Future Perspective for ALK-Positive Anaplastic Large Cell Lymphoma with Initial Central Nervous System (CNS) Involvement: Could Next-Generation ALK Inhibitors Replace Brain Radiotherapy for the Prevention of Further CNS Relapse?
Abstract
:1. Introduction
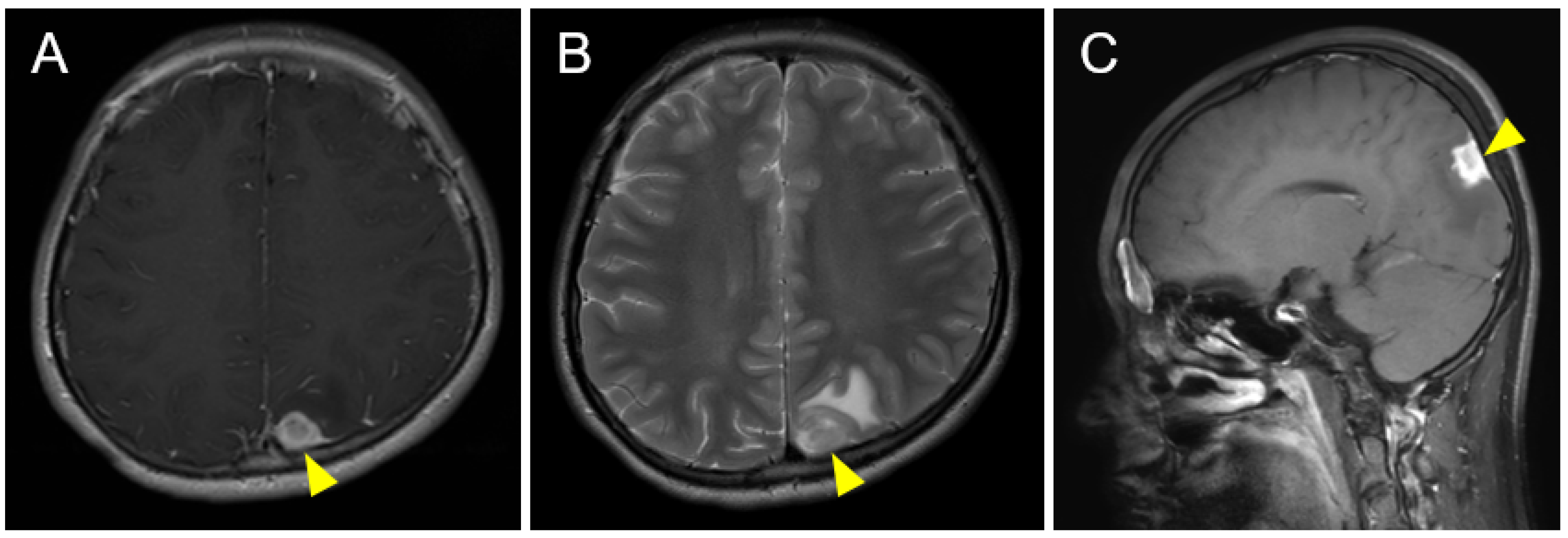
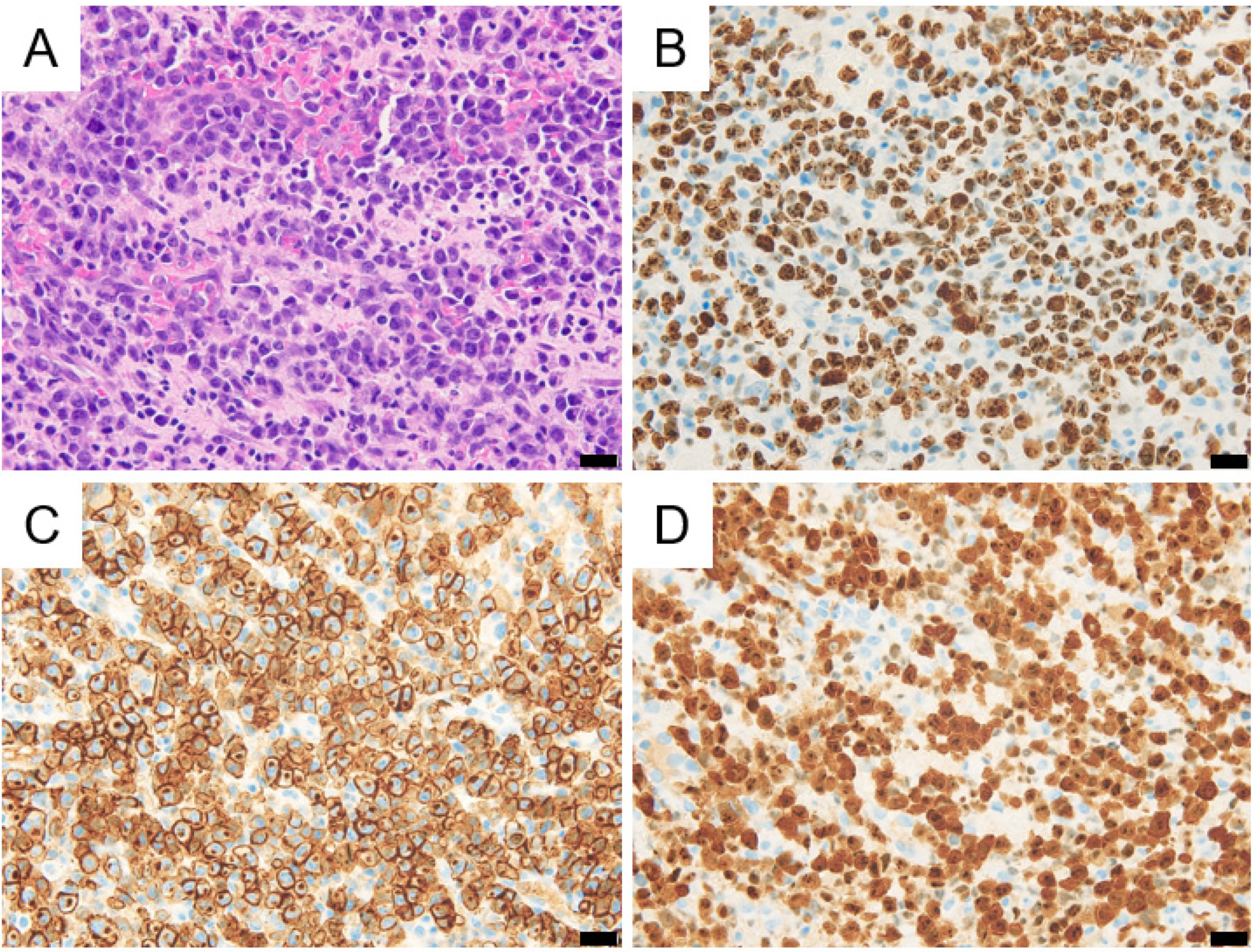
2. Case Description
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alaggio, R.; Amador, C.; Anagnostopoulos, I.; Attygalle, A.D.; Araujo, I.B.O.; Berti, E.; Bhagat, G.; Borges, A.M.; Boyer, D.; Calaminici, M.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia 2022, 36, 1720–1748. [Google Scholar] [CrossRef]
- Burkhardt, B.; Zimmermann, M.; Oschlies, I.; Niggli, F.; Mann, G.; Parwaresch, R.; Riehm, H.; Schrappe, M.; Reiter, A.; BFM Group. The impact of age and gender on biology, clinical features and treatment outcome of non-Hodgkin lymphoma in childhood and adolescence. Br. J. Haematol. 2005, 131, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Brugieres, L.; Le Deley, M.C.; Rosolen, A.; Williams, D.; Horibe, K.; Wrobel, G.; Mann, G.; Zsiros, J.; Uyttebroeck, A.; Marky, I.; et al. Impact of the methotrexate administration dose on the need for intrathecal treatment in children and adolescents with anaplastic large-cell lymphoma: Results of a randomized trial of the EICNHL Group. J. Clin. Oncol. 2009, 27, 897–903. [Google Scholar] [CrossRef]
- Mussolin, L.; Le Deley, M.C.; Carraro, E.; Damm-Welk, C.; Attarbaschi, A.; Williams, D.; Burke, A.; Horibe, K.; Nakazawa, A.; Wrobel, G.; et al. Prognostic Factors in Childhood Anaplastic Large Cell Lymphoma: Long Term Results of the International ALCL99 Trial. Cancers 2020, 12, 2747. [Google Scholar] [CrossRef]
- Le Deley, M.C.; Rosolen, A.; Williams, D.M.; Horibe, K.; Wrobel, G.; Attarbaschi, A.; Zsiros, J.; Uyttebroeck, A.; Marky, I.M.; Lamant, L.; et al. Vinblastine in children and adolescents with high-risk anaplastic large-cell lymphoma: Results of the randomized ALCL99-vinblastine trial. J. Clin. Oncol. 2010, 28, 3987–3993. [Google Scholar] [CrossRef]
- Williams, D.; Mori, T.; Reiter, A.; Woessman, W.; Rosolen, A.; Wrobel, G.; Zsiros, J.; Uyttebroeck, A.; Marky, I.; Le Deley, M.C.; et al. Central nervous system involvement in anaplastic large cell lymphoma in childhood: Results from a multicentre European and Japanese study. Pediatr. Blood Cancer 2013, 60, E118–E121. [Google Scholar] [CrossRef]
- Reiter, A.; Schrappe, M.; Tiemann, M.; Parwaresch, R.; Zimmermann, M.; Yakisan, E.; Dopfer, R.; Bucsky, P.; Mann, G.; Gadner, H.; et al. Successful treatment strategy for Ki-1 anaplastic large-cell lymphoma of childhood: A prospective analysis of 62 patients enrolled in three consecutive Berlin-Frankfurt-Munster group studies. J. Clin. Oncol. 1994, 12, 899–908. [Google Scholar] [CrossRef] [PubMed]
- Seidemann, K.; Tiemann, M.; Schrappe, M.; Yakisan, E.; Simonitsch, I.; Janka-Schaub, G.; Dorffel, W.; Zimmermann, M.; Mann, G.; Gadner, H.; et al. Short-pulse B-non-Hodgkin lymphoma-type chemotherapy is efficacious treatment for pediatric anaplastic large cell lymphoma: A report of the Berlin-Frankfurt-Munster Group Trial NHL-BFM 90. Blood 2001, 97, 3699–3706. [Google Scholar] [CrossRef]
- Mori, T.; Kiyokawa, N.; Shimada, H.; Miyauchi, J.; Fujimoto, J. Anaplastic large cell lymphoma in Japanese children: Retrospective analysis of 34 patients diagnosed at the National Research Institute for Child Health and Development. Br. J. Haematol. 2003, 121, 94–96. [Google Scholar] [CrossRef]
- Wells, E.M.; Ullrich, N.J.; Seidel, K.; Leisenring, W.; Sklar, C.A.; Armstrong, G.T.; Diller, L.; King, A.; Krull, K.R.; Neglia, J.P.; et al. Longitudinal assessment of late-onset neurologic conditions in survivors of childhood central nervous system tumors: A Childhood Cancer Survivor Study report. Neuro Oncol. 2018, 20, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Goldsby, R.E.; Liu, Q.; Nathan, P.C.; Bowers, D.C.; Yeaton-Massey, A.; Raber, S.H.; Hill, D.; Armstrong, G.T.; Yasui, Y.; Zeltzer, L.; et al. Late-occurring neurologic sequelae in adult survivors of childhood acute lymphoblastic leukemia: A report from the Childhood Cancer Survivor Study. J. Clin. Oncol. 2010, 28, 324–331. [Google Scholar] [CrossRef]
- Nathan, P.C.; Patel, S.K.; Dilley, K.; Goldsby, R.; Harvey, J.; Jacobsen, C.; Kadan-Lottick, N.; McKinley, K.; Millham, A.K.; Moore, I.; et al. Guidelines for identification of, advocacy for, and intervention in neurocognitive problems in survivors of childhood cancer: A report from the Children’s Oncology Group. Arch. Pediatr. Adolesc. Med. 2007, 161, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.R.; Chien, P.N.; Nam, S.Y.; Heo, C.Y. Anaplastic Large Cell Lymphoma: Molecular Pathogenesis and Treatment. Cancers 2022, 14, 1650. [Google Scholar] [CrossRef] [PubMed]
- Camidge, D.R.; Dziadziuszko, R.; Peters, S.; Mok, T.; Noe, J.; Nowicka, M.; Gadgeel, S.M.; Cheema, P.; Pavlakis, N.; de Marinis, F.; et al. Updated Efficacy and Safety Data and Impact of the EML4-ALK Fusion Variant on the Efficacy of Alectinib in Untreated ALK-Positive Advanced Non-Small Cell Lung Cancer in the Global Phase III ALEX Study. J. Thorac. Oncol. 2019, 14, 1233–1243. [Google Scholar] [CrossRef]
- Nakagawa, K.; Hida, T.; Nokihara, H.; Morise, M.; Azuma, K.; Kim, Y.H.; Seto, T.; Takiguchi, Y.; Nishio, M.; Yoshioka, H.; et al. Final progression-free survival results from the J-ALEX study of alectinib versus crizotinib in ALK-positive non-small-cell lung cancer. Lung Cancer 2020, 139, 195–199. [Google Scholar] [CrossRef]
- Kassem, L.; Shohdy, K.S.; Lasheen, S.; Abdel-Rahman, O.; Ali, A.; Abdel-Malek, R.R. Safety issues with the ALK inhibitors in the treatment of NSCLC: A systematic review. Crit. Rev. Oncol. Hematol. 2019, 134, 56–64. [Google Scholar] [CrossRef]
- Hou, H.; Sun, D.; Liu, K.; Jiang, M.; Liu, D.; Zhu, J.; Zhou, N.; Cong, J.; Zhang, X. The safety and serious adverse events of approved ALK inhibitors in malignancies: A meta-analysis. Cancer Manag. Res. 2019, 11, 4109–4118. [Google Scholar] [CrossRef] [PubMed]
- Pearson, A.D.J.; Barry, E.; Mosse, Y.P.; Ligas, F.; Bird, N.; de Rojas, T.; Zimmerman, Z.F.; Wilner, K.; Woessmann, W.; Weiner, S.; et al. Second Paediatric Strategy Forum for anaplastic lymphoma kinase (ALK) inhibition in paediatric malignancies: ACCELERATE in collaboration with the European Medicines Agency with the participation of the Food and Drug Administration. Eur. J. Cancer 2021, 157, 198–213. [Google Scholar] [CrossRef] [PubMed]
- Fukano, R.; Mori, T.; Sekimizu, M.; Choi, I.; Kada, A.; Saito, A.M.; Asada, R.; Takeuchi, K.; Terauchi, T.; Tateishi, U.; et al. Alectinib for relapsed or refractory anaplastic lymphoma kinase-positive anaplastic large cell lymphoma: An open-label phase II trial. Cancer Sci. 2020, 111, 4540–4547. [Google Scholar] [CrossRef]
- Rigaud, C.; Abbou, S.; Ducassou, S.; Simonin, M.; Le Mouel, L.; Pereira, V.; Gourdon, S.; Lambilliotte, A.; Geoerger, B.; Minard-Colin, V.; et al. Profound and sustained response with next-generation ALK inhibitors in patients with relapsed or progressive ALK-positive anaplastic large cell lymphoma with central nervous system involvement. Haematologica 2022, 107, 2255–2260. [Google Scholar] [CrossRef]
- Del Baldo, G.; Abbas, R.; Woessmann, W.; Horibe, K.; Pillon, M.; Burke, A.; Beishuizen, A.; Rigaud, C.; Le Deley, M.C.; Lamant, L.; et al. Neuro-meningeal relapse in anaplastic large-cell lymphoma: Incidence, risk factors and prognosis—A report from the European intergroup for childhood non-Hodgkin lymphoma. Br. J. Haematol. 2021, 192, 1039–1048. [Google Scholar] [CrossRef]
- Woessmann, W.; Zimmermann, M.; Lenhard, M.; Burkhardt, B.; Rossig, C.; Kremens, B.; Lang, P.; Attarbaschi, A.; Mann, G.; Oschlies, I.; et al. Relapsed or refractory anaplastic large-cell lymphoma in children and adolescents after Berlin-Frankfurt-Muenster (BFM)-type first-line therapy: A BFM-group study. J. Clin. Oncol. 2011, 29, 3065–3071. [Google Scholar] [CrossRef]
- Iijima-Yamashita, Y.; Mori, T.; Nakazawa, A.; Fukano, R.; Takimoto, T.; Tsurusawa, M.; Kobayashi, R.; Horibe, K. Prognostic impact of minimal disseminated disease and immune response to NPM-ALK in Japanese children with ALK-positive anaplastic large cell lymphoma. Int. J. Hematol. 2018, 107, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Fukano, R.; Saito, A.; Takimoto, T.; Sekimizu, M.; Nakazawa, A.; Tsurusawa, M.; Kobayashi, R.; Horibe, K.; Japanese Pediatric Leukemia/Lymphoma Study Group. Analysis of Japanese Registration from the Randomized International Trial for ChildhoodAnaplastic Large Cell Lymphoma (ALCL99-R1). Rinsho Ketsueki 2014, 55, 526–533. [Google Scholar] [PubMed]
- Thorer, H.; Zimmermann, M.; Makarova, O.; Oschlies, I.; Klapper, W.; Lang, P.; von Stackelberg, A.; Fleischhack, G.; Worch, J.; Juergens, H.; et al. Primary central nervous system lymphoma in children and adolescents: Low relapse rate after treatment according to Non-Hodgkin-Lymphoma Berlin-Frankfurt-Münster protocols for systemic lymphoma. Haematologica 2014, 99, e238–e241. [Google Scholar] [CrossRef]
- Eyre, T.A.; Khan, D.; Hall, G.W.; Collins, G.P. Anaplastic lymphoma kinase-positive anaplastic large cell lymphoma: Current and future perspectives in adult and paediatric disease. Eur. J. Haematol. 2014, 93, 455–468. [Google Scholar] [CrossRef]
- Ruf, S.; Hebart, H.; Hjalgrim, L.L.; Kabickova, E.; Lang, P.; Steinbach, D.; Schwabe, G.C.; Woessmann, W. CNS progression during vinblastine or targeted therapies for high-risk relapsed ALK-positive anaplastic large cell lymphoma: A case series. Pediatr. Blood Cancer 2018, 65, e27003. [Google Scholar] [CrossRef]
- Morris, S.W.; Kirstein, M.N.; Valentine, M.B.; Dittmer, K.G.; Shapiro, D.N.; Saltman, D.L.; Look, A.T. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin’s lymphoma. Science 1994, 263, 1281–1284. [Google Scholar] [CrossRef]
- Perkins, S.L.; Pickering, D.; Lowe, E.J.; Zwick, D.; Abromowitch, M.; Davenport, G.; Cairo, M.S.; Sanger, W.G. Childhood anaplastic large cell lymphoma has a high incidence of ALK gene rearrangement as determined by immunohistochemical staining and fluorescent in situ hybridisation: A genetic and pathological correlation. Br. J. Haematol. 2005, 131, 624–627. [Google Scholar] [CrossRef]
- Mosse, Y.P.; Voss, S.D.; Lim, M.S.; Rolland, D.; Minard, C.G.; Fox, E.; Adamson, P.; Wilner, K.; Blaney, S.M.; Weigel, B.J. Targeting ALK With Crizotinib in Pediatric Anaplastic Large Cell Lymphoma and Inflammatory Myofibroblastic Tumor: A Children’s Oncology Group Study. J. Clin. Oncol. 2017, 35, 3215–3221. [Google Scholar] [CrossRef]
- Gadgeel, S.M.; Gandhi, L.; Riely, G.J.; Chiappori, A.A.; West, H.L.; Azada, M.C.; Morcos, P.N.; Lee, R.M.; Garcia, L.; Yu, L.; et al. Safety and activity of alectinib against systemic disease and brain metastases in patients with crizotinib-resistant ALK-rearranged non-small-cell lung cancer (AF-002JG): Results from the dose-finding portion of a phase 1/2 study. Lancet Oncol. 2014, 15, 1119–1128. [Google Scholar] [CrossRef]
- Costa, D.B.; Shaw, A.T.; Ou, S.H.; Solomon, B.J.; Riely, G.J.; Ahn, M.J.; Zhou, C.; Shreeve, S.M.; Selaru, P.; Polli, A.; et al. Clinical Experience With Crizotinib in Patients With Advanced ALK-Rearranged Non-Small-Cell Lung Cancer and Brain Metastases. J. Clin. Oncol. 2015, 33, 1881–1888. [Google Scholar] [CrossRef]
- Gadgeel, S.; Peters, S.; Mok, T.; Shaw, A.T.; Kim, D.W.; Ou, S.I.; Perol, M.; Wrona, A.; Novello, S.; Rosell, R.; et al. Alectinib versus crizotinib in treatment-naive anaplastic lymphoma kinase-positive (ALK+) non-small-cell lung cancer: CNS efficacy results from the ALEX study. Ann. Oncol. 2018, 29, 2214–2222. [Google Scholar] [CrossRef]
- Nishio, M.; Nakagawa, K.; Mitsudomi, T.; Yamamoto, N.; Tanaka, T.; Kuriki, H.; Zeaiter, A.; Tamura, T. Analysis of central nervous system efficacy in the J-ALEX study of alectinib versus crizotinib in ALK-positive non-small-cell lung cancer. Lung Cancer 2018, 121, 37–40. [Google Scholar] [CrossRef]
- Gambacorti-Passerini, C.; Mussolin, L.; Brugieres, L. Abrupt Relapse of ALK-Positive Lymphoma after Discontinuation of Crizotinib. N. Engl. J. Med. 2016, 374, 95–96. [Google Scholar] [CrossRef]
- Arosio, G.; Sharma, G.G.; Villa, M.; Mauri, M.; Crespiatico, I.; Fontana, D.; Manfroni, C.; Mastini, C.; Zappa, M.; Magistroni, V.; et al. Synergistic Drug Combinations Prevent Resistance in ALK+ Anaplastic Large Cell Lymphoma. Cancers 2021, 13, 4422. [Google Scholar] [CrossRef]
- Brugieres, L.; Afify, Z.; Lowe, E. ALK inhibitors for ALK-altered paediatric malignancies. Lancet Oncol. 2021, 22, 1646–1648. [Google Scholar] [CrossRef]
- Lowe, E.J.; Reilly, A.F.; Lim, M.S.; Gross, T.G.; Saguilig, L.; Barkauskas, D.A.; Wu, R.; Alexander, S.; Bollard, C.M. Crizotinib in Combination With Chemotherapy for Pediatric Patients With ALK+ Anaplastic Large-Cell Lymphoma: The Results of Children’s Oncology Group Trial ANHL12P1. J. Clin. Oncol. 2023, 41, 2043–2053. [Google Scholar] [CrossRef]
- He, Y.; Pei, K.; Zhang, H.; Wang, J.; Su, X.; Gan, W.; Wang, P. Observation of Alectinib- and Crizotinib-included chemotherapy in children with ALK-positive anaplastic large cell lymphoma: A single institutional experience. Cancer Med. 2023, 12, 7182–7188. [Google Scholar] [CrossRef]
- Yang, J.; Li, J.; Gu, W.Y.; Jin, L.; Duan, Y.L.; Huang, S.; Zhang, M.; Wang, X.S.; Liu, Y.; Zhou, C.J.; et al. Central nervous system relapse in a pediatric anaplastic large cell lymphoma patient with CLTC/ALK translocation treated with alectinib: A case report. World J. Clin. Cases 2020, 8, 1685–1692. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanaka, M.; Miura, H.; Ishimaru, S.; Furukawa, G.; Kawamura, Y.; Kozawa, K.; Yamada, S.; Ito, F.; Kudo, K.; Yoshikawa, T. Future Perspective for ALK-Positive Anaplastic Large Cell Lymphoma with Initial Central Nervous System (CNS) Involvement: Could Next-Generation ALK Inhibitors Replace Brain Radiotherapy for the Prevention of Further CNS Relapse? Pediatr. Rep. 2023, 15, 333-340. https://doi.org/10.3390/pediatric15020029
Tanaka M, Miura H, Ishimaru S, Furukawa G, Kawamura Y, Kozawa K, Yamada S, Ito F, Kudo K, Yoshikawa T. Future Perspective for ALK-Positive Anaplastic Large Cell Lymphoma with Initial Central Nervous System (CNS) Involvement: Could Next-Generation ALK Inhibitors Replace Brain Radiotherapy for the Prevention of Further CNS Relapse? Pediatric Reports. 2023; 15(2):333-340. https://doi.org/10.3390/pediatric15020029
Chicago/Turabian StyleTanaka, Makito, Hiroki Miura, Soichiro Ishimaru, Gen Furukawa, Yoshiki Kawamura, Kei Kozawa, Seiji Yamada, Fumitaka Ito, Kazuko Kudo, and Tetsushi Yoshikawa. 2023. "Future Perspective for ALK-Positive Anaplastic Large Cell Lymphoma with Initial Central Nervous System (CNS) Involvement: Could Next-Generation ALK Inhibitors Replace Brain Radiotherapy for the Prevention of Further CNS Relapse?" Pediatric Reports 15, no. 2: 333-340. https://doi.org/10.3390/pediatric15020029
APA StyleTanaka, M., Miura, H., Ishimaru, S., Furukawa, G., Kawamura, Y., Kozawa, K., Yamada, S., Ito, F., Kudo, K., & Yoshikawa, T. (2023). Future Perspective for ALK-Positive Anaplastic Large Cell Lymphoma with Initial Central Nervous System (CNS) Involvement: Could Next-Generation ALK Inhibitors Replace Brain Radiotherapy for the Prevention of Further CNS Relapse? Pediatric Reports, 15(2), 333-340. https://doi.org/10.3390/pediatric15020029





