Moisture Behavior of Pharmaceutical Powder during the Tableting Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Heat Capacity of the Material
2.2.2. Thermal Diffusivity and Conductivity
2.2.3. Powder Retention/Sorption Curve
2.2.4. Relative Tablet Density
2.2.5. Tableting Process
2.2.6. Tablet Surface and Peripheral
2.2.7. NIR-Penetration Depth
2.2.8. Determination of Relative Tablet Moisture Content Density
2.2.9. Tablet Cohesion and Internal Friction Angle Density
3. Numerical Method
3.1. Simulation Model for the Mechanical Behavior of the Powder
3.1.1. Density-Dependent Drucker–Prager Cap Model
- -
- The Mohr–Coulomb shear failure surface (Fs), representing shear flow, defined as
- -
- The transition surface (Ft), representing a mathematical smoothing surface, defined as
- -
- The cap yield surface (Fc) defined as
- -
- The cap evolution parameter, Pa, that represents the volumetric plastic strain-driven hardening/softening, defined as
- -
- The cap eccentricity, R, is a material parameter between 0.0001 and 1000 that controls the shape of the cap and is defined as
- -
- The hardening/softening law is a user-defined piecewise linear function relating the hydrostatic compression yield stress, Pb, and the corresponding volumetric inelastic/plastic strain, :
3.1.2. Density-Dependent Drucker–Prager Cap Model Implemented in COMSOL Multiphyisics®
- -
- The Drucker–Prager yield function, Fs:
- -
- The elliptical cap surface, Fc:
3.1.3. Nonlinear Elastic Law
3.2. Moisture and Heat Transport Simulation Model
3.3. Method of Calculation of Elastic Parameters and Drucker–Prager Cap Model Parameters
3.4. Finite Element Model
4. Results and Discussion
4.1. NIR Penetration Depth
4.2. Tablet Compression Curve
4.3. Mechanical, Heat, and Moisture Simulation during Compaction
4.3.1. Relative Density Evolution and Distribution
4.3.2. Temperature Evolution and Distribution
- Simulated temperature of tablets
- Measured surface and peripheral temperatures of the tablets
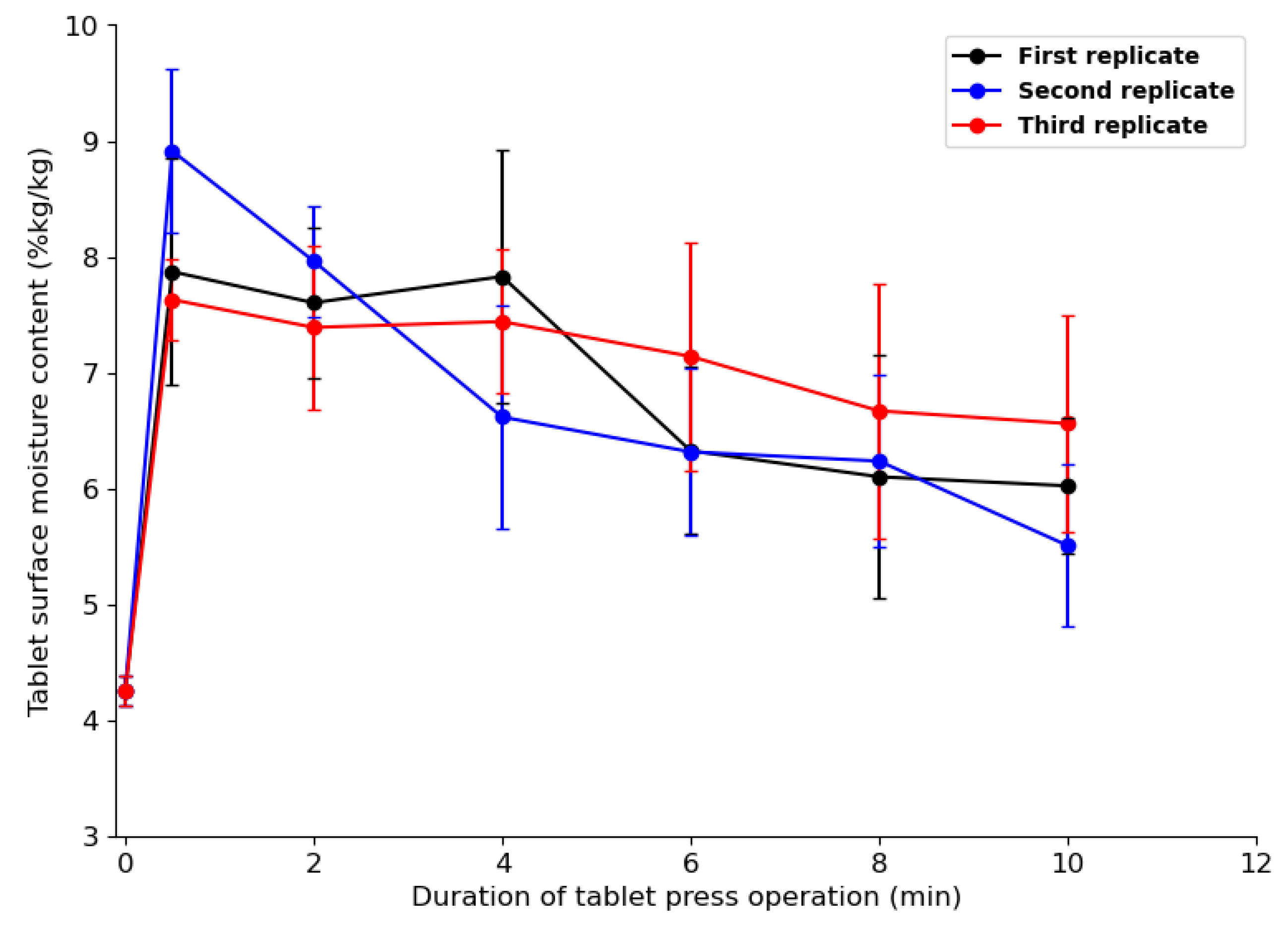
4.3.3. Tablet Moisture Content Evolution and Distribution during Compaction
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chattoraj, S.; Daugherity, P.; McDermott, T.; Olsofsky, A.; Roth, J.W.; Tobyn, M. Sticking and picking in pharmaceutical tablet compression: An IQ Consortium review. J. Pharm. Sci. 2018, 107, 2267–2282. [Google Scholar] [CrossRef]
- Zhoa, J. Compaction of Pharmaceutical Powders on a High-Speed Rotary Press. Master’s Thesis, Faculty of Pharmaceutical Sciences, University of British Columbia, Vancouver, BC, Canada, 2003. Available online: https://open.library.ubc.ca/media/download/pdf/831/1.0090907/2 (accessed on 14 December 2022).
- Denny, P.J. Compaction quations: A compression of the Heckel and Kawakita equations. Powder Technol. 2002, 183, 162–172. [Google Scholar] [CrossRef]
- Leuenberger, H.; Rohera, B.D. Fundamentals of powder compression: 1. The compatibility and compressibility of pharmaceutical powders. Pharm. Res. 1986, 3, 12–22. [Google Scholar] [CrossRef]
- Drucker, D.C.; Prager, W. Soil mechanics and plastic analysis or limit design. Q. Appl. Math. 1952, 10, 157–165. [Google Scholar] [CrossRef] [Green Version]
- Baladi, G.Y.; Rohani, B. Soil plasticity. In Theoretical Foundation for Large-Scale Computations for Nonlinear Material Behavior. Mechanics of Elastic and Inelastic Solids 6; Nemat-Nasser, S., Asaro, R.J., Hegemier, G.A., Eds.; Springer: Dordrecht, The Netherlands, 1984; Volume 6, pp. 317–352. [Google Scholar] [CrossRef]
- Schwer, L.E.; Murray, Y.D. A three-invariant smooth cap model with mixed hardening. Int. J. Numer. Anal. Methods Geomech. 1994, 18, 657–688. [Google Scholar] [CrossRef]
- Foster, C.D.; Regueiro, R.A.; Fossum, A.F.; Borja, R.I. Implicit numerical integration of a three-invariant, isotropic/kinematic hardening cap plasticity model for geomaterials. Comput. Methods Appl. Mech. Eng. 2005, 194, 5109–5138. [Google Scholar] [CrossRef] [Green Version]
- DiMaggio, F.L.; Sandler, I.S. Material model for granular soils. J. Eng. Mech. Div. 1971, 97, 935–950. [Google Scholar] [CrossRef]
- Schofield, A.N.; Wroth, P. Critical State Soil Mechanics; McGraw-Hill: London, UK, 1968; Volume 310, Available online: http://www.civ.eng.cam.ac.uk/geotech_new/publications/schofield8wroth81968.pdf (accessed on 15 March 2020).
- Wu, C.Y.; Ruddy, O.M.; Bentham, A.C.; Hancock, B.C.; Best, S.M.; Elliott, J.A. Modelling the mechanical behaviour of pharmaceutical powders during compaction. Powder Technol. 2005, 152, 107–117. [Google Scholar] [CrossRef]
- Han, L.H.; Elliott, J.A.; Bentham, A.C.; Mills, A.; Amidon, G.E.; Hancock, B.C. A modified Drucker-Prager Cap model for die compaction simulation of pharmaceutical powders. Int. J. Solids Struct. 2008, 45, 3088–3106. [Google Scholar] [CrossRef] [Green Version]
- Si, G.N.; Lan, C. Modeling and simulation of pharmaceutical powder compaction. Appl. Mech. Mater. 2012, 217, 1403–1406. [Google Scholar] [CrossRef]
- Hayashi, Y.; Otoguro, S.; Miura, T.; Onuki, Y.; Obata, Y.; Takayama, K. Effect of process variables on the Drucker–Prager cap model and residual stress distribution of tablets estimated by the finite element method. Chem. Pharm. Bull. 2014, 62, 1062–1072. [Google Scholar] [CrossRef] [Green Version]
- Diarra, H.; Mazel, V.; Busignies, V.; Tchoreloff, P. Investigating the effect of tablet thickness and punch curvature on density distribution using finite elements method. Int. J. Pharm. 2015, 493, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Baroutaji, A.; Lenihan, S.; Bryan, K. Combination of finite element method and Drucker-Prager Cap material model for simulation of pharmaceutical tableting process. Materialwiss. Werkst. 2017, 48, 1133–1145. [Google Scholar] [CrossRef]
- Krok, A.; Peciar, M.; Fekete, R. Numerical investigation into the influence of the punch shape on the mechanical behavior of pharmaceutical powders during compaction. Particuology 2014, 16, 116–131. [Google Scholar] [CrossRef]
- Zavaliangos, A.; Galen, S.; Cunningham, J.; Winstead, D. Temperature evolution during compaction of pharmaceutical powders. J. Pharm. Sci. 2008, 97, 3291–3304. [Google Scholar] [CrossRef]
- Klinzing, G.R.; Zavaliangos, A.; Cunningham, J.; Mascaro, T.; Winstead, D. Temperature and density evolution during compaction of a capsule shaped tablet. Comput. Chem. Eng. 2010, 34, 1082–1091. [Google Scholar] [CrossRef]
- Krok, A.; García-Triñanes, P.; Peciar, M.; Wu, C.Y. Finite element analysis of thermomechanical behaviour of powders during tabletting. Chem. Eng. Res. Des. 2016, 110, 141–151. [Google Scholar] [CrossRef] [Green Version]
- DeCrosta, M.T.; Schwartz, J.B.; Wigent, R.J.; Marshall, K. Thermodynamic analysis of compact formation: Compaction, unloading, and ejection I. Design and development of a compaction calorimeter and mechanical and thermal energy determinations of powder compaction. Int. J. Pharm. 2000, 198, 113–134. [Google Scholar] [CrossRef]
- Ketolainen, J.; IIkka, J.J.; Paronen, P. Temperature changes during tabletting measured using infrared thermoviewer. Int. J. Pharm. 1993, 92, 157–166. [Google Scholar] [CrossRef]
- Krok, A.; Mirtic, A.; Reynolds, G.K.; Schiano, S.; Roberts, R.; Wu, C.-Y. An experimental investigation of temperature rise during compaction of pharmaceutical powders. Int. J. Pharm. 2016, 513, 97–108. [Google Scholar] [CrossRef]
- Wu, C.Y.; Hancock, B.C.; Mills, A.; Bentham, A.C.; Best, S.M.; Elliott, J.A. Numerical and experimental investigation of capping mechanisms during pharmaceutical tablet compaction. Powder Technol. 2008, 181, 121–129. [Google Scholar] [CrossRef]
- Garner, S.; Ruiz, E.; Strong, J.; Zavaliangos, A. Mechanisms of crack formation in die compacted powders during unloading and ejection: An experimental and modeling comparison between standard straight and tapered dies. Powder Technol. 2014, 264, 114–127. [Google Scholar] [CrossRef]
- Mazel, V.; Diarra, H.; Malvestio, J.; Tchoreloff, P. Lamination of biconvex tablets: Numerical and experimental study. Int. J. Pharm. 2018, 542, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Klinzing, G.R.; Troup, G.M. Modeling the air pressure increase within a powder bed during compression—A step toward understanding tablet defects. J. Pharm. Sci. 2019, 108, 1991–2001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waimer, F.; Krumme, M.; Danz, P.; Tenter, U.; Schmidt, P.C. The influence of engravings on the sticking of tablets. Investigations with an instrumented upper punch. Pharm. Dev. Technol. 1999, 4, 369–375. [Google Scholar] [CrossRef]
- Al-Karawi, C.; Leopold, C.S. A comparative study on the sticking tendency of ibuprofen and ibuprofen sodium dihydrate to differently coated tablet punches. Eur. J. Pharm. Biopharm. 2018, 128, 107–118. [Google Scholar] [CrossRef]
- Danjo, K.; Kamiya, K.; Otsuka, A. Effect of temperature on the sticking of low melting point materials. Chem. Pharm. Bull. 1993, 41, 1423–1427. [Google Scholar] [CrossRef] [Green Version]
- Sun, C.C. Materials science tetrahedron—A useful tool for pharmaceutical research and development. J. Pharm. Sci. 2009, 98, 1671–1687. [Google Scholar] [CrossRef]
- Wakins, V.; Chu, E.; Schlam, R.; Sidorenko, A.; Badawy, S.; Yin, S.; Narang, A.S. Molecular basis of crystal morphology dependent adhesion behavior of mefenamic acid during tableting. Pharm. Res. 2014, 31, 160–172. [Google Scholar] [CrossRef]
- Danjo, K.; Kojima, S.; Chen, C.; Sunada, H.; Otsuka, A. Effect of water content on sticking during compression. Chem. Pharm. Bull. 1997, 45, 706–709. [Google Scholar] [CrossRef] [Green Version]
- Roberts, M.; Ford, J.L.; MacLeod, G.S.; Fell, J.T.; Smith, G.W.; Rowe, P.H. Effects of surface roughness and chrome plating of punch tips on the sticking tendencies of model ibuprofen formulations. J. Pharm. Pharmacol. 2003, 55, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M.; Ford, J.L.; MacLeod, G.S.; Fell, J.T.; Smith, G.W.; Rowe, P.H.; Dyas, M.A. Effect of lubricant type and concentration on the punch tip adherence of model ibuprofen formulations. J. Pharm. Pharmacol. 2004, 56, 299–305. [Google Scholar] [CrossRef]
- Roberts, M.; Ford, J.L.; MacLeod, G.S.; Fell, J.T.; Smith, G.W.; Rowe, P.H.; Dyas, M.A. Effect of punch tip geometry and embossment on the punch tip adherence of a model ibuprofen formulation. J. Pharm. Pharmacol. 2004, 56, 947–950. [Google Scholar] [CrossRef]
- Tejedor, M.B.; Nordgren, N.; Schuleit, M.; Millqvist-Fureby, A.; Rutland, M.W. AFM colloidal probe measurements implicate capillary condensation in punch−particle surface interactions during tableting. Langmuir 2017, 33, 13180–13188. [Google Scholar] [CrossRef]
- Pingali, K.C.; Shinbrot, T.; Cuitino, A.; Muzzio, F.J.; Garfunkel, E.; Lifshitz, Y.; Mann, A.B. AFM study of hydrophilicity on acetaminophen crystals. Int. J. Pharm. 2012, 438, 184–190. [Google Scholar] [CrossRef]
- Wang, J.J.; Guillot, M.A.; Bateman, S.D.; Morris, K.R. Modeling of adhesion in tablet compression. II. Compaction studies using a compaction simulator and an instrumented tablet press. J. Pharm. Sci. 2004, 93, 407–417. [Google Scholar] [CrossRef] [PubMed]
- McDermott, T.S.; Farrenkopf, J.; Hlinak, A.; Neilly, J.P.; Sauer, D. A material sparing method for quantitatively measuring tablet sticking. Powder Technol. 2011, 212, 240–252. [Google Scholar] [CrossRef]
- Costa, N.F.; Paulo, M.G.; Diogo, H.P.; Pinto, J.F. Solving a sticking related tablet problem by multivariate statistics and computational tomographic analysis. Powder Technol. 2020, 367, 456–463. [Google Scholar] [CrossRef]
- Freiberg, D.; Zavaliangos, A. Adhesion of powder onto tools during compaction: A numerical study. Powder Technol. 2022, 407, 117644. [Google Scholar] [CrossRef]
- Dembele, M.; Hudon, S.; Simard, J.-S.; Abatzoglou, N.; Gosselin, R. Multivariate data analysis approach to tablet sticking on an industrial scale: A qualitative case study of an ibuprofen-based formulation. Pharm. Dev. Technol. 2022, 27, 1093–1109. [Google Scholar] [CrossRef]
- Boudoin, L.N.; Lambeth, G.H.; Llimatta, E.W.; Hu, P.C. High Content Sodium Ibuprofen Granules, Their Preparation and Their Use in Preparing Non-Effervescent Solid Dosage Forms. U.S. Patent US9629809 B2, 25 April 2017. [Google Scholar]
- Siesler, H.W.; Ozaki, Y.; Kawata, S.; Heise, H.M. (Eds.) Near-Infrared Spectroscopy. Principles, Instruments, Applications; Wiley-VCH: Weinheim, Germany, 2002; 348p, ISBN 3-527-30149-6. [Google Scholar]
- Watanabe, A.; Morita, S.; Ozaki, Y. A study on water adsorption onto microcrystalline cellulose by near-infrared spectroscopy with two-dimensional correlation spectroscopy and principal component analysis. Appl. Spectrosc. 2006, 60, 1054–1061. [Google Scholar] [CrossRef]
- Luck, W.A.P. Structure of Water and Aqueous Solutions. In Proceedings of the international Symposium, Marburg, Germany, 18–28 July 1973; Luck, W.A.P., Ed.; Verlag Chemie: Weinheim, Germany, 1974; pp. 248–284. [Google Scholar]
- Curcio, J.A.; Petty, C.C. The near infrared absorption spectrum of liquid water. J. Opt. Soc. Am. 1951, 41, 302–304. [Google Scholar] [CrossRef]
- Büning-Pfaue, H. Analysis of water in food by near infrared spectroscopy. Food Chem. 2003, 82, 107–115. [Google Scholar] [CrossRef]
- Gupta, A.; Peck, G.E.; Miller, R.W.; Morris, K.R. Real-time near-infrared monitoring of content uniformity, moisture content, compact density, tensile strength, and Young’s modulus of roller compacted powder blends. J. Pharm. Sci. 2005, 94, 1589–1597. [Google Scholar] [CrossRef] [PubMed]
- Mainali, D.; Li, J.; Yehl, P.; Chetwyn, N. Development of a comprehensive near infrared spectroscopy calibration model for rapid measurements of moisture content in multiple pharmaceutical products. J. Pharm. Biomed. Anal. 2014, 95, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Otto, M. Chemometrics. Statistics and Computer Application in Analytical Chemistry; Wiley-VCH: New York, NY, USA, 1998; ISBN 978-3-527-34097-2. [Google Scholar]
- Naes, T.; Isaksson, T.; Fearn, T.; Davies, T. A User-Friendly Guide to Multivariate Calibration and Classification, 2nd ed.; IM Publications Open: Chichester, UK, 2017; Chapter 5. [Google Scholar] [CrossRef]
- Pasquini, C. Near infrared spectroscopy: Fundamentals, practical aspects and analytical applications. J. Braz. Chem. Soc. 2003, 14, 198–219. [Google Scholar] [CrossRef] [Green Version]
- Guy, A. Cellulose Microcrystalline. Available online: https://file.wuxuwang.com/hpe/HPE6/HPE6_59.pdf (accessed on 10 April 2023).
- NETZSCH Proven Excellence. Laser Flash Technique. Available online: https://www.netzsch-thermal-analysis.com/en/contract-testing/methods/laser-flash-technique-lfa-1 (accessed on 3 January 2019).
- COMSOL Documentation. Available online: https://doc.comsol.com/5.6/docserver/#!/com.comsol.help.comsol/helpdesk/helpdesk.html (accessed on 10 March 2021).
- Geladi, P.; Macdougall, D.; Martens, H. Linearization and Scatter Correction for Near Infrared Reflectance Spectra of Meat. Appl. Spectrosc. 1985, 39, 491–500. [Google Scholar] [CrossRef]
- Roger, J.-M.; Mallet, A.; Marini, F. Preprocessing NIR spectra for Aquaphotomics. Molecules 2022, 27, 6795. [Google Scholar] [CrossRef]
- Robert, G.; Gosselin, R. Evaluating the impact of NIR pre-processing methods via multiblock partial least-squares. Anal. Chim. Acta 2022, 1189, 339255. [Google Scholar] [CrossRef]
- Jackson, J.E. A User’s Guide to Principal Components, 1st ed.; John Wiley and Sons: New York, NY, USA, 1991. [Google Scholar] [CrossRef]
- Geladi, P.; Kowalski, B.R. Partial least-squares regression: A tutorial. Anal. Chim. Acta 1986, 185, 1–17. [Google Scholar] [CrossRef]
- Pharmaceutical Tableting Process. Available online: https://www.comsol.com/model/download/943861/models.nsm.pharmaceutical_tableting_process.pdf (accessed on 10 April 2023).
- Michrafy, A.; Ringenbacher, D.; Techoreloff, P. Modelling the compaction behaviour of powders: Application to pharmaceutical powders. Powder Technol. 2002, 127, 257–266. [Google Scholar] [CrossRef] [Green Version]
- Dos Santos, G.H.; Mendes, N. Combined heat, air and moisture (HAM) transfer model for porous building materials. J. Build. Phys. 2009, 32, 203–220. [Google Scholar] [CrossRef]
- Tariku, F.; Kumaran, M.K.; Fazio, P. Transient model for coupled heat, air and moisture transfer through multilayered porous media. Int. J. Heat Mass Transf. 2010, 53, 3035–3044. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Christakis, N.; Patel, M.K.; Cross, M.; Leaper, M.C.A. A computational model of coupled heat and moisture transfer with phase change in granular sugar during varying environmental conditions. Numer. Heat Transf. A-Appl. 2004, 45, 751–776. [Google Scholar] [CrossRef]
- Chao, B.T.; Trigger, K.J.; Zystra, L.B. Thermophysical aspects of metal cutting. Trans. ASME 1952, 74, 1039–1049. [Google Scholar] [CrossRef]
- Grzesik, W. Advanced Machining Processes of Metallic Materials: Theory, Modelling, and Applications, 2nd ed.; Elsevier Science: Amsterdam, The Netherlands, 2008. [Google Scholar] [CrossRef]
- Bever, M.B.; Holt, D.L.; Titchener, A.L. The stored energy of cold work. In Progress in Materials Science; Chalmers, B., Christian, J.W., Massalski, T.B., Eds.; Elsevier: Amsterdam, The Netherlands, 1973; Volume 17, pp. 5–88. [Google Scholar] [CrossRef]
- Von Böckh, P.; Wetzel, T. Heat Transfer: Basics and Practice; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar] [CrossRef]
- Koumbogle, K.; Gitzhofer, F.; Abatzoglou, N. Moisture transport coefficients determination on a model pharmaceutical tablet. Processes 2022, 10, 254. [Google Scholar] [CrossRef]
- Ostuka, M.; Koyama, A.; Hattori, Y. Real-time release monitoring for water content and mean particle size of granules in lab-sized fluid-bed granulator by near-infrared spectroscopy. RSC Adv. 2014, 4, 17461–17468. [Google Scholar] [CrossRef]
- Bogreckci, I.; Lee, W.S. The Effects of Soil Moisture Content on Reflectance Spectra of Soils Using UVVIS-NIR Spectroscopy. In Proceedings of the 7th International Conference on Precision Agriculture, Minneapolis, MN, USA, 25–28 July 2004. [Google Scholar]
- Lambert, J.H. Photometria Sive de Mensura et Gradibus Luminis, Colorum et Umbrae. 1760. Available online: https://www.amazon.com/Photometrie-Photometria-Mensura-Gradibus-Luminis/dp/1279983108 (accessed on 10 May 2023).
- Gou, D.; Li, Y.; An, X.; Yang, R. DEM modelling of particle fragmentation during compaction of particles. Powder Technol. 2022, 398, 117073. [Google Scholar] [CrossRef]
- Buckner, I.S.; Friedman, R.A.; Wurster, D.E. Using compression calorimetry to characterize powder compaction behavior of pharmaceutical materials. J. Pharm. Sci. 2010, 99, 861–870. [Google Scholar] [CrossRef]
- Kadiri, M.S.; Michrafy, A. The effect of punch’s shape on die compaction of pharmaceutical powders. Powder Technol. 2013, 239, 467–477. [Google Scholar] [CrossRef] [Green Version]
- Laity, P.R. Effects of punches with embossed features on compaction behaviour. Powder Technol. 2014, 254, 373–386. [Google Scholar] [CrossRef]
- Bechard, S.R.; Down, G.R.B. Infrared imaging of pharmaceutical materials undergoing compaction. Pharm. Res. 1992, 9, 521–528. [Google Scholar] [CrossRef]
- Nezamabadi, S.; Ghadiri, M.; Delenne, J.-Y.; Radjai, F. Modelling the compaction of plastic particle packings. Comput. Part. Mech. 2021, 9, 45–52. [Google Scholar] [CrossRef]
- Brunauer, S. The Adsorption of Gases and Vapors; Oxford University Press: Oxford, UK, 1943; Volume 1, p. 49, ISBN 10: 1443727369. [Google Scholar]
- Christodoulou, C.; Sorensen, E.; García-Muñoz, S.; Mazzei, L. Mathematical modelling of water absorption and evaporation in a pharmaceutical tablet during film coating. Chem Eng Sci. 2018, 175, 40–55. [Google Scholar] [CrossRef]
- Schiros, T. Water-Metal Surfaces: Insights from Core-Level Spectroscopy and Density Functional Theory; Department of Physics, Stockholm University: Stockholm, Sweden, 2006; p. 72. ISBN 978-91-7155-607-3. [Google Scholar]
- Fernandes, F.W.; Campos, T.M.B.; Cividanes, L.S.; Simonetti, E.A.N.; Thim, G.P. Adsorbed water on iron surface by molecular dynamics. Appl. Surf. Sci. 2016, 362, 70–78. [Google Scholar] [CrossRef]
- Meng, S.; Wang, E.G.; Gao, S. Water adsorption on metal surfaces: A general picture from density functional theory studies. Phys. Rev. B 2004, 69, 195404. [Google Scholar] [CrossRef] [Green Version]
- Charlaix, E.; Ciccotti, M. Capillary Condensation in Confined Media. In Handbook of Nanophysics I: Principles and Methods, 1st ed.; Sattler, K.D., Ed.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2010; Volume 1, pp. 194–210. [Google Scholar]
- Tanaka, H.; Hiratsuka, T.; Nishiyama, N.; Mori, K.; Miyahara, M.T. Capillary condensation in mesoporous silica with surface roughness. Adsorption 2013, 19, 631–641. [Google Scholar] [CrossRef]
- Philip, J.R. Adsorption and capillary condensation on rough surfaces. J. Phys. Chem. 1978, 82, 1379–1385. [Google Scholar] [CrossRef]
- Naidich, Y.V.; Lavrinenko, I.A. Capillary adhesion forces between solid particles with an intermediate liquid layer at the contact. Powder Metall. Met. Ceram. 1967, 6, 709–710. [Google Scholar] [CrossRef]
- De Lazzer, A.; Dreyer, M.; Rath, H.J. Particle-surface capillary forces. Langmuir 1999, 15, 4551–4559. [Google Scholar] [CrossRef]
- Paul, S.; Taylor, L.J.; Murphy, B.; Krzyzaniak, J.F.; Dawson, N.; Mullarney, M.P.; Meenam, P.; Sun, C.C. Mechanism and kinetics of punch sticking of pharmaceuticals. J. Pharm. Sci. 2017, 106, 151–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aoki, S.; Danjo, K. Effect of tableting conditions on the sticking of tablet using ibuprofen. Yakugaku Zasshi 1998, 118, 511–518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kakimi, K.; Niwa, T.; Danjo, K. Influence of compression pressure and velocity on tablet sticking. Chem. Pharm. Bull. 2010, 58, 1565–1568. [Google Scholar] [CrossRef] [PubMed] [Green Version]





















Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koumbogle, K.; Gosselin, R.; Gitzhofer, F.; Abatzoglou, N. Moisture Behavior of Pharmaceutical Powder during the Tableting Process. Pharmaceutics 2023, 15, 1652. https://doi.org/10.3390/pharmaceutics15061652
Koumbogle K, Gosselin R, Gitzhofer F, Abatzoglou N. Moisture Behavior of Pharmaceutical Powder during the Tableting Process. Pharmaceutics. 2023; 15(6):1652. https://doi.org/10.3390/pharmaceutics15061652
Chicago/Turabian StyleKoumbogle, Komlan, Ryan Gosselin, François Gitzhofer, and Nicolas Abatzoglou. 2023. "Moisture Behavior of Pharmaceutical Powder during the Tableting Process" Pharmaceutics 15, no. 6: 1652. https://doi.org/10.3390/pharmaceutics15061652
APA StyleKoumbogle, K., Gosselin, R., Gitzhofer, F., & Abatzoglou, N. (2023). Moisture Behavior of Pharmaceutical Powder during the Tableting Process. Pharmaceutics, 15(6), 1652. https://doi.org/10.3390/pharmaceutics15061652






