Optimization, In Vitro, and In Silico Characterization of Theophylline Inhalable Powder Using Raffinose-Amino Acid Combination as Fine Co-Spray-Dried Carriers
Abstract
:1. Introduction
2. Materials and Methods
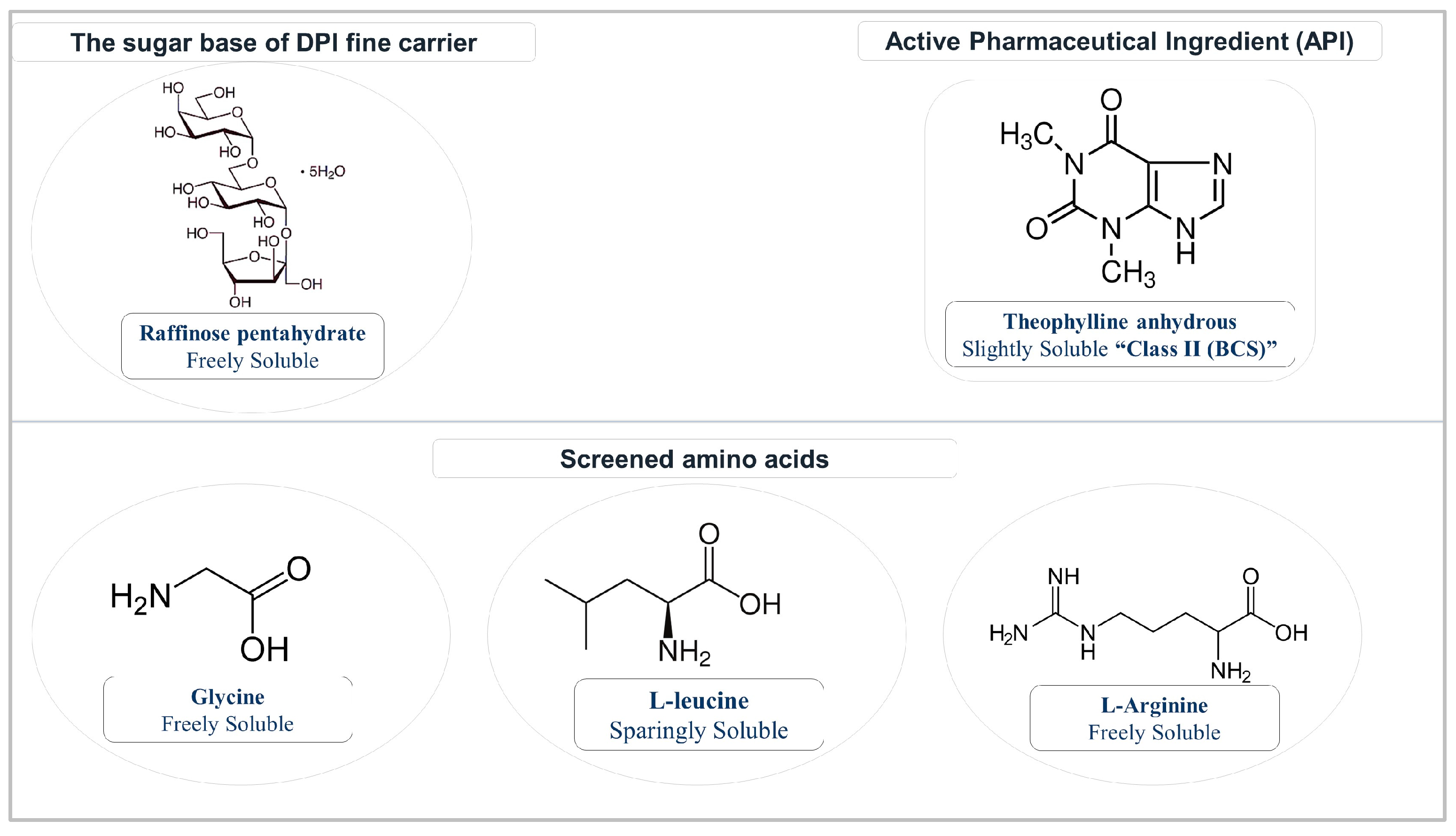
2.1. Materials
2.2. Methods
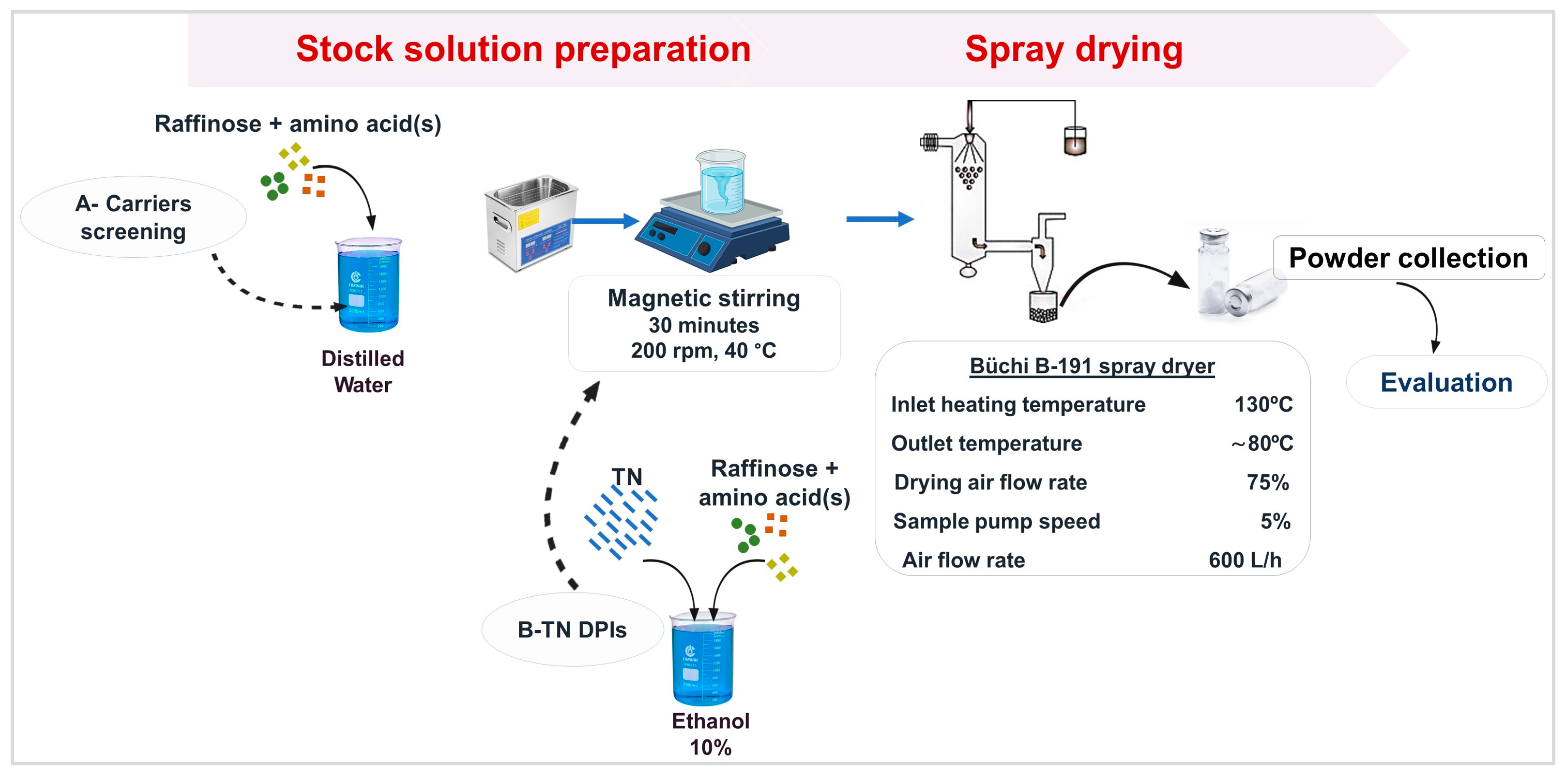
2.2.1. Preparation of Initial Solutions and Spray-Drying Process
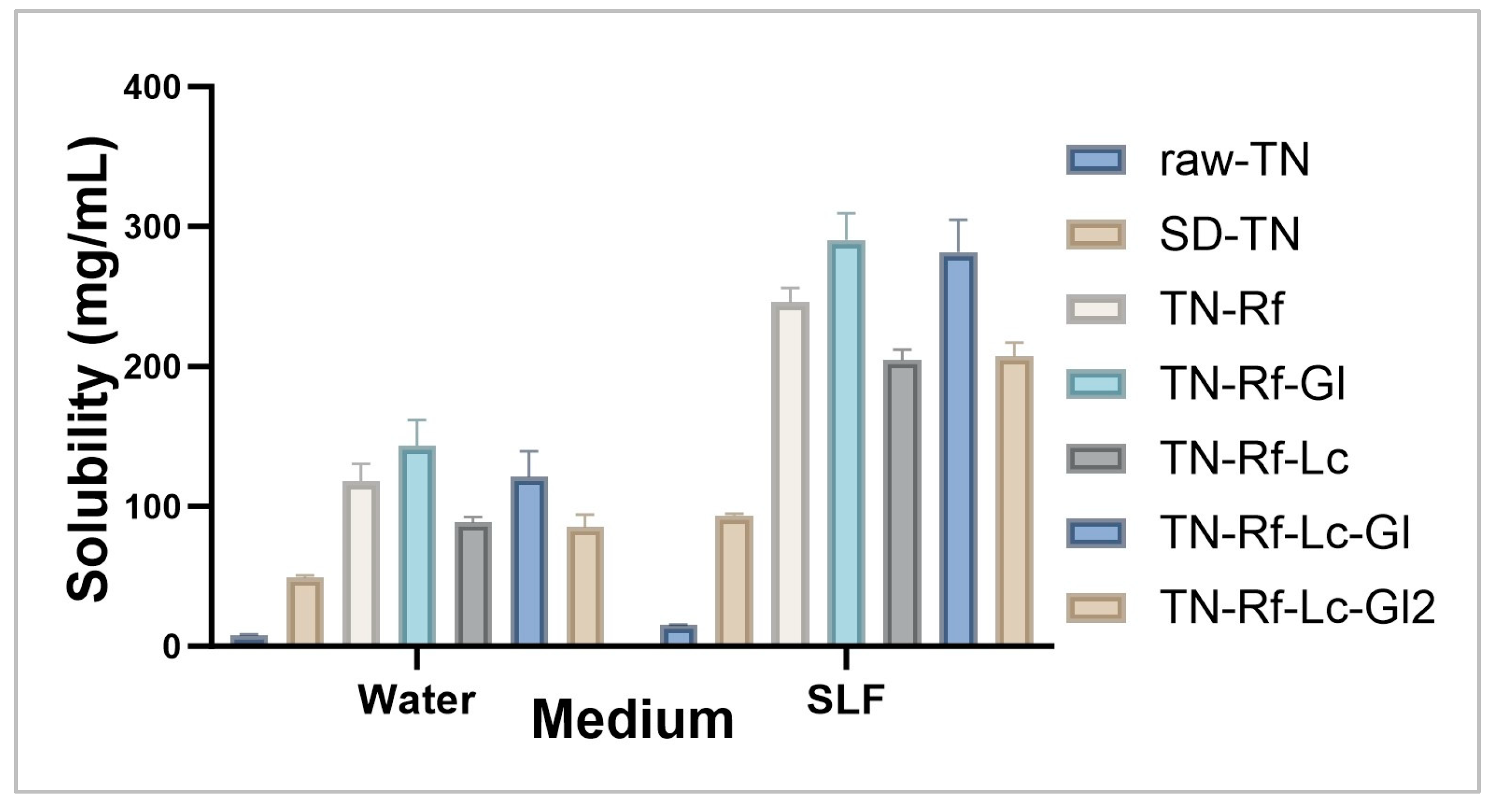
2.2.2. Assessment of TN Solubility
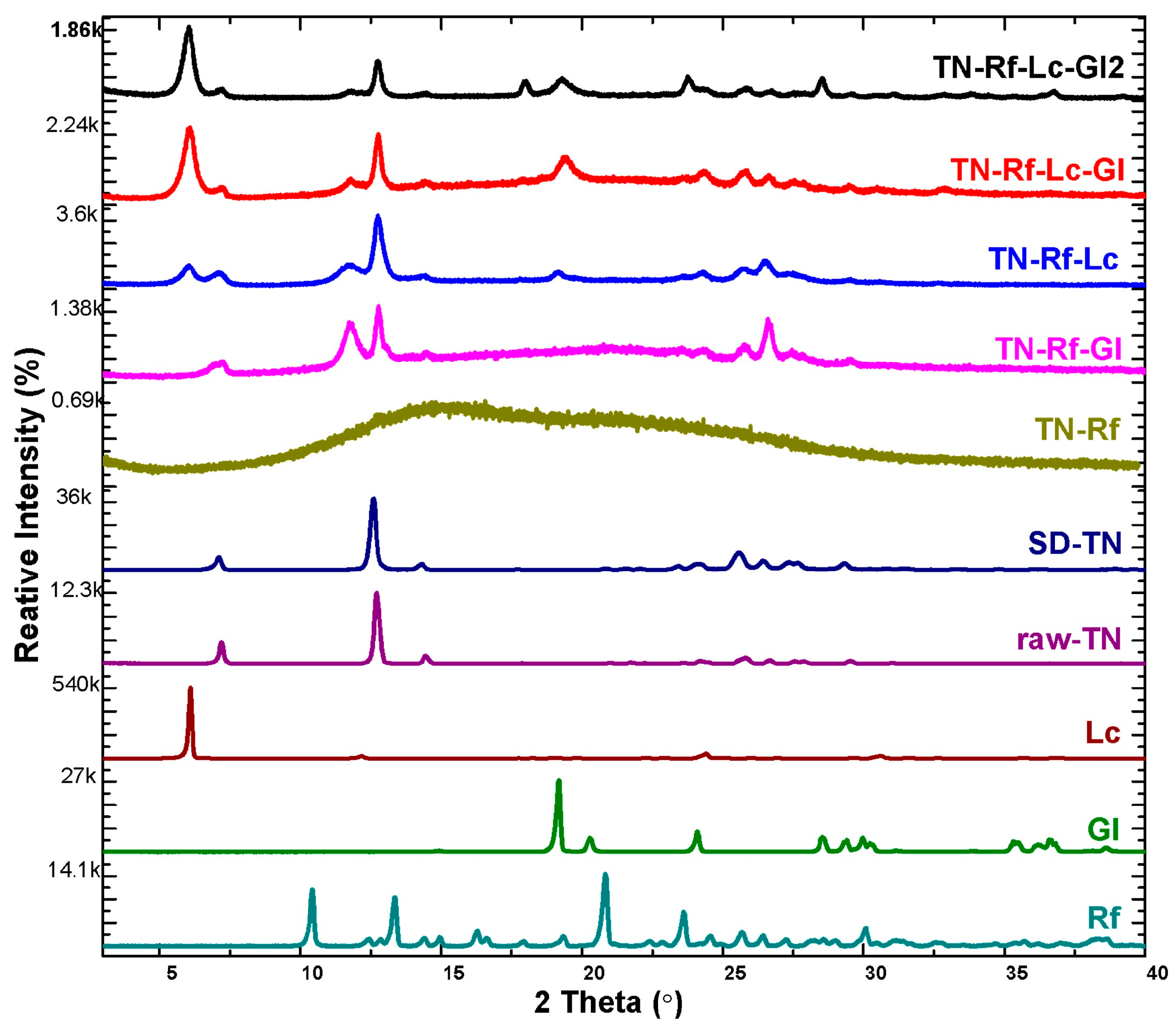
2.2.3. Structural Analysis by X-Ray Powder Diffraction (XRPD)
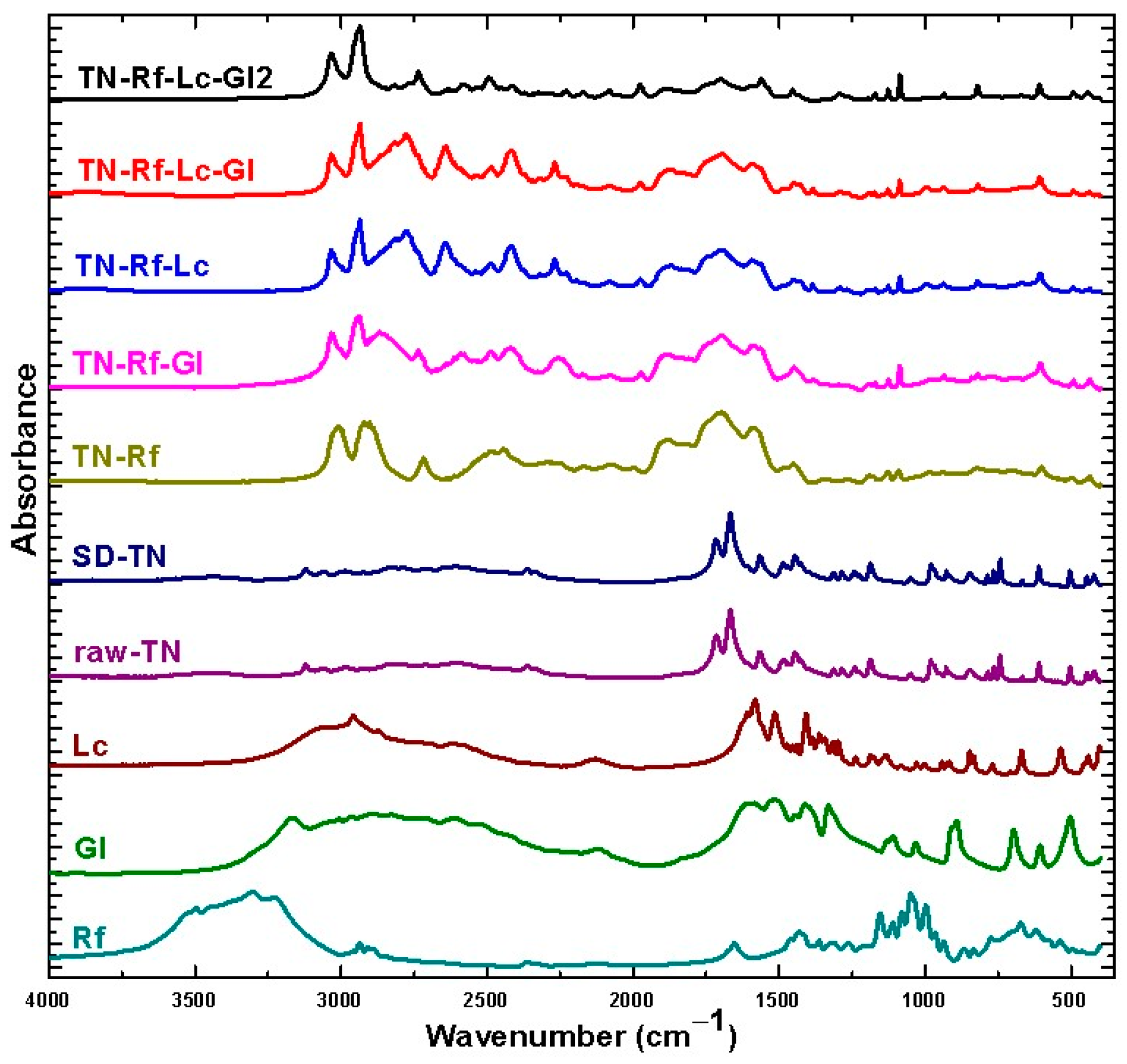
2.2.4. Structural Analysis by Fourier Transform Infrared Spectroscopy (FTIR)
2.2.5. Thermal Analysis by Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA)
2.2.6. Particle Size Distribution Assessment by Laser Diffraction
2.2.7. Morphology Study by Scanning Electron Microscopy (SEM)
2.2.8. Rheology Study
2.2.9. Aerodynamic Assessment
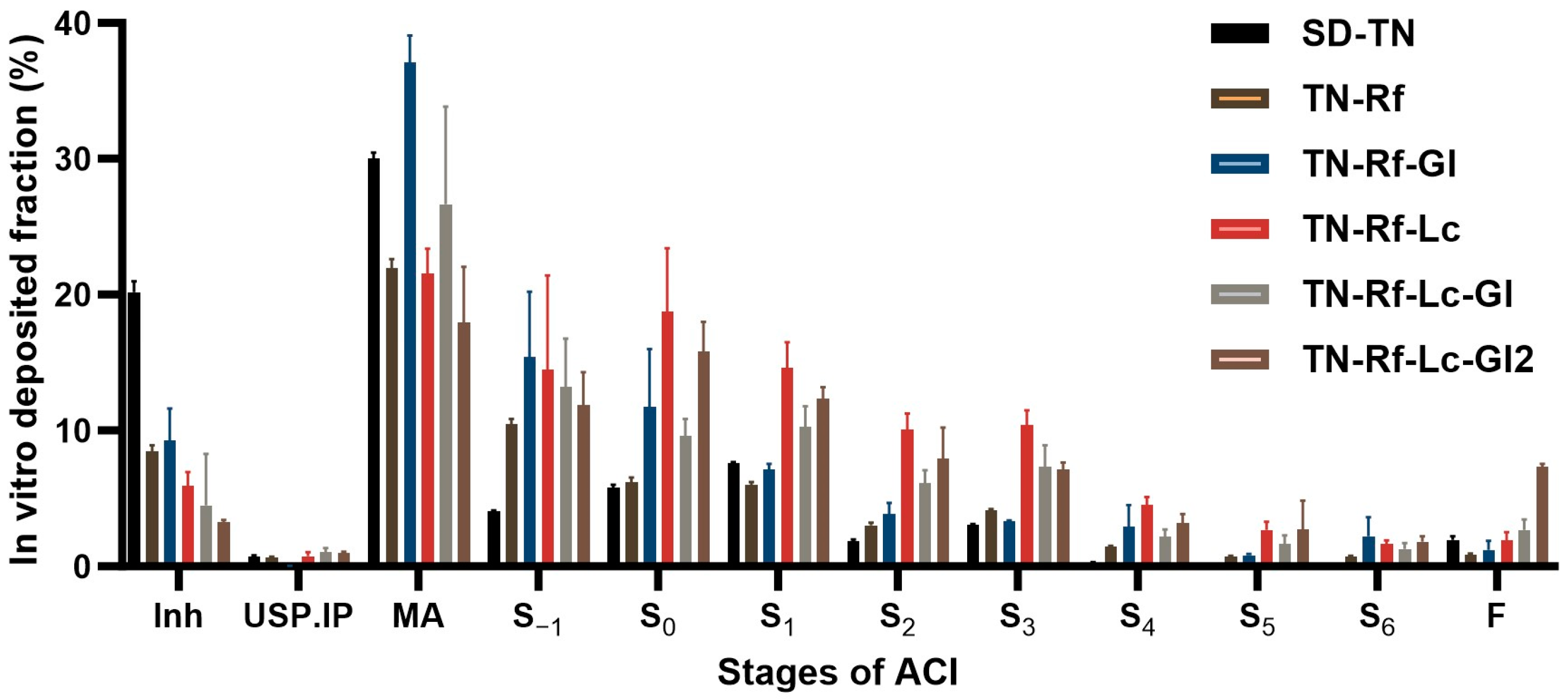
In Vitro Aerodynamic Assessment by Andersen Cascade Impactor
In Silico Aerodynamic Assessment
Aerodynamic Particle Counter (APC) Characterization
2.2.10. In Vitro Dissolution
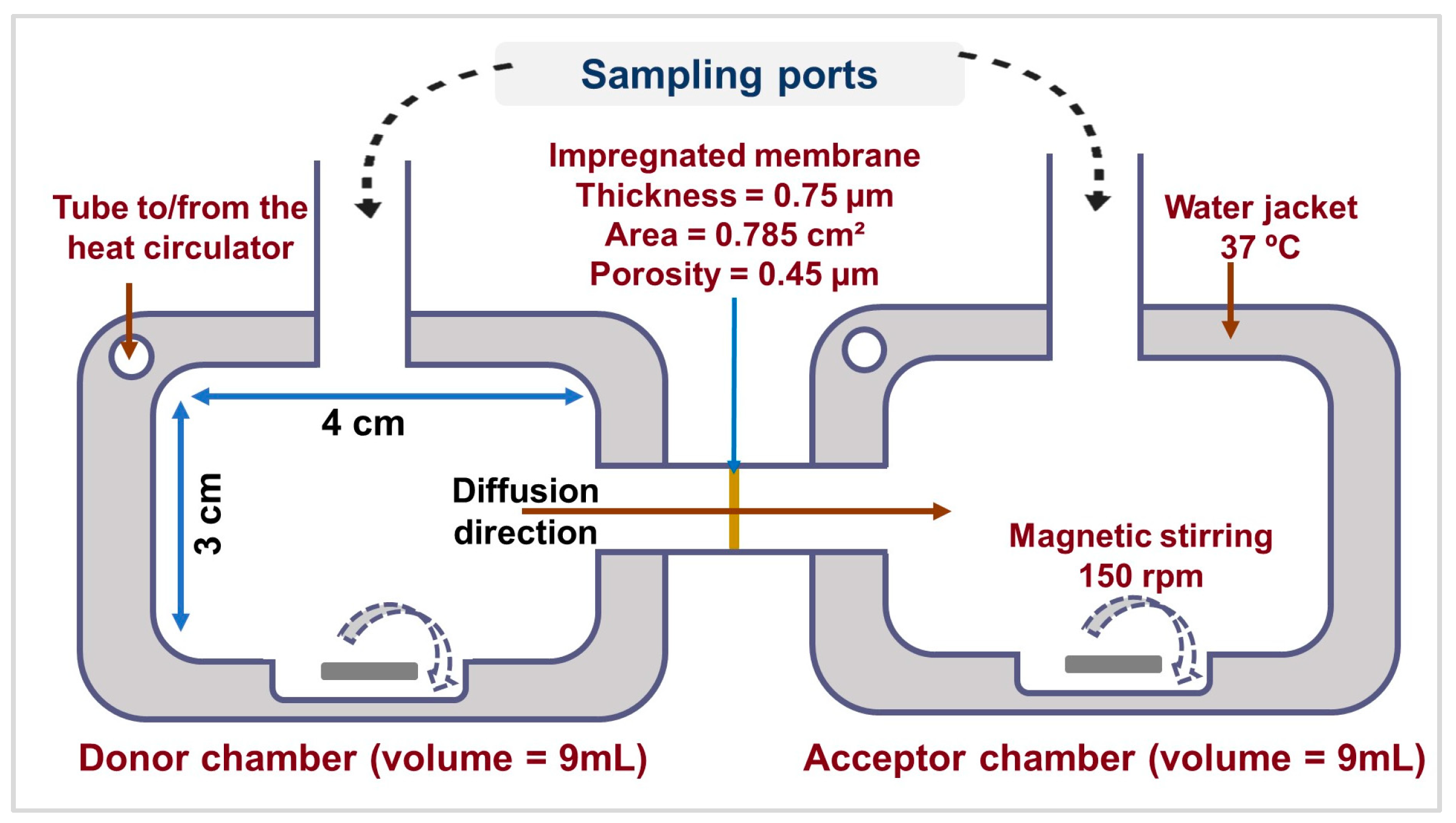
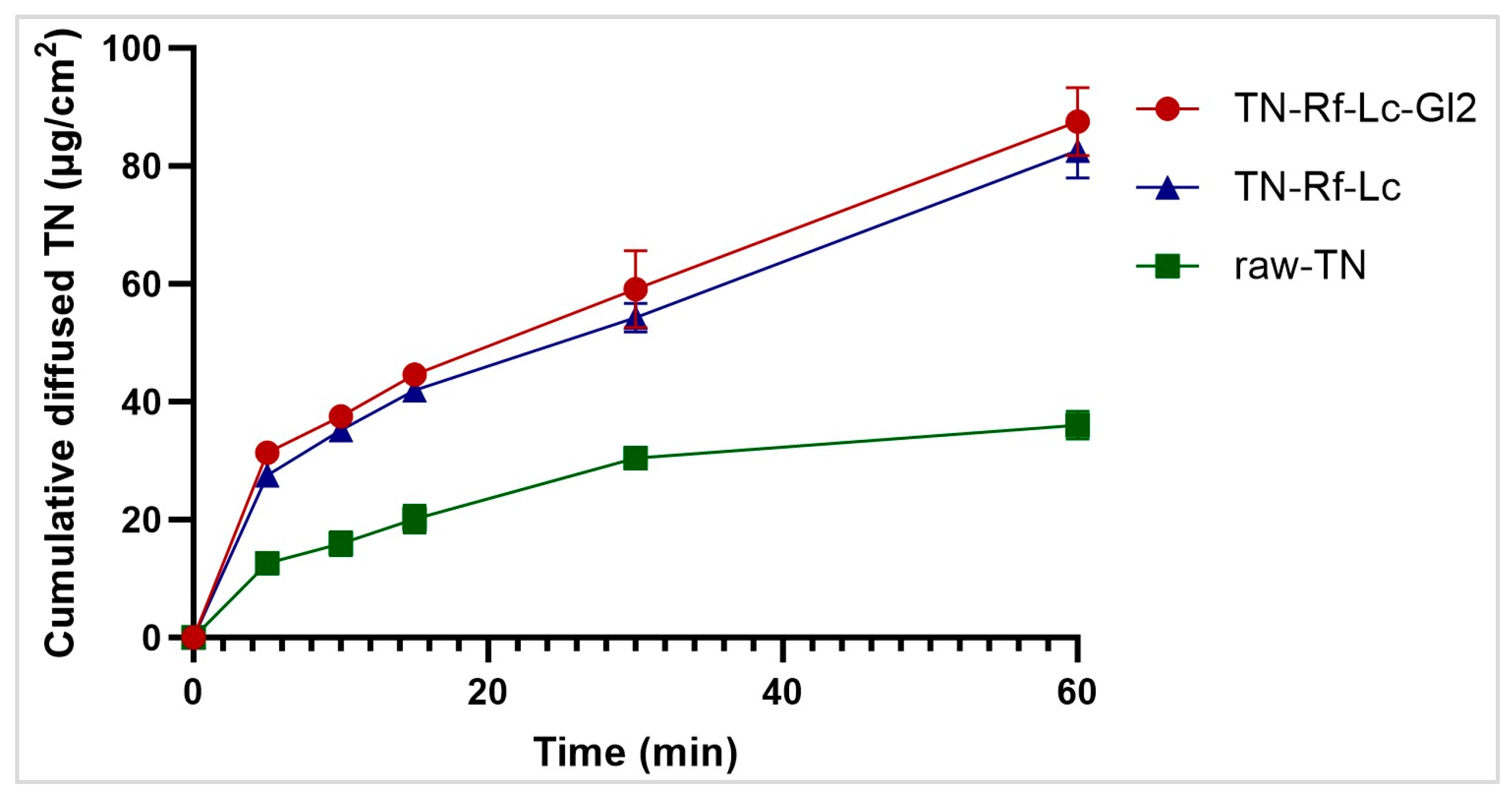
2.2.11. In Vitro Diffusion Evaluation
2.2.12. Statistical Analysis
3. Results
3.1. Screening Results of the Carriers
3.2. Solubility of TN
3.3. X-Ray Diffractograms
3.4. FTIR Spectra
3.5. Thermograms
3.6. Particle Size Distribution by Laser Diffraction
3.7. Morphological Characteristics
3.8. Density and Flowability Properties
3.9. Aerodynamic Properties
3.9.1. In Vitro Aerodynamic Characterization, Andersen Cascade Impactor (ACI)
3.9.2. In Silico Aerodynamic Characterization
3.9.3. Aerodynamic Particle Counter (APC)
3.10. In Vitro Drug Release
3.11. In Vitro Diffusion
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACI | Andersen Cascade Impactor |
| APC | Aerodynamic Particle Counter |
| API | Active Pharmaceutical Ingredient |
| APSD | Aerodynamic Particle Size Distribution |
| CI | Carr’s Index |
| DPI | Dry Powder Inhaler |
| DSC | Differential Scanning Calorimetry |
| EF | Emitted Fraction |
| FPF | Fine Particle Fraction |
| FTIR | Fourier-Transform Infrared |
| HR | Hausner Ratio |
| MMAD | Median Mass Aerodynamic Diameter |
| PDDS | Pulmonary Drug Delivery System |
| PSD | Particle Size Distribution |
| SD | Standard Deviation |
| SEM | Scanning Electron Microscopy |
| SD-TN | Spray Dried Theophylline |
| TGA | Thermogravimetric Analysis |
| TN | Theophylline Anhydrous |
| Xc | Crystallinity Index |
| XRPD | X-Ray Powder Diffraction |
References
- Anderson, S.; Atkins, P.; Bäckman, P.; Cipolla, D.; Clark, A.; Daviskas, E.; Disse, B.; Entcheva-Dimitrov, P.; Fuller, R.; Gonda, I.; et al. Inhaled Medicines: Past, Present, and Future. Pharmacol. Rev. 2022, 74, 48–118. [Google Scholar] [CrossRef] [PubMed]
- Shakshuki, A.; Agu, R.U. Improving the Efficiency of Respiratory Drug Delivery: A Review of Current Treatment Trends and Future Strategies for Asthma and Chronic Obstructive Pulmonary Disease. Pulm. Ther. 2017, 3, 267–281. [Google Scholar] [CrossRef] [PubMed]
- Parihar, A.; Prajapati, B.G.; Paliwal, H.; Shukla, M.; Khunt, D.; Bahadure, S.D.; Dyawanapelly, S.; Junnuthula, V. Advanced Pulmonary Drug Delivery Formulations for the Treatment of Cystic Fibrosis. Drug Discov. Today 2023, 28, 103729. [Google Scholar] [CrossRef] [PubMed]
- Muneer, S.; Wang, T.; Rintoul, L.; Ayoko, G.A.; Islam, N.; Izake, E.L. Development and Characterization of Meropenem Dry Powder Inhaler Formulation for Pulmonary Drug Delivery. Int. J. Pharm. 2020, 587, 119684. [Google Scholar] [CrossRef]
- Pham, D.-D.; Fattal, E.; Tsapis, N. Pulmonary Drug Delivery Systems for Tuberculosis Treatment. Int. J. Pharm. 2015, 478, 517–529. [Google Scholar] [CrossRef]
- Huang, Z.; Kłodzińska, S.N.; Wan, F.; Nielsen, H.M. Nanoparticle-Mediated Pulmonary Drug Delivery: State of the Art towards Efficient Treatment of Recalcitrant Respiratory Tract Bacterial Infections. Drug Deliv. Transl. Res. 2021, 11, 1634–1654. [Google Scholar] [CrossRef]
- Banat, H.; Ambrus, R.; Csóka, I. Drug Combinations for Inhalation: Current Products and Future Development Addressing Disease Control and Patient Compliance. Int. J. Pharm. 2023, 643, 123070. [Google Scholar] [CrossRef]
- Ceschan, N.E.; Bucalá, V.; Ramírez-Rigo, M.V. Levofloxacin Dry Powder Inhaler for High Dose Delivery. Powder Technol. 2024, 432, 119168. [Google Scholar] [CrossRef]
- Khadka, P.; Dummer, J.; Hill, P.C.; Das, S.C. The Quest to Deliver High-Dose Rifampicin: Can the Inhaled Approach Help? Expert Opin. Drug Deliv. 2024, 21, 31–44. [Google Scholar] [CrossRef]
- Party, P.; Piszman, Z.I.; Farkas, Á.; Ambrus, R. Comprehensive In Vitro and In Silico Aerodynamic Analysis of High-Dose Ibuprofen- and Mannitol-Containing Dry Powder Inhalers for the Treatment of Cystic Fibrosis. Pharmaceutics 2024, 16, 1465. [Google Scholar] [CrossRef]
- Shoyele, S.A.; Slowey, A. Prospects of Formulating Proteins/Peptides as Aerosols for Pulmonary Drug Delivery. Int. J. Pharm. 2006, 314, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Masjedi, M.; Montahaei, T.; Sharafi, Z.; Jalali, A. Pulmonary Vaccine Delivery: An Emerging Strategy for Vaccination and Immunotherapy. J. Drug Deliv. Sci. Technol. 2022, 69, 103184. [Google Scholar] [CrossRef]
- Kudo, M.; Ishigatsubo, Y.; Aoki, I. Pathology of Asthma. Front. Microbiol. 2013, 4, 263. [Google Scholar] [CrossRef]
- Boboltz, A.; Kumar, S.; Duncan, G.A. Inhaled Drug Delivery for the Targeted Treatment of Asthma. Adv. Drug Deliv. Rev. 2023, 198, 114858. [Google Scholar] [CrossRef]
- Matera, M.G.; Page, C.P.; Calzetta, L.; Rogliani, P.; Cazzola, M. Pharmacology and Therapeutics of Bronchodilators Revisited. Pharmacol. Rev. 2020, 72, 218–252. [Google Scholar] [CrossRef]
- Barnes, P.J. Theophylline. Am. J. Respir. Crit. Care Med. 2013, 188, 901–906. [Google Scholar] [CrossRef]
- Barnes, P.J. Theophylline: New Perspectives for an Old Drug. Am. J. Respir. Crit. Care Med. 2003, 167, 813–818. [Google Scholar] [CrossRef]
- Weinberger, M.; Hendeles, L. Theophylline in Asthma. N. Engl. J. Med. 1996, 334, 1380–1388. [Google Scholar] [CrossRef]
- Hansel, T.T.; Tennant, R.C.; Tan, A.J.; Higgins, L.A.; Neighbour, H.; Erin, E.M.; Barnes, P.J. Theophylline: Mechanism of Action and Use in Asthma and Chronicobstructive Pulmonary Disease. Drugs Today 2004, 40, 55. [Google Scholar] [CrossRef]
- Barnes, P.; Pauwels, R. Theophylline in the Management of Asthma: Time for Reappraisal? Eur. Respir. J. 1994, 7, 579–591. [Google Scholar] [CrossRef]
- Kawai, M.; Kato, M. Theophylline for the Treatment of Bronchial Asthma: Present Status. Methods Find. Exp. Clin. Pharmacol. 2000, 22, 309. [Google Scholar] [CrossRef]
- Plaunt, A.J.; Nguyen, T.L.; Corboz, M.R.; Malinin, V.S.; Cipolla, D.C. Strategies to Overcome Biological Barriers Associated with Pulmonary Drug Delivery. Pharmaceutics 2022, 14, 302. [Google Scholar] [CrossRef] [PubMed]
- Nelson, H.S. Inhalation Devices, Delivery Systems, and Patient Technique. Ann. Allergy. Asthma. Immunol. 2016, 117, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Islam, N.; Suwandecha, T.; Srichana, T. Dry Powder Inhaler Design and Particle Technology in Enhancing Pulmonary Drug Deposition: Challenges and Future Strategies. DARU J. Pharm. Sci. 2024, 32, 761–779. [Google Scholar] [CrossRef] [PubMed]
- Shahin, H.I.; Chablani, L. A Comprehensive Overview of Dry Powder Inhalers for Pulmonary Drug Delivery: Challenges, Advances, Optimization Techniques, and Applications. J. Drug Deliv. Sci. Technol. 2023, 84, 104553. [Google Scholar] [CrossRef]
- Pilcer, G.; Amighi, K. Formulation Strategy and Use of Excipients in Pulmonary Drug Delivery. Int. J. Pharm. 2010, 392, 1–19. [Google Scholar] [CrossRef]
- Healy, A.M.; Amaro, M.I.; Paluch, K.J.; Tajber, L. Dry Powders for Oral Inhalation Free of Lactose Carrier Particles. Adv. Drug Deliv. Rev. 2014, 75, 32–52. [Google Scholar] [CrossRef]
- Rahimpour, Y.; Kouhsoltani, M.; Hamishehkar, H. Alternative Carriers in Dry Powder Inhaler Formulations. Drug Discov. Today 2014, 19, 618–626. [Google Scholar] [CrossRef]
- Aziz, D.F.; Rostamnezhad, M.; Azad, K.; Ismail Khalil, Y.; Vatanara, A. Screening of Sugars and Cyclodextrins as Carriers in Montelukast Dry Powder Inhalers Processed by Spray Freeze Drying. Adv. Powder Technol. 2023, 34, 104152. [Google Scholar] [CrossRef]
- Pinto, J.T.; Zellnitz, S.; Guidi, T.; Schiaretti, F.; Schroettner, H.; Paudel, A. Spray-Congealing and Wet-Sieving as Alternative Processes for Engineering of Inhalation Carrier Particles: Comparison of Surface Properties, Blending and In Vitro Performance. Pharm. Res. 2021, 38, 1107–1123. [Google Scholar] [CrossRef]
- Ferdynand, M.S.; Nokhodchi, A. Co-Spraying of Carriers (Mannitol-Lactose) as a Method to Improve Aerosolization Performance of Salbutamol Sulfate Dry Powder Inhaler. Drug Deliv. Transl. Res. 2020, 10, 1418–1427. [Google Scholar] [CrossRef] [PubMed]
- Banat, H.; Csóka, I.; Paróczai, D.; Burian, K.; Farkas, Á.; Ambrus, R. A Novel Combined Dry Powder Inhaler Comprising Nanosized Ketoprofen-Embedded Mannitol-Coated Microparticles for Pulmonary Inflammations: Development, In Vitro–In Silico Characterization, and Cell Line Evaluation. Pharmaceuticals 2024, 17, 75. [Google Scholar] [CrossRef]
- Mukhtar, M.; Szakonyi, Z.; Farkas, Á.; Burian, K.; Kókai, D.; Ambrus, R. Freeze-Dried vs Spray-Dried Nanoplex DPIs Based on Chitosan and Its Derivatives Conjugated with Hyaluronic Acid for Tuberculosis: In Vitro Aerodynamic and in Silico Deposition Profiles. Eur. Polym. J. 2021, 160, 110775. [Google Scholar] [CrossRef]
- Amaro, M.I.; Tajber, L.; Corrigan, O.I.; Healy, A.M. Co-Spray Dried Carbohydrate Microparticles: Crystallisation Delay/Inhibition and Improved Aerosolization Characteristics Through the Incorporation of Hydroxypropyl-β-Cyclodextrin with Amorphous Raffinose or Trehalose. Pharm. Res. 2015, 32, 180–195. [Google Scholar] [CrossRef] [PubMed]
- Ógáin, O.N.; Li, J.; Tajber, L.; Corrigan, O.I.; Healy, A.M. Particle Engineering of Materials for Oral Inhalation by Dry Powder Inhalers. I—Particles of Sugar Excipients (Trehalose and Raffinose) for Protein Delivery. Int. J. Pharm. 2011, 405, 23–35. [Google Scholar] [CrossRef]
- Zillen, D.; Beugeling, M.; Hinrichs, W.L.J.; Frijlink, H.W.; Grasmeijer, F. Natural and Bioinspired Excipients for Dry Powder Inhalation Formulations. Curr. Opin. Colloid Interface Sci. 2021, 56, 101497. [Google Scholar] [CrossRef]
- Lu, W.; Rades, T.; Rantanen, J.; Chan, H.-K.; Yang, M. Amino Acids as Stabilizers for Spray-Dried Simvastatin Powder for Inhalation. Int. J. Pharm. 2019, 572, 118724. [Google Scholar] [CrossRef] [PubMed]
- Eedara, B.B.; Alabsi, W.; Encinas-Basurto, D.; Polt, R.; Mansour, H.M. Spray-Dried Inhalable Powder Formulations of Therapeutic Proteins and Peptides. AAPS PharmSciTech 2021, 22, 185. [Google Scholar] [CrossRef]
- Rangnekar, B.; Momin, M.A.M.; Eedara, B.B.; Sinha, S.; Das, S.C. Bedaquiline Containing Triple Combination Powder for Inhalation to Treat Drug-Resistant Tuberculosis. Int. J. Pharm. 2019, 570, 118689. [Google Scholar] [CrossRef]
- Dharmadhikari, A.S.; Kabadi, M.; Gerety, B.; Hickey, A.J.; Fourie, P.B.; Nardell, E. Phase I, Single-Dose, Dose-Escalating Study of Inhaled Dry Powder Capreomycin: A New Approach to Therapy of Drug-Resistant Tuberculosis. Antimicrob. Agents Chemother. 2013, 57, 2613–2619. [Google Scholar] [CrossRef]
- Luinstra, M.; Rutgers, W.; Van Laar, T.; Grasmeijer, F.; Begeman, A.; Isufi, V.; Steenhuis, L.; Hagedoorn, P.; De Boer, A.; Frijlink, H.W. Pharmacokinetics and Tolerability of Inhaled Levodopa from a New Dry-Powder Inhaler in Patients with Parkinson’s Disease. Ther. Adv. Chronic Dis. 2019, 10, 204062231985761. [Google Scholar] [CrossRef]
- Lakio, S.; Morton, D.A.V.; Ralph, A.P.; Lambert, P. Optimizing Aerosolization of a High-Dose L-Arginine Powder for Pulmonary Delivery. Asian J. Pharm. Sci. 2015, 10, 528–540. [Google Scholar] [CrossRef]
- Baldelli, A.; Wong, C.; Oguzlu, H.; Mahvizani, H.; Ong, H.X.; Rodriguez, A.P.; Singhera, G.; Thamboo, A.; Singh, A.; Traini, D.; et al. Impact of Amino Acids on the Properties of Nasal Dry Powders. J. Drug Deliv. Sci. Technol. 2023, 87, 104848. [Google Scholar] [CrossRef]
- Li, H.-Y.; Seville, P.C.; Williamson, I.J.; Birchall, J.C. The Use of Amino Acids to Enhance the Aerosolisation of Spray-Dried Powders for Pulmonary Gene Therapy. J. Gene Med. 2005, 7, 343–353. [Google Scholar] [CrossRef]
- Malamatari, M.; Somavarapu, S.; Kachrimanis, K.; Bloxham, M.; Taylor, K.M.G.; Buckton, G. Preparation of Theophylline Inhalable Microcomposite Particles by Wet Milling and Spray Drying: The Influence of Mannitol as a Co-Milling Agent. Int. J. Pharm. 2016, 514, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Salama, R.O.; Young, P.M.; Traini, D. Concurrent Oral and Inhalation Drug Delivery Using a Dual Formulation System: The Use of Oral Theophylline Carrier with Combined Inhalable Budesonide and Terbutaline. Drug Deliv. Transl. Res. 2014, 4, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Leng, D.; Kissi, E.O.; Löbmann, K.; Thanki, K.; Fattal, E.; Rades, T.; Foged, C.; Yang, M. Design of Inhalable Solid Dosage Forms of Budesonide and Theophylline for Pulmonary Combination Therapy. AAPS PharmSciTech 2019, 20, 137. [Google Scholar] [CrossRef]
- Weers, J.G.; Tarara, T.E.; Clark, A.R. Design of Fine Particles for Pulmonary Drug Delivery. Expert Opin. Drug Deliv. 2007, 4, 297–313. [Google Scholar] [CrossRef]
- Parlati, C. Respirable Microparticles of Aminoglycoside Antibiotics for Pulmonary Administration. Ph.D. Thesis, Universita’ Degli Studi di Parma, Parma, Italy, 2009. [Google Scholar]
- Party, P.; Klement, M.L.; Szabó-Révész, P.; Ambrus, R. Preparation and Characterization of Ibuprofen Containing Nano-Embedded-Microparticles for Pulmonary Delivery. Pharmaceutics 2023, 15, 545. [Google Scholar] [CrossRef]
- Benke, E.; Winter, C.; Szabó-Révész, P.; Roblegg, E.; Ambrus, R. The Effect of Ethanol on the Habit and in Vitro Aerodynamic Results of Dry Powder Inhalation Formulations Containing Ciprofloxacin Hydrochloride. Asian J. Pharm. Sci. 2021, 16, 471–482. [Google Scholar] [CrossRef]
- Benke, E.; Farkas, Á.; Balásházy, I.; Szabó-Révész, P.; Ambrus, R. Stability Test of Novel Combined Formulated Dry Powder Inhalation System Containing Antibiotic: Physical Characterization and In Vitro—In Silico Lung Deposition Results. Drug Dev. Ind. Pharm. 2019, 45, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.B.; Patel, J.K.; Chakraborty, S.; Shukla, D. Revealing Facts behind Spray Dried Solid Dispersion Technology Used for Solubility Enhancement. Saudi Pharm. J. 2015, 23, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Mehanna, M.M.; Mohyeldin, S.M.; Elgindy, N.A. Rifampicin-Carbohydrate Spray-Dried Nanocomposite: A Futuristic Multiparticulate Platform For Pulmonary Delivery. Int. J. Nanomed. 2019, 14, 9089–9112. [Google Scholar] [CrossRef]
- EUROPEAN PHARMACOPOEIA 11.8 Bulk Density Powders; Council of Europe: Strasbourg, France, 2024.
- Thalberg, K.; Lindholm, D.; Axelsson, A. Comparison of Different Flowability Tests for Powders for Inhalation. Powder Technol. 2004, 146, 206–213. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines Preparations for Inhalation: Aerodynamic Assessment of Fine Particles. In European Pharmacopoeia 10.0; Vol. Section 2.9. 18; Council of Europe: Strasbourg, France, 2019; p. 354. ISBN 978-92-871-8912-7.
- Hofmann, W. Modelling Inhaled Particle Deposition in the Human Lung—A Review. J. Aerosol Sci. 2011, 42, 693–724. [Google Scholar] [CrossRef]
- Koblinger, L.; Hofmann, W. Monte Carlo Modeling of Aerosol Deposition in Human Lungs. Part I: Simulation of Particle Transport in a Stochastic Lung Structure. J. Aerosol Sci. 1990, 21, 661–674. [Google Scholar] [CrossRef]
- Farkas, Á.; Lewis, D.; Church, T.; Tweedie, A.; Mason, F.; Haddrell, A.E.; Reid, J.P.; Horváth, A.; Balásházy, I. Experimental and Computational Study of the Effect of Breath-Actuated Mechanism Built in the NEXThaler® Dry Powder Inhaler. Int. J. Pharm. 2017, 533, 225–235. [Google Scholar] [CrossRef]
- Farkas, Á.; Jókay, Á.; Balásházy, I.; Füri, P.; Müller, V.; Tomisa, G.; Horváth, A. Numerical Simulation of Emitted Particle Characteristics and Airway Deposition Distribution of Symbicort® Turbuhaler® Dry Powder Fixed Combination Aerosol Drug. Eur. J. Pharm. Sci. 2016, 93, 371–379. [Google Scholar] [CrossRef]
- Party, P.; Kókai, D.; Burián, K.; Nagy, A.; Hopp, B.; Ambrus, R. Development of Extra-Fine Particles Containing Nanosized Meloxicam for Deep Pulmonary Delivery: In Vitro Aerodynamic and Cell Line Measurements. Eur. J. Pharm. Sci. 2022, 176, 106247. [Google Scholar] [CrossRef]
- Farkas, Á.; Szipőcs, A.; Horváth, A.; Horváth, I.; Gálffy, G.; Varga, J.; Galambos, K.; Kugler, S.; Nagy, A.; Szalai, Z. Establishment of Relationships between Native and Inhalation Device Specific Spirometric Parameters as a Step towards Patient Tailored Inhalation Device Selection. Respir. Med. 2019, 154, 133–140. [Google Scholar] [CrossRef]
- Abadelah, M.; Chrystyn, H.; Larhrib, H. Use of Inspiratory Profiles from Patients with Chronic Obstructive Pulmonary Disease (COPD) to Investigate Drug Delivery Uniformity and Aerodynamic Dose Emission of Indacaterol from a Capsule Based Dry Powder Inhaler. Eur. J. Pharm. Sci. 2019, 134, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Ph. Eur. 2.9.3 Dissolution Test for Solid Dosage Forms. In European Pharmacopeia; EDQM Council of Europe: Strasbourg, France, 2023.
- May, S.; Jensen, B.; Wolkenhauer, M.; Schneider, M.; Lehr, C.M. Dissolution Techniques for In Vitro Testing of Dry Powders for Inhalation. Pharm. Res. 2012, 29, 2157–2166. [Google Scholar] [CrossRef]
- May, S. Dissolution Testing of Powders for Inhalation. Ph.D. Thesis, Faculty of Natural Sciences and Technology, Saarbrücken, Germany, 2013. [Google Scholar]
- Gieszinger, P.; Kiss, T.; Szabó-Révész, P.; Ambrus, R. The Development of an In Vitro Horizontal Diffusion Cell to Monitor Nasal Powder Penetration Inline. Pharmaceutics 2021, 13, 809. [Google Scholar] [CrossRef] [PubMed]
- Sipos, B.; Szabó-Révész, P.; Csóka, I.; Pallagi, E.; Dobó, D.G.; Bélteky, P.; Kónya, Z.; Deák, Á.; Janovák, L.; Katona, G. Quality by Design Based Formulation Study of Meloxicam-Loaded Polymeric Micelles for Intranasal Administration. Pharmaceutics 2020, 12, 697. [Google Scholar] [CrossRef]
- Simon, A.; Amaro, M.I.; Cabral, L.M.; Healy, A.M.; De Sousa, V.P. Development of a Novel Dry Powder Inhalation Formulation for the Delivery of Rivastigmine Hydrogen Tartrate. Int. J. Pharm. 2016, 501, 124–138. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, É.Y.; Amaro, M.I.; De Almeida, G.S.; Cabral, L.M.; Healy, A.M.; De Sousa, V.P. Development of a New Formulation of Roflumilast for Pulmonary Drug Delivery to Treat Inflammatory Lung Conditions. Int. J. Pharm. 2018, 550, 89–99. [Google Scholar] [CrossRef]
- Gharse, S.; Fiegel, J. Large Porous Hollow Particles: Lightweight Champions of Pulmonary Drug Delivery. Curr. Pharm. Des. 2016, 22, 2463–2469. [Google Scholar] [CrossRef]
- Hackl, E.; Darkwah, J.; Smith, G.; Ermolina, I. Effect of Arginine on the Aggregation of Protein in Freeze-Dried Formulations Containing Sugars and Polyol: 1—Formulation Development. AAPS PharmSciTech 2018, 19, 896–911. [Google Scholar] [CrossRef]
- Lewis, D.; Rouse, T.; Singh, D.; Edge, S. Defining the ‘Dose’ for Dry Powder Inhalers: The Challenge of Correlating In-Vitro Dose Delivery Results with Clinical Efficacy. Am. Pharm. Rev. 2017, 20, 54–62. [Google Scholar]
- Newman, S.P. Fine Particle Fraction: The Good and the Bad. J. Aerosol Med. Pulm. Drug Deliv. 2022, 35, 2–10. [Google Scholar] [CrossRef]
- Wolfenden, R.; Andersson, L.; Cullis, P.M.; Southgate, C.C.B. Affinities of Amino Acid Side Chains for Solvent Water. Biochemistry 1981, 20, 849–855. [Google Scholar] [CrossRef]
- Savjani, K.T.; Gajjar, A.K.; Savjani, J.K. Drug Solubility: Importance and Enhancement Techniques. ISRN Pharm. 2012, 2012, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Seton, L.; Khamar, D.; Bradshaw, I.J.; Hutcheon, G.A. Solid State Forms of Theophylline: Presenting a New Anhydrous Polymorph. Cryst. Growth Des. 2010, 10, 3879–3886. [Google Scholar] [CrossRef]
- Li, L.; Leung, S.S.Y.; Gengenbach, T.; Yu, J.; Gao, G.; Tang, P.; Zhou, Q.; Chan, H.-K. Investigation of L-Leucine in Reducing the Moisture-Induced Deterioration of Spray-Dried Salbutamol Sulfate Power for Inhalation. Int. J. Pharm. 2017, 530, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Chan, H.-K.; Gengenbach, T.; Denman, J.A. Protection of Hydrophobic Amino Acids against Moisture-Induced Deterioration in the Aerosolization Performance of Highly Hygroscopic Spray-Dried Powders. Eur. J. Pharm. Biopharm. 2017, 119, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Madarász, J.; Bombicz, P.; Jármi, K.; Bán, M.; Pokol, G.; Gál, S. Thermal, FTIR and XRD Study on Some 1:1 Molecular Compounds of Theophylline. J. Therm. Anal. Calorim. 2002, 69, 281–290. [Google Scholar] [CrossRef]
- Mangal, S.; Nie, H.; Xu, R.; Guo, R.; Cavallaro, A.; Zemlyanov, D.; Zhou, Q. Physico-Chemical Properties, Aerosolization and Dissolution of Co-Spray Dried Azithromycin Particles with L-Leucine for Inhalation. Pharm. Res. 2018, 35, 28. [Google Scholar] [CrossRef]
- Lin, S.-Y.; Chien, J.-L. In Vitro Simulation of Solid-Solid Dehydration, Rehydration, and Solidification of Trehalose Dihydrate Using Thermal and Vibrational Spectroscopic Techniques. Pharm. Res. 2003, 20, 1926–1931. [Google Scholar] [CrossRef]
- Zhu, B.; Haghi, M.; Nguyen, A.; Goud, M.; Yeung, S.; Young, P.M.; Traini, D. Delivery of Theophylline as Dry Powder for Inhalation. Asian J. Pharm. Sci. 2015, 10, 520–527. [Google Scholar] [CrossRef]
- Li, L.; Sun, S.; Parumasivam, T.; Denman, J.A.; Gengenbach, T.; Tang, P.; Mao, S.; Chan, H.-K. L-Leucine as an Excipient against Moisture on in Vitro Aerosolization Performances of Highly Hygroscopic Spray-Dried Powders. Eur. J. Pharm. Biopharm. 2016, 102, 132–141. [Google Scholar] [CrossRef]
- Chew, N.Y.; Chan, H.-K. Effect of Powder Polydispersity on Aerosol Generation. J. Pharm. Pharm. Sci. 2002, 5, 162–168. [Google Scholar] [PubMed]
- Chang, R.Y.K.; Chan, H.-K. Advancements in Particle Engineering for Inhalation Delivery of Small Molecules and Biotherapeutics. Pharm. Res. 2022, 39, 3047–3061. [Google Scholar] [CrossRef] [PubMed]
- Mosharraf, M.; Nyström, C. The Effect of Particle Size and Shape on the Surface Specific Dissolution Rate of Microsized Practically Insoluble Drugs. Int. J. Pharm. 1995, 122, 35–47. [Google Scholar] [CrossRef]
- Chvatal, A.; Ambrus, R.; Party, P.; Katona, G.; Jójárt-Laczkovich, O.; Szabó-Révész, P.; Fattal, E.; Tsapis, N. Formulation and Comparison of Spray Dried Non-Porous and Large Porous Particles Containing Meloxicam for Pulmonary Drug Delivery. Int. J. Pharm. 2019, 559, 68–75. [Google Scholar] [CrossRef]
- Hassan, M.S.; Lau, R.W.M. Effect of Particle Shape on Dry Particle Inhalation: Study of Flowability, Aerosolization, and Deposition Properties. AAPS PharmSciTech 2009, 10, 1252. [Google Scholar] [CrossRef]
- Ordoubadi, M.; Gregson, F.K.A.; Wang, H.; Nicholas, M.; Gracin, S.; Lechuga-Ballesteros, D.; Reid, J.P.; Finlay, W.H.; Vehring, R. On the Particle Formation of Leucine in Spray Drying of Inhalable Microparticles. Int. J. Pharm. 2021, 592, 120102. [Google Scholar] [CrossRef]
- Xu, Y.; Harinck, L.; Lokras, A.G.; Gerde, P.; Selg, E.; Sjöberg, C.-O.; Franzyk, H.; Thakur, A.; Foged, C. Leucine Improves the Aerosol Performance of Dry Powder Inhaler Formulations of siRNA-Loaded Nanoparticles. Int. J. Pharm. 2022, 621, 121758. [Google Scholar] [CrossRef]
- Davis, M.; Walker, G. Recent Strategies in Spray Drying for the Enhanced Bioavailability of Poorly Water-Soluble Drugs. J. Control. Release 2018, 269, 110–127. [Google Scholar] [CrossRef]
- Rissler, J.; Nordin, E.Z.; Eriksson, A.C.; Nilsson, P.T.; Frosch, M.; Sporre, M.K.; Wierzbicka, A.; Svenningsson, B.; Löndahl, J.; Messing, M.E.; et al. Effective Density and Mixing State of Aerosol Particles in a Near-Traffic Urban Environment. Environ. Sci. Technol. 2014, 48, 6300–6308. [Google Scholar] [CrossRef]
- Bosquillon, C.; Lombry, C.; Préat, V.; Vanbever, R. Influence of Formulation Excipients and Physical Characteristics of Inhalation Dry Powders on Their Aerosolization Performance. J. Control. Release 2001, 70, 329–339. [Google Scholar] [CrossRef]
- Berk, Z. Chapter 6—Size Reduction. In Food Process Engineering and Technology; Berk, Z., Ed.; Food Science and Technology; Academic Press: San Diego, CA, USA, 2009; pp. 153–174. ISBN 978-0-12-373660-4. [Google Scholar]
- Naikwade, S. Preparation and In Vitro Evaluation of Budesonide Spray Dried Microparticles for Pulmonary Delivery. Sci. Pharm. 2009, 77, 419–441. [Google Scholar] [CrossRef]
- Benke, E.; Farkas, Á.; Szabó-Révész, P.; Ambrus, R. Development of an Innovative, Carrier-Based Dry Powder Inhalation Formulation Containing Spray-Dried Meloxicam Potassium to Improve the In Vitro and In Silico Aerodynamic Properties. Pharmaceutics 2020, 12, 535. [Google Scholar] [CrossRef]
- Party, P.; Bartos, C.; Farkas, Á.; Szabó-Révész, P.; Ambrus, R. Formulation and In Vitro and In Silico Characterization of “Nano-in-Micro” Dry Powder Inhalers Containing Meloxicam. Pharmaceutics 2021, 13, 211. [Google Scholar] [CrossRef]
- Mukhtar, M.; Pallagi, E.; Csóka, I.; Benke, E.; Farkas, Á.; Zeeshan, M.; Burián, K.; Kókai, D.; Ambrus, R. Aerodynamic Properties and in Silico Deposition of Isoniazid Loaded Chitosan/Thiolated Chitosan and Hyaluronic Acid Hybrid Nanoplex DPIs as a Potential TB Treatment. Int. J. Biol. Macromol. 2020, 165, 3007–3019. [Google Scholar] [CrossRef]
- Hillyer, E.V.; Price, D.B.; Chrystyn, H.; Martin, R.J.; Israel, E.; Van Aalderen, W.M.C.; Papi, A.; Usmani, O.S.; Roche, N.; on behalf of the Respiratory Effectiveness Group. Small Airways Study Group Harmonizing the Nomenclature for Therapeutic Aerosol Particle Size: A Proposal. J. Aerosol Med. Pulm. Drug Deliv. 2018, 31, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Labiris, N.R.; Dolovich, M.B. Pulmonary Drug Delivery. Part II: The Role of Inhalant Delivery Devices and Drug Formulations in Therapeutic Effectiveness of Aerosolized Medications. Br. J. Clin. Pharmacol. 2003, 56, 600–612. [Google Scholar] [CrossRef] [PubMed]
- Borghardt, J.M.; Kloft, C.; Sharma, A. Inhaled Therapy in Respiratory Disease: The Complex Interplay of Pulmonary Kinetic Processes. Can. Respir. J. 2018, 2018, 1–11. [Google Scholar] [CrossRef]
- Bhagwat, S.; Schilling, U.; Chen, M.-J.; Wei, X.; Delvadia, R.; Absar, M.; Saluja, B.; Hochhaus, G. Predicting Pulmonary Pharmacokinetics from In Vitro Properties of Dry Powder Inhalers. Pharm. Res. 2017, 34, 2541–2556. [Google Scholar] [CrossRef]
- Roohpour, N.; Wasikiewicz, J.M.; Moshaverinia, A.; Paul, D.; Grahn, M.F.; Rehman, I.U.; Vadgama, P. Polyurethane Membranes Modified with Isopropyl Myristate as a Potential Candidate for Encapsulating Electronic Implants: A Study of Biocompatibility and Water Permeability. Polymers 2010, 2, 102–119. [Google Scholar] [CrossRef]
- Van Zyl, J.M.; Derendinger, B.; Seifart, H.I.; Van Der Bijl, P. Comparative Diffusion of Drugs through Bronchial Tissue. Int. J. Pharm. 2008, 357, 32–36. [Google Scholar] [CrossRef]
- Kus, M.; Ibragimow, I.; Piotrowska-Kempisty, H. Caco-2 Cell Line Standardization with Pharmaceutical Requirements and In Vitro Model Suitability for Permeability Assays. Pharmaceutics 2023, 15, 2523. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.; Furubayashi, T.; Kataoka, M.; Sakane, T.; Sezaki, H.; Tokuda, H. Optimized Conditions for Prediction of Intestinal Drug Permeability Using Caco-2 Cells. Eur. J. Pharm. Sci. 2000, 10, 195–204. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Guideline on Biopharmaceutics Classification System-Based Biowaivers; Vol. Working document QAS/23.929; WHO: Geneva, Switzerland, 2023. [Google Scholar]





| Formulation | TN (%) | Rf (%) | Lc (%) | Gl (%) | Ar (%) |
|---|---|---|---|---|---|
| Rf | - | 3.5 | - | - | - |
| Rf-Lc | - | 3.5 | 0.75 | - | - |
| Rf-Gl | - | 3.5 | - | 0.75 | - |
| Rf-Ar | - | 3.5 | - | - | 0.75 |
| Rf-Ar-Lc | - | 3.5 | 0.75 | - | 0.75 |
| Rf-Gl-Lc | - | 3.5 | 0.75 | 0.75 | - |
| Rf-Gl-Ar | - | 3.5 | - | 0.75 | 0.75 |
| SD-TN | 1.17 | - | - | - | - |
| TN-Rf | 1.17 | 3.5 | - | - | - |
| TN-Rf-Gl | 1.17 | 3.5 | - | 0.75 | - |
| TN-Rf-Lc | 1.17 | 3.5 | 0.75 | - | - |
| TN-Rf-Lc-Gl | 1.17 | 3.5 | 0.75 | 0.75 | - |
| TN-Rf-Lc-Gl2 | 1.75 | 3.5 | 0.75 | 0.75 | - |
| Formulation | D [0.1] * (µm) | D [0.5] * (µm) | D [0.9] * (µm) | Span * |
|---|---|---|---|---|
| SD-TN | 2.10 ± 0.01 | 4.38 ± 0.05 | 9.85 ± 1.93 | 1.77 ± 0.89 |
| TN-Rf | 2.19 ± 0.09 | 4.89 ± 0.19 | 13.10 ± 3.25 | 2.21 ± 0.55 |
| TN-Rf-Gl | 2.57 ± 0.01 | 5.96 ± 0.22 | 16.21 ± 1.70 | 2.29 ± 0.21 |
| TN-Rf-Lc | 1.72 ± 0.04 | 3.99 ± 0.17 | 13.19 ± 2.94 | 2.86 ± 0.63 |
| TN-Rf-Lc-Gl | 1.88 ± 0.01 | 4.43 ± 0.11 | 19.22 ± 6.63 | 3.90 ± 1.40 |
| TN-Rf-Lc-Gl2 | 1.82 ± 0.03 | 4.23 ± 0.09 | 13.23 ± 3.47 | 2.69 ± 0.77 |
| Formulation | ρbulk * (g/cm3) | ρtapped * (g/cm3) | HR * | CI * (%) |
|---|---|---|---|---|
| Raw-TN | 1.11 ± 0.03 | 1.53 ± 0.07 | 1.37 ± 0.03 | 27.14 ± 1.34 |
| SD-TN | 1.27 ± 0.07 | 1.75 ± 0.09 | 1.38 ± 0.01 | 27.68 ± 0.37 |
| TN-Rf | 0.34 ± 0.03 | 0.45 ± 0.03 | 1.32 ± 0.04 | 24.49 ± 2.25 |
| TN-Rf-Gl | 0.26 ± 0.04 | 0.36 ± 0.05 | 1.40 ± 0.05 | 28.57 ± 2.78 |
| TN-Rf-Lc | 0.36 ± 0.02 | 0.50 ± 0.03 | 1.41 ± 0.03 | 29.24 ± 1.50 |
| TN-Rf-Lc-Gl | 0.44 ± 0.02 | 0.63 ± 0.04 | 1.43 ± 0.05 | 30.25 ± 2.36 |
| TN-Rf-Lc-Gl2 | 0.32 ± 0.04 | 0.46 ± 0.05 | 1.45 ± 0.04 | 31.20 ± 1.88 |
| Formulation | EF * (%) | MMAD * (µm) | FPF * (%) | GSD * | Calculated Delivered Dose (mg) |
|---|---|---|---|---|---|
| SD-TN | 73.30 ± 1.09 | 5.35 ± 0.74 | 32.52 ± 10.63 | 2.05 ± 0.21 | 5.75 ± 0.43 |
| TN-Rf | 86.92 ± 0.45 | 6.44 ± 0.08 | 30.14 ± 0.24 | 2.71 ± 0.06 | 5.62 ± 0.10 |
| TN-Rf-Gl | 90.26 ± 2.56 | 7.20 ± 0.95 | 24.97 ± 4.79 | 2.37 ± 0.01 | 7.35 ± 0.79 |
| TN-Rf-Lc | 94.40 ± 1.05 | 5.33 ± 0.82 | 45.69 ± 4.82 | 2.68 ± 0.15 | 9.58 ± 0.80 |
| TN-Rf-Lc-Gl | 95.08 ± 3.71 | 5.61 ± 0.73 | 38.75 ± 8.02 | 5.36 ± 0.01 | 8.26 ± 0.45 |
| TN-Rf-Lc-Gl2 | 96.45 ± 0.36 | 5.14 ± 0.00 | 47.84 ± 1.36 | 2.72 ± 0.20 | 8.90 ± 1.44 |
| Parameters | Number Particle Size | Surface Particle Size | Volume Particle Size | |||
|---|---|---|---|---|---|---|
| TN-Rf-Lc | TN-Rf-Lc-Gl2 | TN-Rf-Lc | TN-Rf-Lc-Gl2 | TN-Rf-Lc | TN-Rf-Lc-Gl2 | |
| Median * (µm) | 2.01 ± 0.13 | 1.65 ± 0.10 | 3.58 ± 0.15 | 3.49 ± 0.25 | 4.58 ± 0.09 | 4.90 ± 0.09 |
| Mean * (µm) | 2.30 ± 0.11 | 2.02 ± 0.13 | 4.02 ± 0.08 | 4.02 ± 0.13 | 5.12 ± 0.17 | 5.43 ± 0.09 |
| Geometric mean * (µm) | 1.97 ± 0.09 | 1.71 ± 0.10 | 3.54 ± 0.10 | 3.41 ± 0.17 | 4.54 ± 0.10 | 4.71 ± 0.07 |
| Mode * (µm) | 2.49 ± 0.4 | 1.45 ± 0.14 | 3.43 ± 0.23 | 4.33 ± 0.83 | 4.63 ± 0.15 | 5.67 ± 0.35 |
| GSD * | 1.75 ± 0.02 | 1.76 ± 0.03 | 1.67 ± 0.05 | 1.79 ± 0.05 | 1.63 ± 0.06 | 1.73 ± 0.07 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Party, P.; Soliman, L.; Nagy, A.; Farkas, Á.; Ambrus, R. Optimization, In Vitro, and In Silico Characterization of Theophylline Inhalable Powder Using Raffinose-Amino Acid Combination as Fine Co-Spray-Dried Carriers. Pharmaceutics 2025, 17, 466. https://doi.org/10.3390/pharmaceutics17040466
Party P, Soliman L, Nagy A, Farkas Á, Ambrus R. Optimization, In Vitro, and In Silico Characterization of Theophylline Inhalable Powder Using Raffinose-Amino Acid Combination as Fine Co-Spray-Dried Carriers. Pharmaceutics. 2025; 17(4):466. https://doi.org/10.3390/pharmaceutics17040466
Chicago/Turabian StyleParty, Petra, Lomass Soliman, Attila Nagy, Árpád Farkas, and Rita Ambrus. 2025. "Optimization, In Vitro, and In Silico Characterization of Theophylline Inhalable Powder Using Raffinose-Amino Acid Combination as Fine Co-Spray-Dried Carriers" Pharmaceutics 17, no. 4: 466. https://doi.org/10.3390/pharmaceutics17040466
APA StyleParty, P., Soliman, L., Nagy, A., Farkas, Á., & Ambrus, R. (2025). Optimization, In Vitro, and In Silico Characterization of Theophylline Inhalable Powder Using Raffinose-Amino Acid Combination as Fine Co-Spray-Dried Carriers. Pharmaceutics, 17(4), 466. https://doi.org/10.3390/pharmaceutics17040466







