1. Introduction
In recent years, there has been an increased focus on addressing the issue of poorly water-soluble API candidates in pharmaceutical development [
1,
2]. One strategy to overcome this solubility challenge is the nanocrystal approach, which involves reducing the size of the crystalline API to improve dissolution rate and saturation solubility, in accordance with the Noyes–Whitney and Ostwald–Freundlich principles [
1,
3,
4,
5].
There are several FDA-approved products that have utilized the nanocrystal approach, such as Rapamune
® (Pfizer, 2000), which was one of the earliest products. Other examples include Invega Sustenna
® (Janssen, 2009) and Ryanodex
® (Eagle Pharmaceutical, 2014) [
6,
7]. Furthermore, nanocrystal suspensions are commonly employed in the preclinical stages for toxicological studies, highlighting their importance in early formulation development [
8].
Nanocrystal formulations typically consist of an aqueous nanosuspension with particle sizes ranging from 100 to 1000 nm. However, these nanoparticle suspensions require stabilizing excipients due to their high surface energy. There are two types of stabilizers commonly used in the literature: ionic stabilizers for thermodynamic and electrostatic stabilization such as sodium dodecyl sulfate or sodium deoxycholate, and steric stabilizers for kinetic stabilization [
9,
10]. Steric stabilizers can be classified as non-ionic (e.g., polysorbate 80) and polymeric stabilizers (e.g., copovidone). The most effective stabilization is achieved by combining both types, known as electro-steric stabilization, which exhibits a synergistic effect [
11,
12].
Basically, there are two main approaches for size reduction techniques: bottom-up and top-down [
13]. Top-down approaches involve breaking down large particles into smaller ones using methods like media milling or high-pressure homogenization. Bottom-up approaches involve particle growth or formation through processes like precipitation [
14]. However, controlling particle growth in bottom-up approaches is difficult, making them less suitable for larger scales and commercial-scale production in the pharmaceutical industry [
14].
Aqueous suspensions of nanocrystals can pose certain risks of instabilities, including physical instabilities like Ostwald ripening, sedimentation, and agglomeration [
15]. Moreover, chemical instabilities such as hydrolysis or microbial growth can limit the product shelf life [
16]. Additionally, the oral administration of the liquid nanosuspension can lead to dosing errors and reduced patient compliance and requires suitable dosing devices. To address these shortcomings, the liquid nanocrystal suspension can be converted into a solid dosage form, such as tablets or capsules filled with powder. This can be accomplished through drying/solidification techniques like spray drying, spray coating (also referred to as spray granulation), and freeze drying [
17,
18]. Each drying technique has its own advantages and limitations.
Independent of the drying technique used, drying protectants (also known as bulking agents, dispersants/protectants or embedding matrix material) are commonly added to the crystalline nanosuspension stabilized with ionic/steric stabilizers prior to the thermally stressful drying step to avoid particle growth/agglomeration. Drying protectants are known from lyophilization and are mostly sugars (sucrose, trehalose, or lactose) or sugar alcohols (mannitol) acting as spacers to avoid crystal-to-crystal contact [
19]. To ensure the positive impact of nanosizing on dissolution, it is crucial to consider the nanoparticulate redispersibility and the preservation of crystallinity in both the solidified intermediate and final solid dosage form.
In summary, stabilization is necessary in two steps:
- (a)
To produce a stable nanosuspension using techniques like wet ball milling, where ionic and/or steric stabilizers are employed.
- (b)
To solidify the nanosuspension through a drying process using drying protectants/bulking agents (such as sugars or sugar alcohols), which act as spacers to prevent crystal-to-crystal contact.
The redispersibility is defined as the amount of the nano proportion after the redispersion of the dried intermediate/final drug product, thus referring to the particle size distribution.
Several methods are available for characterizing nanocrystals in terms of size, but each of them has certain disadvantages regarding accuracy of particle size characterization, when applied as single size characterizing method. Therefore, a combination of at least two methods is recommended [
20,
21]: (1) indirect particle size characterization applying laser-based methods such as dynamic light scattering (DLS) or laser diffraction (LD) and (2) direct particle size characterization applying microscopic methods such as electron microscopy or light microscopy. Combining the two techniques allows characterization of a very large particle number (laser-based methods) including some visual information on particle shape and size of microscopically visual particles.
LD is the preferred method for characterizing nanocrystals regarding particle size distribution. The LD technique has a broader detection range between approximately 10 nm and 3500 µm, but it has inferior resolution compared with DLS in the lower nanometer range. DLS, on the other hand, is not able to detect particles > 3 µm due to the absence of Brownian motion and the related sedimentation. Additionally, DLS has an inherent difficulty in properly analyzing polydisperse samples. Consequently, inhomogeneous “nanoparticle samples” (consisting of various particle fractions having different particle sizes > 1 µm) cannot be detected accurately using DLS. This limitation of the technique should be considered as it represents an issue when DLS is applied as the only method used to investigate nanoparticle suspensions.
In contrast to laser-based methods, microscopic methods can only provide a very small sample of the total particle population. The resolution of light microscopes is limited to around 200 nm, making light microscopy useful only for particles above 1 µm. Images of particles can be misleading when drawing conclusions about the total particle population. However, in combination with laser-based methods, the images can be helpful to verify and complement the laser-based measurements.
Several published studies reported on the solidification of crystalline nanosuspensions [
14,
18]. Czyz, Wewers [
22] investigated the impact of spray drying temperature, the type of drying, and the API particle size on the redispersibility of naproxen and itraconazole. Wewers, Finke [
23] described the formulation parameters on the redispersiblity of naproxen nanoparticles from granules produced in a fluidized bed process. Another study focused on freeze drying, assessing formulation and process factors on the redispersibility of cilostazol nanocrystals [
24]. A comparative study on silybin nanosuspension dried via freeze and spray drying was described by Ma, Gao [
25]. Fluid bed granulation was also investigated to convert indomethacin nanosuspensions into solid dosage forms [
26].
Recently, vacuum drum drying was introduced as a novel drying technique in the manufacturing of ASD-based drug products [
27] as well as for the solidification of nanocrystal suspensions [
28]. However, an experimental study comparing vacuum drum drying with spray drying as a commonly used drying technique for crystalline nanosuspensions and exploring benefits and distinctions of each technique is missing. In the literature, several studies investigated the manufacturability and dissolution of nanocrystalline-based tablets [
26,
29]. However, the specific impact of the compaction pressure on the redispersibility of a solidified nanocrystalline intermediate addressing even a mechanistic understanding by considering tablets with tensile strengths higher than usually targeted was not reported.
Thus, the present study compares spray drying (small-scale) with vacuum drum drying (pilot-scale) in terms of manufacturability (yield, visual appearance as, e.g., product spreading on drums (VDD) or string formation (SD)), redispersibility and downstream-processability of resulting intermediates including compression analysis. For this study, solid nanocrystal powders were manufactured at different drug loads (22%, 33%, 44%
w/
w (solid)) with different commonly used drying protectants (mannitol, mannitol/trehalose mix (1:1) or trehalose). Czyz, Wewers [
22] and Schönfeld, Westedt [
28] have previously reported that the glass transition temperature (
Tg) of formulations can affect redispersibility, depending on the drying process parameters employed. In order to assess a wide range of
Tg values for the resulting solids, trehalose was selected as a component with a high
Tg (115 °C [
30]), mannitol with a low
Tg (87 °C [
30]), and a 1:1 mixture with a
Tg in-between. Subsequently, the solid intermediates were characterized for drying effectiveness and the extent to which the nanoparticulate nature of the drug product was preserved after drying. In addition, the impact of tableting on the redispersibility of these solid intermediates was assessed at increasing compaction pressures.
Vacuum drum drying is a continuous drying process where the solution/suspension is being dispensed between the gap of two counter-rotating drums. This leads to the formation of a thin film on both drums, which is subsequently dried through the application of vacuum and drum heating. After approximately three-fourths of a drum turn, the dried film is scraped off by knives. The drying effectiveness relies on various factors including the temperature of the drums, their rotation speed (which determines the duration of residence time), the vacuum conditions, and the formulation properties such as the solid load of the liquid. During spray drying, the feed solution/suspension is sprayed into a hot gas stream to evaporate the solvent. In the pharmaceutical industry, spray drying is a commonly used standard drying process, whereas VDD is rarely employed. However, VDD can offer advantages over SD including cost-effectiveness, higher yields and suitability for early phase development. Additionally, VDD normally eliminates the need for a secondary drying step and does not have viscosity limitations, making it suitable for processing even pastes. This allows for high solid loads in the solution/suspension, reducing solvent and solvent recovery costs.
ABT-199, also known as Venetoclax, was chosen as example compound for this study with the following physicochemical properties (form I): poor water solubility (<4 ng/mL at pH 7.4 in aqueous buffer) [
31], melting point of 145 °C and glass transition temperature of 121 °C. Crystalline ABT-199 nanosuspensions were stabilized by sodium deoxycholate and copovidone and were prepared by wet ball milling. ABT-199 is a potent and selective BCL-2 inhibitor approved for CLL (chronic lymphocytic leukemia), SLL (small lymphocytic lymphoma) and acute myeloid leukemia and marketed by AbbVie Inc., North Chicago, IL, USA [
32,
33,
34]. Sodium deoxycholate functions as an ionic stabilizer, while copovidone acts as a polymeric steric stabilizer. When combined, they work synergistically to provide electro-steric stabilization of the nanosuspension during the wet ball milling process. The stabilizer selection was based on previously performed miniaturized formulation screening.
4. Discussion
This study comparing a commonly used spray drying technique versus a vacuum drum drying technique for the solidification of nanocrystals revealed three main findings. First, the drying technique can impact the nanoparticulate nature and related redispersibility behavior and affects the bulk properties of the intermediates when the same formulation compositions are compared to each other. Second, varying the drug load and the related changes in excipient levels to stabilize the nanoparticles during drying affects the redispersibility of the solid intermediates by influencing the glass transition temperature of the formulation. But the results suggested that this impact can be mitigated by applying the appropriate processing temperature and not exceeding the glass transition temperature of the formulation during drying. Third, the drying technique impacts the bulk properties and related compression behavior and therewith the downstream processability of the nanocrystal-based solid intermediate to a tablet.
Overall, an ABT-199 nanocrystalline suspension was successfully manufactured with good reproducibility through a wet ball milling technique utilizing zirconium oxide beads in a pilot-scale bead agitator mill. The d50 and d90 values were in a comparable range for all three sub-baches, indicating the desired quality for a liquid nanosuspension (d90 < 1 µm targeted) before drying to a solid intermediate via spray drying or VDD.
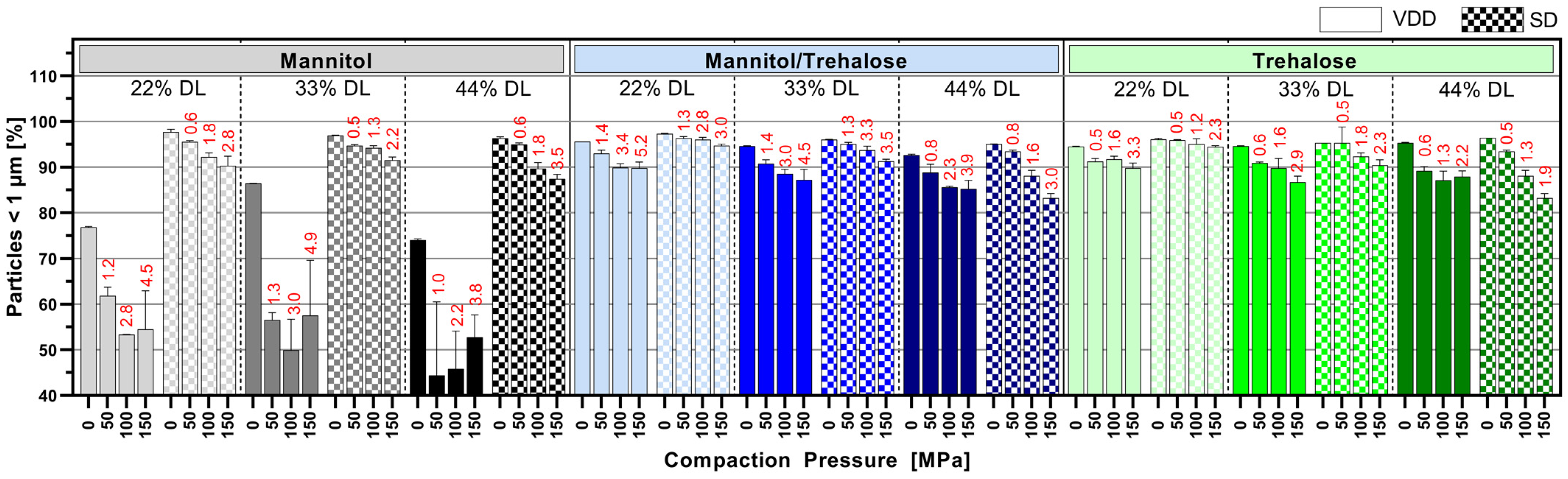
Redispersibility behavior and maintaining the nanoparticulate nature in the solid intermediates seem to be a complex interplay between applied drying technique as well as drug load in the formulation and drying protectant type used. In an assessment of the impact of drug load (22%, 33%, 44%) and the respective drying protectant types and proportions within the formulation on redispersibility, the data implied that redispersibility was mainly influenced by the type of drying protectant and the drying technique itself. The impact of the drying technique was already described by Czyz, Wewers [
22] for naproxen and itraconazole, where the spray drying process boundary (outlet temperature) for redispersibility to a nanocrystalline suspension correlates with the glass transition temperature of the pure drying protectant, as shown for trehalose, sucrose, and lactose. Similar observations were made by Schönfeld, Westedt [
28] for vacuum drum drying in an investigation of mannitol, trehalose, and lactose as drying protectants for a ritonavir nanosuspension. Therefore, a lower process temperature in VDD was initially applied for the mannitol-only (55 °C) compared to the mannitol/trehalose or trehalose-only formulation (75 °C) because the glass transition temperature of mannitol is lower than that of trehalose [
30]. However, the drum temperature of 55 °C for the mannitol formulation was obviously above the critical temperature in the present study because the particle size data of the redispersed VDD intermediate suggest the formation of agglomerates/particle growth. Consequently, the drying of the mannitol formulation was repeated at an even lower drum temperature of 45 °C. But at this temperature, the nanosuspension showed less adherence to the drum surface, so the nanosuspension could not be dried. At a drum temperature of 50 °C, the nanosuspension was processed, but again, redispersibility properties were not enhanced. In contrast, SD mannitol formulations demonstrated good redispersibility and maintained their initial PSD after nano milling in this study. The preservation of the nanoparticulate nature/size can be explained by the lower outlet temperature (40 °C) during spray drying and shorter residence time of each particle within the spray dryer, resulting in reduced exposure to thermal stress during drying. The mean residence time in a Büchi B-290 spray dryer is about 1.0 to 1.5 s according to the vendor’s technical datasheet [
35]. In contrast, the VDD drum speed was set to 0.3 rpm, resulting in a considerably longer mean residence time of approximately 120 to 150 s.
However, a slight trend towards a loss of nano proportion with increasing ABT-199 drug load was observed in the present study for both drying techniques and for all drying protectant types used. Because the amount of drying protectant in the formulation composition decreases with an increase in the API load, nanoparticle aggregation might be facilitated by the reduced spacer effect of the drying protectant as Zuo, Sun [
36] described previously. According to their explanation, water-soluble additives like mannitol can create hydrophilic bridges between the nanoparticles to prevent crystal-to-crystal contact and ultimately crystal growth. In addition, Wewers, Finke [
23] showed that there is a correlation between the concentration-dependent nanoparticle distance within a solid intermediate and its nano-dispersibility. This same stabilizing concentration-dependent effect can be observed in the lyophilization processes of nanoparticles, where sugar is utilized as a so-called “cryo-protectant” to prevent agglomeration by acting as a spacer [
37]. Moreover, Malamatari, Somavarapu [
38] described the mannitol-to-drug ratio as a critical parameter affecting redispersibility, and similar observations were made by Sun, Ni [
39], explaining the decrease in nano proportion in the presented study.
In the present case, the most favorable ABT-199 drug load for the target final dosage form “tablets” seems to be 22% considering redispersibility. Often, formulations with high drug loads are desired to minimize pill burden for patients, especially if high dosage strengths are required. Despite the feasibility of manufacturing high-drug-loaded nanocrystalline powders, the redispersibility of resulting tablets might not be given to its full extent since the applied compaction pressure may destroy the nanoparticulate structure by resulting in agglomeration. However, the impact of a minor loss of the nano proportion (e.g., down to 80–90%) due to compression on bioavailability has not been extensively studied yet. Many studies assessed the dissolution of nanocrystal-based tablets without confirming their quality in terms of nano-dispersibility after compression. For instance, dissolution profiles were beneficial for an itraconazole nanocrystal formulation dried via spray drying compared to a microcrystalline formulation [
30]. Li, Zhou [
40] demonstrated that tablets consisting of loratadine nanocrystals showed similar or better dissolution behavior, depending on the dissolution medium used, and favorable bioavailability compared to a marketed formulation and crude drug tablet. But the presence of nanocrystals and the exact nanocrystalline proportion after redispersion of manufactured tablets has not been assessed in both cases.
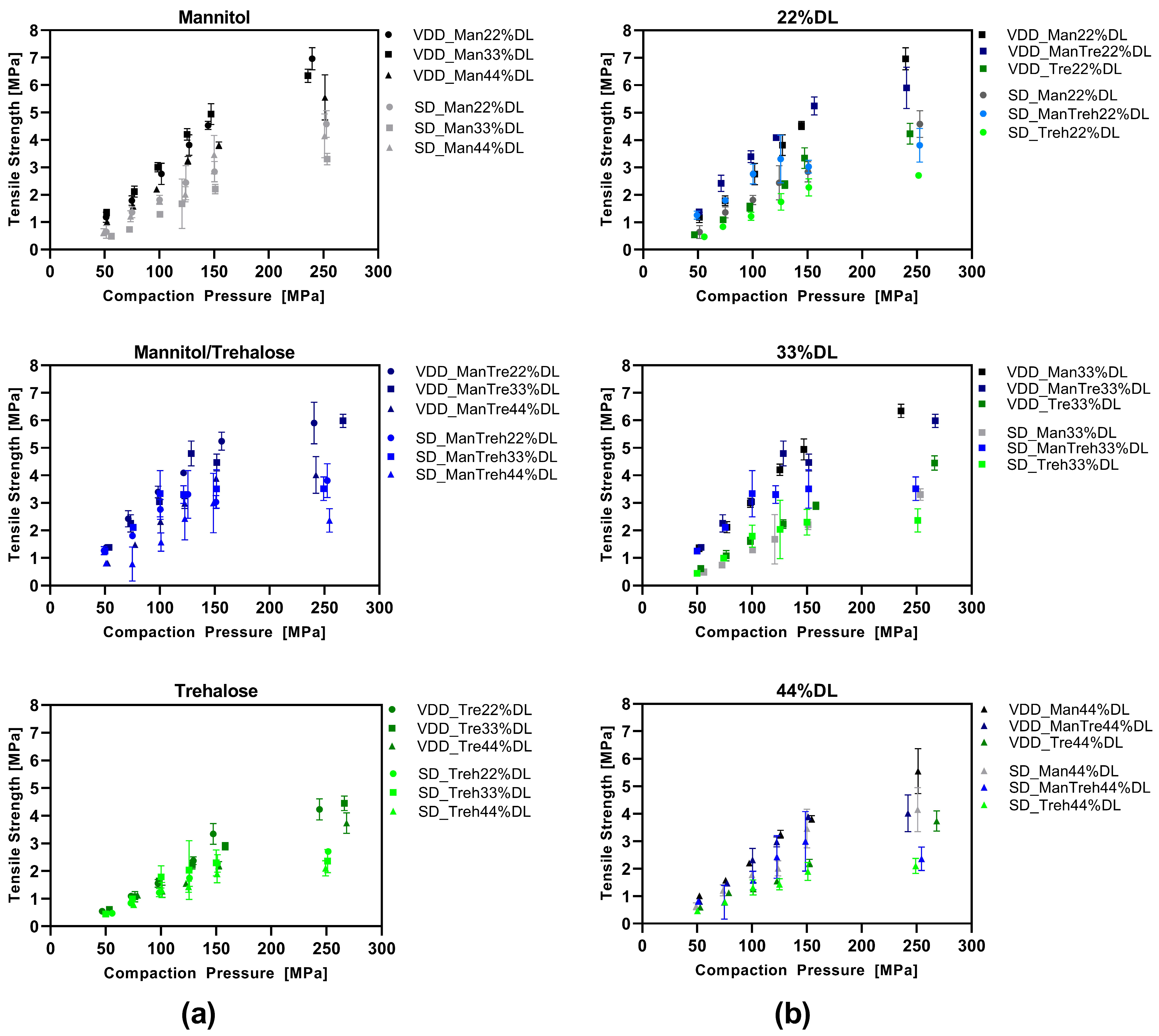
The data presented here indicated a certain impact of compaction pressure resulting in poorer redispersibility. In particular, for the 44% drug-loaded formulations with trehalose or mannitol/trehalose, it was discovered that a higher compaction pressure results in lower redispersibility due to agglomeration of the nanocrystals. However, when comparing different drug loads and drying protectants in formulations processed by different drying techniques to each other, it needs to be considered that applied compaction pressures resulted in different tensile strength values. Thus, the respective tablet formulations have different mechanical strengths which may impact the redispersibility and the proportion of particles maintaining their nanoparticulate nature. Thus, a direct comparison of applied compaction pressure versus redispersibility behavior needs to consider the difference in the tensile strength of the tested tablets. In all cases, the compression of VDD intermediates led to higher tensile strength values at tested compaction pressures. Since the mechanical strength of a tablet is correlated with the inner porosity of a tablet (also in the present case, see
Figure S2), the propensity for crystal-to-crystal contact is higher for tablets with higher mechanical strengths. A tensile strength of approximately 1.5 MPa is the desired target for drug product development. In most SD and VDD tablets, a tensile strength of ~1.5 MPa was already achieved at low compaction pressures of 50–100 MPa. In contrast to compaction pressures > 100 MPa, the application of a compaction pressure of 50–100 MPa showed no or only minor impact on the redispersed nano proportion (particles <1 µm: >80%). Interestingly, as soon as the solid intermediate showed poor redispersibility after drying like the VDD pure mannitol formulations, the impact of compaction pressure on redispersibility was even more pronounced. Consequently, it seems to be crucial to generate a physically stable solid intermediate first where the nanoparticulate structure is fully maintained after drying. The impact of target tensile strengths up to 2.0 MPa has been assessed by Schönfeld, Westedt [
28] for solidified ritonavir nanocrystals. In contrast to the present study, there was no impact of the tableting process on redispersibility observed for tensile strengths up to 2.0 MPa. Although the risk of agglomeration may theoretically increase with increasing compaction pressure, the threshold where the tableting process affects the redispersibility of nanocrystals seems to be formulation- and API-specific and therefore needs to be considered in drug product development. Similar API-specific characteristics for redispersibility were observed for granules loaded with nanoparticles by Wewers, Finke [
23]. They reported a minimum mean nanoparticle distance to avoid agglomeration specifically for itraconazole (100 nm) and naproxen (80 nm). This minimum mean nanoparticle distance might be dominated by API particle morphology factors, such as shape or size, and physicochemical properties. Future studies should also focus on evaluating the reproducibility of the tableting process, especially on larger-scale equipment in a long-term run to obtain a comprehensive understanding of the tableting process for solidified nanocrystals. However, to avoid tableting and related redispersion issues of the nano proportion, encapsulation is known to be a less stressful unit operation applying no or only minor compaction pressures. Thus, a powder-filled capsule can be considered as an alternative as a final dosage form unit for nanocrystal products.
In a comparison of downstream processability to tablets of intermediates manufactured either via SD or VDD, the VDD intermediates showed generally favorable powder bulk properties such as improved flowability, lower LOD values after drying and higher bulk density. The SD intermediates consisted of small, spherical particles as is common for the spray drying process [
41,
42]. Such particles exhibit a high agglomeration tendency as a solid indicated by a bimodal particle size distribution (
Figure 4b) and unfavorable electrostatic behavior. These factors contribute to poor flowability and low bulk density. In contrast, VDD intermediates were larger and more platelet-like as observed in previous studies [
27]. The VDD bulk density for ABT-199 intermediates was in the similar range as previously reported for solidified ritonavir nanocrystal intermediates processed with similar drying protectants [
28]. This suggests that the powder properties might be mainly affected by the drying protectant type and drying technique, with the API having less impact. Despite the absence of secondary drying, the LOD values, and consequently the moisture content values for the VDD intermediates, were significantly lower compared to the spray-dried ones. This difference can be attributed to the longer total drying time in the VDD process compared to the SD process (VDD: 120–150 s, SD: 1.0–1.5 s, see above) and the selected SD process conditions with a relatively low outlet temperature.
The VDD intermediates demonstrated enhanced manufacturability in terms of die filling due to their favorable bulk properties in comparison to the SD intermediates. Furthermore, the VDD intermediates exhibited improved tabletability characterized by higher tensile strength values at comparable compaction pressures and the absence of tablet defects, as demonstrated by the compression analysis. This superior tabletability for the VDD intermediate was unexpected since the bulk particle size of the SD intermediates was much smaller than that of the VDD intermediates. A potential explanation could be that the irregular-shaped particles enable mechanical interlocking during compression combined with plastic deformation of the polymeric components. In future studies, measurements of the specific surface area would be of interest to see if there are differences in total surface area and, thereby, total bonding area.
The LOD values for pure mannitol-containing formulations were comparable for SD and VDD but much higher for mannitol/trehalose SD intermediates compared to VDD. Usually, a water content in a certain range improves the tabletability of a bulk material compared to dryer bulk materials [
43,
44]. As shown by Sun [
43] for microcrystalline cellulose, the optimum water content considering tensile strength was between 3.3 and 5.6% moisture. However, tabletability was improved independent of the type of drying protectants for VDD intermediates, indicating that water content is less likely to be the primary factor and the lower water content of the VDD intermediates did not adversely affect tabletability. Furthermore, tablets containing mannitol exhibited the highest tensile strength and mechanical strength. Mannitol is a commonly used tablet binder in the pharmaceutical industry due to its favorable binding properties [
45], while trehalose is not typically employed for this purpose. The tabletability generally decreased as the drug load increased, which can be attributed to the fact that the API has inferior compactability properties in comparison to the drying protectant, even though the proportion of copovidone, a good binder, also increased with increasing drug load [
46,
47]. A potential explanation is that crystalline structures require higher compaction pressures compared to amorphous sugar-based drying protectants, which were dissolved in the nanosuspension before drying.
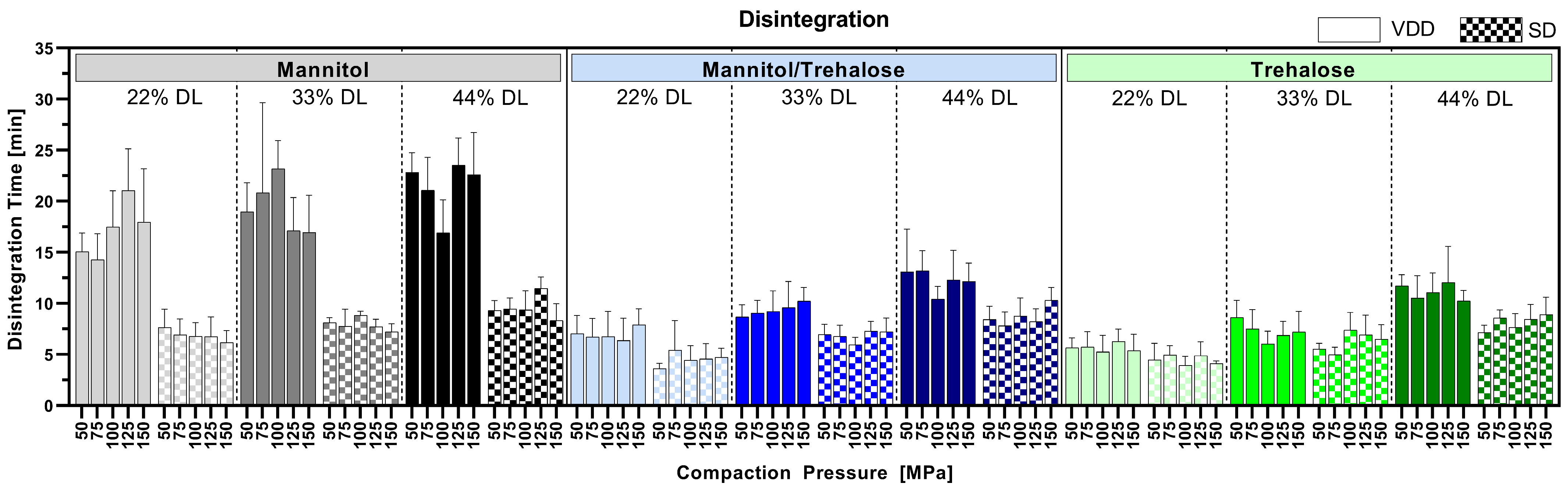
The tablet disintegration time was mainly influenced by the type of drying protectant resulting in the following disintegration time rank order: trehalose < mannitol/trehalose mix < mannitol. The disintegration time was less affected by the final drug load of the formulation or the applied drying process. Interestingly, VDD mannitol formulations showed slower disintegration compared to mannitol/trehalose mix or trehalose formulations. This can be explained by the interplay of several factors. First, both drying protectants are soluble in water, with mannitol having higher water solubility compared to trehalose (mannitol: 180 mg/mL, trehalose: 50 mg/mL) [
48]. However, a long residence time on the drums for mannitol formulations with a low glass transition temperature might lead to a softening and densification of the material itself on the drums. Thus, high-density particles might decelerate the disintegration due to the reduced surface area/porosity. Second, the present study showed larger particle sizes for VDD mannitol formulations compared to SD mannitol intermediates, resulting in a decreased surface area and inner tablet microstructure correlating even more with an increased disintegration time. The impact of particle size (d90) on disintegration time for different mannitol grades has been described by Kosugi, Leong [
49], whereas Skelbæk-Pedersen, Al-Sharabi [
50] observed a difference in water ingress into tablets for different particle sizes of the powder particles based on the predominant powder deformation behavior during compression. Third, tablet hardness is known to impact disintegration [
51]. Therefore, the disintegration times of VDD mannitol tablets are increased due to their highest tensile strengths amongst all others at tested compaction pressures. Fourth, different polymorphs of mannitol might be present in the VDD and SD intermediates in different proportions, potentially influencing the solubility of mannitol in water and therefore influencing the disintegration time. Yang, Liu [
52] described the impact of processing technique on the polymorphism of mannitol and its impact on physicochemical properties such as solubility. Last, the slight trend towards slower disintegration time with increasing ABT-199 drug load might be related to the decreasing proportion of the water-soluble drying protectant. As the amount of water-insoluble ABT-199 proportion increased, the wettability of the tablet was reduced, resulting in a slower disintegration process. Drying protectants are known to improve wettability [
53], leading to enhanced water intake and disintegration. Nevertheless, all intermediates, except for the VDD mannitol ones, were within the USP/Ph. Eur. specification for uncoated immediate-release tablets. Overall, disintegration is not fully understood so far [
54] and is affected by a complex interplay of powder bulk properties, process parameters/manufacturing techniques, tablet properties and tablet microstructure as stated by Sun [
55]. It can be assumed that the presence of nanocrystals adds another variable affecting the disintegration of tablets.
5. Conclusions
The present study demonstrated the applicability of vacuum drum drying (VDD) for the solidification of ABT-199 nanocrystalline suspension into a solid nano-dispersible powder as an alternative to the commonly used spray drying (SD) process.
Maintaining the nanoparticulate nature upon drying was identified as a key quality attribute of the solid nanocrystal-based intermediates in a comparison of both drying techniques using different drying protectants at varying drug loads. Except for vacuum drum drying of mannitol formulations, the redispersibility of intermediates showed that nanoparticulate nature was maintained in both drying techniques at specific process parameters for formulations containing mannitol, a mannitol/trehalose mix or trehalose as a drying protectant. It was discovered that the redispersibility was slightly worse with a decreasing proportion of drying protectants (and increasing drug load) within the formulation. In contrast to VDD, SD intermediates showed good redispersibility and maintained the nanoparticulate structure independent of the applied drying protectant type or amount in the formulation (and correlating drug load 22–33–44%). Thus, the shorter drying process of SD (SD: approx. 1.0–1.5 s; VDD: approx. 120–150 s) is beneficial for avoiding thermal stress to the nanoparticles. The lower thermal stress may enable the achievement of a re-nanodispersible intermediate with higher drug load levels (>44%) compared to VDD. Furthermore, the SD intermediates do not require an additional comminution step for downstream processing but rather a sieving/deagglomeration step since the primary particle size of SD intermediates is essentially smaller compared to VDD intermediates. A subsequent sieving/deagglomeration step generates less mechanical stress and may be less stressful for the nanoparticles compared to a comminution step as applied to VDD intermediates.
The redispersibility of tablets was impacted by the compression process and can result in a loss of nano proportion depending on the intermediate drug load as well as the drying protectant type. For the redispersibility of SD and VDD tablets, compaction pressure during tableting and an increase in the size of nanoparticles after redispersion were correlated, while the disintegration was not impacted. Although SD and VDD intermediates behaved similarly in relation to redispersibility, it was clearly shown that processing via VDD has benefits for downstream processing. VDD processing resulted in more favorable bulk properties like improved flowability, higher bulk density and finally better tabletability at a given manufacturability due to constant automatic die filling. Overall, VDD intermediates containing the mannitol/trehalose mix as a drying protectant combined good redispersibility with good downstream processability by means of good flowability, high bulk density and good tabletability. However, in terms of tablet disintegration, the SD tablets revealed generally faster disintegration compared to the VDD tablets, which may result in faster dissolution after the tablet release compared to the VDD tablets. Overall, comparing VDD to SD, the data indicated further benefits for VDD like higher yield values despite the small batch size for a pilot-scale machine and no need for a secondary drying step, and the particle size distribution is more definable by subsequent milling step.
In conclusion, high-drug-loaded (44%) ABT-199 nanoparticulate tablet formulations processed via VDD or SD were feasible in terms of drying and compression but also maintained the nanoparticulate nature in the final dosage form unit. However, considering the impact of the tableting process on the redispersibility, targeting lower drug loads, and applying low compaction pressures are highly recommended to reduce the risk of agglomeration during processing.