrous membranes as selective cell adhering wound dressings in the treatment of chemically burned corneas. J. Mater. Chem. B 2014, 2, 4226–4236. [Google Scholar] [CrossRef]
- Zhou, Z.; Long, D.; Hsu, C.-C.; Liu, H.; Chen, L.; Slavin, B.; Lin, H.; Li, X.; Tang, J.; Yiu, S.; et al. Nanofiber-reinforced decellularized amniotic membrane improves limbal stem cell transplantation in a rabbit model of corneal epithelial defect. Acta Biomater. 2019, 97, 310–320. [Google Scholar] [CrossRef]
- Holan, V.; Trosan, P.; Cejka, C.; Javorkova, E.; Zajicova, A.; Hermankova, B.; Chudickova, M.; Cejkova, J. A Comparative Study of the Therapeutic Potential of Mesenchymal Stem Cells and Limbal Epithelial Stem Cells for Ocular Surface Reconstruction. Stem Cells Transl. Med. 2015, 4, 1052–1063. [Google Scholar] [CrossRef]
- Krishna, L.; Nilawar, S.; Ponnalagu, M.; Subramani, M.; Jayadev, C.; Shetty, R.; Chatterjee, K.; Das, D. Fiber Diameter Differentially Regulates Function of Retinal Pigment and Corneal Epithelial Cells on Nanofibrous Tissue Scaffolds. ACS Appl. Bio Mater. 2020, 3, 823–837. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Hu, Y.; Yu, F.; Zhang, W.; Fu, Y. A novel application of electrospun silk fibroin/poly(l-lactic acid-co-ε-caprolactone) scaffolds for conjunctiva reconstruction. RSC Adv. 2018, 8, 18372–18380. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zhao, B.; Liu, X.; Li, X.; Zeng, C.; Shi, H.; Xu, X.; Lin, T.; Dai, L.; Liu, Y. Aligned Nanofibers from Polypyrrole/Graphene as Electrodes for Regeneration of Optic Nerve via Electrical Stimulation. ACS Appl. Mater. Interfaces 2016, 8, 6834–6840. [Google Scholar] [CrossRef] [PubMed]
- Jasty, S.; Suriyanarayanan, S.; Krishnakumar, S. Influence of self-assembling peptide nanofibre scaffolds on retinal differentiation potential of stem/progenitor cells derived from ciliary pigment epithelial cells. J. Tissue Eng. Regen. Med. 2017, 11, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Baradaran-Rafii, A.; Biazar, E.; Heidari-keshel, S. Cellular Response of Limbal Stem Cells on Polycaprolactone Nanofibrous Scaffolds for Ocular Epithelial Regeneration. Curr. Eye Res. 2015, 41, 326–333. [Google Scholar] [CrossRef]
- Lai, J.-Y.; Lue, S.J.; Cheng, H.-Y.; Ma, D.H.-K. Effect of Matrix Nanostructure on the Functionality of Carbodiimide Cross-Linked Amniotic Membranes as Limbal Epithelial Cell Scaffolds. J. Biomed. Nanotechnol. 2013, 9, 2048–2062. [Google Scholar] [CrossRef]
- Sharma, S.; Gupta, D.; Mohanty, S.; Jassal, M.; Agrawal, A.K.; Tandon, R. Surface-Modified Electrospun Poly(ε-Caprolactone) Scaffold With Improved Optical Transparency and Bioactivity for Damaged Ocular Surface Reconstruction. Investig. Opthalmol. Vis. Sci. 2014, 55, 899. [Google Scholar] [CrossRef]
- Biazar, E.; Baradaran-Rafii, A.; Heidari-keshel, S.; Tavakolifard, S. Oriented nanofibrous silk as a natural scaffold for ocular epithelial regeneration. J. Biomater. Sci. Polym. Ed. 2015, 26, 1139–1151. [Google Scholar] [CrossRef]
- Sun, N.; Dou, X.; Tang, Z.; Zhang, D.; Ni, N.; Wang, J.; Gao, H.; Ju, Y.; Dai, X.; Zhao, C.; et al. Bio-inspired chiral self-assemblies promoted neuronal differentiation of retinal progenitor cells through activation of metabolic pathway. Bioact. Mater. 2021, 6, 990–997. [Google Scholar] [CrossRef]
- Jung, S.; Pant, B.; Climans, M.; Curtis Shaw, G.; Lee, E.-J.; Kim, N.; Park, M. Transformation of electrospun Keratin/PVA nanofiber membranes into multilayered 3D Scaffolds: Physiochemical studies and corneal implant applications. Int. J. Pharm. 2021, 610, 121228. [Google Scholar] [CrossRef]
- Fee, T.; Surianarayanan, S.; Downs, C.; Zhou, Y.; Berry, J. Nanofiber Alignment Regulates NIH3T3 Cell Orientation and Cytoskeletal Gene Expression on Electrospun PCL+Gelatin Nanofibers. PLoS ONE 2016, 11, e0154806. [Google Scholar] [CrossRef] [PubMed]
- Senturk, B.; Cubuk, M.O.; Ozmen, M.C.; Aydin, B.; Guler, M.O.; Tekinay, A.B. Inhibition of VEGF mediated corneal neovascularization by anti-angiogenic peptide nanofibers. Biomaterials 2016, 107, 124–132. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).