Pharmaceutical Pollutants: Ecotoxicological Impacts and the Use of Agro-Industrial Waste for Their Removal from Aquatic Environments
Abstract
:1. Introduction
2. Methodological Approach
2.1. Information Search
2.2. Drug Mass Removal Rate
- DMRR = drug mass removal rate (mg/g·h);
- Ci = initial concentration of the pharmaceutical molecule (mg/L);
- Cf = final concentration of the pharmaceutical molecule (mg/L);
- CB = concentration of biosorbent material (g/L);
- RT = removal time (h).
3. Emerging Pollutants
4. Occurrence of Pharmaceuticals as Pollutants in the Environment
4.1. General Information about Pharmaceutical Molecules
4.2. Pharmaceutical Molecules as Contaminants
5. Ecotoxicological Effects of Pharmaceutical Pollutants in Aquatic Environments
5.1. Antibiotics
5.2. Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
5.3. Hormones
5.4. Psychotropic Drugs
6. Pharmaceutical Pollutant Degradation and Removal Strategies
- Advanced oxidation processes allow for pharmaceutical drugs to be degraded using strong oxidant agents such as ozone (O3), hydroxyl radicals (-OH−1), and hydrogen peroxide (H2O2); or a combination of compounds such as titanium oxide (TiO2) with UV light (photocatalysis); or iron salts and H2O2 (Fenton) among others [169,170,171,172].
- Ionic exchange resins: Pharmaceutical pollutants in water are removed through electrostatic interaction between charged functional groups in the resin and those in the drug structure. Mineral (clays/zeolite), organic (peat/lignite), and synthetic materials (acrylic acid polymers/Sephadex) are commonly used in these processes [79,173,174].
- Chemical precipitation: The addition of adequate chemicals/additives such as alum, marine salt, or metallic salts (e.g., FeCl3, AlCl3, MgCl2, CaCl2) to drug-polluted water generates insoluble chemical complexes/particles that precipitate as sludge, facilitating their removal from the water solution [175,176].
- Biosorption is an approach for pharmaceutical pollutant removal from water employing sorbents of biological origin (biomass); these include lignocellulosic materials, such as agro-industrial wastes, woody biomass, or vegetable peels, but also other biological origin materials, including algae, manure, mussel and crustacean shells, or bird feathers. In these materials, pollutants are passively bound through ionic, chemical, or physical mechanisms [191,192,193].
6.1. Pharmaceutical Removal from Water through Biosorption Using Agro-Industrial Wastes
| Pharmaceutical Drug | Chemical Structure | Ci (mg/L) | Agro-Industrial Waste | Agro-Industrial Waste Treatment | Biosorbent Concentration (g/L) | Time (h) | Removal Percentage | qmax (mg/g) Langmuir Model | Reference | DMRR (mg/g·h) |
|---|---|---|---|---|---|---|---|---|---|---|
| Sulfamethoxazole | 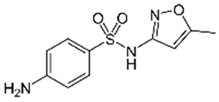 | 1 | Miscanthus x giganteus biochar | Air-dried, cut, pyrolyzed at 360 °C. | 2.0 | 4 | 32 | - | [211] | 0.3 |
| 3.8 | Rice husk | Rice husks were washed, dried at 60 °C for 48 h, grounded, sieved and pyrolyzed at 500 °C (RH-500) and 800 °C (RH-800) for 2 h. RH-800 was activated with NaOH, heated at 800 °C. | 0.2 | 0.25 | - | - | [212] | - | ||
| 10–60 | Sugarcane bagasse | Untreated sugarcane bagasse was used as an adsorbent, was washed with ultrapure water, dried at 60 °C for 8 h, ground, and triturated. | 0.02 | 0.083 | 51 | 1.43 | [213] | - | ||
| 20 | Wheat straw ashes | The acidification of ashes was performed using 2 M HCl, mixture at 70 °C, filtered, dried at 105 °C. | 1 | 0.5 | 22 | - | [214] | 8.8 | ||
| Sulfamethoxazole | 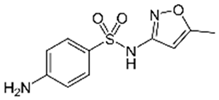 | 100 | Activated biochar of forage bermudagrass | Dried and sieved, pyrolyzed at 300 °C, activated with NaOH, washed with 3 M HCl and deionized water, dried at 105 °C. | 0.01 | 72 | 91.86 | 424.66 | [215] | 127.6 |
| 0.5–40 | Activated carbon of walnut shell | Pyrolyzed at 300 °C, impregnated with K2CO3, heated at 105 °C and 900 °C. | 0.01 | 48 | - | 93.5 | [216] | - | ||
| 250 | Biochar of malt rootlets | Dried at 50 °C, sieved, heated at 900 °C. | 0.09 | 1.5 | 94 | - | [207] | 1740.7 | ||
| 10 | Biochar of bagasse | Pyrolyzed at 300, 400, and 600 °C, washed with deionized water, dried, crushed, and sieved. | 0.1 | 24 | 41.55 | 54.38 | [217] | 1.8 | ||
| 30 | Almond shell activated biochar | Pyrolyzed at 700 °C and mixed with H2O2. | 0.005 | 3 | 96.88 | 344.8 | [206] | 1940.0 | ||
| 0.5–50 | Bamboo biochar | Cut into 0.6–2 mm size particles, pyrolyzed at 380 °C, cooled, grinded, washed, and dried. Added H3PO4 at 50 °C, heated to 600 °C, cooled, washed with distilled water, and dryed at 120 °C. | 0.1 | 24 | - | 34.01 | [218] | - | ||
| 0.00005 | Pelletized pine forestry waste biochar | Pyrolyzed at 850 °C, ground and sieved, dried at 105 °C. | 40 | 1 | 75 | - | [219] | 0.0000009 | ||
| 0–80 | Arundo donax stems biochar | Pyrolyzed at 300–600 °C, milled, washed with deionized water, and other portion was pulverized for demineralization with HCl and HCL-HF and heated to 750 °C. | 0.1 | 48 | - | 0.473–0.778 * | [220] | - | ||
| Ciprofloxacin |  | 50 | Olive stones | Olive stone waste was cleaned with distilled water, dried at 105 °C; was pulverized, washed, and dried at 105 °C; was carbonized at 450 °C; added KOH and activating at 550, 650, and 750 °C. | 1 | 1.25 | 99 | 294.98 | [221] | 39.6 |
| 25 | Activated biochar of barley bagasse | The barley bagasse was dried at 60–70 °C, crushed, sieved, and heated at 105 and 400 °C; activated biochar of barley bagasse with 2 M of K2CO3, dried and washed with distilled water. | 0.5 | 24 | 82 | 104.9 | [222] | 1.7 | ||
| 5.0 | Rice husk | Rice husks were washed, dried at 60 °C for 48 h, grounded, sieved and pyrolyzed 500 °C (RH-500) and 800 °C (RH-800) for 2 h. RH-800 was activated with NaOH, heated at 800 °C. | 0.2 | 0.25 | - | 584.4 | [212] | - | ||
| 10–60 | Sugarcane bagasse | Untreated sugarcane bagasse was used as an adsorbent, was washed with ultrapure water, dried at 60 °C for 8 h, ground, and triturated. | 5 | 0.083 | 99 | 2.61 | [213] | - | ||
| 250 | Dialium guineense seed waste (DGS) | Washed with distilled water, dried in open air, heated at 105 °C, dried, grinded, and sieved. | 0.1 | 2 | 42.9 | 9.17 | [209] | 536.0 | ||
| Dialium guineense seed waste sodium hydroxide modified form (NH-DGS) | Washed with distilled water, dried in open air, heated at 105 °C, dried, grinded, and sieved. Mixed with NaOH, filtered and washed with deionized water, dried at 80–120 °C. | 61.0 | 120.34 | 762.5 | ||||||
| 60 | Banyan aerial roots | Cut 1 cm, washed with deionized water, dried at 80 °C, mixed with 1 M C6H8O7, and heated at 150 °C. | 0.03 | 48 | 90.66 | 65.70 | [223] | 37.8 | ||
| 20 | Biochar of rice straw | Washed, dried at 80 °C, crushed, heated to 700 °C, washed with deionized water. | 1.6 | 24 | 55.0 | 48.80 | [224] | 0.3 | ||
| 100 | Carbon from date palm leaflets | Cut, carbonized with sulfuric acid, heated at 160 °C. | 0.4 | 48 | - | 133.3 | [225] | - | ||
| Tetracycline |  | 100 | Alfalfa biochar | Pyrolyzed at 500 °C, washed with deionized water and filtered (0.45 µm), and washed with 0.1 M HCl. | 0.01 | 48 | - | 372.31 | [226] | - |
| Bermudagrass biochar | - | 44.24 | - | |||||||
| 10 | Activated carbon from peach stones (AC-PS) | Washed with ultrapure water, dried at 110 °C, and chemically activated with H3PO4. | 0.8 | 3 | 96.1 | 845.9 | [227] | 4.0 | ||
| 100 | Activated carbon of hard Shell of apricot stone | Washed with distilled water, ground, added H3PO4, heated to 100 °C and after to 400 °C, washed with distilled water, dried at 80 °C. | 0.3 | 24 | 100 | 308.33 | [228] | 13.9 | ||
| 20 | Activated nanobiochar from vine wood | Washed, pyrolyzed at 600 °C, activated with NaOH 5% w/w and NaCl (3 M). | 0.4 | 8 | 88.17 | 1.98 | [201] | 5.5 | ||
| 100 | Spent black tea leaves (SBTL) | Steep under stirring with boiling water 100 °C, filtered, washed with distilled water, dried at 100 °C, ground. | 0.2 | 3 | 78 | - | [229] | 130.0 | ||
| Pomegranate peel (PP) | Ground and pulverized, steeped under stirring with boiling water 100 °C, filtered, washed with distilled water, dried at 100 °C, ground. | 0.3 | 0.5 | 90 | - | 600.0 | ||||
| 50 | Bamboo charcoal | Carbonized at 150–250 °C, 250–400 °C and 400–700 °C, ground and sieved, washed, dried at 105 °C. | 1 | 24 | 87.6 | 22.7 | [230] | 1.8 | ||
| Amoxicillin |  | 25 | Banana peel activated carbon | Activated carbon by H3PO4, carbonized at 350 °C, 450 and 500 °C. | 1.5 | 2 | 96.02 | 393.70 | [231] | 8.0 |
| 100 | Activated biochar from corn cob | Corn cobs were used for activated biochar with ZnCl2, dried at 120 °C for 12 h, pyrolyzed at 700 °C for 2 h. | 1 | 6 | 65.88 | 175.86 | [232] | 11.0 | ||
| 700 | Bertholletia excelsa capsules | Milled, mixed with ZnCl2, dried at 80 °C, pyrolyzed from 25 up to 600 or 700 °C, cooled, and refluxed with HCl. | 1.5 | 0.5 | 98.60 | 388.4 | [208] | 920.3 | ||
| 25 | Olive stone | Washed with tap water and impregnated with H3PO4 at 100 °C for 9 h, dried at 170 °C and 380 °C, washed with distilled water, and dyed at 110 °C. | 1.0 | 100 | 93 | 57.04 | [233] | 0.2 | ||
| 20 | Activated nanobiochar from vine wood | Washed, pyrolyzed at 600 °C, activated with NaOH 5% w/w and NaCl (3 M). | 0.4 | 8 | 60.23 | 2.69 | [201] | 3.8 | ||
| Norfloxacin |  | 15–150 | Activated carbons of red mombin seeds | The red mombin seeds, corn cob, coffee husk, internal and external parts of mango seeds and ice cream beans were washed with potable water and dried at 80 °C, were grounded and sieved, all raw materials were mixed with ZnCl2 and were pyrolyzed at 600 °C for 2 h. | 0.01 | 7 | - | 404 | [202] | - |
| Activated carbons of corn cob | - | 264 | - | |||||||
| Activated carbons of coffee husk | - | 235 | - | |||||||
| Activated carbons of internal parts of mango seeds | - | 221 | - | |||||||
| Activated carbons of external parts of mango seeds | - | 262 | - | |||||||
| Activated carbons of ice cream beans | - | 84 | - | |||||||
| 5 | Rice husk | Washed with distilled water, dried at 60 °C for 48 h, ground to powder and sieved. | 0.2 | 3 | 59.51 | - | [234] | 5.0 | ||
| Coffee husk | 70.83 | - | 5.8 | |||||||
| 10 | Biochar from potato stem | Washed, dried at 80 °C, chopped, crushed, sieved, pyrolyzed at 500 °C. | 0.1 | 36 | - | 5.24 | [235] | - | ||
| Ofloxacin |  | 25–250 | Activated carbons of red mombin seeds | The red mombin seeds, corn cob, coffee husk, internal and external parts of mango seeds and ice cream beans were washed with potable water and dried at 80 °C, were grounded and sieved, all raw materials were mixed with ZnCl2 and were pyrolyzed at 600 °C for 2 h. | 0.01 | 7 | - | 380 | [202] | - |
| Activated carbons of corn cob | - | 267 | - | |||||||
| Activated carbons of coffee husk | - | 202 | - | |||||||
| Activated carbons of internal parts of mango seeds | - | 176 | - | |||||||
| Activated carbons of external parts of mango seeds | - | 254 | - | |||||||
| Activated carbons of ice cream beans | - | 77 | - | |||||||
| 100 | Bamboo sawdust biochar | Pyrolyzed at 500 °C, washed with distilled water, dried at 105 °C. | 0.05 | 96 | - | 45.11 | [236] | - | ||
| 50 | Moringa oleifera pod husks (AMOP) | Air-dried, pulverized, sieved, activated with NH4Cl, filtered, washed with distilled water, and air-dried. | 2.5 | 4 | 90.98 | 3.597 | [237] | 4.5 | ||
| Biochar of Moringa oleifera pod husks (CMOP) | Air-dried, pulverized, sieved, activated with NH4Cl, filtered, washed with distilled water, air-dried, pyrolyzed at 350 °C, washed with distilled water, air-dried. | 99.84 | 5.051 | 5.0 | ||||||
| Chloramphenicol | 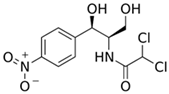 | 5–100 | Wheat straw | Pyrolyzed at 550 °C and 700 °C, respectively, crushed and sieved, washed with HCl and deionized water. | 0.05 | 24 | - | 11.3 | [238] | - |
| Softwood | - | 8.8 | - | |||||||
| 50 | Bamboo charcoal | Carbonized at 150–250 °C, 250–400 °C and 400–700 °C, ground and sieved, washed, dried at 105 °C. | 1.0 | 24 | 70.3 | 8.1 | [230] | 1.5 | ||
| Doxycycline | 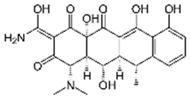 | 20 | Biochar of rice straw | Washed, dried at 80 °C, crushed, heated to 700 °C, washed with deionized water. | 1.6 | 24 | 90.0 | 170.36 | [224] | 0.5 |
| 100 | Spent black tea leaves (SBTL) | Steep under stirring with boiling water 100 °C, filtered, washed with distilled water, dried at 100 °C, ground. | 0.05 | 2.5 | 89 | - | [229] | 672.0 | ||
| Pomegranate peel (PP) | Ground and pulverized steep under stirring with boiling water 100 °C, filtered, washed with distilled water, dried at 100 °C, ground. | 0.15 | 1.5 | 83 | - | 368.9 | ||||
| Metronidazole |  | 100 | Cassava starch waste | Dried at 60 °C, activated with H3PO4, NaOH and combination of both, heated at 105 °C, washed with water, dried at 105 °C, pyrolyzed at 900 °C. | 0.1 | 24 | 55 | - | [239] | 22.9 |
| 0.5–40 | Activated carbon of walnut Shell | Pyrolyzed at 300 °C, impregnated with K2CO3, heated at 105 °C and 900 °C. | 0.01 | 48 | - | 93.5 | [216] | - | ||
| Azithromycin | 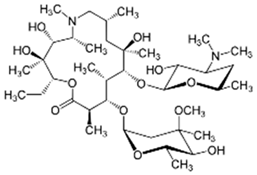 | 200 | Biochar from Terminalla chebula (CBC) | Cleaned, dried, and peeled the bagasse was shredded, washed, dried 72 h at 70 °C, heated for pyrolysis at 500 °C for 1 h; after cooling, washed and dried at 70 °C for 12 h. | 200 | 2 | 57.17 | 21.36 | [240] | 0.29 |
| Biochar from sugarcane bagasse (BBC) | 60.03 | 17.95 | 0.30 | |||||||
| Cefazolin | 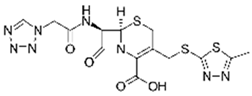 | 30 | Alperujo | Alperujo dried were covered with distilled water, heated at 160–220 °C, dried at 50 °C, and sieved. | 0.125 | 24 | - | 1572.73 | [241] | - |
| Sulfadiazine |  | 20 | Activated carbons from olive pomace | Washed with hot distilled water, dried at 103 °C, ground and sieved, pyrolyzed at 450 °C, impregnated with KOH, stirred at 60 °C, dried at 103 °C. The mixture was activated at 560, 700, and 840 °C, cooled, washed with distilled water, and dried at 103 °C. | 0.8 | 2 | 99 | 66.2 | [242] | 12.3 |
| Sulfamethazine | 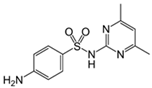 | 0.5–50 | Bamboo biochar | Cut into 0.6–2 mm size particles, pyrolyzed at 380 °C, cooled, grinded, washed, and dried. Adding H3PO4 at heated 50 °C and heated at 600 °C, cooled, washed with distilled water and drying at 120 °C. | 0.1 | 24 | - | 40.11 | [218] | - |
| Enrofloxacin | 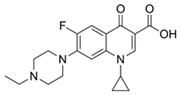 | 100 | Bamboo sawdust biochar | Pyrolyzed at 500 °C, washed with distilled water, dried at 105 °C. | 0.05 | 96 | - | 45.88 | [236] | - |
| Trimethoprim |  | 29 | Charcoal from feathers | Washed, drying at 80 °C, crushed, heated at 600 °C, dried 105 °C, sieved to 100–160 mesh. | 0.2 | 60 | 90 | 164 | [243] | 2.2 |
| Cephalexin | 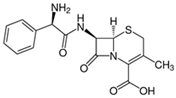 | 20 | Activated nanobiochar from vine wood | Washed, pyrolyzed at 600 °C and activated with NaOH 5% w/w and NaCl (3 M). | 0.4 | 8 | 76.02 | 7.08 | [201] | 4.8 |
| Penicillin G | 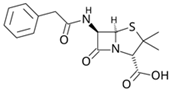 | 20 | Activated nanobiochar from vine wood | Washed, pyrolyzed at 600 °C and activated with NaOH 5% w/w and NaCl (3 M). | 0.4 | 8 | 73.94 | 8.41 | [201] | 4.6 |
| Pharmaceutical Drug | Chemical Structure | Ci (mg/L) | Agro-Industrial Waste | Agro-Industrial Waste Treatment | Biosorbent Concentration (g/L) | Time (h) | Removal Percentage | qmax (mg/g) Langmuir Model | Reference | DMRR (mg/g·h) |
|---|---|---|---|---|---|---|---|---|---|---|
| Diclofenac | 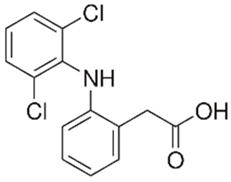 | 50 | Olive stones | Olive stone waste was cleaned with distilled water; dried at 105 °C; pulverized, washed, and dried at 105 °C; carbonized at 450 °C; and KOH was added and activated at 550, 650, and 750 °C. | 1 | 1.25 | 99 | 256.41 | [221] | 39.6 |
| 25 | Chestnut shells biochar | The chestnut shells were dried at 60 °C for 8 h and ground; 50 g of water chestnut shells were soaked in FeSO4.7H2O, pyrolyzed at 600 °C, sieved, and dried at 80 °C. | 0.5 | 1 | 98 | 75.9 | [248] | 49.0 | ||
| 4.4 | Rice husk | Rice husks were washed, dried at 60 °C for 48 h, grounded, sieved, and pyrolyzed 500 °C (RH-500) and 800 °C (RH-800) for 2 h. RH-800 was activated with NaOH, heated at 800 °C. | 0.2 | 0.25 | - | - | [212] | - | ||
| 100 | Rice husk ash | Rice husk ashes were heated at 105 °C for 1.5 h and sieved. | 1 | 4 | - | 2.3 | [249] | - | ||
| 10–60 | Sugarcane bagasse | Untreated sugarcane bagasse was used as an adsorbent, washed with ultrapure water, dried at 60 °C for 8 h, ground, and triturated. | 0.017 | 0.083 | 77 | 1.81 | [213] | - | ||
| 5–100 | Wheat straw | Pyrolyzed at 550 °C and 700 °C, respectively, crushed and sieved, washed with HCl and deionized water. | 0.05 | 24 | - | 6.7 | [238] | - | ||
| Softwood | - | 5.5 | - | |||||||
| 12.5 | Fique Bagasse | Dried at 100 °C, sieved, and biochar produced through pyrolyzed at 650, 750, and 850 °C. | 0.05 | 24 | 56 | 5.4 | [250] | 5.8 | ||
| 20 | Wheat straw ashes | The acidification of ashes was carried out using 2 M HCl, mixture at 70 °C, filtered, dried at 105 °C. | 1 | 0.5 | 0 | - | [214] | 0.0 | ||
| 0.5 | Pinewood microbiochar | Pyrolyzed at 525 °C, ground and sieved. | 2 | 48 | 70 | 1.106 ** | [251] | 0.004 | ||
| 20–350 | Moringa oleifera seed husks | Husks were peeled and ground, washed, dried at 105 °C, mixed with methyl alcohol, washed with purified water and stirred in HNO3, washed with purified water and dried at 105 °C, dried at 300 °C, washed with purified water, filtered, and dried at 105 °C. | 0.025 | 24 | - | 72.77 | [252] | - | ||
| 0.5 | Pinewood microbiochar | Pyrolyzed at 525 °C, ground, and sieved. | 0.1 | 4.5 | 68 | 526.3 | [253] | 0.8 | ||
| 10 | Activated onion skin | Washed, dried and ground (0.25 mm), pretreated with H2SO4, and dried at 50 °C. | 0.05 | 3.6 | 65.99 | 134.0035 * | [254] | 36.7 | ||
| 30 | Activated carbon from cocoa pod husks | Sun-dried cocoa pods were pulverized, the powder was sieved, soaked for 24 h with H2SO4, washed with deionized water, and dried at 120 °C. | 0.25 | 0.25 | 93.6 | 0.47 | [247] | 449.3 | ||
| 0.1 | Cellulose fiber (CF) decorated with polypyrrole (PPy) | CF mixed with FeCl3.6H2O, filtered, washed with water/methanol (1:1, v/v), dried at 60 °C. | 0.025 | 0.25 | 93 | 210.07 | [255] | 14.9 | ||
| 50 | Activated carbon from potato peel waste | Dried at 60 °C, milled and sieved, carbon activation was carried out with K2CO3, dried at 100 °C, heated at 700 °C, cooled, washed with distilled water, and dried at 100 °C. | 0.01 | 24 | 70 | 68.5 | [256] | 145.8 | ||
| 14.80 | Activated carbon of olive waste cake | Impregnated with H3PO4, pyrolyzed to 450 °C, cooled, washed with hot distilled water, dried at 105 °C, and ground. | 0.3 | 26 | - | 56.2 | [244] | - | ||
| Acetaminophen | 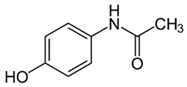 | 25–100 | Tingui shells | Tingui shells underwent thermal decomposition at 550 °C for 2 h and activation with carbon dioxide, and distilled water was added; it was kept at 200 °C for 24 h and activated with carbon dioxide; called HT-CO2. | 20 | 24 | - | 383.2 | [257] | - |
| 50 | Activated carbon from artichoke leaves | Pomegranate peels and artichoke leaves were washed with deionized water and dried for 36 h at 90 °C, crushed and sieved, and dried at 115 °C for 24 h. The pyrolysis was carried out at 450 °C and peels were mixed with 1 M HCl at 80 °C for 1.5 h. | 0.5 | 1 | 98.1 | 154.9 | [258] | 49.1 | ||
| Activated carbon from pomegranate peels | - | 258.9 | - | |||||||
| 2.3 | Rice husk | Rice husks were washed, dried at 60 °C for 48 h, grounded, sieved, and pyrolyzed at 500 °C (RH-500) and 800 °C (RH-800) for 2 h. RH-800 was activated with NaOH, heated at 800 °C. | 0.2 | 0.25 | - | 209.6 | [212] | - | ||
| Acetaminophen | 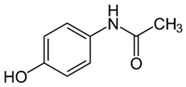 | 12.5–200 | Purple basil (Ocimum basilicum L.) waste | The plant was collected, washed, dried for 5 days, pulverized, mixed with distilled water, heated at 85 °C for 60 min, cooled, and filtered. | 0.5 | 1–800 | - | 0.023 | [259] | - |
| 100 | Activated biochar from corn cob | Corn cobs were used for activated biochar with ZnCl2, dried at 120 °C for 12 h, and pyrolyzed at 700 °C for 2 h. | 1 | 6 | 69.37 | 332.08 | [232] | 11.6 | ||
| 100 | Dende coconuts (DND) | The carbons derived from babassu coconut biomass and dende coconut were sieved, washed with water, and dried at 60 °C for 24 h. | 10 | 300 | - | 149 | [260] | - | ||
| babassu coconuts (BBS) | - | 128 | - | |||||||
| 100 | Gundelia tournefortii seeds | Gundelia tournefortii seeds were washed with distilled water and dried. After, the seed waste was soaked for 24 h in H3PO4, washed with distilled water, dried at 105 °C for 24 h, and then carbonized for 1 h at 700 °C, cooled, washed, and dried at 105 °C. | 0.25 | 0.5 | 98.31 | 14.34 | [246] | 786.5 | ||
| 150 | Mango seeds | Mango seeds were cut, rinsed in distilled water, drained, and immersed in HCl for 24 h. After, mango seeds were washed with distilled water and dried at 105 °C; crushed, sieved, and treated with phosphoric acid (H3PO4) for 24 h; dried at 105 °C for 24 h; and carbonized at 400 °C for 1 h. Rinsed with NaOH and dried at 105 °C. | 1.95 | 1.1 | 94.01 | 7.23 | [261] | 65.7 | ||
| 45 | Activated carbon from residual pods of Erythrina speciosa | Washed with tap water, broken, dried at 50 °C, grounded, mixed with ZnCl2, dried, heated at 10 °C, pyrolyzed at 800 °C, washed with HCl and distilled water, dried at 50 °C. | 1.2 | 2 | 86.49 | 60.83 | [262] | 16.2 | ||
| 450 | Activated carbon of Jatoba bark | Washed with distilled water, dried at 70 °C, ground, activated with KOH, heated at 500 °C. | 0.025 | 4 | - | 356.25 | [263] | - | ||
| 100 | Activated carbon from Quercus brantii (Oak) | Pyrolysis at 600 °C and activation with NaOH, KOH, NH4Cl, ZnCl2, and H3PO4. | 10 | 2.5 | 89.55 | 45.45 | [264] | 3.6 | ||
| 120 | Activated carbon of banana leaves | Cleaned with tap water, washed, dried, crushed, and immersed in H2SO4; carbonized at 150 °C, cooled, washed with NaOH and distilled water, and dried at 60 °C. | 0.01 | 24 | 83.8 | 142.2 | [265] | 419.2 | ||
| 0.007 | Activated carbon of olive stones | Grinded, washed with distilled water, dried at 100 °C, impregnated with H3PO4, heated at 500 °C, washed with distilled water at 60 °C, heated at 900 °C, grinded, and sieved. | 0.01 | 240 | - | 108.3 | [266] | - | ||
| Ibuprofen | 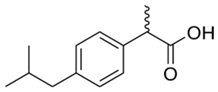 | 100 | Rice husk ash | Rice husk ashes were heated at 105 °C for 1.5 h and sieved. | 1 | 4 | - | 2.3 | [249] | - |
| 10–60 | Sugarcane bagasse | Untreated sugarcane bagasse was used as an adsorbent; washed with ultrapure water, dried at 60 °C for 8 h, ground, and triturated. | 0.02 | 0.083 | 65 | 1.62 | [213] | - | ||
| 10 | Olive pomace | The olive pomace was dried at 105 °C for 24 h and sieved, activated with zinc chloride and calcium hydroxide (1:0.8:0.2), and pyrolyzed at 550 °C for 0.5 h; after, the treated material underwent acid leaching with HCl 6 mol L−1 and was washed with water, dried at 105 °C for 24 h. | 0.5 | 0.8 | 95.28 | 360.607 | [267] | 2.8 | ||
| 75 | Activated carbon from rose geranium (Pelargonium graveolens L.) leaves | The rose geranium (Pelargonium graveolens L.) leaves were pulverized, pyrolyzed at 600 °C for 2 h; the activation of carbon leaves was with orthophosphoric acid (H3PO4); after, the mixture was rinsed with ultrapure water several times and dried at 70 °C. | 0.5 | 1.5 | 83.12 | 113.76 | [268] | 83.1 | ||
| 50 | Carbon activated of bean husks | Washed with distilled water, sun-dried, pulverized, sieved H3PO4 added, heated at 105 °C, cooled and washed with deionized water, and dried at 105 °C. | 0.1 | 1.16 | 78.17 | 50.00 | [269] | 336.9 | ||
| 15 | Wood apple biochar (WAB) | Pyrolyzed at 650 °C, heated to 65 °C, and cooled. | 0.33 | 24 | 90 | 5 | [270] | 1.7 | ||
| 30 | Wood apple steam activated biochar (WASAB) | 1 | 95 | 12.658 | 1.2 | |||||
| 100 | Activated carbon from Quercus brantii (Oak) | Pyrolysis at 600 °C and activation with NaOH, KOH, NH4Cl, ZnCl2, and H3PO4. | 10 | 2 | 100 | 96.15 | [264] | 5.0 | ||
| 10 | Activated onion skin | Washed, dried and ground (0.25 mm), pretreated with H2SO4, and dried at 50 °C. | 0.05 | 3.6 | 81.90 | 147.058 | [254] | 45.6 | ||
| 10 | Activated carbon from rice husk (AC-RH) | Washed with ultrapure water, dried at 110 °C, and chemically activated with H3PO4. | 0.8 | 3 | 97.2 | 239.8 | [227] | 4.0 | ||
| 20 | Activated biochar from mung bean husk | Washed with distilled water, dried, pyrolyzed at 550 °C, and heated up to 650 °C; steam was passed for activate biochar; was grinded and sieved. | 0.1 | 2 | 99.16 | 62.5 | [271] | 99.2 | ||
| 100 | Pinewood biochar | Pyrolyzed at 425 °C, washed with distilled water, sieved, heated at 110 °C. | 4 | 16 | 72.0 | 10.74 | [272] | 1.2 | ||
| 10.04 | Activated carbon of olive waste cake | Impregnated with H3PO4, pyrolyzed to 450 °C, cooled, washed with hot distilled water, dried at 105 °C, and ground. | 0.3 | 26 | 70.07 | 12.6 | [244] | 0.9 | ||
| Dipyrone | 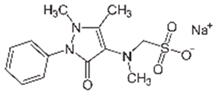 | 100 | Yeast waste (YW) | The wastes YW, CP, and CG were pyrolyzed at 800 °C for 2 h. For functionalization, natural deep eutectic solvent (NADES) was added to the biochar, also mixing citric acid, sucrose, and water (1:1:10); was stirred at 220 rpm for 2 h at 50 °C. The suspensions were ultrasonicated at 65 °C for 2 h and dried at 65 °C. | 0.01 | 0.5 | 31 | 2.71 | [245] | 6200 |
| Cork Powder (CP) | 0.5 | 52.10 | 14.66 | 10,420 | ||||||
| Old coffee grounds (CG) | 0.5 | 47 | 40.78 | 9400 | ||||||
| Ketoprofen | 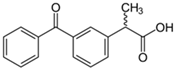 | 19.28 | Activated carbon of olive waste cake | Impregnated with H3PO4, pyrolyzed to 450 °C, cooled, washed with hot distilled water, dried at 105 °C, and ground. | 0.3 | 26 | 88.40 | 24.7 | [244] | 2.2 |
| Naproxen | 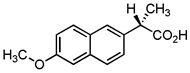 | 19.78 | Activated carbon of olive waste cake | Impregnated with H3PO4, pyrolyzed to 450 °C, cooled, washed with hot distilled water, dried at 105 °C, and ground. | 0.3 | 26 | 90.45 | 39.5 | [244] | 2.3 |
| Pharmaceutical Drug | Chemical Structure | Ci (mg/L) | Agro-Industrial Waste | Agro-Industrial Waste Treatment | Biosorbent Concentration (g/L) | Time (h) | Removal Percentage | qmax (mg/g) Langmuir Model | Reference | DMRR (mg/g·h) |
|---|---|---|---|---|---|---|---|---|---|---|
| Neurological/Psychoactive | ||||||||||
| Carbamazepine 1 | 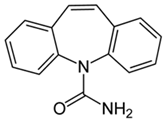 | 25 | Banana peel activated carbon | Activated carbon by H3PO4, carbonized at 350 °C, 450 °C, and 500 °C. | 1.5 | 2 | 90.62 | 338.98 | [231] | 7.6 |
| 5–100 | Wheat straw | Pyrolyzed at 550 °C and 700 °C, respectively, crushed and sieved, washed with HCl and deionized water. | 0.05 | 24 | - | 15.9 | [238] | - | ||
| Softwood | - | 20.5 | - | |||||||
| 4.7 | Biochar of paper mill sludge | Pyrolyzed at 800 °C, washed with HCl and distilled water, and dried at 105 °C. | 0.15 | 0.25 | - | 17.48 | [273] | - | ||
| 100 | Activated biochar from grapefruit peel | Pyrolyzed at 400 °C, mixed with KOH. | 0.01 | 24 | 58.6 | 286.5 | [274] | 244.2 | ||
| 0.005 | Pinewood nanobiochar | Pyrolyzed at 525 °C; nanobiochar with an average size of 60 ± 20 nm. | 0.005 | 3 | 95 | 0.074 | [275] | 0.3 | ||
| 100 | Activated carbons from peach stones | Crushed and sieved, added H3PO4, calcined at 435 °C, washed with ultrapure water, and dried at 110 °C. | 0.12 | 4 | - | 335 | [276] | - | ||
| 5 | Rice straw | Dried, cut, pulverized, sieved, and dried at 60 °C. | 60 | 2 | 75.3 | 40.0 | [277] | 1.2 | ||
| Fluoxetine 2 | 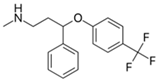 | 30 | Alperujo | Dried alperujo was covered with distilled water, heated at 160–220 °C, dried at 50 °C, and sieved. | 0.125 | 24 | - | 33.24 | [241] | - |
| Paroxetine 2 | 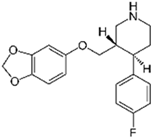 | 6.6 | Biochar of paper mill sludge | Pyrolyzed at 800 °C, washed with HCl and distilled water, and dried at 105 °C. | 0.15 | 0.25 | - | 21.08 | [273] | - |
| Sertraline 2 | 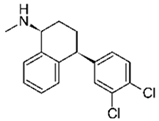 | 20 | Mango stones | Stones were washed with deionized water and dried at 105 °C for 24 h, ground, sieved, and FeSO4.7H2O was added; the biosorbent was filtered, washed, and dried at 105 °C. | 0.01 | 24 | 76.97 | 64.79 | [278] | 64.1 |
| Diazepam 3 | 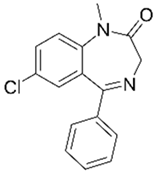 | 0.00034 | Bagasse | Biochar of bagasse was pelletized, and two natural draft gasifier stoves (N1 and N2) were used. | 0.05 | 24 | 78 | - | [279] | 0.00022 |
| Oxazepam 3 | 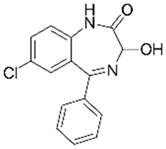 | 5.7 | Biochar of paper mill sludge | Pyrolyzed at 800 °C, washed with HCl and distilled water, and dried at 105 °C. | 0.15 | 0.25 | - | 20.07 | [273] | - |
| Pramipexole 4 | 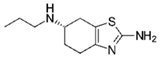 | 50 | Activated carbon from potato peel | Washed with distilled water, dried 120 °C, heated to 200 °C, mixed with KOH, filtered, dried at 100 °C, and activated at 400, 600, and 800 °C. | 1 | 24 | 61 | 98 | [280] | 1.3 |
| Antimicrobials/Antiparasitics | ||||||||||
| Triclosan | 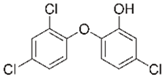 | 3–60 | Wheat straw | Pyrolyzed at 550 °C and 700 °C, respectively, crushed and sieved, washed with HCl and deionized water. | 0.05 | 24 | - | 20.3 | [238] | - |
| Softwood | - | 30.2 | - | |||||||
| 20 | Wheat straw ashes | The acidification of ashes was carried out using 2 M HCl, mixture at 70 °C, filtered, dried at 105 °C. | 1 | 0.5 | 30 | - | [214] | 12.2 | ||
| Chloroquine | 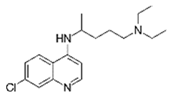 | 20 | Mango stones | Stones were washed with deionized water and dried at 105 °C for 24 h, ground, sieved, and added with FeSO4.7H2O; the biosorbent was filtered, washed, and dried at 105 °C. | 0.01 | 24 | 95.95 | 49.92 | [278] | 80.0 |
| 100 | Sugarcane bagasse | Sugarcane bagasse was dried 24 h at 100 °C, and then was milled, sieved, and stored in a freezer at −2 °C until being modified as hydrochar. | 0.02 | 1 | 63.69 | 73.45 | [281] | 3184.5 | ||
| Albendazole |  | 75 | Activated carbons of acorns of cork | Pyrolyzed at 500 °C, impregnated with H3PO4. | 2.4 | 1 | 99.97 | 137.2 | [282] | 31.2 |
| Sulfapyridine | 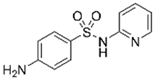 | 10 | Biochar of bagasse | Pyrolyzed at 300, 400, and 600 °C; washed with deionized water; dried; crushed; and sieved. | 0.1 | 24 | 69.64 | 8.60 | [217] | 2.9 |
| Sulfathiazole | 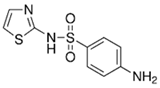 | 0.5–50 | Bamboo biochar | Cut into 0.6–2 mm size particles, pyrolyzed at 380 °C, cooled, ground, washed, and dried. Adding with H3PO4, heated 50 °C and heated at 600 °C, cooled, washed with distilled water, and dried at 120 °C. | 0.1 | 24 | - | 25.11 | [218] | - |
| Hormones | ||||||||||
| 17α-ethinyl estradiol | 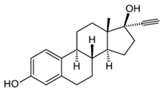 | 0.2632 | Raw peanut shells (Arachis hypogaea) | Raw peanut shells underwent washing, drying, grinding, sieving, and chemical treatments. | 2 | 24 | 90 | 0.0173 | [283] | 0.0049 |
| 20 | Wheat straw ashes | The acidification of ashes was accomplished using 2 M HCl, mixture at 70 °C, filtering, drying at 105 °C. | 1 | 0.5 | 15 | - | [214] | 6.0 | ||
| 17β-estradiol |  | 5 | Rice husk | The biochar of rice husks was obtained by pyrolysis at 600 °C. | 0.5 | 1 | 60.1 | - | [284] | 6.0 |
| 11.3 | Rice husk | Washed with ultrapure water, agitated to 200 rpm for 24 h, and filtered in cellulose ester membranes (0.45 µm). | 12 | 1 | 94.9 | 1.649 | [285] | 0.4 | ||
| Estriol | 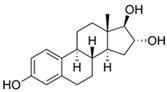 | 8.0 | Rice husk | Washed with ultrapure water, agitated to 200 rpm for 24 h, and filtered in cellulose ester membranes (0.45 µm). | 12 | 1.5 | 82.5 | 0.979 | [285] | 0.3 |
| Estrone | 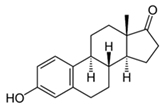 | 5 | Rice husk | The biochar of rice husks was obtained by pyrolysis at 600 °C. | 0.5 | 1 | 44.6 | - | [284] | 4.5 |
| 10.5 | Rice husk | Washed with ultrapure water, agitated to 200 rpm for 24 h, and filtered in cellulose ester membranes (0.45 µm). | 4 | 0.5 | 86.3 | 2.698 | [285] | 2.3 | ||
| Dexamethasone 5 |  | 4 | Activated biochar of Syzygium cumini leaves | Washed with potable water, dried at 60 °C, ground, sieved, mixed with ZnCl2, and pyrolyzed at 600 °C. | 5 | 3 | 53.02 | 0.673 | [286] | 0.1 |
| Other | ||||||||||
| Octocrylene 6 | 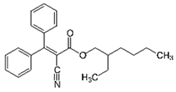 | 5 | Rice husk | The biochar of rice husks was obtained by pyrolysis at 600 °C. | 0.5 | 1 | 59.4 | - | [284] | 5.9 |
| Oxybenzone 6 | 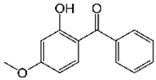 | 5 | Rice husk | The biochar of rice husks was obtained by pyrolysis at 600 °C. | 0.5 | 1 | 37.4 | - | [284] | 3.7 |
| Salicylic acid 7 | 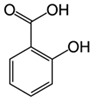 | 100 | Pinewood biochar | Pyrolyzed at 425 °C, washed with distilled water, sieved, heated at 110 °C. | 4 | 16 | 76.0 | 22.70 | [272] | 1.2 |
| Atenolol 8 | 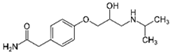 | 1 | Miscanthus x giganteus biochar | Air-dried, cut, pyrolyzed at 360 °C. | 0.5 | 4 | 69 | - | [211] | 0.3 |
| Carboplatin 9 |  | 100 | Rice husk ash | Rice husk ashes were heated at 105 °C for 1.5 h and sieved. | 1 | 1 | 36.52 | 9.9 | [249] | 36.5 |
| Clofibric acid 10 | 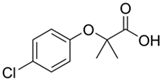 | 5 | Rice straw | Dried, cut, pulverized, sieved, and dried at 60 °C. | 30 | 2 | 42.5 | 126.3 | [277] | 0.04 |
| Dorzolamide 11 | 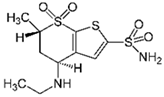 | 50 | Activated carbon from potato peel | Washed with distilled water, dried at 120 °C, heated at 200 °C, mixed with KOH, filtered, dried at 100 °C, and activated at 400, 600, and 800 °C. | 1 | 24 | 55 | 92 | [280] | 1.1 |
| Metformin 12 | 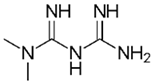 | 30 | Seed husks of Moringa oleifera Lam. | Moringa oleifera Lam. husks were peeled and washed with deionized water at 45 °C, transferred to nitric acid for 1 h, dried for 12 h, placed in an oven at 300 °C for 1 h, cooled, ground, and sieved. | 0.03 | 15 | 93.54 | 28.05 | [287] | 62.4 |
| Metoclopramide 13 | 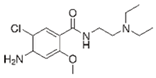 | 0.000299 | Bagasse | Biochar of bagasse was pelletized and used two natural draft gasifier stoves (N1 and N2). | 0.05 | 24 | 97 | - | [279] | 0.00024 |
| Ranitidine 14 | 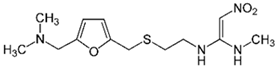 | 100 | Activated carbon of mung bean husk (MBH) | Washed with distilled water, dried, carbonized at 550 °C. | 0.75 | 1.5 | 99.16 | 28 | [288] | 88.1 |
6.2. Management and Treatment Alternatives for Biosorbent Materials Employed for Pharmaceutical Pollutants Removal from Water
7. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Pison, G.; Couppié, E.; Caporali, A. The population of the world. Popul. Soc. 2022, 603, 1–8. [Google Scholar] [CrossRef]
- UN, World Population Prospects—Population Division—United Nations [Internet]. Available online: https://population.un.org/wpp/ (accessed on 17 July 2024).
- Weber, H.; Sciubba, J.D. The effect of population growth on the environment: Evidence from European regions. Eur. J. Popul. 2019, 35, 379–402. [Google Scholar] [CrossRef] [PubMed]
- Gu, D.; Andreev, K.; Dupre, M.E. Major trends in population growth around the world. China CDC Wkly. 2021, 3, 604. [Google Scholar] [CrossRef] [PubMed]
- Sadigov, R. Rapid growth of the world population and its socioeconomic results. Sci. World J. 2022, 1, 8110229. [Google Scholar] [CrossRef] [PubMed]
- Jayachandran, S. How economic development influences the environment. Annu. Rev. Econom. 2022, 14, 229–252. [Google Scholar] [CrossRef]
- Parra-Henao, G.; Henao, E.; Escobar, J.P. Environmental health and infectious diseases: Challenges and perspectives in Latin America. Ther. Adv. Infect. Dis. 2023, 10, 1–4. [Google Scholar] [CrossRef]
- Aware, C.B.; Patil, D.N.; Suryawanshi, S.S.; Mali, P.R.; Rane, M.R.; Gurav, R.G.; Jadhav, J.P. Natural bioactive products as promising therapeutics: A review of natural product-based drug development. S. Afr. J. Bot. 2022, 151, 512–528. [Google Scholar] [CrossRef]
- Dzobo, K. The role of natural products as sources of therapeutic agents for innovative drug discovery. Compr. Pharmacol. 2022, 408–422. [Google Scholar] [CrossRef]
- Assidi, M.; Buhmeida, A.; Budowle, B. Medicine and health of 21st Century: Not just a high biotech-driven solution. NPJ Genom. Med. 2022, 7, 67. [Google Scholar] [CrossRef]
- Urquhart, L. Top companies and drugs by sales in 2021. Nat. Rev. Drug Discov. 2022, 21, 251. [Google Scholar] [CrossRef]
- Larsson, D.G.J. Pollution from drug manufacturing: Review and perspectives. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369, 20130571. [Google Scholar] [CrossRef] [PubMed]
- Lenzen, M.; Malik, A.; Li, M.; Fry, J.; Weisz, H.; Pichler, P.P.; Moreira-Chavez, L.S.; Capon, A.; Pencheon, D. The environmental footprint of health care: A global assessment. Lancet Planet. Health 2020, 4, e271–e279. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Mehmood, S.; Rasheed, T.; Iqbal, H.M. Antibiotics traces in the aquatic environment: Persistence and adverse environmental impact. Curr. Opin. Environ. Sci. Health 2020, 13, 68–74. [Google Scholar] [CrossRef]
- Samal, K.; Mahapatra, S.; Ali, M.H. Pharmaceutical wastewater as Emerging Contaminants (EC): Treatment technologies, impact on environment and human health. Energy Nexus 2022, 6, 100076. [Google Scholar] [CrossRef]
- Aus der Beek, T.; Weber, F.A.; Bergmann, A.; Hickmann, S.; Ebert, I.; Hein, A.; Küster, A. Pharmaceuticals in the environment—Global occurrences and perspectives. Environ. Toxicol. Chem. 2016, 35, 823–835. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, P.; Shukla, P.; Giri, B.S.; Chowdhary, P.; Chandra, R.; Gupta, P.; Pandey, A. Prevalence and hazardous impact of pharmaceutical and personal care products and antibiotics in environment: A review on emerging contaminants. Environ. Res. 2021, 194, 110664. [Google Scholar] [CrossRef] [PubMed]
- Boxall, A.B.; Wilkinson, J.L.; Bouzas-Monroy, A. Medicating nature: Are human-use pharmaceuticals poisoning the environment? One Earth 2022, 5, 1080–1084. [Google Scholar] [CrossRef]
- Wang, H.; Xi, H.; Xu, L.; Jin, M.; Zhao, W.; Liu, H. Ecotoxicological effects, environmental fate and risks of pharmaceutical and personal care products in the water environment: A review. Sci. Total Environ. 2021, 788, 147819. [Google Scholar] [CrossRef]
- Wilkinson, J.L.; Boxall, A.B.; Kolpin, D.W.; Leung, K.M.; Lai, R.W.; Galbán-Malagón, C.; Adell, A.D.; Mondon, J.; Metian, M.; Marchant, R.A.; et al. Pharmaceutical pollution of the world’s rivers. Proc. Natl. Acad. Sci. USA 2022, 119, e2113947119. [Google Scholar] [CrossRef]
- Hawash, H.B.; Moneer, A.A.; Galhoum, A.A.; Elgarahy, A.M.; Mohamed, W.A.; Samy, M.; El-Seedi, H.R.; Gaballah, M.S.; Mubarak, M.F.; Attia, N.F. Occurrence and spatial distribution of pharmaceuticals and personal care products (PPCPs) in the aquatic environment, their characteristics, and adopted legislations. J. Water Process Eng. 2023, 52, 103490. [Google Scholar] [CrossRef]
- Puri, M.; Gandhi, K.; Kumar, M.S. Emerging environmental contaminants: A global perspective on policies and regulations. J. Environ. Manag. 2023, 332, 117344. [Google Scholar] [CrossRef] [PubMed]
- Taoufik, N.; Boumya, W.; Janani, F.Z.; Elhalil, A.; Mahjoubi, F.Z. Removal of emerging pharmaceutical pollutants: A systematic mapping study review. J. Environ. Chem. Eng. 2020, 8, 104251. [Google Scholar] [CrossRef]
- Quesada, H.B.; Baptista, A.T.A.; Cusioli, L.F.; Seibert, D.; de Oliveira Bezerra, C.; Bergamasco, R. Surface water pollution by pharmaceuticals and an alternative of removal by low-cost adsorbents: A review. Chemosphere 2019, 222, 766–780. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Chaudhry, S.A. Adsorption of pharmaceutical pollutants using lignocellulosic materials. In Green Materials for Wastewater Treatment, Environmental Chemistry for a Sustainable World; Naushad, M., Lichtfouse, E., Eds.; Springer: Cham, Switzerland, 2020; Volume 38, pp. 277–289. [Google Scholar] [CrossRef]
- Vinayagam, V.; Murugan, S.; Kumaresan, R.; Narayanan, M.; Sillanpää, M.; Dai Viet, N.V.; Kushwaha, O.S.; Potdar, P.; Gadiya, S. Sustainable adsorbents for the removal of pharmaceuticals from wastewater: A review. Chemosphere 2022, 300, 134597. [Google Scholar] [CrossRef]
- Chen, C.; Wang, Y.; Qian, Y.; Zhao, X.; Wang, Q. The synergistic toxicity of the multiple chemical mixtures: Implications for risk assessment in the terrestrial environment. Environ. Int. 2015, 77, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Martin, O.; Scholze, M.; Ermler, S.; McPhie, J.; Bopp, S.K.; Kienzler, A.; Parissis, N.; Kortenkamp, V. Ten years of research on synergisms and antagonisms in chemical mixtures: A systematic review and quantitative reappraisal of mixture studies. Environ. Int. 2021, 146, 106206. [Google Scholar] [CrossRef]
- Gustavsson, M.; Molander, S.; Backhaus, T.; Kristiansson, E. Estimating the release of chemical substances from consumer products, textiles and pharmaceuticals to wastewater. Chemosphere 2022, 287, 131854. [Google Scholar] [CrossRef]
- Martin, O.V. Synergistic effects of chemical mixtures: How frequent is rare? Curr. Opin. Toxicol. 2023, 36, 100424. [Google Scholar] [CrossRef]
- Posthuma, L.; Zijp, M.C.; De Zwart, D.; Van de Meent, D.; Globevnik, L.; Koprivsek, M.; Focks, A.; van Gils, J.; Birk, S. Chemical pollution imposes limitations to the ecological status of European surface waters. Sci. Rep. 2020, 10, 14825. [Google Scholar] [CrossRef]
- Ukaogo, P.O.; Ewuzie, U.; Onwuka, C.V. Environmental pollution: Causes, effects, and the remedies. In Microorganisms for Sustainable Environment and Health; Chowdhary, P., Raj, A., Verma, D., Akhter, Y., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 419–429. [Google Scholar] [CrossRef]
- Naidu, R.; Biswas, B.; Willett, I.R.; Cribb, J.; Singh, B.K.; Nathanail, C.P.; Coulon, F.; Semple, K.T.; Jones, K.C.; Barclay, A.; et al. Chemical pollution: A growing peril and potential catastrophic risk to humanity. Environ. Int. 2021, 156, 106616. [Google Scholar] [CrossRef]
- Geissen, V.; Mol, H.; Klumpp, E.; Umlauf, G.; Nadal, M.; van der Ploeg, M.; van de Zee, M.; van de Zee, S.E.A.T.M.; Ritsema, C.J. Emerging pollutants in the environment: A challenge for water resource management. Int. Soil Water Conserv. Res. 2015, 3, 57–65. [Google Scholar] [CrossRef]
- Krishnakumar, S.; Singh, D.S.H.; Godson, P.S.; Thanga, S.G. Emerging pollutants: Impact on environment, management, and challenges. Environ. Sci. Pollut. Res. 2022, 29, 72309–72311. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Jin, R.; Chen, Q.; Du, X.; Yang, J.; Liu, M. Organic contaminants of emerging concern in global estuaries: Environmental occurrence, fate, and bioavailability. Crit. Rev. Environ. Sci. Technol. 2023, 53, 550–575. [Google Scholar] [CrossRef]
- Mishra, R.K.; Mentha, S.S.; Misra, Y.; Dwivedi, N. Emerging pollutants of severe environmental concern in water and wastewater: A comprehensive review on current developments and future research. Water-Energy Nexus 2023, 6, 74–95. [Google Scholar] [CrossRef]
- Wang, F.; Xiang, L.; Leung, K.S.; Elsner, M.; Zhang, Y.; Guo, Y.; Pan, B.; Sun, H.; An, T.; Ying, G.; et al. Emerging contaminants: A One Health perspective. Innovation 2024, 5, 100612. [Google Scholar] [CrossRef] [PubMed]
- Sanganyado, E. Policies and regulations for the emerging pollutants in freshwater ecosystems: Challenges and opportunities. In Emerging Freshwater Pollutants; Dalu, T., Tavengwa, N.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 361–372. [Google Scholar] [CrossRef]
- Tavengwa, N.T.; Moyo, B.; Musarurwa, H.; Dalu, T. Challenges and future directions in the analysis of emerging pollutants in aqueous environments. In Emerging Freshwater Pollutants; Dalu, T., Tavengwa, N.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 373–379. [Google Scholar] [CrossRef]
- Pereira, L.C.; de Souza, A.O.; Bernardes, M.F.F.; Pazin, M.; Tasso, M.J.; Pereira, P.H.; Dorta, D.J. A perspective on the potential risks of emerging contaminants to human and environmental health. Environ. Sci. Pollut. Res. 2015, 22, 13800–13823. [Google Scholar] [CrossRef]
- Lei, M.; Zhang, L.; Lei, J.; Zong, L.; Li, J.; Wu, Z.; Wang, Z. Overview of emerging contaminants and associated human health effects. BioMed Res. Int. 2015, 2015, 404796. [Google Scholar] [CrossRef]
- Abdulrazaq, Y.; Abdulsalam, A.; Rotimi, A.L.; Abdulbasit, A.A.; Clifford, O.; Abdulsalam, O.A.; Racheal, O.N.; Joy, A.A.; Victor, F.O.; Johannes, Z.M.; et al. Classification, potential routes and risk of emerging pollutants/contaminant. In Emerging Contaminants; Nuro, A., Ed.; IntechOpen: Rijeka, Croatia, 2020. [Google Scholar] [CrossRef]
- Vasilachi, I.C.; Asiminicesei, D.M.; Fertu, D.I.; Gavrilescu, M. Occurrence and fate of emerging pollutants in water environment and options for their removal. Water 2021, 13, 181. [Google Scholar] [CrossRef]
- NORMAN Network. 2024. Available online: https://www.norman-network.com (accessed on 17 July 2024).
- Khan, S.; Naushad, M.; Govarthanan, M.; Iqbal, J.; Alfadul, S.M. Emerging contaminants of high concern for the environment: Current trends and future research. Environ. Res. 2022, 207, 112609. [Google Scholar] [CrossRef]
- de Araújo, L.C.A.; da Silva, S.M.; Lima, A.V.A.; Barbosa, A.V.; dos Santos Silva, J.; Leite, K.M.; Júnior, W.J.D.N.; Silveira-Filho, V.D.M.; Mendes-Marques, C.L.; de Oliveira, M.B.M. Effects of Antibiotics on Impacted Aquatic Environment Microorganisms. In Emerging Contaminants; Nuro, A., Ed.; IntechOpen: Rijeka, Croatia, 2020. [Google Scholar] [CrossRef]
- Ortúzar, M.; Esterhuizen, M.; Olicón-Hernández, D.R.; González-López, J.; Aranda, E. Pharmaceutical pollution in aquatic environments: A concise review of environmental impacts and bioremediation systems. Front. Microbiol. 2022, 13, 869332. [Google Scholar] [CrossRef]
- Felis, E.; Kalka, J.; Sochacki, A.; Kowalska, K.; Bajkacz, S.; Harnisz, M.; Korzeniewska, E. Antimicrobial pharmaceuticals in the aquatic environment-occurrence and environmental implications. Eur. J. Pharmacol. 2020, 866, 172813. [Google Scholar] [CrossRef] [PubMed]
- Yazdan, M.M.S.; Kumar, R.; Leung, S.W. The environmental and health impacts of steroids and hormones in wastewater effluent, as well as existing removal technologies: A review. Ecologies 2022, 3, 206–224. [Google Scholar] [CrossRef]
- Bean, T.G.; Chadwick, E.A.; Herrero-Villar, M.; Mateo, R.; Naidoo, V.; Rattner, B.A. Do pharmaceuticals in the environment pose a risk to wildlife? Environ. Toxicol. Chem. 2024, 43, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Pires, P.; Pereira, A.M.; Pena, A.; Silva, L.J. Non-Steroidal Anti-Inflammatory Drugs in the Aquatic Environment and Bivalves: The State of the Art. Toxics 2024, 12, 415. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Zhang, J.; Hu, L.; Yu, Z.; Lai, S.; Liu, Y.; Mai, Z.; Xu, M. Trace analysis of 47 psychotropic medications in environmental samples by ultra-performance liquid chromatography tandem mass spectrometry (UPLC-MS/MS). J. Chromatogr. A 2024, 1715, 464627. [Google Scholar] [CrossRef] [PubMed]
- Currie, G.M. Pharmacology, part 1: Introduction to pharmacology and pharmacodynamics. J. Nucl. Med. Technol. 2018, 46, 81–86. [Google Scholar] [CrossRef]
- Santiago, M.; Davis, E.A.; Hinton, T.; Angelo, T.A.; Shield, A.; Babey, A.M.; Kemp-Harper, B.; Maynard, G.; Al-Sallami, H.S.; Musgrave, I.F.; et al. Defining and unpacking the core concepts of pharmacology education. Pharmacol. Res. Pers. 2021, 9, e00894. [Google Scholar] [CrossRef]
- Abrantes, C.G.; Duarte, D.; Reis, C.P. An overview of pharmaceutical excipients: Safe or not safe? J. Pharm. Sci. 2016, 105, 2019–2026. [Google Scholar] [CrossRef]
- Urbina, J.A.J.; Solano, J.A.V. Los contaminantes emergentes de las aguas residuales de la industria farmacéutica y su tratamiento por medio de la ozonización. Inf. Tec. 2020, 84, 249–263. [Google Scholar] [CrossRef]
- Elder, D.P.; Kuentz, M.; Holm, R. Pharmaceutical excipients—Quality, regulatory and biopharmaceutical considerations. Eur. J. Pharm. Sci. 2016, 87, 88–99. [Google Scholar] [CrossRef]
- Patel, R.; Barker, J.; ElShaer, A. Pharmaceutical excipients and drug metabolism: A mini-review. Int. J. Mol. Sci. 2020, 21, 8224. [Google Scholar] [CrossRef] [PubMed]
- Pockle, R.D.; Masareddy, R.S.; Patil, A.S.; Patil, P.D. A comprehensive review on pharmaceutical excipients. Ther. Deliv. 2023, 14, 443–458. [Google Scholar] [CrossRef]
- Majumder, A.; Gupta, B.; Gupta, A.K. Pharmaceutically active compounds in aqueous environment: A status, toxicity and insights of remediation. Environ. Res. 2019, 176, 108542. [Google Scholar] [CrossRef] [PubMed]
- Berdigaliyev, N.; Aljofan, M. An overview of drug discovery and development. Future Med. Chem. 2020, 12, 939–947. [Google Scholar] [CrossRef]
- Charuaud, L.; Jarde, E.; Jaffrezic, A.; Thomas, M.F.; Le Bot, B. Veterinary pharmaceutical residues from natural water to tap water: Sales, occurrence and fate. J. Hazard. Mater. 2019, 361, 169–186. [Google Scholar] [CrossRef] [PubMed]
- Domingo-Echaburu, S.; Dávalos, L.M.; Orive, G.; Lertxundi, U. Drug pollution & sustainable development goals. Sci. Total Environ. 2021, 800, 149412. [Google Scholar] [CrossRef] [PubMed]
- Kairigo, P.; Ngumba, E.; Sundberg, L.R.; Gachanja, A.; Tuhkanen, T. Contamination of surface water and river sediments by antibiotic and antiretroviral drug cocktails in low and middle-income countries: Occurrence, risk and mitigation strategies. Water 2020, 12, 1376. [Google Scholar] [CrossRef]
- Desai, M.; Njoku, A.; Nimo-Sefah, L. Comparing environmental policies to reduce pharmaceutical pollution and address disparities. Int. J. Environ. Res. Public Health 2022, 19, 8292. [Google Scholar] [CrossRef]
- Delgado, N.; Orozco, J.; Zambrano, S.; Casas-Zapata, J.C.; Marino, D. Veterinary pharmaceutical as emerging contaminants in wastewater and surface water: An overview. J. Hazard. Mater. 2023, 460, 132431. [Google Scholar] [CrossRef]
- Islas-Flores, H.; Gómez-Oliván, L.M. Legislation Controlling the Discharge of Pharmaceuticals into the Environment. In Ecopharmacovigilance; Gómez-Oliván, L., Ed.; The Handbook of Environmental Chemistry; Springer: Cham, Switzerland, 2017; Volume 66, pp. 95–117. [Google Scholar] [CrossRef]
- de Oliveira Souza, H.; dos Santos Costa, R.; Quadra, G.R.; dos Santos Fernandez, M.A. Pharmaceutical pollution and sustainable development goals: Going the right way? Sustain. Chem. Pharm. 2021, 21, 100428. [Google Scholar] [CrossRef]
- Miettinen, M.; Khan, S.A. Pharmaceutical pollution: A weakly regulated global environmental risk. Rev. Eur. Comp. Int. Environ. Law 2022, 31, 75–88. [Google Scholar] [CrossRef]
- González-Peña, O.I.; López-Zavala, M.Á.; Cabral-Ruelas, H. Pharmaceuticals market, consumption trends and disease incidence are not driving the pharmaceutical research on water and wastewater. Int. J. Environ. Res. 2021, 18, 2532. [Google Scholar] [CrossRef]
- Freitas, L.D.A.A.; Radis-Baptista, G. Pharmaceutical pollution and disposal of expired, unused, and unwanted medicines in the Brazilian context. J. Xenobiot. 2021, 11, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Fu, Y.H.; Sheng, H.J.; Topp, E.; Jiang, X.; Zhu, Y.G.; Tiedje, J.M. Antibiotic resistance in the soil ecosystem: A One Health perspective. Curr. Opin. Environ. Sci. Health 2021, 20, 100230. [Google Scholar] [CrossRef]
- Fabbri, E. Pharmaceuticals in the environment: Expected and unexpected effects on aquatic fauna. Ann. N. Y. Acad. Sci. 2015, 1340, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Mezzelani, M.; Gorbi, S.; Regoli, F. Pharmaceuticals in the aquatic environments: Evidence of emerged threat and future challenges for marine organisms. Mar. Environ. Res. 2018, 140, 41–60. [Google Scholar] [CrossRef]
- Srain, H.S.; Beazley, K.F.; Walker, T.R. Pharmaceuticals and personal care products and their sublethal and lethal effects in aquatic organisms. Environ. Rev. 2021, 29, 142–181. [Google Scholar] [CrossRef]
- Wang, C.; Ye, D.; Li, X.; Jia, Y.; Zhao, L.; Liu, S.; Xu, J.; Du, J.; Tian, L.; Li, J.; et al. Occurrence of pharmaceuticals and personal care products in bottled water and assessment of the associated risks. Environ. Int. 2021, 155, 106651. [Google Scholar] [CrossRef]
- Papagiannaki, D.; Belay, M.H.; Gonçalves, N.P.; Robotti, E.; Bianco-Prevot, A.; Binetti, R.; Calza, P. From monitoring to treatment, how to improve water quality: The pharmaceuticals case. Chem. Eng. J. Adv. 2022, 10, 100245. [Google Scholar] [CrossRef]
- Akhtar, J.; Amin, N.A.S.; Shahzad, K. A review on removal of pharmaceuticals from water by adsorption. Desalination Water Treat. 2016, 57, 12842–12860. [Google Scholar] [CrossRef]
- de Andrade, J.R.; Oliveira, M.F.; da Silva, M.G.; Vieira, M.G. Adsorption of pharmaceuticals from water and wastewater using nonconventional low-cost materials: A review. Ind. Eng. Chem. Res. 2018, 57, 3103–3127. [Google Scholar] [CrossRef]
- Guo, J.; Ren, J.; Chang, C.; Duan, Q.; Li, J.; Kanerva, M.; Yang, F.; Mo, J. Freshwater crustacean exposed to active pharmaceutical ingredients: Ecotoxicological effects and mechanisms. Environ. Sci. Pollut. Res. 2023, 30, 48868–48902. [Google Scholar] [CrossRef] [PubMed]
- Daughton, C.G.; Ruhoy, I.S. The afterlife of drugs and the role of pharmEcovigilance. Drug Saf. 2008, 31, 1069–1082. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.L.; Song, C.; He, L.Y.; Wang, B.; Gao, F.Z.; Zhang, M.; Ying, G.G. Antibiotics in soil and water: Occurrence, fate, and risk. Curr. Opin. Environ. Sci. Health 2023, 32, 100437. [Google Scholar] [CrossRef]
- Li, M.; Yang, L.; Yen, H.; Zhao, F.; Wang, X.; Zhou, T.; Feng, Q.; Chen, L. Occurrence, spatial distribution and ecological risks of antibiotics in soil in urban agglomeration. J. Environ. Sci. 2023, 125, 678–690. [Google Scholar] [CrossRef]
- Yang, Q.; Gao, Y.; Ke, J.; Show, P.L.; Ge, Y.; Liu, Y.; Guo, R.; Chen, J. Antibiotics: An overview on the environmental occurrence, toxicity, degradation, and removal methods. Bioengineered 2021, 12, 7376–7416. [Google Scholar] [CrossRef]
- da Silva Freitas, L.; Honscha, L.C.; Volcão, L.M.; de Lima Brum, R.; da Silva Júnior, F.M.R.; Ramos, D.F. Antibiotics in the Environment: Prescribing risks to non-target organisms. Pollutants 2022, 2, 435–443. [Google Scholar] [CrossRef]
- Xu, K.; Wang, J.; Gong, H.; Li, Y.; Zhou, L.; Yan, M. Occurrence of antibiotics and their associations with antibiotic resistance genes and bacterial communities in Guangdong coastal areas. Ecotoxicol. Environ. Saf. 2019, 186, 109796. [Google Scholar] [CrossRef]
- Siedlewicz, G.; Żak, A.; Sharma, L.; Kosakowska, A.; Pazdro, K. Effects of oxytetracycline on growth and chlorophyll a fluorescence in green algae (Chlorella vulgaris), diatom (Phaeodactylum tricornutum) and cyanobacteria (Microcystis aeruginosa and Nodularia spumigena). Oceanologia 2020, 62, 214–225. [Google Scholar] [CrossRef]
- Isidori, M.; Lavorgna, M.; Nardelli, A.; Pascarella, L.; Parrella, A. Toxic and genotoxic evaluation of six antibiotics on non-target organisms. Sci. Total Environ. 2005, 346, 87–98. [Google Scholar] [CrossRef]
- Kovalakova, P.; Cizmas, L.; McDonald, T.J.; Marsalek, B.; Feng, M.; Sharma, V.K. Occurrence and toxicity of antibiotics in the aquatic environment: A review. Chemosphere 2020, 251, 126351. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Song, G.; Lim, W. A review of the toxicity in fish exposed to antibiotics. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2020, 237, 108840. [Google Scholar] [CrossRef] [PubMed]
- Buelow, E.; Ploy, M.C.; Dagot, C. Role of pollution on the selection of antibiotic resistance and bacterial pathogens in the environment. Curr. Opin. Microbiol. 2021, 64, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Feng, G.; Huang, H.; Chen, Y. Effects of emerging pollutants on the occurrence and transfer of antibiotic resistance genes: A review. J. Hazard. Mater. 2021, 420, 126602. [Google Scholar] [CrossRef] [PubMed]
- Larsson, D.G.; Flach, C.F. Antibiotic resistance in the environment. Nat. Rev. Microbiol. 2022, 20, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Tolba, R. Nonsteroidal anti-inflammatory drugs (NSAIDs). In Treatment of Chronic Pain Conditions; Pope, J., Deer, T., Eds.; Springer: New York, NY, USA, 2017; pp. 77–79. [Google Scholar] [CrossRef]
- Gunaydin, C.; Bilge, S.S. Effects of nonsteroidal anti-inflammatory drugs at the molecular level. Eurasian J. Med. 2018, 50, 116–121. [Google Scholar] [CrossRef]
- Bindu, S.; Mazumder, S.; Bandyopadhyay, U. Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochem. Pharmacol. 2020, 180, 114147. [Google Scholar] [CrossRef]
- Kołecka, K.; Gajewska, M.; Caban, M. From the pills to environment–Prediction and tracking of non-steroidal anti-inflammatory drug concentrations in wastewater. Sci. Total Environ. 2022, 825, 153611. [Google Scholar] [CrossRef]
- Izadi, P.; Izadi, P.; Salem, R.; Papry, S.A.; Magdouli, S.; Pulicharla, R.; Brar, S.K. Non-steroidal anti-inflammatory drugs in the environment: Where were we and how far we have come? Environ. Pollut. 2020, 267, 115370. [Google Scholar] [CrossRef]
- He, K.; Borthwick, A.G.; Lin, Y.; Li, Y.; Fu, J.; Wong, Y.; Liu, W. Sale-based estimation of pharmaceutical concentrations and associated environmental risk in the Japanese wastewater system. Environ. Int. 2020, 139, 105690. [Google Scholar] [CrossRef]
- Hiba, Z.I.N.D.; Mondamert, L.; Remaury, Q.B.; Cleon, A.; Leitner, N.K.V.; Labanowski, J. Occurrence of carbamazepine, diclofenac, and their related metabolites and transformation products in a French aquatic environment and preliminary risk assessment. Water Res. 2021, 196, 117052. [Google Scholar] [CrossRef]
- Sánchez-Sánchez, E.; Fernández-Cerezo, F.L.; Díaz-Jimenez, J.; Rosety-Rodriguez, M.; Díaz, A.J.; Ordonez, F.J.; Rosety, M.A.; Rosety, I. Consumption of over-the-counter drugs: Prevalence and type of drugs. Int. J. Environ. Res. Public Health 2021, 18, 5530. [Google Scholar] [CrossRef] [PubMed]
- Świacka, K.; Michnowska, A.; Maculewicz, J.; Caban, M.; Smolarz, K. Toxic effects of NSAIDs in non-target species: A review from the perspective of the aquatic environment. Environ. Pollut. 2021, 273, 115891. [Google Scholar] [CrossRef]
- Bonnesen, K.; Ehrenstein, V.; Grønkjær, M.S.; Pedersen, L.; Lash, T.L.; Schmidt, M. Impact of lifestyle and socioeconomic position on use of non-steroidal anti-inflammatory drugs: A population-based cohort study. Pharmacoepidemiol. Drug Saf. 2023, 32, 455–467. [Google Scholar] [CrossRef]
- Trombini, C.; Blasco, J.; Hampel, M. Ibuprofen and Diclofenac: Effects on Freshwater and Marine Aquatic Organisms—Are They at Risk? In Non-Steroidal Anti-Inflammatory Drugs in Water. The Handbook of Environmental Chemistry; Gómez-Oliván, L.M., Ed.; Springer: Cham, Switzerland, 2020; pp. 161–189. [Google Scholar] [CrossRef]
- Muñiz-González, A.B. Ibuprofen as an emerging pollutant on non-target aquatic invertebrates: Effects on Chironomus riparius. Environ. Toxicol. Pharmacol. 2021, 81, 103537. [Google Scholar] [CrossRef]
- Karki, B.K.; Philip, L. Fate of pharmaceuticals and personal care products like metronidazole, naproxen, and methylparaben and their effect on the physiological characteristics of two wetland plants. Chem. Eng. J. 2024, 483, 149180. [Google Scholar] [CrossRef]
- Batucan, N.S.P.; Tremblay, L.A.; Northcott, G.L.; Matthaei, C.D. Medicating the environment? A critical review on the risks of carbamazepine, diclofenac and ibuprofen to aquatic organisms. Environ. Adv. 2022, 7, 100164. [Google Scholar] [CrossRef]
- Jan-Roblero, J.; Cruz-Maya, J.A. Ibuprofen: Toxicology and biodegradation of an emerging contaminant. Molecules 2023, 28, 2097. [Google Scholar] [CrossRef]
- Das, S.A.; Karmakar, S.; Chhaba, B.; Rout, S.K. Ibuprofen: Its toxic effect on aquatic organisms. J. Exp. Zool. India 2019, 22, 1125–1131. [Google Scholar]
- Lonappan, L.; Brar, S.K.; Das, R.K.; Verma, M.; Surampalli, R.Y. Diclofenac and its transformation products: Environmental occurrence and toxicity—A review. Environ. Int. 2016, 96, 127–138. [Google Scholar] [CrossRef]
- Bonnefille, B.; Gomez, E.; Courant, F.; Escande, A.; Fenet, H. Diclofenac in the marine environment: A review of its occurrence and effects. Mar. Pollut. Bull. 2018, 131, 496–506. [Google Scholar] [CrossRef]
- Nunes, B.; Daniel, D.; Canelas, G.G.; Barros, J.; Correia, A.T. Toxic effects of environmentally realistic concentrations of diclofenac in organisms from two distinct trophic levels, Hediste diversicolor and Solea senegalensis. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2020, 231, 108722. [Google Scholar] [CrossRef] [PubMed]
- Mirzaee, S.A.; Noorimotlagh, Z.; Ahmadi, M.; Rahim, F.; Martinez, S.S.; Nourmohammadi, A.; Jaafarzadeh, N. The possible oxidative stress and DNA damage induced in Diclofenac-exposed Non-target organisms in the aquatic environment: A systematic review. Ecol. Indic. 2021, 131, 108172. [Google Scholar] [CrossRef]
- Nunes, B.; Antunes, S.C.; Santos, J.; Martins, L.; Castro, B.B. Toxic potential of paracetamol to freshwater organisms: A headache to environmental regulators? Ecotoxicol. Environ. Saf. 2014, 107, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Nunes, B. Ecotoxicological effects of the drug paracetamol: A critical review of past ecotoxicity assessments and future perspectives. In Non-Steroidal Anti-Inflammatory Drugs in Water. The Handbook of Environmental Chemistry; Gómez-Oliván, L.M., Ed.; Springer: Cham, Switzerland, 2020; Volume 96, pp. 131–145. [Google Scholar] [CrossRef]
- Vieira, Y.; Spode, J.E.; Dotto, G.L.; Georgin, J.; Franco, D.S.P.; dos Reis, G.S.; Lima, E.C. Paracetamol environmental remediation and ecotoxicology: A review. Environ. Chem. Lett. 2024, 22, 2343–2373. [Google Scholar] [CrossRef]
- Wojcieszyńska, D.; Guzik, U. Naproxen in the environment: Its occurrence, toxicity to nontarget organisms and biodegradation. Appl. Microbiol. Biotechnol. 2020, 104, 1849–1857. [Google Scholar] [CrossRef]
- Mezzelani, M.; Gorbi, S.; Da Ros, Z.; Fattorini, D.; d’Errico, G.; Milan, M.; Bargelloni, L.; Regoli, F. Ecotoxicological potential of non-steroidal anti-inflammatory drugs (NSAIDs) in marine organisms: Bioavailability, biomarkers and natural occurrence in Mytilus galloprovincialis. Mar. Environ. Res. 2016, 121, 31–39. [Google Scholar] [CrossRef]
- Parolini, M. Toxicity of the Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) acetylsalicylic acid, paracetamol, diclofenac, ibuprofen and naproxen towards freshwater invertebrates: A review. Sci. Total Environ. 2020, 740, 140043. [Google Scholar] [CrossRef]
- Negro, R.; Attanasio, R.; Nagy, E.V.; Papini, E.; Perros, P.; Hegedüs, L. Use of thyroid hormones in hypothyroid and euthyroid patients; the 2019 Italian survey. Eur. Thyroid J. 2020, 9, 25–31. [Google Scholar] [CrossRef]
- Zucchi, R. Thyroid hormone analogues: An update. Thyroid 2020, 30, 1099–1105. [Google Scholar] [CrossRef]
- Hirsch, I.B.; Juneja, R.; Beals, J.M.; Antalis, C.J.; Wright Jr, E.E. The evolution of insulin and how it informs therapy and treatment choices. Endocr. Rev. 2020, 41, 733–755. [Google Scholar] [CrossRef] [PubMed]
- Sims, E.K.; Carr, A.L.; Oram, R.A.; DiMeglio, L.A.; Evans-Molina, C. 100 years of insulin: Celebrating the past, present and future of diabetes therapy. Nat. Med. 2021, 27, 1154–1164. [Google Scholar] [CrossRef] [PubMed]
- Aktas, G.; Taslamacioglu Duman, T. Current usage of long-acting insulin analogs in patients with type 2 diabetes mellitus. Expert Rev. Endocrinol. Metab. 2024, 19, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Kapugi, M.; Cunningham, K. Corticosteroids. Orthop. Nurs. 2019, 38, 336–339. [Google Scholar] [CrossRef]
- Mustafa, S.S. Steroid-induced secondary immune deficiency. Ann. Allerg. Asthma Immunol. 2023, 130, 713–717. [Google Scholar] [CrossRef]
- Borzutzky, C.; Jaffray, J. Diagnosis and management of heavy menstrual bleeding and bleeding disorders in adolescents. JAMA Pediatr. 2020, 174, 186–194. [Google Scholar] [CrossRef]
- Jewson, M.; Purohit, P.; Lumsden, M.A. Progesterone and abnormal uterine bleeding/menstrual disorders. Best Pract. Res. Clin. Obstet. Gynaecol. 2020, 69, 62–73. [Google Scholar] [CrossRef]
- Minkin, M.J. Menopause: Hormones, lifestyle, and optimizing aging. Obstet. Gynecol. Clin. 2019, 46, 501–514. [Google Scholar] [CrossRef]
- Flores, V.A.; Pal, L.; Manson, J.E. Hormone therapy in menopause: Concepts, controversies, and approach to treatment. Endocr. Rev. 2021, 42, 720–752. [Google Scholar] [CrossRef]
- Bhasin, S.; Brito, J.P.; Cunningham, G.R.; Hayes, F.J.; Hodis, H.N.; Matsumoto, A.M.; Snyder, P.J.; Swerdloff, R.S.; Wu, F.C.; Yialamas, M.A. Testosterone therapy in men with hypogonadism: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2018, 103, 1715–1744. [Google Scholar] [CrossRef] [PubMed]
- Klein, K.O.; Phillips, S.A. Review of hormone replacement therapy in girls and adolescents with hypogonadism. J. Pediatr. Adolesc. Gynecol. 2019, 32, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Rey, R.A. Recent advancement in the treatment of boys and adolescents with hypogonadism. Ther. Adv. Endocrinol. Metab. 2022, 13, 20420188211065660. [Google Scholar] [CrossRef] [PubMed]
- Rodprasert, W.; Virtanen, H.E.; Mäkelä, J.A.; Toppari, J. Hypogonadism and cryptorchidism. Front. Endocrinol. 2020, 10, 906. [Google Scholar] [CrossRef]
- Sun, T.; Xu, W.; Xu, H.; Chen, Y.; Niu, Y.; Wang, D.; Wang, T.; Yang, W.; Liu, J. Hormonal therapy is effective and safe for cryptorchidism caused by idiopathic hypogonadotropic hypogonadism in adult males. Front. Endocrinol. 2023, 13, 1095950. [Google Scholar] [CrossRef] [PubMed]
- Ulm, M.; Ramesh, A.V.; McNamara, K.M.; Ponnusamy, S.; Sasano, H.; Narayanan, R. Therapeutic advances in hormone-dependent cancers: Focus on prostate, breast and ovarian cancers. Endocr. Connect. 2019, 8, R10–R26. [Google Scholar] [CrossRef] [PubMed]
- Emons, G. Hormone-dependent cancers: Molecular mechanisms and Therapeutical implications. Cells 2022, 12, 110. [Google Scholar] [CrossRef]
- Chimento, A.; De Luca, A.; Avena, P.; De Amicis, F.; Casaburi, I.; Sirianni, R.; Pezzi, V. Estrogen receptors-mediated apoptosis in hormone-dependent cancers. Int. J. Mol. Sci. 2022, 23, 1242. [Google Scholar] [CrossRef] [PubMed]
- Festin, M.P.R. Overview of modern contraception. Best Pract. Res. Clin. Obstet. Gynaecol. 2020, 66, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.J.; Johnston, D.S. A brief history and future prospects of contraception. Science 2023, 380, 154–158. [Google Scholar] [CrossRef]
- Adeel, M.; Song, X.; Wang, Y.; Francis, D.; Yang, Y. Environmental impact of estrogens on human, animal and plant life: A critical review. Environ. Int. 2017, 99, 107–119. [Google Scholar] [CrossRef]
- Ciślak, M.; Kruszelnicka, I.; Zembrzuska, J.; Ginter-Kramarczyk, D. Estrogen pollution of the European aquatic environment: A critical review. Water Res. 2023, 229, 119413. [Google Scholar] [CrossRef] [PubMed]
- Itana, D.D.; Duguma, A. The role and impacts of growth hormones in maximizing animal production—A review. Turk. J. Agric. Food Sci. Technol. 2021, 9, 975–981. [Google Scholar] [CrossRef]
- Qaid, M.M.; Abdoun, K.A. Safety and concerns of hormonal application in farm animal production: A review. J. Appl. Anim. Res. 2022, 50, 426–439. [Google Scholar] [CrossRef]
- Skoupá, K.; Šťastný, K.; Sládek, Z. Anabolic Steroids in Fattening Food-Producing Animals—A Review. Animals 2022, 12, 2115. [Google Scholar] [CrossRef] [PubMed]
- Hirpessa, B.B.; Ulusoy, B.H.; Hecer, C. Hormones and hormonal anabolics: Residues in animal source food, potential public health impacts, and methods of analysis. J. Food Qual. 2020, 2020, 5065386. [Google Scholar] [CrossRef]
- Evans, H.C.; Briggs, E.F.; Burnett, R.H.; Contreras-Correa, Z.E.; Duvic, M.A.; Dysart, L.M.; Gilmore, A.A.; Messman, R.D.; Reid, D.; Ugur, M.R.; et al. Harnessing the value of reproductive hormones in cattle production with considerations to animal welfare and human health. J. Anim. Sci. 2022, 100, skac177. [Google Scholar] [CrossRef]
- Cheng, D.; Ngo, H.H.; Guo, W.; Chang, S.W.; Nguyen, D.D.; Liu, Y.; Liu, Y.; Wei, Q.; Wei, D. A critical review on antibiotics and hormones in swine wastewater: Water pollution problems and control approaches. J. Hazard. Mater. 2020, 387, 121682. [Google Scholar] [CrossRef]
- Sacdal, R.; Madriaga, J.; Espino, M.P. Overview of the analysis, occurrence and ecological effects of hormones in lake waters in Asia. Environ. Res. 2020, 182, 109091. [Google Scholar] [CrossRef]
- Zhong, R.; Zou, H.; Gao, J.; Wang, T.; Bu, Q.; Wang, Z.L.; Hu, M.; Wang, Z. A critical review on the distribution and ecological risk assessment of steroid hormones in the environment in China. Sci. Total Environ. 2021, 786, 147452. [Google Scholar] [CrossRef]
- Bilal, M.; Barceló, D.; Iqbal, H.M. Occurrence, environmental fate, ecological issues, and redefining of endocrine disruptive estrogens in water resources. Sci. Total Environ. 2021, 800, 149635. [Google Scholar] [CrossRef]
- Ojoghoro, J.O.; Scrimshaw, M.D.; Sumpter, J.P. Steroid hormones in the aquatic environment. Sci. Total Environ. 2021, 792, 148306. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wang, X.L.; Zhang, J.W.; Wu, K.S. Impact of endocrine-disrupting chemicals on reproductive function in zebrafish (Danio rerio). Reprod. Domest. Anim. 2015, 50, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Azizi-Lalabadi, M.; Pirsaheb, M. Investigation of steroid hormone residues in fish: A systematic review. Process Saf. Environ. Prot. 2021, 152, 14–24. [Google Scholar] [CrossRef]
- Jenila, J.S.; Issac, P.K.; Lam, S.S.; Oviya, J.C.; Jones, S.; Munusamy-Ramanujam, G.; Chang, S.W.; Ravindran, B.; Mannacharaju, M.; Ghotekar, S.; et al. Deleterious effect of gestagens from wastewater effluent on fish reproduction in aquatic environment: A review. Environ. Res. 2023, 236, 116810. [Google Scholar] [CrossRef] [PubMed]
- Adeola, A.O.; Ore, O.T.; Fapohunda, O.; Adewole, A.H.; Akerele, D.D.; Akingboye, A.S.; Oloye, F.F. Psychotropic drugs of emerging concerns in aquatic systems: Ecotoxicology and remediation approaches. Chem. Afr. 2022, 5, 481–508. [Google Scholar] [CrossRef]
- Nogueira, A.F.; Nunes, B. Acute and chronic effects of diazepam on the polychaete Hediste diversicolor: Antioxidant, metabolic, pharmacologic, neurotoxic and behavioural mechanistic traits. Environ. Toxicol. Pharmacol. 2021, 82, 103538. [Google Scholar] [CrossRef]
- Oggier, D.M.; Weisbrod, C.J.; Stoller, A.M.; Zenker, A.K.; Fent, K. Effects of diazepam on gene expression and link to physiological effects in different life stages in zebrafish Danio rerio. Environ. Sci. Technol. 2010, 44, 7685–7691. [Google Scholar] [CrossRef]
- Chen, K.; Wu, M.; Chen, C.; Xu, H.; Wu, X.; Qiu, X. Impacts of chronic exposure to sublethal diazepam on behavioral traits of female and male zebrafish (Danio rerio). Ecotoxicol. Environ. Saf. 2021, 208, 111747. [Google Scholar] [CrossRef]
- Qiu, X.; Wang, S.; Zhuo, M.; Shi, Y.; Chen, K. Diazepam at environmentally relevant concentrations disturbed social interactions and brain neurotransmitters in adult Japanese medaka (Oryzias latipes). Aquat. Toxicol. 2023, 265, 106773. [Google Scholar] [CrossRef]
- Overturf, C.L.; Overturf, M.D.; Huggett, D.B. Bioconcentration and endocrine disruption effects of diazepam in channel catfish, Ictalurus punctatus. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2016, 183, 46–52. [Google Scholar] [CrossRef]
- Baali, H.; Cosio, C. Effects of carbamazepine in aquatic biota. Environ. Sci. Process. Impacts 2022, 24, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Desbiolles, F.; Moreau, X.; de Jong, L.; Malleret, L.; Grandet-Marchant, Q.; Wong-Wah-Chung, P.; Laffont-Schwob, I. Advances and limits of two model species for ecotoxicological assessment of carbamazepine, two by-products and their mixture at environmental level in freshwater. Water Res. 2020, 169, 115267. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.; Soares, Â.M.; Esteves, V.I.; Freitas, R. Occurrence of the antiepileptic carbamazepine in water and bivalves from marine environments: A review. Environ. Toxicol. Pharmacol. 2021, 86, 103661. [Google Scholar] [CrossRef] [PubMed]
- Nkoom, M.; Lu, G.; Liu, J.; Dong, H. Biological uptake, depuration and biochemical effects of diclofenac and carbamazepine in Carassius carassius. Ecotoxicol. Environ. Saf. 2020, 205, 111106. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, M.; Mashinchian Moradi, A.; Mortazavi, P.; Jamili, S. Effects of chronic exposure to carbamazepine on hematological parameters in Cyprinus carpio. Iran. J. Fish. Sci. 2020, 19, 443–456. [Google Scholar] [CrossRef]
- Nikhil, J.; Maneesha, P.; Chitra, K.C. Neurotoxic effects of carbamazepine on the mosquitofish Gambusia affinis. Drug Chem. Toxicol. 2024, 1–15. [Google Scholar] [CrossRef]
- Cuerda-Correa, E.M.; Alexandre-Franco, M.F.; Fernández-González, C. Advanced oxidation processes for the removal of antibiotics from water. An overview. Water 2019, 12, 102. [Google Scholar] [CrossRef]
- Bermúdez, L.A.; Pascual, J.M.; Martínez, M.D.M.M.; Poyatos Capilla, J.M. Effectiveness of advanced oxidation processes in wastewater treatment: State of the art. Water 2021, 13, 2094. [Google Scholar] [CrossRef]
- Mansouri, F.; Chouchene, K.; Roche, N.; Ksibi, M. Removal of pharmaceuticals from water by adsorption and advanced oxidation processes: State of the art and trends. App. Sci. 2021, 11, 6659. [Google Scholar] [CrossRef]
- da Silva, T.L.; Costa, C.S.D.; da Silva, M.G.C.; Vieira, M.G.A. Overview of non-steroidal anti-inflammatory drugs degradation by advanced oxidation processes. J. Clean. Prod. 2022, 346, 131226. [Google Scholar] [CrossRef]
- Huang, L.; Shen, R.; Shuai, Q. Adsorptive removal of pharmaceuticals from water using metal-organic frameworks: A review. J. Environ. Manag. 2021, 277, 111389. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Sadia, M.; Azeem, M.; Ahmad, M.Z.; Umar, M.; Abbas, Z.U. Ion Exchange Resins and their Applications in Water Treatment and Pollutants Removal from Environment: A Review: Ion Exchange Resins and their Applications. Futur. Biotechnol. 2023, 3, 12–19. [Google Scholar] [CrossRef]
- Rashed, I.G.A.A.; Afify, H.A.; Ahmed, A.E.M.; Ayoub, M.A.E.S. Optimization of chemical precipitation to improve the primary treatment of wastewater. Desalination Water Treat. 2013, 51, 7048–7056. [Google Scholar] [CrossRef]
- Sun, W.; Ma, G.; Sun, Y.; Liu, Y.; Song, N.; Xu, Y.; Zheng, H. Effective treatment of high phosphorus pharmaceutical wastewater by chemical precipitation. Can. J. Chem. Eng. 2017, 95, 1585–1593. [Google Scholar] [CrossRef]
- Zaied, B.K.; Rashid, M.; Nasrullah, M.; Zularisam, A.W.; Pant, D.; Singh, L. A comprehensive review on contaminants removal from pharmaceutical wastewater by electrocoagulation process. Sci. Total Environ. 2020, 726, 138095. [Google Scholar] [CrossRef] [PubMed]
- Bampos, G.; Petala, A.; Frontistis, Z. Recent trends in pharmaceuticals removal from water using electrochemical oxidation processes. Environments 2021, 8, 85. [Google Scholar] [CrossRef]
- Wu, C.; Ge, J.; Gu, F.; Bai, L. Electrochemical oxidation technique to pharmaceutical pollutants removal. Chemosphere 2023, 337, 139373. [Google Scholar] [CrossRef] [PubMed]
- Heberer, T.; Feldmann, D. Removal of Pharmaceutical Residues from Contaminated Raw Water Sources by Membrane Filtration. In Pharmaceuticals in the Environment; Kümmerer, K., Ed.; Springer: Berlin/Heidelberg, Germany, 2004; pp. 391–410. [Google Scholar] [CrossRef]
- Gómez-Espinosa, R.M.; Arizmendi-Cotero, D. Role of Membrane on Emerging Contaminant Removal. In Ecopharmacovigilance; Gómez-Oliván, L., Ed.; The Handbook of Environmental Chemistry; Springer: Cham, Switzerland, 2017; Volume 66, pp. 157–174. [Google Scholar] [CrossRef]
- Żyłła, R.; Foszpańczyk, M.; Kamińska, I.; Kudzin, M.; Balcerzak, J.; Ledakowicz, S. Impact of polymer membrane properties on the removal of pharmaceuticals. Membranes 2022, 12, 150. [Google Scholar] [CrossRef]
- Mansour, F.; Al-Hindi, M.; Yahfoufi, R.; Ayoub, G.M.; Ahmad, M.N. The use of activated carbon for the removal of pharmaceuticals from aqueous solutions: A review. Rev. Environ. Sci. Biotechnol. 2018, 17, 109–145. [Google Scholar] [CrossRef]
- Köpping, I.; McArdell, C.S.; Borowska, E.; Böhler, M.A.; Udert, K.M. Removal of pharmaceuticals from nitrified urine by adsorption on granular activated carbon. Water Res. X 2020, 9, 100057. [Google Scholar] [CrossRef]
- Rocha, L.S.; Pereira, D.; Sousa, É.; Otero, M.; Esteves, V.I.; Calisto, V. Recent advances on the development and application of magnetic activated carbon and char for the removal of pharmaceutical compounds from waters: A review. Sci. Total Environ. 2020, 718, 137272. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xu, J.; Liu, X.; Sheng, L. Preparation of straw activated carbon and its application in wastewater treatment: A review. J. Clean. Prod. 2021, 283, 124671. [Google Scholar] [CrossRef]
- Palma, T.L.; Magno, G.; Costa, M.C. Biodegradation of paracetamol by some gram-positive bacterial isolates. Curr. Microbiol. 2021, 78, 2774–2786. [Google Scholar] [CrossRef] [PubMed]
- Grignet, R.D.S.; Barros, M.G.; Panatta, A.A.; Bernal, S.P.; Ottoni, J.R.; Passarini, M.R.; da CS Gonçalves, C. Medicines as an emergent contaminant: The review of microbial biodegration potential. Folia Microbiol. 2022, 67, 157–174. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, M.; El-Sheekh, M.; Ma, Y.; Pugazhendhi, A.; Natarajan, D.; Kandasamy, G.; Sathiyan, G.; Geetha, R.; Paulraj, B.; Liu, G.; et al. Current status of microbes involved in the degradation of pharmaceutical and personal care products (PPCPs) pollutants in the aquatic ecosystem. Environ. Pollut. 2022, 300, 118922. [Google Scholar] [CrossRef]
- Rastogi, A.; Tiwari, M.K. Microbial Degradation of Pharmaceuticals. In New Trends in Emerging Environmental Contaminants. Energy, Environment, and Sustainability; Singh, S.P., Agarwal, A.K., Gupta, T., Maliyekkal, S.M., Eds.; Springer: Singapore, 2022; pp. 183–210. [Google Scholar] [CrossRef]
- Magesh, N.; Annam Renita, A.; Senthil Kumar, P. Practice on treating pharmaceutical compounds (antibiotics) present in wastewater using biosorption techniques with different biowaste compounds. A review. Environ. Prog. Sustain. Energy 2020, 39, e13429. [Google Scholar] [CrossRef]
- Torres, E. Biosorption: A review of the latest advances. Processes 2020, 8, 1584. [Google Scholar] [CrossRef]
- Rehman, M.U.; Taj, M.B.; Carabineiro, S.A. Biogenic adsorbents for removal of drugs and dyes: A comprehensive review on properties, modification and applications. Chemosphere 2023, 338, 139477. [Google Scholar] [CrossRef]
- Filote, C.; Roșca, M.; Hlihor, R.M.; Cozma, P.; Simion, I.M.; Apostol, M.; Gavrilescu, M. Sustainable application of biosorption and bioaccumulation of persistent pollutants in wastewater treatment: Current practice. Processes 2021, 9, 1696. [Google Scholar] [CrossRef]
- Yaashikaa, P.R.; Kumar, P.S.; Saravanan, A.; Vo, D.V.N. Advances in biosorbents for removal of environmental pollutants: A review on pretreatment, removal mechanism and future outlook. J. Hazard. Mater. 2021, 420, 126596. [Google Scholar] [CrossRef]
- Ramesh, B.; Saravanan, A.; Kumar, P.S.; Yaashikaa, P.R.; Thamarai, P.; Shaji, A.; Rangasamy, G. A review on algae biosorption for the removal of hazardous pollutants from wastewater: Limiting factors, prospects and recommendations. Environ. Pollut. 2023, 327, 121572. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Park, Y.K. Applications of modified biochar-based materials for the removal of environment pollutants: A mini review. Sustainability 2020, 12, 6112. [Google Scholar] [CrossRef]
- Liang, L.; Xi, F.; Tan, W.; Meng, X.; Hu, B.; Wang, X. Review of organic and inorganic pollutants removal by biochar and biochar-based composites. Biochar 2021, 3, 255–281. [Google Scholar] [CrossRef]
- Eduardo, S.R.; Dávila-Guzmán, N.E.; Margarita, L.C. Agro-wastes as precursors of biochar, a cleaner adsorbent to remove pollutants from aqueous solutions. In Biomass-Derived Materials for Environmental Applications; Anastopoulos, I., Lima, E., Meili, L., Giannakoudakis, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 349–376. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, B.; Theng, B.K.; Lee, X.; Zhang, X.; Chen, M.; Xu, P. Removal performance, mechanisms, and influencing factors of biochar for air pollutants: A critical review. Biochar 2022, 4, 30. [Google Scholar] [CrossRef]
- Pouretedal, H.R.; Sadegh, N. Effective removal of amoxicillin, cephalexin, tetracycline and penicillin G from aqueous solutions using activated carbon nanoparticles prepared from vine wood. J. Water Process Eng. 2014, 1, 64–73. [Google Scholar] [CrossRef]
- Bednárek, J.; Matějová, L.; Koutník, I.; Vráblová, M.; Cruz, G.J.F.; Strašák, T.; Šiler, P.; Hrbáč, J. Revelation of high-adsorption-performance activated carbon for removal of fluoroquinolone antibiotics from water. Biomass Convers. Biorefin. 2022, 14, 2585–2599. [Google Scholar] [CrossRef]
- Tan, X.F.; Liu, S.B.; Liu, Y.G.; Gu, Y.L.; Zeng, G.M.; Hu, X.J.; Wang, X.; Liu, S.H.; Jiang, L.H. Biochar as potential sustainable precursors for activated carbon production: Multiple applications in environmental protection and energy storage. Bioresour. Technol. 2017, 227, 359–372. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Preparation, modification and environmental application of biochar: A review. J. Clean. Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Panwar, N.L.; Pawar, A. Influence of activation conditions on the physicochemical properties of activated biochar: A review. Biomass Convers. Biorefin. 2020, 12, 925–947. [Google Scholar] [CrossRef]
- Zbair, M.; Ahsaine, H.A.; Anfar, Z. Porous carbon by microwave assisted pyrolysis: An effective and low-cost adsorbent for sulfamethoxazole adsorption and optimization using response surface methodology. J. Clean. Prod. 2018, 202, 571–581. [Google Scholar] [CrossRef]
- Kemmou, L.; Frontistis, Z.; Vakros, J.; Manariotis, I.D.; Mantzavinos, D. Degradation of antibiotic sulfamethoxazole by biochar-activated persulfate: Factors affecting the activation and degradation processes. Catal. Today 2018, 313, 128–133. [Google Scholar] [CrossRef]
- Lima, D.R.; Lima, E.C.; Umpierres, C.S.; Thue, P.S.; El-Chaghaby, G.A.; da Silva, R.S.; Pavan, F.A.; Dias, S.L.P.; Biron, C. Removal of amoxicillin from simulated hospital effluents by adsorption using activated carbons prepared from capsules of cashew of Para. Environ. Sci. Pollut. Res. 2019, 26, 16396–16408. [Google Scholar] [CrossRef] [PubMed]
- Ezekoye, O.M.; Akpomie, K.G.; Eze, S.I.; Chukwujindu, C.N.; Ani, J.U.; Ujam, O.T. Biosorptive interaction of alkaline modified Dialium guineense seed powders with ciprofloxacin in contaminated solution: Central composite, kinetics, isotherm, thermodynamics, and desorption. Int. J. Phytoremed. 2020, 22, 1028–1037. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.A.; Ali, F.J. Equilibrium, thermodynamics and kinetics study of doxycycline adsorption from aqueous solution using spent black tea leaves and pomegranate peel wastes. Int. J. Dev. Res. 2014, 4, 129–135. [Google Scholar]
- Burdová, H.; Brázová, V.; Kwoczynski, Z.; Snow, J.; Trögl, J.; Kříženecká, S. Miscanthus x giganteus biochar: Effective adsorption of pharmaceuticals from model solution and hospital wastewater. J. Clean. Prod. 2024, 460, 142545. [Google Scholar] [CrossRef]
- Romero-Hernandez, J.J.; Paredes-Laverde, M.; Silva-Agredo, J.; Mercado, D.F.; Ávila-Torres, Y.; Torres-Palma, R.A. Pharmaceutical adsorption on NaOH-treated rice husk-based activated carbons: Kinetics, thermodynamics, and mechanisms. J. Clean. Prod. 2024, 434, 139935. [Google Scholar] [CrossRef]
- Peñafiel, M.E.; Flores, D. Competitive adsorption of drugs from a multi-component mixture on sugarcane bagasse. Water 2023, 15, 2127. [Google Scholar] [CrossRef]
- Guérin, T.; Ghinet, A.; Hossart, M.; Waterlot, C. Wheat and ryegrass biomass ashes as effective sorbents for metallic and organic pollutants from contaminated water in lab-engineered cartridge filtration system. Bioresour. Technol. 2020, 318, 124044. [Google Scholar] [CrossRef]
- Zeng, S.; Kan, E. Chemical activation of forage grass-derived biochar for treatment of aqueous antibiotic sulfamethoxazole. ACS Omega 2020, 5, 13793–13801. [Google Scholar] [CrossRef]
- Teixeira, S.; Delerue-Matos, C.; Santos, L. Application of experimental design methodology to optimize antibiotics removal by walnut shell based activated carbon. Sci. Total Environ. 2019, 646, 168–176. [Google Scholar] [CrossRef]
- Yao, Y.; Zhang, Y.; Gao, B.; Chen, R.; Wu, F. Removal of sulfamethoxazole (SMX) and sulfapyridine (SPY) from aqueous solutions by biochars derived from anaerobically digested bagasse. Environ. Sci. Pollut. Res. 2018, 25, 25659–25667. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W.; Johir, M.A.H.; Sornalingam, K. Single and competitive sorption properties and mechanism of functionalized biochar for removing sulfonamide antibiotics from water. Chem. Eng. J. 2016, 311, 348–358. [Google Scholar] [CrossRef]
- Shimabuku, K.K.; Kearns, J.P.; Martinez, J.E.; Mahoney, R.B.; Moreno-Vasquez, L.; Summers, R.S. Biochar sorbents for sulfamethoxazole removal from surface water, stormwater, and wastewater effluent. Water Res. 2016, 96, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Wang, Z.; Zhao, J.; Herbert, S.; Xing, B. Sorption of antibiotic sulfamethoxazole varies with biochars produced at different temperatures. Environ. Pollut. 2013, 181, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Al-Sareji, O.J.; Grmasha, R.A.; Meiczinger, M.; Al-Juboori, R.A.; Somogyi, V.; Stenger-Kovács, C.; Hashim, K.S. A sustainable and highly efficient fossil-free carbon from olive stones for emerging contaminants removal from different water matrices. Chemosphere 2024, 351, 141189. [Google Scholar] [CrossRef] [PubMed]
- Meseguer, V.F.; Ortuño, J.F.; Aguilar, M.I.; Lloréns, M.; Pérez-Marín, A.B.; Fuentes, E. Ciprofloxacin Uptake from an Aqueous Solution via Adsorption with K2CO3-Activated Biochar Derived from Brewing Industry Bagasse. Processes 2024, 12, 199. [Google Scholar] [CrossRef]
- Fan, H.; Ma, Y.; Wan, J.; Wang, Y.; Li, Z.; Chen, Y. Adsorption properties and mechanisms of novel biomaterials from banyan aerial roots via simple modification for ciprofloxacin removal. Sci. Total Environ. 2019, 708, 134630. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.W.; Tan, X.F.; Liu, Y.G.; Tian, S.R.; Zeng, G.M.; Jiang, L.H.; Liu, S.B.; Li, J.; Liu, N.; Yin, Z.H. Comprehensive adsorption studies of doxycycline and ciprofloxacin antibiotics by biochars prepared at different temperatures. Front. Chem. 2018, 6, 80. [Google Scholar] [CrossRef]
- El-Shafey, E.S.I.; Al-Lawati, H.; Al-Sumri, A.S. Ciprofloxacin adsorption from aqueous solution onto chemically prepared carbon from date palm leaflets. J. Environ. Sci. 2012, 24, 1579–1586. [Google Scholar] [CrossRef]
- Jang, H.M.; Kan, E. A novel hay-derived biochar for removal of tetracyclines in water. Bioresour. Technol. 2019, 274, 162–172. [Google Scholar] [CrossRef]
- Álvarez-Torrellas, S.; Rodríguez, A.; Ovejero, G.; García, J. Comparative adsorption performance of ibuprofen and tetracycline from aqueous solution by carbonaceous materials. Chem. Eng. J. 2016, 283, 936–947. [Google Scholar] [CrossRef]
- Marzbali, M.H.; Esmaieli, M.; Abolghasemi, H.; Marzbali, M.H. Tetracycline adsorption by H3PO4-activated carbon produced from apricot nut shells: A batch study. Process Saf. Environ. Prot. 2016, 102, 700–709. [Google Scholar] [CrossRef]
- Hassan, S.A.; Ali, F.J. Usability study of spent black tea leaves and pomegranate peel in adsorption of tetracycline hydrochloride antibiotic. Int. J. Curr. Res. 2013, 5, 2530–2538. [Google Scholar]
- Liao, P.; Zhan, Z.; Dai, J.; Wu, X.; Zhang, W.; Wang, K.; Yuan, S. Adsorption of tetracycline and chloramphenicol in aqueous solutions by bamboo charcoal: A batch and fixed-bed column study. Chem. Eng. J. 2013, 228, 496–505. [Google Scholar] [CrossRef]
- Al-Sareji, O.J.; Grmasha, R.A.; Meiczinger, M.; Al-Juboori, R.A.; Somogyi, V.; Hashim, K.S. A Sustainable Banana Peel Activated Carbon for Removing Pharmaceutical Pollutants from Different Waters: Production, Characterization, and Application. Materials 2024, 17, 1032. [Google Scholar] [CrossRef]
- Varela, C.F.; Moreno-Aldana, L.C.; Agámez-Pertuz, Y.Y. Adsorption of pharmaceutical pollutants on ZnCl2-activated biochar from corn cob: Efficiency, selectivity and mechanism. J. Bioresour. Bioprod. 2024, 9, 58–73. [Google Scholar] [CrossRef]
- Limousy, L.; Ghouma, I.; Ouederni, A.; Jeguirim, M. Amoxicillin removal from aqueous solution using activated carbon prepared by chemical activation of olive stone. Environ. Sci. Pollut. Res. 2016, 24, 9993–10004. [Google Scholar] [CrossRef] [PubMed]
- Paredes-Laverde, M.; Silva-Agredo, J.; Torres-Palma, R.A. Removal of norfloxacin in deionized, municipal water and urine using rice (Oryza sativa) and coffee (Coffea arabica) husk wastes as natural adsorbents. J. Environ. Manag. 2018, 213, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, Z.; Xie, X.; Zhu, J.; Li, R.; Qin, T. Removal of Norfloxacin from aqueous solution by clay-biochar composite prepared from potato stem and natural attapulgite. Colloids Surf. A Physicochem. Eng. Asp. 2017, 514, 126–136. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, J.; Wu, J.; Liu, Q.; Zhang, H.; Jin, S. Adsorptive removal of fluoroquinolone antibiotics using bamboo biochar. Sustainability 2015, 7, 12947–12957. [Google Scholar] [CrossRef]
- Wuana, R.A.; Sha’Ato, R.; Iorhen, S. Aqueous phase removal of ofloxacin using adsorbents from Moringa oleifera pod husks. Adv. Environ. Res. 2015, 4, 49–68. [Google Scholar] [CrossRef]
- Kozyatnyk, I.; Oesterle, P.; Wurzer, C.; Mašek, O.; Jansson, S. Removal of contaminants of emerging concern from multicomponent systems using carbon dioxide activated biochar from lignocellulosic feedstocks. Bioresour. Technol. 2021, 340, 125561. [Google Scholar] [CrossRef] [PubMed]
- Schneider, L.T.; Staudt, J.; Borba, C.E.; Scheufele, F.B.; Nivaldo, A. Produção e Aplicação de Carvões de Resíduo de Fecularia na Remoção de Metronidazol em Solução. 2° Cogresso Sul-Americano de Residuos Sólidos e Sustentabilidade, Foz do Lguaçu/PR, 2019; pp. 1–5. Available online: https://www.ibeas.org.br/conresol/conresol2019/I-048.pdf (accessed on 20 August 2024).
- Patil, M.; Singh, S.; Kumari, D.; Daverey, A.; Dutta, K. Adsorption of azithromycin antibiotic from water onto biochar derived from Terminalia chebula and sugarcane bagasse. Water Pract. Technol. 2024, 19, 2973–2990. [Google Scholar] [CrossRef]
- Escudero-Curiel, S.; Pazos, M.; Sanromán, A. Facile one-step synthesis of a versatile nitrogen-doped hydrochar from olive oil production waste, “alperujo”, for removing pharmaceuticals from wastewater. Environ. Pollut. 2023, 330, 121751. [Google Scholar] [CrossRef] [PubMed]
- Aslan, S.; Şirazi, M. Adsorption of sulfonamide antibiotic onto activated carbon prepared from an agro-industrial by-product as low-cost adsorbent: Equilibrium, thermodynamic, and kinetic studies. Water Air Soil Pollut. 2020, 231, 222. [Google Scholar] [CrossRef]
- Cheng, C.; Zhang, J.; Zhang, C.; Liu, H.; Liu, W. Preparation and characterization of charcoal from feathers and its application in trimethoprim adsorption. Desalination Water Treat. 2014, 52, 5401–5412. [Google Scholar] [CrossRef]
- Baccar, R.; Sarrà, M.; Bouzid, J.; Feki, M.; Blánquez, P. Removal of pharmaceutical compounds by activated carbon prepared from agricultural by-product. Chem. Eng. J. 2012, 211, 310–317. [Google Scholar] [CrossRef]
- Guimarães, T.G.; Barros, L.A.; Silva, R.S.; Gonzalez, M.H.; Carrilho, E.N.; Labuto, G. Synthesis and characterization of biochars modified with natural deep eutectic solvent (NADES) for dipyrone removal from aqueous medium. Sustain. Chem. Pharm. 2023, 35, 101205. [Google Scholar] [CrossRef]
- Mokhtaryan, S.; Khodabakhshi, A.; Sadeghi, R.; Nourmoradi, H.; Shakeri, K.; Hemati, S.; Mohammadi-Moghadam, F. New activated carbon derived from Gundelia tournefortii seeds for effective removal of acetaminophen from aqueous solutions: Adsorption performance. Arab. J. Chem. 2023, 16, 105253. [Google Scholar] [CrossRef]
- De Luna, M.D.G.; Budianta, W.; Rivera, K.K.P.; Arazo, R.O. Removal of sodium diclofenac from aqueous solution by adsorbents derived from cocoa pod husks. J. Environ. Chem. Eng. 2017, 5, 1465–1474. [Google Scholar] [CrossRef]
- Bano, A.; Aziz, M.K.; Mishra, R.; Dave, H.; Prasad, B.; Kumari, M.; Dubey, D.; Meili, L.; Shah, M.P.; Prasad, K.S. Response surface methodology–based optimisation of adsorption of diclofenac and treatment of pharmaceutical effluent using combined coagulation-adsorption onto nFe2O3 decorated water chestnut shells biochar. Environ. Sci. Pollut. Res. 2024, 31, 55317–55335. [Google Scholar] [CrossRef] [PubMed]
- Grefa, D.D.A.; Sánchez, J.E.G.; Sánchez, L.R.B.; Alfonso, M.S.P.; Tagle, M.E.V. Rice husk ash as sorbent for solid phase extraction of diclofenac, ibuprofen and carboplatin residues from waters. Microchem. J. 2023, 195, 109361. [Google Scholar] [CrossRef]
- Correa-Navarro, Y.M.; Giraldo, L.; Moreno-Piraján, J.C. Biochar from fique bagasse for remotion of caffeine and diclofenac from aqueous solution. Molecules 2020, 25, 1849. [Google Scholar] [CrossRef] [PubMed]
- Lonappan, L.; Rouissi, T.; Liu, Y.; Brar, S.K.; Surampalli, R.Y. Removal of diclofenac using microbiochar fixed-bed column bioreactor. J. Environ. Chem. Eng. 2019, 7, 102894. [Google Scholar] [CrossRef]
- Araujo, L.A.; Bezerra, C.O.; Cusioli, L.F.; Silva, M.F.; Nishi, L.; Gomes, R.G.; Bergamasco, R. Moringa oleifera biomass residue for the removal of pharmaceuticals from water. J. Environ. Chem. Eng. 2018, 6, 7192–7199. [Google Scholar] [CrossRef]
- Lonappan, L.; Rouissi, T.; Brar, S.K.; Verma, M.; Surampalli, R.Y. An insight into the adsorption of diclofenac on different biochars: Mechanisms, surface chemistry, and thermodynamics. Bioresour. Technol. 2018, 249, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Abbas, G.; Javed, I.; Iqbal, M.; Haider, R.; Hussain, F.; Qureshi, N. Adsorption of non-steroidal anti-inflammatory drugs (diclofenac and ibuprofen) from aqueous medium onto activated onion skin. Desalination Water Treat. 2017, 95, 274–285. [Google Scholar] [CrossRef]
- Pires, B.C.; Dutra, F.V.A.; Nascimento, T.A.; Borges, K.B. Preparation of PPy/cellulose fibre as an effective potassium diclofenac adsorbent. React. Funct. Polym. 2017, 113, 40–49. [Google Scholar] [CrossRef]
- Bernardo, M.; Rodrigues, S.; Lapa, N.; Matos, I.; Lemos, F.; Batista, M.K.S.; Carvalho, A.P.; Fonseca, I. High efficacy on diclofenac removal by activated carbon produced from potato peel waste. Int. J. Environ. Sci. Technol. 2016, 13, 1989–2000. [Google Scholar] [CrossRef]
- dos Santos, D.; Moreira, W.; de Araújo, T.; Bernardo, M.; Fonseca, I.; Ostroski, I.; de Barros, M.A. Removal of Emerging Contaminants from Water by Using Carbon Materials Derived from Tingui Shells. Separations 2024, 11, 215. [Google Scholar] [CrossRef]
- Melliti, A.; Touihri, M.; Kofroňová, J.; Hannachi, C.; Sellaoui, L.; Bonilla-Petriciolet, A.; Vurm, R. Sustainable removal of caffeine and acetaminophen from water using biomass waste-derived activated carbon: Synthesis, characterization, and modelling. Chemosphere 2024, 355, 141787. [Google Scholar] [CrossRef] [PubMed]
- Solmaz, A.; Turna, T.; Baran, A. Removal of paracetamol from aqueous solution with zinc oxide nanoparticles obtained by green synthesis from purple basil (Ocimum basilicum L.) waste. Biomass Convers. Biorefin. 2024, 14, 10771–10789. [Google Scholar] [CrossRef]
- Ferreira, R.C.; de Araújo, T.P.; Dias, D.; Bernardo, M.; Lapa, N.; Fonseca, I.M.; de Barros, M.A. Removal of paracetamol and Cu2+ from water by using porous carbons derived from agrowastes. Processes 2023, 11, 2146. [Google Scholar] [CrossRef]
- Preglo, A.R.; Namata, J.; Caculba, J.; Sanchez, G.; Joyno, C.; Pagalan, E.; Arazo, R.O. Paracetamol removal from aqueous solution through activated carbon from mango seeds. Chem. Afr. 2023, 6, 699–710. [Google Scholar] [CrossRef]
- Georgin, J.; Franco, D.S.; Netto, M.S.; Manzar, M.S.; Zubair, M.; Meili, L.; Piccillo, D.G.A.; Silva, L.F. Adsorption of the Paracetamol First-Line Covid Treatment Drug onto Activated Carbon from Residual Pods of Erythrina Speciosa. SSRN 2022. to be submitted. [Google Scholar] [CrossRef]
- Spessato, L.; Bedin, K.C.; Cazetta, A.L.; Souza, I.P.; Duarte, V.A.; Crespo, L.H.; Silva, M.C.; Pontes, R.M.; Almeida, V.C. KOH-super activated carbon from biomass waste: Insights into the paracetamol adsorption mechanism and thermal regeneration cycles. J. Hazard. Mater. 2019, 371, 499–505. [Google Scholar] [CrossRef]
- Nourmoradi, H.; Moghadam, K.F.; Jafari, A.; Kamarehie, B. Removal of acetaminophen and ibuprofen from aqueous solutions by activated carbon derived from Quercus Brantii (Oak) acorn as a low-cost biosorbent. J. Environ. Chem. Eng. 2018, 6, 6807–6815. [Google Scholar] [CrossRef]
- Parmar, S.; Sharma, S.; Sharma, A.; Verma, S. Removal of Acetaminophen from Waste Water using Low Cost Adsorbent. Methods 2018, 5, 1025–1031. [Google Scholar]
- García-Mateos, F.J.; Ruiz-Rosas, R.; Marqués, M.D.; Cotoruelo, L.M.; Rodríguez-Mirasol, J.; Cordero, T. Removal of paracetamol on biomass-derived activated carbon: Modeling the fixed bed breakthrough curves using batch adsorption experiments. Chem. Eng. J. 2015, 279, 18–30. [Google Scholar] [CrossRef]
- Raupp, Í.N.; Filho, A.V.; Arim, A.L.; Muniz, A.R.C.; da Rosa, G.S. Ibuprofen adsorption onto olive pomace activated carbon. Chem. Eng. Technol. 2023, 46, 2395–2403. [Google Scholar] [CrossRef]
- Thabede, P.M.; Mtunzi, F.; Nyamukamba, P. Sorption behaviour of ibuprofen using activated carbon derived from rose geranium (Pelargonium graveolens L.) leaves. Appl. Sci. 2023, 13, 5133. [Google Scholar] [CrossRef]
- Bello, O.S.; Alao, O.C.; Alagbada, T.C.; Olatunde, A.M. Biosorption of ibuprofen using functionalized bean husks. Sustain. Chem. Pharm. 2019, 13, 100151. [Google Scholar] [CrossRef]
- Chakraborty, P.; Banerjee, S.; Kumar, S.; Sadhukhan, S.; Halder, G. Elucidation of ibuprofen uptake capability of raw and steam activated biochar of Aegle marmelos shell: Isotherm, kinetics, thermodynamics and cost estimation. Process Saf. Environ. Prot. 2018, 118, 10–23. [Google Scholar] [CrossRef]
- Mondal, S.; Bobde, K.; Aikat, K.; Halder, G. Biosorptive uptake of ibuprofen by steam activated biochar derived from mung bean husk: Equilibrium, kinetics, thermodynamics, modeling and eco-toxicological studies. J. Environ. Manag. 2016, 182, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Essandoh, M.; Kunwar, B.; Pittman, C.U., Jr.; Mohan, D.; Mlsna, T. Sorptive removal of salicylic acid and ibuprofen from aqueous solutions using pine wood fast pyrolysis biochar. Chem. Eng. J. 2015, 265, 219–227. [Google Scholar] [CrossRef]
- Calisto, V.; Jaria, G.; Silva, C.P.; Ferreira, C.I.; Otero, M.; Esteves, V.I. Single and multi-component adsorption of psychiatric pharmaceuticals onto alternative and commercial carbons. J. Environ. Manag. 2017, 192, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Xie, S.; Chen, C.; Quan, H.; Hua, L.; Luo, X.; Guo, L. Activated biochar derived from pomelo peel as a high-capacity sorbent for removal of carbamazepine from aqueous solution. RSC Adv. 2017, 7, 54969–54979. [Google Scholar] [CrossRef]
- Naghdi, M.; Taheran, M.; Pulicharla, R.; Rouissi, T.; Brar, S.K.; Verma, M.; Surampalli, R.Y. Pine-wood derived nanobiochar for removal of carbamazepine from aqueous media: Adsorption behavior and influential parameters. Arab. J. Chem. 2016, 12, 5292–5301. [Google Scholar] [CrossRef]
- Torrellas, S.Á.; Lovera, R.G.; Escalona, N.; Sepúlveda, C.; Sotelo, J.L.; García, J. Chemical-activated carbons from peach stones for the adsorption of emerging contaminants in aqueous solutions. Chem. Eng. J. 2015, 279, 788–798. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, X.; Chen, X.; Dai, C.; Zhang, J.; Zhang, Y. Biosorption of clofibric acid and carbamazepine in aqueous solution by agricultural waste rice straw. J. Environ. Sci. 2013, 25, 2384–2395. [Google Scholar] [CrossRef]
- Ribeiro, A.C.; Januário, E.F.D.; Vidovix, T.B.; Vieira, A.M.S.; Bergamasco, R. Synthesis of a novel functionalized biosorbent from mango stone and its application in the pharmaceutical’s removal from water and a synthetic mixture. Chemosphere 2024, 346, 140520. [Google Scholar] [CrossRef] [PubMed]
- Mukarunyana, B.; Boman, C.; Kabera, T.; Lindgren, R.; Fick, J. The ability of biochars from cookstoves to remove pharmaceuticals and personal care products from hospital wastewater. Environ. Technol. Innov. 2023, 32, 103391. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Deliyanni, E.A. Modified activated carbons from potato peels as green environmental-friendly adsorbents for the treatment of pharmaceutical effluents. Chem. Eng. Res. Des. 2015, 97, 135–144. [Google Scholar] [CrossRef]
- de Moura, A.A.; Straioto, H.; Martins, W.M.; de Araújo, T.P.; Diório, A.; Bergamasco, R.; Moises, M.P.; Barros, M.A.S.D. Synthesis of low-cost hydrochar using agro-industrial residues for the removal of pharmaceutical drug chloroquine. Desalination Water Treat. 2023, 306, 178–188. [Google Scholar] [CrossRef]
- Zekkaoui, C.; Berrama, T.; Kadmi, Y.; Souames, S.; Dadou, S.; Beriber, A. Insights into the efficient elimination of veterinary drug from water by adsorption on activated carbon: Optimization study. Int. J. Environ. Sci. Technol. 2021, 18, 3635–3650. [Google Scholar] [CrossRef]
- Procópio, A.M.D.S.; Andrade, F.V.D.; Silva, F.S.; de Andrade, S.J. Removal of 17 α-ethinylestradiol (EE2) from aqueous solutions by peanut shells (A rachis hypogaea): Adsorption kinetic, isothermal, and thermodynamic studies. Water Pract. Technol. 2023, 18, 2543–2560. [Google Scholar] [CrossRef]
- Amaral, M.A.F.; Fusinato, M.D.; da Cunha, M.E.; Coll, J.P.R.; Lacerda, J.H.G.; Sanches Filho, P.J. Evaluation of the Adsorbent Potential of Biochar obtained by Pyrolysis to Remove Emerging Contaminants. Braz. J. Anal. Chem. 2023, 10, 80–89. [Google Scholar] [CrossRef]
- Ferandin, H.J.; Marcia, T.V.; Ramos, S.P.Y.; Ferri, P.C.; Sloboda, R.E.; Granhen, T.C.R. Adsorption of naturals hormones estrone, 17β-estradiol, and estriol by rice husk: Monocomponent and multicomponent kinetics and equilibrium. Environ. Technol. 2018, 41, 1075–1092. [Google Scholar] [CrossRef]
- Londero, M.C.; Pavoski, G.; Espinosa, D.C.R.; de Vasconcellos, N.J.S.; da Silva, W.L. Potential application of alternative materials for organic pollutant removal. Water Air Soil Pollut. 2022, 233, 65. [Google Scholar] [CrossRef]
- Cusioli, L.F.; Mantovani, D.; Bergamasco, R.; Tusset, A.M.; Lenzi, G.G. Preparation of a New Adsorbent Material from Agro-Industrial Waste and Comparison with Commercial Adsorbent for Emerging Contaminant Removal. Processes 2023, 11, 2478. [Google Scholar] [CrossRef]
- Mondal, S.; Sinha, K.; Aikat, K.; Halder, G. Adsorption thermodynamics and kinetics of ranitidine hydrochloride onto superheated steam activated carbon derived from mung bean husk. J. Environ. Chem. Eng. 2015, 3, 187–195. [Google Scholar] [CrossRef]
- Ronda, A.; Calero, M.; Blázquez, G.; Pérez, A.; Martín-Lara, M.A. Optimization of the use of a biosorbent to remove heavy metals: Regeneration and reuse of exhausted biosorbent. J. Taiwan Inst. Chem. Eng. 2015, 51, 109–118. [Google Scholar] [CrossRef]
- Bădescu, I.S.; Bulgariu, D.; Ahmad, I.; Bulgariu, L. Valorisation possibilities of exhausted biosorbents loaded with metal ions–a review. J. Environ. Manag. 2018, 224, 288–297. [Google Scholar] [CrossRef]
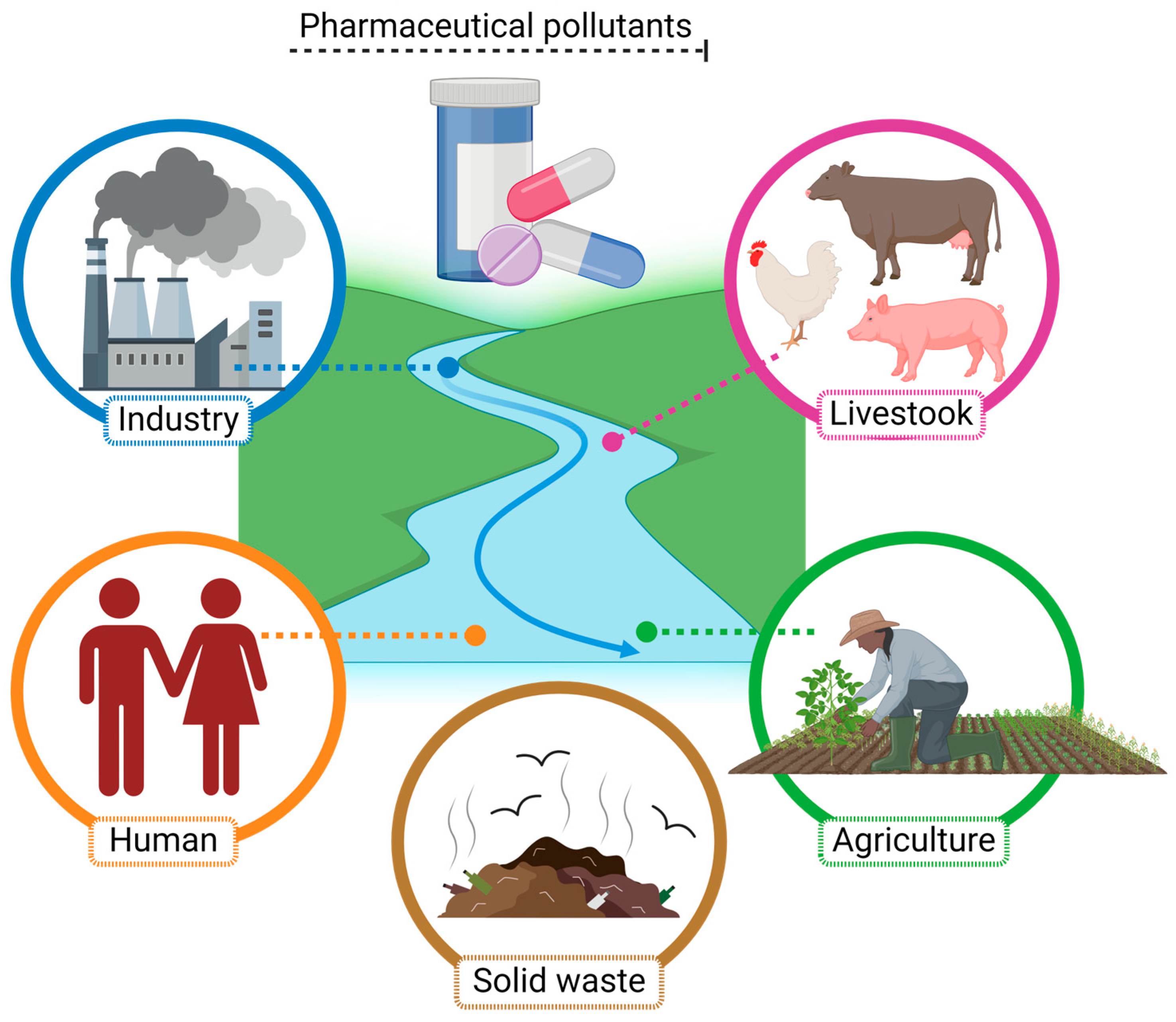
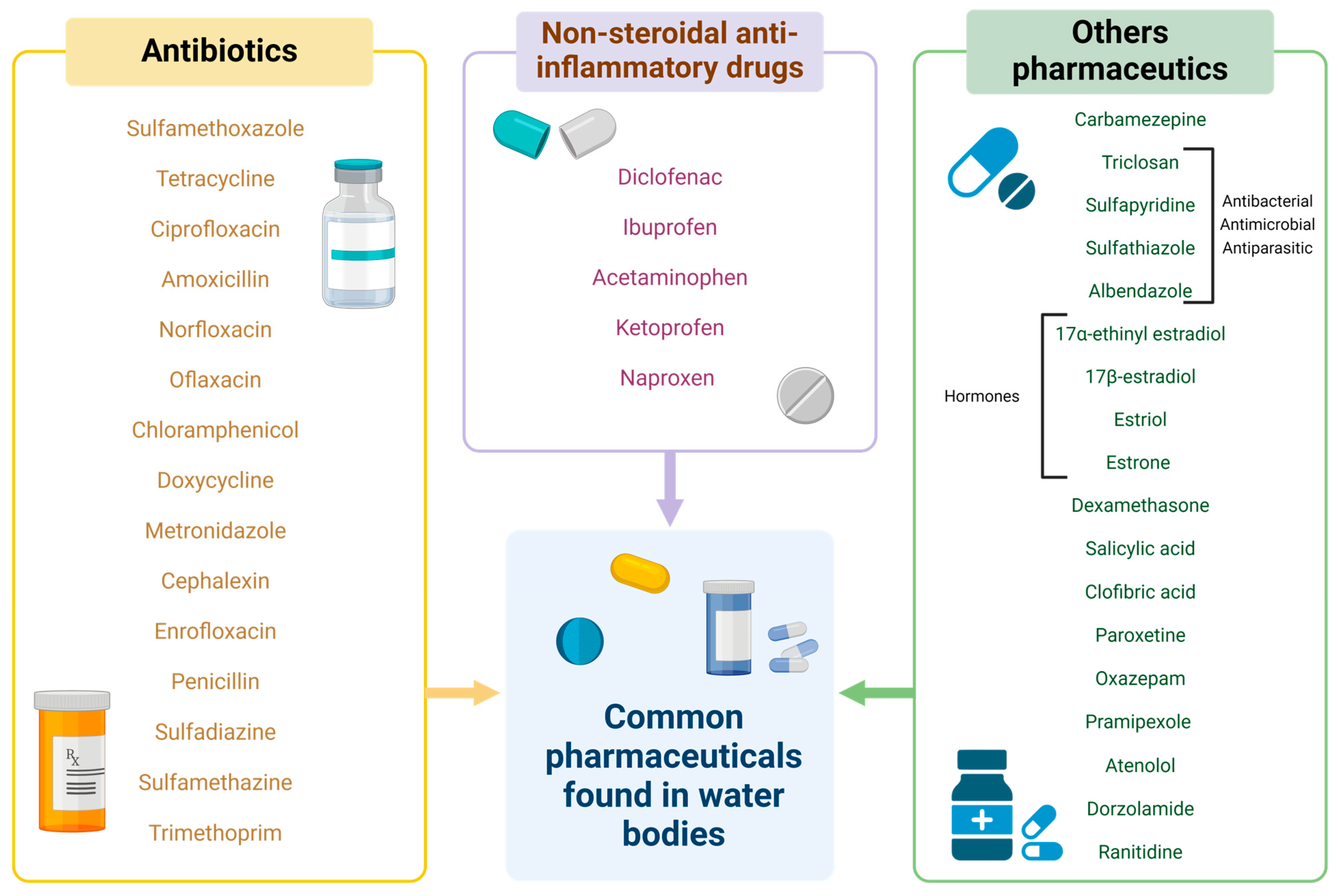
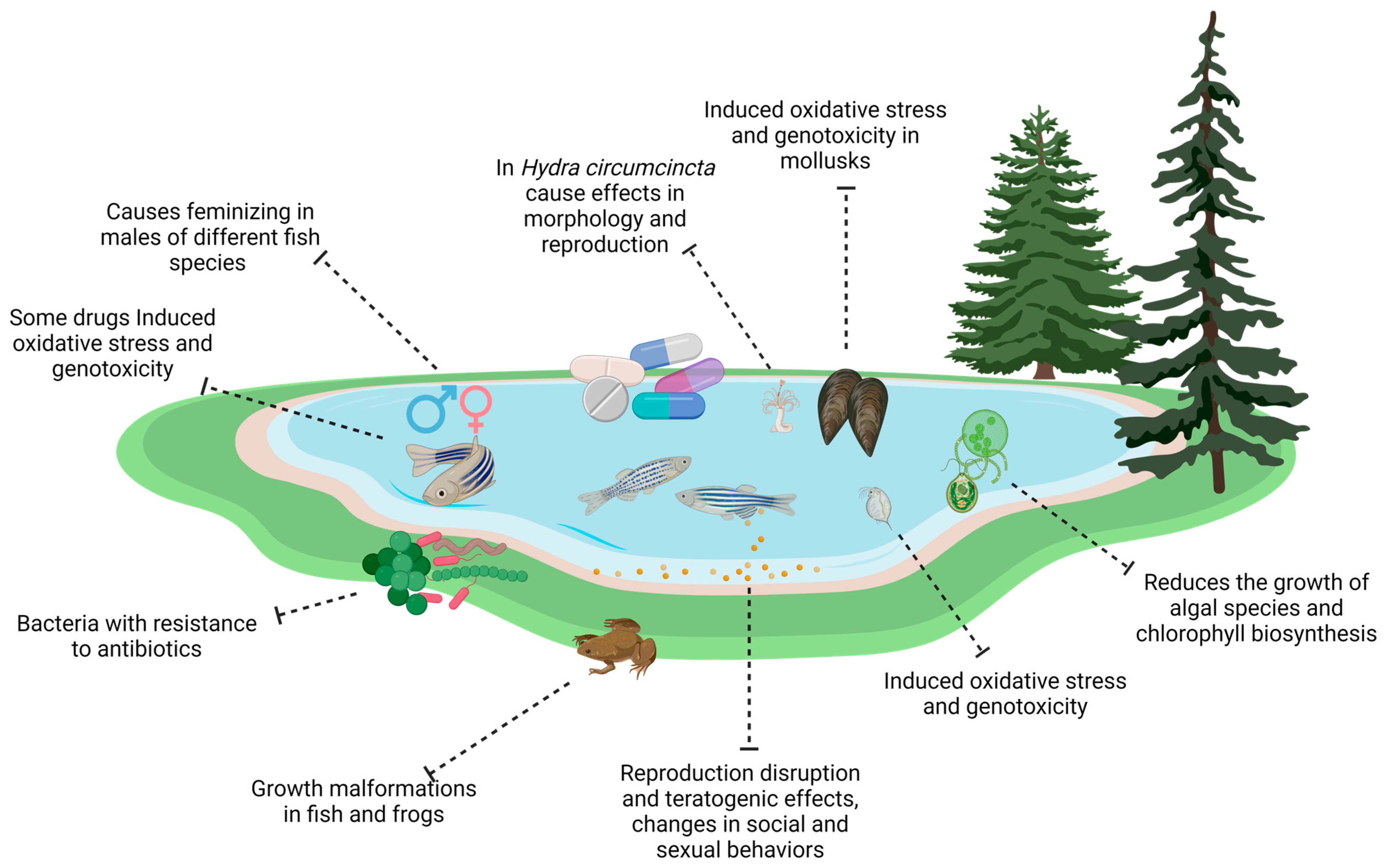
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estrada-Almeida, A.G.; Castrejón-Godínez, M.L.; Mussali-Galante, P.; Tovar-Sánchez, E.; Rodríguez, A. Pharmaceutical Pollutants: Ecotoxicological Impacts and the Use of Agro-Industrial Waste for Their Removal from Aquatic Environments. J. Xenobiot. 2024, 14, 1465-1518. https://doi.org/10.3390/jox14040082
Estrada-Almeida AG, Castrejón-Godínez ML, Mussali-Galante P, Tovar-Sánchez E, Rodríguez A. Pharmaceutical Pollutants: Ecotoxicological Impacts and the Use of Agro-Industrial Waste for Their Removal from Aquatic Environments. Journal of Xenobiotics. 2024; 14(4):1465-1518. https://doi.org/10.3390/jox14040082
Chicago/Turabian StyleEstrada-Almeida, Ana Gabriela, María Luisa Castrejón-Godínez, Patricia Mussali-Galante, Efraín Tovar-Sánchez, and Alexis Rodríguez. 2024. "Pharmaceutical Pollutants: Ecotoxicological Impacts and the Use of Agro-Industrial Waste for Their Removal from Aquatic Environments" Journal of Xenobiotics 14, no. 4: 1465-1518. https://doi.org/10.3390/jox14040082
APA StyleEstrada-Almeida, A. G., Castrejón-Godínez, M. L., Mussali-Galante, P., Tovar-Sánchez, E., & Rodríguez, A. (2024). Pharmaceutical Pollutants: Ecotoxicological Impacts and the Use of Agro-Industrial Waste for Their Removal from Aquatic Environments. Journal of Xenobiotics, 14(4), 1465-1518. https://doi.org/10.3390/jox14040082










