Solitary and Synergistic Effects of Different Hydrophilic and Hydrophobic Phospholipid Moieties on Rat Behaviors
Abstract
:1. Introduction
2. Materials and Methods
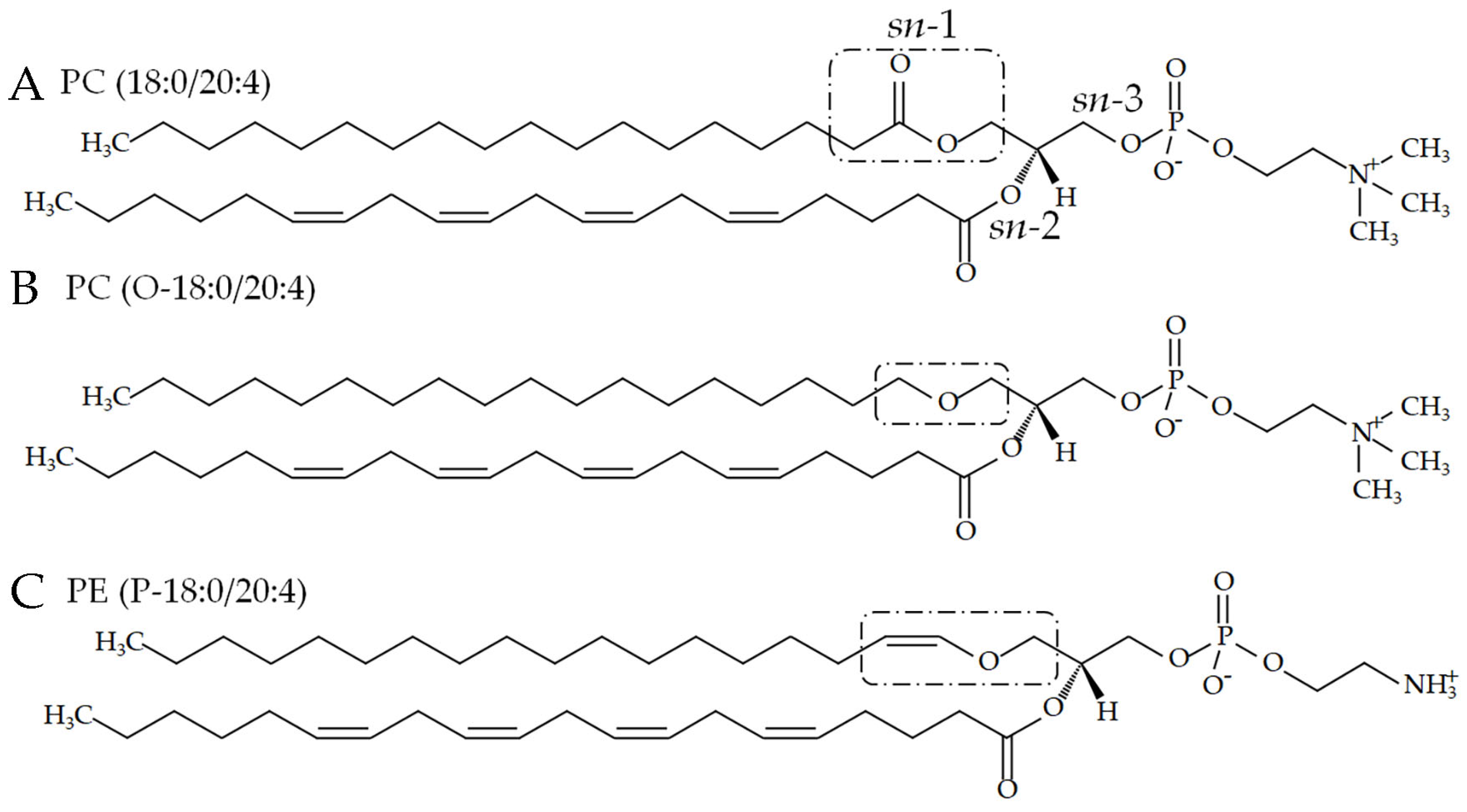
2.1. Phospholipids with an Ester or Vinyl–Ether Bond at sn-1
2.2. Phospholipid Synthesis
2.2.1. PC(18:0/20:0)
2.2.2. Plasmanylcholine (PC with an Alkyl–Ether Bond at sn-1)
2.2.3. Plasmanylethanolamine (PE with an Alkyl–Ether Bond at sn-1)
2.3. Phospholipid Analysis
2.3.1. Nuclear Magnetic Resonance Spectroscopy
2.3.2. Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry
2.3.3. Liquid Chromatography Mass Spectrometry (LC-MS)
2.4. Animals
2.5. Preparation of Liposomes
2.6. Brain Imaging
2.7. Blood–Brain Barrier Permeability
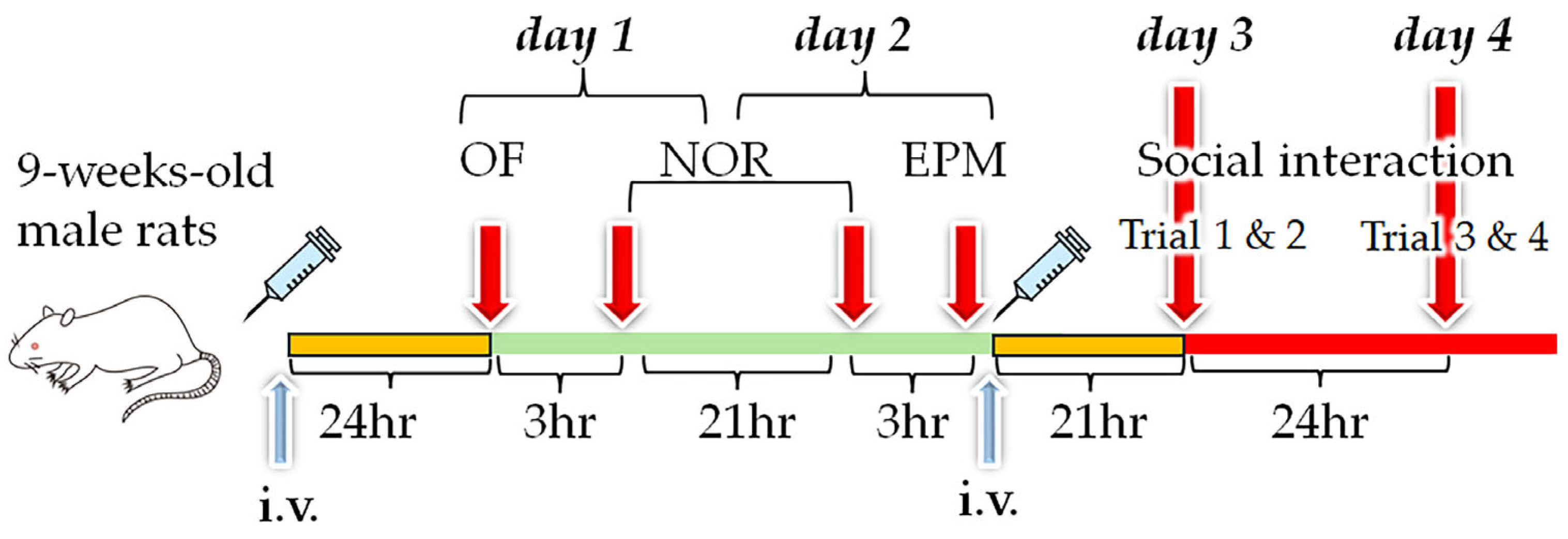
2.8. Liposome Injections for Behavioral Tests
2.9. Behavioral Tests
2.9.1. Open-Field Test
2.9.2. Novel Object Recognition Test
2.9.3. Elevated plus Maze Test
2.9.4. Social Interaction
2.10. Statistics
3. Results
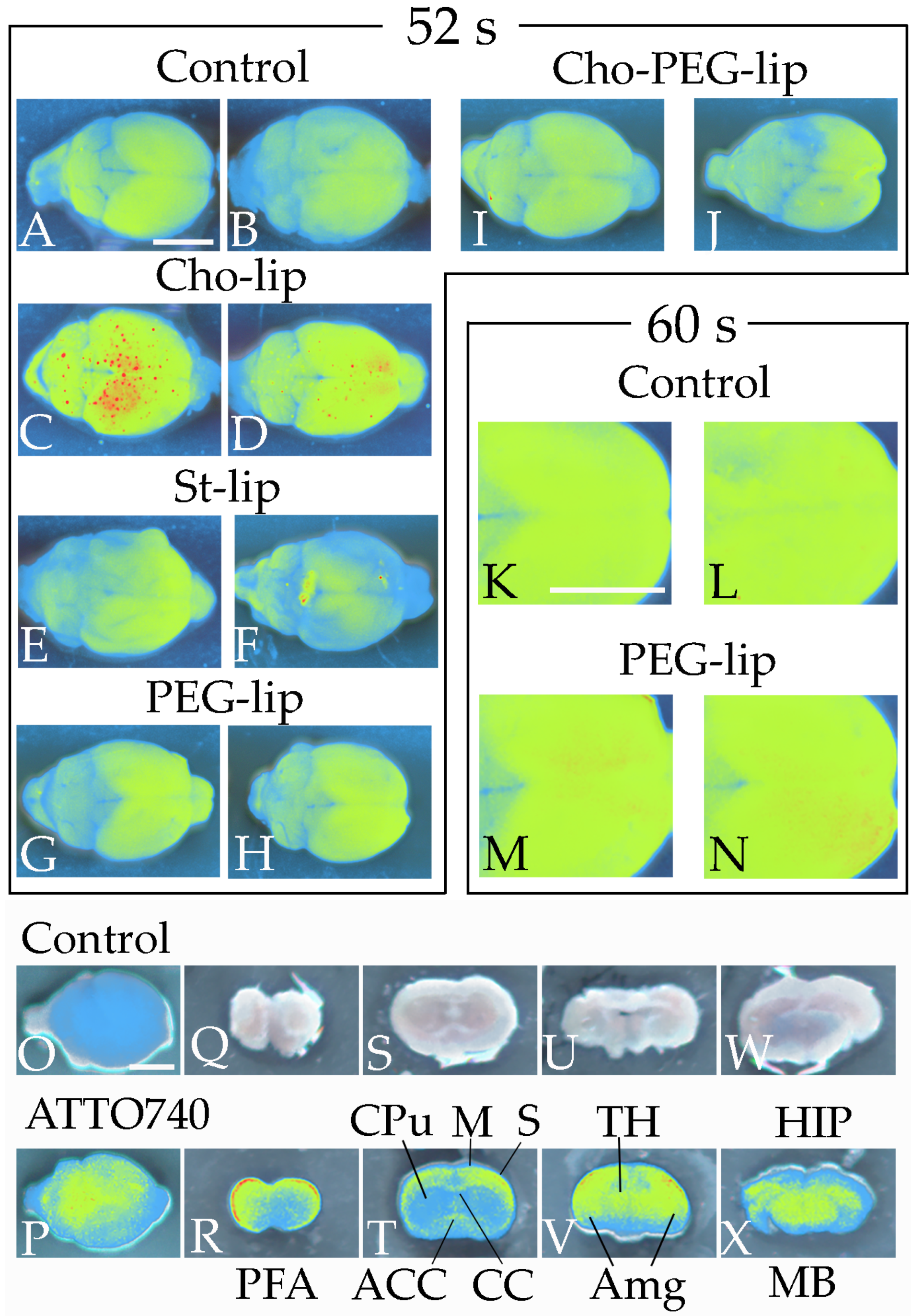
3.1. Phospholipid Localization in the Brain after Liposome Injection
3.2. Blood–Brain Barrier Permeability and Liposome Uptake into the Brain
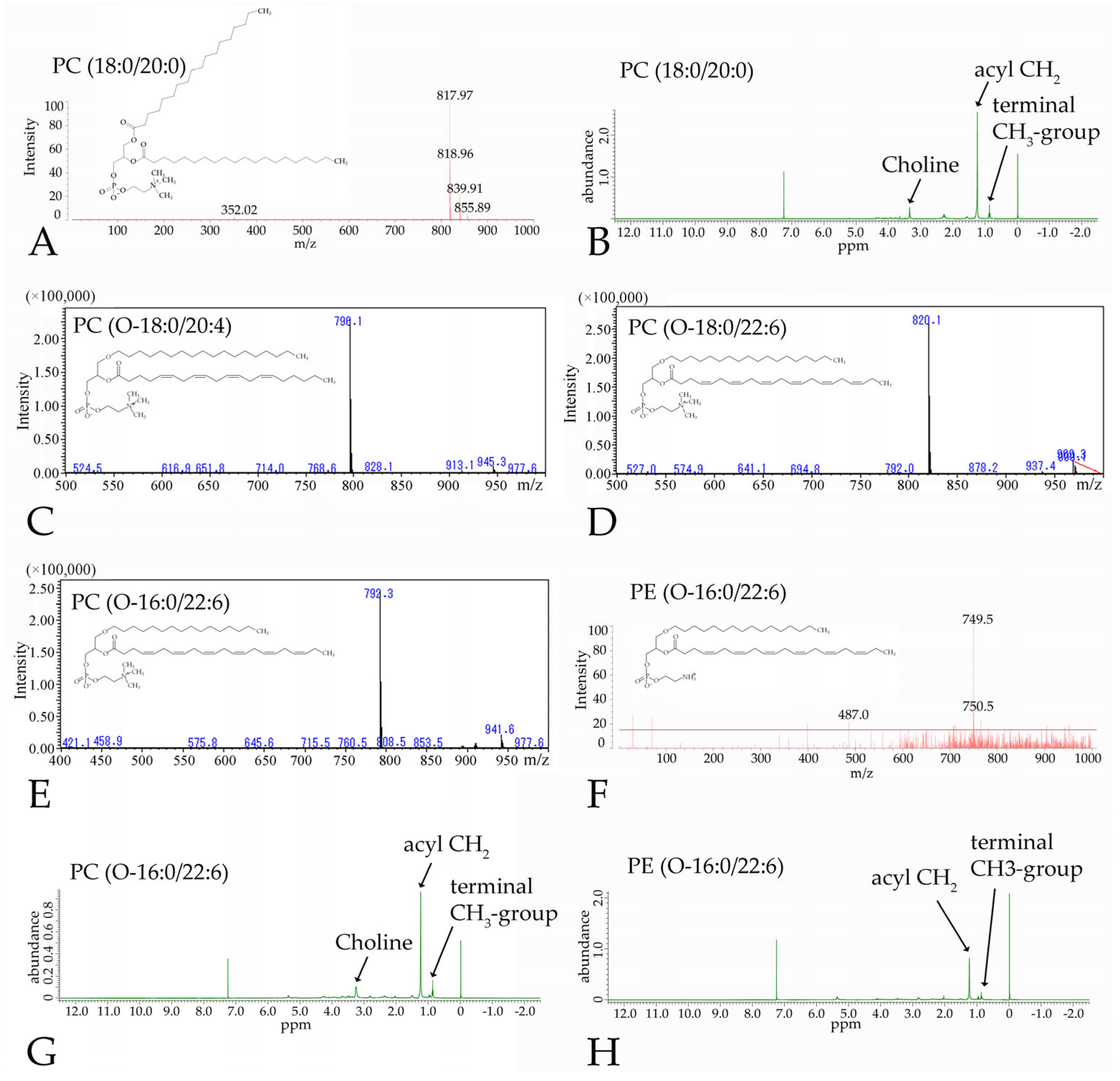
3.3. Nuclear Magnetic Resonance and Mass Spectometry of the Synthesized Phospholipids
3.4. Behavioral Changes Induced by Phospholipid Injection
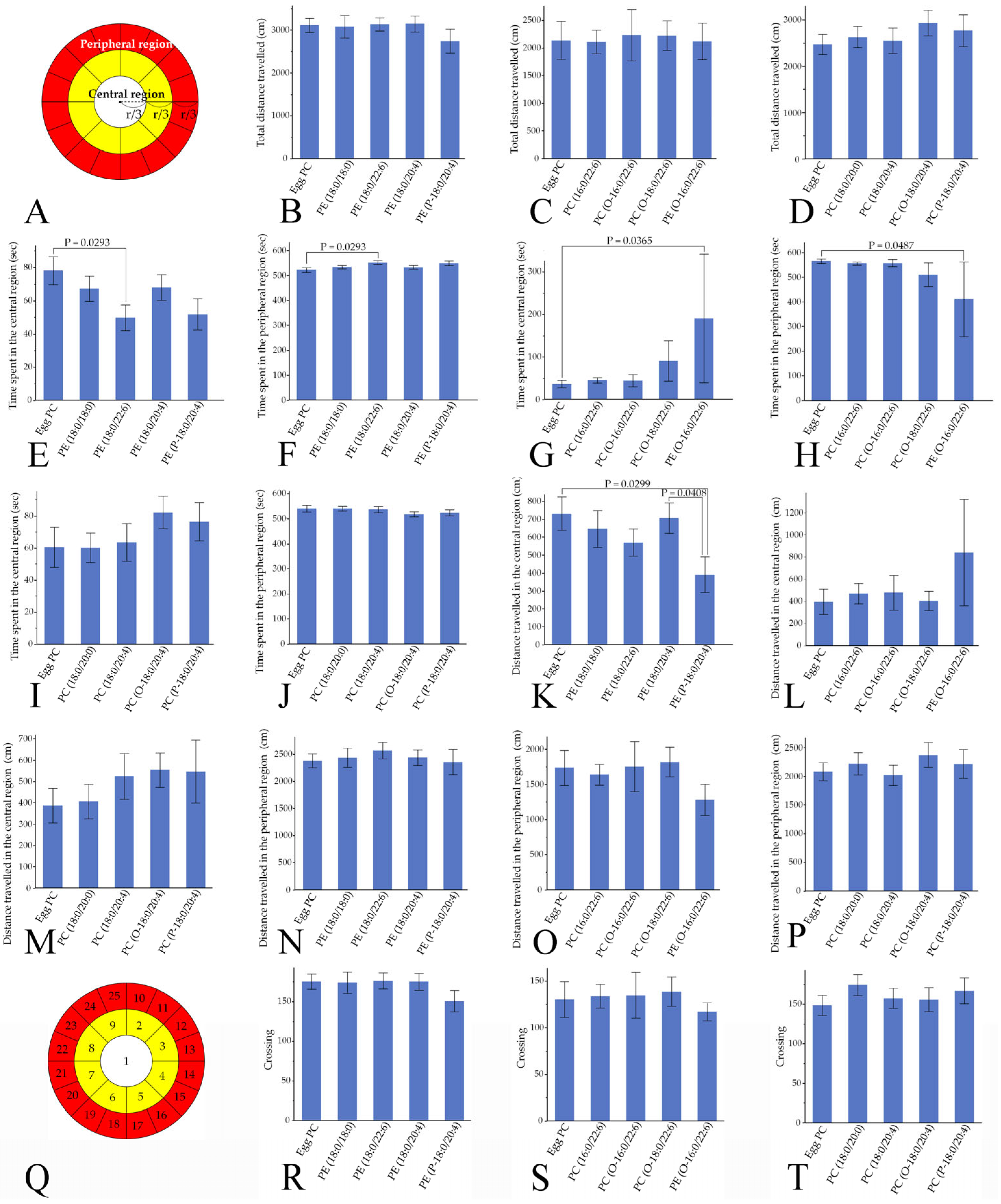
3.4.1. Locomotor Activities
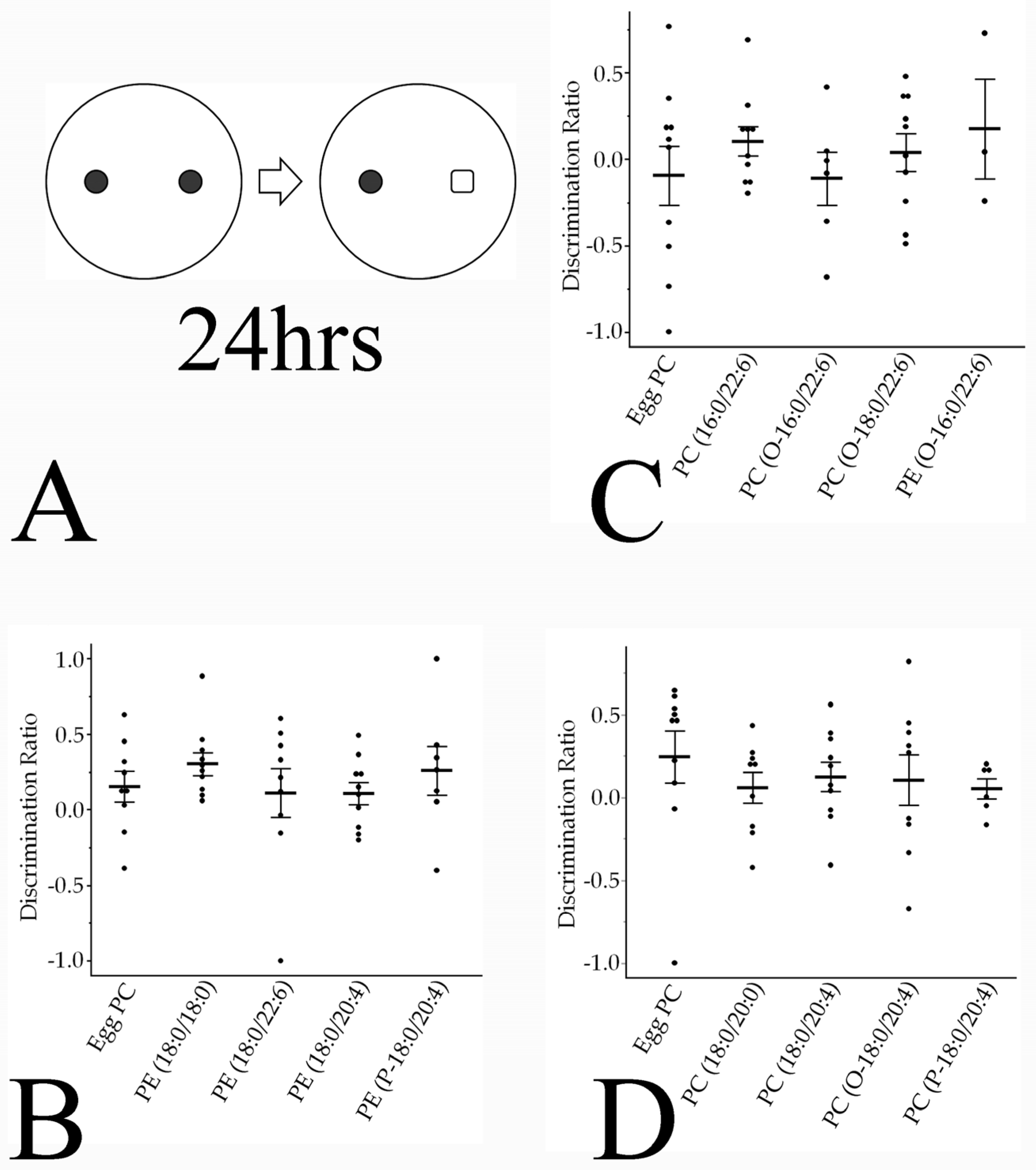
3.4.2. Novel Object Recognition Memory
3.4.3. Anxiety-like Behaviors
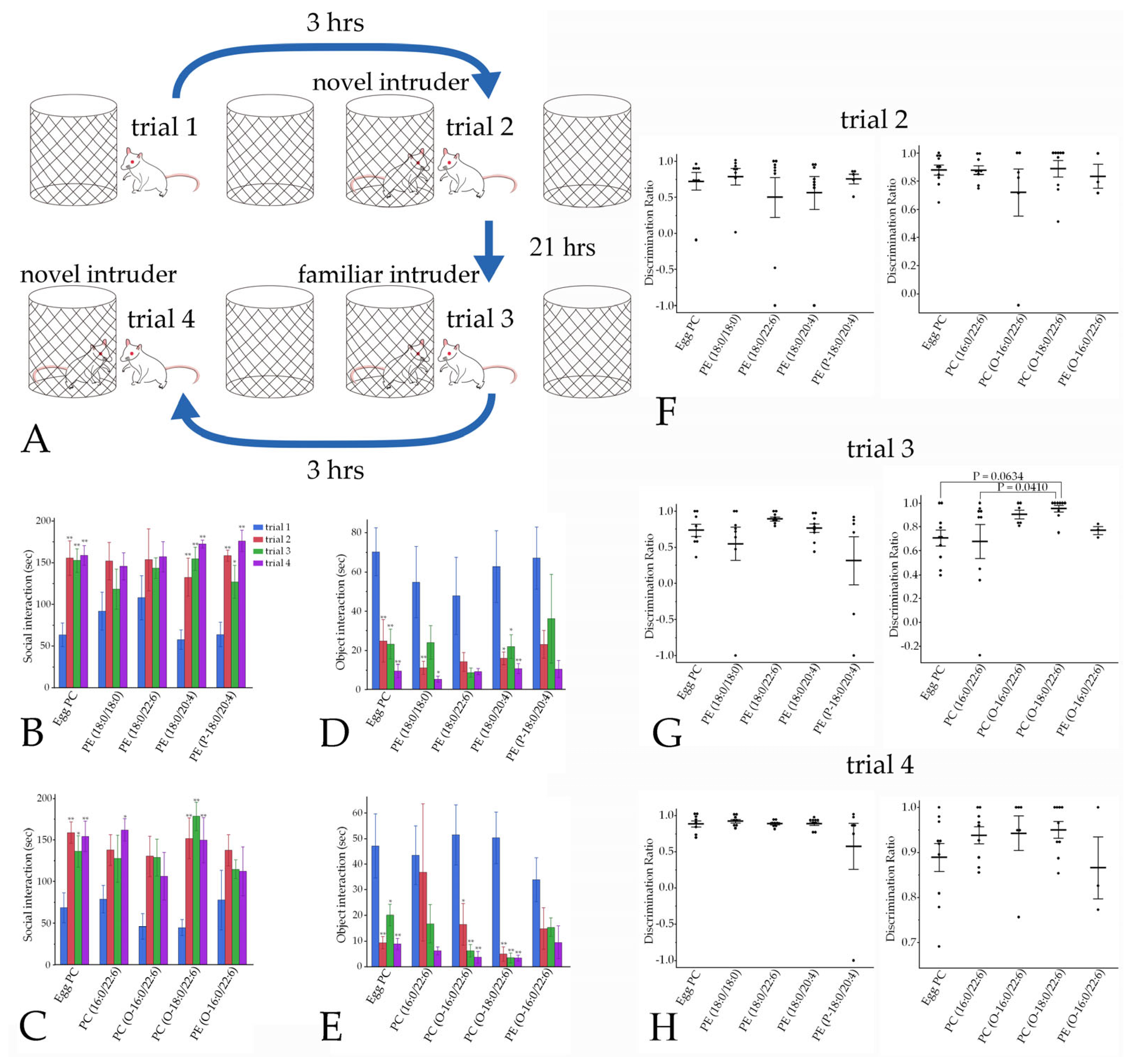
3.4.4. Social Interaction
4. Discussion
4.1. Phospholipid Moieties Alter Locomotor Activities
4.2. Phospholipid Moieties Reduce Anxiety-like Behaviors
4.3. Involvement of Phospholipid Moieties in Social Interaction and Recognition Memory
5. Conclusions, Limitations, and Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Yeagle, P.L. Chapter 2—The Lipids of Biological Membranes. In The Membranes of Cells, 3rd ed.; Yeagle, P.L., Ed.; Academic Press: Boston, MA, USA, 2016; pp. 27–56. [Google Scholar]
- Braverman, N.E.; Moser, A.B. Functions of plasmalogen lipids in health and disease. Biochim. Biophys. Acta 2012, 1822, 1442–1452. [Google Scholar] [CrossRef] [PubMed]
- Horrocks, L.A.; Sharma, M. Chapter 2 Plasmalogens and O-alkyl glycerophospholipids. In New Comprehensive Biochemistry; Hawthorne, J.N., Ansell, G.B., Eds.; Elsevier: Amsterdam, The Netherlands, 1982; Volume 4, pp. 51–93. [Google Scholar]
- Koivuniemi, A. The biophysical properties of plasmalogens originating from their unique molecular architecture. FEBS Lett. 2017, 591, 2700–2713. [Google Scholar] [CrossRef] [PubMed]
- Udagawa, J.; Hino, K. Plasmalogen in the brain: Effects on cognitive functions and behaviors attributable to its properties. Brain Res. Bull. 2022, 188, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Ladd, S.L.; Sommer, S.A.; LaBerge, S.; Toscano, W. Effect of phosphatidylcholine on explicit memory. Clin. Neuropharmacol. 1993, 16, 540–549. [Google Scholar] [CrossRef]
- Chin, E.W.M.; Lim, W.M.; Ma, D.; Rosales, F.J.; Goh, E.L.K. Choline Rescues Behavioural Deficits in a Mouse Model of Rett Syndrome by Modulating Neuronal Plasticity. Mol. Neurobiol. 2019, 56, 3882–3896. [Google Scholar] [CrossRef] [PubMed]
- Brydges, C.R.; Bhattacharyya, S.; Dehkordi, S.M.; Milaneschi, Y.; Penninx, B.; Jansen, R.; Kristal, B.S.; Han, X.; Arnold, M.; Kastenmüller, G.; et al. Metabolomic and inflammatory signatures of symptom dimensions in major depression. Brain Behav. Immun. 2022, 102, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Mangold, H.K.; Weber, N. Biosynthesis and biotransformation of ether lipids. Lipids 1987, 22, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.S.; Mawatari, S.; Fujino, T. Plasmalogens, the Vinyl Ether-Linked Glycerophospholipids, Enhance Learning and Memory by Regulating Brain-Derived Neurotrophic Factor. Front. Cell Dev. Biol. 2022, 10, 828282. [Google Scholar] [CrossRef]
- Azad, A.K.; Sheikh, A.M.; Haque, M.A.; Osago, H.; Sakai, H.; Shibly, A.Z.; Yano, S.; Michikawa, M.; Hossain, S.; Tabassum, S.; et al. Time-Dependent Analysis of Plasmalogens in the Hippocampus of an Alzheimer’s Disease Mouse Model: A Role of Ethanolamine Plasmalogen. Brain Sci. 2021, 11, 1613. [Google Scholar] [CrossRef]
- Fallatah, W.; Smith, T.; Cui, W.; Jayasinghe, D.; Di Pietro, E.; Ritchie, S.A.; Braverman, N. Oral administration of a synthetic vinyl-ether plasmalogen normalizes open field activity in a mouse model of rhizomelic chondrodysplasia punctata. Dis. Model. Mech. 2020, 13, dmm042499. [Google Scholar] [CrossRef]
- Andriambelo, B.; Stiffel, M.; Roke, K.; Plourde, M. New perspectives on randomized controlled trials with omega-3 fatty acid supplements and cognition: A scoping review. Ageing Res. Rev. 2023, 85, 101835. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, Y.; Sakurai, Y.; Damnjanović, J. A simple chemo-enzymatic synthesis of alkyl-acyl (plasmanyl) phospholipids. Biocatal. Agric. Biotechnol. 2020, 26, 101625. [Google Scholar] [CrossRef]
- Damnjanović, J.; Nakano, H.; Iwasaki, Y. Simple and Efficient Profiling of Phospholipids in Phospholipase D-modified Soy Lecithin by HPLC with Charged Aerosol Detection. J. Am. Oil Chem. Soc. 2013, 90, 951–957. [Google Scholar] [CrossRef]
- Morita, S.Y.; Deharu, Y.; Takata, E.; Nakano, M.; Handa, T. Cytotoxicity of lipid-free apolipoprotein B. Biochim. Biophys. Acta 2008, 1778, 2594–2603. [Google Scholar] [CrossRef] [PubMed]
- Hino, K.; Kaneko, S.; Harasawa, T.; Kimura, T.; Takei, S.; Shinohara, M.; Yamazaki, F.; Morita, S.Y.; Sato, S.; Kubo, Y.; et al. Change in Brain Plasmalogen Composition by Exposure to Prenatal Undernutrition Leads to Behavioral Impairment of Rats. J. Neurosci. 2019, 39, 7689–7702. [Google Scholar] [CrossRef] [PubMed]
- Zenika, N.M.; Kikuchi, S.; Uchimura, Y.; Yoshioka, M.; Morita, S.-y.; Kobayashi, T.; Kinoshita, Y.; Furusho, Y.; Tamiaki, H.; Yanagisawa, D.; et al. Ethanolamine and vinyl-ether moieties in brain phospholipids modulate behavior in rats. Int. J. Mol. Sci. 2024. submitted. [Google Scholar]
- Krüttner, S.; Falasconi, A.; Valbuena, S.; Galimberti, I.; Bouwmeester, T.; Arber, S.; Caroni, P. Absence of familiarity triggers hallmarks of autism in mouse model through aberrant tail-of-striatum and prelimbic cortex signaling. Neuron 2022, 110, 1468–1482.e5. [Google Scholar] [CrossRef] [PubMed]
- Hamezah, H.S.; Durani, L.W.; Ibrahim, N.F.; Yanagisawa, D.; Kato, T.; Shiino, A.; Tanaka, S.; Damanhuri, H.A.; Ngah, W.Z.W.; Tooyama, I. Volumetric changes in the aging rat brain and its impact on cognitive and locomotor functions. Exp. Gerontol. 2017, 99, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Sivakumaran, M.H.; Mackenzie, A.K.; Callan, I.R.; Ainge, J.A.; O’Connor, A.R. The Discrimination Ratio derived from Novel Object Recognition tasks as a Measure of Recognition Memory Sensitivity, not Bias. Sci. Rep. 2018, 8, 11579. [Google Scholar] [CrossRef]
- Ono, T.; Hino, K.; Kimura, T.; Uchimura, Y.; Ashihara, T.; Higa, T.; Kojima, H.; Murakami, T.; Udagawa, J. Excessive folic acid intake combined with undernutrition during gestation alters offspring behavior and brain monoamine profiles. Congenit. Anom. 2022, 62, 169–180. [Google Scholar] [CrossRef]
- Zhou, Z.; Sayer, B.G.; Hughes, D.W.; Stark, R.E.; Epand, R.M. Studies of Phospholipid Hydration by High-Resolution Magic-Angle Spinning Nuclear Magnetic Resonance. Biophys. J. 1999, 76, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Mika, A.; Kaczynski, Z.; Stepnowski, P.; Kaczor, M.; Proczko-Stepaniak, M.; Kaska, L.; Sledzinski, T. Potential Application of 1H NMR for Routine Serum Lipidome Analysis –Evaluation of Effects of Bariatric Surgery. Sci. Rep. 2017, 7, 15530. [Google Scholar] [CrossRef] [PubMed]
- Hahnefeld, L.; Vogel, A.; Gurke, R.; Geisslinger, G.; Schäfer, M.K.E.; Tegeder, I. Phosphatidylethanolamine Deficiency and Triglyceride Overload in Perilesional Cortex Contribute to Non-Goal-Directed Hyperactivity after Traumatic Brain Injury in Mice. Biomedicines 2022, 10, 914. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Friedmann, D.; Xiong, J.; Liu, C.D.; Ferguson, B.R.; Weerakkody, T.; DeLoach, K.E.; Ran, C.; Pun, A.; Sun, Y.; et al. Anatomically Defined and Functionally Distinct Dorsal Raphe Serotonin Sub-systems. Cell 2018, 175, 472–487.e20. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.J.; Kwon, K.J.; Shin, C.Y.; Lee, G.S.; Moon, J.H.; Lee, T.G.; Yoon, S. Investigation of Phospholipid Differences in Valproic Acid-Induced Autistic Mouse Model Brain Using Mass Spectrometry Imaging. Metabolites 2023, 13, 178. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Xiao, L.; Sun, Y.; He, M.; Gao, C.; Zhu, C.; Chang, H.; Ding, J.; Li, W.; Wang, Y.; et al. The GABAB receptor agonist STX209 reverses the autism-like behaviour in an animal model of autism induced by prenatal exposure to valproic acid. Mol. Med. Rep. 2022, 25, 154. [Google Scholar] [CrossRef] [PubMed]
- Schneider, T.; Turczak, J.; Przewłocki, R. Environmental enrichment reverses behavioral alterations in rats prenatally exposed to valproic acid: Issues for a therapeutic approach in autism. Neuropsychopharmacology 2006, 31, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Miao, G.; Deen, J.; Struzeski, J.B.; Chen, M.; Zhang, Y.; Cole, S.A.; Fretts, A.M.; Lee, E.T.; Howard, B.V.; Fiehn, O.; et al. Plasma lipidomic profile of depressive symptoms: A longitudinal study in a large sample of community-dwelling American Indians in the strong heart study. Mol. Psychiatry 2023, 28, 2480–2489. [Google Scholar] [CrossRef]
- Zhang, F.; Guo, L.; Shi, J.; Jiang, H.; Zhou, F.; Zhou, Y.; Lv, B.; Xu, M. Choline metabolism in regulating inflammatory bowel disease-linked anxiety disorders: A multi-omics exploration of the gut-brain axis. Neurobiol. Dis. 2023, 191, 106390. [Google Scholar] [CrossRef]
- Weele, C.M.V.; Siciliano, C.A.; Tye, K.M. Dopamine tunes prefrontal outputs to orchestrate aversive processing. Brain Res. 2019, 1713, 16–31. [Google Scholar] [CrossRef]
- Ohmura, Y.; Tsutsui-Kimura, I.; Sasamori, H.; Nebuka, M.; Nishitani, N.; Tanaka, K.F.; Yamanaka, A.; Yoshioka, M. Different roles of distinct serotonergic pathways in anxiety-like behavior, antidepressant-like, and anti-impulsive effects. Neuropharmacology 2019, 167, 107703. [Google Scholar] [CrossRef] [PubMed]
- Heydendael, W.; Sharma, K.; Iyer, V.; Luz, S.; Piel, D.; Beck, S.; Bhatnagar, S. Orexins/hypocretins act in the posterior paraventricular thalamic nucleus during repeated stress to regulate facilitation to novel stress. Endocrinology 2011, 152, 4738–4752. [Google Scholar] [CrossRef] [PubMed]
- Cho, D.; O’Berry, K.; Possa-Paranhos, I.C.; Butts, J.; Palanikumar, N.; Sweeney, P. Paraventricular thalamic MC3R circuits link energy homeostasis with anxiety-related behavior. J. Neurosci. 2023, 43, 6280–6296. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; He, L.; Huang, A.J.Y.; Boehringer, R.; Robert, V.; Wintzer, M.E.; Polygalov, D.; Weitemier, A.Z.; Tao, Y.; Gu, M.; et al. A hypothalamic novelty signal modulates hippocampal memory. Nature 2020, 586, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Gunaydin, L.A.; Grosenick, L.; Finkelstein, J.C.; Kauvar, I.V.; Fenno, L.E.; Adhikari, A.; Lammel, S.; Mirzabekov, J.J.; Airan, R.D.; Zalocusky, K.A.; et al. Natural neural projection dynamics underlying social behavior. Cell 2014, 157, 1535–1551. [Google Scholar] [CrossRef] [PubMed]
- Leithead, A.B.; Godino, A.; Barbier, M.; Harony-Nicolas, H. Social Interaction Elicits Activity in Glutamatergic Neurons in the Posterior Intralaminar Complex of the Thalamus. Biol. Psychiatry 2024, 95, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.N.; Hsu, T.T.; Lin, M.H.; Chuang, H.C.; Hu, H.T.; Sun, C.P.; Tao, M.H.; Lin, J.Y.; Hsueh, Y.P. Interhemispheric Connectivity Potentiates the Basolateral Amygdalae and Regulates Social Interaction and Memory. Cell Rep. 2019, 29, 34–48.e34. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.N.; Chuang, H.C.; Chou, W.H.; Chen, C.Y.; Wang, H.F.; Chou, S.J.; Hsueh, Y.P. Tbr1 haploinsufficiency impairs amygdalar axonal projections and results in cognitive abnormality. Nat. Neurosci. 2014, 17, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; He, Z.; Zubkov, D.; Huang, S.; Kurochkin, I.; Yang, X.; Halene, T.; Willmitzer, L.; Giavalisco, P.; Akbarian, S.; et al. Lipidome alterations in human prefrontal cortex during development, aging, and cognitive disorders. Mol. Psychiatry 2020, 25, 2952–2969. [Google Scholar] [CrossRef]
- Han, X.; Holtzman, D.M.; McKeel, D.W. Plasmalogen deficiency in early Alzheimer’s disease subjects and in animal models: Molecular characterization using electrospray ionization mass spectrometry. J. Neurochem. 2001, 77, 1168–1180. [Google Scholar] [CrossRef]



| Liposomes | Egg PC (mg/mL) | DSPE (mg/mL) | DiIC18 (μg/mL) | Cholesterol (mg/mL) | Methyl PEG2000-DSPE (mg/mL) |
|---|---|---|---|---|---|
| St-lip | 8 | 2 | 100 | 0 | 0 |
| Cho-lip | 8 | 2 | 100 | 1 | 0 |
| PEG-lip | 8 | 1.5 | 100 | 0 | 0.5 |
| Cho-PEG-lip | 8 | 1.5 | 100 | 1 | 0.5 |
| Control liposomes | 10 | 0 | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kikuchi, S.; Iwasaki, Y.; Yoshioka, M.; Hino, K.; Morita, S.-y.; Tada, R.; Uchimura, Y.; Kubo, Y.; Kobayashi, T.; Kinoshita, Y.; et al. Solitary and Synergistic Effects of Different Hydrophilic and Hydrophobic Phospholipid Moieties on Rat Behaviors. Pharmaceutics 2024, 16, 762. https://doi.org/10.3390/pharmaceutics16060762
Kikuchi S, Iwasaki Y, Yoshioka M, Hino K, Morita S-y, Tada R, Uchimura Y, Kubo Y, Kobayashi T, Kinoshita Y, et al. Solitary and Synergistic Effects of Different Hydrophilic and Hydrophobic Phospholipid Moieties on Rat Behaviors. Pharmaceutics. 2024; 16(6):762. https://doi.org/10.3390/pharmaceutics16060762
Chicago/Turabian StyleKikuchi, Shuhei, Yugo Iwasaki, Mina Yoshioka, Kodai Hino, Shin-ya Morita, Ryu Tada, Yasuhiro Uchimura, Yoshinori Kubo, Tomoya Kobayashi, Yusuke Kinoshita, and et al. 2024. "Solitary and Synergistic Effects of Different Hydrophilic and Hydrophobic Phospholipid Moieties on Rat Behaviors" Pharmaceutics 16, no. 6: 762. https://doi.org/10.3390/pharmaceutics16060762
APA StyleKikuchi, S., Iwasaki, Y., Yoshioka, M., Hino, K., Morita, S.-y., Tada, R., Uchimura, Y., Kubo, Y., Kobayashi, T., Kinoshita, Y., Hayashi, M., Furusho, Y., Tamiaki, H., Ishiyama, H., Kuroda, M., & Udagawa, J. (2024). Solitary and Synergistic Effects of Different Hydrophilic and Hydrophobic Phospholipid Moieties on Rat Behaviors. Pharmaceutics, 16(6), 762. https://doi.org/10.3390/pharmaceutics16060762







