Abstract
A long-term, sustainable solution to weed infestation is extremely desirable because weeds have the potential to reduce crop productivity and the aesthetic appeal of the environment. In this study, the impacts of mowing and varying degrees of trampling pressure on the suppression of weeds, alongside wound-induced changes in the allelopathic potential, of the rhizosphere soil and the root exudates of southern crabgrass (Digitaria ciliaris) and Asian flatsedge (Cyperus microiria) were evaluated under both field and greenhouse conditions. The field study results showed that all trampling treatments induced the relative suppression of weed growth. Grass weeds showed higher resistance to trampling than broad-leaved weeds. However, laboratory bioassays showed that light trampling caused a significant increase in the growth-inhibitory effects of southern crabgrass rhizosphere soil on lettuce. Moreover, mowing (9.11% of control) and trampling (16.4% of control) resulted in a marginal increase in the growth-inhibitory effects of root exudates released from southern crabgrass. Furthermore, the growth-inhibitory activities of the Asian flatsedge rhizosphere soil were significantly reduced after heavy trampling pressure. Moreover, mowing and trampling resulted in marginal reductions in the growth-inhibitory activities of root exudates released from Asian flatsedge against lettuce (i.e., 18.7% and 28.5%, respectively). In general, mowing and varying degrees of trampling induced contrasting and integrated impacts on weed suppression as well as the allelopathic potential of both southern crabgrass and Asian flatsedge.
1. Introduction
The application of herbicides and mowing to mitigate the challenges of weed infestation often leads to health and environmental problems in the long term [1,2]. For instance, intense or prolonged mowing has been reported to further induce grass weed infestation [3,4], along with severe accidental injury to mowing machine operators [5,6]. In addition, mowing needs a long-standing, hardworking labor force [7]. Moreover, excessive pesticide inputs harm both life on Earth and the whole environment [8]. When humans or animals become directly or indirectly exposed to synthetic agrichemicals for a long period of time, they commonly develop several health conditions, including both respiratory and reproductive impairments, diabetes, neurological disorders, and cancer [9]. Furthermore, the inappropriate use of pesticides pollutes water bodies, interferes with soil health, and results in the development of pesticide-resistant weeds as well [10]. Hence, there is a strong need for alternate weed control techniques to ensure sustainable weed management.
In previous studies, the potential use of allelopathic species has been explored in the control of weeds. The extensive and effective implementation of bioherbicides released directly from allelopathic plants or manufactured indirectly from allelopathic compounds could, in fact, be a better and more sustainable means of strengthening global crop production, along with a reduction in the health and environmental hazards caused by synthetic herbicides [10]. Furthermore, studies have been performed on how mechanical stimulation, including trampling, rolling, and roll chopping, can sustainably suppress weeds’ growth [4,11]. Moreover, studies on the effects of human [12,13], animal [14], and machine trampling [11] on weed control, soil compaction, and vegetation composition have indicated that light treading pressure possesses more desirable impacts on both weed suppression and soil health than intense treading pressure [13,15]. This can be linked to the fact that mechanical stimulation (i.e., touching, cutting, and pressuring), herbivory, and some environmental factors (such as drought and nutrient availability) induce the release of volatile organic compounds (VOCs), such as ethylene, which, depending on its concentration, stimulates or suppresses both growth and senescence in plants [16,17,18]. It also induces transient increases in the root exudation of organic carbon, amino acids, ammonium cations, phenolics, and proteins [19,20,21]. Thus, the desirable weed suppression impacted by light trampling might not only be due to the outcome of the physical top-down pressure on weeds but also a complex process involving the influence of allelochemicals released from touched or wounded plants.
Root exudates are major sources for the direct input of plant chemicals into the rhizosphere, making them one of the most important sources of allelochemicals released into the rhizosphere soil [22]. Both mowing and trampling are long-established methods of weeding that could (along with other mechanical stimulation) hypothetically influence the allelopathic activity and subsequent suppression of weeds in the field. As an example of the enhancement in allelopathic compound release through mechanical means, Yang et al. [23] reported that the use of a mist system on the roots of sorghum (Sorghum bicolor) increased sorgoleone exudation through the induction of abundant root hair production. Sorgoleone is a strong allelochemical as well as a potent bioherbicide produced in the root hairs of sorghum plants [24,25]. Allelochemicals are released into the environment through several routes, including volatilization, leachates, exudation, and decomposition. Specific bioassays have been designed to effectively evaluate the growth-inhibitory effects of compounds released through these routes. These include plant-box [26] and rhizosphere soil [27] methods for root exudates, the dish pack method [28] for volatiles, and the sandwich method [29] for leachates. In this study, the plant-box and rhizosphere soil methods were adopted to assess wound-induced variations in the allelopathic effects of candidate plants.
The objective of this study was to assess the impacts of mowing and varying degrees of intensity of trampling on the suppression of weed growth, along with variations in the allelopathic potential, of both the rhizosphere soil and the root exudates of southern crabgrass (Digitaria ciliaris) and Asian flatsedge (Cyperus microiria). Both Southern crabgrass (annual plant) and Asian flatsedge (perennial plant) are common, widespread, and noxious weeds, and they all aggressively grow in open fields, soybean fields, and both upland and paddy fields [30,31,32]. In addition, biochemical compounds (i.e., veratric acid, maltol, and (−)-loliolide) released in the root exudates of crabgrass (Digitaria sanguinalis), which belongs to the Digitaria family, were reported to inhibit the growth of wheat, maize, and soybean alongside the growth of soil bacteria, actinobacteria, and fungi [33]. On the other hand, several terpenes, including α-cyperone, β-selinene, and α-humulene, were extracted from the tubers and rhizomes of whitehead spikesedge (Cyperus kyllingia), which belongs to the Cyperus family, and they all indicated growth inhibition effects against lettuce seedlings [34]. This study presents the outcome of preliminary research conducted on common weeds as an initial stage of a large, ongoing research project.
2. Materials and Methods
All field and greenhouse experiments, along with laboratory bioassays, were conducted at the Tokyo University of Agriculture and Technology, Saiwai-cho, Fuchu, Tokyo, Japan (35°41′ N, 139°28′ E). Southern crabgrass and Asian flatsedge were selected as candidate species for laboratory bioassays because the two weeds were the most dominant weeds within the field. They also possess stronger stems, which gave them more resistance and allowed the more successful uprooting of their roots from the soil. All the other weeds (i.e., oriental water willow (Justicia procumbens), horsenettle (Solanum carolinense), and giant foxtail (Setaria faberi)) were difficult to pull out of the ground because they had become extremely broken up, particularly after heavy trampling pressure.
In addition, a greenhouse study was carried out in order to evaluate the potential allelopathic effects of the candidate species in a controlled environment. In the field, rainfall (Appendix A) might interfere with the allelopathic potential in the soil by leaching water-soluble allelochemicals into deeper soil profiles [35]. The greenhouse experiment also ensured that the growth-inhibitory effects of only the root exudates from Southern crabgrass and Asian flatsedge were evaluated, and not those from other organisms.
2.1. Planting Conditions and Treatments
This section describes how the weed species were grown in both the field and greenhouse studies, along with how they were treated.
2.1.1. Field Study
Beginning in July 2018, the experimental field was established after mowing an uncultivated area of land consisting of 24 plots. The soil type was Andosol (also known as Kuroboku soil), which is a common, humus-rich, light black soil in the Kanto Plain, Central Japan, developed from volcanic ash, and which was texturally classified as clay loam, with 29.6% of sand, 33.4% of silt, and 23.4% of clay [36,37]. This study used a randomized complete block design with four replications. The plot size was 1.05 m2 (0.70 × 1.50 m), and the distance between plots was 1.0 m. Treatments consisted of mowing, trampling 25 times (T25), trampling 50 times (T50), trampling 100 times (T100), and trampling 200 times (T200). The control plots were left undisturbed. Weed species grew naturally for around three months before being treated. Mowing and trampling experiments were carried out once after the weed survey. The weeds were trimmed to 2~5 cm using a shoulder-type lawn mower (MBC231DWB, Makita Co., Ltd., Kagawa, Japan) and the leaf cuttings were immediately removed from all mown plots. The trampling was conducted by rolling a 69.5 kg grass roller (SL-003 International Trading Co., Ltd., Yangjiang, China) back and forth from one end of the plot to the other.
2.1.2. Greenhouse Study
A greenhouse was used to grow southern crabgrass and Asian flatsedge plants both in the soil (i.e., for assessing the allelopathic influences of the rhizosphere soil) and in the sand (i.e., for assessing the allelopathic influences of the root exudates). Southern crabgrass was grown with commercially available seeds (ESPEC MIC Corporation, Aichi, Japan), while Asian flatsedge was grown with transplants collected from the field. Both species were grown for around four months. The T15 treatment was included based on the outcome of the field study, which suggested that light trampling pressure induced higher allelopathic impacts than heavy trampling pressure.
- Soil cultivation (for assessing allelopathic influences of the rhizosphere soil)
Between six and eight Southern crabgrass and Asian flatsedge plants were grown in clay pots (21 cm dia. × 17 cm depth) using commercially available, pre-fertilized, and granulated soil (JA Nippi No. 1, Ninon Hiryo Co., Ltd., Tokyo, Japan). Treatments consisted of mowing, trampling 15 times (T15), trampling 25 times (T25), and trampling 50 times (T50). The control pots were left undisturbed. All treatments, including the control pots, were replicated three times. The weeds were trimmed down to 2~5 cm using a garden shear, and the leaf cuttings were immediately removed from all the clipped pots. To ensure that each pot received the same amount of trampling force per treatment, trampling was strictly carried out on the same day by a single person (~50 kg), who evenly stamped on all weeds with a boot-shod leg.
- Sand cultivation (for assessing allelopathic influences of the root exudates)
Between six and eight southern crabgrass and Asian flatsedge plants were grown in clay pots (21 cm dia. × 17 cm depth) using commercially available 100% natural river sand (Miyuki Shoko Co., Ltd., Saitama, Japan). The use of natural river sand allowed the easy removal of plants from the clay pots without destroying the roots [26]. Treatments consisted of mowing and trampling 15 times (T15). All control plants were left intact or untouched.
2.2. Field Experiments
This section describes all experiments carried out in the field, including the weed survey, and the calculation of the frequency percentages of all identified weeds, the calculation of the multiplied dominance ratio (MDR) of the 5 most frequent weeds, the soil hardness test, and the gathering of rainfall data as well.
2.2.1. Weed Survey and Calculation of the Frequency Percentage
The assessment of the suppression of weed growth began by documenting and computing the frequency percentage of all spotted weeds within all 24 plots of the experimental field (two days before treatment). Frequency (%) measurement is an easy, fast, and reliable method because only the presence or absence of a species is recorded to calculate the percentage of all sampling units (e.g., quadrants or plots) in which the target species is found, and it is calculated as follows [38]:
The number of sampling units referred to the number of all plots in which a given weed was found, while the total number of sampling units was 24 (all 24 plots of the field). In addition, the frequency percentages were used to select the candidate weeds for multiplied dominance ratio (MDR) calculation and allelopathic activity bioassays.
2.2.2. Multiplied Dominance Ratio (MDR)
One day before treatment, which was considered zero weeks after treatment (0 WAT), two weeks after treatment (2 WAT), and four weeks after treatment (4 WAT), the percentage coverage and height of the 5 most frequent weeds were recorded within a 0.25 m2 quadrant (0.50 × 0.50 m) placed in the center of each plot. In each plot, the height of three mature individuals per species was randomly measured using a ruler (from soil to shoot apex) in three different places within the quadrant. The plants measured at 0 WAT were not marked to ensure randomness; therefore, they could not be recognized at both 2 WAT and 4 WAT. Afterwards, the MDR was calculated to express the impacts of mowing and trampling on weed volume [39] for the 5 most frequent weeds. The MDR is a common weed dominance index, calculated by multiplying the percentage coverage and height of each target species [40].
MDR (m3 m−2) = coverage (m2 m−2) × height (m)
2.2.3. Soil Hardness Test
Variations in the hardness of the soil are common indicators of changes in the levels of soil compaction [41,42]. Therefore, at 3 WAT, 6 WAT, and 13 WAT, soil hardness was recorded using a soil penetrometer (Hardness tester, Fujiwara Scientific Co., Ltd., Tokyo, Japan) to quantify the impacts of trampling and mowing on soil compaction. Three consecutive sunny days were awaited to record 10 samples per plot, because the soil hardness test is conducted best on moist, but not too wet, soil.
2.2.4. Gathering of the Rainfall Data
The data on daily rainfall in Fuchu, Tokyo, Japan, between 1 September 2018 and 31 December 2018 (Appendix A), were obtained using the Automated Meteorological Data Acquisition System (AMeDAS) [43].
2.3. Laboratory Bioassays
This section describes the laboratory bioassays (i.e., the rhizosphere soil and plant-box methods) used to assess the allelopathic potential of rhizosphere soil alongside the root exudates of the target weed species.
2.3.1. Rhizosphere Soil Method
The allelopathic effects of rhizosphere soil from both field-collected and greenhouse-grown weed species were investigated using the rhizosphere soil method described by Fujii et al. [27]. In all field and greenhouse studies, rhizosphere soil was collected on the fourth day after mowing and trampling. Rhizosphere soil is commonly defined as the soil adhering to plant roots after being shaken thoroughly [44].
Fifteen mature plants per species per treatment were gently pulled out of the ground by hand and subsequently taken into the laboratory for soil sampling, along with allelopathic analyses. Afterwards, all surface soil was shaken off the plants, and the rhizosphere soil was gently collected from the surface roots using a soft brush. Three grams of soil (sieved with a 1.0-mm sieve) was placed into a 6-well multi-dish. Subsequently, 5.0 mL of 0.75% agar was poured on top of the soil. After the gelatinization of the soil–agar mixture, an additional 3.2 mL of agar was added to the mixture. Lettuce seeds (Lactuca sativa L. var. Legacy; Takii Company, Kyoto, Japan) were planted on the gelatinized soil–agar mixture. The six-well multi-dishes were closed and incubated in a dark incubator (NTS Model MI-25S) at 25 °C for 3 days. Subsequently, the lengths of the lettuce radicles were measured.
The percentage of inhibition of rhizosphere soil growth before treatment (i.e., using intact plants) was determined by considering the growth of lettuce seedlings grown in the agar medium (gelling temperature 30–31 °C, Nacalai Tesque, Kyoto, Japan) as 100%. Furthermore, changes in the allelopathic effects of rhizosphere soil after treatments were determined by comparing the length of lettuce seedlings grown in the soil of mown and trampled plants with the length of lettuce seedlings grown in the soil of intact plants.
2.3.2. Plant-Box Method
The allelopathic effect of root exudates was assessed using the plant-box method described by Fujii et al. [26]. It was carried out in order to gain further insight into how the wound-induced changes in root exudation processes influence the allelopathic potential of the target weeds.
In this context, mature plants were mown or trampled and slowly pulled out of the pots by hand. The plants were immediately taken into the laboratory for the allelopathic assessment of root exudates. Afterwards, the roots of the plants were gently and thoroughly washed with distilled water. Then, the plants were placed into the root zone separating tubes and fixed in their positions in the plant boxes using cellophane tape. The agar solution was slowly poured into the boxes (to avoid bubbles) up to the 6.5 cm level. The boxes were cooled down immediately by dipping them in ice-chilled water (for approximately 30 min) and leaving them to stand at room temperature for a few more minutes. Lettuce seeds (Lactuca sativa L. var. Legacy; Takii Company, Kyoto, Japan) were seeded on the surface of the agar (narrowed tip downward). All boxes were covered with polyethylene and incubated in a growth chamber (BiOTRON. Type LH-350SP, NK System, Taiwan) at 25 °C for 5 days (12 h of light and 12 h of darkness). After the incubation period, the lengths of lettuce radicles and hypocotyls were measured.
The percentage of inhibition of root exudate growth before treatment (i.e., using intact plants) was determined by considering the growth of lettuce seedlings grown on the agar medium (gelling temperature 30–31 °C, Nacalai Tesque, Kyoto, Japan) as 100%. In addition, the changes in the allelopathic effects of the root exudates were determined by comparing the percentage growth inhibition of both mown and trampled plants with that of intact plants.
2.4. Statistical Analysis
Statistical analyses and graphs were obtained using IBM SPSS Statistics 27 (IBM® SPSS®, Armonk, NY, USA) along with Microsoft Excel (Microsoft, Redmond, Washington, DC, USA). Tukey’s HSD test, Dunnett’s test, and analysis of variance (ANOVA) were conducted. The significance level was 0.05.
3. Results
3.1. Weed Survey
In the field, seventeen weed species were identified before treatment and the frequency percentages of these species were calculated (Table 1). The five most frequent weed species were southern crabgrass (Digitaria ciliaris), Asian flatsedge (Cyperus microiria), oriental water willow (Justicia procumbens), horsenettle (Solanum carolinense), and giant foxtail (Setaria faberi). The five least frequent weeds were yellow foxtail (Setaria glauca), mulberry (Morus alba), dallisgrass (Paspalum dilatatum), spotted spurge (Euphorbia maculata), and annual bluegrass (Poa annua). The five most frequent weed species served to evaluate the impacts of both mowing and trampling on the MDRs of the weeds.

Table 1.
Frequency percentages of weed species identified inside all 24 experimental field plots.
3.2. Multiplied Dominance Ratio (MDR)
A wide-ranging dataset of the multiplied dominance ratios (MDRs) of all the five most frequent weeds is shown in Table 2. The high variability observed in the MDRs is related to the larger variations in the elongation (height) along with the expansion (coverage area) of candidate weed species. In comparison to all the other weeds, it was found that southern crabgrass was taller and had a larger coverage area, resulting in a significantly higher MDR. Despite being slightly taller than Asian flatsedge and oriental water willow plants, horsenettle and giant foxtail still occupied a smaller area inside most plots, which resulted in their MDRs becoming noticeably lower than the MDR of southern crabgrass. Additionally, an analysis of variance (ANOVA) of the MDRs also showed that the difference between species was highly significant. In comparison to the controls, only mowing induced significant decreases in the MDR of southern crabgrass (a vs. b) at both 2 WAT and 4 WAT. None of the trampling treatments significantly affected the MDRs of any target grass weeds (southern crabgrass, Asian flatsedge, and giant foxtail). On the other hand, the MDR of oriental water willow became significantly reduced due to mowing and T200 at 2 WAT. Moreover, the MDR of horsenettle was decreased by mowing, T50, T100, and T200 at 2 WAT. Throughout the study, the most frequent and most abundant weed was southern crabgrass.

Table 2.
Multiplied dominance ratio (MDR) of the five most frequent weed species in the experimental field.
Impacts of Mowing and Trampling on MDR of the Five Most Frequent Weeds
The results of the field study showed that mowing caused sharp and prolonged reductions in the MDRs of all the weed species. Moreover, it was observed that the various degrees of trampling induced uneven changes in the MDRs of the weeds (Table 2). In general, the ANOVA of the MDRs indicated that the differences between treatments and the species*treatments interaction were all highly significant at both 2 WAT and 4 WAT. Significant reductions in the MDR induced by both mowing and trampling were recorded at 2 WAT on horsenettle (mowing, T50, T100, and T200) along with oriental water willow (mowing and T200). Furthermore, the results indicated that, at both 2 WAT and 4 WAT, there were changes in the MDR of southern crabgrass induced by both mowing and all trampling treatments. However, significant changes (reductions) were only noted between mowing and the control (a vs. b). T25, T50, T100, and T200 were not significantly different from the control or mowing (ab). Furthermore, the results of the statistical analysis showed that (in comparison to controls) there were no significant reductions in the MDRs of all graminoids (i.e., southern crabgrass, Asian flatsedge, and giant foxtail) due to trampling. On the contrary, at 2 WAT, the MDR of the shrub (oriental water willow) had become significantly narrowed in T200, while the MDR of the forb (horsenettle) had also been significantly decreased in T50, T100, and T200. These findings show that graminoids (especially southern crabgrass) have stronger resistance to trampling than shrubs, along with forbs.
Resistance is commonly assessed based on the resistance indices, which are the number of passes required to reduce the vegetation cover or height by 50% [12,45]. In this study, resistance was assessed depending on the number of passes (trampling times) needed to reduce the MDR of a weed species by 50%. The resistance indices for southern crabgrass were 200 passes (T200) at 2 WAT and both 50 passes (T50) and 200 passes (T200) at 4 WAT. The resistance indices for most of the other weeds were 25 passes (T25) at both 2 WAT and 4 WAT. However, the results did not show a 50% reduction in the MDR of Asian flatsedge in both T50 and T100 at 2 WAT, along with the MDR of giant foxtail in T100 at both 2 WAT and 4 WAT. Although the height of the Asian flatsedge was reduced, the plant did not die and thus showed some resistance to trampling. On the other hand, most horsenettle, most oriental water willow, and most giant foxtail plants died shortly after trampling. In trampled plots, the MDR gradually declined over time. The MDR at 4 WAT was somewhat lower than the MDR at 2 WAT. However, the MDR increased marginally with time in the control plots.
3.3. Soil Hardness Test
An ANOVA of soil hardness indicated that the differences between treatment and measurement times were all highly significant (Figure 1). At 3 WAT, soil hardness in T100 was significantly higher compared to the control, mowing, T25, and T50. Furthermore, soil hardness in T200 was significantly higher compared to the control and mowing. At 6 WAT, only T100 showed significantly higher soil hardness than the control. Significant differences in soil hardness were not detected at 13 WAT.

Figure 1.
Mean soil hardness (N/cm2). The capped bars indicate the standard errors of four replications (n = 40). The means within each measurement time followed by the same letter are not significantly different (p < 0.05). 3 WAT, 6 WAT, and 13 WAT (weeks after treatment). Control, undisturbed plots; Mowing, mown plots; T25, trampling 25 times; T50, trampling 50 times; T100, trampling 100 times; and T200, trampling 200 times.
3.4. Results of the Allelopathic Potential of Selected Weed Species
This section introduces the results of the evaluation of the allelopathic activity of the studied weed species.
3.4.1. The Allelopathic Influences of Rhizosphere Soil
The results of the evaluation of the rhizosphere soil for potential allelopathic effects showed that the field-collected rhizosphere soil of intact southern crabgrass and Asian flatsedge induced the significant growth inhibition of lettuce radicles at 72.0% and 73.8%, respectively (Table 3 and Table 4).

Table 3.
A summary of one-way ANOVA of growth inhibition percentages of lettuce radicles assessed using the rhizosphere soil method.

Table 4.
Dunnett’s test of mean growth inhibition percentages of lettuce radicles assessed using the rhizosphere soil method.
Furthermore, an ANOVA of variations in the allelopathic activity of field-collected rhizosphere soil showed that the differences between species, treatments, and the species*treatments interaction were all highly significant (Figure 2A).

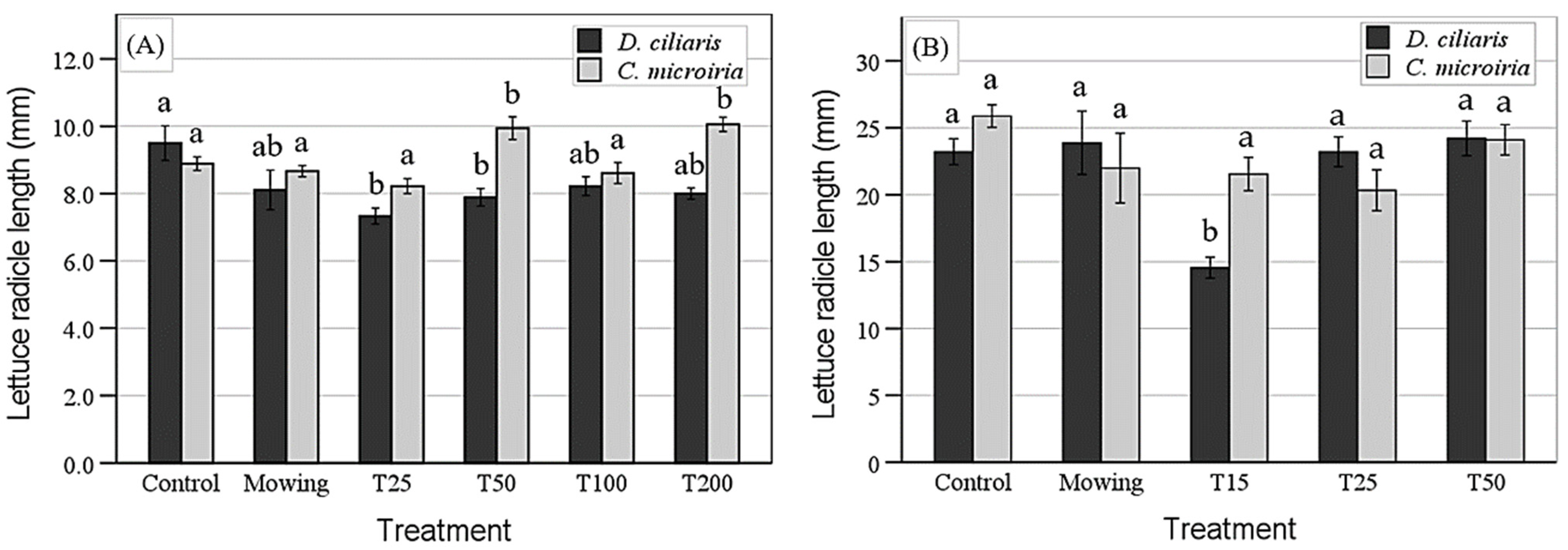
Figure 2.
Mean length of lettuce radicles grown in the rhizosphere soil of southern crabgrass (D. ciliaris) and Asian flatsedge (C. microiria). (A) The soil from the field; (B) the soil from the greenhouse. The capped bars indicate the standard errors of three replications (n = 9). The means of each weed species followed by the same letter are insignificantly different (p < 0.05). Control, intact plants; Mowing, mown plants; T15, trampling 15 times; T25, trampling 25 times; T50, trampling 50 times; T100, trampling 100 times; T200, trampling 200 times.
The lettuce radicles in T25 and T50 were significantly shorter concerning southern crabgrass than the controls. No significant difference was observed among the control, mowing, T25, and T100 treatments of Asian flatsedge. The lettuce radicles in T50 and T200 were significantly taller than the controls. In addition, an ANOVA of changes in the allelopathic activity of greenhouse-collected rhizosphere soil (Figure 2B) indicated that the differences between treatments and the species*treatments interaction were all significant. When the rhizosphere soil of southern crabgrass was used, the lettuce radicle in T15 was significantly shorter in comparison with the control, mowing, T25, and T50. No treatments significantly affected the allelopathic effects of the rhizosphere soil of Asian flatsedge from the greenhouse.
3.4.2. The Allelopathic Influences of Root Exudates
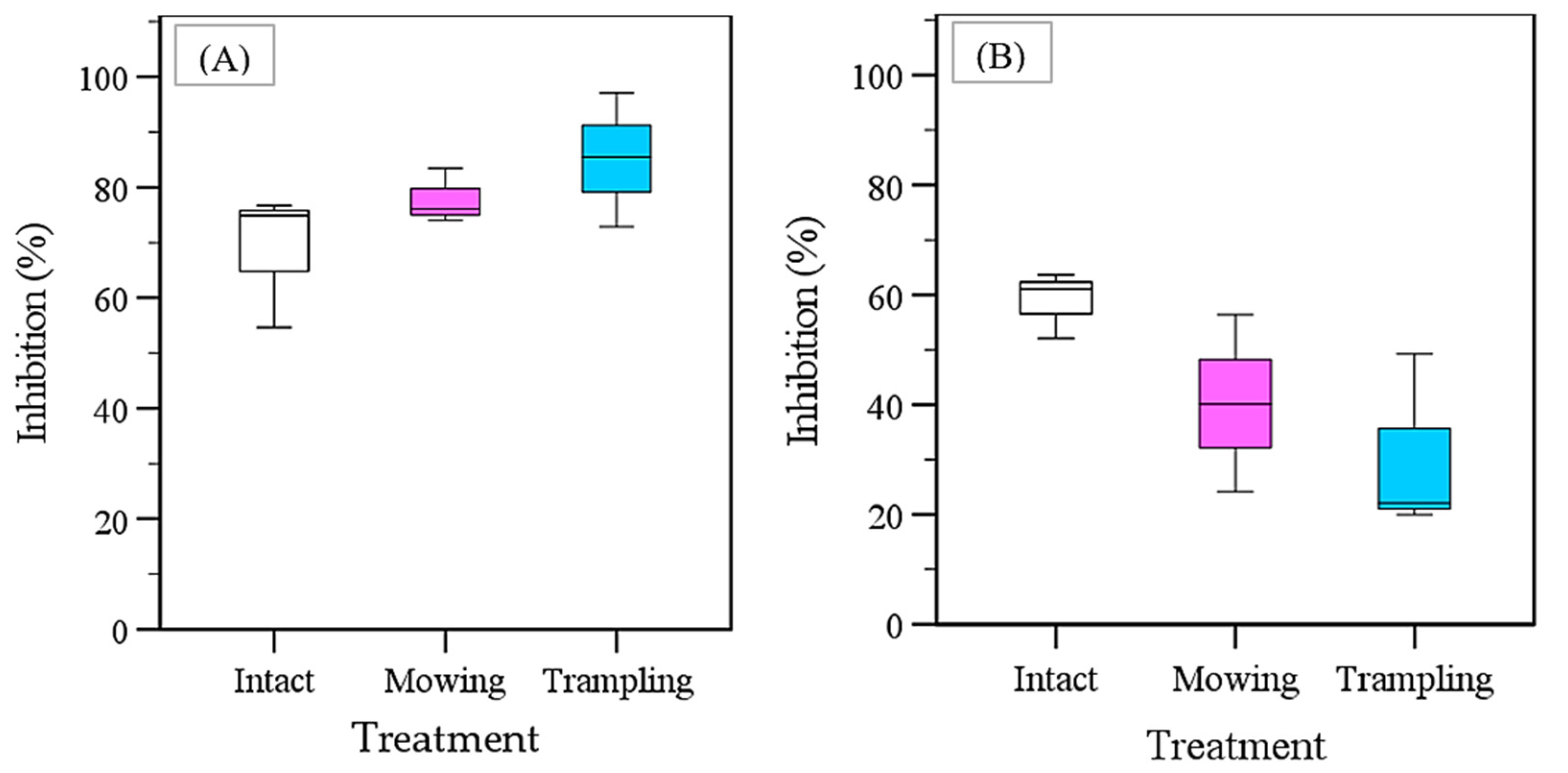
The results of the plant-box method showed that the root exudates of intact southern crabgrass and intact Asian flatsedge induced 77.9% and 58.9% growth inhibition in the lettuce radicle, respectively (Table 5 and Table 6 and Figure 3). Additionally, an ANOVA showed that southern crabgrass significantly inhibited the growth of lettuce when compared to the control. Furthermore, mowing and trampling increased the growth-inhibitory effects of southern crabgrass root exudates on lettuce radicles by 9.11% and 16.4%, respectively (Figure 3A). In contrast, mowing and trampling reduced the growth-inhibitory effects of Asian flatsedge against lettuce radicles by 18.7% and 28.5%, respectively (Figure 3B). Moreover, an ANOVA showed that the differences between species, treatments, and the species*treatment interaction were all highly significant. The root exudates of both mown and trampled southern crabgrass did not significantly inhibit the growth of lettuce seedlings. The growth of lettuce radicles was significantly stimulated by the root exudates of trampled Asian flatsedge. In addition, in comparison to the controls, lettuce seedlings incubated with mown Asian flatsedge had increased radicle lengths; however, all increases were insignificant.

Table 5.
A summary of one-way ANOVA of elongation percentages of lettuce radicles assessed using the plant-box method.

Table 6.
Dunnett’s test of mean elongation percentages of lettuce radicles assessed using the plant-box method.

Figure 3.
The percentage growth suppression of root exudates from sand-grown untouched, mown, and 15-times trampled southern crabgrass (D. ciliaris) alongside Asian flatsedge (C. microiria) against the elongation of lettuce radicles. (A) Southern crabgrass; (B) Asian flatsedge. The figures display the means of all three replications of the plant-box method outcome.
4. Discussion
The results of the weed survey and the values of MDR showed that the grass weeds, particularly southern crabgrass, strongly dominated all the other weeds in the no-tillage study field. Similarly, Kobayashi et al. [40,46] reported that the emergence of grass weeds is higher than in broad-leaved weeds in summer and no-tillage fields in Japan. In the tilled field, the weed emergence along with species composition was correlated strongly with the reservoir of weed seeds in the soil or the weed seedbank for both grass and broad-leaved weeds [47,48].
Although larger variations in the MDRs of target weeds were recorded before and after both mowing and trampling, relative reductions in the MDRs of all weeds were recorded at 2 WAT and 4 WAT in all trampled plots. These findings suggest that all trampling treatments impacted (depending on intensity and species) the suppression of growth (i.e., height and coverage area) in all weeds. Suppression of weed growth through mechanical means such as trampling or treading and rolling has also been reported in past studies [4,11].
Furthermore, southern crabgrass, alongside the other graminoids (Asian flatsedge and giant foxtail), demonstrated higher resistance to trampling than the shrub (oriental water willow) and the forb (horsenettle), which suggests that the higher resistance of the graminoids to trampling pressure was due to their morphological characteristics, i.e., graminoids’ growing points are commonly under the soil surface, rendering them more resilient to trampling pressure. In previous studies, it has also been indicated that the easily bendable stems, greater leaf tensile strength, low-to-ground growing points, and below-ground reproductive structure of the graminoids—southern crabgrass in particular—render them more resistant to trampling than shrubs and forbs, which are more vulnerable and characterized by broad leaves, woody stems, and reproductive structures high on the plant [45,49,50].
Increases in the soil hardness, largely inside the intensely trampled plots (T100 and T200), were revealed by the outcome of the soil hardness test, which indicated that the soil compaction increased with the increasing trampling intensity. Similarly, da Silva et al. [41] and Panda and Yamamoto [42] also reported that soil compaction depends on the increase in trampling intensity, and it is signaled by the variations in hydraulic conductivity along with soil hardness. The disappearance of significant differences in the soil hardness among the mown, trampled, and untouched plots (observed at 13 WAT) suggested that the soil had recovered by the end of the study. Moreover, previous studies have indicated that soils take between 85 and 165 days (depending on soil type) to recover from short-term trampling impacts through natural processes [51,52,53,54,55]. In addition, soil compaction occurs in the time for which soil particles are pressed tightly together, causing the pore space in between them to become narrower, and the soil bulk density to become higher [56]. Soil physical stresses due to compaction, along with drought, result in perturbations in the root exudation processes of stressed plants [57], reductions in root size, deceleration of root penetration, and decreases in the availability of plant nutrients.
The assessment outcome for the allelopathic potential indicated that both southern crabgrass and Asian flatsedge significantly inhibited the growth of lettuce radicles by over 70%. Therefore, both weeds contain compounds that suppress lettuce radicle growth. Similarly, Ito et al. [58] reported that soil in which southern crabgrass had been grown inhibited the growth of several crops, especially cucumber. The root exudates and extracts of the roots and aerial parts of Cyperus rotundus [59] and Cyperus iria [60] were reported to have phytotoxic effects on tomatoes, cucumbers, rice, and soybeans.
Furthermore, the growth suppression activities of the rhizosphere soil of southern crabgrass were significantly increased by the lowest trampling intensity under both greenhouse (T15) and field conditions (T25 and T50). However, the treatments did not significantly affect the growth-inhibitory effects of the rhizosphere soil of Asian flatsedge. The inhibitory effect of the root exudates of southern crabgrass was not significantly affected by mowing and T15. In contrast, T50 and T200, under field conditions, significantly reduced the growth suppression activities of Asian flatsedge rhizosphere soil. Additionally, trampling and mowing only slightly decreased the growth-suppressing abilities of Asian flatsedge root exudates, by 18.7% and 28.5%, respectively. These findings suggest that mowing and trampling induced uneven influences on the allelopathic potential of the two weed species, and that mowing and heavy trampling treatments (over T50) significantly increased the carbon demand, resulting in a lower concentration of allelochemicals available for rhizodeposition. Previous studies have shown that wounding in plants induces a transient disruption in the root exudation processes due to the turnover of storage compounds during remobilization [61,62], leading to the utilization of stored assimilates to support the maintenance respiration [63,64]. In addition, transient increases in the rhizodeposition of perennial ryegrass (Lolium perenne) following defoliation were reported by Paterson and Sim [38]. Meanwhile, decreases in the root exudates of timothy (Phleum pratense) [65] and maize plants [66] were reported after defoliation.
All trampling treatments induced relative growth suppression (i.e., reductions in the MDR) in all the target weeds. Moreover, a light trampling intensity resulted in higher increases in the allelopathic potential of the target weeds. These findings suggest that, apart from the pressing force of the roller, trampling induced the release of allelopathic compounds from the weeds and/or nearby organisms (i.e., volatile organic compounds such as ethylene and other allelochemicals in root exudates) to affect the growth of surrounding weed species. Jaffe [16], Chehab et al. [67], and Sunohora et al. [68] found that when plants such as Plantago asiatica, Cucumis sativus, Mimosa pudica, and Ricinus communis become mechanically stimulated due to fingers sliding along them or touching or trampling, they rapidly attempt to overcome the intrusion by undergoing diverse biochemical reactions, including the release of natural growth inhibitors such as ethylene, jasmonates, and abscisic acid (ABA), along with several morphological reactions, such as the acceleration of leaf senescence processes and fast cessation of shoot elongation. These types of touch-induced responses are commonly termed thigmomorphogenis [16,67]. In addition, similar findings on the release of phytotoxic compounds (such as organic carbon, phenolics, and sorgeolone) through the roots of mechanically stimulated or trampled plants have previously been published [23,61].
The evaluation of the potential allelopathic effect of root exudates using the plant-box method showed that after mowing and T15, the growth suppression activity of southern crabgrass increased, whereas that of Asian flatsedge decreased. These findings indicate that the differences in allelopathic effects between the two weeds could be due to the solubility of their root exudates. Hydrophobic compounds cannot travel in water-based media, including the plant-box and rhizosphere soil methods. Fujii et al. [26] found that plant species with hydrophobic chemicals possessed decreased allelopathic effects in water-based media compared to species with hydrophilic chemicals. Root exudates from Digitaria sanguinalis are largely composed of hydrophilic allelochemicals, such as veratric acid, maltol, and (−)-loliolide [33]. Meanwhile, root exudates from the rhizomes of Cyperus species are largely composed of essential oils, such as cyperol, α-cyperone [69], and methyl esters of acyclic terpenic acids [70], which are hydrophobic.
Moreover, the results showed that changes in the growth suppression activities of southern crabgrass and Asian flatsedge in the field after both mowing and various degrees of trampling differed from changes in their growth suppression activities in the greenhouse. These results suggest that the environmental conditions impacted the variations in the allelochemical exudation processes from the roots of the two weeds in response to mowing and varying trampling pressure. Similar to the outcome of this study, Yang et al. [71] also reported that the root exudation rate of some plant species, such as Pinus koraiensis, Larix gmelinii, and Betula platyphylla, was influenced by environmental factors, including the site, temperature, latitude, organic matter content, and moisture content. This short-term study involved single-year field and greenhouse research; however, it led to key insights into how and why long-term studies need to be carried out in the future to take full advantage of the impacts of trampling on the enhancement in the allelopathic potential of plants, along with suppression of weed growth, sustainably.
5. Conclusions
This is the first study to show that southern crabgrass and Asian flatsedge possess allelopathic effects on lettuce radicle growth. Mowing and varying trampling intensities resulted in contrasting impacts on both weed suppression and the allelopathic activity of southern crabgrass and Asian flatsedge. A forthcoming article will present the long-term impacts of trampling on weed suppression, alongside the allelopathic potential of cover crops. Future studies should also focus on identifying and quantifying the allelochemicals in southern crabgrass, Asian flatsedge, and other plant species for their potential utilization in sustainable weed management in combination with proper mowing and trampling.
Author Contributions
Conceptualization, B.B., T.C. and Y.F.; methodology, Y.F. and B.B.; software, B.B. and Y.F.; formal analysis, B.B., K.S.A. and Y.F.; investigation, B.B.; resources, Y.F., T.C. and B.B.; writing—initial draft preparation, K.S.A. and B.B.; writing—review and editing, K.S.A., S.T., T.C., B.B. and Y.F.; supervision, Y.F., S.T. and T.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially supported by JST-CREST, Grant Number JPMJCR17O2.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets of the present study are available from the corresponding authors upon reasonable request.
Acknowledgments
The authors would like to express their gratitude to the Japanese Ministry of Education, Culture, Sports, Science, and Technology (MEXT), Japan, for providing the scholarship to the first author at the Tokyo University of Agriculture and Technology, Tokyo, Japan.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A

Figure A1.
The total daily rainfall in Fuchu, Tokyo, Japan, between 1 September 2018 and 31 December 2018. The data were obtained using the Automated Meteorological Data Acquisition System (AMeDAS).
Figure A1.
The total daily rainfall in Fuchu, Tokyo, Japan, between 1 September 2018 and 31 December 2018. The data were obtained using the Automated Meteorological Data Acquisition System (AMeDAS).

References
- Ghanizadeh, H.; Harrington, K.C. Non-Target Site Mechanisms of Resistance to Herbicides. Crit. Rev. Plant Sci. 2017, 36, 24–34. [Google Scholar] [CrossRef]
- Hussain, M.; Farooq, S.; Merfield, C.; Jabran, K. Mechanical Weed Control. In Non-Chemical Weed Control; Elsevier Academic Press: Cambridge, MA, USA, 2018; pp. 133–155. ISBN 978-0-12-809881-3. [Google Scholar]
- Fynn, R.W.S.; Morris, C.D.; Edwards, T.J. Effect of Burning and Mowing on Grass and Forb Diversity in a Long-Term Grassland Experiment. Appl. Veg. Sci. 2004, 7, 1–10. [Google Scholar] [CrossRef]
- Inagaki, H.; Inagaki, S.; Kato, Y.; Kawai, M.; Sunakawa, T. Effects of Tread Pressure Treatment on Vegetation Surrounding Rice Fields. J. Jpn. Soc. Reveg. Technol. 2017, 43, 183–186. [Google Scholar] [CrossRef][Green Version]
- Kogler, R.; Quendler, E.; Boxberger, J. Occupational Accidents with Mowing Machines in Austrian Agriculture. Ann. Agric. Environ. Med. 2015, 22, 137–141. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wu, B.; Wu, Y.; Aoki, Y.; Nishimura, S. Mowing Patterns Comparison: Analyzing the Mowing Behaviors of Elderly Adults on an Inclined Plane via a Motion Capture Device. IEEE Access 2020, 8, 216623–216633. [Google Scholar] [CrossRef]
- Börklü, H.R.; Erdemir, F. Conceptual design of an innovative lawn mower machine. Düzce Üniversitesi Bilim Ve Teknol. Derg./Duzce Univ. J. Sci. Amp; Technol. 2019, 7, 15–26. [Google Scholar] [CrossRef]
- FAO. Pesticides and Environmental Incidents: Rotterdam Convention on the Prior Informed Consent Procedure for Certain Hazardous Chemicals and Pesticides in International Trade; FAO: Rome, Italy, 2022; ISBN 978-92-5-134937-3. [Google Scholar]
- Rani, L.; Thapa, K.; Kanojia, N.; Sharma, N.; Singh, S.; Grewal, A.S.; Srivastav, A.L.; Kaushal, J. An Extensive Review on the Consequences of Chemical Pesticides on Human Health and Environment. J. Clean. Prod. 2021, 283, 124657. [Google Scholar] [CrossRef]
- Mehdizadeh, M.; Mushtaq, W. Chapter 9—Biological Control of Weeds by Allelopathic Compounds from Different Plants: A BioHerbicide Approach. In Natural Remedies for Pest, Disease and Weed Control; Egbuna, C., Sawicka, B., Eds.; Elsevier Academic Press: Cambridge, MA, USA, 2020; pp. 107–117. ISBN 978-0-12-819304-4. [Google Scholar]
- Creamer, N.G.; Dabney, S.M. Killing Cover Crops Mechanically: Review of Recent Literature and Assessment of New Research Results. Am. J. Altern. Agric. 2002, 17, 32–40. [Google Scholar] [CrossRef]
- Cole, D.N.; Bayfield, N.G. Recreational Trampling of Vegetation: Standard Experimental Procedures. Biol. Conserv. 1993, 63, 209–215. [Google Scholar] [CrossRef]
- Mihretie, F.A.; Tsunekawa, A.; Haregeweyn, N.; Adgo, E.; Tsubo, M.; Masunaga, T.; Meshesha, D.T.; Tsuji, W.; Ebabu, K.; Tassew, A. Tillage and Sowing Options for Enhancing Productivity and Profitability of Teff in a Sub-Tropical Highland Environment. Field Crop. Res. 2021, 263, 108050. [Google Scholar] [CrossRef]
- Warren, S.D.; Nevill, M.B.; Blackburn, W.H.; Garza, N.E. Soil Response to Trampling under Intensive Rotation Grazing. Soil Sci. Soc. Am. J. 1986, 50, 1336–1341. [Google Scholar] [CrossRef]
- Burden, R.F.; Randerson, P.F. Quantitative Studies of the Effects of Human Trampling on Vegetation as an Aid to the Management of Semi-Natural Areas. J. Appl. Ecol. 1972, 9, 439–457. [Google Scholar] [CrossRef]
- Jaffe, M.J. Thigmomorphogenesis: The Response of Plant Growth and Development to Mechanical Stimulation. Planta 1973, 114, 143–157. [Google Scholar] [CrossRef]
- Hatanaka, A. The Biogeneration of Green Odour by Green Leaves. Phytochemistry 1993, 34, 1201–1218. [Google Scholar] [CrossRef]
- Iqbal, N.; Khan, N.A.; Ferrante, A.; Trivellini, A.; Francini, A.; Khan, M.I.R. Ethylene Role in Plant Growth, Development and Senescence: Interaction with Other Phytohormones. Front Plant Sci 2017, 8, 475. [Google Scholar] [CrossRef] [PubMed]
- Bardgett, R.D.; Mawdsley, J.L.; Edwards, S.; Hobbs, P.J.; Rodwell, J.S.; Davies, W.J. Plant Species and Nitrogen Effects on Soil Biological Properties of Temperate Upland Grasslands. Funct. Ecol. 1999, 13, 650–660. [Google Scholar] [CrossRef]
- Paterson, E.; Thornton, B.; Sim, A.; Pratt, S. Effects of Defoliation and Atmospheric CO2 Depletion on Nitrate Acquisition, and Exudation of Organic Compounds by Roots of Festuca rubra. Plant Soil 2003, 250, 293–305. [Google Scholar] [CrossRef]
- Paterson, E.; Thornton, B.; Midwood, A.J.; Sim, A. Defoliation Alters the Relative Contributions of Recent and Non-Recent Assimilate to Root Exudation from Festuca rubra. Plant Cell Environ. 2005, 28, 1525–1533. [Google Scholar] [CrossRef]
- Bertin, C.; Yang, X.; Weston, L.A. The Role of Root Exudates and Allelochemicals in the Rhizosphere. Plant Soil 2003, 256, 67–83. [Google Scholar] [CrossRef]
- Yang, X.; Owens, T.G.; Scheffler, B.E.; Weston, L.A. Manipulation of Root Hair Development and Sorgoleone Production in Sorghum Seedlings. J. Chem. Ecol. 2004, 30, 199–213. [Google Scholar] [CrossRef] [PubMed]
- Czarnota, M.A.; Rimando, A.M.; Weston, L.A. Evaluation of Root Exudates of Seven Sorghum Accessions. J. Chem. Ecol. 2003, 29, 2073–2083. [Google Scholar] [CrossRef] [PubMed]
- Trezzi, M.M.; Vidal, R.A. Potential of Sorghum and Pearl Millet Cover Crops in Weed Suppression in the Field: II—Mulching Effect. Planta Daninha 2004, 22, 1–10. [Google Scholar] [CrossRef]
- Fujii, Y.; Pariasca, D.; Shibuya, T.; Yasuda, T.; Kahn, B.; Waller, G.R. Plant-box Method: A Specific Bioassay to Evaluate Allelopathy through Root Exudates. In Allelopathy New Concept and Methodology; Fujii, Y., Hiradate, S., Eds.; Science Publishers: Enfield, CT, USA, 2007; pp. 39–56. [Google Scholar]
- Fujii, Y.; Furubayashi, A.; Hiradate, S. Rhizosphere Soil Method: A New Bioassay to Evaluate Allelopathy in the Field. In Proceedings of the 4th World Congress on Allelopathy, “Establishing the Scientific Base”, Wagga Wagga, Australia, 21–26 August 2005; pp. 493–497. [Google Scholar]
- Fujii, Y.; Matsuyama, M.; Hiradate, S.; Shimozawa, H. Dish Pack Method: A New Bioassay for Volatile Allelopathy. Thymus 2005, 2, 493–497. [Google Scholar]
- Fujii, Y.; Parvez, S.S.; Parvez, M.M.; Ohmae, Y.; Iida, O. Screening of 239 Medicinal Plant Species for Allelopathic Activity Using the Sandwich Method. Weed Biol. Manag. 2003, 3, 233–241. [Google Scholar] [CrossRef]
- Noguchi, K. Upland Weeds and Their Management in Soybean in Japan. In Pest Management in Soybean; Copping, L.G., Green, M.B., Rees, R.T., Eds.; Springer: Dordrecht, The Netherlands, 1992; pp. 299–307. ISBN 978-94-011-2870-4. [Google Scholar]
- Ito, M.; Ichikawa, E. Nitrification Inhibition by Roots of Digitaria adscendens (H. B. K.) Henr. J. Weed Sci. Technol. 1994, 39, 125–127. [Google Scholar] [CrossRef]
- Chozin, M.A.; Yasuda, S. Possibility of Natural Hybridization between Cyperus iria L. and Cyperus microiria Steud. J. Weed Sci. Technol. 1991, 36, 282–289. [Google Scholar] [CrossRef]
- Zhou, B.; Kong, C.-H.; Li, Y.-H.; Wang, P.; Xu, X.-H. Crabgrass (Digitaria sanguinalis) Allelochemicals that Interfere with Crop Growth and the Soil Microbial Community. J. Agric. Food Chem. 2013, 61, 5310–5317. [Google Scholar] [CrossRef]
- Komai, K.; Tang, C.-S. Chemical Constituents and Inhibitory Activities of Essential Oils from Cyperus brevifolius And C. kyllingia. J Chem Ecol 1989, 15, 2171–2176. [Google Scholar] [CrossRef]
- Chou, C.-H.; Leu, L.-L. Allelopathic Substances and Interactions of Delonix regia (Boj) Raf. J. Chem. Ecol. 1992, 18, 2285–2303. [Google Scholar] [CrossRef]
- Jaikaew, P.; Boulange, J.; Thuyet, D.Q.; Malhat, F.; Ishihara, S.; Watanabe, H. Potential Impacts of Seasonal Variation on Atrazine and Metolachlor Persistence in Andisol Soil. Env. Monit Assess 2015, 187, 760. [Google Scholar] [CrossRef]
- IUSS Working Group, W.R.B. World Reference Base for Soil Resources 2014: International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; FAO: Rome, Italy, 2014; ISBN 978-92-5-108370-3. [Google Scholar]
- Booth, B.D.; Murphy, S.D.; Swanton, C.J. Weed Ecology in Natural and Agricultural Systems; CAB International: Wallingford, UK, 2003; ISBN 978-0-85199-528-1. [Google Scholar]
- Kobayashi, H. Seasonal Dynamics of Weed Biomass in Upland Fields, Periodically Cultivated Fields and Weeded Fields without Soil Disturbance: Estimation Based on Plant Coverage and Height. Tohoku Agri. Res. 2000, 53, 93–94. [Google Scholar]
- Kobayashi, H.; Nakamura, Y.; Watanabe, Y. Analysis of Weed Vegetation of No-Tillage Upland Fields Based on the Multiplied Dominance Ratio. Weed Biol. Manag. 2003, 3, 77–92. [Google Scholar] [CrossRef]
- da Silva, A.P.; Imhoff, S.; Corsi, M. Evaluation of Soil Compaction in an Irrigated Short-Duration Grazing System. Soil Tillage Res. 2003, 70, 83–90. [Google Scholar] [CrossRef]
- Pande, T.N.; Yamamoto, H. Cattle Treading Effects on Plant Growth and Soil Stability in the Mountain Grassland of Japan. Land Degrad. Dev. 2006, 17, 419–428. [Google Scholar] [CrossRef]
- Japan Meteorological Agency|AMeDAS. Available online: https://www.jma.go.jp/jma/en/Activities/amedas/amedas.html (accessed on 28 October 2022).
- Kang, S.; Mills, A.L. Soil Bacterial Community Structure Changes Following Disturbance of the Overlying Plant Community. Soil Sci. 2004, 169, 55–65. [Google Scholar] [CrossRef]
- Hill, R.; Pickering, C. Differences in Resistance of Three Subtropical Vegetation Types to Experimental Trampling. J. Environ. Manag. 2009, 90, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Miura, S.; Oyanagi, A. Effects of Winter Barley as a Cover Crop on the Weed Vegetation in a No-Tillage Soybean. Weed Biol. Manag. 2004, 4, 195–205. [Google Scholar] [CrossRef]
- Rahman, A.; James, T.K.; Grbavac, N. Potential of Weed Seedbanks for Managing Weeds: A Review of Recent New Zealand Research. Weed Biol. Manag. 2001, 1, 89–95. [Google Scholar] [CrossRef]
- Rahman, A.; James, T.K.; Grbavac, N. Correlation between the Soil Seed Bank and Weed Populations in Maize Fields. Weed Biol. Manag. 2006, 6, 228–234. [Google Scholar] [CrossRef]
- Sun, D.; Liddle, M.J. Trampling Resistance, Stem Flexibility and Leaf Strength in Nine Australian Grasses and Herbs. Biol. Conserv. 1993, 65, 35–41. [Google Scholar] [CrossRef]
- Kobayashi, T.; Hori, Y.; Nomoto, N. Effects of Trampling and Vegetation Removal on Species Diversity and Micro-Environment under Different Shade Conditions. J. Veg. Sci. 1997, 8, 873–880. [Google Scholar] [CrossRef]
- Greenland, D.J. Soil Management and Soil Degradation. J. Soil Sci. 1981, 32, 301–322. [Google Scholar] [CrossRef]
- Dexter, A.R. Amelioration of Soil by Natural Processes. Soil Tillage Res. 1991, 20, 87–100. [Google Scholar] [CrossRef]
- Jordan, D.; Li, F.; Ponder Jr, F.; Berry, E.C.; Hubbard, V.C.; Kim, K.Y. The Effects of Forest Practices on Earthworm Populations and Soil Microbial Biomass in a Hardwood Forest in Missouri. Appl. Soil Ecol. 1999, 13, 31–38. [Google Scholar] [CrossRef]
- Elliott, A.H.; Tian, Y.Q.; Rutherford, J.C.; Carlson, W.T. Effect of Cattle Treading on Interrill Erosion from Hill Pasture: Modelling Concepts and Analysis of Rainfall Simulator Data. Soil Res. 2002, 40, 963–976. [Google Scholar] [CrossRef]
- Drewry, J.J.; Paton, R.J.; Monaghan, R.M. Soil Compaction and Recovery Cycle on a Southland Dairy Farm: Implications for Soil Monitoring. Soil Res. 2004, 42, 851–856. [Google Scholar] [CrossRef]
- Reintam, E.; Kuht, J. Weed Responses to Soil Compaction and Crop Management. In Weed Control; Price, A., Ed.; InTech: London, UK, 2012; ISBN 978-953-51-0159-8. [Google Scholar]
- More, S.S.; Shinde, S.E.; Kasture, M.C. Root Exudates a Key Factor for Soil and Plant: An Overview. Pharma Innov. J 2020, 8, 449–459. [Google Scholar]
- Ito, M.; Kobayashi, H.; Ueki, K. Allelopathic Potential of Digitaria adscendens: Inhibitory Effects of Previously Grown Soil on Crop Growth and Weed Emergence. In Proceedings of the 11th Asian Pacific Weed Science Society Conference, Taipei, Taiwan, China, 29 November–5 December 1987; Volume 2, pp. 607–612. [Google Scholar]
- Alsaadawi, I.S.; Salih, N.M.M. Allelopathic Potential of Cyperus rotundas LI Interference with Crops. Allelopath. J. 2009, 23, 297–303. [Google Scholar]
- Chopra, N.; Tewari, G.; Tewari, L.M.; Upreti, B.; Pandey, N. Allelopathic Effect of Echinochloa colona L. and Cyperus iria L. Weed Extracts on the Seed Germination and Seedling Growth of Rice and Soyabean. Adv. Agric. 2017, 2017, 5748524. [Google Scholar] [CrossRef]
- Paterson, E.; Sim, A. Rhizodeposition and C-Partitioning of Lolium perenne in Axenic Culture Affected by Nitrogen Supply and Defoliation. Plant Soil 1999, 216, 155–164. [Google Scholar] [CrossRef]
- Danckwerts, J.E.; Gordon, A.J. Long-Term Partitioning, Storage and Re-Mobilization of 14C Assimilated by Lolium perenne (Cv. Melle). Ann. Bot. 1987, 59, 55–66. [Google Scholar] [CrossRef]
- Evans, G.C. The Quantitative Analysis of Plant Growth; University of California Press: Berkeley, CA, USA, 1972; Volume 1. [Google Scholar]
- Bokhari, U.G.; Singh, J.S. Effects of Temperature and Clipping on Growth, Carbohydrate Reserves, and Root Exudation of Western Wheatgrass in Hydroponic Culture1. Crop Sci. 1974, 14, 790–794. [Google Scholar] [CrossRef]
- Mikola, J.; Kytöviita, M.-M. Defoliation and the Availability of Currently Assimilated Carbon in the Phleum pratense Rhizosphere. Soil Biol. Biochem. 2002, 34, 1869–1874. [Google Scholar] [CrossRef]
- Nguyen, C.; Henry, F. A Carbon-14-Glucose Assay to Compare Microbial Activity between Rhizosphere Samples. Biol. Fertil. Soils 2002, 35, 270–276. [Google Scholar] [CrossRef]
- Chehab, E.W.; Eich, E.; Braam, J. Thigmomorphogenesis: A Complex Plant Response to Mechano-Stimulation. J. Exp. Bot. 2009, 60, 43–56. [Google Scholar] [CrossRef]
- Sunohara, Y.; Ikeda, H.; Tsukagoshi, S.; Murata, Y.; Sakurai, N.; Noma, Y. Effects of Trampling on Morphology and Ethylene Production in Asiatic Plantain. Weed Sci. 2002, 50, 479–484. [Google Scholar] [CrossRef]
- Stephane, F.F.Y.; Juleshttps, B.K.J. Terpenoids as Important Bioactive Constituents of Essential Oils. In Essential Oils-Bioactive Compounds, New Perspectives and Applications; IntechOpen: London, UK, 2020; ISBN 978-1-83962-697-5. [Google Scholar]
- Iwamur, J.; Hosotsubo, H.; Hirao, N. Mass Spectra for Molecular Structure(4)Mass Spectra of 2-Alkenoic Acid Methyl Esters. Mass Spectrosc. 1978, 26, 269–274. [Google Scholar] [CrossRef]
- Yang, L.; Wang, X.; Mao, Z.; Jiang, Z.; Gao, Y.; Chen, X.; Aubrey, D.P. Root Exudation Rates Decrease with Increasing Latitude in Some Tree Species. Forests 2020, 11, 1045. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).